Abbreviations
AAT, aortic arch thrombosis; AIS, acute ischemic stroke; AMI, acute myocardial infarction; ANA, antinuclear antibodies; APL, acute promyelocytic leukemia; APLA, antiphospholipid antibody; ARDS, acute respiratory distress syndrome; BCS, Budd-Chiari syndrome; CVST, cerebral venous sinus thrombosis; DES, diffuse encephalopathic stroke; DIC, disseminated intravascular coagulation; “DIC”, false disseminated intravascular coagulation; DVT, deep venous thrombosis; ECs, endothelial cells; EVT, extravascular tissue; HERNS, hereditary endotheliopathy, retinopathy, nephropathy and stroke; HIT-WCS, heparin-induced thrombocytopenia with white clot syndrome; HUS, hemolytic-uremic syndrome; IFN, interferon; IL, interleukin; ITP, immune thrombocytopenic purpura; ITP-LS, ITP-like syndrome; MAC, membrane attack complex; MAHA, microangiopathic hemolytic anemia; IVCT/SVCT, inferior vena cava thrombosis/superior vena cava thrombosis; MODS, multiorgan dysfunction syndrome, MOIS, multiorgan inflammatory syndrome; MVMI, microvascular myocardial infarction; NETs, neutrophil extracellular traps; PF4, platelet factor 4; PNH, paroxysmal nocturnal hemoglobinuria; PT, pulmonary thrombosis/prothrombin time; PTE, pulmonary thromboembolism; aPTT, activated partial thromboplastin time; PVT, portal vein thrombosis; RAT, renal artery thrombosis; SET, subendothelial tissue; SOS, sinusoidal obstruction syndrome; SPG, symmetrical peripheral gangrene; TCIP, thrombocytopenia in critically ill patients; TF, tissue factor; TIA, transient ischemic attack; TNF, tumor necrosis factor; TTP, thrombotic thrombocytopenic purpura; ULVWF, ultra-large von Willebrand factor; VCCS, venous circulatory congestion syndrome; VMTD, vascular microthrombotic disease; EA-VMTD, endotheliopathy-associated VMTD; aEA-VMTD, arterial EA-VMTD; vEA-VMTD, venous EA-VMTD; VOD, veno-occlusive disease; VTE, venous thromboembolism; VWF, von Willebrand factor.
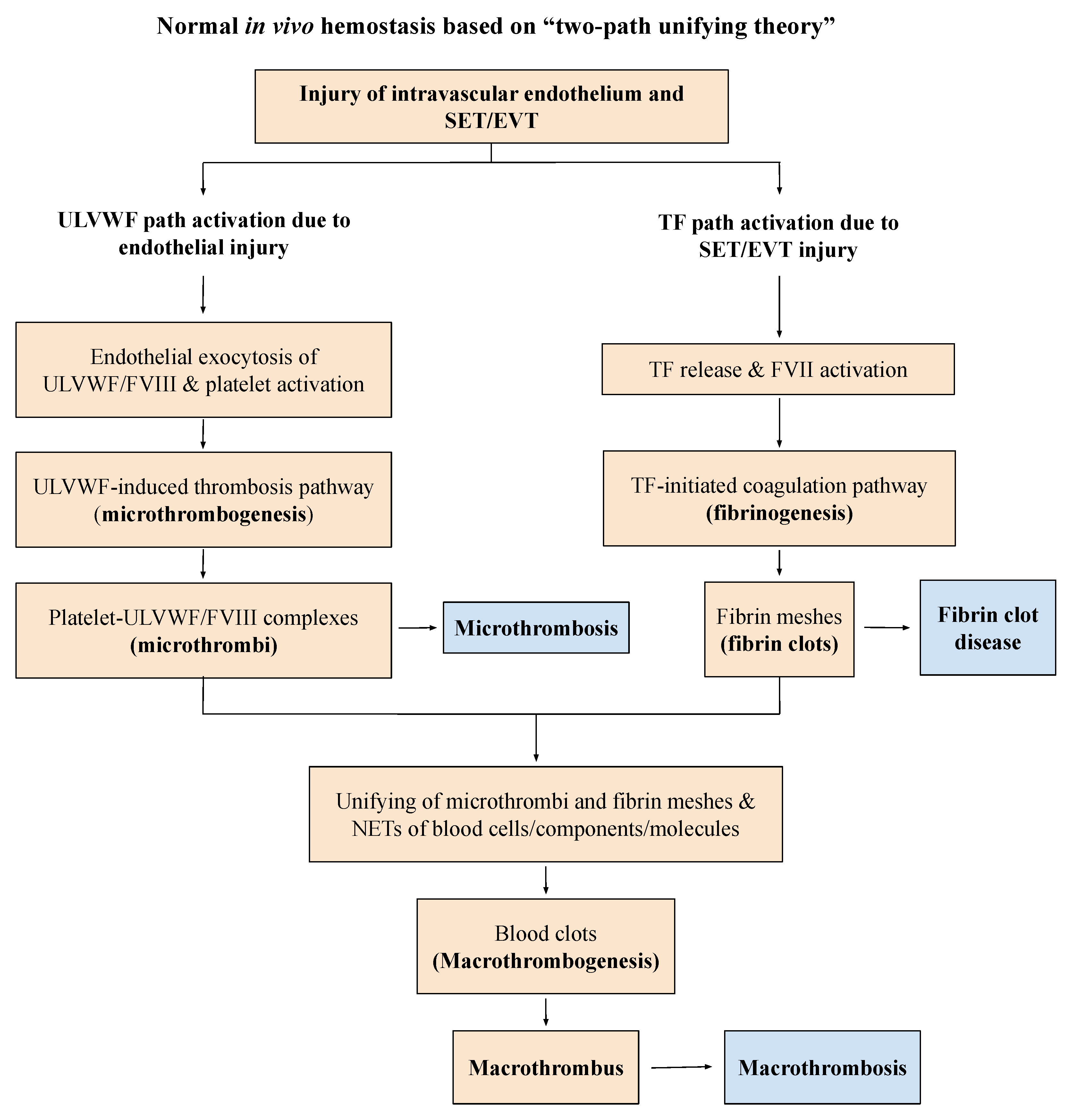
Figure 1.
Normal hemostasis in vivo based on “two-path unifying theory” (Reproduced and modified with permission from Chang JC. Thrombosis Journal. 2019;17:10). Following a vascular injury, hemostatic system in vivo activates two independent sub-hemostatic paths: microthrombotic (ULVWF path) and fibrinogenetic (TF path). The former is initiated by the damage of ECs and the latter by that of SET/EVT in external bodily injury and intravascular injury. In activated ULVWF path, ULVWF/FVIII are released and recruit platelets, and produce microthrombi strings via microthrombogenesis, but in activated TF path, TF is released and activates FVII. The TF-FVIIa complex produces fibrin meshes via fibrinogenesis of extrinsic coagulation cascade. The final path of in vivo hemostasis is macrothrombogenesis, in which microthrombi strings and fibrin meshes become unified together with the incorporation of NETs, including red blood cells, neutrophils, DNAs and histones. This unifying event “macrothrombogenesis” promotes the hemostatic plug and wound healing in external bodily injury and produces macrothrombus causing macrothrombosis in intravascular injury. Abbreviations: EA-VMTD, endotheliopathy-associated vascular microthrombotic disease: ECs, endothelial cells; EVT, extravascular tissue; NETs, neutrophil extracellular traps; SET, subendothelial tissue; TF, tissue factor; ULVWF, ultra large von Willebrand factor multimers.
Figure 1.
Normal hemostasis in vivo based on “two-path unifying theory” (Reproduced and modified with permission from Chang JC. Thrombosis Journal. 2019;17:10). Following a vascular injury, hemostatic system in vivo activates two independent sub-hemostatic paths: microthrombotic (ULVWF path) and fibrinogenetic (TF path). The former is initiated by the damage of ECs and the latter by that of SET/EVT in external bodily injury and intravascular injury. In activated ULVWF path, ULVWF/FVIII are released and recruit platelets, and produce microthrombi strings via microthrombogenesis, but in activated TF path, TF is released and activates FVII. The TF-FVIIa complex produces fibrin meshes via fibrinogenesis of extrinsic coagulation cascade. The final path of in vivo hemostasis is macrothrombogenesis, in which microthrombi strings and fibrin meshes become unified together with the incorporation of NETs, including red blood cells, neutrophils, DNAs and histones. This unifying event “macrothrombogenesis” promotes the hemostatic plug and wound healing in external bodily injury and produces macrothrombus causing macrothrombosis in intravascular injury. Abbreviations: EA-VMTD, endotheliopathy-associated vascular microthrombotic disease: ECs, endothelial cells; EVT, extravascular tissue; NETs, neutrophil extracellular traps; SET, subendothelial tissue; TF, tissue factor; ULVWF, ultra large von Willebrand factor multimers.
![Biomedicines 10 02706 g001]()
Figure 2.
Pathogenesis of endotheliopathy based on “two-activation theory of the endothelium”. Endothelial molecular pathogenesis is initiated by the activated complement system following exposure to pathogen, toxin, drug, poison, venom, vaccine, polytrauma, hyperglycemia, severe hypertension, and others. Endotheliopathy releases inflammatory cytokines and hemostatic factors, and activates two clinically distinctive molecular pathways: inflammatory and microthrombotic. Both arterial endotheliolpathy and venous endotheliopathy provoke inflammation via inflammatory pathway leading to the inflammatory syndrome called MOIS due to cytokines, but arterial endotheliopathy promotes microthrombosis via microthrombotic pathway and produces aEA-VMTD due to activation of ULVWF path of hemostasis (shown in
Figure 1). Arterial endotheliopathy is characterized by the triad of consumptive thrombocytopenia, MAHA, and MODS, which is called “TTP-like syndrome”, but venous endotheliopathy is characterized by ITP/“ITP-like syndrome” due to silent microthrombi with consumptive thrombocytopenia as explained in the text. The distinguishing features are caused by different anatomy, physiological function and hemodynamic characteristics between arterial system and venous system. These are very important pathological features in the understanding of the complexity of variable thrombotic phenotypes occurring endotheliopathic syndromes. Abbreviations: EA-VMTD, endotheliopathy-associated vascular microthrombotic disease; ITP, immune thrombocytopenic purpura; MAC, membrane attack complex; IFN, interferon; IL, interleukin; MAHA, microangiopathic hemolytic anemia; MODS, multiorgan dysfunction syndrome; MOIS, multiorgan inflammatory syndrome; TNF, tumor necrosis factor; TTP, thrombotic thrombocytopenic purpura; aEA-VMTD, arterial EA-VMTD; vEA-VMTD, venous EA-VMTD; ULVWF, ultra large von Willebrand factor.
Figure 2.
Pathogenesis of endotheliopathy based on “two-activation theory of the endothelium”. Endothelial molecular pathogenesis is initiated by the activated complement system following exposure to pathogen, toxin, drug, poison, venom, vaccine, polytrauma, hyperglycemia, severe hypertension, and others. Endotheliopathy releases inflammatory cytokines and hemostatic factors, and activates two clinically distinctive molecular pathways: inflammatory and microthrombotic. Both arterial endotheliolpathy and venous endotheliopathy provoke inflammation via inflammatory pathway leading to the inflammatory syndrome called MOIS due to cytokines, but arterial endotheliopathy promotes microthrombosis via microthrombotic pathway and produces aEA-VMTD due to activation of ULVWF path of hemostasis (shown in
Figure 1). Arterial endotheliopathy is characterized by the triad of consumptive thrombocytopenia, MAHA, and MODS, which is called “TTP-like syndrome”, but venous endotheliopathy is characterized by ITP/“ITP-like syndrome” due to silent microthrombi with consumptive thrombocytopenia as explained in the text. The distinguishing features are caused by different anatomy, physiological function and hemodynamic characteristics between arterial system and venous system. These are very important pathological features in the understanding of the complexity of variable thrombotic phenotypes occurring endotheliopathic syndromes. Abbreviations: EA-VMTD, endotheliopathy-associated vascular microthrombotic disease; ITP, immune thrombocytopenic purpura; MAC, membrane attack complex; IFN, interferon; IL, interleukin; MAHA, microangiopathic hemolytic anemia; MODS, multiorgan dysfunction syndrome; MOIS, multiorgan inflammatory syndrome; TNF, tumor necrosis factor; TTP, thrombotic thrombocytopenic purpura; aEA-VMTD, arterial EA-VMTD; vEA-VMTD, venous EA-VMTD; ULVWF, ultra large von Willebrand factor.
![Biomedicines 10 02706 g002]()
Figure 3.
Schematic illustration of cross section of blood vessel histology and hemostatic components (Reproduced and modified with permission from Chang JC. Clin Appl Thromb Hemost 2019 Jan-Dec; 25:1076029619887437). The blood vessel wall is the site of hemostasis (coagulation) in vivo to produce blood clots (hemostatic plug) and stop hemorrhage in the external bodily injury. It is also the site of hemostasis (thrombogenesis) to produce intravascular blood clots (thrombus) in the intravascular injury to cause thrombosis. Its histologic components are divided into the endothelium, tunica intima, tunica media and tunica externa, and each component has its function contributing to molecular hemostasis. As illustrated, ECs damage triggers the exocytosis of ULVWF/FVIII and SET damage promotes the release of sTF from the tunica intima, tunica media and tunica externa. EVT damage releases of eTF from the outside of blood vessel wall. This depth of blood vessel wall injury contributes to the genesis of different thrombotic disorders such as microthrombosis, macrothrombosis, fibrin clots, thrombo-hemorrhagic clots and various endotheliopathic syndromes. This concept based on the blood vessel wall model is especially important in the understanding of different phenotypes of stroke and heart attack. Abbreviations: EVT, extravascular tissue; eTF, extravascular tissue factor; SET, subendothelial tissue; sTF, subendothelial tissue factor; RBC, red blood cells; ULVWF, ultra large von Willebrand factor.
Figure 3.
Schematic illustration of cross section of blood vessel histology and hemostatic components (Reproduced and modified with permission from Chang JC. Clin Appl Thromb Hemost 2019 Jan-Dec; 25:1076029619887437). The blood vessel wall is the site of hemostasis (coagulation) in vivo to produce blood clots (hemostatic plug) and stop hemorrhage in the external bodily injury. It is also the site of hemostasis (thrombogenesis) to produce intravascular blood clots (thrombus) in the intravascular injury to cause thrombosis. Its histologic components are divided into the endothelium, tunica intima, tunica media and tunica externa, and each component has its function contributing to molecular hemostasis. As illustrated, ECs damage triggers the exocytosis of ULVWF/FVIII and SET damage promotes the release of sTF from the tunica intima, tunica media and tunica externa. EVT damage releases of eTF from the outside of blood vessel wall. This depth of blood vessel wall injury contributes to the genesis of different thrombotic disorders such as microthrombosis, macrothrombosis, fibrin clots, thrombo-hemorrhagic clots and various endotheliopathic syndromes. This concept based on the blood vessel wall model is especially important in the understanding of different phenotypes of stroke and heart attack. Abbreviations: EVT, extravascular tissue; eTF, extravascular tissue factor; SET, subendothelial tissue; sTF, subendothelial tissue factor; RBC, red blood cells; ULVWF, ultra large von Willebrand factor.
![Biomedicines 10 02706 g003]()
Figure 4.
Pathogenesis of thrombosis producing its primary and secondary phenotypes based on “two-path unifying theory of hemostasis. Thrombogenesis following an intravascular injury may take two different paths. For example, in sepsis the intravascular injury is limited to ECs but disseminated. The damage releases ULVWF/FVIII and activates ULVWF path (microthrombotic pathway) that produces microthrombi strings, eventually leading to microthrombosis (e.g., aEA-VMTD). However, in a local trauma the intravascular injury is extended from ECs to SET/EVT but localized, which releases TF and small amount of ULVWF/FVIII and activates TF path (traumatic pathway) that produces blood clots composed of fibrin meshes, small amount of ULVWF/FVIII and platelets, and NETs, leading to macrothrombosis (e.g., DVT). Sometimes the septic patient with disseminated microthrombi strings due to endotheliopathy admitted to ICU could also develop another unrelated serious vascular damage to SET/EVT due to an indwelling vascular device in ICU care, which releases TF and promote fibrinogenesis that forms fibrin meshes. In this case, combined micro-macrothrombi composed of ternary components of ULVWF/FVIII, platelets, and fibrin meshes could be formed and lead to combined micro-macrothrombosis shown in the Figure with clinical phenotypes. The thrombosis due to EA-VMTD and another vascular injury is a complex form of micro-macrothrombosis. Their thrombogeneses are logical and has been explained in the text and my previous publications [
14,
25]. Abbreviations: AAT, aortic arch thrombosis; aEA-VMTD, arterial endotheliopathy-associated vascular microthrombotic disease; AIS, acute ischemic stroke; AMI, acute myocardial infarction; ARDS, acute respiratory distress syndrome; BCS, Budd-Chiari syndrome; DES, diffuse encephalopathic stroke; CVST, cerebral venous sinus thrombosis; DVT, deep venous thrombosis; ECs, Endothelial cells; EVT, extravascular tissue; HERNS, hereditary endotheliopathy, retinopathy, retinopathy and stroke syndromes; HUS, hemolytic-uremic syndrome; ICU, intensive care unit; ITP-LS, ITP-like syndrome; IVCT/SVCT, inferior vena cava thrombosis/superior vena cava thrombosis; MODS, multiorgan dysfunction syndrome; NETs, neutrophil extracellular traps; PDIS, peripheral digit ischemic syndrome; PNH, paroxysmal nocturnal hemoglobinuria; PT, pulmonary thrombosis; PTE, pulmonary thromboembolism; PVT, portal vein thrombosis; RAT, renal artery thrombosis; SET, subendothelial tissue; SOS, sinus obstruction syndrome; SPG, symmetrical peripheral gangrene; SVT, splanchnic vein thrombosis; TF, tissue factor; TIA, transient ischemic attack; TTP-LS, TTP-like syndrome; ULVWF, ultra large von Willebrand factor; VOD, veno-occlusive disease; VTE, venous thromboembolism.
Figure 4.
Pathogenesis of thrombosis producing its primary and secondary phenotypes based on “two-path unifying theory of hemostasis. Thrombogenesis following an intravascular injury may take two different paths. For example, in sepsis the intravascular injury is limited to ECs but disseminated. The damage releases ULVWF/FVIII and activates ULVWF path (microthrombotic pathway) that produces microthrombi strings, eventually leading to microthrombosis (e.g., aEA-VMTD). However, in a local trauma the intravascular injury is extended from ECs to SET/EVT but localized, which releases TF and small amount of ULVWF/FVIII and activates TF path (traumatic pathway) that produces blood clots composed of fibrin meshes, small amount of ULVWF/FVIII and platelets, and NETs, leading to macrothrombosis (e.g., DVT). Sometimes the septic patient with disseminated microthrombi strings due to endotheliopathy admitted to ICU could also develop another unrelated serious vascular damage to SET/EVT due to an indwelling vascular device in ICU care, which releases TF and promote fibrinogenesis that forms fibrin meshes. In this case, combined micro-macrothrombi composed of ternary components of ULVWF/FVIII, platelets, and fibrin meshes could be formed and lead to combined micro-macrothrombosis shown in the Figure with clinical phenotypes. The thrombosis due to EA-VMTD and another vascular injury is a complex form of micro-macrothrombosis. Their thrombogeneses are logical and has been explained in the text and my previous publications [
14,
25]. Abbreviations: AAT, aortic arch thrombosis; aEA-VMTD, arterial endotheliopathy-associated vascular microthrombotic disease; AIS, acute ischemic stroke; AMI, acute myocardial infarction; ARDS, acute respiratory distress syndrome; BCS, Budd-Chiari syndrome; DES, diffuse encephalopathic stroke; CVST, cerebral venous sinus thrombosis; DVT, deep venous thrombosis; ECs, Endothelial cells; EVT, extravascular tissue; HERNS, hereditary endotheliopathy, retinopathy, retinopathy and stroke syndromes; HUS, hemolytic-uremic syndrome; ICU, intensive care unit; ITP-LS, ITP-like syndrome; IVCT/SVCT, inferior vena cava thrombosis/superior vena cava thrombosis; MODS, multiorgan dysfunction syndrome; NETs, neutrophil extracellular traps; PDIS, peripheral digit ischemic syndrome; PNH, paroxysmal nocturnal hemoglobinuria; PT, pulmonary thrombosis; PTE, pulmonary thromboembolism; PVT, portal vein thrombosis; RAT, renal artery thrombosis; SET, subendothelial tissue; SOS, sinus obstruction syndrome; SPG, symmetrical peripheral gangrene; SVT, splanchnic vein thrombosis; TF, tissue factor; TIA, transient ischemic attack; TTP-LS, TTP-like syndrome; ULVWF, ultra large von Willebrand factor; VOD, veno-occlusive disease; VTE, venous thromboembolism.
![Biomedicines 10 02706 g004]()
Figure 5.
Pathogenesis of multiple thrombotic disorders encountering in COVID-19 sepsis. Unlike other sepsis, including bacterial, viral, fungal, rickettsial and parasitic sepsis, COVID-19 has been highlighted by the mystery of extensive thrombotic diseases, which has included ARDS, DVT, VTE, PTE, complicated gangrene syndromes such as SPG, limb gangrene and acrocyanosis, which pathogeneses are summarized and thrombotic syndromes are exemplified in the bottom section of the Figure even though all of these syndromes have been rarely but previously described in other sepsis as well. This author does not think these thrombotic syndromes are the unique feature of COVID-19 but more likely have occurred as the result of intensive intervention utilizing numerous vascular devices and accesses in ICU following the directive from hyped psychology of political and medical communities in pandemic and also were highlighted by enthusiastic publications of the cases in medical literature. To emphasize the importance of vascular damage contributing to the heightened role inducing thrombogenesis in ICU setting, I have summarized the pathogenesis of several thrombotic syndromes that have occurred in COVID-19 infection. This Figure can be easily applied to other sepsis in place of COVID-19 sepsis. Abbreviations: ANF, acute necrotizing fasciitis; ARDS, acute respiratory distress syndrome; BCS, Budd-Chiari syndrome; CVST, cerebral venous sinus thrombosis; DVT, deep venous thrombosis; ECs, endothelial cells; EVT, extravascular tissue; ICU, intensive care unit; ITP, immune thrombocytopenic purpura; IVCT/SVCT, inferior vena cava thrombosis/superior vena cava thrombosis; MAHA, microangiopathic hemolytic anemia; MODS, multiorgan dysfuncion syndrome; MMT, micromacrothrombosis; MVT, mesenteric vein thrombosis; PTE, pulmonary thromboembolism; PVT, portal vein thrombosis; SET, subendothelial tissue; SPG, symmetrical peripheral gangrene; SVT, splanchnic vein thrombosis; TF, tissue factor; TTP, thrombotic thrombocytopenic purpura; ULVWF, ultra large von Willebrand factor; aEA-VMTD, arterial endotheliopathy-asasociated vascular microthrombotic disdase; vEA-VMTD, venous EA-VMTD; VCCS, venous circulatory congestion syndrome; VTE, venous throm-boembolism.
Figure 5.
Pathogenesis of multiple thrombotic disorders encountering in COVID-19 sepsis. Unlike other sepsis, including bacterial, viral, fungal, rickettsial and parasitic sepsis, COVID-19 has been highlighted by the mystery of extensive thrombotic diseases, which has included ARDS, DVT, VTE, PTE, complicated gangrene syndromes such as SPG, limb gangrene and acrocyanosis, which pathogeneses are summarized and thrombotic syndromes are exemplified in the bottom section of the Figure even though all of these syndromes have been rarely but previously described in other sepsis as well. This author does not think these thrombotic syndromes are the unique feature of COVID-19 but more likely have occurred as the result of intensive intervention utilizing numerous vascular devices and accesses in ICU following the directive from hyped psychology of political and medical communities in pandemic and also were highlighted by enthusiastic publications of the cases in medical literature. To emphasize the importance of vascular damage contributing to the heightened role inducing thrombogenesis in ICU setting, I have summarized the pathogenesis of several thrombotic syndromes that have occurred in COVID-19 infection. This Figure can be easily applied to other sepsis in place of COVID-19 sepsis. Abbreviations: ANF, acute necrotizing fasciitis; ARDS, acute respiratory distress syndrome; BCS, Budd-Chiari syndrome; CVST, cerebral venous sinus thrombosis; DVT, deep venous thrombosis; ECs, endothelial cells; EVT, extravascular tissue; ICU, intensive care unit; ITP, immune thrombocytopenic purpura; IVCT/SVCT, inferior vena cava thrombosis/superior vena cava thrombosis; MAHA, microangiopathic hemolytic anemia; MODS, multiorgan dysfuncion syndrome; MMT, micromacrothrombosis; MVT, mesenteric vein thrombosis; PTE, pulmonary thromboembolism; PVT, portal vein thrombosis; SET, subendothelial tissue; SPG, symmetrical peripheral gangrene; SVT, splanchnic vein thrombosis; TF, tissue factor; TTP, thrombotic thrombocytopenic purpura; ULVWF, ultra large von Willebrand factor; aEA-VMTD, arterial endotheliopathy-asasociated vascular microthrombotic disdase; vEA-VMTD, venous EA-VMTD; VCCS, venous circulatory congestion syndrome; VTE, venous throm-boembolism.
![Biomedicines 10 02706 g005]()
Table 1.
Conceptual characteristics of the terms: hemostasis, coagulation and thrombosis.
Table 1.
Conceptual characteristics of the terms: hemostasis, coagulation and thrombosis.
| | Hemostasis | Coagulation | Thrombosis + |
|---|
| Term concept | Philosophical | Physiological | Structural |
| Implied meaning | Natural process in vivo | Artificial process in vitro | Pathological process in vivo |
| | | Physiologic process at bleeding site | |
| Involved location | Blood vessel wall | Laboratory test tube | Intravascular lumen |
| | | Vessel wall in extravascular trauma | |
| End products | Hemostatic plug | Fibrin mesh/fibrin clot * | Microthrombi/macrothrombus |
| Critical role in | Vascular wall injury | Coagulation test | Thrombogenesis |
| | | Hemorrhage | |
| Involved components | Endothelium | TF/thromboplastin | ULVWF/FVIII from ECs |
| | SET | Coagulation factors | Platelets from circulation |
| | EVT | Fibrinogen | Coagulation proteins/serine proteases |
| | Blood from circulation | | Fibrinogen |
| | | | TF from SET/EVT |
| End results | Determined by the depth | Determined by participating | Determined by ULVWF/FVIII, platelets, TF, |
| | of vascular damage | coagulation factors | hemostatic factors and unifying mechanism |
| Inciting events | Endotheliopathy | Coagulation tests for PT and aPTT | Microthrombosis (i.e., EA-VMTD) |
| | Vascular injury | | Macrothrombosis (e.g., DVT) |
| | | | Combined micro-macrothrombosis (e.g., VTE) |
Table 2.
Three fundamentals in normal and abnormal hemostasis.
Table 2.
Three fundamentals in normal and abnormal hemostasis.
| (1) Hemostatic principles |
| (1) Hemostasis can be activated only by vascular injury. |
| (2) Hemostasis must be activated through ULVWF path and/or TF path. |
| (3) Hemostasis is the same process in both hemorrhage and thrombosis. |
| (4) Hemostasis is the same process in both arterial thrombosis and venous thrombosis. |
| (5) Level of vascular damage (endothelium/SET/EVT) determines different clinical phenotypes of hemorrhage and thrombosis. |
| (2) Major participating components |
| Components | Origin | Mechanism |
| (1) ECs/SET/EVT | Blood vessel wall/EVT | Protective barrier |
| (2) ULVWF/FVIII | ECs | Endothelial exocytosis/anchoring and microthrombogenesis |
| (3) Platelets | Circulation | Adhesion to ULVWF to form microthrombi/assembling and microthrombogenesis |
| (4) TF | SET and EVT | Release from tissue due to vascular injury/leading to fibrinogenesis |
| (5) Coagulation factors | Circulation | Activation to fibrin mesh and fibrin clot/participating in fibrinogenesis |
| (3) Vascular injury and hemostatic phenotypes |
| Injury-induced damage | Involved hemostatic path | Level of Vascular Injury and examples |
| (1) Endothelium | ULVWF | Level 1 damage—microthrombosis (e.g., TIA [focal]; Heyde’s syndrome [local]; EA-VMTD [disseminated]) |
| (2) Endothelium/SET | ULVWF + sTF | Level 2 damage—macrothrombosis (e.g., AIS; DVT; PTE; AA) |
| (3) Endothelium/SET/EVT | ULVWF + eTF | Level 3 damage—macrothrombosis with hemorrhage (e.g., THS; THMI) |
| (4) EVT alone | eTF | Level e damage—fibrin clot disease (e.g., AHS [e.g., SDH; EDH]; ICH) |
| Hemostatic phenotypes | Causes | Genesis |
| (1) Hemorrhage | External bodily injury | Trauma-induced external bleeding (e.g., accident; assault; self-inflicted) |
| (2) Hematoma | Internal EVT injury | Obtuse trauma-induced bleeding (e.g., tissue and cavitary hematoma) |
| (3) Thrombosis | Intravascular injury | Intravascular injury (e.g., atherosclerosis; sepsis; indwelling catheter; surgery) |
Table 3.
Vascular system contributing to the phenotype expression of thrombosis.
Table 3.
Vascular system contributing to the phenotype expression of thrombosis.
| The depth of intravascular wall injury |
| ECs injury: ULVWF and FVIII release (e.g., sepsis causing EA-VMTD) |
| ECs and SET injury: ULVWF and FVIII and TF release (e.g., vascular trauma causing DVT) |
| ECs and SET/EVT injury: ULVWF and FVIII and TF (e.g., vascular causing THS) |
| The extent of injury affecting vascular tree system |
| Focal/local/multifocal (e.g., TIA, DVT, Susac syndrome) |
| Regional (e.g., vascular trauma, Kasabach-Merritt syndrome) |
| Disseminated (e.g., endothelial damage/dysfunction due to sepsis, vaccination, envenomation and others) |
| The vascular milieux system |
| Venous (e.g., ITP-like syndrome, VOD, DVT, IVCT/SVCT, VTE, PTE, CVST) |
| Arterial (e.g., TTP-like syndrome, arterial thrombosis, AMI, AIS, SPG, diabetic gangrene) |
| Microvasculature (e.g., capillaries, arterioles, venules, and hepatic sinusoids, causing microthrombosis such as ARDS, encephalopathy, ALF, HUS, WFS) |
| Macrovasculature (e.g., aorta, artery and “minute” arteries, and vena cava, and vein, causing macrothrombosis such as aortic aneurysm, AIS, AMI, SPG, DVT, IVC/SVC, CVST) |
| The locality of vascular injury |
| Tropism (e.g., bacteria, virus, fungus, parasite causing organotropism such as WFS syndrome due to N. menningococcus) |
| Endothelial heterogeneity (e.g., gene expression of host in specific vascular system such as ITP-like syndrome in venous system) |
| Trauma site |
| Interaction with non-hemostatic factors |
| Environmental factor (e.g., pathogen, vaccine, venom, toxin, drug, chemical, trauma) |
| Hemostasis altering genetic factor (e.g., thrombophilic genes such as PC, PS, FV-Leiden, antithrombin, ADAMTS13, and others) |
| Hereditary disease (e.g., Fabry’s disease, HERNS syndrome, Degos disease, hereditary hemorrhagic telangiectasia) |
Table 4.
Endotheliopathy in arterial and venous milieux determining the clinical phenotype of thrombotic disorders.
Table 4.
Endotheliopathy in arterial and venous milieux determining the clinical phenotype of thrombotic disorders.
| Clinical Phenotype | Arterial Endotheliopathy | Venous Endothelipathy |
|---|
| Underlying pathology | aEA-VMTD | vEA-VMTD |
| Physiological/hemodynamic difference in vascular milieu | Efferent circulation from the heart (O2 delivery) | Afferent circulation into the heart (CO2 disposal) |
| | Tissue hypoxia (e.g., microthromboangiitis obliterans) | Pulmonary circulatory congestion (e.g., ARDS) |
| | High pressure flow | Low pressure flow |
| | High shear stress | Low shear stress |
| | Capillary and arteriolar microvascular event | Venous and pulmonary microvascular event |
| | Insignificant role of NETosis | Significant role of NETosis |
| Primary cause | | |
| Vascular injury (ECs) | Sepsis-induced arterial microvascular endotheliopathy | Sepsis-induced venous endotheliopathy |
| | | Vaccine-induced venous endotheliopathy |
| Vascular pathology site | Disseminated aEA-VMTD at microvasculature | Transient or “silent” vEA-VMTD at venous system |
| Activated hemostatic path | ULVWF path | ULVWF path |
| Thrombosis component | Microthrombi strings in the microvasculature | Microthrombi strings in venous system |
| Microthrombotic event | Disseminated VMTD | Silent microthrombosis with microthrombolysis |
| Clinical phenotypes | TTP-like syndrome | ITP-like syndrome |
| | consumptive thrombocytopenia | consumptive thrombocytopenia |
| | MAHA | MAHA (mild and rare: e.g., ARDS) |
| | MODS/MOIS | MOIS |
Table 5.
Classification of primary and secondary phenotype examples of the thrombotic disorder.
Table 5.
Classification of primary and secondary phenotype examples of the thrombotic disorder.
| Microthrombosis |
| ● aEA-VMTD |
| Mechanism: via microthrombogenesis from activated ULVWF path due to ECs injury of capillaries and arterioles |
| Disseminated aEA-VMTD (e.g., TTP-like syndrome, MODS, MVMI, microthrombotic encephalopathy, HUS) |
| Regional aEA-VMTD (e.g., Kasabach-Merritt syndrome, Heyde’s syndrome) |
| Multifocal aEA-VMTD (e.g., HERNS syndrome, Susac syndrome, diabetic retinal microaneurysm) |
| Focal aEA-VMTD (e.g., TIA) |
| ● vEA-VMTD |
| Mechanism: via microthrombogenesis from activated ULVWF path due to venous ECs injury |
| Disseminated vEA-VMTD (e.g., “silent” ITP-like syndrome, ARDS) |
| Regional vEA-VMTD (e.g., VOD) |
| Macrothrombosis |
| ● Arterial macrothrombosis |
| Mechanism: via macrothrombogenesis from activated ULVWF path and TF path due to arterial ECs and SET/EVT injury |
| Localized arterial thrombosis (e.g., AIS, THS, AMI, aortic aneurysm-associated thrombosis) |
| Multifocal partially obstructive macrothrombosis (e.g., chronic atherosclerosis?) |
| ● Venous macrothrombosis |
| Mechanism: via macrothrombogenesis from activated ULVWF path and TF path due to venous ECs and SET/EVT injury |
| Localized venous thrombosis (e.g., distal DVT, superficial venous thrombosis) |
| Fibrin clot disease |
| ● Arterial/venous fibrin clot disease |
| Mechanism: via pathologic fibrinogenesis from activated aberrant TF path due to overexpressed TF in APL |
| Disseminated fibrin clot disease and hemorrhagic syndrome (e.g., true DIC in APL) |
| Localized hematoma (?) (e.g., internal bleeding including SDH, EDH, AHS) |
| Combined micro-macrothrombosis (due to two different vascular injuries: e.g., sepsis and vascular injury in ICU ) |
| ● Arterial combined micro-macrothrombosis |
| Mechanism: via combined micro-macrothrombogenesis from activated ULVWF path and TF path in arterial system |
| Peripheral gangrene syndrome (e.g., SPG, PDIS, Buerger’s disease, Fournier’s disease, purpura fulminans, acrocyanosis) |
| ● Venous combined micro-macrothrombosis |
| Mechanism: via combined micro-macrothrombogenesis from activated ULVWF path and TF path in venous system |
| Venous circulatory congestion syndrome (e.g., VTE, PTE, IVCT/SVCT, SVT, BCS, PVT, CVST) |
| Non-hemostatic thrombotic syndromes |
| ● Disseminated VMTD |
| Mechanism: excess of ULVWF due to ADAMTS13 deficiency leading to intravascular microthrombogenesis |
| ADAMTS13 gene mutation-associated vascular microthrombotic disease (GA-VMTD: Hereditary TTP) |
| ADAMTS13 gene antibody-associated vascular microthrombotic disease (AA-VMTD: Acquired TTP) |
| ● HIT-WCS |
| Mechanism: platelet thrombi due to heparin-PF4 complex reacting with heparin-PF4 complex antibody |
| White clot syndrome |
| ● Fibrin clot disease in APL * |
| Mechanism: activated aberrant TF path from overexpressed TF of APL without vascular injury |
| APL coagulopathy with hemorrhagic syndrome (i.e., fibrin clot disease; true DIC) |
Table 6.
Expected laboratory and clinical markers of different phenotypes of thrombosis based on two hemostatic theories in vivo.
Table 6.
Expected laboratory and clinical markers of different phenotypes of thrombosis based on two hemostatic theories in vivo.
| | aEA-VMTD | vEA-VMTD | aMT | vMT | FCD | Combined aMMT | Combined vMMT |
|---|
| Typical example | TTP-like syndrome | ITP-like syndrome | AIS | Distal DVT | APL | SPG | VTE |
| Thrombocytopenia | + | + | - | - | + (due to leukemia) | + | + |
| Fibrinogen | Increased/decreased | Increased/decreased | Normal | Normal | Decreased | Markedly increased | May be increased |
| ULVWF/VWF antigen | Markedly increased | May be increased | Normal | Normal | Normal expected | Markedly increased | Maybe increased |
| FVIII | Markedly increased | Increased | Normal | Normal | Markedly decreased | Increased | May be increased |
| FV | Normal | Normal | Normal | Normal | Decreased | Decreased (?) | Decreased (?) |
| ADAMTS13 | Low | Low | Normal | Normal | Normal expected | Likely low | Likely low |
| FSPs | +/- | +/- | Normal | Normal | Positive | Positive | Positive |
| D-dimer | Normal | Normal | +/- | +/- | Increased | Markedly increased | Markedly increased |
| MAHA | + | +/- | - | - | - | + | - |
| MODS | + | +/- | - | - | - | + | - |
| Inflammation | + | + | - | - | ? | + | + |
| C5b-9 involvement | Expected to be + | Expected to be + | - | - | ? | Expected to be + | Expected to be + |