Determination of Phenethyl Isothiocyanate, Erucin, Iberverin, and Erucin Nitrile Concentrations in Healthy and Pieris rapae-Infected Broccoli Tissues Using Gas Chromatography-Mass Spectrometry
Abstract
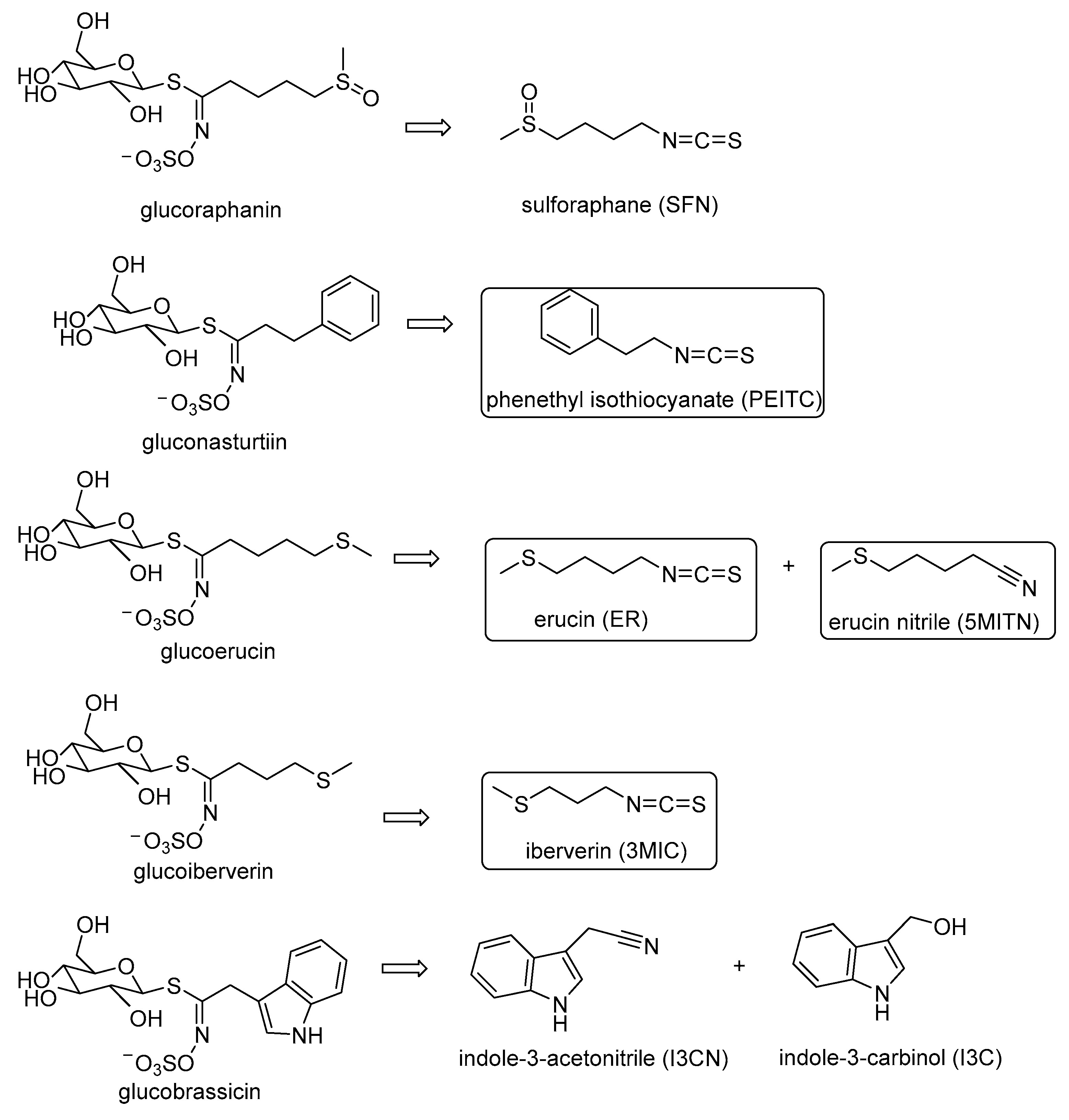
:1. Introduction
2. Materials and Methods
2.1. Reagents and Materials
2.2. Standard Solutions
2.3. Sampling
2.4. GLS Hydrolysis and Extraction Conditions
2.5. Analysis of Glucosinolate Hydrolysis Products
2.5.1. GC-MS Method
2.5.2. Validation, Linearity, and Detection Limits
3. Results
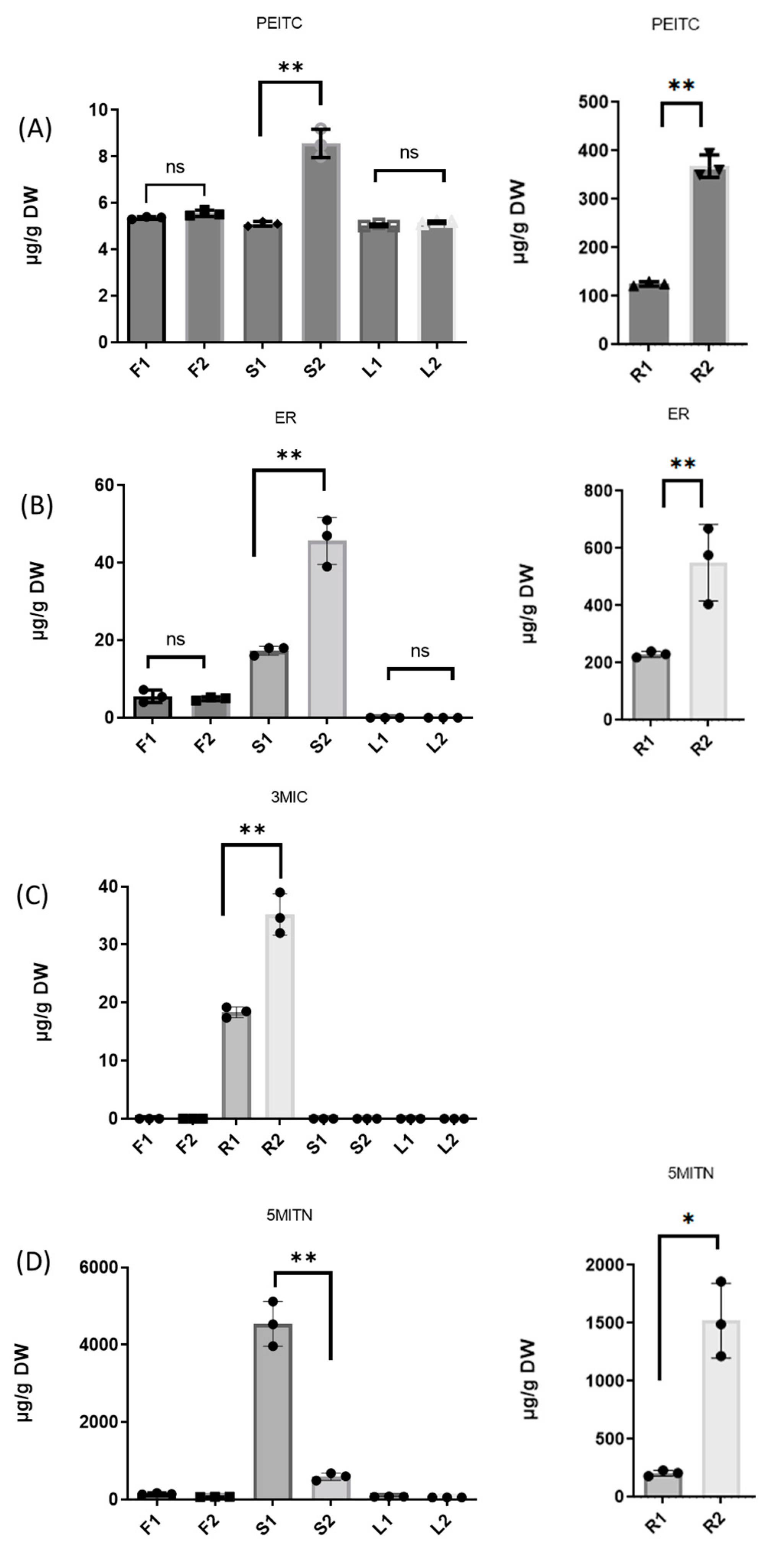
3.1. Broccoli Plants
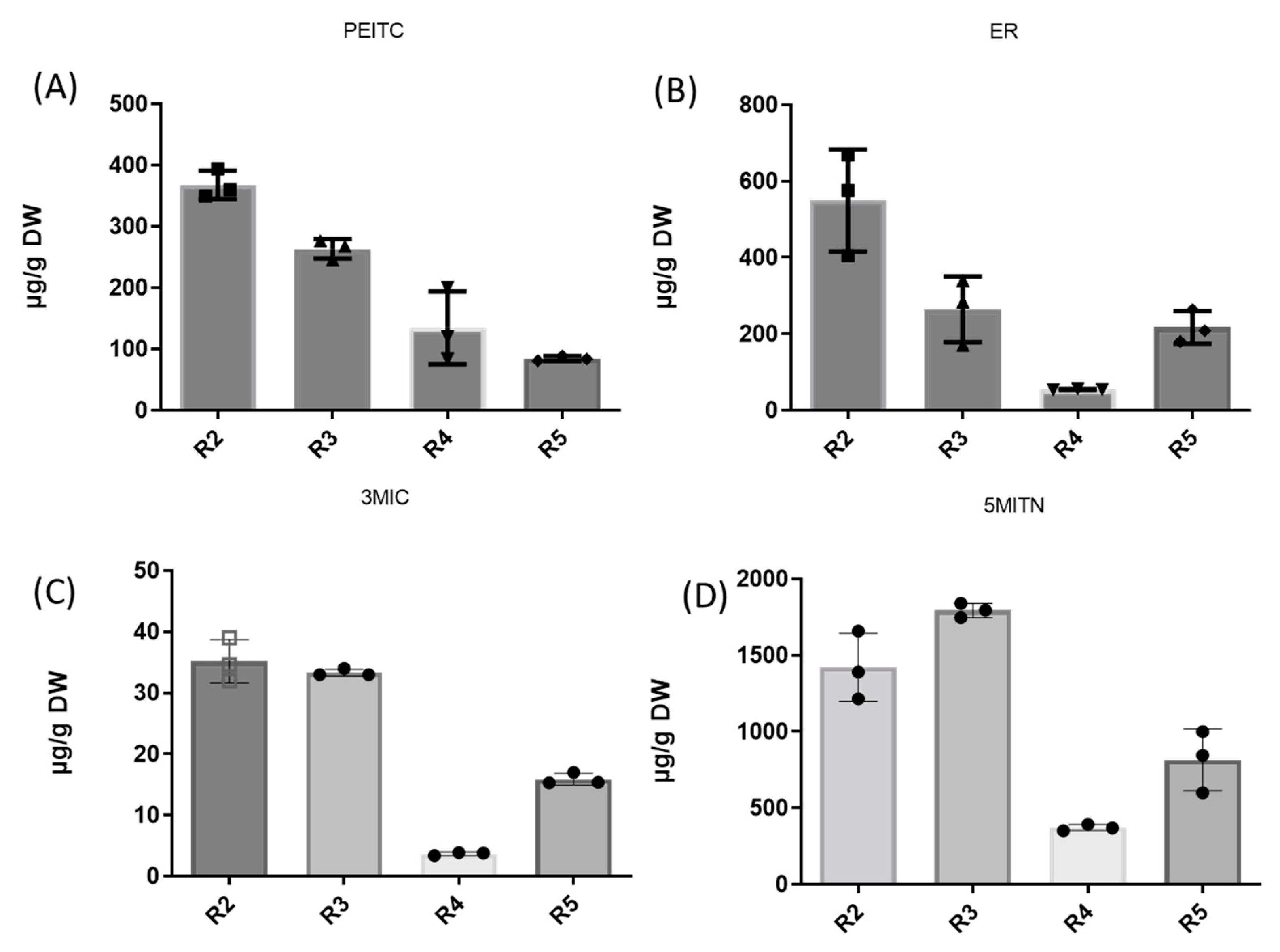
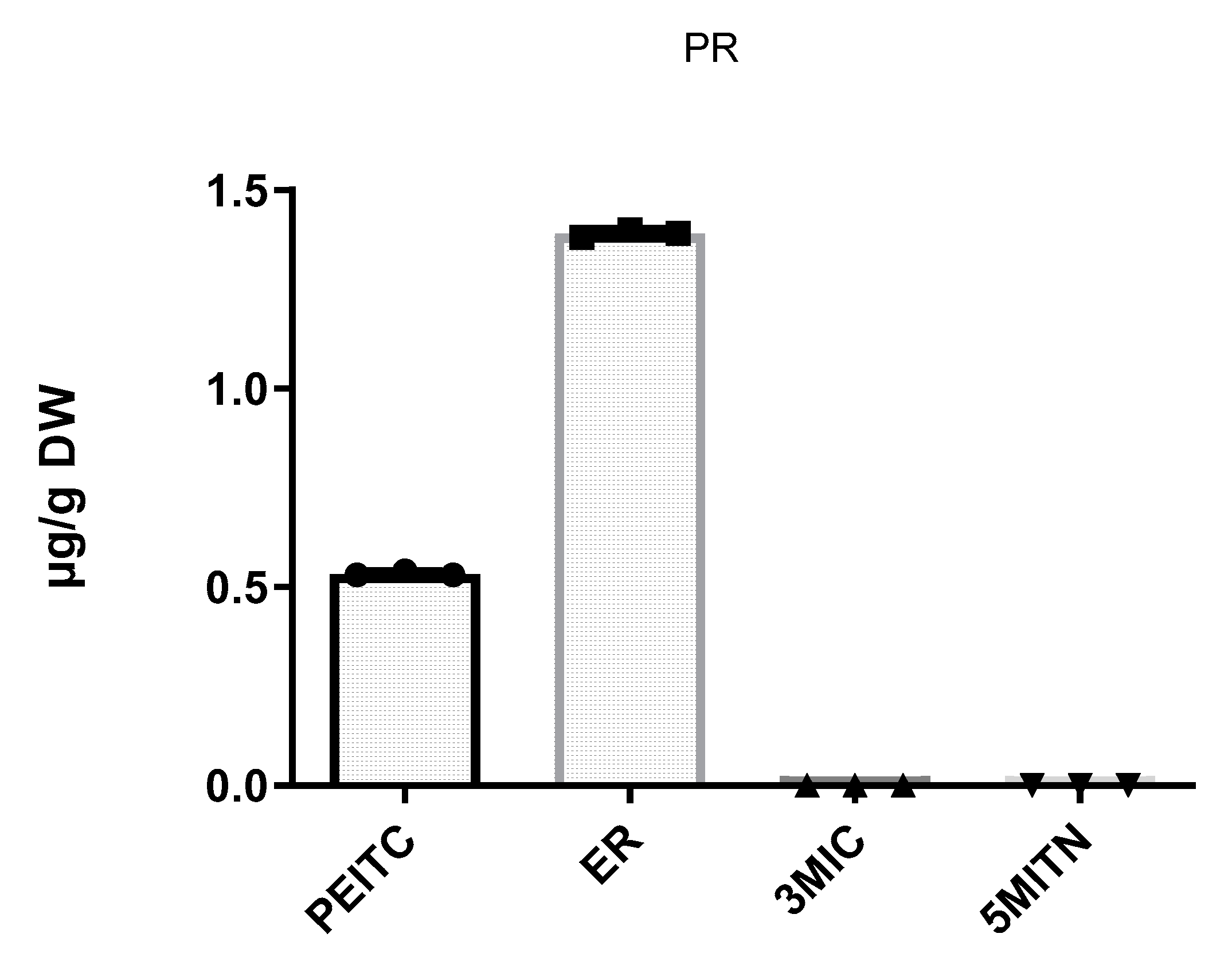
3.2. Extraction of Isothiocyanates PEITC, ER, 3MIC, and Nitrile 5MITN from Broccoli Samples, and Determination via GC-MS
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Shen, Y.; Wang, J.; Shaw, R.K.; Yu, H.; Sheng, X.; Zhao, Z.; Li, S.; Gu, H. Development of GBTS and KASP panels for genetic diversity, population structure, and fingerprinting of a large collection of broccoli (Brassica oleracea L. var. italica) in China. Front. Plant Sci. 2021, 12, 655254. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, V.P.T.; Stewart, J.; Lopez, M.; Ioannou, I.; Allais, F. Glucosinolates: Natural occurrence, biosynthesis, accessibility, isolation, structures, and biological activities. Molecules 2020, 25, 4537. [Google Scholar] [CrossRef]
- Li, H.; Xia, Y.; Liu, H.-Y.; Guo, H.; He, X.-Q.; Liu, Y.; Wu, D.-T.; Mai, Y.-H.; Li, H.-B.; Zou, L.; et al. Nutritional values, beneficial effects, and food applications of broccoli (Brassica oleracea var. italica Plenck). Trends Food Sci. Technol. 2022, 119, 288–308. [Google Scholar] [CrossRef]
- Wittstock, U.; Kurzbach, E.; Herfurth, A.-M.; Stauber, E. Glucosinolate breakdown. Adv. Bot. Res. 2016, 80, 125–169. [Google Scholar]
- Ilahy, R.; Tlili, I.; Pek, Z.; Montefusco, A.; Siddiqui, M.W.; Homa, F.; Hdider, C.; R’Him, T.; Lajos, H.; Lenucci, M.S. Pre- and post-harvest factors affecting glucosinolate content in broccoli. Front. Nutr. 2020, 7, 147. [Google Scholar] [CrossRef] [PubMed]
- Palliyaguru, D.L.; Yuan, J.M.; Kensler, T.W.; Fahey, J.W. Isothiocyanates: Translating the power of plants to people. Mol. Nutr. Food Res. 2018, 62, 1700965. [Google Scholar] [CrossRef] [PubMed]
- Eagles, S.K.; Gross, A.S.; McLachlan, A.J. The effects of cruciferous vegetable-enriched diets on drug metabolism: A systematic review and meta-analysis of dietary intervention trials in humans. Clin. Pharmacol. Therap. 2020, 108, 212–227. [Google Scholar] [CrossRef] [PubMed]
- Radošević, K.; Srček, V.G.; Bubalo, M.C.; Brnčić, S.R.; Takács, K.; Redovniković, I.R. Assessment of glucosinolates, antioxidative and antiproliferative activity of broccoli and collard extracts. J. Food Comp. 2017, 61, 59–66. [Google Scholar] [CrossRef]
- Kaiser, A.E.; Baniasadi, M.; Giansiracusa, D.; Giansiracusa, M.; Garcia, M.; Fryda, Z.; Wong, T.L.; Bishayee, A. Sulforaphane: A broccoli bioactive phytocompound with cancer preventive potential. Cancers 2021, 13, 4796. [Google Scholar] [CrossRef]
- Zhang, Y.; Talalay, P.; Cho, C.G.; Posner, G.H. A major inducer of anticarcinogenic protective enzymes from broccoli: Isolation and elucidation of structure. Proc. Natl. Acad. Sci. USA 1992, 89, 2399–2403. [Google Scholar] [CrossRef]
- Ernst Insa, M.A.; Palani, K.; Esatbeyoglu, T.; Schwarz, K.; Rimbach, G. Synthesis and Nrf2-inducing activity of the isothiocyanates iberverin, iberin and cheirolin. Pharmacol. Res. 2013, 70, 155–162. [Google Scholar] [CrossRef] [PubMed]
- Melchini, A.; Traka, M.H.; Catania, S.; Miceli, N.; Taviano, M.F.; Maimone, P.; Francisco, M.; Mithen, R.F.; Costa, C. Antiproliferative activity of the dietary isothiocyanate erucin, a bioactive compound from cruciferous vegetables, on human prostate cancer cells. Nutr. Cancer 2013, 65, 132–138. [Google Scholar] [CrossRef] [PubMed]
- Manici, L.M.; Lazzeri, L.; Baruzzi, G.; Leoni, O.; Galletti, S.; Palmieri, S. Suppressive activity of some glucosinolate enzyme degradation products on Pythium irregulare and Rhizoctonia solani in sterile soil. Pest Manag. Sci. 2000, 56, 921–926. [Google Scholar] [CrossRef]
- Crowley, E.; Rowan, N.J.; Faller, D.; Friel, A.M. Natural and synthetic isothiocyanates possess anticancer potential against liver and prostate cancer in vitro. Anticancer Res. 2019, 39, 3469–3485. [Google Scholar] [CrossRef] [PubMed]
- Chuang, W.-T.; Yun-Ta, L.; Huang, C.-S.; Lo, C.-W.; Yao, H.-T.; Chen, H.-W.; Lii, C.-K. Benzyl isothiocyanate and phenethyl isothiocyanate inhibit adipogenesis and hepatosteatosis in mice with obesity induced by a high-fat diet. J. Agric. Food Chem. 2019, 67, 7136–7146. [Google Scholar] [CrossRef] [PubMed]
- Neubauer, C.; Heitmann, B.; Müller, C. Biofumigation potential of Brassicaceae cultivars to Verticillium dahliae. Eur. J. Plant Pathol. 2014, 140, 341–352. [Google Scholar] [CrossRef]
- Hooker, W.J.; Walker, J.C.; Smith, F.G. Toxicity of beta-phenethyl isothiocyanate to certain fungi. Am. J. Bot. 1943, 30, 632–637. [Google Scholar]
- Wilson, A.E.; Bergaentzlé, M.; Bindler, F.; Marchioni, E.; Lintz, A.; Ennahar, S. In vitro efficacies of various isothiocyanates from cruciferous vegetables as antimicrobial agents against foodborne pathogens and spoilage bacteria. Food Control 2013, 30, 318–324. [Google Scholar] [CrossRef]
- Baena, R.; Salinas, H. Cancer chemoprevention by dietary phytochemicals: Epidemiological evidence. Maturitas 2016, 94, 13–19. [Google Scholar] [CrossRef]
- Belitz, H.-D.; Grosch, W.; Schieberle, P. Food Chemistry, 4th ed.; Springer: Berlin/Heidelberg, Germany, 2009. [Google Scholar]
- Ghawi, S.K.; Methven, L.; Niranjan, K. The potential to intensify sulforaphane formation in cooked broccoli (Brassica oleracea var. italica) using mustard seeds (Sinapis alba). Food Chem. 2013, 138, 1734–1741. [Google Scholar] [CrossRef]
- Torres-Contreras, A.M.; Nair, V.; Cisneros-Zevallos, L.; Jacobo-Velázquez, D.A. Stability of bioactive compounds in broccoli as affected by cutting styles and storage time. Molecules 2017, 22, 636. [Google Scholar] [CrossRef] [PubMed]
- Xiao, J.; Pei, Y.; Qi, A.; Jingnan, R.; Gang, F.; Zelan, W.; Xixiang, L.; Siyi, P. Identification of glucosinolates and volatile odor compounds in microwaved radish (Raphanus sativus L.) seeds and the corresponding oils by UPLC-IMS-QTOF-MS and GC × GC-qMS analysis. Food Res. Int. 2023, 169, 112873. [Google Scholar]
- Hanschen, F.S.; Schreiner, M. Isothiocyanates, nitriles, and epithionitriles from glucosinolates are affected by genotype and developmental stage in Brassica oleracea varieties. Front. Plant Sci. 2017, 8, 1095. [Google Scholar] [CrossRef] [PubMed]
- Aires, A.; Rosa, E.; Carvalho, R. Effect of nitrogen and sulfur fertilization on glucosinolates in the leaves and roots of broccoli sprouts (Brassica oleracea var. italica). J. Sci. Food Agric. 2006, 86, 1512–1516. [Google Scholar] [CrossRef]
- Zhang, Y.; Makaza, N.; Jiang, C.; Wu, Y.; Nishanbaev, S.Z.; Zou, L.; Sun, J.; Song, X.; Wu, Y. Supplementation of cooked broccoli with exogenous moringa myrosinase enhanced isothiocyanate formation. Food Chem. 2022, 395, 133651. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.G.; Lim, S.; Kim, J.; Lee, E.J. The mechanism of deterioration of the glucosinolate-myrosynase system in radish roots during cold storage after harvest. Food Chem. 2017, 233, 60–68. [Google Scholar] [CrossRef] [PubMed]
- Krishna Moorthy, P.N.; Prasannakumar, N.R.; Mani, M.; Saroja, S.; Ranganath, H.R. Pests and their management in cruciferous vegetables (cabbage, cauliflower, knol khol, broccoli, radish, turnip, beet root). In Trends in Horticultural Entomology; Mani, M., Ed.; Spinger: Berlin/Heidelberg, Germany, 2022; pp. 997–1011. [Google Scholar]
- Renwick, J.A.A.; Radke, C.D.; Sachdev-Gupta, K.; Städler, E. Leaf surface chemicals stimulating oviposition by Pieris rapae (Lepidoptera: Pieridae) on cabbage. Chemoecology 1992, 3, 33–38. [Google Scholar] [CrossRef]
- Van Loon, J.J.A.; Blaakmeer, A.; Griepink, F.C.; Van Beek, T.A.; Schoonhoven, L.M.; De Groot, A. Leaf surface compound from Brassica oleracea (Cruciferae) induces oviposition by Pieris brassicae (Lepidoptera: Pieridae). Chemoecology 1992, 3, 39–44. [Google Scholar] [CrossRef]
- Huang, X.; Renwick, J.A.A. Relative activities of glucosinolates as oviposition stimulants for Pieris rapae and P. napi oleracea. J. Chem. Ecol. 1994, 20, 1025–1037. [Google Scholar] [CrossRef]
- Miles, C.I.; del Campo, M.L.; Renwick, J.A. Behavioral and chemosensory responses to a host recognition cue by larvae of Pieris rapae. J. Comp. Physiol. A Neuroethol. Sens. Neural Behav. Physiol. 2005, 191, 147–155. [Google Scholar] [CrossRef]
- Stadler, E.; Renwick, J.; Radke, C.D.; Sachdev-Gupta, K. Tarsal contact chemoreceptor response to glucosinolates and cardenolides mediating oviposition in Pieris rape. Physiol. Entomol. 1995, 20, 175–187. [Google Scholar] [CrossRef]
- Badenes-Pérez, F.R. Plant glucosinolate content and host-plant preference and suitability in the small white butterfly (Lepidoptera: Pieridae) and comparison with another specialist lepidopteran. Plants 2023, 12, 2148. [Google Scholar] [CrossRef] [PubMed]
- Kokotou, M.G.; Revelou, P.K.; Pappas, C.; Constantinou-Kokotou, V. High resolution mass spectrometry studies of sulforaphane and indole-3-carbinol in broccoli. Food Chem. 2017, 237, 566–573. [Google Scholar] [CrossRef] [PubMed]
- Ciska, E.; Drabinska, N.; Honke, J.; Narwojsz, A. Boiled Brussels sprouts: A rich source of glucosinolates and the corresponding nitriles. J. Funct Foods 2015, 19, 91–99. [Google Scholar] [CrossRef]
- Karanikolopoulou, S.; Revelou, P.-K.; Xagoraris, M.; Kokotou, M.G.; Constantinou-Kokotou, V. Current Methods for the extraction and analysis of isothiocyanates and indoles in cruciferous vegetables. Analytica 2021, 2, 93–120. [Google Scholar] [CrossRef]
- Arora, R.; Sharma, D.; Kumar, R.; Singh, B.; Vig, A.P.; Arora, S. Evaluating extraction conditions of glucosinolate hydrolytic products from seeds of Eruca sativa (Mill.) Thell. using GC-MS. J. Food Sci. 2014, 79, C1964–C1969. [Google Scholar] [CrossRef] [PubMed]
- Mukerjee, A.K.; Ashare, R. Isothiocyanates in the chemistry of heterocycles. Chem. Rev. 1991, 91, 1–24. [Google Scholar] [CrossRef]
- FAOSTAT. Food and Agriculture Organization Corporate Statistical Database. 2021. Available online: http://www.fao.org/faostat/en/#data/QC/visualize (accessed on 5 October 2023).
- Wittstock, U.; Agerbirk, N.; Stauber, E.J.; Olsen, C.E.; Hippler, M.; Mitchell-Olds, T.; Gershenzon, J.; Vogel, H. Successful herbivore attack due to metabolic diversion of a plant chemical defense. Proc. Natl. Acad. Sci. USA 2004, 101, 4859–4864. [Google Scholar] [CrossRef]
- Kaur, S.; Mahesh Kumar Samota, M.K.; Choudhary, M.; Choudhary, M.; Pandey, A.K.; Sharma, A.; Thakur, J. How do plants defend themselves against pathogens-Biochemical mechanisms and genetic interventions. Physiol. Mol. Biol. Plants 2022, 28, 485–504. [Google Scholar] [CrossRef]
- Itoh, Y.; Okumura, Y.; Fujii, T.; Ishikawa, Y.; Omura, H. Effects of mating on host selection by female small white butterflies Pieris rapae (Lepidoptera: Pieridae). J. Comp. Phys. A 2018, 204, 245–255. [Google Scholar] [CrossRef]
- Lund, M.; Brainard, D.C.; Szendrei, Z. Cue hierarchy for host plant selection in Pieris rapae. Entomol. Exp. Appl. 2019, 167, 330–340. [Google Scholar] [CrossRef]
- Yang, J.; Guo, H.; Jiang, N.-J.; Tang, R.; Li, G.-C.; Huang, L.-Q.; Van Loon, J.J.; Wang, C.-Z. Identification of a gustatory receptor tuned to sinigrin in the cabbage butterfly Pieris rapae. PLoS Genet. 2021, 17, e1009527. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Hernández, M.C.; Medina, S.; Gil-Izquierdo, A.; Martínez-Ballesta, C.; Moreno, D.A. Broccoli isothiocyanates content and in vitro availability according to variety and origin. Mac. J. Chem. Chem. Eng. 2013, 32, 251–264. [Google Scholar] [CrossRef]
- Brown, P.D.; Morra, M.J. Control of soil-borne plant pests using glucosinolate-containing plants. Adv. Agron. 1997, 61, 167–231. [Google Scholar]
- Rosa, E.A.S.; Heaney, R.K.; Fenwick, G.R.; Portas, C.A.M. Glucosinolates in Crop Plants; Horticultural Reviews; Wiley & Sons, Inc.: Hoboken, NJ, USA, 1997. [Google Scholar]
- Burow, M.; Losansky, A.; Muller, R.; Plock, A.; Kliebenstein, D.J.; Wittstock, U. The genetic basis of constitutive and herbivore-induced ESP-independent nitrile formation in arabidopsis. Plant Physiol. 2009, 149, 561–574. [Google Scholar] [CrossRef]
- Eugui, D.; Velasco, P.; Abril-Urías, P.; Escobar, C.; Gómez-Torres, O.; Caballero, S.; Poveda, J. Glucosinolate-extracts from residues of conventional and organic cultivated broccoli leaves (Brassica oleracea var. italica) as potential industrially-scalable efficient biopesticides against fungi, oomycetes and plant parasitic nematodes. Ind. Crops Prod. 2003, 200, 116841. [Google Scholar] [CrossRef]
- Szymandera-Buszka, K.; Piechocka, J.; Zaremba, A.; Przeor, M.; Jędrusek-Golinska, A. Pumpkin, cauliflower and broccoli as new carriers of thiamine compounds for food fortification. Foods 2021, 10, 578. [Google Scholar] [CrossRef]
- Shi, M.; Ying, D.-Y.; Hlaing, M.M.; Ye, J.-H.; Sanguansri, L.; Augustin, M.A. Development of broccoli by-products as carriers for delivering EGCG. Food Chem. 2019, 301, 125301. [Google Scholar] [CrossRef]
- Krupa-Kozak, U.; Drabinska, N.; Rosell, C.M.; Fadda, C.; Anders, A.; Jelinski, T.; Ostaszyk, A. Broccoli leaf powder as an attractive by-product ingredient: Effect on batter behaviour, technological properties and sensory quality of gluten-free mini sponge cake. Int. J. Food Sci. Technol. 2019, 54, 1121–1129. [Google Scholar] [CrossRef]
- Krupa-Kozak, U.; Drabinska, N.; Bączek, N.; Simkova, K.; Starowicz, M.; Jelinski, T. Application of broccoli leaf powder in gluten-free bread: An innovative approach to improve its bioactive potential and technological quality. Foods 2021, 10, 819. [Google Scholar] [CrossRef]
| Mean Value ± SD μg/g DW | ||||
|---|---|---|---|---|
| Broccoli Tissue | PEITC | ER | 3MIC | 5MITN |
| F1 | 5.3 ± 0.06 | 5.1 ± 1.86 | n.d | 137.2 ± 5.73 |
| F2 | 5.6 ± 0.14 | 5.0 ± 0.49 | n.d. | 72.6 ± 9.92 |
| R1 | 126.7 ± 4.83 | 230.5 ± 10.14 | 18.5 ± 1.07 | 208.3 ± 27.98 |
| R2 | 367.6 ± 23.43 | 576.3 ± 148.51 | 34.6 ± 3.74 | 1491.1 ± 331.55 |
| S1 | 5.1 ± 0.02 | 18.0 ± 1.37 | n.d. | 4534.7 ± 578.98 |
| S2 | 8.7 ± 0.73 | 47.4 ± 7.62 | n.d. | 604.1 ± 99.25 |
| L1 | 5.0 ± 0.01 | n.d. | n.d. | 75.5 ± 3.94 |
| L2 | 5.2 ± 0.03 | n.d. | n.d. | 56.8 ± 1.17 |
| Mean Value ± SD µg/g DW | ||||
|---|---|---|---|---|
| Broccoli Root | PEITC | ER | 3MIC | 5MITN |
| R2 | 367.6 ± 23.43 | 576.3 ± 148.51 | 34.6 ± 3.74 | 1491.1 ± 331.55 |
| R3 | 272.8 ± 112.53 | 284.0 ± 108.71 | 33.1 ± 0.66 | 1795.1 ± 65.60 |
| R4 | 134.3 ± 46.40 | 54.7 ± 1.67 | 3.9 ± 0.08 | 371.3 ± 21.03 |
| R5 | 86.5 ± 2.43 | 209.0 ± 71.68 | 15.4 ± 2.52 | 846.2 ± 270.02 |
| Mean ± SD | 215 ± 90.21 | 280.75 ± 120.54 | 21.75 ± 8.76 | 1125.75 ± 540.11 |
| Mean Value ± SD μg/g DW | ||||
|---|---|---|---|---|
| PEITC | ER | 3MIC | 5MITN | |
| Pieris rapae | 0.5 ± 0.00 | 1.4 ± 0.02 | n.d. | n.d. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mesimeri, I.-D.; Revelou, P.-K.; Constantinou-Kokotou, V.; Kokotou, M.G. Determination of Phenethyl Isothiocyanate, Erucin, Iberverin, and Erucin Nitrile Concentrations in Healthy and Pieris rapae-Infected Broccoli Tissues Using Gas Chromatography-Mass Spectrometry. Chemosensors 2024, 12, 16. https://doi.org/10.3390/chemosensors12010016
Mesimeri I-D, Revelou P-K, Constantinou-Kokotou V, Kokotou MG. Determination of Phenethyl Isothiocyanate, Erucin, Iberverin, and Erucin Nitrile Concentrations in Healthy and Pieris rapae-Infected Broccoli Tissues Using Gas Chromatography-Mass Spectrometry. Chemosensors. 2024; 12(1):16. https://doi.org/10.3390/chemosensors12010016
Chicago/Turabian StyleMesimeri, Irene-Dimitra, Panagiota-Kyriaki Revelou, Violetta Constantinou-Kokotou, and Maroula G. Kokotou. 2024. "Determination of Phenethyl Isothiocyanate, Erucin, Iberverin, and Erucin Nitrile Concentrations in Healthy and Pieris rapae-Infected Broccoli Tissues Using Gas Chromatography-Mass Spectrometry" Chemosensors 12, no. 1: 16. https://doi.org/10.3390/chemosensors12010016
APA StyleMesimeri, I. -D., Revelou, P. -K., Constantinou-Kokotou, V., & Kokotou, M. G. (2024). Determination of Phenethyl Isothiocyanate, Erucin, Iberverin, and Erucin Nitrile Concentrations in Healthy and Pieris rapae-Infected Broccoli Tissues Using Gas Chromatography-Mass Spectrometry. Chemosensors, 12(1), 16. https://doi.org/10.3390/chemosensors12010016










