Analyzing the Dynamics of a Periodic Typhoid Fever Transmission Model with Imperfect Vaccination
Abstract
1. Introduction
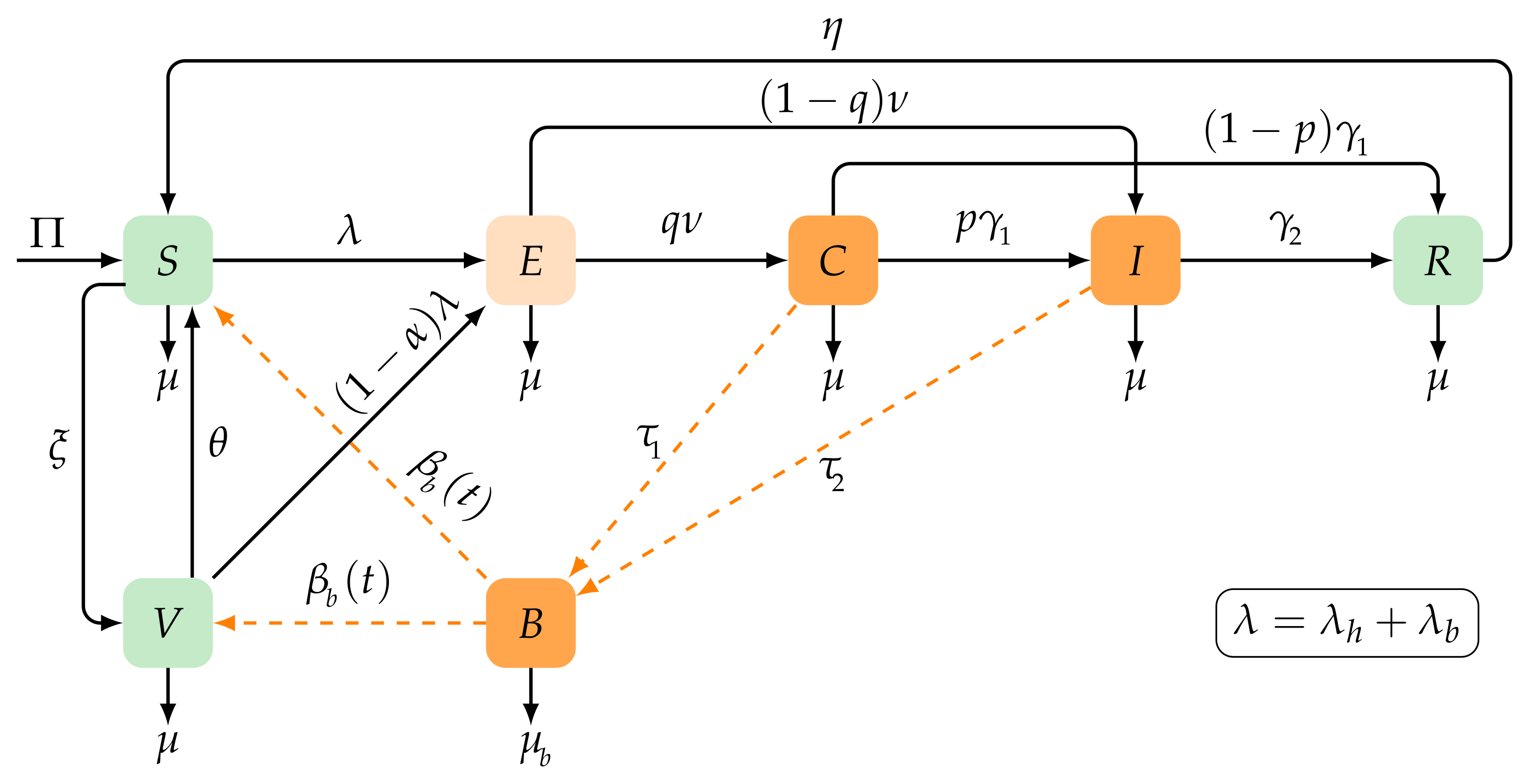
2. Seasonal Typhoid Fever Model
2.1. Basic Properties
2.2. Disease-Free Solution
3. Threshold Dynamics
3.1. Basic Reproduction Number
- 1.
- if and only if ;
- 2.
- if and only if .
3.2. Local Stability of
3.3. Global Stability of
3.4. Existence of Positive Periodic Solutions
4. Model Calibration, Sensitivity Analysis, and Prediction
4.1. Model Fitting to Typhoid Fever Data from Taiwan
4.2. Sensitivity Analysis
4.3. Prediction of New Typhoid Infections in Taiwan
5. Numerical Simulations
5.1. Extinction and Persistence
5.2. Vaccine Coverage and Efficiency
6. Discussion and Conclusions
- Identifying key parameters: Through a sensitivity analysis, our study identified crucial factors that influence typhoid spread. Public health departments can prioritize interventions and allocate resources accordingly. Taking steps to reduce human-to-human transmission rates and improve hygiene and sanitation practices helps control transmission.
- Predicting future epidemics: Our periodic model and simulations allow the prediction of future typhoid epidemics. Public health departments can anticipate and prepare for outbreaks by implementing preventive measures such as a better surveillance, public awareness campaigns, and improved response strategies.
- Vaccination: The study emphasized the importance of vaccination in reducing cases of typhoid. Public health departments can use this information to develop targeted vaccination campaigns, focusing on areas with low coverage. Increasing vaccination rates and ensuring access to effective vaccines can mitigate the risk of typhoid outbreaks.
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- James, S.L.; Abate, D.; Abate, K.H.; Abay, S.M.; Abbafati, C.; Abbasi, N.; Abbastabar, H.; Abd-Allah, F.; Abdela, J.; Abdelalim, A.; et al. Global, regional, and national incidence, prevalence, and years lived with disability for 354 diseases and injuries for 195 countries and territories, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet 2018, 392, 1789–1858. [Google Scholar]
- Crump, J.A.; Sjölund-Karlsson, M.; Gordon, M.A.; Parry, C.M. Epidemiology, clinical presentation, laboratory diagnosis, antimicrobial resistance, and antimicrobial management of invasive Salmonella infections. Clin. Microbiol. Rev. 2015, 28, 901–937. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention. Typhoid Fever and Paratyphoid Fever. Available online: https://www.cdc.gov/typhoid-fever/index.html (accessed on 1 June 2023).
- World Health Organization. Typhoid. Available online: https://www.who.int/news-room/fact-sheets/detail/typhoid (accessed on 1 June 2023).
- Mogasale, V.; Maskery, B.; Ochiai, R.L.; Lee, J.S.; Mogasale, V.V.; Ramani, E.; Kim, Y.E.; Park, J.K.; Wierzba, T.F. Burden of typhoid fever in low-income and middle-income countries: A systematic, literature-based update with risk-factor adjustment. Lancet Glob. Health 2014, 2, e570–e580. [Google Scholar] [CrossRef] [PubMed]
- Bhutta, Z.A. Current concepts in the diagnosis and treatment of typhoid fever. BMJ 2006, 333, 78–82. [Google Scholar] [CrossRef]
- Ackers, M.L.; Puhr, N.D.; Tauxe, R.V.; Mintz, E.D. Laboratory-based surveillance of Salmonella serotype Typhi infections in the United States: Antimicrobial resistance on the rise. JAMA 2000, 283, 2668–2673. [Google Scholar] [CrossRef]
- Mirza, S.; Beechmg, N.; Hart, C. Multi-drug resistant typhoid: A global problem. J. Med Microbiol. 1996, 44, 317–319. [Google Scholar] [CrossRef]
- L’Organisation mondiale de la Santé; World Health Organization. Typhoid vaccines: WHO position paper–March 2018–Vaccins antityphoïdiques: Note de synthèse de l’OMS–mars 2018. Wkly. Epidemiol. Rec. = Relevé Épidémiologique Hebd. 2018, 93, 153–172. [Google Scholar]
- Mushayabasa, S. A simple epidemiological model for typhoid with saturated incidence rate and treatment effect. Int. J. Math. Comput. Sci. 2013, 6, 688–695. [Google Scholar]
- Edward, S.; Nyerere, N. Modelling typhoid fever with education, vaccination and treatment. Eng. Math. 2016, 1, 44–52. [Google Scholar]
- Karunditu, J.W.; Kimathi, G.; Osman, S. Mathematical modeling of typhoid fever disease incorporating unprotected humans in the spread dynamics. J. Adv. Math. Comput. Sci. 2019, 32, 1–11. [Google Scholar] [CrossRef]
- Mutua, J.M.; Wang, F.B.; Vaidya, N.K. Modeling malaria and typhoid fever co-infection dynamics. Math. Biosci. 2015, 264, 128–144. [Google Scholar] [CrossRef]
- Tilahun, G.T.; Makinde, O.D.; Malonza, D. Co-dynamics of pneumonia and typhoid fever diseases with cost effective optimal control analysis. Appl. Math. Comput. 2018, 316, 438–459. [Google Scholar] [CrossRef]
- González-Guzmán, J. An epidemiological model for direct and indirect transmission of typhoid fever. Math. Biosci. 1989, 96, 33–46. [Google Scholar] [CrossRef] [PubMed]
- Mushanyu, J.; Nyabadza, F.; Muchatibaya, G.; Mafuta, P.; Nhawu, G. Assessing the potential impact of limited public health resources on the spread and control of typhoid. J. Math. Biol. 2018, 77, 647–670. [Google Scholar] [CrossRef]
- Shaikh, A.S.; Nisar, K.S. Transmission dynamics of fractional order Typhoid fever model using Caputo–Fabrizio operator. Chaos Solitons Fractals 2019, 128, 355–365. [Google Scholar] [CrossRef]
- Mushayabasa, S. Modeling the impact of optimal screening on typhoid dynamics. Int. J. Dyn. Control 2016, 4, 330–338. [Google Scholar] [CrossRef]
- Edward, S. A deterministic mathematical model for direct and indirect transmission dynamics of typhoid fever. Open Access Libr. J. 2017, 4, 75873. [Google Scholar] [CrossRef]
- Tilahun, G.T.; Makinde, O.D.; Malonza, D. Modelling and optimal control of typhoid fever disease with cost-effective strategies. Comput. Math. Methods Med. 2017, 2017, 2324518. [Google Scholar] [CrossRef]
- Peter, O.J.; Ibrahim, M.O.; Edogbanya, H.O.; Oguntolu, F.A.; Oshinubi, K.; Ibrahim, A.A.; Ayoola, T.A.; Lawal, J.O. Direct and indirect transmission of typhoid fever model with optimal control. Results Phys. 2021, 27, 104463. [Google Scholar] [CrossRef]
- Musa, S.S.; Zhao, S.; Hussaini, N.; Usaini, S.; He, D. Dynamics analysis of typhoid fever with public health education programs and final epidemic size relation. Results Appl. Math. 2021, 10, 100153. [Google Scholar] [CrossRef]
- Mondal, J. Influence of awareness programs by media in the typhoid fever: A study based on mathematical modeling. J. Math. Model. 2018, 6, 1–26. [Google Scholar]
- Pitzer, V.E.; Bowles, C.C.; Baker, S.; Kang, G.; Balaji, V.; Farrar, J.J.; Grenfell, B.T. Predicting the impact of vaccination on the transmission dynamics of typhoid in South Asia: A mathematical modeling study. PLoS Negl. Trop. Dis. 2014, 8, e2642. [Google Scholar] [CrossRef] [PubMed]
- Abboubakar, H.; Kombou, L.K.; Koko, A.D.; Fouda, H.P.E.; Kumar, A. Projections and fractional dynamics of the typhoid fever: A case study of Mbandjock in the Centre Region of Cameroon. Chaos Solitons Fractals 2021, 150, 111129. [Google Scholar] [CrossRef]
- Sinan, M.; Shah, K.; Kumam, P.; Mahariq, I.; Ansari, K.J.; Ahmad, Z.; Shah, Z. Fractional order mathematical modeling of typhoid fever disease. Results Phys. 2022, 32, 105044. [Google Scholar] [CrossRef]
- Abboubakar, H.; Racke, R. Mathematical modeling, forecasting, and optimal control of typhoid fever transmission dynamics. Chaos Solitons Fractals 2021, 149, 111074. [Google Scholar] [CrossRef]
- Peter, O.; Ibrahim, M.; Oguntolu, F.; Akinduko, O.; Akinyemi, S. Direct and indirect transmission dynamics of typhoid fever model by differential transform method. J. Sci. Technol. Educ. 2018, 6, 167–177. [Google Scholar]
- Pitzer, V.E.; Feasey, N.A.; Msefula, C.; Mallewa, J.; Kennedy, N.; Dube, Q.; Denis, B.; Gordon, M.A.; Heyderman, R.S. Mathematical modeling to assess the drivers of the recent emergence of typhoid fever in Blantyre, Malawi. Clin. Infect. Dis. 2015, 61, S251–S258. [Google Scholar] [CrossRef]
- Syed, K.A.; Saluja, T.; Cho, H.; Hsiao, A.; Shaikh, H.; Wartel, T.A.; Mogasale, V.; Lynch, J.; Kim, J.H.; Excler, J.L.; et al. Review on the recent advances on typhoid vaccine development and challenges ahead. Clin. Infect. Dis. 2020, 71, S141–S150. [Google Scholar] [CrossRef]
- Wang, W.; Zhao, X.Q. Threshold dynamics for compartmental epidemic models in periodic environments. J. Dyn. Differ. Equ. 2008, 20, 699–717. [Google Scholar] [CrossRef]
- Tian, J.P.; Wang, J. Some results in Floquet theory, with application to periodic epidemic models. Appl. Anal. 2015, 94, 1128–1152. [Google Scholar] [CrossRef]
- Zhang, F.; Zhao, X.Q. A periodic epidemic model in a patchy environment. J. Math. Anal. Appl. 2007, 325, 496–516. [Google Scholar] [CrossRef]
- Smith, H.L.; Waltman, P. The Theory of the Chemostat: Dynamics of Microbial Competition; Cambridge University Press: Cambridge, UK, 1995. [Google Scholar]
- Zhao, X.Q. Dynamical Systems in Population Biology, 2nd ed.; Springer: New York, NY, USA, 2017. [Google Scholar]
- Zhao, X.Q. Dynamical Systems in Population Biology; Springer: New York, NY, USA, 2003. [Google Scholar]
- Geoba.se. Population Website. Available online: http://www.geoba.se/country.php?cc=TW&year=2023 (accessed on 1 June 2023).
- Taiwan National Infectious Disease Statistics System. Typhoid Fever. Available online: https://nidss.cdc.gov.tw/en/SingleDisease.aspx?dc=1&dt=2&disease=002 (accessed on 1 June 2023).
- McKay, M.D.; Beckman, R.J.; Conover, W.J. Comparison of three methods for selecting values of input variables in the analysis of output from a computer code. Technometrics 1979, 21, 239–245. [Google Scholar]
- Mitchell, C.; Kribs, C. A comparison of methods for calculating the basic reproductive number for periodic epidemic systems. Bull. Math. Biol. 2017, 79, 1846–1869. [Google Scholar] [CrossRef]
- Hornick, R.; Greisman, S.; Woodward, T.; DuPont, H.; Hawkins, A.; Snyder, M. Typhoid fever: Pathogenesis and immunologic control. N. Engl. J. Med. 1970, 283, 739–746. [Google Scholar] [CrossRef]
- Wain, J.; Diep, T.S.; Ho, V.A.; Walsh, A.M.; Hoa, N.T.T.; Parry, C.M.; White, N.J. Quantitation of bacteria in blood of typhoid fever patients and relationship between counts and clinical features, transmissibility, and antibiotic resistance. J. Clin. Microbiol. 1998, 36, 1683–1687. [Google Scholar] [CrossRef]
- Mushayabasa, S.; Bhunu, C.P.; Mhlanga, N.A. Modeling the transmission dynamics of typhoid in malaria endemic settings. Appl. Appl. Math. Int. J. (AAM) 2014, 9, 9. [Google Scholar]
- Cho, J.C.; Kim, S.J. Viable, but non-culturable, state of a green fluorescence protein-tagged environmental isolate of Salmonella typhi in groundwater and pond water. FEMS Microbiol. Lett. 1999, 170, 257–264. [Google Scholar] [CrossRef] [PubMed]
- Saltelli, A. Sensitivity analysis for importance assessment. Risk Anal. 2002, 22, 579–590. [Google Scholar] [CrossRef]
- Arriola, L.; Hyman, J.M. Sensitivity analysis for uncertainty quantification in mathematical models. In Mathematical and Statistical Estimation Approaches in Epidemiology; Springer: Dordrecht, The Netherlands, 2009; pp. 195–247. [Google Scholar]
- Van den Driessche, P.; Watmough, J. Reproduction numbers and sub-threshold endemic equilibria for compartmental models of disease transmission. Math. Biosci. 2002, 180, 29–48. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention. Vaccine Information Statements (VISs). Typhoid VIS. Available online: https://www.cdc.gov/vaccines/hcp/vis/vis-statements/typhoid.html (accessed on 1 June 2023).
- Irena, T.K.; Gakkhar, S. A dynamical model for HIV-typhoid co-infection with typhoid vaccine. J. Appl. Math. Comput. 2021, 67, 641–670. [Google Scholar] [CrossRef]










| Parameters | Description |
|---|---|
| Human birth rate | |
| Natural mortality rate | |
| Human-to-human transmission rates | |
| Environment to human infection rate | |
| Bacteria excretion (carriers, infectious) | |
| Recovery rates | |
| Rate of progression to carriers | |
| q | Probability of exposed group E transitioning into carriers C |
| p | Probability of carriers C to become infected I |
| Disease-induced mortality | |
| Rate of transition from recovered to susceptible classes | |
| Vaccine efficacy | |
| Vaccination waning rate | |
| Vaccination rate | |
| Bacterial decay rate |
| Parameters | Baseline Values | Range | Units | Source |
|---|---|---|---|---|
| - | Persons per day | [37] | ||
| - | [37] | |||
| 0–0.1 | Per day | [22,29] | ||
| 0–0.1 | Per day | [22,29] | ||
| 0–1 | Per day | [24,29] | ||
| 0–0.1 | Per day | [24,29] | ||
| 0–1 | - | [24,29] | ||
| 1–10 | - | [24,29] | ||
| p | 0–1 | [22,27] | ||
| q | 0–1 | [22,27] | ||
| 0–1 | [22,27] | |||
| 0–1 | [41,42] | |||
| 0–0.5 | [41] | |||
| 0.5–0.956 | [27,30] | |||
| 0.00833–0.6 | [41,43] | |||
| 0.05–0.5 | [27,29] | |||
| 0.001–0.5 | [29,44] |
| Parameter | ||||||
| Sensitivity index | ||||||
| Parameter | q | |||||
| Sensitivity index | ||||||
| Parameter | p | |||||
| Sensitivity index |
| Parameters | |||||||
| Values | |||||||
| Parameters | p | q | |||||
| Values | |||||||
| Parameters | |||||||
| Values | 2.04735 |
| Parameters | |||||||
| Values | |||||||
| Parameters | p | q | |||||
| Values | |||||||
| Parameters | |||||||
| Values |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alharbi, M.H.; Alalhareth, F.K.; Ibrahim, M.A. Analyzing the Dynamics of a Periodic Typhoid Fever Transmission Model with Imperfect Vaccination. Mathematics 2023, 11, 3298. https://doi.org/10.3390/math11153298
Alharbi MH, Alalhareth FK, Ibrahim MA. Analyzing the Dynamics of a Periodic Typhoid Fever Transmission Model with Imperfect Vaccination. Mathematics. 2023; 11(15):3298. https://doi.org/10.3390/math11153298
Chicago/Turabian StyleAlharbi, Mohammed H., Fawaz K. Alalhareth, and Mahmoud A. Ibrahim. 2023. "Analyzing the Dynamics of a Periodic Typhoid Fever Transmission Model with Imperfect Vaccination" Mathematics 11, no. 15: 3298. https://doi.org/10.3390/math11153298
APA StyleAlharbi, M. H., Alalhareth, F. K., & Ibrahim, M. A. (2023). Analyzing the Dynamics of a Periodic Typhoid Fever Transmission Model with Imperfect Vaccination. Mathematics, 11(15), 3298. https://doi.org/10.3390/math11153298








