Cultured Autologous Corneal Epithelia for the Treatment of Unilateral Limbal Stem Cell Deficiency: A Case Series of 15 Patients
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Study Population
2.3. Baseline Evaluation
2.4. Histological Analysis
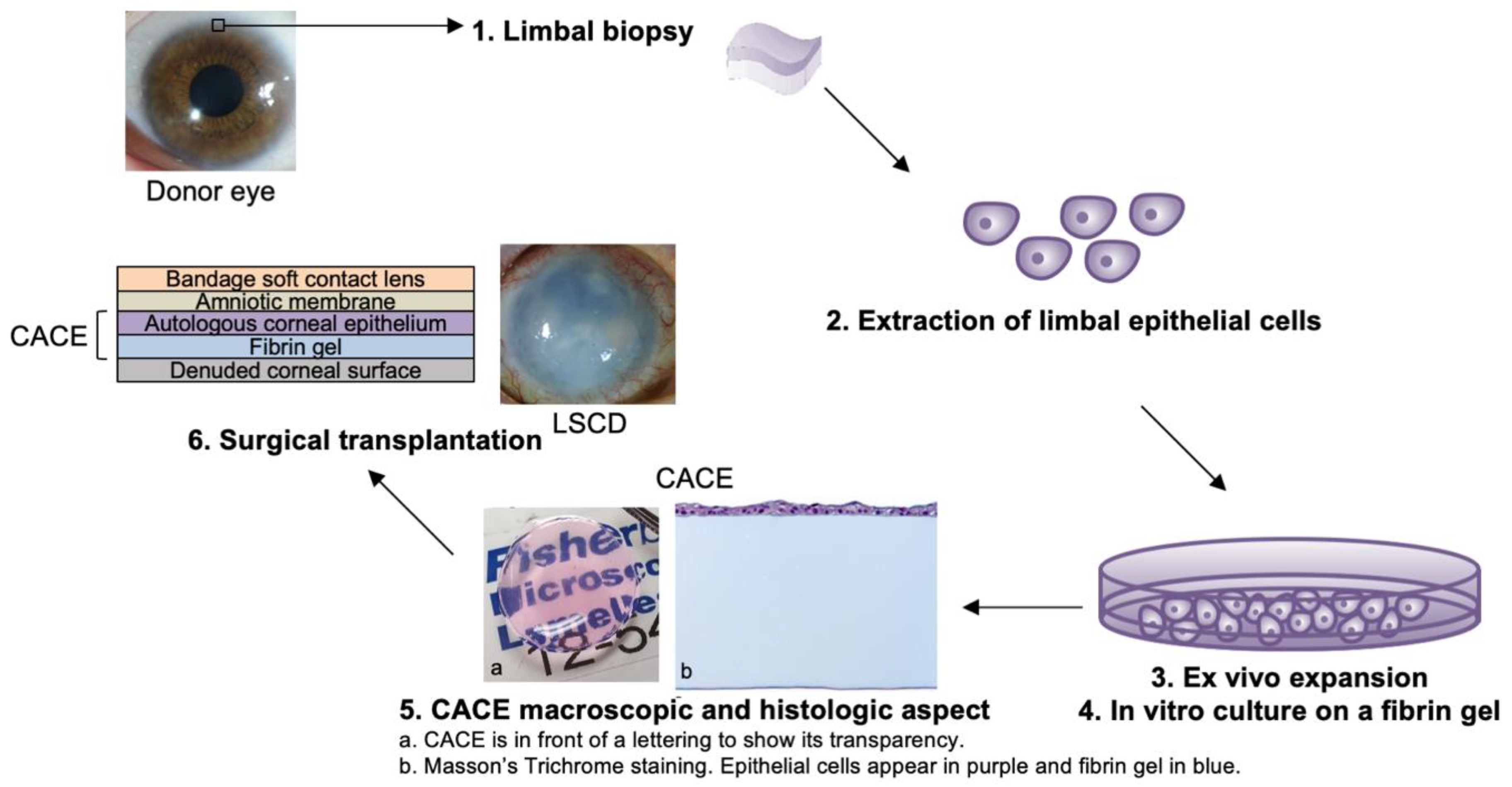
2.5. Cell Isolation and Culture
2.6. Surgical Procedure
2.7. Follow-Up and Outcome Assessment
2.8. Statistics
3. Results
3.1. Study Population
3.2. Adverse Events
3.3. Baseline Evaluation
3.4. Anatomical Outcomes
3.5. Functional Outcomes
3.6. Causes for Poor Visual Recovery
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- DelMonte, D.W.; Kim, T. Anatomy and physiology of the cornea. J. Cataract. Refract. Surg. 2011, 37, 588–598. [Google Scholar] [CrossRef]
- Thoft, R.A.; Friend, J. The X, Y, Z hypothesis of corneal epithelial maintenance. Investig. Ophthalmol. Vis. Sci. 1983, 24, 1442–1443. [Google Scholar]
- Castro-Munozledo, F. Review: Corneal epithelial stem cells, their niche and wound healing. Mol. Vis. 2013, 19, 1600–1613. [Google Scholar] [PubMed]
- Le, Q.; Xu, J.; Deng, S.X. The diagnosis of limbal stem cell deficiency. Ocul. Surf. 2018, 16, 58–69. [Google Scholar] [CrossRef] [PubMed]
- Dua, H.S.; Azuara-Blanco, A. Limbal stem cells of the corneal epithelium. Surv. Ophthalmol. 2000, 44, 415–425. [Google Scholar] [CrossRef]
- Sejpal, K.; Bakhtiari, P.; Deng, S.X. Presentation, diagnosis and management of limbal stem cell deficiency. Middle East Afr. J. Ophthalmol. 2013, 20, 5–10. [Google Scholar] [CrossRef] [PubMed]
- Kenyon, K.R.; Tseng, S.C. Limbal autograft transplantation for ocular surface disorders. Ophthalmology 1989, 96, 709–722; discussion 703–722. [Google Scholar] [CrossRef]
- Holland, E.J. Epithelial transplantation for the management of severe ocular surface disease. Trans. Am. Ophthalmol. Soc. 1996, 94, 677–743. [Google Scholar] [CrossRef]
- Lang, S.J.; Bohringer, D.; Geerling, G.; Reinhard, T. Long-term results of allogenic penetrating limbo-keratoplasty: 20 years of experience. Eye 2017, 31, 372–378. [Google Scholar] [CrossRef]
- Kolli, S.; Ahmad, S.; Lako, M.; Figueiredo, F. Successful clinical implementation of corneal epithelial stem cell therapy for treatment of unilateral limbal stem cell deficiency. Stem Cells 2010, 28, 597–610. [Google Scholar] [CrossRef]
- Pellegrini, G.; Traverso, C.E.; Franzi, A.T.; Zingirian, M.; Cancedda, R.; De Luca, M. Long-term restoration of damaged corneal surfaces with autologous cultivated corneal epithelium. Lancet 1997, 349, 990–993. [Google Scholar] [CrossRef]
- Rama, P.; Matuska, S.; Paganoni, G.; Spinelli, A.; De Luca, M.; Pellegrini, G. Limbal stem-cell therapy and long-term corneal regeneration. N. Engl. J. Med. 2010, 363, 147–155. [Google Scholar] [CrossRef] [PubMed]
- Prabhasawat, P.; Ekpo, P.; Uiprasertkul, M.; Chotikavanich, S.; Tesavibul, N. Efficacy of cultivated corneal epithelial stem cells for ocular surface reconstruction. Clin. Ophthalmol. 2012, 6, 1483–1492. [Google Scholar] [CrossRef] [PubMed]
- Scholz, S.L.; Thomasen, H.; Hestermann, K.; Dekowski, D.; Steuhl, K.P.; Meller, D. Long-term results of autologous transplantation of limbal epithelium cultivated ex vivo for limbal stem cell deficiency. Ophthalmol. Z. Dtsch. Ophthalmol. Ges. 2016, 113, 321–329. [Google Scholar] [CrossRef]
- Shortt, A.J.; Secker, G.A.; Rajan, M.S.; Meligonis, G.; Dart, J.K.; Tuft, S.J.; Daniels, J.T. Ex vivo expansion and transplantation of limbal epithelial stem cells. Ophthalmology 2008, 115, 1989–1997. [Google Scholar] [CrossRef] [PubMed]
- Mittal, V.; Jain, R.; Mittal, R. Ocular surface epithelialization pattern after simple limbal epithelial transplantation: An in vivo observational study. Cornea 2015, 34, 1227–1232. [Google Scholar] [CrossRef]
- Sangwan, V.S.; Basu, S.; MacNeil, S.; Balasubramanian, D. Simple limbal epithelial transplantation (SLET): A novel surgical technique for the treatment of unilateral limbal stem cell deficiency. Br. J. Ophthalmol. 2012, 96, 931–934. [Google Scholar] [CrossRef] [PubMed]
- Vazirani, J.; Ali, M.H.; Sharma, N.; Gupta, N.; Mittal, V.; Atallah, M.; Amescua, G.; Chowdhury, T.; Abdala-Figuerola, A.; Ramirez-Miranda, A.; et al. Autologous simple limbal epithelial transplantation for unilateral limbal stem cell deficiency: Multicentre results. Br. J. Ophthalmol. 2016, 100, 1416–1420. [Google Scholar] [CrossRef]
- Germain, L.; Auger, F.A.; Grandbois, E.; Guignard, R.; Giasson, M.; Boisjoly, H.; Guerin, S.L. Reconstructed human cornea produced in vitro by tissue engineering. Pathobiology 1999, 67, 140–147. [Google Scholar] [CrossRef]
- Talbot, M.; Carrier, P.; Giasson, C.J.; Deschambeault, A.; Guerin, S.L.; Auger, F.A.; Bazin, R.; Germain, L. Autologous transplantation of rabbit limbal epithelia cultured on fibrin gels for ocular surface reconstruction. Mol. Vis. 2006, 12, 65–75. [Google Scholar]
- Carrier, P.; Deschambeault, A.; Audet, C.; Talbot, M.; Gauvin, R.; Giasson, C.J.; Auger, F.A.; Guerin, S.L.; Germain, L. Impact of cell source on human cornea reconstructed by tissue engineering. Investig. Ophthalmol. Vis. Sci. 2009, 50, 2645–2652. [Google Scholar] [CrossRef] [PubMed]
- Carrier, P.; Deschambeault, A.; Talbot, M.; Giasson, C.J.; Auger, F.A.; Guerin, S.L.; Germain, L. Characterization of wound reepithelialization using a new human tissue-engineered corneal wound healing model. Investig. Ophthalmol. Vis. Sci. 2008, 49, 1376–1385. [Google Scholar] [CrossRef] [PubMed]
- Proulx, S.; d’Arc Uwamaliya, J.; Carrier, P.; Deschambeault, A.; Audet, C.; Giasson, C.J.; Guérin, S.L.; Auger, F.A.; Germain, L. Reconstruction of a human cornea by the self-assembly approach of tissue engineering using the three native cell types. Mol. Vis. 2010, 16, 2192–2201. [Google Scholar] [PubMed]
- Le-Bel, G.; Guerin, L.P.; Carrier, P.; Mouriaux, F.; Germain, L.; Guerin, S.L.; Bazin, R. Grafting of an autologous tissue-engineered human corneal epithelium to a patient with limbal stem cell deficiency (LSCD). Am. J. Ophthalmol. Case Rep. 2019, 15, 100532. [Google Scholar] [CrossRef] [PubMed]
- Guérin, L.P.; Le-Bel, G.; Desjardins, P.; Couture, C.; Gillard, E.; Boisselier, É.; Bazin, R.; Germain, L.; Guérin, S.L. The human tissue-engineered cornea (hTEC): Recent progress. Int. J. Mol. Sci. 2021, 22, 1291. [Google Scholar] [CrossRef]
- Shanbhag, S.S.; Nikpoor, N.; Donthineni, P.R.; Singh, V.; Chodosh, J.; Basu, S. Autologous limbal stem cell transplantation: A systematic review of clinical outcomes with different surgical techniques. Br. J. Ophthalmol. 2020, 104, 247–253. [Google Scholar] [CrossRef] [PubMed]
- Barabino, S.; Raghavan, A.; Loeffler, J.; Dana, R. Radiotherapy-induced ocular surface disease. Cornea 2005, 24, 909–914. [Google Scholar] [CrossRef] [PubMed]
- Shortt, A.J.; Secker, G.A.; Notara, M.D.; Limb, G.A.; Khaw, P.T.; Tuft, S.J.; Daniels, J.T. Transplantation of ex vivo cultured limbal epithelial stem cells: A review of techniques and clinical results. Surv. Ophthalmol. 2007, 52, 483–502. [Google Scholar] [CrossRef] [PubMed]
- Shortt, A.J.; Tuft, S.J.; Daniels, J.T. Ex vivo cultured limbal epithelial transplantation. A clinical perspective. Ocul. Surf. 2010, 8, 80–90. [Google Scholar] [CrossRef]
- Zhao, Y.; Ma, L. Systematic review and meta-analysis on transplantation of ex vivo cultivated limbal epithelial stem cell on amniotic membrane in limbal stem cell deficiency. Cornea 2015, 34, 592–600. [Google Scholar] [CrossRef]
- Haagdorens, M.; Van Acker, S.I.; Van Gerwen, V.; Ni Dhubhghaill, S.; Koppen, C.; Tassignon, M.J.; Zakaria, N. Limbal Stem Cell deficiency: Current treatment options and emerging therapies. Stem Cells Int. 2016, 2016, 9798374. [Google Scholar] [CrossRef] [PubMed]
- Biber, J.M.; Skeens, H.M.; Neff, K.D.; Holland, E.J. The cincinnati procedure: Technique and outcomes of combined living-related conjunctival limbal allografts and keratolimbal allografts in severe ocular surface failure. Cornea 2011, 30, 765–771. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, T.; Kinoshita, S. Ocular surface reconstruction using cultivated mucosal epithelial stem cells. Cornea 2003, 22, S75–S80. [Google Scholar] [CrossRef] [PubMed]
- Nishida, K.; Yamato, M.; Hayashida, Y.; Watanabe, K.; Yamamoto, K.; Adachi, E.; Nagai, S.; Kikuchi, A.; Maeda, N.; Watanabe, H.; et al. Corneal reconstruction with tissue-engineered cell sheets composed of autologous oral mucosal epithelium. N. Engl. J. Med. 2004, 351, 1187–1196. [Google Scholar] [CrossRef]
- Ricardo, J.R.; Cristovam, P.C.; Filho, P.A.; Farias, C.C.; de Araujo, A.L.; Loureiro, R.R.; Covre, J.L.; de Barros, J.N.; Barreiro, T.P.; dos Santos, M.S.; et al. Transplantation of conjunctival epithelial cells cultivated ex vivo in patients with total limbal stem cell deficiency. Cornea 2013, 32, 221–228. [Google Scholar] [CrossRef]
- Miotti, G.; Parodi, P.C.; Zeppieri, M. Stem cell therapy in ocular pathologies in the past 20 years. World J. Stem Cells 2021, 13, 366–385. [Google Scholar] [CrossRef]
- Susaimanickam, P.J.; Maddileti, S.; Pulimamidi, V.K.; Boyinpally, S.R.; Naik, R.R.; Naik, M.N.; Reddy, G.B.; Sangwan, V.S.; Mariappan, I. Generating minicorneal organoids from human induced pluripotent stem cells. Development 2017, 144, 2338–2351. [Google Scholar] [CrossRef]
- Reinshagen, H.; Auw-Haedrich, C.; Sorg, R.V.; Boehringer, D.; Eberwein, P.; Schwartzkopff, J.; Sundmacher, R.; Reinhard, T. Corneal surface reconstruction using adult mesenchymal stem cells in experimental limbal stem cell deficiency in rabbits. Acta Ophthalmol. 2011, 89, 741–748. [Google Scholar] [CrossRef]
- Garzón, I.; Martín-Piedra, M.A.; Alfonso-Rodríguez, C.; González-Andrades, M.; Carriel, V.; Martínez-Gómez, C.; Campos, A.; Alaminos, M. Generation of a biomimetic human artificial cornea model using Wharton’s jelly mesenchymal stem cells. Investig. Ophthalmol. Vis. Sci. 2014, 55, 4073–4083. [Google Scholar] [CrossRef]
- Hassan, N.T.; AbdelAziz, N.A. Oral mucosal stem cells, human immature dental pulp stem cells and hair follicle bulge stem cells as adult stem cells able to correct limbal stem cell deficiency. Curr. Stem Cell Res. Ther. 2018, 13, 356–361. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Ren, H.; Guo, X.; Hu, C.; Fu, J. Radiation-induced skin injury: Pathogenesis, treatment, and management. Aging 2020, 12, 23379–23393. [Google Scholar] [CrossRef] [PubMed]








| Patients | Age | Sex | Cause of LSCD | Baseline VA | Baseline Corneal Opacity Score | Baseline Central Vascularization Score | Follow-Up Time (Months) |
|---|---|---|---|---|---|---|---|
| 1 | 37 | M | Chemical burn | HM | 3 | 1 | 81.3 |
| 2 | 18 | F | Idiopathic | FC | 2 | 2 | 24.7 |
| 3 | 83 | F | Glaucoma medication | HM | 0.5 | 0 | 71.0 |
| 4 | 69 | M | Tumor excision | 6/15 | 1 | 1 | 74.8 |
| 5 | 38 | F | Idiopathic | FC | 0.5 | 0 | 62.4 |
| 6 | 80 | M | Glaucoma medication | 6/15+1 | 0 | 0 | 40.9 |
| 7 | 32 | F | Idiopathic | LP | 4 | 4 | 59.6 |
| 8 | 33 | M | Chemical burn | 6/9-2 | 0.5 | 2 | 56.8 |
| 9 | 64 | F | Radiotherapy for mucosa associated lymphoid tumor | HM | 2 | 2.5 | 52.9 |
| 10 | 70 | M | CIN (conjunctival intraepithelial neoplasia) Interferon and Mitomycin treatment | FC | 1 | 0 | 46.3 |
| 11 | 79 | F | CIN (conjunctival intraepithelial neoplasia) CO2 laser treatment | LP | 4 | 4 | 64.2 |
| 12 | 40 | F | Glaucoma medication and surgeries | FC | 2.5 | 0 | 55.5 |
| 13 | 57 | M | Mycosis fungoides | FC | 4 | 2 | 19.1 |
| 14 | 51 | F | Chemical burn | 6/15+2 | 1.5 | 0 | 16.8 |
| 15 | 68 | F | Multiple surgeries | 6/18+2 | 2 | 0 | 43.9 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| BCL related event | 1 | 1 | 4 | 1 | 2 | 1 | 2 | ||||||||
| Superficial punctate keratitis | 1 | ||||||||||||||
| Epithelial defect | 2 | 3 | 1 | 1 | 1 | 4 | 2 | 1 | 2 | 2 | 1 | ||||
| Epithelial irregularity | 1 | 3 | 1 | 2 | 1 | ||||||||||
| Eyelash trichiasis | 1 | 1 | |||||||||||||
| Ocular surface disorder | 1 | 2 | 3 | 1 | 1 | ||||||||||
| Amniotic membrane related event | 1 | 1 | 1 | ||||||||||||
| Corneal surface infection | 2 | 2 | 1 | 1 | |||||||||||
| Eyelid cellulitis | 1 | ||||||||||||||
| Corneal opacity | 1 | 1 | |||||||||||||
| Descemetocele without leak | 1 | ||||||||||||||
| Glaucoma | 1 | ||||||||||||||
| Post-operative hemorrhage | 1 | ||||||||||||||
| Pseudopterygium | 1 | ||||||||||||||
| Symblepharon recurrence | 3 | ||||||||||||||
| Fall | 1 | ||||||||||||||
| Shock: blow with forceps | 1 | ||||||||||||||
| Number of events | 4 | 2 | 6 | 6 | 5 | 4 | 7 | 4 | 7 | 6 | 4 | 2 | 8 | 1 | 6 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cataract surgery | 1 | 1 | 1 | 1 | |||||||||||
| Lamellar Keratoplasty | 1 | 1 | 1 | ||||||||||||
| Superficial Keratectomy | 1 | 1 | 1 | ||||||||||||
| DSAEK | 1 | ||||||||||||||
| Penetrating keratoplasty | 1 | 1 | |||||||||||||
| Laser iridotomy | 1 | ||||||||||||||
| Retina reattachment | 1 | ||||||||||||||
| Boston Keratoprosthesis | 11 | ||||||||||||||
| Repeat CACE | 1 | 1 | |||||||||||||
| Tarsorrhaphy | 1 | ||||||||||||||
| Lid taping | 1 | ||||||||||||||
| Number of procedures | 0 | 0 | 1 | 1 | 0 | 0 | 3 | 0 | 3 | 1 | 3 | 2 | 1 | 2 | 3 |
| Patients | Corneal Opacity 1 | Central Vascularization 1 | Anatomical Outcome 3 | BCVA 2 | Functional Outcome 4 | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Pre-op | 1y | 3y | 5y | Pre-Op | 1y | 3y | 5y | Pre-op | 1y post-Op | Longest FU | |||
| 1 | 3 | 3.5 | 3 | 0 | 1 | 2 | 2 | 0 | F | HM | HM | 6/45 | F |
| 2 | 2 | 0 | 2 | 0 | S | FC 30 cm | 6/24 | 6/9 | S | ||||
| 3a 6 | 0.5 | 0 | 1.5 | 1.5 | 0 | 0 | 0 | 0 | S | HM | 6/45 | 6/45 | S |
| 3b 6 | 1.5 | 0 | ND 5 | 0 | 0 | ND 5 | S | 6/45 | 6/120 | 6/120 | F | ||
| 4 | 1 | 0 | 0 | 0 | 1 | 0 | 0 | 0 | S | 6/15 | 6/6 | 6/7.5 | S |
| 5 | 0.5 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | S | CF 60 cm | 6/18 | 6/24 | S |
| 6 | 0 | 0 | 0 | 0 | 0 | 0 | S | 6/15+1 | 6/45 | 6/60 | F | ||
| 7 | 4 | 2 | 2 | 0 | 4 | 0 | 0 | 0 | S | LP | 6/120 | HM | PS |
| 8 | 0.5 | 0 | 0 | 0.5 | 2 | 0 | 0 | 0 | S | 6/9-2 | 6/18 | 6/9 | F |
| 9 | 2 | 1.5 | 2 | 0 | 2.5 | 0 | 0 | 0 | S | HM | CF 1 m | 6/120 | F |
| 10 | 1 | 0.5 | 0 | 0 | 0 | 0 | S | CF | 6/21 | 6/18 | S | ||
| 11 | 4 | 3.25 | 0 | 0 | 4 | 0 | 0 | 0 | S | LP | LP | LP | F |
| 12 | 2.5 | 1.75 | 0 | 0 | 0 | 0 | 0 | 0 | S | CF | 6/45 | 6/18 | S |
| 13 | 4 | 1 | 2 | 0 | S | CF | 6/60 | 6/60 | S | ||||
| 14 | 1.5 | 0 | 0 | 0 | S | 6/15+2 | 6/45 | 6/12 | F | ||||
| 15 | 2 | 0 | 0 | 0 | 0 | 0 | S | 6/18+2 | 6/30 | 6/30 | F | ||
| Mean Score 7 | 1.9 | 0.7 | 0.7 | 0.1 | 1.2 | 0.1 | 0.2 | 0.0 | |||||
| Mean Score Improvement 8 | 1.2 * | 1.2 * | 1.8 * | 1.1 * | 1.0 | 1.2 * | |||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guérin, L.-P.; Larouche, D.; Morcos, M.W.; Faucher, A.; Auger, F.A.; Knoppers, B.M.; Kyrillos, R.; Bazin, R.; Germain, L. Cultured Autologous Corneal Epithelia for the Treatment of Unilateral Limbal Stem Cell Deficiency: A Case Series of 15 Patients. Biomedicines 2022, 10, 1958. https://doi.org/10.3390/biomedicines10081958
Guérin L-P, Larouche D, Morcos MW, Faucher A, Auger FA, Knoppers BM, Kyrillos R, Bazin R, Germain L. Cultured Autologous Corneal Epithelia for the Treatment of Unilateral Limbal Stem Cell Deficiency: A Case Series of 15 Patients. Biomedicines. 2022; 10(8):1958. https://doi.org/10.3390/biomedicines10081958
Chicago/Turabian StyleGuérin, Louis-Philippe, Danielle Larouche, Mohib W. Morcos, Anne Faucher, François A. Auger, Bartha M. Knoppers, Ralph Kyrillos, Richard Bazin, and Lucie Germain. 2022. "Cultured Autologous Corneal Epithelia for the Treatment of Unilateral Limbal Stem Cell Deficiency: A Case Series of 15 Patients" Biomedicines 10, no. 8: 1958. https://doi.org/10.3390/biomedicines10081958
APA StyleGuérin, L.-P., Larouche, D., Morcos, M. W., Faucher, A., Auger, F. A., Knoppers, B. M., Kyrillos, R., Bazin, R., & Germain, L. (2022). Cultured Autologous Corneal Epithelia for the Treatment of Unilateral Limbal Stem Cell Deficiency: A Case Series of 15 Patients. Biomedicines, 10(8), 1958. https://doi.org/10.3390/biomedicines10081958







