Aloperine: A Potent Modulator of Crucial Biological Mechanisms in Multiple Diseases
Abstract
:1. Introduction
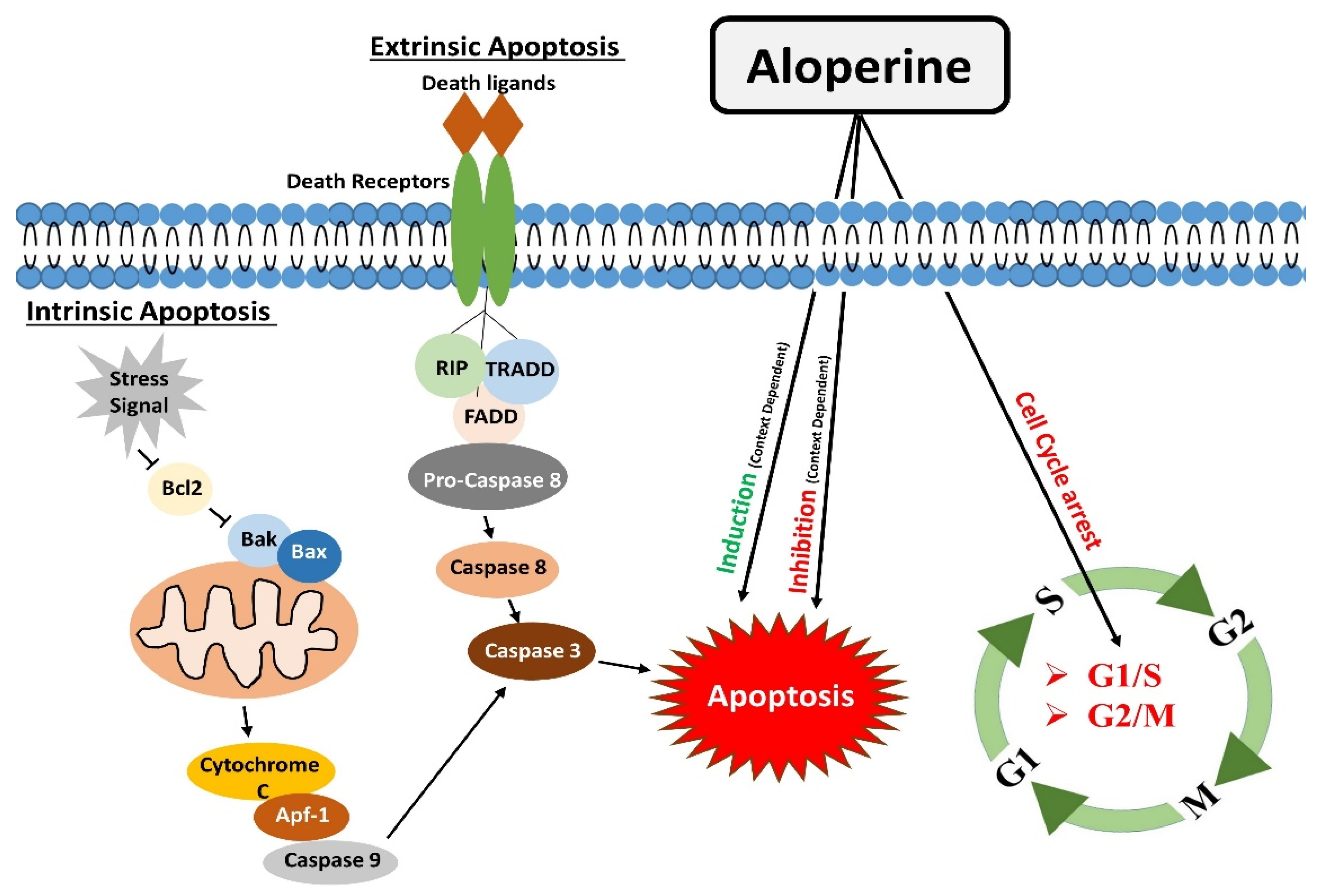
2. Regulation of Apoptosis
3. Modulatory Effects on the Cell Cycle
4. Modulation of Autophagy
5. Inhibitory Effects of Aloperine on Tumor Cell Invasion and Migration
| Apoptosis | ||||||
|---|---|---|---|---|---|---|
| Pathological Conditions | Cell Lines | Animal Model | Dosage | Regulatory Effects of Aloperine | Ref. | |
| In Vitro (µM) | In Vivo | |||||
| Multiple Myeloma | U266 and MM.1S | SCID NOD mice | 50/100/250/500 | 20 mg/kg | Induced Caspase-dependent apoptosis | [12] |
| Prostate cancer | PC3, DU145 and LNCaP | BALB/C mice | 100/200 | 30 mg/kg | Induced Caspase dependent apoptosis | [22] |
| Hepatocellular carcinoma | Hep3B and Huh7 | Zebrafish embryo | 200/350/500 | 100 µM, 150 µM | Induced Mitochondria-dependent apoptosis | [23] |
| Osteosarcoma | MG-63 and U2OS | --------- | 100/200 | --------- | Induced Mitochondria-dependent apoptosis | [11] |
| Colon cancer | HCT116 | --------- | 250/500 | -------- | Induced Mitochondria-dependent apoptosis | [14] |
| Breast cancer | MCF-7 and MDA-MB-231 | --------- | 100/200/400 | --------- | Induced Mitochondria-dependent apoptosis | [26] |
| I/R-Induced Renal Injury | RAW264.7 and HK2 | C57BL/6 mice | 500 | 50 mg/kg | Inhibition of Apoptosis | [15] |
| Thyroid Cancer | IHH-4,8505c and KMH-2 | --------- | 100/200 | --------- | Induced Caspase-dependent apoptosis | [30] |
| Leukemia | HL-60 | --------- | 50/100 | --------- | Induced Mitochondria-dependent apoptosis | [7] |
| Alzheimer’s disease | N2a/Swe.D9 | --------- | 100 | --------- | Induced Mitochondria-dependent apoptosis | [43] |
| Non-small cell lung cancer | H1944 and NCI-H1869 | BALB/C nude mice | 250 | 30 mg/kg | Induced Mitochondria-dependent apoptosis | [24] |
| Intervertebral disc degeneration | Nucleus Pulposus cells | Sprague-Dawley rats | 100 | --------- | Inhibition of Apoptosis | [44] |
| Bladder Cancer | EJ cells | --------- | 25/50/100 | --------- | Induced Mitochondria-dependent apoptosis | [59] |
| OGD/RP neuronal injury | Hippocampal Neuronal cells | Sprague-Dawley rats | 100/200/400 | --------- | Inhibition of Apoptosis | [60] |
| Colorectal Cancer | SW480 and HT29 | --------- | 200/400/800/1000 | --------- | Induced Mitochondria-dependent apoptosis | [40] |
| Early brain injury | --------- | Sprague-Dawley rats | --------- | 75/150 mg/kg | Inhibition of Apoptosis | [17] |
| I/R-Induced Cerebral injury | --------- | Sprague-Dawley rats | --------- | 2/25/50 mg/kg | Inhibition of Apoptosis | [16] |
| Retinal pigment epithelial cells injury | ARPE-19 | --------- | 6.25/12.5/25 | --------- | Inhibition of Apoptosis | [19] |
| DSS-Induced Colitis | Jurkat Cells | C57BL/6 mice | 250/500 | 40 mg/kg | Inhibition of Apoptosis | [29] |
| Microembolisation-Induced cardiac Injury | --------- | Sprague-Dawley rats | --------- | 200 mg/kg | Inhibition of Apoptosis | [61] |
| Cell Cycle | ||||||
| Prostate cancer | PC3, DU145 and LNCaP | BALB/C mice | 100/200 | 30 mg/kg | G1 phase arrest | [22] |
| Hepatocellular carcinoma | Hep3B and Huh7 | Zebrafish embryo | 200/350/500 | 100 µM, 150 µM | G2 phase arrest | [23] |
| Colon cancer | HCT116 | --------- | 250/500 | --------- | G2 phase arrest | [14] |
| Thyroid Cancer | IHH-4,8505c and KMH-2 | --------- | 100/200 | --------- | No impact on Cell Cycle | [30] |
| Non-small cell lung cancer | H1944 and NCI-H1869 | BALB/C nude mice | 250 | 30 mg/kg | G1 phase arrest | [24] |
| Liver cancer | SNU-182 | --------- | 5 | --------- | G2 phase arrest | [21] |
| Autophagy | ||||||
| Thyroid Cancer | KMH-2 and IHH-4 | --------- | 200 | --------- | Autophagy induction | [27] |
| Thyroid Cancer | 8505c | --------- | 200 | --------- | Autophagy inhibition | [27] |
| Leukaemia | HL-60 | --------- | 50/100 | --------- | Autophagy induction | [7] |
| Migration and Invasion | ||||||
| Breast cancer | MCF-7 and MDA-MB-231 | --------- | 100/200/400 | --------- | Inhibition of Migration and Invasion | [26] |
| Liver cancer | SNU-182 | --------- | 5 | --------- | Inhibition of Migration and Invasion | [21] |

6. Modulatory Effects on PI3K/Akt/mTOR Signaling
7. Inhibition of NF-κB Signaling
8. Activation of Nrf2 Signaling
9. Inhibition of Ras Signaling
| PI3K/Akt and Other Downstream Molecules Signaling | ||||||
|---|---|---|---|---|---|---|
| Pathological Conditions | Cell Lines | Animal Model | Dosage | Regulatory Effects of Aloperine | Ref. | |
| In Vitro (µM) | In Vivo | |||||
| Prostate cancer | PC3, DU145 and LNCaP | BALB/C mice | 100/200 | 30 mg/kg | Inhibition of Akt/ERK signaling | [22] |
| Hepatocellular carcinoma | Hep3B and Huh7 | Zebrafish embryo | 200/350/500 | 100 µM, 150 µM | Inhibition of PI3K/Akt signaling | [23] |
| Osteosarcoma | MG-63 and U2OS | --------- | 100/200 | --------- | Inhibition of PI3K/Akt signaling | [11] |
| Colon cancer | HCT116 | --------- | 250/500 | --------- | Inhibition of PI3K/Akt signaling | [14] |
| I/R-Induced Renal Injury | RAW264.7 and HK2 | C57BL/6 mice | 500 | 50 mg/kg | Inhibition of PI3K/Akt/mTOR signaling | [15] |
| Thyroid Cancer | KMH-2 and IHH-4 | --------- | 200 | --------- | Inhibition of Akt/mTOR signaling | [27] |
| Thyroid Cancer | IHH-4,8505c and KMH-2 | -------- | 100/200 | ------- | Inhibition of Akt signaling | [30] |
| DSS-Induced Colitis | Jurkat Cells | C57BL/6 mice | 250/500 | 40 mg/kg | Inhibition of PI3K/Akt/mTOR signaling | [29] |
| Microembolisation-Induced cardiac Injury | --------- | Sprague-Dawley rats | --------- | 200 mg/kg | Activation of the PI3K/Akt signaling | [61] |
| I/R-Induced Cerebral injury | --------- | Sprague-Dawley rats | --------- | 2/25/50 mg/kg | Activation of the PI3K/Akt signaling | [16] |
| NF-κB Signaling | ||||||
| Allergic airway inflammation | --------- | BALB/c mice | --------- | 100/200 mg/kg | Inhibition of NF-κB signaling | [18] |
| Neuropathic pain | --------- | ICR mice | --------- | 80 mg/kg | Inhibition of NF-κB signaling | [31] |
| Intervertebral disc degeneration | Nucleus Pulposus cells | Sprague-Dawley rats | 100 | ------- | Inhibition of NF-κB signaling | [44] |
| Pulmonary arterial hypertension | --------- | Sprague-Dawley rats | --------- | 25/50/100 mg/kg | Inhibition of NF-κB signaling | [124] |
| Osteoporosis | RAW264.7 | C57BL/6 mice | 20 | 30 mg/Kg | Inhibition of NF-κB signaling | [121] |
| LPS-induced macrophage activation | RAW264.7 | --------- | 50/100 | --------- | Inhibition of NF-κB signaling | [111] |
| Nrf2/HO-1 Signaling | ||||||
| Allergic airway inflammation | --------- | BALB/c mice | --------- | 100/200 mg/kg | Activation of Nrf2/HO-1 Signaling | [18] |
| Retinal pigment epithelial cells injury | ARPE-19 | --------- | 6.25/12.5/25 | --------- | Activation of Nrf2/HO-1 Signaling | [19] |
| High Glucose induced Schwann cells injury | RSC96 cells | --------- | 1/10/50 | --------- | Activation of Nrf2/HO-1 Signaling | [21] |
| CCl4 induced mouse hepatic injury | --------- | C57BL/6 mice | --------- | 50/100 mg/kg | Activation of Nrf2/HO-1 Signaling | [147] |
| Ras Signaling | ||||||
| Breast cancer | MCF-7 and MDA-MB-231 | --------- | 100/200/400 | --------- | Inhibition of Ras signaling | [26] |
| Bladder Cancer | EJ cells | ---------- | 25/50/100 | --------- | Inhibition of Ras signaling | [59] |


10. Conclusions and Future Prospects
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AHR | Airway hyper-responsiveness |
| ALP | Alkaline phosphatase |
| ALT | Alanine transaminase |
| AMD | Age-related macular degeneration |
| Apaf-1 | Protease activating factor-1 |
| ARE | Antioxidant response element |
| AST | Aspartate aminotransferase |
| ATP | Adenosine triphosphate |
| BAFFR | B-cell activating factor receptor |
| Bax | Bcl2-associated X protein |
| Bcl2 | B-cell lymphoma 2 |
| BMM | Bone Marrow-Derived Macrophages |
| CCI | Chronic constriction injury (CCI) |
| CCl4 | Carbon Tetrachloride |
| CD40 | Cluster of differentiation 40 |
| Cdc2 | Cell-Division Cycle 2 |
| Cdc25C | Cell division cycle 25 |
| CDK | Cyclin-dependent protein kinase |
| cFLIP | Cellular FLICE (FADD-like IL-1β-converting enzyme)-inhibitory protein |
| CL | Clearance |
| CME | Coronary micro-embolization |
| CNC | Cap ‘n’ collar |
| CO | Carbon monoxide |
| DSS | Dextran sodium sulfate |
| E2F1 | E2F Transcription Factor 1 |
| EGF | Epidermal growth factor |
| EGFR | Epidermal growth factor receptor |
| EMT | Epithelial-mesenchymal transition |
| ERK | Extracellular signal-regulated kinase |
| ERK1/2 | Extracellular signal-regulated kinases 1 and 2 |
| FADD | Fas-associated death domain |
| GRO1 | Growth Regulated Oncogene 1 |
| GST | Glutathione S-transferase |
| GTP | guanosine 5’-triphosphate |
| H2O2 | Hydrogen peroxide |
| HO-1 | Heme oxygenase-1 |
| IkBα | Nuclear factor of kappa light polypeptide gene enhancer in B-cells inhibitor, alpha |
| IKK | IκB kinase |
| IL-13 | Interleukin-13 |
| IL-4 | Interleukin-4 |
| IL-5 | Interleukin-5 |
| IL-6 | Interleukin-6 |
| IR | Ischemia and reperfusion |
| LC3 | 1A/1B-light chain 3 |
| LTβR | Lymphotoxin beta receptor |
| MAPK | Mitogen-activated protein kinase |
| MCAO | Middle cerebral artery occlusion |
| MDA | Malondialdehyde |
| MEK | Mitogen-activated protein kinase kinase |
| MEK1/2 | Mitogen-activated protein kinase kinases 1 and 2 |
| MMP | Matrix metalloproteinases |
| mTOR | Mammalian target of rapamycin |
| NADPH | Nicotinamide adenine dinucleotide phosphate |
| NF-kB | Nuclear factor kappa-light-chain-enhancer of activated B cells |
| NIK | NF-κB-inducing kinase |
| NQO1 | NAD(P)H: quinone oxidoreductase 1 |
| Nrf2 | Nuclear factor erythroid 2-related factor 2 |
| NSCLC | Non-small cell lung cancer. |
| OGD-RP | Oxygen-glucose deprivation-reperfusion |
| PAH | Pulmonary arterial hypertension |
| PARP | Poly ADP ribose polymerase |
| PI | Propidium Iodide |
| PI3K | Phosphatidylinositol-3 kinase and PI3 kinase. |
| Rb | Retinoblastoma Tumor Suppressor Protein |
| PRR | Pattern recognition receptor |
| RANK | Receptor activator of nuclear factor κB |
| RANKL | Receptor activator of nuclear factor κB ligand |
| ROS | Reactive oxygen species |
| RPE | Retinal pigment epithelium |
| RTK | Receptor tyrosine kinase |
| SFDA | Chinese state food and drug administration |
| Snail | Zinc finger protein SNAI1 |
| Th17 | T helper cell 17 |
| T1/2 | Half-life |
| Tmax | Time to reach maximum concentration |
| TNFR | TNF receptors TNFR1 |
| TNF-α | Tumor Necrosis Factor alpha |
| TRAIL | Tumor Necrosis Factor-Alpha-Related Apoptosis-Inducing Ligand |
| TRAIL-R1/2 | Tumor Necrosis Factor-related Apoptosis-inducing Ligand Receptor 1/2 |
| Tregs | Regulatory T-cells |
| Twist1 | Twist-related protein 1 |
| Vd | Apparent volume of distribution |
| XAR | Xenobiotic-activated receptor (XAR) |
References
- Chen, X.; Yi, C.; Yang, X.; Wang, X. Liquid chromatography of active principles in Sophora fla-vescens root. J. Chromatogr. B 2004, 812, 149–163. [Google Scholar] [CrossRef]
- Wang, H.; Guo, S.; Qian, D.; Qian, Y.; Duan, J.-A. Comparative analysis of quinolizidine alkaloids from different parts of Sophora alopecuroides seeds by UPLC–MS/MS. J. Pharm. Biomed. Anal. 2012, 67–68, 16–21. [Google Scholar] [CrossRef]
- Kangli, M.; Jianzhong, Z.; Ying, D.; Yufei, X. Research progress on the chemical compounds and pharmacology of Sophora flavescens. Nat. Prod. Res. Dev. 2001, 13, 69–73. [Google Scholar]
- Tolkachev, O.; Monakhova, T.; Sheichenko, V.; Kabanov, V.; Fesenko, O.; Proskurnina, N. Alka-loids of a new type from Sophora alopecuroides L. Chem. Nat. Compd. 1975, 11, 29–34. [Google Scholar] [CrossRef]
- Brosius, A.; Ziller, J.; Zhang, Q. Relative and absolute configuration of aloperine. Acta Crystallogr. Sect. C Cryst. Struct. Commun. 1997, 53, 1510–1512. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, L.; Chen, J.; Lin, Y. Clinical observation on the efficacy of mateling injection combined with radiotherapy in treating nasopharyngeal tumors. Strait Pharm. J. 1996, 8, 41–43. [Google Scholar]
- Lin, Z.; Huang, C.-F.; Liu, X.-S.; Jiang, J. In Vitro Anti-Tumour Activities of Quinolizidine Alkaloids Derived from Sophora Flavescens Ait. Basic Clin. Pharmacol. Toxicol. 2010, 108, 304–309. [Google Scholar] [CrossRef] [PubMed]
- Zhou, C.C.; Gao, H.B.; Sun, X.B.; Shi, H.B.; Liu, W.; Yuan, H.N.; Wang, Z.X. Anti-inflammatory and anti-allergic action of aloperine. Zhongguo Yao Li Xue Bao Acta Pharmacol. Sin. 1989, 10, 360–365. [Google Scholar]
- Li Fan, S.; Zhang, S. Antiviral effect of aloperine. J. Zhong Cao Yao 1998, 29, 253–254. [Google Scholar]
- Elmore, S. Apoptosis: A review of programmed cell death. Toxicol. Pathol. 2007, 35, 495–516. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Jin, Z.; Dai, L.; Wu, H.; Wang, J.; Wang, L.; Zhou, Z.; Yang, L.; Gao, W. Aloperine in-duces apoptosis and inhibits invasion in MG-63 and U2OS human osteosarcoma cells. Biomed. Pharmacother. 2018, 97, 45–52. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Yang, S.; Zhou, H.; Sun, M.; Du, L.; Wei, M.; Luo, M.; Huang, J.; Deng, H.; Feng, Y. Aloperine executes antitumor effects against multiple myeloma through dual apoptotic mecha-nisms. J. Hematol. Oncol. 2015, 8, 1–13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xu, Z.; Yan, Y.; Zeng, S.; Qian, L.; Dai, S.; Xiao, L.; Wang, L.; Yang, X.; Xiao, Y.; Gong, Z. Re-ducing autophagy and inducing G1 phase arrest by aloperine enhances radio-sensitivity in lung cancer cells. Oncol. Rep. 2017. [Google Scholar] [CrossRef]
- Zhang, L.; Zheng, Y.; Deng, H.; Liang, L.; Peng, J. Aloperine induces G2/M phase cell cycle arrest and apoptosis in HCT116 human colon cancer cells. Int. J. Mol. Med. 2014, 33, 1613–1620. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hu, S.; Zhang, Y.; Zhang, M.; Guo, Y.; Yang, P.; Zhang, S.; Simsekyilmaz, S.; Xu, J.-F.; Li, J.; Xiang, X. Aloperine protects mice against ischemia-reperfusion (IR)-induced renal injury by regu-lating PI3K/AKT/mTOR signaling and AP-1 activity. Mol. Med. 2015, 21, 912–923. [Google Scholar] [CrossRef]
- Li, Z.; Cao, X.; Xiao, L.; Zhou, R. Aloperine protects against cerebral ischemia/reperfusion injury via activating the PI3K/AKT signaling pathway in rats. Exp. Ther. Med. 2021, 22, 1–8. [Google Scholar] [CrossRef]
- Song, S.; Chen, Y.; Han, F.; Dong, M.; Xiang, X.; Sui, J.; Li, Y.; Yang, H.; Liu, J. Aloperine acti-vates the Nrf2-ARE pathway when ameliorating early brain injury in a subarachnoid hemorrhage model. Exp. Ther. Med. 2018, 15, 3847–3855. [Google Scholar]
- Wang, C.; Choi, Y.H.; Xian, Z.; Zheng, M.; Piao, H.; Yan, G. Aloperine suppresses allergic airway inflammation through NF-κB, MAPK, and Nrf2/HO-1 signaling pathways in mice. Int. Immunopharmacol. 2018, 65, 571–579. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Zhou, H.; Chen, J.; Lv, X.; Liu, H. Aloperine protects human retinal pigment epithelial cells against hydrogen peroxide–induced oxidative stress and apoptosis through activation of Nrf2/HO-1 pathway. J. Recept. Signal Transduct. 2022, 42, 88–94. [Google Scholar] [CrossRef]
- Casimiro, M.C.; Crosariol, M.; Loro, E.; Li, Z.; Pestell, R.G. Cyclins and cell cycle control in cancer and disease. Genes Cancer 2012, 3, 649–657. [Google Scholar] [CrossRef]
- Huang, H.; Cao, Y.; Huang, L.; Lu, R.; Wang, J.; Zhou, Y. Aloperine suppresses the proliferation, migration and invasion of human liver cancer cells via induction of G2/M cell cycle arrest and inhibition of GROα expression. All Life 2021, 14, 392–400. [Google Scholar] [CrossRef]
- Ling, Z.; Guan, H.; You, Z.; Wang, C.; Hu, L.; Zhang, L.; Wang, Y.; Chen, S.; Xu, B.; Chen, M. Aloperine executes antitumor effects through the induction of apoptosis and cell cycle arrest in prostate cancer n vitro and in vivo. OncoTargets Ther. 2018, 11, 2735. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, J.-S.; Huo, C.-Y.; Cao, H.-H.; Fan, C.-L.; Hu, J.-Y.; Deng, L.-J.; Lu, Z.-B.; Yang, H.-Y.; Yu, L.-Z.; Mo, Z.-X. Aloperine induces apoptosis and G2/M cell cycle arrest in hepatocellular carcinoma cells through the PI3K/Akt signaling pathway. Phytomedicine 2019, 61, 152843. [Google Scholar] [CrossRef] [PubMed]
- Muhammad, T.; Sakhawat, A.; Khan, A.A.; Huang, H.; Khan, H.R.; Huang, Y.; Wang, J. Aloperine in combination with therapeutic adenoviral vector synergistically suppressed the growth of non-small cell lung cancer. J. Cancer Res. Clin. Oncol. 2020, 146, 861–874. [Google Scholar] [CrossRef] [PubMed]
- Lv, W.; Liu, Q.; An, J.; Song, X. Aloperine prevents hypoxia-induced epithelial-mesenchymal transition in bladder cancer cells through regulating the mTOR/p70S6K/4E-BP1 pathway. Preprint 2020. [Google Scholar] [CrossRef] [Green Version]
- Tian, D.; Li, Y.; Li, X.; Tian, Z. Aloperine inhibits proliferation, migration and invasion and induces apoptosis by blocking the Ras signaling pathway in human breast cancer cells. Mol. Med. Rep. 2018, 18, 3699–3710. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yu, H.-I.; Shen, H.-C.; Chen, S.-H.; Lim, Y.-P.; Chuang, H.-H.; Tai, T.-S.; Kung, F.-P.; Lu, C.-H.; Hou, C.-Y.; Lee, Y.-R. Autophagy Modulation in Human Thyroid Cancer Cells following Aloperine Treatment. Int. J. Mol. Sci. 2019, 20, 5315. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Holz, R.W.; Fisher, S.K. Synaptic transmission and cellular signaling: An overview. Basic Neurochem. 2012, 235–257. [Google Scholar]
- Fu, X.; Sun, F.; Wang, F.; Zhang, J.; Zheng, B.; Zhong, J.; Yue, T.; Zheng, X.; Xu, J.-F.; Wang, C.-Y. Aloperine protects mice against DSS-induced colitis by PP2A-mediated PI3K/Akt/mTOR signaling suppression. Mediat. Inflamm. 2017, 2017, 5706152. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, Y.-R.; Chen, S.-H.; Lin, C.-Y.; Chao, W.-Y.; Lim, Y.-P.; Yu, H.-I.; Lu, C.-H. In Vitro Antitumor Activity of Aloperine on Human Thyroid Cancer Cells through Caspase-Dependent Apoptosis. Int. J. Mol. Sci. 2018, 19, 312. [Google Scholar] [CrossRef] [Green Version]
- Xu, Y.-Q.; Jin, S.-J.; Liu, N.; Li, Y.-X.; Zheng, J.; Ma, L.; Du, J.; Zhou, R.; Zhao, C.-J.; Niu, Y. Aloperine attenuated neuropathic pain induced by chronic constriction injury via anti-oxidation activity and suppression of the nuclear factor kappa B pathway. Biochem. Biophys. Res. Commun. 2014, 451, 568–573. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Ma, T.; Wang, Z.; Jia, L.; Zhang, X.; He, Q.; Liu, S. Aloperine attenuates high glucose-induced oxidative injury in Schwann cells via activation of NRF2/HO-1 pathway. Trop. J. Pharm. Res. 2020, 19, 1147–1152. [Google Scholar] [CrossRef]
- Shin, S.-S.; Park, Y.-J.; Hwang, B.; Park, S.L.; Han, S.-W.; Park, S.-S.; Choi, Y.H.; Kim, W.-J.; Moon, S.-K. Triacanthine exerts antitumor effects on bladder cancer in vitro and in vivo. Phytomedicine 2019, 64, 153069. [Google Scholar] [CrossRef] [PubMed]
- D’Arcy, M.S. Cell death: A review of the major forms of apoptosis, necrosis and autophagy. Cell Biol. Int. 2019, 43, 582–592. [Google Scholar] [CrossRef] [PubMed]
- Bertheloot, D.; Latz, E.; Franklin, B.S. Necroptosis, pyroptosis and apoptosis: An intricate game of cell death. Cell. Mol. Immunol. 2021, 18, 1106–1121. [Google Scholar] [CrossRef] [PubMed]
- Jan, R.; Chaudhry, G.-E.-S. Understanding Apoptosis and Apoptotic Pathways Targeted Cancer Therapeutics. Adv. Pharm. Bull. 2019, 9, 205–218. [Google Scholar] [CrossRef] [Green Version]
- Xu, Z.; Wang, X.; Chen, X.; Zeng, S.; Qian, L.; Wei, J.; Gong, Z.; Yan, Y. Identification of Aloperine as an anti-apoptotic Bcl2 protein inhibitor in glioma cells. PeerJ 2019, 7, e7652. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Porter, A.G.; Jänicke, R.U. Emerging roles of caspase-3 in apoptosis. Cell Death Differ. 1999, 6, 99–104. [Google Scholar] [CrossRef] [PubMed]
- Tsujimoto, Y. Role of Bcl-2 family proteins in apoptosis: Apoptosomes or mitochondria? Genes Cells 1998, 3, 697–707. [Google Scholar] [CrossRef]
- Han, W.; Kong, D.; Lu, Q.; Zhang, W.; Fan, Z. Aloperine inhibits proliferation and promotes apoptosis in colorectal cancer cells by regulating the circNSUN2/miR-296-5p/STAT3 pathway. Drug Des. Dev. Ther. 2021, 15, 857. [Google Scholar] [CrossRef] [PubMed]
- Schrier, R.W.; Wang, W. Acute Renal Failure and Sepsis. N. Engl. J. Med. 2004, 351, 159–169. [Google Scholar] [CrossRef]
- Liang, H.L.; Arsenault, J.; Mortensen, J.; Park, F.; Johnson, C.P.; Nilakantan, V. Partial attenuation of cytotoxicity and apoptosis by SOD1 in ischemic renal epithelial cells. Apoptosis 2009, 14, 1176–1189. [Google Scholar] [CrossRef] [Green Version]
- Zhao, J.; Zhang, G.; Li, M.; Luo, Q.; Leng, Y.; Liu, X. Neuro-protective effects of aloperine in an Alzheimer’s disease cellular model. Biomed. Pharmacother. 2018, 108, 137–143. [Google Scholar] [CrossRef] [PubMed]
- Ren, D.; Ma, W.; Guo, B.; Wang, S. Aloperine attenuates hydrogen peroxide-induced injury via anti-apoptotic activity and suppression of the nuclear factor-κB signaling pathway. Exp. Ther. Med. 2017, 13, 315–320. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hartwell, L.H.; Kastan, M.B. Cell cycle control and cancer. Science 1994, 266, 1821–1828. [Google Scholar] [CrossRef] [PubMed]
- Boward, B.; Wu, T.; Dalton, S. Concise review: Control of cell fate through cell cycle and pluripo-tency networks. Stem Cell. 2016, 34, 1427–1436. [Google Scholar] [CrossRef] [Green Version]
- Mizushima, N. Autophagy: Process and function. Genes Dev. 2007, 21, 2861–2873. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Suzuki, K.; Ohsumi, Y. Molecular machinery of autophagosome formation in yeast, Saccharomyces cerevisiae. FEBS Lett. 2007, 581, 2156–2161. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Badadani, M. Autophagy Mechanism, Regulation, Functions, and Disorders. ISRN Cell Biol. 2012, 2012, 927064. [Google Scholar] [CrossRef] [Green Version]
- Glick, D.; Barth, S.; MacLeod, K.F. Autophagy: Cellular and molecular mechanisms. J. Pathol. 2010, 221, 3–12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Klionsky, D.J.; Abdel-Aziz, A.K.; Abdelfatah, S.; Abdellatif, M.; Abdoli, A.; Abel, S.; Abeliovich, H.; Abildgaard, M.H.; Abudu, Y.P.; Acevedo-Arozena, A.; et al. Guidelines for the use and interpretation of assays for monitoring autophagy (4th edition). Autophagy 2020, 17, 1–382. [Google Scholar] [CrossRef]
- Cheung, K.J.; Ewald, A.J. A collective route to metastasis: Seeding by tumor cell clusters. Science 2016, 352, 167–169. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brown, G.T.; Murray, G.I. Current mechanistic insights into the roles of matrix metalloproteinases in tumour invasion and metastasis. J. Pathol. 2015, 237, 273–281. [Google Scholar] [CrossRef]
- Curran, S.; Murray, G.I. Matrix metalloproteinases in tumour invasion and metastasis. J. Pathol. 1999, 189, 300–308. [Google Scholar] [CrossRef]
- Curran, S.; Murray, G.I. Matrix metalloproteinases: Molecular aspects of their roles in tumour in-vasion and metastasis. Eur. J. Cancer 2000, 36, 1621–1630. [Google Scholar] [CrossRef]
- Hollestelle, A.; Peeters, J.K.; Smid, M.; Timmermans, M.; Verhoog, L.C.; Westenend, P.J.; Heine, A.A.J.; Chan, A.; Sieuwerts, A.M.; Wiemer, E.; et al. Loss of E-cadherin is not a necessity for epithelial to mesenchymal transition in human breast cancer. Breast Cancer Res. Treat. 2013, 138, 47–57. [Google Scholar] [CrossRef]
- Kalluri, R.; Weinberg, R.A. The basics of epithelial-mesenchymal transition. J. Clin. Investig. 2009, 119, 1420–1428. [Google Scholar] [CrossRef] [Green Version]
- Bonnitcha, P.; Grieve, S.; Figtree, G. Clinical imaging of hypoxia: Current status and future directions. Free. Radic. Biol. Med. 2018, 126, 296–312. [Google Scholar] [CrossRef]
- Zhang, L.; Liang, J.; Liu, X.; Wu, J.; Tan, D.; Hu, W. Aloperine Exerts Antitumor Effects on Bladder Cancer in vitro. OncoTargets Ther. 2020, 13, 10351–10360. [Google Scholar] [CrossRef] [PubMed]
- Ma, N.-T.; Zhou, R.; Chang, R.-Y.; Hao, Y.-J.; Ma, L.; Jin, S.-J.; Du, J.; Zheng, J.; Zhao, C.-J.; Niu, Y.; et al. Protective effects of aloperine on neonatal rat primary cultured hippocampal neurons injured by oxygen–glucose deprivation and reperfusion. J. Nat. Med. 2015, 69, 575–583. [Google Scholar] [CrossRef] [PubMed]
- Mao, Q.; Guo, F.; Liang, X.; Wu, Y.; Lu, Y. Aloperine activates the PI3K/Akt pathway and protects against coronary micro-embolisation-induced myocardial injury in rats. Pharmacology 2019, 104, 90–97. [Google Scholar] [CrossRef]
- Fruman, D.A.; Meyers, R.E.; Cantley, L.C. Phosphoinositide kinases. Annu. Rev. Biochem. 1998, 67, 481–507. [Google Scholar] [CrossRef] [PubMed]
- Guo, H.; German, P.; Bai, S.; Barnes, S.; Guo, W.; Qi, X.; Lou, H.; Liang, J.; Jonasch, E.; Mills, G.B.; et al. The PI3K/AKT Pathway and Renal Cell Carcinoma. J. Genet. Genom. 2015, 42, 343–353. [Google Scholar] [CrossRef] [Green Version]
- Vanhaesebroeck, B.; Guillermet-Guibert, J.; Graupera, M.; Bilanges, B. The emerging mechanisms of isoform-specific PI3K signalling. Nat. Rev. Mol. Cell Biol. 2010, 11, 329–341. [Google Scholar] [CrossRef]
- Auger, K.R.; Serunian, L.A.; Soltoff, S.P.; Libby, P.; Cantley, L.C. PDGF-dependent tyrosine phosphorylation stimulates pro-duction of novel polyphosphoinositides in intact cells. Cell 1989, 57, 167–175. [Google Scholar] [CrossRef]
- Fruman, D.A.; Rommel, C. PI3K and cancer: Lessons, challenges and opportunities. Nat. Rev. Drug Discov. 2014, 13, 140–156. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Osaki, M.; Oshimura, M.; Ito, H. PI3K-Akt pathway: Its functions and alterations in human cancer. Apoptosis 2004, 9, 667–676. [Google Scholar] [CrossRef]
- Ruderman, N.B.; Kapeller, R.; White, M.F.; Cantley, L.C. Activation of phosphatidylinositol 3-kinase by insulin. Proc. Natl. Acad. Sci. USA 1990, 87, 1411–1415. [Google Scholar] [CrossRef] [Green Version]
- Thorpe, L.M.; Yuzugullu, H.; Zhao, J.J. PI3K in cancer: Divergent roles of isoforms, modes of activation and therapeutic tar-geting. Nat. Rev. Cancer 2015, 15, 7–24. [Google Scholar] [CrossRef]
- Hua, H.; Zhu, Y.; Song, Y.-H. Ruscogenin suppressed the hepatocellular carcinoma metastasis via PI3K/Akt/mTOR signaling pathway. Biomed. Pharmacother. 2018, 101, 115–122. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.-H.; Tian, X.-Y.; An, Q.-M.; Guan, X.-Y.; Hao, C.-Y. LINC00963 promotes hepatocellular carcinoma progression by activating PI3K/AKT pathway. Eur. Rev. Med. Pharmacol. Sci. 2018, 22, 1645–1652. [Google Scholar]
- Psyrri, A.; Arkadopoulos, N.; Vassilakopoulou, M.; Smyrniotis, V.; Dimitriadis, G. Pathways and targets in hepatocellular carcinoma. Expert Rev. Anticancer Ther. 2012, 12, 1347–1357. [Google Scholar] [CrossRef] [PubMed]
- Benjamin, R.S. Osteosarcoma: Better treatment through better trial design. Lancet Oncol. 2015, 16, 12–13. [Google Scholar] [CrossRef]
- Walkley, C.R.; Qudsi, R.; Sankaran, V.G.; Perry, J.A.; Gostissa, M.; Roth, S.I.; Rodda, S.J.; Snay, E.; Dunning, P.; Fahey, F.H.; et al. Conditional mouse osteosarcoma, dependent on p53 loss and potentiated by loss of Rb, mimics the human disease. Genes Dev. 2008, 22, 1662–1676. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- DeSantis, C.; Naishadham, D.; Jemal, A. Cancer statistics for African Americans, 2013. CA Cancer J. Clin. 2013, 63, 151–166. [Google Scholar] [CrossRef]
- Schrier, R.W.; Wang, W.; Poole, B.; Mitra, A. Acute renal failure: Definitions, diagnosis, pathogenesis, and therapy. J. Clin. Investig. 2004, 114, 5–14. [Google Scholar] [CrossRef] [Green Version]
- Amura, C.R.; Renner, B.; Lyubchenko, T.; Faubel, S.; Simonian, P.L.; Thurman, J.M. Complement activation and toll-like re-ceptor-2 signaling contribute to cytokine production after renal ischemia/reperfusion. Mol. Immunol. 2012, 52, 249–257. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fan, L.-H.; He, L.; Cao, Z.-Q.; Xiang, J.; Liu, L. Effect of ischemia preconditioning on renal ischemia/reperfusion injury in rats. Int. Braz. J. Urol. 2012, 38, 842–854. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kusch, A.; Hoff, U.; Bubalo, G.; Zhu, Y.; Fechner, M.; Schmidt-Ullrich, R.; Marko, L.; Müller, D.; Schmidt-Ott, K.; Gürgen, D. Novel signalling mechanisms and targets in renal ischaemia and reperfusion injury. Acta Physiol. 2013, 208, 25–40. [Google Scholar] [CrossRef]
- Sabbahy, M.E.; Vaidya, V.S. Ischemic kidney injury and mechanisms of tissue repair. Wiley Interdiscip. Rev. Syst. Biol. Med. 2011, 3, 606–618. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Siegel, R.; Miller, K.; Jemal, A. Cancer Statistics, 2017. CA Cancer J. Clin. 2017, 67, 7–30. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Robbins, H.L.; Hague, A. The PI3K/Akt Pathway in Tumors of Endocrine Tissues. Front. Endocrinol. 2016, 6, 188. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baumgart, D.C.; Carding, S.R. Inflammatory bowel disease: Cause and immunobiology. Lancet 2007, 369, 1627–1640. [Google Scholar] [CrossRef]
- Mirlekar, B.; Ghorai, S.; Khetmalas, M.; Bopanna, R.; Chattopadhyay, S. Nuclear matrix protein SMAR1 control regulatory T-cell fate during inflammatory bowel disease (IBD). Mucosal Immunol. 2015, 8, 1184–1200. [Google Scholar] [CrossRef] [PubMed]
- Chi, H. Regulation and function of mTOR signalling in T cell fate decisions. Nat. Rev. Immunol. 2012, 12, 325–338. [Google Scholar] [CrossRef]
- Huynh, A.; DuPage, M.; Priyadharshini, B.; Sage, P.T.; Quiros, J.; Borges, C.M.; Townamchai, N.; Gerriets, V.; Rathmell, J.C.; Sharpe, A.H.; et al. Control of PI(3) kinase in Treg cells maintains homeostasis and lineage stability. Nat. Immunol. 2015, 16, 188–196. [Google Scholar] [CrossRef] [PubMed]
- Cagnol, S.; Chambard, J.C. ERK and cell death: Mechanisms of ERK-induced cell death–apoptosis, autophagy and senes-cence. FEBS J. 2010, 277, 2–21. [Google Scholar] [CrossRef]
- Strozyk, E.; Kulms, D. The role of AKT/mTOR pathway in stress response to UV-irradiation: Implication in skin carcinogene-sis by regulation of apoptosis, autophagy and senescence. Int. J. Mol. Sci. 2013, 14, 15260–15285. [Google Scholar] [CrossRef] [Green Version]
- Heusch, G.; Skyschally, A.; Kleinbongard, P. Coronary microembolization and microvascular dysfunction. Int. J. Cardiol. 2018, 258, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Liang, J.; Li, L.; Sun, Y.; He, W.; Wang, X.; Su, Q. The protective effect of activating Nrf2/HO-1 signaling pathway on cardi-omyocyte apoptosis after coronary microembolization in rats. BMC Cardiovasc. Disord. 2017, 17, 1–9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Collaborators, G. A systematic analysis for the global burden of disease study 2017. Lancet 2018, 392, 1789–1858. [Google Scholar]
- Wang, Z.; Zhou, W.; Dong, H.; Ma, X.; He, Z. Dexmedetomidine pretreatment inhibits cerebral ischemia/reperfusion-induced neuroinflammation via activation of AMPK. Mol. Med. Rep. 2018, 18, 3957–3964. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jung, J.E.; Kim, G.S.; Chen, H.; Maier, C.M.; Narasimhan, P.; Song, Y.S.; Niizuma, K.; Katsu, M.; Okami, N.; Yoshioka, H.; et al. Reperfusion and Neurovascular Dysfunction in Stroke: From Basic Mechanisms to Potential Strategies for Neuroprotection. Mol. Neurobiol. 2010, 41, 172–179. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhao, Y.; Xu, J. Sanggenon C ameliorates cerebral ischemia-reperfusion injury by inhibiting inflammation and oxidative stress through regulating RhoA-ROCK signaling. Inflammation 2020, 43, 1476–1487. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oeckinghaus, A.; Ghosh, S. The NF-κB family of transcription factors and its regulation. Cold Spring Harb. Perspect. Biol. 2009, 1, a000034. [Google Scholar] [CrossRef] [PubMed]
- Miraghazadeh, B.; Cook, M.C. Nuclear factor-kappaB in autoimmunity: Man and mouse. Front. Immunol. 2018, 9, 613. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kuhn, L.B.; Zapf, S.; Kristina, D.; Strobl, D.C.; Weih, F.; Blum, H.; Weigert, O.; Strobl, L.J.; Ursula, Z.-S. The non-canonical NF-kappaB Signaling Pathway Contributes to the Expansion and Lymphomagenesis of CD40-activated B Cells. Blood 2018, 132, 1340. [Google Scholar] [CrossRef]
- Beinke, S.; Ley, S.C. Functions of NF-kappaB 1 and NF-kappaB2 in immune cell biology. Biochem. J. 2004, 382, 393–409. [Google Scholar] [CrossRef] [PubMed]
- Vallabhapurapu, S.; Karin, M. Regulation and function of NF-κB transcription factors in the immune system. Annu. Rev. Immunol. 2009, 27, 693–733. [Google Scholar] [CrossRef]
- Zhang, H.; Sun, S.-C. NF-κB in inflammation and renal diseases. Cell Biosci. 2015, 5, 1–12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hayden, M.S.; Ghosh, S. Shared principles in NF-κB signaling. Cell 2008, 132, 344–362. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Karin, M.; Delhase, M. The IκB kinase (IKK) and NF-κB: Key elements of proinflammatory signalling. Semin. Immunol. 2000, 12, 85–98. [Google Scholar] [CrossRef] [PubMed]
- Sun, S.-C. Non-canonical NF-κB signaling pathway. Cell Res. 2011, 21, 71–85. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sun, S.-C.; Liu, Z.-G. A special issue on NF-κB signaling and function. Cell Res. 2011, 21, 1–2. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, T. The nuclear factor NF-kappaB pathway in inflammation. Cold Spring Harb. Perspect. Biol. 2009, 1, a1651. [Google Scholar] [CrossRef] [Green Version]
- Sutterwala, F.S.; Haasken, S.; Cassel, S.L. Mechanism of NLRP3 inflammasome activation. Ann. N. Y. Acad. Sci. 2014, 1319, 82–95. [Google Scholar] [CrossRef] [PubMed]
- Tak, P.P.; Firestein, G.S. NF-κB: A key role in inflammatory diseases. J. Clin. Investig. 2001, 107, 7–11. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Zhao, W.; Zhang, X.; Chen, X. Neocryptotanshinone inhibits lipopolysaccharide-induced inflammation in RAW264.7 macrophages by suppression of NF-κB and iNOS signaling pathways. Acta Pharm. Sin. B 2015, 5, 323–329. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, L.; Guo, H.; Li, Y.; Meng, X.; Yan, L.; Zhang, D.; Wu, S.; Zhou, H.; Peng, L.; Xie, Q. Oleoylethanolamide exerts anti-inflammatory effects on LPS-induced THP-1 cells by enhancing PPARα signaling and inhibiting the NF-κB and ERK1/2/AP-1/STAT3 pathways. Sci. Rep. 2016, 6, 1–12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jiang, W.-Y.; Jeon, B.-H.; Kim, Y.-C.; Lee, S.H.; Sohn, D.H.; Seo, G.S. PF2401-SF, standardized fraction of Salvia miltiorrhiza shows anti-inflammatory activity in macrophages and acute arthritis in vivo. Int. Immunopharmacol. 2013, 16, 160–164. [Google Scholar] [CrossRef]
- Ye, Y.; Wang, Y.; Yang, Y.; Tao, L. Aloperine suppresses LPS-induced macrophage activation through inhibiting the TLR4/NF-κB pathway. Inflamm. Res. 2020, 69, 375–383. [Google Scholar] [CrossRef] [PubMed]
- Galli, S.J.; Tsai, M.; Piliponsky, A.M. The development of allergic inflammation. Nature 2008, 454, 445–454. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Medoff, B.D.; Thomas, S.Y.; Luster, A.D. T Cell Trafficking in Allergic Asthma: The Ins and Outs. Annu. Rev. Immunol. 2008, 26, 205–232. [Google Scholar] [CrossRef]
- Beitha, I.D.; Kemp, A.; Kenyon, J.; Prout, M.; Chestnut, T.J. Identifying neuropathic back and leg pain: A cross-sectional study. Pain 2011, 152, 1511–1516. [Google Scholar] [CrossRef] [PubMed]
- Sun, T.; Luo, J.; Jia, M.; Li, H.; Li, K.; Fu, Z. Small interfering RNA-mediated knockdown of NF-κBp65 attenuates neuro-pathic pain following peripheral nerve injury in rats. Eur. J. Pharmacol. 2012, 682, 79–85. [Google Scholar] [CrossRef]
- Wang, H.-Q.; Samartzis, D. Clarifying the nomenclature of intervertebral disc degeneration and displacement: From bench to bedside. Int. J. Clin. Exp. Pathol. 2014, 7, 1293–1298. [Google Scholar] [PubMed]
- Jackson, R.D.; Mysiw, W.J. Insights into the epidemiology of postmenopausal osteoporosis: The Women’s Health Initiative. Semin. Reprod. Med. 2014, 32, 454–462. [Google Scholar] [PubMed]
- Nakashima, T.; Hayashi, M.; Fukunaga, T.; Kurata, K.; Oh-Hora, M.; Feng, J.Q.; Bonewald, L.F.; Kodama, T.; Wutz, A.; Wagner, E.F.; et al. Evidence for osteocyte regulation of bone homeostasis through RANKL expression. Nat. Med. 2011, 17, 1231–1234. [Google Scholar] [CrossRef]
- Ono, T.; Hayashi, M.; Sasaki, F.; Nakashima, T. RANKL biology: Bone metabolism, the immune system, and beyond. Inflamm. Regen. 2020, 40, 1–16. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Takayanagi, H. Osteoclast differentiation and activation. Clin. Calcium 2007, 17, 484–492. [Google Scholar] [PubMed]
- Hu, R.; Chen, L.; Chen, X.; Xie, Z.; Xia, C.; Chen, Y. Aloperine improves osteoporosis in ovariectomized mice by inhibiting RANKL-induced NF-κB, ERK and JNK approaches. Int. Immunopharmacol. 2021, 97, 107720. [Google Scholar] [CrossRef] [PubMed]
- Zhu, S.; Wang, J.; Wang, X.; Zhao, J. Protection against monocrotaline-induced pulmonary arterial hypertension and caveo-lin-1 downregulation by fluvastatin in rats. Mol. Med. Rep. 2018, 17, 3944–3950. [Google Scholar] [PubMed] [Green Version]
- Liu, A.; Philip, J.; Vinnakota, K.C.; Van den Bergh, F.; Tabima, D.M.; Hacker, T.; Beard, D.A.; Chesler, N.C. Estrogen maintains mitochondrial content and function in the right ventricle of rats with pulmonary hypertension. Physiol. Rep. 2017, 5, e13157. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Zhou, F.; Dong, J.; Dong, Q.; Luan, H.; Li, L.; Hao, Y. Therapeutic effects of aloperine on the pulmonary arterial hyper-tension. Farmacia 2019, 67, 691–701. [Google Scholar] [CrossRef]
- Moi, P.; Chan, K.; Asunis, I.; Cao, A.; Kan, Y.W. Isolation of NF-E2-related factor 2 (Nrf2), a NF-E2-like basic leucine zipper transcriptional activator that binds to the tandem NF-E2/AP1 repeat of the beta-globin locus control region. Proc. Natl. Acad. Sci. USA 1994, 91, 9926–9930. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Itoh, K.; Chiba, T.; Takahashi, S.; Ishii, T.; Igarashi, K.; Katoh, Y.; Oyake, T.; Hayashi, N.; Satoh, K.; Hatayama, I. An Nrf2/small Maf heterodimer mediates the induction of phase II detoxifying enzyme genes through antioxidant response elements. Biochem. Biophys. Res. Commun. 1997, 236, 313–322. [Google Scholar] [CrossRef] [PubMed]
- Venugopal, R.; Jaiswal, A.K. Nrf1 and Nrf2 positively and c-Fos and Fra1 negatively regulate the human antioxidant response element-mediated expression of NAD (P) H: Quinone oxidoreductase1 gene. Proc. Natl. Acad. Sci. USA 1996, 93, 14960–14965. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nguyen, T.; Sherratt, P.J.; Pickett, C.B. Regulatory mechanisms controlling gene expression mediated by the antioxidant re-sponse element. Annu. Rev. Pharmacol. Toxicol. 2003, 43, 233–260. [Google Scholar] [CrossRef]
- Ma, Q. Xenobiotic-Activated Receptors: From Transcription to Drug Metabolism to Disease. Chem. Res. Toxicol. 2008, 21, 1651–1671. [Google Scholar] [CrossRef]
- Kensler, T.W.; Wakabayashi, N.; Biswal, S. Cell survival responses to environmental stresses via the Keap1-Nrf2-ARE path-way. Annu. Rev. Pharmacol. Toxicol. 2007, 47, 89–116. [Google Scholar] [CrossRef] [PubMed]
- Talalay, P.; Dinkova-Kostova, A.T.; Holtzclaw, W.D. Importance of phase 2 gene regulation in protection against electrophile and reactive oxygen toxicity and carcinogenesis. Adv. Enzym. Regul. 2003, 43, 121–134. [Google Scholar] [CrossRef]
- Hayes, J.D.; McMahon, M.; Chowdhry, S.; Dinkova-Kostova, A.T. Cancer Chemoprevention Mechanisms Mediated Through the Keap1–Nrf2 Pathway. Antioxid. Redox Signal. 2010, 13, 1713–1748. [Google Scholar] [CrossRef] [PubMed]
- Chi, X.; Yao, W.; Xia, H.; Jin, Y.; Li, X.; Cai, J.; Hei, Z. Elevation of HO-1 Expression Mitigates Intestinal Ischemia-Reperfusion Injury and Restores Tight Junction Function in a Rat Liver Transplantation Model. Oxidative Med. Cell. Longev. 2015, 2015, 986075. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ahmed, S.M.U.; Luo, L.; Namani, A.; Wang, X.J.; Tang, X. Nrf2 signaling pathway: Pivotal roles in inflammation. Biochim. Biophys. Acta Mol. Basis Dis. 2017, 1863, 585–597. [Google Scholar] [CrossRef] [PubMed]
- Hayes, J.D.; Dinkova-Kostova, A.T. The Nrf2 regulatory network provides an interface between redox and intermediary me-tabolism. Trends Biochem. Sci. 2014, 39, 199–218. [Google Scholar] [CrossRef]
- Malhotra, D.; Portales-Casamar, E.; Singh, A.; Srivastava, S.; Arenillas, D.; Happel, C.; Shyr, C.; Wakabayashi, N.; Kensler, T.W.; Wasserman, W.W. Global mapping of binding sites for Nrf2 identifies novel targets in cell survival response through ChIP-Seq profiling and network analysis. Nucleic Acids Res. 2010, 38, 5718–5734. [Google Scholar] [CrossRef]
- Papp, D.; Lenti, K.; Modos, D.; Fazekas, D.; Dúl, Z.; Turei, D.; Földvári-Nagy, L.; Nussinov, R.; Csermely, P.; Korcsmáros, T. The NRF2-related interactome and regulome contain multifunctional proteins and fine-tuned autoregulatory loops. FEBS Lett. 2012, 586, 1795–1802. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gemenetzi, M.; Lotery, A.J. Epigenetics in age-related macular degeneration: New discoveries and future perspectives. Cell. Mol. Life Sci. 2020, 77, 807–818. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jonas, J.B.; Cheung, C.M.G.; Panda-Jonas, S. Updates on the Epidemiology of Age-Related Macular Degeneration. Asia-Pacific J. Ophthalmol. 2017, 6, 493–497. [Google Scholar] [CrossRef]
- Ambati, J.; Fowler, B.J. Mechanisms of Age-Related Macular Degeneration. Neuron 2012, 75, 26–39. [Google Scholar] [CrossRef] [Green Version]
- Lambros, M.L.; Plafker, S.M. Oxidative stress and the Nrf2 anti-oxidant transcription factor in age-related macular degenera-tion. Retin. Degener. Dis. 2016, 854, 67–72. [Google Scholar]
- Strauss, O. The Retinal Pigment Epithelium in Visual Function. Physiol. Rev. 2005, 85, 845–881. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ao, J.; Wood, J.P.; Chidlow, G.; Gillies, M.C.; Casson, R.J. Retinal pigment epithelium in the pathogenesis of age-related macular degeneration and photobiomodulation as a potential therapy? Clin. Exp. Ophthalmol. 2018, 46, 670–686. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Babizhayev, M.A.; Strokov, I.A.; Nosikov, V.V.; Savel’yeva, E.L.; Sitnikov, V.F.; Yegorov, Y.E.; Lankin, V.Z. The role of oxi-dative stress in diabetic neuropathy: Generation of free radical species in the glycation reaction and gene polymorphisms en-coding antioxidant enzymes to genetic susceptibility to diabetic neuropathy in population of type I diabetic patients. Cell Biochem. Biophys. 2015, 71, 1425–1443. [Google Scholar] [PubMed]
- Fang, X.; Zhang, C.; Zhang, C.; Cai, Y.; Yu, Z.; Huang, Z.; Li, W.; Zhang, W. Reactivation of Denervated Schwann Cells by Embryonic Spinal Cord Neurons to Promote Axon Regeneration and Remyelination. Stem Cells Int. 2019, 2019, 7378594. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jaeschke, H.; McGill, M.R.; Ramachandran, A. Oxidant stress, mitochondria, and cell death mechanisms in drug-induced liver injury: Lessons learned from acetaminophen hepatotoxicity. Drug Metab. Rev. 2012, 44, 88–106. [Google Scholar] [CrossRef] [Green Version]
- Xiong, R.; Shan, S.; Wang, X.; Zhang, X.; Yu, H.; Shi, H.; Wang, X. Aloperine attenuates carbon tetrachloride-induced mouse hepatic injury via Nrf2/HO-1 pathway. Trop. J. Pharm. Res. 2020, 19, 983–988. [Google Scholar] [CrossRef]
- Liu, L.; Shang, Y.; Li, M.; Han, X.; Wang, J.; Wang, J. Curcumin ameliorates asthmatic airway inflammation by activating nuclear factor-E2-related factor 2/haem oxygenase (HO)-1 signalling pathway. Clin. Exp. Pharmacol. Physiol. 2015, 42, 520–529. [Google Scholar] [CrossRef] [PubMed]
- Lowy, D.R.; Willumsen, B.M. Function and regulation of ras. Annu. Rev. Biochem. 1993, 62, 851–891. [Google Scholar] [CrossRef] [PubMed]
- Bos, J.L. ras oncogenes in human cancer: A review. Cancer Res. 1989, 49, 4682–4689. [Google Scholar] [PubMed]
- Campbell, S.L.; Khosravi-Far, R.; Rossman, K.L.; Clark, G.J.; Der, C.J. Increasing complexity of Ras signaling. Oncogene 1998, 17, 1395–1413. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Daub, H.; Weiss, F.U.; Wallasch, C.; Ullrich, A. Role of transactivation of the EGF receptor in signalling by G-protein-coupled receptors. Nature 1996, 379, 557–560. [Google Scholar] [CrossRef]
- Chung, E.; Kondo, M. Role of Ras/Raf/MEK/ERK signaling in physiological hematopoiesis and leukemia development. Immunol. Res. 2010, 49, 248–268. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Zhou, X.; Shen, H.; Wang, D.; Wang, Y. Phosphorylated ERK is a potential predictor of sensitivity to sorafenib when treating hepatocellular carcinoma: Evidence from an in vitrostudy. BMC Med. 2009, 7, 41. [Google Scholar] [CrossRef] [Green Version]
- Kim, D.; Kim, S.Y.; Lee, J.S.; Hong, Y.S.; Kim, J.E.; Kim, K.P.; Kim, J.; Jang, S.J.; Yoon, Y.-K.; Kim, T.W. Primary tumor loca-tion predicts poor clinical outcome with cetuximab in RAS wild-type metastatic colorectal cancer. BMC Gastroenterol. 2017, 17, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Zhang, S.; Chen, X.; Zheng, X.; Yao, Y.; Lu, G.; Zhou, J. Palbociclib, a selective CDK4/6 inhibitor, enhances the effect of selumetinib in RAS-driven non-small cell lung cancer. Cancer Lett. 2017, 408, 130–137. [Google Scholar] [CrossRef]
- Pan, Q.; Liu, R.; Banu, H.; Ma, L.; Li, H. Inhibition of isoprenylcysteine carboxylmethyltransferase sensitizes common chemo-therapies in cervical cancer via Ras-dependent pathway. Biomed. Pharmacother. 2018, 99, 169–175. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Ding, Y.; Wang, J.; Wu, Y. Identification of the key factors related to bladder cancer by lncRNA-miRNA-mRNA three-layer network. Front. Genet. 2020, 10, 1398. [Google Scholar] [CrossRef]
- Yuan, X.-Y.; Liu, W.; Zhang, P.; Wang, R.-Y.; Guo, J.-Y. Effects and mechanisms of aloperine on 2, 4-dinitrofluorobenzene-induced allergic contact dermatitis in BALB/c mice. Eur. J. Pharmacol. 2010, 629, 147–152. [Google Scholar] [CrossRef] [Green Version]
- Dang, Z.; Xie, H.; Zhu, L.; Zhang, Q.; Li, Z.; Huang, L.; Chen, C.-H. Structure Optimization of Aloperine Derivatives as HIV-1 Entry Inhibitors. ACS Med. Chem. Lett. 2017, 8, 1199–1203. [Google Scholar] [CrossRef]
- Lin, W.-C.; Lin, J.-Y. Five Bitter Compounds Display Different Anti-inflammatory Effects through Modulating Cytokine Secretion Using Mouse Primary Splenocytes in Vitro. J. Agric. Food Chem. 2010, 59, 184–192. [Google Scholar] [CrossRef] [PubMed]
- Song, X.L.; Zhang, Y.J.; Wang, X.F.; Zhang, W.J.; Wang, Z.; Zhang, F.; Zhang, Y.J.; Lu, J.H.; Mei, J.W.; Hu, Y.P. Casticin induces apoptosis and G0/G1 cell cycle arrest in gallbladder cancer cells. Cancer Cell Int. 2017, 17, 1–10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yin, W.; Han, J.; Zhang, Z.; Han, Z.; Wang, S. Aloperine protects mice against bleomycin-induced pulmonary fibrosis by at-tenuating fibroblast proliferation and differentiation. Sci. Rep. 2018, 8, 1–10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, X.; Lv, X.Q.; Tang, S.; Mei, L.; Li, Y.H.; Zhang, J.P.; Jiang, J.D.; Peng, Z.G.; Song, D.Q. Discovery and evolution of aloperine derivatives as a new family of HCV inhibitors with novel mechanism. Eur. J. Med. Chem. 2018, 143, 1053–1065. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.-S.; Kim, J.-E.; Lim, D.Y.; Huang, Z.; Chen, H.; Langfald, A.; Lubet, R.A.; Grubbs, C.J.; Dong, Z.; Bode, A.M. Naproxen Induces Cell-Cycle Arrest and Apoptosis in Human Urinary Bladder Cancer Cell Lines and Chemically Induced Cancers by Targeting PI3K. Cancer Prev. Res. 2014, 7, 236–245. [Google Scholar] [CrossRef] [Green Version]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2018. CA Cancer J. Clin. 2018, 68, 7–30. [Google Scholar] [CrossRef]
- van den Anker, J.; Reed, M.D.; Allegaert, K.; Kearns, G.L. Developmental changes in pharmacokinetics and pharmacodynamics. J. Clin. Pharmacol. 2018, 58, S10–S25. [Google Scholar] [CrossRef] [Green Version]
- Huang, S.; Zhang, Y.; Zhang, Y.; Liu, J.; Liu, Z.; Wang, X. Establishment of LC-MS/MS method for determination of aloperine in rat plasma and its application in preclinical pharmacokinetics. J. Chromatogr. B 2021, 1173, 122671. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tahir, M.; Ali, S.; Zhang, W.; Lv, B.; Qiu, W.; Wang, J. Aloperine: A Potent Modulator of Crucial Biological Mechanisms in Multiple Diseases. Biomedicines 2022, 10, 905. https://doi.org/10.3390/biomedicines10040905
Tahir M, Ali S, Zhang W, Lv B, Qiu W, Wang J. Aloperine: A Potent Modulator of Crucial Biological Mechanisms in Multiple Diseases. Biomedicines. 2022; 10(4):905. https://doi.org/10.3390/biomedicines10040905
Chicago/Turabian StyleTahir, Muhammad, Sakhawat Ali, Wenting Zhang, Boqiang Lv, Wenge Qiu, and Juan Wang. 2022. "Aloperine: A Potent Modulator of Crucial Biological Mechanisms in Multiple Diseases" Biomedicines 10, no. 4: 905. https://doi.org/10.3390/biomedicines10040905






