Abstract
A mathematical model of cerebral blood flow in the form of a dynamical system is studied. The cerebral blood flow autoregulation modeling problem is treated as a nonlinear control problem and the potential and applicability of the nonlinear control theory techniques are analyzed in this respect. It is shown that the cerebral hemodynamics model in question is differentially flat. Then, the integrator backstepping approach combined with barrier Lyapunov functions is applied to construct the control laws that recover the cerebral autoregulation performance of a healthy human. Simulation results confirm the good performance and flexibility of the suggested cerebral blood flow autoregulation design. The conducted research should enrich our understanding of the mathematics behind the cerebral blood flow autoregulation mechanisms and medical treatments to compensate for impaired cerebral autoregulation, e.g., in preterm infants.
Keywords:
intracranial hemodynamics; cerebral autoregulation; biomechanical system; nonlinear dynamics; nonlinear control; differential flatness; output tracking; integrator backstepping MSC:
93C10; 93C95
1. Introduction
Understanding the mathematics behind the cerebral autoregulation process is of primary importance in the mathematical modeling of cerebral blood circulation and regulation. Impaired cerebral blood flow autoregulation is one of the crucial factors that can cause cerebral hemorrhage in preterm infants. An impaired cerebral autoregulation mechanism is unable to maintain constant cerebral blood flow despite the changes in systemic arterial pressure. As a result, an increase in cerebral blood flow caused by an acute systemic arterial pressure increase can lead to bleeding in the germinal matrix of a preterm newborn [1], which is a specific region in the immature brain between the thalamus and caudate nucleus, with high vascularity and a fragile capillary network [2]. Among a variety of cerebral blood flow models (see, e.g., [3,4,5,6,7,8,9,10,11]), one can highlight lumped parameter models based on the analogy to electric circuits [4,5,6,7,8,9,10]. In [9,10], the influence of the germinal matrix on blood flow is taken into account by modeling the capillary level as two parallel connected lumped objects describing the germinal matrix and the remaining part of the brain. In [4], a cerebral blood flow model is presented in the form of nonlinear ordinary differential equations, which can be seen as a starting point to the automatic control theory oriented cerebral autoregulation modeling, because of its simplicity and, at the same time, ability to reproduce various clinical results [4].
The first steps towards considering the cerebral blood flow autoregulation modeling problem as a feedback control problem were undertaken in [7,8]. In [7], maintenance of the cerebral autoregulatory function was studied as an optimal conflict control problem. In [8], a mathematical model of cerebral autoregulation was proposed in the form of a heuristic feedback controller, verified using the techniques of the viability theory [12].
In the current work, we continue to develop the automatic control theory-based autoregulation considerations in a systemic way. Based on the cerebral blood flow dynamical model of [4], the nonlinear control theory tools are applied to construct the feedback control laws that describe the mathematics behind the cerebral autoregulation mechanisms. The main results of this paper originate from the suggested idea to interpret the cerebral blood flow autoregulation modeling challenge as an automatic control problem. This is the first time, at least to our knowledge, that the cerebral blood flow model introduced in [4] is studied as a dynamical system with control input and its controllability properties are analyzed. Because of the model’s intrinsic nonlinearity, such a well-known and effective nonlinear control tool as integrator backstepping combined with barrier Lyapunov functions is used to construct the control laws that recover the cerebral autoregulation performance of healthy humans.
The remaining part of the paper is organized as follows. In Section 2, the cerebral hemodynamics model equations presented in [4] are revisited and written in the form of a nonlinear dynamical system with control input. The cerebral blood flow autoregulation modeling problem is formulated as a nonlinear output tracking control problem. In Section 3, we show the differential flatness property of the cerebral hemodynamics in question, which is used further in Section 4 to construct the state feedback control laws that model the cerebral autoregulation performance. Numerical simulation results of the suggested cerebral blood flow autoregulation scheme are given in Section 4. Finally, Section 5 concludes with some remarks.
2. Problem Formalization
In this paper, we consider the cerebral hemodynamics model introduced in [4]. For convenience’s sake, let us first summarize the model equations. The model accounts for the hemodynamics of the arterial–arteriolar cerebrovascular bed and large cerebral veins, cerebrospinal fluid circulation and intracranial pressure dynamics; see Figure 1. The arterial–arteriolar cerebrovascular bed is modeled as a windkessel and is described by means of the hydraulic compliance (storage capacity) and the hydraulic resistance variables [4]. The arterial–arteriolar blood volume is calculated as below
where and stand for the systemic arterial and intracranial pressure variables, respectively. The difference represents transmural pressure in the arterioles. The rate q of blood flow through the arterial–arteriolar cerebrovascular bed that enters the skull is written as
where denotes the capillary pressure variable, and the difference is the perfusion pressure of the arterioles. Here, by the Hagen–Poiseuille law, the arterial–arteriolar resistance is inversely proportional to the second power of the blood volume with a coefficient [4], i.e.,

Figure 1.
The cerebral hemodynamics model components and quantities.
For the large intracranial veins, autoregulatory mechanisms and venous elasticity are neglected. The transmural pressure in the cerebral veins and the venous blood volume are supposed to remain constant. Then, the rate of the blood flow through the venous cerebrovascular bed is described using a hydraulic resistance as
with the venous hydraulic compliance being ignored and treated as a constant [4]. Here, in (4), the difference represents the perfusion pressure of the cerebral veins. Similarly, cerebrospinal fluid production at the cerebral capillaries and reabsorption at the dural sinuses are modeled as static processes. The cerebrospinal fluid production and reabsorption rates and are characterized by constant hydraulic resistances and , respectively, as below
where is the venous sinus pressure, which is taken as a constant [4]. The quantities q, and given by the Formulae (2), (4) and (5) are linked through the algebraic equation
The intracranial pressure dynamics by Monro–Kellie doctrine are written as follows [4]:
where is the intracranial compliance (craniospinal storage capacity), and stands for the constant rate of possible mock cerebrospinal fluid injection in surgery. In (7), the intracranial storage capacity is inversely proportional to the intracranial pressure with a craniospinal compartment elastance coefficient , i.e.,
Moreover, one should note that the following condition on the pressure values is required to hold for all the above considerations to be valid [4].
Finally, within the cerebral hemodynamics model in question, cerebral blood flow autoregulation is supposed to function only at the level of arterioles and is described in terms of the arterial–arteriolar compliance . Vasodilation or vasoconstriction of the arterioles is modeled through positive or negative values of the compliance rate , respectively. In [4], the following heuristic cerebral autoregulation model is suggested:
where is a sigmoidal function with saturation, is a time constant, and denotes a basal value of the arterial–arteriolar blood flow rate q required for tissue metabolism.
Notice that the cerebral autoregulation model (9) is intuitively clear but the authors do not provide any mathematical proof or strict mathematical considerations of its validity in [4]. In this paper, as is done in [7], the arterial–arteriolar compliance rate is considered as a control input. Then, the control purpose is to force the nonzero values of the difference to zero using nonlinear control theory tools and, thus, provide rigorous mathematical insights into the cerebral blood flow autoregulation mechanism of a healthy human.
In the current work, we adopt the arterial–arteriolar blood volume and the intracranial pressure as system state variables, instead of considering the and dynamics, as is done in [4]. This choice of the state variables is more suitable for a thorough analysis of the cerebral hemodynamics in question and control design since is a crucial quantity defining the behavior of the overall system. In view of (1), the blood volume dynamics are governed by the formula
and are determined by the cerebral autoregulation mechanism , the intracranial pressure dynamics and the systemic arterial pressure alterations rate .
After rearranging the terms in the differential-algebraic Equations (7) and (10) and taking into account the relations (5) and (8), one can avoid algebraic loops and obtain the following dynamical system:
which describes time behavior of the cerebral blood volume and the intracranial pressure . Using the Formulae (1)–(6), the capillary pressure and the arterial–arteriolar compliance quantities in the right-hand side of the system (11) can be represented as functions of the system state variables and in the following way:
Notice that the functions in the right-hand side of the system (11) depend on the systemic arterial pressure time behavior. In this paper, we suppose that the arterial blood pressure dynamics are in a steady state, i.e., , and the arterial pressure has a constant value, which is possibly different from the basal one. Then, the choice of the arterial–arteriolar compliance rate as a control input u results in the cerebral hemodynamics model
with the arterial-,arteriolar blood flow rate q being considered as a system output function and in view of (2), (3) and (12), written as
Thus, in summary, the cerebrovascular autoregulation modeling problem in question can be formulated as a constrained (e.g., asymptotic) output regulation control problem for the nonlinear dynamical system (13), i.e., we find a feedback control law such that
for all reasonable initial values , of the system state variables. In addition, for a proper range of the systemic arterial pressure values , the quantities and are required to remain positive during transients and within reasonable bounds
3. Differential Flatness of Cerebral Hemodynamics
First, let us check the controllability properties of the nonlinear dynamical system (13) by showing that it is differentially flat [13]. Recall that a dynamical system of the form (13) is differentially flat if and only if there exists a scalar function of the system state variables , and, in a general case, of the control input u with its time derivatives , , ⋯, for some finite natural number such that , and u can be represented as functions of and its time derivatives of up to some finite order [13]. Then, it is well known that differentially flat systems possess good controllability properties (see, e.g., [13,14]). To find such a function , called the flat output, one can exploit the linearity of the functions on the right-hand side of the system (13) with respect to the control variable u. The coefficients of u in (13) form the vector field
As a flat output candidate for the system (13), one can take a first integral of the vector field (17) (see, e.g., [15]). To find the first integrals of given by (17), consider an auxiliary system of ordinary differential equations written in the symmetric form
Integration of (18) results in the following function:
which has constant values on solutions of the auxiliary system (18), with its Lie derivative [15] along the vector field being identically zero. Then, the first- and second-order time derivatives of (19) along solutions of the dynamical system (13) can be written, respectively, as
where and are corresponding functions of their arguments.
Let , and . One can show that the Jacobian matrix of the mapping defined by the relations (19) and (20) is nonsingular at a point , if and only if the following condition holds:
Hence, the map is a diffeomorphism defined for all values , such that the inequality (22) is satisfied. The relationships (19) and (20) qualify as a change of coordinates in the state space of the system (13), with its inversion being written as . Thus, the system state variables , are expressed as functions of the flat output given by (19) and its time derivative defined as (20). Finally, from (21), we deduce that the control variable u can be represented for the nonzero values of as below
where
4. Nonlinear Output Regulation Control Design
To guarantee the cerebral blood flow regulation (15), one can first try to find the constant reference values and such that the condition holds. Then, a reasonable control strategy would be to force the differences and to zero as in a controllable way to meet the constraints (16) by the choice of a state feedback .
In this paper, the reference value of the intracranial pressure variable let us select to nullify the rate given by (20) under the basal value of the arterial-arteriolar blood flow rate, i.e.,
One can easily check that the right-hand side of the expression (25) under the basal values of model parameters given in [4] and revised for convenience’s sake in Table 1 coincides with a basal value of the intracranial pressure in a healthy human [16]. Note that, in case some of the model parameter values in (25) differ from the basal ones of a healthy human, still, one could use, for instance, the constant rate of mock cerebrospinal fluid injection to obtain a reference value of the intracranial pressure that stays within medically reasonable bounds [16].

Table 1.
Model parameters in basal conditions.
Then, from the relations (14) and , it is deduced that
Note that, for the reference blood volume value (26) to be correctly defined, the following conditions on the model parameters and the intracranial pressure reference value have to be satisfied:
It is worthwhile to indicate that the validity of the relationships (27) is inherently related to the model parameters’ consistency and can be easily verified for the parameter values given in Table 1 within the systemic arterial pressure autoregulatory lower and upper limits mmHg and mmHg, respectively [4].
In what follows, we exploit the differential flatness property of the cerebral hemodynamics model (13) conceived in the above section. In the new coordinates defined by the Formulae (19) and (20), the dynamical system (13) takes the form
with the functions on its right-hand side being introduced in (24). Further, in view of (19) for the variable, we take the reference value , where and are given by (25) and (26), respectively. Moreover, by combining the relations (20) and (25), one obtains as the reference value of the variable.
To achieve the regulation and as , a straightforward control selection would be the state feedback linearization-based design
which results in the following regulation error dynamics:
Then, for any positive gain coefficients and , the equilibrium point , of the system (30) is (globally) asymptotically stable.
Notice that the control law (29) and, hence, the resultant closed-loop dynamics (30) are defined whenever the control coefficient in (28) is not zero. It can be shown that the inequality holds for the reference values , of the arterial–arteriolar blood volume and intracranial pressure variables defined as (25) and (26), respectively, under the autoregulatory range of the systemic arterial pressure values mmHg and model parameter basal values given in Table 1. Hence, due to the continuity property of the function , the condition is satisfied at least in some neighborhood of the point , of the system (28) state space.
It is well known that the control law (29) cannot explicitly guarantee that the nontrivial trajectories , of the closed-loop dynamics (30) (and, hence, the variables and during the cerebral autoregulation transients) remain bounded within the prescribed bounds for all or do not approach and become stuck in the set ().
To avoid the control singularity, i.e., obtaining at some , let us redesign the control law (29) to provide the conditions
with proper positive bounds , consistent with the constraints (16). Notice that, since , which, by virtue of the relations (24), is equivalent to the inequality , there always exist positive constants , such that, from (31), it can be deduced that for all . Then, since the change of coordinates given by (19) and (20) defines a diffeomorphism whenever , the inequalities (31) can be rewritten in the variables , as
with some relevant bounds and .
To provide the regulation , as and satisfy the conditions (32) on the transients simultaneously, we suggest to redesign the control law (29) by using the integrator backstepping approach [17] based on barrier Lyapunov functions; see, e.g., [18].
To this end, consider first a (barrier) function
where , , is a positive design constant. Introduce the error variable . Here, is a continuously differentiable function to be defined later, which accounts for the desired reference behavior of the variable.
The time derivative of along solutions of the system (28) is written as
The choice , where is a positive gain coefficient, results in
Hence, is negative definite if .
Then, as a Lyapunov function candidate for the whole system, employ the barrier function
where , is some positive design constant. Let the values and , be such that . Note that the function is positive definite in the domain and grows unbounded as or/and .
The time derivative of along solutions of the system (28) is calculated as below
The control selection
where is a positive gain coefficient, yields
By completing the squares, one obtains
Hence, the time derivative is negative definite in the domain . Moreover, for any positive gain coefficients , in the control law (33), by taking the ratio of the positive design parameters , within the Lyapunov function small enough, one can obtain the values of , which are arbitrary close to .
Thus, the equilibrium point , of the system (28) under the control (33) is asymptotically stable with the domain of attraction , which is positively invariant (see, e.g., [19]).
Notice that the difference between the integrator backstepping control law (33) and the basic feedback linearization-based design (29) under and is in the presence of the extra term in (33), which resulted in the desired boundedness with the required bounds property of the transients.
Finally, in summary, in view of the relationships and (33), one obtains the following cerebral blood flow autoregulation mathematics:
The numerical simulation results of the cerebral blood flow autoregulation design (34) performance under the model parameter values indicated in Table 1 are shown in Figure 2, Figure 3, Figure 4, Figure 5, Figure 6 and Figure 7. First, the arterial pressure steady state value mmHg, which is deviated from the basal quantity mmHg of a healthy adult [4], was considered. Figure 2, Figure 3, Figure 4 and Figure 5 demonstrate the autoregulation response to high arterial pressure for various control gain coefficients , and the selected bounds , based on initial values of the system state variables.

Figure 2.
Arterial–arteriolar blood flow rate q time behavior (solid red line for , , , ; solid blue line for , , , ; solid green line for , , , ) and its reference value (dashed line).

Figure 3.
Arterial–arteriolar blood volume time behavior (solid red line for , , , ; solid blue line for , , , ; solid green line for , , , ) and its reference value (dashed line).

Figure 4.
Intracranial pressure time behavior (solid red line for , , , ; solid blue line for , , , ; solid green line for , , , ) and its reference value (dashed line).
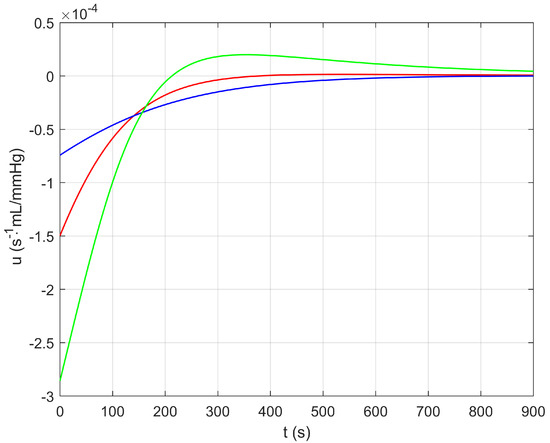
Figure 5.
Control input (arterial–arteriolar compliance rate) versus time (solid red line for , , , ; solid blue line for , , , ; solid green line for , , , ).

Figure 6.
Sensitivity of arterial–arteriolar blood flow rate q autoregulation response to changes in systemic arterial pressure steady state values (solid red line for a step decrease to mmHg, , , , ; solid blue line for a step decrease to mmHg, , , , ; solid green line for a step increase to mmHg, , , , ; solid magenta line for a step increase to mmHg, , , , ; solid black line for a step increase to mmHg, , , , ; dashed line for the reference value ).

Figure 7.
Arterial–arteriolar blood flow rate q autoregulation response (solid black line) to a step systemic arterial pressure increase to mmHg under , , , (dashed line for the reference value ).
Figure 6 and Figure 7 illustrate the sensitivity of the arterial–arteriolar blood flow rate q autoregulation response to changes in systemic arterial pressure steady state values. All arterial pressure changes started from the basal steady state value mmHg.
Notice that the simulation results show the good performance and flexibility of the cerebral blood flow autoregulation scheme (34). By adjusting parameters , and , of the control law (33), one can provide medically reasonable transients within required regulation times and bounds. Even if one control parameter set fails to yield satisfactory autoregulation responses for the whole range of arterial blood pressure values mmHg, as shown in Figure 6, one can readily adjust, e.g., the gain coefficient , as demonstrated in Figure 7, to obtain reasonable cerebral blood flow autoregulation time behavior.
5. Discussion
In this paper, we proposed to treat the cerebral blood flow autoregulation modeling problem as an output tracking automatic control problem. This is the first time, at least to our knowledge, that the cerebral blood flow lumped parameter model introduced in [4] was studied as a dynamical system with control input and its controllability properties were analyzed. It was shown that the cerebral hemodynamics model in question is differentially flat. This fact reveals good potential for applying a variety of automatic control tools to design the impaired cerebral autoregulation compensation mathematical algorithms. Within the current research, due to the model’s nonlinearity, such a well-known and effective nonlinear control approach as integrator backstepping combined with barrier Lyapunov functions was used to construct the control laws that recover the cerebral autoregulation performance of a healthy human. Additionally, the backstepping-based design in question can be further strengthened to account not only for the model nonlinearities but also for various model uncertainties; see, e.g., [17].
The cerebral autoregulation model was developed in terms of arterial–arteriolar compliance time behavior in the form of a feedback control law that utilizes information on the intracranial pressure and arterial–arteriolar blood volume time profiles. It is worthwhile to notice that the intracranial pressure and arterial–arteriolar blood volume values are not available for direct measurements during clinical maneuvers. Hence, future research can be focused on the estimation of the unmeasured quantities using, e.g., state observer construction techniques [20] based on measurements of the arterial–arteriolar blood flow rate values.
Author Contributions
Conceptualization, A.G., A.K. and R.L.; methodology, A.G. and A.K.; formal analysis, A.G.; investigation, A.G.; validation, A.G.; writing—original draft preparation, A.G. and A.K.; writing—review and editing, A.G., A.K. and R.L.; supervision, R.L.; project administration, R.L.; funding acquisition, R.L. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the Klaus Tschira Foundation (grant number 00.302.2016), Buhl-Strohmaier Foundation, Würth Foundation and by the Russian Foundation of Basic Research (project 20-07-00279).
Acknowledgments
The authors are thankful to Varvara Turova for the valuable comments and suggestions.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Hambleton, G.; Wigglesworth, J.S. Origin of intraventricular haemorrhage in the preterm infant. Arch. Dis. Child. 1976, 51, 651–659. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Volpe, J.J. Neurology of the Newborn; Elsevier Health Sciences: Amsterdam, The Netherlands, 2008. [Google Scholar]
- Paulson, O.B.; Strandgaard, S.; Edvinsson, L. Cerebral autoregulation. Cerebrovasc. Brain Metab. Rev. 1990, 2, 161–192. [Google Scholar] [PubMed]
- Ursino, M.; Lodi, C.A. A simple mathematical model of the interaction between intracranial pressure and cerebral hemodynamics. J. Appl. Physiol. 1997, 82, 1256–1269. [Google Scholar] [CrossRef] [PubMed]
- Olufsen, M.S.; Nadim, A.; Lipsitz, L.A. Dynamics of cerebral blood flow regulation explained using a lumped parameter model. Am. J. Physiol. Regulatory Integrative Comp. Physiol. 2002, 282, R611–R622. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Spronck, B.; Martens, H.J.; Gommer, E.D.; Vosse, F.N. A lumped parameter model of cerebral blood flow control combining cerebral autoregulation and neurovascular coupling. Am. J. Physiol. Heart Circ. Physiol. 2012, 303, H1143–H1153. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lampe, R.; Botkin, N.; Turova, V.; Blumenstein, T.; Alves-Pinto, A. Mathematical modelling of cerebral blood circulation and cerebral autoregulation: Towards preventing intracranial hemorrhages in preterm newborns. Comp. Math. Meth. Med. 2014, 2014, 965275. [Google Scholar] [CrossRef] [PubMed]
- Botkin, N.; Turova, V.; Lampe, R. Feedback control of impaired cerebral autoregulation in preterm infants: Mathematical modelling. In Proceedings of the 25th Mediterranean Conference on Control and Automation (MED), Valletta, Malta, 3–6 July 2017; pp. 229–234. [Google Scholar]
- Botkin, N.D.; Kovtanyuk, A.E.; Turova, V.L.; Sidorenko, I.N.; Lampe, R. Direct modeling of blood flow through the vascular network of the germinal matrix. Comput. Biol. Med. 2018, 92, 147–155. [Google Scholar] [CrossRef] [PubMed]
- Botkin, N.D.; Kovtanyuk, A.E.; Turova, V.L.; Sidorenko, I.N.; Lampe, R. Accounting for tube hematocrit in modeling of blood flow in cerebral capillary networks. Comp. Math. Meth. Med. 2019, 2019, 4235937. [Google Scholar] [CrossRef] [PubMed]
- Payne, S. Cerebral Autoregulation. Control of Blood Flow in the Brain; Springer: Berlin/Heidelberg, Germany, 2016. [Google Scholar]
- Botkin, N.; Turova, V.; Diepolder, J.; Bittner, M.; Holzapfel, F. Aircraft control during cruise flight in windshear conditions: Viability approach. Dyn. Games Appl. 2017, 7, 594–608. [Google Scholar] [CrossRef] [Green Version]
- Fliess, M.; Lévine, J.; Martin, P.; Rouchon, P. Flatness and defect of non-linear systems: Introductory theory and examples. Int. J. Contr. 1995, 61, 1327–1361. [Google Scholar] [CrossRef] [Green Version]
- Chetverikov, V.N. Controllability of flat systems. Diff. Eq. 2007, 43, 1558–1568. [Google Scholar] [CrossRef]
- Isidori, A. Nonlinear Control Systems, 3rd ed.; Springer: Berlin/Heidelberg, Germany, 1995. [Google Scholar]
- Rangel-Castilla, L.; Gopinath, S.; Robertson, C.S. Management of intracranial hypertension. Neurol. Clin. 2008, 26, 521–541. [Google Scholar] [CrossRef] [PubMed]
- Krstić, M.; Kanellakopoulos, I.; Kokotović, P.V. Nonlinear and Adaptive Control Design; John Wiley and Sons: Hoboken, NJ, USA, 1995; p. 563. [Google Scholar]
- Ngo, K.B.; Mahony, R.; Jiang, Z.P. Integrator backstepping using barrier functions for systems with multiple state constraints. In Proceedings of the 44th IEEE Conference on Decision and Control, and the European Control Conference, Seville, Spain, 12–15 December 2005; pp. 8306–8312. [Google Scholar]
- Khalil, H.K. Nonlinear Systems, 3rd ed.; Prentice Hall: Hoboken, NJ, USA, 2002. [Google Scholar]
- Besançon, G. Nonlinear Observers and Applications; Springer: Berlin/Heidelberg, Germany, 2007. [Google Scholar]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).







