Abstract
Antipsychotic drugs (APs) aim to treat schizophrenia, bipolar mania, and behavioral symptoms. In child psychiatry, despite limited evidence regarding their efficacy and safety, APs are increasingly subject to off-label use. Studies investigating addictology-related symptoms in young people being scarce, we aimed to characterize the different patterns of AP misuse and withdrawal in children and adolescents relying on the WHO pharmacovigilance database (VigiBase®, Uppsala Monitoring Centre, Sweden). Using the standardized MedDRA Query ‘drug abuse, dependence and withdrawal’, disproportionality for each AP was assessed with the reporting odds ratio and the information component. A signal was detected when the lower end of the 95% confidence interval of the information component was positive. Results revealed mainly withdrawal symptoms in infants (under 2 years), intentional misuse in children (2 to 11 years), and abuse in adolescents (12 to 17 years). Olanzapine, risperidone, aripiprazole, and quetiapine were disproportionately reported in all age groups, with quetiapine being subject to a specific abuse signal in adolescents. Thus, in adolescents, the evocation of possible recreational consumption may lead to addiction-appropriate care. Further, in young patients with a history of AP treatment, a careful anamnesis may allow one to identify misuse and its role in the case of new-onset symptoms.
1. Introduction
Since the discovery of chlorpromazine in France in the 1950s [1], antipsychotic drugs (APs) have aimed to relieve disorganized thoughts and behaviors, hallucinations, and delusions. Accordingly, they are used to treat schizophrenia, bipolar mania, and behavioral symptoms, inter alia. The common mechanism underlying the efficacy of most APs is considered to be the antagonism of brain dopamine D2 receptors [2]. In contrast with older drugs, referred to as ‘typical (or first-generation) antipsychotics’ (TAPs), the more recent ones (‘atypical (or second-generation) antipsychotics’, AAPs) are characterized by a stronger binding affinity to the serotonin 5HT2 receptor, compared to dopamine D2 receptors [3].
In child psychiatry, despite limited evidence regarding their efficacy and safety profiles, APs are increasingly subject to off-label use [4,5,6]. This phenomenon stems from a global increase in the prescribing of APs [7,8], a restricted pattern of marketing authorizations, as well as the lack of guidelines for their use in this population [9,10]. Aside from drug tolerance (which frequently leads to an increase in dosage), patients treated with APs are at risk of drug dependency [11,12,13]. Whether resulting from prescription or recreative purposes (such as seeking euphoria or relaxation), this consumption may lead to abuse, intentional misuse, but also withdrawal phenomena [14,15]. In younger populations, ‘pharming’, which involves the non-medical use and misuse of, mainly psychoactive, medication [16], may be favored in the growing role of the Internet and social media, especially regarding the easier accessibility of drugs [17,18]. Accordingly, the ‘psychonauts’ [14,16], ‘pharming’ users that try various psychoactive drugs and then share their experiences on social media, appeal to an ever-expanding audience, potentially influencing children and adolescents.
Studies investigating addictology-related symptoms in young people being scarce, beyond reports of accidental overdosage, we tried to characterize the different patterns of antipsychotic misuse and withdrawal, relying on an analysis of the World Health Organization (WHO) safety database (VigiBase®, Uppsala Monitoring Centre, Sweden) [19]. While withdrawal cases in infants and abuse cases in adolescents might be expected, there is still a grey area regarding middle-aged children. As antipsychotics are more and more subject to off-label use and illicit consumption in children and youth, we also aimed to identify potential drug safety signals regarding antipsychotic-related abuse, dependence, and withdrawal in this population. Further, we tried to shed some light on the most involved drugs, while suspecting the existence of different consumption patterns for each one, depending on the age group.
2. Materials and Methods
2.1. Data Source
The WHO mandates the Uppsala Monitoring Centre (UMC) to oversee drug safety [20]. This independent center aims to gather evidence about adverse drug reactions (ADRs), therefore leading to the identification of safety signals [21]. Indeed, VigiBase® (UMC, Sweden), the WHO safety database, collects Individual Case Safety Reports issued by more than 172 national pharmacovigilance network members, along with pharmaceutical companies. The preservation of the anonymity of patients and notifiers is ensured by VigiBase® (UMC, Sweden) [19].
Each individual Case Safety Report features sociodemographic characteristics of the patients (age, sex), administrative information (country, reporter qualification), suspected drug (indication, start and cessation dates, dose), concomitant drug(s), and characteristics of the ADR(s)’ (effect(s), seriousness, onset, outcome). In pharmacovigilance, an ADR is considered to be serious if it justified a hospitalization or its prolongation, caused a congenital malformation, resulted in persistent or significant disability or incapacity, was life threatening, resulted in death, or required significant medical intervention to prevent one of these outcomes [22,23].
2.2. Query
In the Medical Dictionary for Regulatory Activities (MedDRA, version 25.0 [24]), a Standardised MedDRA Query (SMQ) is an exhaustive, validated, predetermined collection of Preferred Terms (PTs) intended to help in investigating drug safety issues in pharmacovigilance [25]. A PT expresses a single medical concept in the most clinically accurate way [24].
We first queried VigiBase® (UMC, Sweden) for all reports featuring the narrow SMQ ‘drug abuse, dependence and withdrawal’ (Medical Dictionary for Regulatory Activities, MedDRA 25.0 [24]) registered between 14 November 1967 (first reports in VigiBase®, (UMC, Sweden)) and 4 August 2022 and involving all antipsychotics reported in the database (acepromazine, acetophenazine, amisulpride, amperozide, aripiprazole, asenapine, benperidol, bifeprunox, blonanserin, brexpipirazole, bromperidol, butaperazine, carfenazine, cariprazine, chlorphenetazine, chlorproethazine, chlorpromazine, chlorprothixene, ciclofenazine, clopenthixol, clorotepine, clotiapine, clozapine, cyamemazine, dixyrazine, droperidol, fluanisone, flupentixol, fluphenazine, fluspirilene, haloperidol, iloperidone, lenperone, levomepromazine, levosulpiride, loxapine, lumateperone, lurasidone, melperone, mesoridazine, methopromazine, metofenazate, molindone, moperone, mosapramine, nemonapride, olanzapine, oxypertine, oxyprothepin, paliperidone, pecazine, penfluridol, perazine, periciazine, perospirone, perphenazine, pimavanserin, pimozide, pipamperone, piperacetazine, pipotiazine, pomaglumetad methionil, prochlorperazine, promazine, prothipendyl, quetiapine, raclopride, remoxipride, risperidone, sarizotan, sertindole, setoperone, sonepiprazole, spiperone, sulforidazine, sulpiride, sultopride, thiopropazate, thioproperazine, thioridazine, tiapride, timiperone, tiotixene, trifluoperazine, trifluperidol, triflupromazine, veralipride, ziprasidone, zotepine, and zuclopenthixol).
Then, queried reports involving patients aged under 18 were classified into 3 age groups: under 23 months, 2 to 11 years, and 12 to 17 years. For each group, records were aggregated depending on the PTs, into one 1 of 3 categories: misuse, abuse, or withdrawal [18].
‘Misuse’ can be defined as the intentional and inappropriate use of a product other than as prescribed or not in accordance with validated drug labels. It included the PTs ‘Intentional overdose’ and ‘Intentional product misuse’.
‘Abuse’ is the intentional non-therapeutic use of a product for the experience or feeling elicited (e.g., euphoria). It included the PTs ‘Drug abuse’, ‘Drug abuser’, ‘Drug dependence’, ‘Drug use disorder’, ‘Neonatal complications of substance abuse’, ‘Substance abuse’, ‘Substance abuser’, ‘Substance dependence’, and ‘Substance use disorder’.
‘Withdrawal’ describes symptoms or signs related to the cessation of a drug, either physiological withdrawal reaction to the drug or exacerbation of the underlying disease itself [26]. It included the PTs ’Drug withdrawal convulsions’, ‘Drug withdrawal headache’, ’Drug withdrawal syndrome’, and ‘Drug withdrawal syndrome neonatal’.
2.3. Statistical Analyses
Quantitative variables were described in terms of means with standard deviations (±SD). Qualitative variables were described with proportions. Statistical analyses were performed using GraphPad Prism version 8.0.2. Thereupon, we performed a disproportionality analysis, to partially mitigate the impact of potential confounding factors [27]. Potential pharmacovigilance signals were sought for all antipsychotics (as described above, see Section 2.2. Query), for each age group:
- Using the narrow SMQ ‘Drug abuse, dependence, and withdrawal’ when the chosen drug accounted for ≥2 cases and >1% of the reports of the respective age group;
- Using the most represented combination of PTs (in each age group), when the chosen drug accounted for ≥10% of the reports and/or was quetiapine, olanzapine, risperidone, and aripiprazole. This particular analysis aimed to clarify the specific ADRs driving any possible signal arising from the disproportionality analysis of the SMQ.
The disproportionality analysis relied on the reporting odds ratio (ROR) and the information component (IC).
As an approximate of the odds ratio (used in case-control studies), the ROR is estimated, in case–non-case studies, to assess the strength of disproportionality. An ROR equal to 1 indicates the absence of signal: the ADR is equally reported with the drug of interest as with the other drugs. In contrast, an ROR greater than 1 suggests the existence of a signal, as cases appear to be more frequently reported with the drug of interest than with the others. The higher the ROR, the stronger the association. A 95% confidence interval (95% CI) reflects the precision of the approximate ROR. Therefore, an ROR is deemed to be statistically significant when the lower bound of its 95% CI is greater than 1 [28].
The IC allows one to compare observed and expected values for the combination of a given drug and an ADR, to check for a potential association. It favors the reduction in the risk of false-positive signals, especially if the chosen ADR has a very low expected frequency in the database (therefore, mechanically increasing the ROR). The positivity of the IC reflects that the number of observed reports is higher than expected. The IC025, which is the bottom end of the 95% CI of the information component, is required to statistically confirm the detection of a signal in VigiBase® (UMC, Sweden) [19,29].
In this disproportionality analysis, potential drug–ADR associations were selected by using IC025. Then, the ROR of each drug–effect association allowed us to assess the strength of respective suggested signals. For this purpose, we used Microsoft® Excel® 2019 Version 2210.
3. Results
3.1. Drug Abuse, Dependence, and Withdrawal
As of 4 August 2022, 16,054 reports belonging to the narrow SMQ ‘Drug abuse, dependence and withdrawal’ and involving consumers of antipsychotics were collected in VigiBase® (UMC, Sweden). Among these reports, 1023 (6.4%) involved patients below 18 years of age, mostly belonging to the 12-to-17-year group (732, 71.6%). Records with patients aged under 18 mostly originated from the United States (398, 38.9%). Healthcare professionals issued 84.8% of the cases, with a majority of physicians (617, 60.3%). Details regarding the characteristics of the reports are provided in Table 1.

Table 1.
Characteristics of the reports of patients with abuse, dependence, or withdrawal.
Four antipsychotics accounted for 839 (82.0%) reports of abuse, dependence, and withdrawal: quetiapine (368, 36.0%), risperidone (224, 21.9%), aripiprazole (129, 12.6%), and olanzapine (118, 11.5%). The number of reports for all ADRs in patients aged under 18 involving each of these antipsychotics is displayed in Table S1.
Seriousness was assessed in 925 (90.4%) reports: 846 (91.5%) were deemed serious, among which 488 (57.6%) ADRs caused/prolonged hospitalizations, 78 (9.2%) life-threatening reactions, 40 (4.7%) deaths, and 30 (3.5%) congenital anomalies/birth defects. When details regarding the outcome were available (429, 41.6%), 356 (83.0%) patients recovered or were recovering, 40 (9.3%) did not recover, and 6 (1.4%) recovered with sequelae.
In patients aged under 18, antipsychotics were subject to a disproportionate reporting for the SMQ ‘Drug abuse, dependence and withdrawal’ (ROR 5.5; IC025 2.2).
3.2. Patients Aged between 0 Days and 23 Months
Among patients below 18 years of age, 198 (19.4%) reports of ADRs related to antipsychotic abuse, dependence, and withdrawal and involving infants aged from 0 days to 23 months were found in VigiBase® (UMC, Sweden) In this age group, males represented 51.5% (n = 102) and their mean age was 2.3 (±2.9) months. Most of them (146, 73.7%) were newborns (aged under 28 days). The most frequently co-reported MedDRA terms were fetal exposure during pregnancy (63, 31.8%), premature baby (27, 12.1%), and tremor (17, 8.6%). Quetiapine was suspected in 75 records (37.8%), followed by aripiprazole (30, 15.2%), risperidone (28, 14.1%), and olanzapine (20, 10.1%). The complete list of involved antipsychotics is provided in Table S2.
Co-reported drugs were found in 177 cases (89.4%). The most frequently suspected co-reported active ingredients were other psychotropic drugs, such as antidepressants sertraline (20, 10.1%), clomipramine and fluoxetine (18 cases each, 9.1%), venlafaxine (17, 8.6%), and the hypnotic drug zopiclone (14, 7.1%).
When available, 140 (96.5%) reports were deemed serious, among which 89 (63.6%) ADRs caused/prolonged hospitalizations, 30 (21.4%) congenital anomalies/birth defects, 13 (9.3%) life-threatening reactions, and 1 (0.7%) death. Among reports with available follow-up, 145 (91.8%) infants recovered or were recovering, 10 (6.3%) were not recovering, and 2 (1.3%) recovered with sequelae.
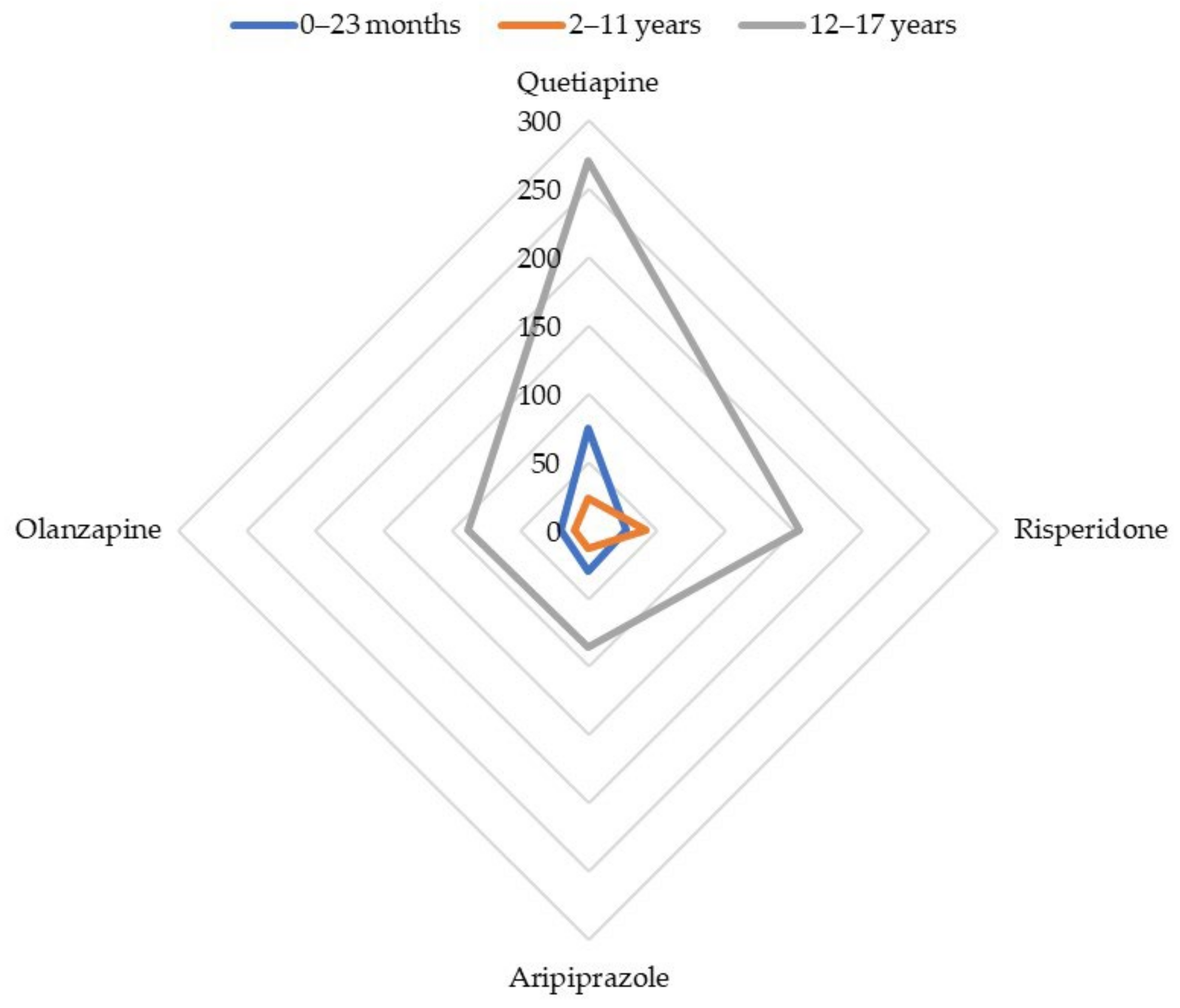
The most often involved antipsychotics (absolute number of reports) were disproportionately reported with the SMQ ‘Drug abuse, dependence, and withdrawal’: quetiapine (ROR 68.3; 95% CI: 53.2–87.8), risperidone (ROR 35.2; 95% CI: 23.8–52.0), aripiprazole (ROR 25.1; 95% CI: 17.2–36.4), and olanzapine (ROR 23.4; 95% CI: 14.9–36.9). Apart from quetiapine, the highest RORs were reached by cyamemazine (ROR 82.1; 95% CI: 47.5–141.8), amisulpride (ROR 68.3; 95% CI: 23.3–200.1), zuclopenthixol (ROR 60.3; 95% CI: 17.6–205.8), and levomepromazine (ROR 58.2; 95% CI: 28.6–118.0). The whole disproportionality analysis for cases of infants aged between 0 and 23 months is displayed in Table 2. Figure 1 summarizes the disproportionality analysis for the main antipsychotics involved in reports of abuse, dependence, or withdrawal, depending on the age range.

Table 2.
Disproportionality analysis for reports of abuse, dependence, or withdrawal in patients aged between 0 days and 23 months, ranked by ROR.
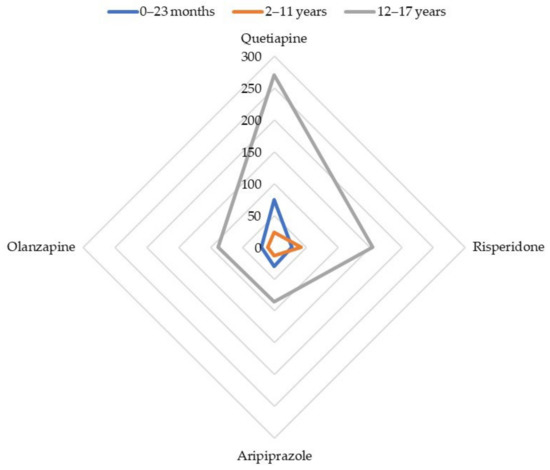
Figure 1.
Cases’ distribution for main antipsychotics involved in reports of abuse, dependence, or withdrawal.
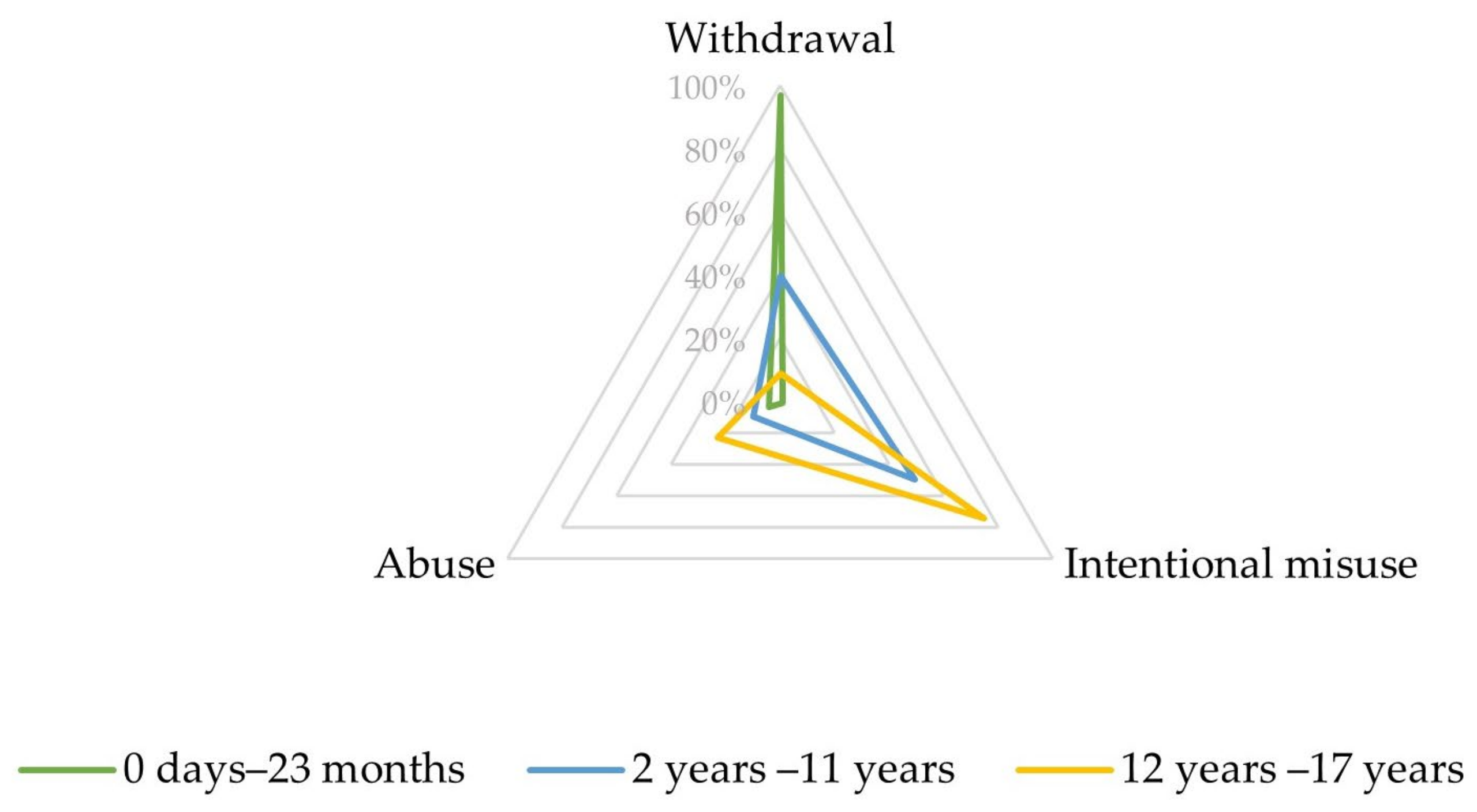
Upon closer inspection, in the narrow SMQ ‘Drug abuse, dependence, and withdrawal’, most reports involving infants aged between 0 days and 23 months referred to PTs related to withdrawal (192, 96.7%), as displayed in Figure 2 and Table S3.
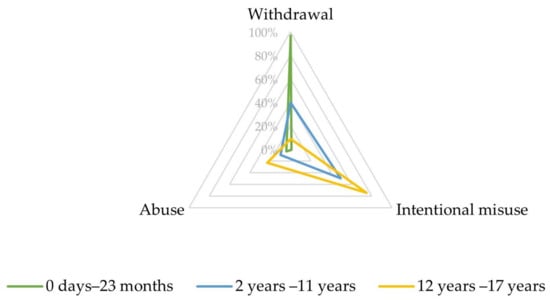
Figure 2.
Characteristics of the combined Preferred Terms (PTs) ‘abuse’, ‘intentional misuse’, and ‘withdrawal’ depending on age.
The combination of ‘withdrawal’ PTs (drug withdrawal headache, drug withdrawal syndrome, drug withdrawal syndrome neonatal) showed disproportionate reporting for quetiapine (ROR 86.8; 95% CI: 67.4–111.8), risperidone (ROR 43.3; 95% CI: 29.1–64.5), aripiprazole (ROR 28.7; 95% CI: 19.4–42.5), and olanzapine (ROR 28.4; 95% CI: 17.8–45.3), as shown in Table 3.

Table 3.
Disproportionality analysis for main antipsychotics involved in reports of withdrawal in patients aged between 0 days and 23 months, ranked by ROR.
3.3. Patients Aged between 2 and 11 Years
Children aged between 2 and 11 years accounted for 93 (9.1%) reports involving patients aged under 18 years. The majority of patients in this age group were male (65, 69.9%), with a mean age of 8.4 years (±2.2). Dyskinesia (16, 17.2%), dystonia (13, 14.0%), and aggression (11, 11.8%) were the most frequently co-reported MedDRA terms. In this age range, risperidone was the most frequently suspected antipsychotic (42, 45.2%), followed by quetiapine (23, 24.7%), aripiprazole 13 (13, 14.0%), and olanzapine (10, 10.8%). The complete list is available in Table S4.
Co-reported drugs were found in 63 cases (67.7%). The most frequently suspected co-reported active ingredients were methylphenidate (10, 10.8%), levothyroxine (6, 6.5%), citalopram (6, 6.5%), valproic acid (5, 5.4%), and omeprazole (5, 5.4%).
When available, 61 cases (79.2%) were considered to be serious, with 36 (46.8%) causing/prolonging hospitalizations, 8 (10.4%) deaths, and 4 (5.2%) life-threatening reactions. Among reports with follow-up, 19 (63.3%) infants recovered or were recovering, 4 (13.3%) were not recovering, and 2 (6.7%) recovered with sequelae.
Quetiapine (ROR 19.3; 95% CI: 12.7–29.4), olanzapine (ROR 10.0; 95% CI: 5.3–18.7), risperidone (ROR 5.0; 95% CI: 3.7–6.3), and aripiprazole (ROR 3.1; 95% CI: 1.8–5.3) were subject to a disproportionate reporting with the SMQ ‘Drug abuse, dependence, and withdrawal’ (Table 4, Figure 1).

Table 4.
Disproportionality analysis for reports of abuse, dependence, or withdrawal in patients aged between 2 years and 11 years, ranked by ROR.
In the SMQ ‘Drug abuse, dependence and withdrawal’, most reports involving children aged between 2 and 11 years referred to PTs related to intentional misuse (46, 49.5%) and withdrawal (37, 39.8%) (Figure 2, Table S5).
The ‘intentional misuse’ combination of PTs (intentional overdose, intentional product misuse) showed disproportionate reporting for quetiapine (ROR 34.4; 95% CI: 22.0–54.0), followed by olanzapine (ROR 12.2; 95% CI: 5.4–27.2) and risperidone (ROR 4.3; 95% CI: 2.7–6.9) (Table 5). No report was found for aripiprazole in this category. The median time to onset for intentional misuse-related ADRs was 730 days (IQR 122–1460).

Table 5.
Disproportionality analysis for main antipsychotics involved in reports of intentional misuse in patients aged between 2 years and 11 years, ranked by ROR.
The ‘withdrawal’ combination of PTs (drug withdrawal syndrome, drug withdrawal convulsions) was subject to disproportionate reporting for olanzapine (ROR 13.7; 95% CI: 5.1–36.7), aripiprazole (ROR 10.0; 95% CI: 5.6–17.7), and risperidone (ROR 7.8; 95% CI: 4.9–12.4) (Table 6).

Table 6.
Disproportionality analysis for main antipsychotics involved in reports of withdrawal in patients aged between 2 years and 11 years, ranked by ROR.
3.4. Patients Aged between 12 and 17 Years
Adolescents aged between 12 and 17 years accounted for 732 (71.6%) of the queried reports. In this age group, most patients were female (408, 55.7%) and their mean age was 15.3 (±1.5) years, with more than one-quarter being 17-year-old adolescents (199, 27.2%). The most frequently co-reported MedDRA terms were suicide attempt (226, 30.9%), somnolence (156, 21.3%), and tachycardia (62, 8.5%). Quetiapine accounted for 270 records (36.9%), followed by risperidone (154, 21.0%), olanzapine (88, 12.0%), and aripiprazole (86, 11.7%). The complete list of suspected antipsychotics for this age range is available in Table S6.
Other drugs were co-reported in 537 cases (73.4%). Fluoxetine (54, 7.4%), alprazolam (39, 5.3%), sertraline (36, 4.9%), paracetamol (35, 4.8%), and diazepam (28, 3.8%) were the most frequently suspected co-reported active ingredients.
When this information was available, 645 (61.7%) reports were deemed serious, including 363 (51.6%) causing/prolonging hospitalizations, 61 (8.6%) life-threatening reactions, and 31 (4.4%) deaths. Among reports with follow-up, 192 (76.7%) infants recovered or were recovering, 26 (10.7%) were not recovering, and 2 (0.8%) recovered with sequelae.
The most frequently involved antipsychotics were disproportionately reported with the narrow SMQ ‘Drug abuse, dependence and withdrawal’: quetiapine (ROR 8.9; 95% CI: 7.8–10.1), olanzapine (ROR 2.8; 95% CI: 2.3–3.5), risperidone (ROR 1.8; 95% CI: 1.5–2.1), and aripiprazole (ROR 1.7; 95% CI: 1.4–2.1) (Table 7, Figure 1). Beyond quetiapine, promazine (ROR 97.5; 95% CI: 56.0–169.8), chlorprothixene (ROR 35.5; 95% CI: 22.1–57.3), pipamperone (ROR 14.6; 95% CI: 8.5–25.0), and cyamemazine (ROR 4.2; 95% CI: 2.6–6.6) showed the greatest RORs.

Table 7.
Disproportionality analysis for reports of abuse, dependence, or withdrawal in patients aged between 12 years and 17 years, ranked by ROR.
Most of these reports referred to PTs related to intentional misuse (545, 74.5%) and abuse (168, 23.0%), as displayed in Figure 2 and Table S7.
The ‘intentional misuse’ combination of PTs (intentional overdose, intentional product misuse) was subject to disproportionate reporting for quetiapine (ROR 10.7; 95% CI: 9.3–12.3), olanzapine (ROR 3.6; 95% CI: 2.8–4.6), risperidone (ROR 2.1; 95% CI: 1.7–2.5), and aripiprazole (ROR 1.9; 95% CI: 1.5–2.5) (Table 8).

Table 8.
Disproportionality analysis for main antipsychotics involved in reports of intentional misuse in patients aged between 12 years and 17 years, ranked by ROR.
The ‘abuse’ combination of PTs (drug abuse, drug dependence, substance abuse, drug abuser, drug use disorder, substance abuser, substance dependence, substance use disorder) was only disproportionately reported for quetiapine (ROR 4.8; 95% CI: 3.7–6.3), (Table 9).

Table 9.
Disproportionality analysis for main antipsychotics involved in reports of abuse in patients aged between 12 years and 17 years, ranked by ROR.
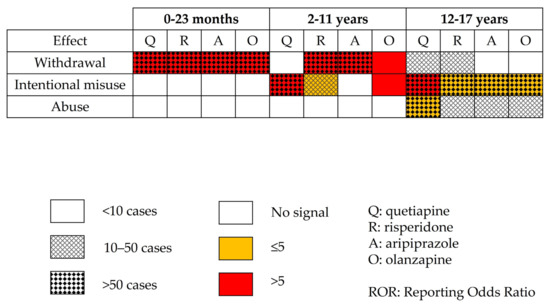
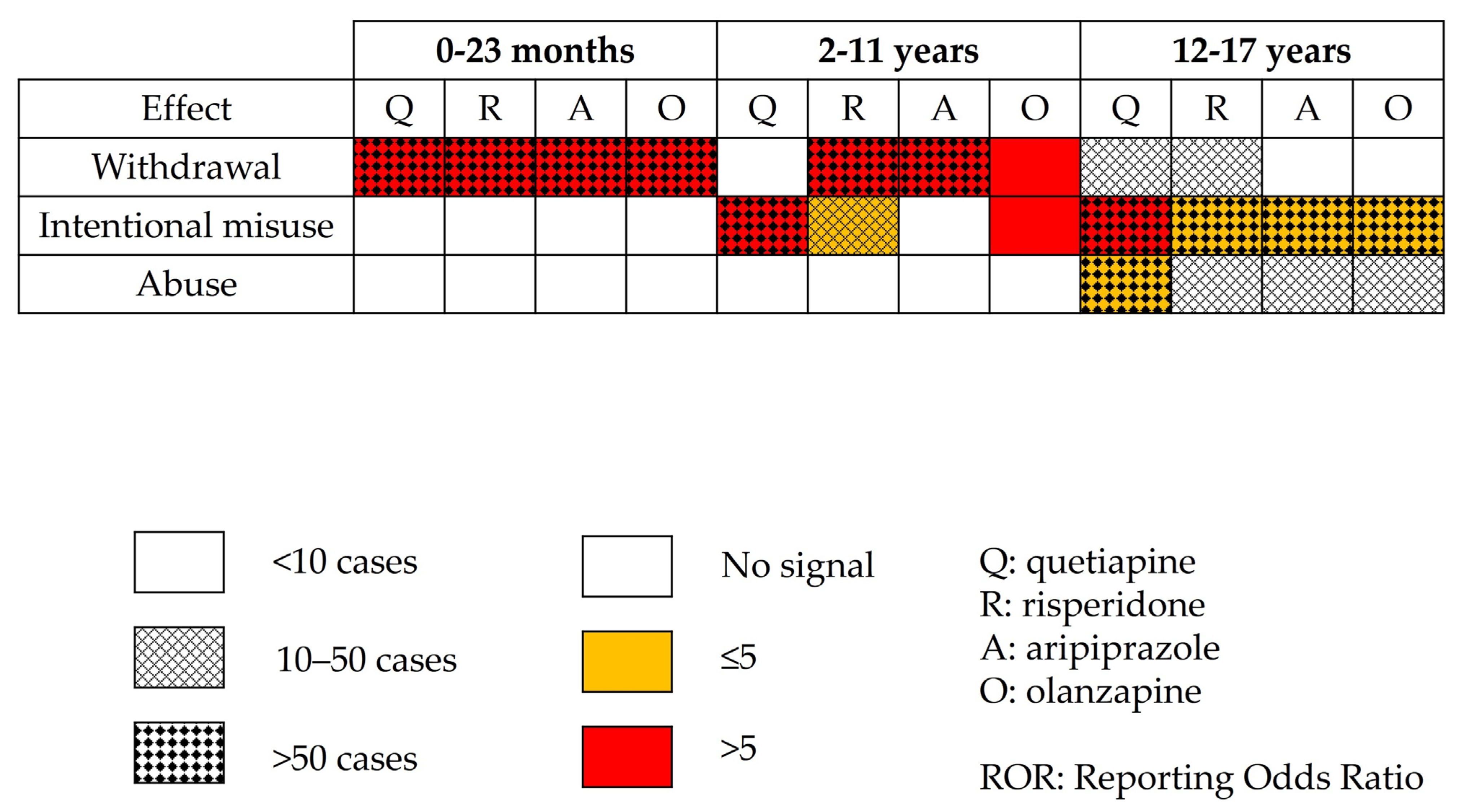
Figure 3.
Disproportionality analysis using combined Preferred Terms for most involved antipsychotics.
4. Discussion
Our analysis of the WHO pharmacovigilance database brings to light varying profiles for abuse, dependence, and withdrawal related to antipsychotics. Indeed, in infants aged under 2 years, almost all ADRs were understandably related to withdrawal symptoms. In children aged between 2 and 11 years, intentional misuse was at the forefront, with fewer cases of withdrawal, frequently complicated by extrapyramidal symptoms. Lastly, adolescents aged from 12 to 17 years were subject to intentional misuse, but also to abuse issues.
In infants aged from 0 days to 23 months, withdrawal syndrome reflects prenatal maternal exposure to APs. AAPs (quetiapine, risperidone, aripiprazole, olanzapine) prevailed, both in terms of absolute number of cases and disproportionality, which is consistent with existing literature regarding their respective safety profiles and prescribing trends during pregnancy [30,31]. However, their drug labels indicate a risk of withdrawal in exposed neonates and recommend avoiding these drugs during pregnancy, unless their use is an absolute necessity [32,33,34,35]. Quetiapine showed the strongest signal in this population. Indeed, in women previously stabilized with quetiapine, it often remains prescribed during pregnancy [36,37]. At birth, the newborn being severed from the maternal blood supply and quetiapine being rapidly dissociated from the D2 receptors [38,39], its abrupt discontinuation may lead to greater risks of withdrawal at birth [40]. The frequent maternal cotreatment with antidepressants (e.g., mood disorders) that we observed may also reinforce withdrawal symptoms in infants [40,41,42].
In children aged between 2 and 11 years, the age and gender distribution regarding treatment with antipsychotics in the literature concurs with our findings [43,44,45]. Indeed, significant disproportionality was found for AAP only, subject to a dramatic increase in consumption in children, circumscribed by the rise of off-label use in this population [43,44,46,47]. Regarding risperidone, which is the leading drug in terms of absolute number of reports, both the Food and Drug Administration (FDA) and the European Medicines Agency (EMA) have granted it marketing authorization in irritability occurring in patients with an autistic disorder from the age of 5 [35,48]. Aripiprazole is authorized by the FDA from the age of 6 [34] in the same indication, but its use is only recommended from the age of 13 by the EMA (manic episodes of bipolar I disorder) [49]. Lastly, marketing authorization for olanzapine and for quetiapine is granted by the FDA in acute mixed or manic episodes of bipolar I disorder from the age of 10 [32,33]. However, the EMA stated that their safety and efficacy in children under the age of 18 have not been established yet and, therefore, that they should not be used in this group until further data become available [50,51]. In more than one-quarter of the reports, abuse, dependence, or withdrawal occurred concomitantly with dystonia and/or dyskinesia. These effects may reflect either overdosing following intentional misuse or withdrawal syndrome [52,53], subsequent to dopamine receptor hypersensitivity, GABA insufficiency, or cellular degeneration (neurotoxicity) [54,55]. In cases of withdrawal, the strongest disproportionality signals concerned risperidone and aripiprazole. This may indicate abrupt treatment discontinuations that could be favored by the urge of appeasing irritability and/or aggressive symptoms via the initiation of a new medication (e.g., in patients suffering from autistic disorders). In addition, possible impulsive and voluntary intoxications may also occur, especially in children presenting with behavioral disorders.
Adolescents aged from 12 to 17 years accounted for the vast majority of the reports belonging to the SMQ ‘drug abuse, dependence and withdrawal’. Paracetamol was quite frequently co-reported as suspect or interacting, which may reflect the fact that suicide attempts accounted for nearly one-third of the reports in this age range [56]. Further, ingestion of co-reported substances, such as antidepressants and benzodiazepines, might also concur with phenomena of intentional misuse or abuse or designate a population of patients receiving long-term treatment with psychotropic drugs. In this context, the AAPs quetiapine, olanzapine, risperidone, and aripiprazole were subject to disproportionate reporting, to a greater degree than TAPs, such as promazine, chlorprothixene, and cyamemazine. Regarding TAPs, cyamemazine has been granted marketing authorization in France (Agence Nationale du Médicament et des produits de santé—ANSM) for behavioral disorders with psychomotor agitation and aggressivity for children aged 3 years or older [57]. Promazine and chlorprothixene were withdrawn from the market in the United States [58,59]. However, commonly prescribed AAPs may be ingested for recreative purposes, as Novel Psychoactive Substances (NPSs) [12,18]. According to the United Nations Office on Drugs and Crime (UNODC), NPSs are ‘substances of abuse, either in a pure form or a preparation, that are not controlled by the 1961 Single Convention on Narcotic Drugs or the 1961 Convention on Psychotropic Substances, but which may pose a public health threat’ [60]. Compared to ‘conventional’ illicit substances, NPSs are distinguished by greater affordability and ease of online purchase, but also lower detectability and social stigma [61]. In younger populations, both appeasement of other consumption symptoms and search of emotional anesthesia are becoming more and more accessible, as these phenomena are further exacerbated by the ‘pharming’ and ‘psychonauts’ trends. As abuse and intentional misuse have been described for risperidone and aripiprazole [13,62], we extend this potential signal to adolescent populations. In line with previous findings (irrespective of the age range) [13,15], olanzapine was disproportionately reported for intentional misuse. Olanzapine can be consumed as a psychedelic or for its sedative effect, with rewarding properties involving glutamatergic stimulation of dopaminergic neurons of the ventral tegmental area [63]. Our findings confirm that quetiapine, already involved in pharmacovigilance signals of intentional misuse and abuse, raises also issues in adolescents. Indeed, it is the leading drug in terms of absolute number of reports. Quetiapine has multiple street names, such as ‘quell’, ‘Susie-Q’, ‘baby heroin’, and ‘Q-ball’ (when associated with cocaine) [64]. Consumed for anxiolytic properties, its popularity as an NPS may be linked to H1 and α1 receptor antagonism. The same rationale may apply to olanzapine, more and more available on the black market [13,64,65].
While other studies aimed to assess the importance of antipsychotic abuse and misuse in different pharmacovigilance databases [13,15,66], our analysis was the first to focus on children and youth. It highlighted different profiles, depending on the age of the patients. Nevertheless, this study is hindered by the inherent flaws of spontaneous reporting systems and post-marketing pharmacovigilance approaches, such as incomplete data and reporting bias. The strict definitions of the terms, from a pharmacovigilance perspective, might be unclear for notifiers, which may have led to a substantial coding heterogeneity, especially regarding intentional misuse and abuse. However, most reports were notified by healthcare professionals, which limits the risk of coding errors. Further, lack of follow-up and under-reporting, although usual in pharmacovigilance [67], may have been heightened by the extent of overall consumption and the acute nature of manifestations (especially related to withdrawal or intentional overdose). Then, associated medications were recorded (co-reported active ingredients), but we were not able to retrieve data on the consumption of other illegal substances, as this is outside the scope of VigiBase®(UMC, Sweden) Lastly, pharmaco-epidemiological studies aim to raise awareness about possible drug safety signals and no definite causality can be drawn from our findings.
Regarding implications for practice and research, different areas have to be taken into account. During pregnancy, as recommended by the safety agencies [32,33,34,35,48,49,50,51], each antipsychotic treatment must be introduced or maintained following a careful assessment of the benefit/risk ratio. In children, off-label prescription should be properly substantiated, considering the scarcity of data regarding safety in efficacy in this population [50,51]. In adolescents, the risk of antipsychotic abuse and its characteristics should be further investigated. In addition, particular heed should be paid to specialized social networks, underlying new consumption patterns [14,16].
5. Conclusions
In this study, relying on a comprehensive analysis of drug abuse, dependence, and withdrawal reports with APs from the WHO safety database, we suggest potential pharmacovigilance signals involving several AAPs, especially quetiapine, olanzapine, risperidone, and aripiprazole. Defining three profiles according to the patients’ age ranges, we highlighted the potential role of these drugs in withdrawal phenomena among infants, but also in intentional misuse behaviors among children and adolescents. We confirmed existing signals of intentional misuse of APs and the growing importance of quetiapine as an NPS. In young patients with a history of AP treatment, a careful anamnesis may allow one to identify the role of these medications in the case of new-onset symptoms. Indeed, given the variety in their manifestations, the hypothesis of an antipsychotic-related ADR should be systematically evoked. In adolescents, the evocation of a possible recreational consumption may lead to addiction-appropriate care, therefore, reducing further morbidity.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/biomedicines10112972/s1, Table S1: Reports of any adverse drug reaction involving risperidone, aripiprazole, quetiapine, and olanzapine in children and adolescents (<18 years old); Table S2: Reported suspected antipsychotics in patients aged between 0 days and 23 months with abuse, dependence, or withdrawal; Table S3: Main Preferred Terms (PTs) reported in cases of abuse, dependence, or withdrawal in patients aged between 0 days and 23 months; Table S4: Reported suspected antipsychotics in patients aged between 2 and 11 years with abuse, dependence, or withdrawal; Table S5: Main PTs reported in cases of abuse, dependence, or withdrawal in patients aged between 2 years and 11 years; Table S6: Reported suspected antipsychotics in patients aged between 12 and 17 years with abuse, dependence, or withdrawal; Table S7: Main PTs reported in cases of abuse, dependence, or withdrawal in patients aged between 12 years and 17 years.
Author Contributions
Conceptualization, D.M., A.O.G. and S.T.; methodology, D.M. and A.O.G.; validation, S.T. and M.-D.D.; formal analysis, D.M.; investigation, D.M.; data curation, D.M. and A.O.G.; writing—original draft preparation, D.M.; writing—review and editing, A.O.G., S.T., M.-D.D., A.D. and F.A.; visualization, D.M. and A.O.G.; supervision, S.T. and M.-D.D. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
In accordance with European regulation, this observational study did not need approval from an institutional review board/independent ethics committee. The views expressed in this article are the authors’ personal views and may not be understood or quoted as being made on behalf of or reflect the position of the ANSM, the EMA, or one of their committees or working parties.
Informed Consent Statement
Not applicable.
Data Availability Statement
The data that support the findings of this study are available from Uppsala Monitoring Center (UMC) but restrictions apply to the availability of these data, which were used under license for the current study and so are not publicly available. Access to VigiBase® is available without fees to Fanny Rocher. Data are, however, available from the authors upon reasonable request and with permission of UMC.
Acknowledgments
The authors acknowledge the UMC, which manages and provided the data used in the present study. Access to VigiBase® is available without fees to Fanny Rocher.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Delay, J.; Deniker, P. Chlorpromazine and Neuroleptic Treatments in Psychiatry. J. Clin. Exp. Psychopathol. 1956, 17, 19–24. [Google Scholar] [PubMed]
- Seeman, P. Dopamine Receptors and the Dopamine Hypothesis of Schizophrenia. Synapse 1987, 1, 133–152. [Google Scholar] [CrossRef] [PubMed]
- Leucht, S.; Corves, C.; Arbter, D.; Engel, R.R.; Li, C.; Davis, J.M. Second-Generation versus First-Generation Antipsychotic Drugs for Schizophrenia: A Meta-Analysis. Lancet 2009, 373, 31–41. [Google Scholar] [CrossRef]
- Sohn, M.; Moga, D.C.; Blumenschein, K.; Talbert, J. National Trends in Off-Label Use of Atypical Antipsychotics in Children and Adolescents in the United States. Medicine 2016, 95, e3784. [Google Scholar] [CrossRef] [PubMed]
- Merino, D.; Fernandez, A.; Gérard, A.O.; Ben Othman, N.; Rocher, F.; Askenazy, F.; Verstuyft, C.; Drici, M.-D.; Thümmler, S. Adverse Drug Reactions of Olanzapine, Clozapine and Loxapine in Children and Youth: A Systematic Pharmacogenetic Review. Pharmaceuticals 2022, 15, 749. [Google Scholar] [CrossRef]
- Menard, M.-L.; Auby, P.; Cruzel, C.; Cohen, D.; Bonnot, O.; Askenazy, F.; Thümmler, S. Emergence of Psychiatric Adverse Events during Antipsychotic Treatment in AP-Naïve Children and Adolescents. Child Adolesc. Psychiatry Ment. Health 2022, 16, 83. [Google Scholar] [CrossRef]
- Patel, N.C.; Crismon, M.L.; Hoagwood, K.; Johnsrud, M.T.; Rascati, K.L.; Wilson, J.P.; Jensen, P.S. Trends in the Use of Typical and Atypical Antipsychotics in Children and Adolescents. J. Am. Acad. Child Adolesc. Psychiatry 2005, 44, 548–556. [Google Scholar] [CrossRef]
- Olfson, M.; Blanco, C.; Liu, L.; Moreno, C.; Laje, G. National Trends in the Outpatient Treatment of Children and Adolescents With Antipsychotic Drugs. Arch. Gen. Psychiatry 2006, 63, 679–685. [Google Scholar] [CrossRef]
- Rachamallu, V.; Elberson, B.W.; Vutam, E.; Aligeti, M. Off-Label Use of Clozapine in Children and Adolescents-A Literature Review. Am. J. Ther. 2019, 26, e406–e416. [Google Scholar] [CrossRef]
- EMA. EMA: Leponex (Clozapine). Available online: https://www.ema.europa.eu/en/medicines/human/referrals/leponex (accessed on 11 April 2022).
- Samaha, A.-N. Can Antipsychotic Treatment Contribute to Drug Addiction in Schizophrenia? Prog. Neuropsychopharmacol. Biol. Psychiatry 2014, 52, 9–16. [Google Scholar] [CrossRef]
- Malekshahi, T.; Tioleco, N.; Ahmed, N.; Campbell, A.N.C.; Haller, D. Misuse of Atypical Antipsychotics in Conjunction with Alcohol and Other Drugs of Abuse. J. Subst. Abus. Treat. 2015, 48, 8–12. [Google Scholar] [CrossRef]
- Evoy, K.E.; Teng, C.; Encarnacion, V.G.; Frescas, B.; Hakim, J.; Saklad, S.; Frei, C.R. Comparison of Quetiapine Abuse and Misuse Reports to the FDA Adverse Event Reporting System With Other Second-Generation Antipsychotics. Subst. Abus. 2019, 13, 1178221819844205. [Google Scholar] [CrossRef]
- Orsolini, L.; Papanti, G.D.; Francesconi, G.; Schifano, F. Mind Navigators of Chemicals’ Experimenters? A Web-Based Description of E-Psychonauts. Cyberpsychol. Behav. Soc. Netw. 2015, 18, 296–300. [Google Scholar] [CrossRef]
- Roy, S.; Charreteur, R.; Peries, M.; Kheloufi, F.; Eiden, C.; Nagot, N.; Donnadieu-Rigole, H.; Micallef, J.; Peyrière, H. Abuse and Misuse of Second-generation Antipsychotics: An Analysis Using VigiBase, the World Health Organisation Pharmacovigilance Database. Br. J. Clin. Pharmacol. 2022, 88, 4646–4653. [Google Scholar] [CrossRef]
- Chiappini, S.; Schifano, F. What about “Pharming”? Issues Regarding the Misuse of Prescription and Over-the-Counter Drugs. Brain Sci. 2020, 10, 736. [Google Scholar] [CrossRef]
- Mackey, T.K.; Nayyar, G. Digital Danger: A Review of the Global Public Health, Patient Safety and Cybersecurity Threats Posed by Illicit Online Pharmacies. Br. Med. Bull. 2016, 118, 110–126. [Google Scholar] [CrossRef]
- Schifano, F. Coming Off Prescribed Psychotropic Medications: Insights from Their Use as Recreational Drugs. Psychother. Psychosom. 2020, 89, 274–282. [Google Scholar] [CrossRef]
- Lindquist, M. VigiBase, the WHO Global ICSR Database System: Basic Facts. Ther. Innov. Regul. Sci. 2008, 42, 409–419. [Google Scholar] [CrossRef]
- Uppsala Monitoring Centre About VigiBase. Available online: https://who-umc.org/vigibase/ (accessed on 26 September 2022).
- Centre, U.M. Uppsala Monitoring Centre. Available online: https://who-umc.org/ (accessed on 28 April 2022).
- Gautron, S.; Wentzell, J.; Kanji, S.; Nguyen, T.; Kobewka, D.M.; MacDonald, E. Characterization of Serious Adverse Drug Reactions in Hospital to Determine Potential Implications of Mandatory Reporting. Can. J. Hosp. Pharm. 2018, 71, 316–323. [Google Scholar] [CrossRef]
- CFR—Code of Federal Regulations Title 21. Available online: https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfcfr/CFRSearch.cfm?fr=312.32 (accessed on 29 April 2022).
- MedDRA Hierarchy|MedDRA. Available online: https://www.meddra.org/how-to-use/basics/hierarchy (accessed on 3 September 2021).
- Standardised MedDRA Queries|MedDRA. Available online: https://www.meddra.org/standardised-meddra-queries (accessed on 9 August 2020).
- Markides, K. Adverse Drug Withdrawal Events. In Encyclopedia of Health & Aging; SAGE Publications, Inc.: Thousand Oaks, CA, USA, 2007; pp. 11–12. [Google Scholar]
- Bate, A.; Evans, S.J.W. Quantitative Signal Detection Using Spontaneous ADR Reporting: QUANTITATIVE SIGNAL DETECTION. Pharmacoepidemiol. Drug Saf. 2009, 18, 427–436. [Google Scholar] [CrossRef]
- Faillie, J.-L. Case–Non-Case Studies: Principle, Methods, Bias and Interpretation. Therapies 2019, 74, 225–232. [Google Scholar] [CrossRef] [PubMed]
- Bate, A.; Lindquist, M.; Edwards, I.R.; Olsson, S.; Orre, R.; Lansner, A.; De Freitas, R.M. A Bayesian Neural Network Method for Adverse Drug Reaction Signal Generation. Eur. J. Clin. Pharmacol. 1998, 54, 315–321. [Google Scholar] [CrossRef] [PubMed]
- Kulkarni, J.; Storch, A.; Baraniuk, A.; Gilbert, H.; Gavrilidis, E.; Worsley, R. Antipsychotic Use in Pregnancy. Expert Opin. Pharmacother. 2015, 16, 1335–1345. [Google Scholar] [CrossRef] [PubMed]
- Damkier, P.; Videbech, P. The Safety of Second-Generation Antipsychotics During Pregnancy: A Clinically Focused Review. CNS Drugs 2018, 32, 351–366. [Google Scholar] [CrossRef] [PubMed]
- FDA-Approved Drugs: Zyprexa. Available online: https://www.accessdata.fda.gov/scripts/cder/daf/index.cfm?event=overview.process&ApplNo=020592 (accessed on 17 September 2022).
- FDA-Approved Drugs: Seroquel. Available online: https://www.accessdata.fda.gov/scripts/cder/daf/index.cfm?event=overview.process&ApplNo=020639 (accessed on 17 September 2022).
- FDA-Approved Drugs: Abilify. Available online: https://www.accessdata.fda.gov/scripts/cder/daf/index.cfm?event=overview.process&ApplNo=021436 (accessed on 17 September 2022).
- FDA-Approved Drugs: Risperdal. Available online: https://www.accessdata.fda.gov/scripts/cder/daf/index.cfm?event=overview.process&ApplNo=020272 (accessed on 17 September 2022).
- Clark, C.T. Psychotropic Drug Use in Perinatal Women with Bipolar Disorder. Semin. Perinatol. 2020, 44, 151230. [Google Scholar] [CrossRef] [PubMed]
- Hebert, M.; Easterling, T.; Kirby, B.; Carr, D.; Buchanan, M.; Rutherford, T.; Thummel, K.; Fishbein, D.; Unadkat, J. Effects of Pregnancy on CYP3A and P-glycoprotein Activities as Measured by Disposition of Midazolam and Digoxin: A University of Washington Specialized Center of Research Study. Clin. Pharmacol. Ther. 2008, 84, 248–253. [Google Scholar] [CrossRef]
- Markowitz, J.S.; Brown, C.S.; Moore, T.R. Atypical Antipsychotics. Part I: Pharmacology, Pharmacokinetics, and Efficacy. Ann. Pharmacother. 1999, 33, 73–85. [Google Scholar] [CrossRef]
- Kapur, S.; Seeman, P. Does Fast Dissociation from the Dopamine d(2) Receptor Explain the Action of Atypical Antipsychotics?: A New Hypothesis. Am. J. Psychiatry 2001, 158, 360–369. [Google Scholar] [CrossRef]
- Cosci, F.; Chouinard, G. Acute and Persistent Withdrawal Syndromes Following Discontinuation of Psychotropic Medications. Psychother. Psychosom. 2020, 89, 283–306. [Google Scholar] [CrossRef]
- Gentile, S. On Categorizing Gestational, Birth, and Neonatal Complications Following Late Pregnancy Exposure to Antidepressants: The Prenatal Antidepressant Exposure Syndrome. CNS Spectr. 2010, 15, 167–185. [Google Scholar] [CrossRef]
- Holland, J.; Brown, R. Neonatal Venlafaxine Discontinuation Syndrome: A Mini-Review. Eur. J. Paediatr. Neurol. 2017, 21, 264–268. [Google Scholar] [CrossRef]
- Rani, F.; Murray, M.L.; Byrne, P.J.; Wong, I.C.K. Epidemiologic Features of Antipsychotic Prescribing to Children and Adolescents in Primary Care in the United Kingdom. Pediatrics 2008, 121, 1002–1009. [Google Scholar] [CrossRef]
- Alessi-Severini, S.; Biscontri, R.G.; Collins, D.M.; Sareen, J.; Enns, M.W. Ten Years of Antipsychotic Prescribing to Children: A Canadian Population-Based Study. Can. J. Psychiatry 2012, 57, 52–58. [Google Scholar] [CrossRef]
- Ronsley, R.; Scott, D.; Warburton, W.P.; Hamdi, R.D.; Louie, D.C.; Davidson, J.; Panagiotopoulos, C. A Population-Based Study of Antipsychotic Prescription Trends in Children and Adolescents in British Columbia, from 1996 to 2011. Can. J. Psychiatry 2013, 58, 361–369. [Google Scholar] [CrossRef]
- Cooper, W.O.; Arbogast, P.G.; Ding, H.; Hickson, G.B.; Fuchs, D.C.; Ray, W.A. Trends in Prescribing of Antipsychotic Medications for US Children. Ambul. Pediatr. 2006, 6, 79–83. [Google Scholar] [CrossRef]
- Patten, S.B.; Waheed, W.; Bresee, L. A Review of Pharmacoepidemiologic Studies of Antipsychotic Use in Children and Adolescents. Can. J. Psychiatry 2012, 57, 717–721. [Google Scholar] [CrossRef]
- EMA Risperdal. Available online: https://www.ema.europa.eu/en/medicines/human/referrals/risperdal (accessed on 17 September 2022).
- EMA Abilify. Available online: https://www.ema.europa.eu/en/medicines/human/EPAR/abilify (accessed on 17 September 2022).
- EMA Zyprexa. Available online: https://www.ema.europa.eu/en/medicines/human/EPAR/zyprexa (accessed on 15 April 2022).
- EMA Seroquel/XR and Associated Names. Available online: https://www.ema.europa.eu/en/medicines/human/referrals/seroquel-seroquel-xr-associated-names (accessed on 17 September 2022).
- Gardos, G.; O’Cole, J.; Tarsy, D. Withdrawal syndromes associated with antipsychotic drugs. Am. J. Psychiatry 1978, 135, 1321–1324. [Google Scholar] [CrossRef]
- Karaş, H.; Güdük, M.; Saatcioğlu, Ö. Withdrawal-Emergent Dyskinesia and Supersensitivity Psychosis Due to Olanzapine Use. Noro Psikiyatr Ars. 2016, 53, 178–180. [Google Scholar] [CrossRef]
- Chouinard, G. Severe Cases of Neuroleptic-Induced Supersensitivity Psychosis: Diagnostic Criteria for the Disorder and Its Treatment. Schizophr. Res. 1991, 5, 21–33. [Google Scholar] [CrossRef]
- Chouinard, G.; Chouinard, V.-A. Atypical Antipsychotics: CATIE Study, Drug-Induced Movement Disorder and Resulting Iatrogenic Psychiatric-Like Symptoms, Supersensitivity Rebound Psychosis and Withdrawal Discontinuation Syndromes. Psychother. Psychosom. 2008, 77, 69–77. [Google Scholar] [CrossRef]
- Gunnell, D.; Murray, V.; Hawton, K. Use of Paracetamol (Acetaminophen) for Suicide and Nonfatal Poisoning: Worldwide Patterns of Use and Misuse. Suicide Life-Threat. Behav. 2000, 30, 313–326. Available online: https://onlinelibrary.wiley.com/doi/abs/10.1111/j.1943-278X.2000.tb01098.x?casa_token=Noeo2wZ941UAAAAA:nBdMBagR3s8ozo6is4cOIR-gq5rb3YJqvHZqi4PFBkwqyQLMckCykkUrlSqCwsYmHviK849ZDIUd5ggF (accessed on 17 September 2022). [PubMed]
- Résumé Des Caractéristiques Du Produit—TERCIAN 40 Mg/Ml, Solution Buvable En Gouttes—Base de Données Publique Des Médicaments. Available online: https://base-donnees-publique.medicaments.gouv.fr/affichageDoc.php?specid=66615713&typedoc=R#RcpPosoAdmin (accessed on 1 October 2022).
- FDA-Approved Drugs: Sparine. Available online: https://www.accessdata.fda.gov/scripts/cder/daf/index.cfm?event=overview.process&ApplNo=010942 (accessed on 17 September 2022).
- FDA-Approved Drugs: Truxal. Available online: https://www.accessdata.fda.gov/scripts/cder/daf/index.cfm?event=overview.process&ApplNo=012486 (accessed on 17 September 2022).
- What Are NPS? Available online: https://www.unodc.org/LSS/Page/NPS (accessed on 17 September 2022).
- Soussan, C.; Andersson, M.; Kjellgren, A. The Diverse Reasons for Using Novel Psychoactive Substances—A Qualitative Study of the Users’ Own Perspectives. Int. J. Drug Policy 2018, 52, 71–78. [Google Scholar] [CrossRef] [PubMed]
- Bénard-Laribière, A.; Noize, P.; Girodet, P.-O.; Lassalle, R.; Dureau-Pournin, C.; Droz-Perroteau, C.; Fourrier-Réglat, A.; Salvo, F.; Bezin, J.; Pariente, A. Monitoring of Drug Misuse or Potential Misuse in a Nationwide Healthcare Insurance Database: A Cross-Sectional Study in France. Therapies 2019, 74, 469–476. [Google Scholar] [CrossRef] [PubMed]
- Egerton, A.; Ahmad, R.; Hirani, E.; Grasby, P.M. Modulation of Striatal Dopamine Release by 5-HT2A and 5-HT2C Receptor Antagonists: [11C]Raclopride PET Studies in the Rat. Psychopharmacology 2008, 200, 487–496. [Google Scholar] [CrossRef]
- Sansone, R.A.; Sansone, L.A. Is Seroquel Developing an Illicit Reputation for Misuse/Abuse? Psychiatry 2010, 7, 13–16. [Google Scholar]
- Mattson, M.E.; Albright, V.A.; Yoon, J.; Council, C.L. Emergency Department Visits Involving Misuse and Abuse of the Antipsychotic Quetiapine: Results from the Drug Abuse Warning Network (DAWN). Subst. Abus. 2015, 9, SART-S22233. [Google Scholar] [CrossRef]
- Klein, L.; Bangh, S.; Cole, J.B. Intentional Recreational Abuse of Quetiapine Compared to Other Second-Generation Antipsychotics. West. J. Emerg. Med. Integr. Emerg. Care Popul. Health 2017, 18, 243–250. [Google Scholar] [CrossRef]
- Hazell, L.; Shakir, S.A.W. Under-Reporting of Adverse Drug Reactions: A Systematic Review. Drug Saf 2006, 29, 385–396. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).