Identification of Specific Plasma miRNAs as Potential Biomarkers for Major Depressive Disorder
Abstract
:1. Introduction
1.1. Major Depressive Disorder (MDD)
1.2. A Genetic Perspective on Major Depressive Disorder
1.3. Implication of miRNA in Major Depressive Disorder
1.4. Pathogenic Mechanisms of miRNA in Major Depressive Disorder
2. Materials and Methods
2.1. Study Sample and Design
2.2. Profiling of miRNAs
2.3. Statistical Analysis
2.4. Bioinformatic Analysis and Identification of the Relationships between miRNAs and Potential Pathways from MDD
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Chand, S.P.; Arif, H. Depression. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2024. [Google Scholar]
- Otte, C.; Gold, S.M.; Penninx, B.W.; Pariante, C.M.; Etkin, A.; Fava, M.; Mohr, D.C.; Schatzberg, A.F. Major Depressive Disorder. Nat. Rev. Dis. Primer 2016, 2, 16065. [Google Scholar] [CrossRef] [PubMed]
- Pitsillou, E.; Bresnehan, S.M.; Kagarakis, E.A.; Wijoyo, S.J.; Liang, J.; Hung, A.; Karagiannis, T.C. The Cellular and Molecular Basis of Major Depressive Disorder: Towards a Unified Model for Understanding Clinical Depression. Mol. Biol. Rep. 2020, 47, 753–770. [Google Scholar] [CrossRef] [PubMed]
- Bain, N.; Abdijadid, S. Major Depressive Disorder. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2024. Available online: https://www.ncbi.nlm.nih.gov/books/NBK559078/ (accessed on 20 June 2024).
- Ortega, M.A.; Alvarez-Mon, M.A.; García-Montero, C.; Fraile-Martinez, O.; Lahera, G.; Monserrat, J.; Muñoz-Merida, L.; Mora, F.; Rodríguez-Jiménez, R.; Fernandez-Rojo, S.; et al. MicroRNAs as Critical Biomarkers of Major Depressive Disorder: A Comprehensive Perspective. Biomedicines 2021, 9, 1659. [Google Scholar] [CrossRef] [PubMed]
- Kendler, K.S. The Origin of Our Modern Concept of Depression—The History of Melancholia From 1780–1880: A Review. JAMA Psychiatry 2020, 77, 863. [Google Scholar] [CrossRef] [PubMed]
- Paykel, E.S. Basic Concepts of Depression. Dialogues Clin. Neurosci. 2008, 10, 279–289. [Google Scholar] [CrossRef] [PubMed]
- Kawatake-Kuno, A.; Murai, T.; Uchida, S. The Molecular Basis of Depression: Implications of Sex-Related Differences in Epigenetic Regulation. Front. Mol. Neurosci. 2021, 14, 708004. [Google Scholar] [CrossRef] [PubMed]
- Ricci, V.; Di Salvo, G.; Maina, G. Lurasidone in First-Episode Psychosis with Predominant Depressive Symptoms: A Case Report. Int. Clin. Psychopharmacol. 2023, 38, 275–280. [Google Scholar] [CrossRef]
- Ventriglio, A.; Bhugra, D.; Sampogna, G.; Luciano, M.; De Berardis, D.; Sani, G.; Fiorillo, A. From Dysthymia to Treatment-Resistant Depression: Evolution of a Psychopathological Construct. Int. Rev. Psychiatry 2020, 32, 471–476. [Google Scholar] [CrossRef]
- Fries, G.R.; Zhang, W.; Benevenuto, D.; Quevedo, J. MicroRNAs in Major Depressive Disorder. In Reviews on Biomarker Studies in Psychiatric and Neurodegenerative Disorders; Advances in Experimental Medicine and Biology; Guest, P.C., Ed.; Springer International Publishing: Cham, Switzerland, 2019; Volume 1118, pp. 175–190. ISBN 978-3-030-05541-7. [Google Scholar]
- Bockmühl, Y.; Patchev, A.V.; Madejska, A.; Hoffmann, A.; Sousa, J.C.; Sousa, N.; Holsboer, F.; Almeida, O.F.X.; Spengler, D. Methylation at the CpG Island Shore Region Upregulates Nr3c1 Promoter Activity after Early-Life Stress. Epigenetics 2015, 10, 247–257. [Google Scholar] [CrossRef]
- Li, M.; Fu, X.; Xie, W.; Guo, W.; Li, B.; Cui, R.; Yang, W. Effect of Early Life Stress on the Epigenetic Profiles in Depression. Front. Cell Dev. Biol. 2020, 8, 867. [Google Scholar] [CrossRef]
- Almeida, M.I.; Reis, R.M.; Calin, G.A. MicroRNA History: Discovery, Recent Applications, and next Frontiers. Mutat. Res. Mol. Mech. Mutagen. 2011, 717, 1–8. [Google Scholar] [CrossRef]
- O’Brien, J.; Hayder, H.; Zayed, Y.; Peng, C. Overview of MicroRNA Biogenesis, Mechanisms of Actions, and Circulation. Front. Endocrinol. 2018, 9, 402. [Google Scholar] [CrossRef]
- Mazziotta, C.; Cervellera, C.F.; Lanzillotti, C.; Touzé, A.; Gaboriaud, P.; Tognon, M.; Martini, F.; Rotondo, J.C. MicroRNA Dysregulations in Merkel Cell Carcinoma: Molecular Mechanisms and Clinical Applications. J. Med. Virol. 2023, 95, e28375. [Google Scholar] [CrossRef] [PubMed]
- Reinhart, B.J.; Weinstein, E.G.; Rhoades, M.W.; Bartel, B.; Bartel, D.P. MicroRNAs in Plants. Genes Dev. 2002, 16, 1616–1626. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Peng, J.; Yi, C. The Epitranscriptome of Small Non-Coding RNAs. Non-Coding RNA Res. 2021, 6, 167–173. [Google Scholar] [CrossRef]
- Homorogan, C.; Nitusca, D.; Seclaman, E.; Enatescu, V.; Marian, C. Uncovering the Roles of MicroRNAs in Major Depressive Disorder: From Candidate Diagnostic Biomarkers to Treatment Response Indicators. Life 2021, 11, 1073. [Google Scholar] [CrossRef]
- Prodan-Bărbulescu, C.; Şeclăman, E.P.; Enătescu, V.; Faur, I.F.; Ghenciu, L.A.; Tuţac, P.; Paşca, P.; Grigoriţă, L.O. Evaluating the Connection between MicroRNAs and Long Non-Coding RNAs for the Establishment of the Major Depressive Disorder Diagnosis. Biomedicines 2024, 12, 516. [Google Scholar] [CrossRef] [PubMed]
- Turk, A.; Calin, G.A.; Kunej, T. MicroRNAs in Leukemias: A Clinically Annotated Compendium. Int. J. Mol. Sci. 2022, 23, 3469. [Google Scholar] [CrossRef] [PubMed]
- Thomas, K.T.; Gross, C.; Bassell, G.J. microRNAs Sculpt Neuronal Communication in a Tight Balance That Is Lost in Neurological Disease. Front. Mol. Neurosci. 2018, 11, 455. [Google Scholar] [CrossRef]
- Żurawek, D.; Turecki, G. The miRNome of Depression. Int. J. Mol. Sci. 2021, 22, 11312. [Google Scholar] [CrossRef]
- Kato, M.; Ogata, H.; Tahara, H.; Shimamoto, A.; Takekita, Y.; Koshikawa, Y.; Nishida, K.; Nonen, S.; Higasa, K.; Kinoshita, T. Multiple Pre-Treatment miRNAs Levels in Untreated Major Depressive Disorder Patients Predict Early Response to Antidepressants and Interact with Key Pathways. Int. J. Mol. Sci. 2022, 23, 3873. [Google Scholar] [CrossRef] [PubMed]
- Ding, R.; Su, D.; Zhao, Q.; Wang, Y.; Wang, J.-Y.; Lv, S.; Ji, X. The Role of microRNAs in Depression. Front. Pharmacol. 2023, 14, 1129186. [Google Scholar] [CrossRef] [PubMed]
- Su, B.; Cheng, S.; Wang, L.; Wang, B. MicroRNA-139-5p Acts as a Suppressor Gene for Depression by Targeting Nuclear Receptor Subfamily 3, Group C, Member 1. Bioengineered 2022, 13, 11856–11866. [Google Scholar] [CrossRef] [PubMed]
- Suento, W.J.; Kunisawa, K.; Wulaer, B.; Kosuge, A.; Iida, T.; Fujigaki, S.; Fujigaki, H.; Yamamoto, Y.; Tanra, A.J.; Saito, K.; et al. Prefrontal Cortex miR-874-3p Prevents Lipopolysaccharide-induced Depression-like Behavior through Inhibition of Indoleamine 2,3-dioxygenase 1 Expression in Mice. J. Neurochem. 2021, 157, 1963–1978. [Google Scholar] [CrossRef] [PubMed]
- Chen, E.Y.; Tan, C.M.; Kou, Y.; Duan, Q.; Wang, Z.; Meirelles, G.V.; Clark, N.R.; Ma’ayan, A. Enrichr: Interactive and collaborative HTML5 gene list enrichment analysis tool. BMC Bioinform. 2013, 14, 128. [Google Scholar] [CrossRef]
- Kuleshov, M.V.; Jones, M.R.; Rouillard, A.D.; Fernandez, N.F.; Duan, Q.; Wang, Z.; Koplev, S.; Jenkins, S.L.; Jagodnik, K.M.; Lachmann, A.; et al. Enrichr: A comprehensive gene set enrichment analysis web server 2016 update. Nucleic Acids Res. 2016, 44, W90–W97. [Google Scholar] [CrossRef]
- Xie, Z.; Bailey, A.; Kuleshov, M.V.; Clarke, D.J.B.; Evangelista, J.E.; Jenkins, S.L.; Lachmann, A.; Wojciechowicz, M.L.; Kropiwnicki, E.; Jagodnik, K.M.; et al. Gene set knowledge discovery with Enrichr. Curr. Protoc. 2021, 1, e90. [Google Scholar] [CrossRef]
- Mendes-Silva, A.P.; Pereira, K.S.; Tolentino-Araujo, G.T.; Nicolau, E.D.S.; Silva-Ferreira, C.M.; Teixeira, A.L.; Diniz, B.S. Shared Biologic Pathways Between Alzheimer Disease and Major Depression: A Systematic Review of MicroRNA Expression Studies. Am. J. Geriatr. Psychiatry 2016, 24, 903–912. [Google Scholar] [CrossRef]
- Maffioletti, E.; Cattaneo, A.; Rosso, G.; Maina, G.; Maj, C.; Gennarelli, M.; Tardito, D.; Bocchio-Chiavetto, L. Peripheral Whole Blood microRNA Alterations in Major Depression and Bipolar Disorder. J. Affect. Disord. 2016, 200, 250–258. [Google Scholar] [CrossRef]
- Eisch, A.J.; Petrik, D. Depression and Hippocampal Neurogenesis: A Road to Remission? Science 2012, 338, 72–75. [Google Scholar] [CrossRef]
- Liu, X.; Zhang, L.; Cheng, K.; Wang, X.; Ren, G.; Xie, P. Identification of Suitable Plasma-Based Reference Genes for miRNAome Analysis of Major Depressive Disorder. J. Affect. Disord. 2014, 163, 133–139. [Google Scholar] [CrossRef]
- Zhang, H.; Xue, X.; Pan, J.; Song, X.; Chang, X.; Mao, Q.; Lu, Y.; Zhao, H.; Wang, Y.; Chi, X.; et al. Integrated Analysis of the Chemical-Material Basis and Molecular Mechanisms for the Classic Herbal Formula of Lily Bulb and Rehmannia Decoction in Alleviating Depression. Chin. Med. 2021, 16, 107. [Google Scholar] [CrossRef]
- Braicu, V.; Stelian, P.; Fulger, L.; Verdes, G.; Brebu, D.; Duta, C.; Fizedean, C.; Ignuta, F.; Danila, A.I.; Cozma, G.V. Impact of Systemic Treatments on Outcomes and Quality of Life in Patients with RAS-Positive Stage IV Colorectal Cancer: A Systematic Review. Diseases 2024, 12, 79. [Google Scholar] [CrossRef] [PubMed]
- Faur, I.F.; Dobrescu, A.; Clim, I.A.; Pasca, P.; Prodan-Barbulescu, C.; Tarta, C.; Neamtu, A.-A.; Brebu, D.; Neamtu, C.; Rosu, M.; et al. The Predictive Role of Serum Lipid Levels, P53 and Ki-67, According to Molecular Subtypes in Breast Cancer: A Randomized Clinical Study. Int. J. Mol. Sci. 2024, 25, 3911. [Google Scholar] [CrossRef]
- Brebu, D.; Prodan-Bărbulescu, C.; Braicu, V.; Pașca, P.; Borcean, G.; Florea, S.; Bîrlog, C.; Dobrescu, A.; Cornianu, M.; Lazăr, F.; et al. Surgical Treatment of Lithiasis of the Main Pancreatic Duct: A Challenging Case and a Literature Review. Diseases 2024, 12, 86. [Google Scholar] [CrossRef]
- Braicu, V.; Fulger, L.; Nelluri, A.; Maganti, R.K.; Shetty, U.S.A.; Verdes, G.; Brebu, D.; Dumitru, C.; Toma, A.-O.; Rosca, O.; et al. Three-Year Analysis of the Rectal Cancer Care Trajectory after the COVID-19 Pandemic. Diseases 2023, 11, 181. [Google Scholar] [CrossRef] [PubMed]
- Ionut Faur, F.; Clim, A.; Nati, I.; Dobrescu, A. Lipid Profile an Important Risk Factor in Patients with Ovarian Tumors: A Meta-Analysis. Obstet. Gynecol. Reprod. Sci. 2022, 6, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Horgos, M.S.; Pop, O.L.; Sandor, M.; Borza, I.L.; Negrean, R.A.; Cote, A.; Neamtu, A.-A.; Grierosu, C.; Sachelarie, L.; Huniadi, A. Platelets Rich Plasma (PRP) Procedure in the Healing of Atonic Wounds. J. Clin. Med. 2023, 12, 3890. [Google Scholar] [CrossRef]
- Gecys, D.; Dambrauskiene, K.; Simonyte, S.; Patamsyte, V.; Vilkeviciute, A.; Musneckis, A.; Butkute-Sliuoziene, K.; Lesauskaite, V.; Zemaitis, L.; Usaite, D.; et al. Circulating Hsa-Let-7e-5p and Hsa-miR-125a-5p as Possible Biomarkers in the Diagnosis of Major Depression and Bipolar Disorders. Dis. Markers 2022, 2022, 3004338. [Google Scholar] [CrossRef] [PubMed]
- Xu, F.-L.; Wang, B.-J. Hsa-miR-874-3p Reduces Endogenous Expression of RGS4-1 Isoform In Vitro. Genes 2024, 15, 1057. [Google Scholar] [CrossRef]
- Gururajan, A.; Naughton, M.E.; Scott, K.A.; O’Connor, R.M.; Moloney, G.; Clarke, G.; Dowling, J.; Walsh, A.; Ismail, F.; Shorten, G.; et al. MicroRNAs as Biomarkers for Major Depression: A Role for Let-7b and Let-7c. Transl. Psychiatry 2016, 6, e862. [Google Scholar] [CrossRef] [PubMed]
- Roumans, S.; Sundquist, K.; Memon, A.A.; Hedelius, A.; Sundquist, J.; Wang, X. Association of Circulating Let-7b-5p with Major Depressive Disorder: A Nested Case-Control Study. BMC Psychiatry 2021, 21, 616. [Google Scholar] [CrossRef]
- Hussen, B.M.; Abdullah, S.R.; Rasul, M.F.; Jawhar, Z.H.; Faraj, G.S.H.; Kiani, A.; Taheri, M. MiRNA-93: A Novel Signature in Human Disorders and Drug Resistance. Cell Commun. Signal. 2023, 21, 79. [Google Scholar] [CrossRef] [PubMed]
- Camkurt, M.A.; Acar, Ş.; Coşkun, S.; Güneş, M.; Güneş, S.; Yılmaz, M.F.; Görür, A.; Tamer, L. Comparison of Plasma MicroRNA Levels in Drug Naive, First Episode Depressed Patients and Healthy Controls. J. Psychiatr. Res. 2015, 69, 67–71. [Google Scholar] [CrossRef] [PubMed]
- Smalheiser, N.R.; Lugli, G.; Zhang, H.; Rizavi, H.; Cook, E.H.; Dwivedi, Y. Expression of microRNAs and Other Small RNAs in Prefrontal Cortex in Schizophrenia, Bipolar Disorder and Depressed Subjects. PLoS ONE 2014, 9, e86469. [Google Scholar] [CrossRef]
- Zou, Z.-L.; Ye, Y.; Zhou, B.; Zhang, Y. Identification and Characterization of Noncoding RNAs-Associated Competing Endogenous RNA Networks in Major Depressive Disorder. World J. Psychiatry 2023, 13, 36–49. [Google Scholar] [CrossRef]
- Shi, Y.; Wang, Q.; Song, R.; Kong, Y.; Zhang, Z. Non-Coding RNAs in Depression: Promising Diagnostic and Therapeutic Biomarkers. eBioMedicine 2021, 71, 103569. [Google Scholar] [CrossRef]
- Serafini, G.; Trabucco, A.; Corsini, G.; Escelsior, A.; Amerio, A.; Aguglia, A.; Nasrallah, H.; Amore, M. The Potential of microRNAs as Putative Biomarkers in Major Depressive Disorder and Suicidal Behavior. Biomark. Neuropsychiatry 2021, 5, 100035. [Google Scholar] [CrossRef]
- Li, Y.-J.; Xu, M.; Gao, Z.-H.; Wang, Y.-Q.; Yue, Z.; Zhang, Y.-X.; Li, X.-X.; Zhang, C.; Xie, S.-Y.; Wang, P.-Y. Alterations of Serum Levels of BDNF-Related miRNAs in Patients with Depression. PLoS ONE 2013, 8, e63648. [Google Scholar] [CrossRef] [PubMed]
- Sun, S.; Yang, S.; Mao, Y.; Jia, X.; Zhang, Z. Reduced Cholesterol Is Associated with the Depressive-like Behavior in Rats through Modulation of the Brain 5-HT1A Receptor. Lipids Health Dis. 2015, 14, 22. [Google Scholar] [CrossRef]
- Xu, F. MicroRNA-132 May Play a Role in Coexistence of Depression and Cardiovascular Disease: A Hypothesis. Med. Sci. Monit. 2013, 19, 438–443. [Google Scholar] [CrossRef] [PubMed]
- Fang, Y.; Qiu, Q.; Zhang, S.; Sun, L.; Li, G.; Xiao, S.; Li, X. Changes in miRNA-132 and miR-124 Levels in Non-Treated and Citalopram-Treated Patients with Depression. J. Affect. Disord. 2018, 227, 745–751. [Google Scholar] [CrossRef] [PubMed]
- Tri, B.D.; Shashni, B.; Matsui, H.; Nagasaki, Y. Designing Poly(Gamma-Aminobutyric Acid)-Based Nanoparticles for the Treatment of Major Depressive Disorders. J. Control. Release 2023, 360, 110–121. [Google Scholar] [CrossRef]
- Prodan-Bărbulescu, C.; Watz, C.-G.; Moacă, E.-A.; Faur, A.-C.; Dehelean, C.-A.; Faur, F.I.; Grigoriţă, L.O.; Maghiari, A.L.; Tuţac, P.; Duţă, C.; et al. A Preliminary Report Regarding the Morphological Changes of Nano-Enabled Pharmaceutical Formulation on Human Lung Carcinoma Monolayer and 3D Bronchial Microtissue. Medicina 2024, 60, 208. [Google Scholar] [CrossRef]
- He, X.-L.; Yang, L.; Wang, Z.-J.; Huang, R.-Q.; Zhu, R.-R.; Cheng, L.-M. Solid Lipid Nanoparticles Loading with Curcumin and Dexanabinol to Treat Major Depressive Disorder. Neural Regen. Res. 2021, 16, 537. [Google Scholar] [CrossRef]
- Zhu, T.; Wang, H.; Gu, H.; Ju, L.; Wu, X.; Pan, W.; Zhao, M.; Yang, J.; Liu, P. Melanin-like Polydopamine Nanoparticles Mediating Anti-Inflammatory and Rescuing Synaptic Loss for Inflammatory Depression Therapy. J. Nanobiotechnol. 2023, 21, 52. [Google Scholar] [CrossRef]
- Cutler, A.J.; Mattingly, G.W.; Maletic, V. Understanding the Mechanism of Action and Clinical Effects of Neuroactive Steroids and GABAergic Compounds in Major Depressive Disorder. Transl. Psychiatry 2023, 13, 228. [Google Scholar] [CrossRef]
- Sforzini, L.; Worrell, C.; Kose, M.; Anderson, I.M.; Aouizerate, B.; Arolt, V.; Bauer, M.; Baune, B.T.; Blier, P.; Cleare, A.J.; et al. A Delphi-method-based consensus guideline for definition of treatment-resistant depression for clinical trials. Mol. Psychiatry 2022, 27, 1286–1299. [Google Scholar] [CrossRef] [PubMed]
- Baumgartner, A.; Campos-Barros, A.; Meinhold, H. Thyroid hormones and depressive illness: Implications for clinical and basic research. Acta Medica Austriaca 1992, 19 (Suppl. S1), 98–102. [Google Scholar] [PubMed]
- Markova, N.; Chernopiatko, A.; Schroeter, C.A.; Malin, D.; Kubatiev, A.; Bachurin, S.; Costa-Nunes, J.; Steinbusch, H.M.; Strekalova, T. Hippocampal gene expression of deiodinases 2 and 3 and effects of 3,5-diiodo-L-thyronine T2 in mouse depression paradigms. BioMed Res. Int. 2013, 2013, 565218. [Google Scholar] [CrossRef]
- Yang, R.; Zhu, F.; Yue, Y.; Lu, X.; Zhu, P.; Li, Z.; Zhao, X.; Yang, X.; Zhou, Y.; Du, X. Association between thyroid function and psychotic symptoms in adolescents with major depressive disorder: A large sample sized cross-sectional study in China. Heliyon 2023, 9, e16770. [Google Scholar] [CrossRef] [PubMed]
- Ogata, H.; Higasa, K.; Kageyama, Y.; Tahara, H.; Shimamoto, A.; Takekita, Y.; Koshikawa, Y.; Nonen, S.; Kato, T.; Kinoshita, T.; et al. Relationship between circulating mitochondrial DNA and microRNA in patients with major depression. J. Affect. Disord. 2023, 339, 538–546. [Google Scholar] [CrossRef]
- Glavan, D.; Gheorman, V.; Gresita, A.; Hermann, D.M.; Udristoiu, I.; Popa-Wagner, A. Identification of transcriptome alterations in the prefrontal cortex, hippocampus, amygdala and hippocampus of suicide victims. Sci. Rep. 2021, 11, 18853. [Google Scholar] [CrossRef] [PubMed]
- Pettai, K.; Milani, L.; Tammiste, A.; Võsa, U.; Kolde, R.; Eller, T.; Nutt, D.; Metspalu, A.; Maron, E. Whole-genome expression analysis reveals genes associated with treatment response to escitalopram in major depression. Eur. Neuropsychopharmacol. 2016, 26, 1475–1483. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.S.; Li, X.J.; Liu, C.Y.; Xu, Q.; Huang, J.Q.; Gu, S.; Chen, J.X. Effects of Histone Modification in Major Depressive Disorder. Curr. Neuropharmacol. 2022, 20, 1261–1277. [Google Scholar] [CrossRef] [PubMed]
- Gordon-Smith, K.; Jones, L.A.; Burge, S.M.; Munro, C.S.; Tavadia, S.; Craddock, N. The neuropsychiatric phenotype in Darier disease. Br. J. Dermatol. 2010, 163, 515–522. [Google Scholar] [CrossRef]
- Aljabali, A.A.A.; Alkaraki, A.K.; Gammoh, O.; Tambuwala, M.M.; Mishra, V.; Mishra, Y.; Hassan, S.S.; El-Tanani, M. Deciphering Depression: Epigenetic Mechanisms and Treatment Strategies. Biology 2024, 13, 638. [Google Scholar] [CrossRef]
- Fuchikami, M.; Yamamoto, S.; Morinobu, S.; Okada, S.; Yamawaki, Y.; Yamawaki, S. The potential use of histone deacetylase inhibitors in the treatment of depression. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2016, 64, 320–324. [Google Scholar] [CrossRef]
- Rosu, L.M.; Prodan-Bărbulescu, C.; Maghiari, A.L.; Bernad, E.S.; Bernad, R.L.; Iacob, R.; Stoicescu, E.R.; Borozan, F.; Ghenciu, L.A. Current Trends in Diagnosis and Treatment Approach of Diabetic Retinopathy during Pregnancy: A Narrative Review. Diagnostics 2024, 14, 369. [Google Scholar] [CrossRef]
- Babur, E.; Altunkaya, M.; Tufan, E.; Süer, C.; Dursun, N. Hyperthyroidism-Induced Upregulation of Neurodegeneration-Related Gene Expression in Metaplasticity-Induced Hippocampus. Neuroendocrinology 2024, 114, 400–410. [Google Scholar] [CrossRef]
- Cardinali, D.P.; Stern, J.E. Peripheral neuroendocrinology of the cervical autonomic nervous system. Braz. J. Med. Biol. Res. Rev. Bras. Pesqui. Medicas Biol. 1994, 27, 573–599. [Google Scholar]
- Redei, E.E.; Ciolino, J.D.; Wert, S.L.; Yang, A.; Kim, S.; Clark, C.; Zumpf, K.B.; Wisner, K.L. Pilot validation of blood-based biomarkers during pregnancy and postpartum in women with prior or current depression. Transl. Psychiatry 2021, 11, 68. [Google Scholar] [CrossRef] [PubMed]
- Cao, H.; Li, S.; Baranova, A.; Zhang, F. Shared Genetic Liability Between Major Depressive Disorder and Atopic Diseases. Front. Immunol. 2021, 12, 665160. [Google Scholar] [CrossRef]
- Verhoeven, W.M.; Tuinier, S.; Kuijpers, H.J.; Egger, J.I.; Brunner, H.G. Psychiatric profile in rubinstein-taybi syndrome. A review and case report. Psychopathology 2010, 43, 63–68. [Google Scholar] [CrossRef]
- Dall’Aglio, L.; Lewis, C.M.; Pain, O. Delineating the Genetic Component of Gene Expression in Major Depression. Biol. Psychiatry 2021, 89, 627–636. [Google Scholar] [CrossRef]
- Rijlaarsdam, J.; Cosin-Tomas, M.; Schellhas, L.; Abrishamcar, S.; Malmberg, A.; Neumann, A.; Felix, J.F.; Sunyer, J.; Gutzkow, K.B.; Grazuleviciene, R.; et al. DNA methylation and general psychopathology in childhood: An epigenome-wide meta-analysis from the PACE consortium. Mol. Psychiatry 2023, 28, 1128–1136. [Google Scholar] [CrossRef] [PubMed]
- Rana, T.; Behl, T.; Sehgal, A.; Sachdeva, M.; Mehta, V.; Sharma, N.; Singh, S.; Bungau, S. Exploring Sonic Hedgehog Cell Signaling in Neurogenesis: Its Potential Role in Depressive Behavior. Neurochem. Res. 2021, 46, 1589–1602. [Google Scholar] [CrossRef]
- Qi, D.; Chen, K. Bioinformatics Analysis of Potential Biomarkers and Pathway Identification for Major Depressive Disorder. Comput. Math. Methods Med. 2021, 2021, 3036741. [Google Scholar] [CrossRef]
- Zhang, Z.; Chen, G. A logical relationship for schizophrenia, bipolar, and major depressive disorder. Part 1: Evidence from chromosome 1 high density association screen. J. Comp. Neurol. 2020, 528, 2620–2635. [Google Scholar] [CrossRef]
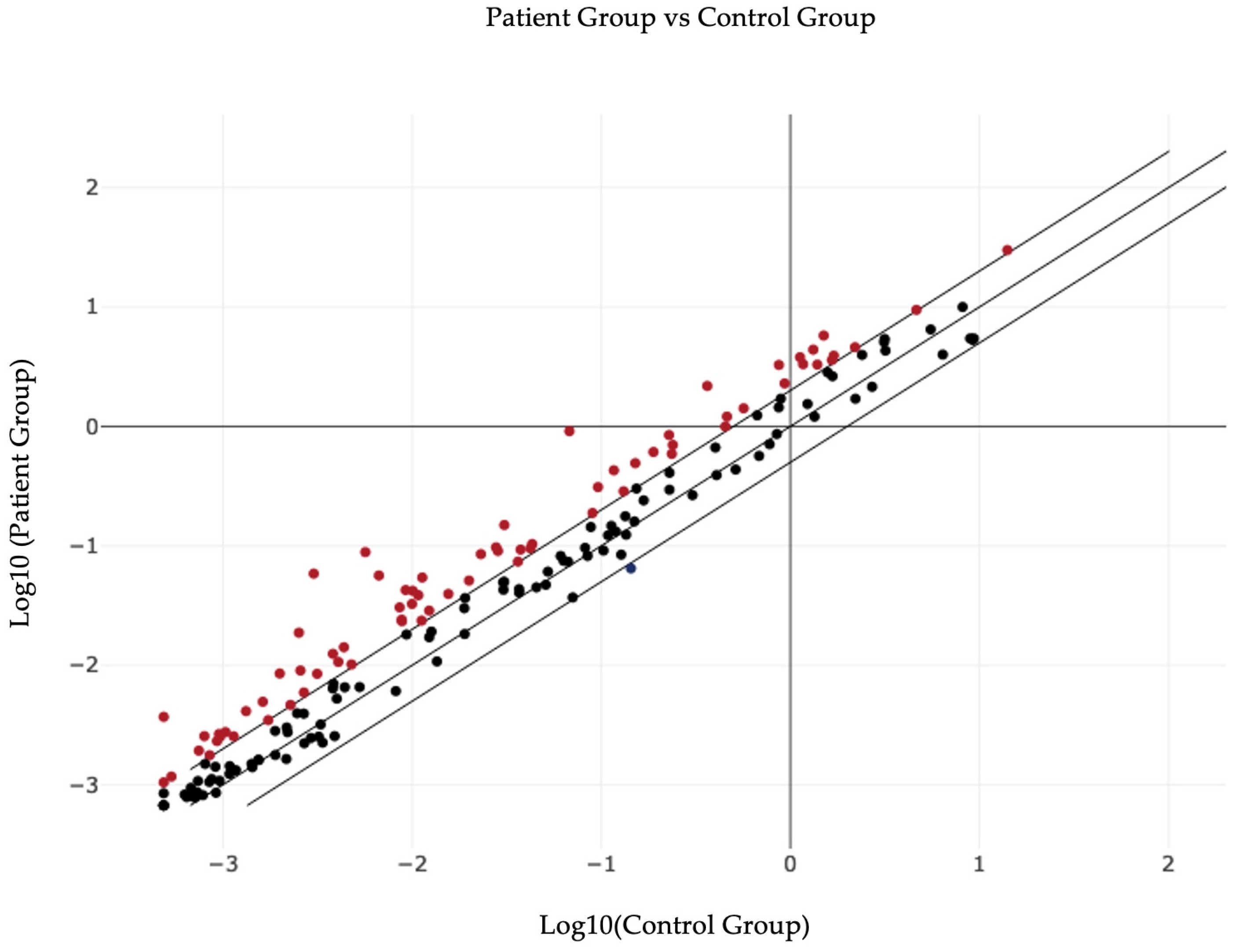


| Number | miRNA ID | Fold Regulation | p-Value |
|---|---|---|---|
| 1 | hsa-miR-874-3p | 19.4 | 0.013566 |
| 2 | hsa-miR-574-3p | 6.01 | 0.019013 |
| 3 | hsa-miR-451a | 1.86 | 0.019841 |
| 4 | hsa-miR-93-3p | 8.47 | 0.020517 |
| 5 | hsa-let-7d-5p | 13.45 | 0.021807 |
| 6 | hsa-miR-125a-5p | 2.59 | 0.022523 |
| 7 | hsa-miR-132-3p | 3.76 | 0.026436 |
| 8 | hsa-miR-141-3p | 3.19 | 0.033533 |
| 9 | hsa-miR-140-5p | 4.8 | 0.03412 |
| 10 | hsa-miR-376a-3p | 2.45 | 0.042657 |
| 11 | hsa-miR-423-3p | 2.1 | 0.04844 |
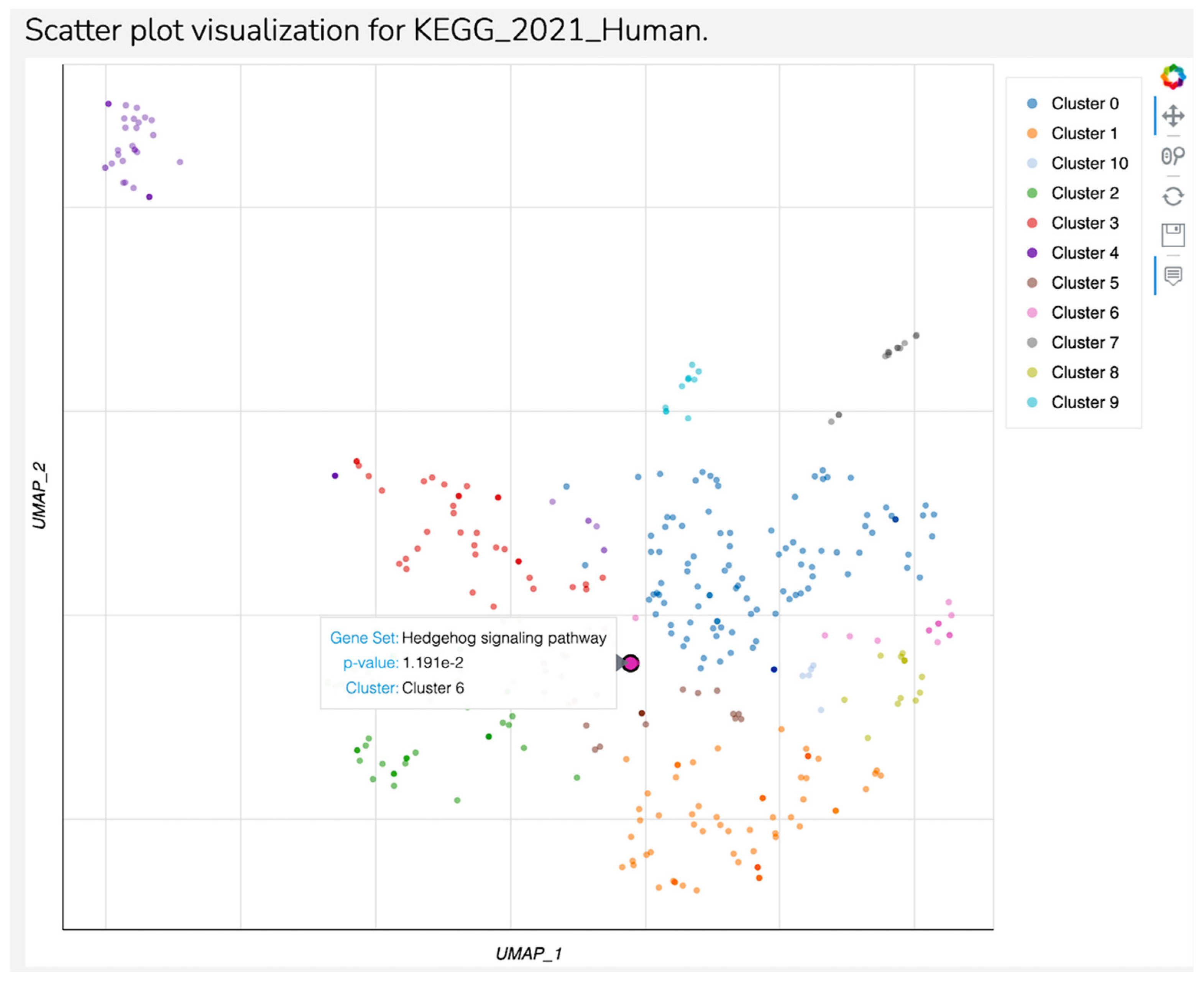
| Index | Name | p-Value | Adjusted p-Value | Odds Ratio | Combined Score |
|---|---|---|---|---|---|
| 1 | Thyroid hormone signaling pathway | 0.006023 | 0.1782 | 8.69 | 44.42 |
| 2 | Pathways of neurodegeneration | 0.01480 | 0.2182 | 3.70 | 15.58 |
| 3 | Arachidonic acid metabolism | 0.01492 | 0.2182 | 11.42 | 48.03 |
| 4 | Prolactin signaling pathway | 0.01935 | 0.2441 | 9.91 | 39.08 |
| 5 | Transcriptional misregulation in cancer | 0.02086 | 0.2441 | 5.41 | 20.92 |
| 6 | Alzheimer disease | 0.02611 | 0.2777 | 3.76 | 13.72 |
| 7 | Serotonergic synapse | 0.04666 | 0.3211 | 6.06 | 18.56 |
| Index | Name | p-Value | Adjusted p-Value | Odds Ratio | Combined Score |
|---|---|---|---|---|---|
| 1 | Hedgehog signaling pathway | 0.01151 | 0.5293 | 13.15 | 58.72 |
| 2 | Maturity-onset diabetes of the young | 0.07277 | 0.5293 | 13.98 | 36.63 |
| 3 | Cellular senescence | 0.07528 | 0.5293 | 4.59 | 11.87 |
| 4 | Axon guidance | 0.09785 | 0.5293 | 3.92 | 9.11 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Prodan-Bărbulescu, C.; Alin, C.D.; Faur, I.F.; Bujor, G.C.; Şeclăman, E.P.; Enătescu, V.; Dănilă, A.-I.; Dăescu, E.; Hajjar, R.; Ghenciu, L.A.; et al. Identification of Specific Plasma miRNAs as Potential Biomarkers for Major Depressive Disorder. Biomedicines 2024, 12, 2165. https://doi.org/10.3390/biomedicines12102165
Prodan-Bărbulescu C, Alin CD, Faur IF, Bujor GC, Şeclăman EP, Enătescu V, Dănilă A-I, Dăescu E, Hajjar R, Ghenciu LA, et al. Identification of Specific Plasma miRNAs as Potential Biomarkers for Major Depressive Disorder. Biomedicines. 2024; 12(10):2165. https://doi.org/10.3390/biomedicines12102165
Chicago/Turabian StyleProdan-Bărbulescu, Cătălin, Cristian Daniel Alin, Ionuţ Flaviu Faur, Georgeta Cristiana Bujor, Edward Paul Şeclăman, Virgil Enătescu, Alexandra-Ioana Dănilă, Ecaterina Dăescu, Rami Hajjar, Laura Andreea Ghenciu, and et al. 2024. "Identification of Specific Plasma miRNAs as Potential Biomarkers for Major Depressive Disorder" Biomedicines 12, no. 10: 2165. https://doi.org/10.3390/biomedicines12102165







