Contrasting Disease Progression, Microglia Reactivity, Tolerance, and Resistance to Toxoplasma gondii Infection in Two Mouse Strains
Abstract
1. Introduction
1.1. Host’s Immune Response, Tolerance, and Resistance
1.2. Sickness Behavior and Microglial Response
1.3. Microglia and Toxoplasmosis
2. Materials and Methods
2.1. Animal Handling, Anesthesia, and the Instillation of T. gondii into the Eye
2.2. Behavioral Testing: Open Field and Olfactory Discrimination
- -
- Open Field Test
- -
- Olfactory Discrimination Test
2.3. Histological and Immunohistochemical Procedures
2.4. Stereological Counting Procedures
- -
- Microscopy and Optical Fractionator
- -
- Area and objects of interest
2.5. Three-Dimensional Reconstructions
2.6. Stereological Sampling Approach and Hierarchical Cluster Analysis to Classify Microglia
3. Results
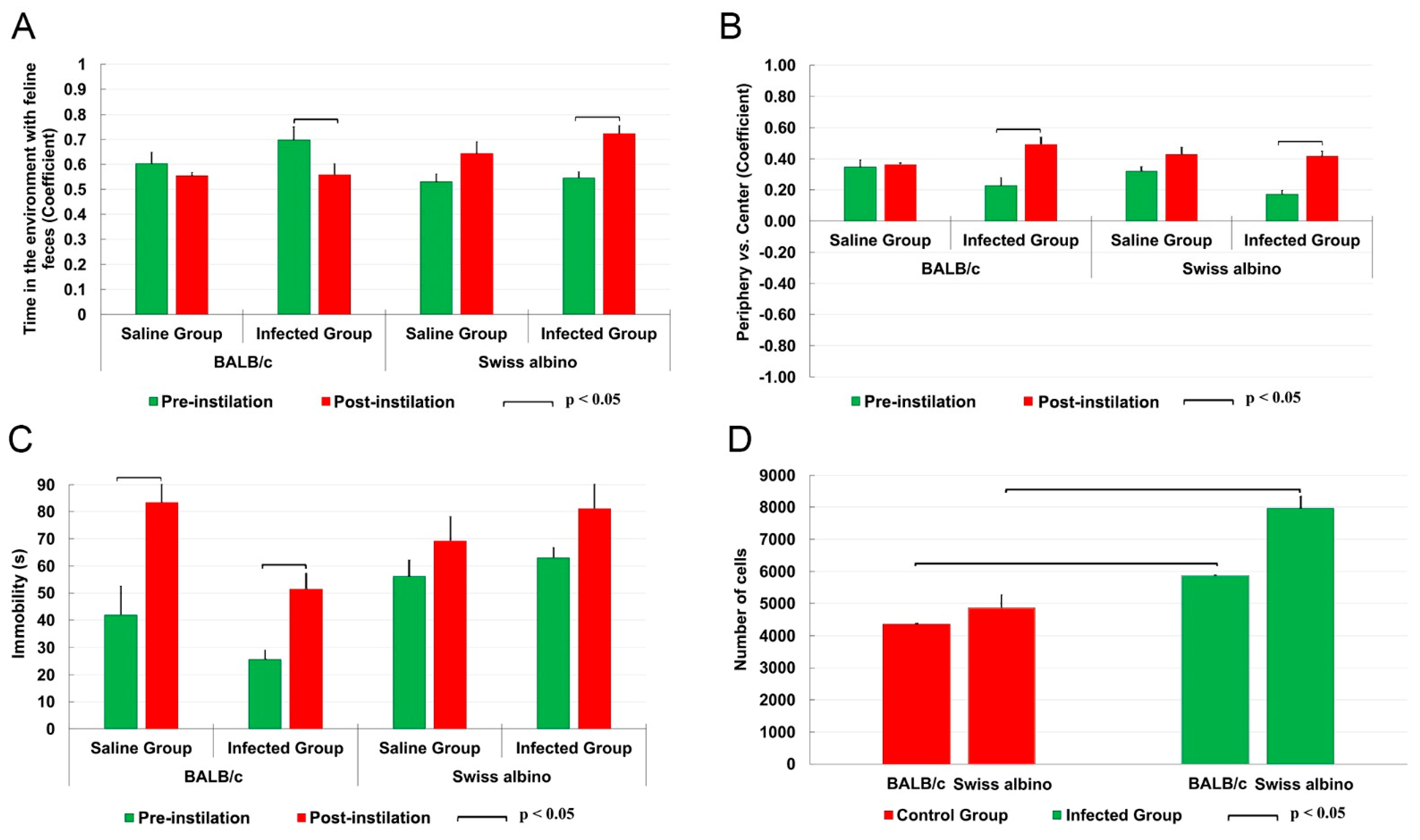
3.1. Comparative Microglial Numerical Changes in Control and T. gondii-Infected Mice
3.2. Behavioral Changes
- Mouse preference in the three-chamber olfactory test and in the open arena
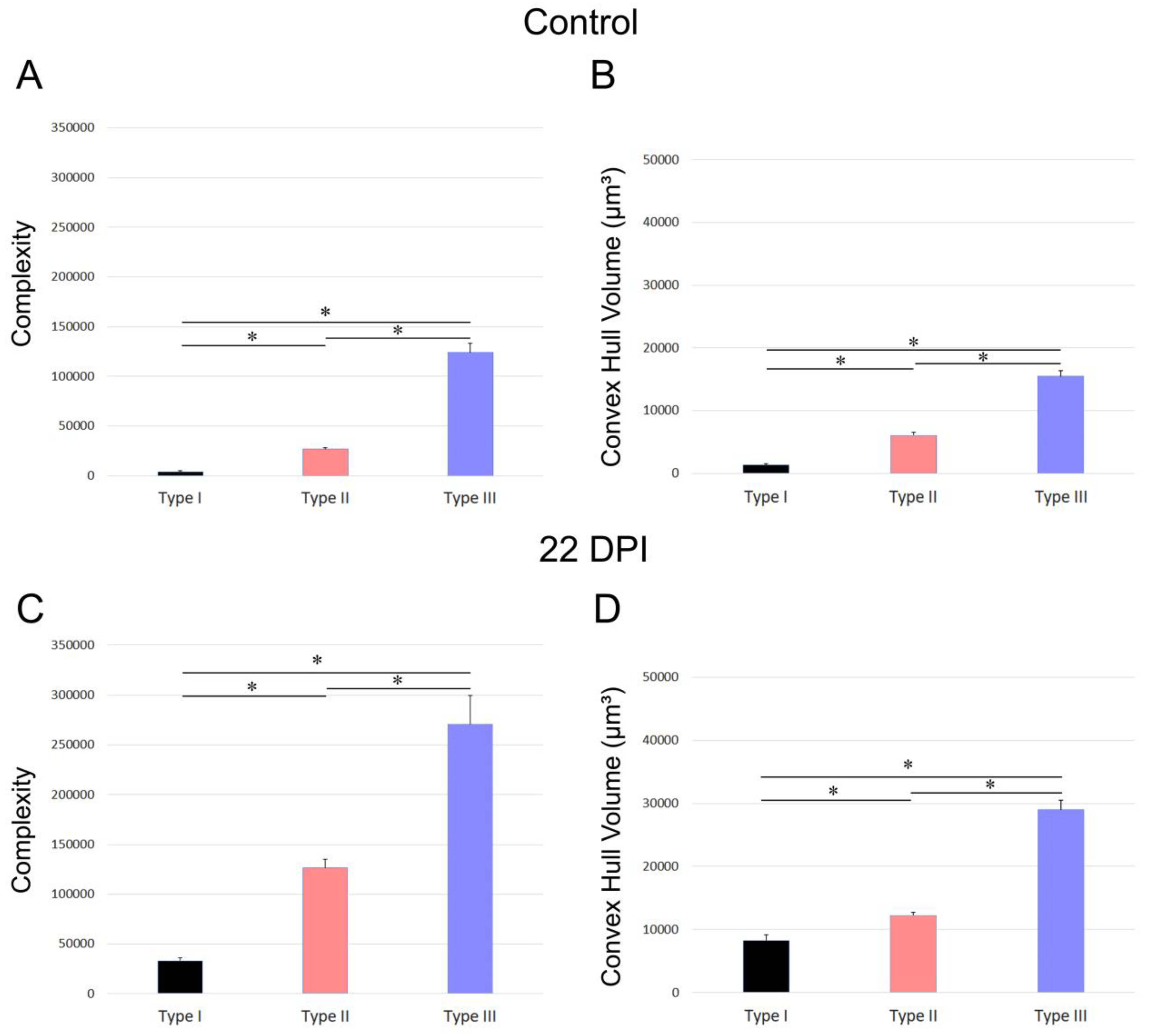
3.3. Microglial Morphotypes in T. gondii-Infected and Control Mice
4. Discussion
- -
- Technical limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Molan, A.; Nosaka, K.; Hunter, M.; Wang, W. Global status of Toxoplasma gondii infection: Systematic review and prevalence snapshots. Trop. Biomed. 2019, 36, 898–925. [Google Scholar] [PubMed]
- Ben-Harari, R.R.; Connolly, M.P. High burden and low awareness of toxoplasmosis in the United States. Postgrad. Med. 2019, 131, 103–108. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Han, Y.; Pan, Z.; Wang, H.; Yuan, M.; Lin, H. Seroprevalence of Toxoplasma gondii infection in blood donors in mainland China: A systematic review and meta-analysis. Parasite 2018, 25, 36. [Google Scholar] [CrossRef] [PubMed]
- Owusu-Dommey, A.; Pogreba-Brown, K.; Villa-Zapata, L. Seroprevalence of Toxoplasma gondii in the U.S.: Evidence from a representative cross-sectional survey. Parasitol. Int. 2020, 79, 102175. [Google Scholar] [PubMed]
- Aerts, R.; Mehra, V.; Groll, A.H.; Martino, R.; Lagrou, K.; Robin, C.; Perruccio, K.; Blijlevens, N.; Nucci, M.; Slavin, M.; et al. Guidelines for the management of Toxoplasma gondii infection and disease in patients with haematological malignancies and after haematopoietic stem-cell transplantation: Guidelines from the 9th European Conference on Infections in Leukaemia, 2022. Lancet Infect. Dis. 2023, 24, e291–e306. [Google Scholar] [CrossRef] [PubMed]
- Medzhitov, R.; Schneider, D.S.; Soares, M.P. Disease tolerance as a defense strategy. Science 2012, 335, 936–941. [Google Scholar] [CrossRef] [PubMed]
- Rivas, F.V.; Chervonsky, A.V.; Medzhitov, R. ART and immunology. Trends Immunol. 2014, 35, 451. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Gullett, J.M.; Tweedell, R.E.; Kanneganti, T.D. Innate immune inflammatory cell death: PANoptosis and PANoptosomes in host defense and disease. Eur. J. Immunol. 2023, 53, 2250235. [Google Scholar] [CrossRef] [PubMed]
- Ahmadpour, E.; Babaie, F.; Kazemi, T.; Mehrani Moghaddam, S.; Moghimi, A.; Hosseinzadeh, R.; Nissapatorn, V.; Pagheh, A.S. Overview of apoptosis, autophagy, and inflammatory processes in Toxoplasma gondii infected cells. Pathogens 2023, 12, 253. [Google Scholar] [CrossRef] [PubMed]
- Johnson, H.J.; Koshy, A.A. Latent Toxoplasmosis Effects on Rodents and Humans: How Much is Real and How Much is Media Hype? mBio 2020, 11, e02164-19. [Google Scholar] [CrossRef]
- Tong, W.H.; Pavey, C.; O’Handley, R.; Vyas, A. Behavioral biology of Toxoplasma gondii infection. Parasites Vectors 2021, 14, 77. [Google Scholar] [CrossRef]
- Suzuki, Y. The immune system utilizes two distinct effector mechanisms of T cells depending on two different life cycle stages of a single pathogen, Toxoplasma gondii, to control its cerebral infection. Parasitol. Int. 2020, 76, 102030. [Google Scholar] [CrossRef]
- Watson, G.F.; Davis, P.H. Systematic review and meta-analysis of variation in Toxoplasma gondii cyst burden in the murine model. Exp. Parasitol. 2019, 196, 55–62. [Google Scholar] [CrossRef]
- Melchor, S.J.; Ewald, S.E. Disease Tolerance in Toxoplasma Infection. Front. Cell Infect. Microbiol. 2019, 9, 185. [Google Scholar] [CrossRef]
- Melchor, S.J.; Saunders, C.M.; Sanders, I.; Hatter, J.A.; Byrnes, K.A.; Coutermarsh-Ott, S.; Ewald, S.E. IL-1R regulates disease tolerance and cachexia in Toxoplasma gondii infection. J. Immunol. 2020, 204, 3329–3338. [Google Scholar] [CrossRef]
- Silva, R.C.M.C.; Travassos, L.H.; Paiva, C.N.; Bozza, M.T. Heme oxygenase-1 in protozoan infections: A tale of resistance and disease tolerance. PLoS Pathog. 2020, 16, e1008599. [Google Scholar] [CrossRef]
- Melo, M.B.; Jensen, K.D.; Saeij, J.P. Toxoplasma gondii effectors are master regulators of the inflammatory response. Trends Parasitol. 2011, 27, 487–495. [Google Scholar] [CrossRef]
- Piotti, P.; Pierantoni, L.; Albertini, M.; Pirrone, F. Inflammation and Behavior Changes in Dogs and Cats. Vet. Clin. N. Am. Small Anim. Pract. 2024, 54, 1–16. [Google Scholar] [CrossRef]
- Kealy, J.; Murray, C.; Griffin, E.W.; Lopez-Rodriguez, A.B.; Healy, D.; Tortorelli, L.S.; Lowry, J.P.; Watne, L.O.; Cunningham, C. Acute Inflammation Alters Brain Energy Metabolism in Mice and Humans: Role in Suppressed Spontaneous Activity, Impaired Cognition, and Delirium. J. Neurosci. 2020, 40, 5681–5696. [Google Scholar] [CrossRef]
- Mahmoud, M.E.; Fereig, R.; Nishikawa, Y. Involvement of Host Defense Mechanisms against Toxoplasma gondii Infection in Anhedonic and Despair-Like Behaviors in Mice. Infect. Immun. 2017, 85, e00007-17. [Google Scholar] [CrossRef]
- Estato, V.; Stipursky, J.; Gomes, F.; Mergener, T.C.; Frazão-Teixeira, E.; Allodi, S.; Tibiriçá, E.; Barbosa, H.S.; Adesse, D. The Neurotropic Parasite Toxoplasma gondii Induces Sustained Neuroinflammation with Microvascular Dysfunction in Infected Mice. Am. J. Pathol. 2018, 188, 2674–2687. [Google Scholar] [CrossRef]
- Castaño Barrios, L.; Da Silva Pinheiro, A.P.; Gibaldi, D.; Silva, A.A.; Machado Rodrigues e Silva, P.; Roffê, E.; da Costa Santiago, H.; Tostes Gazzinelli, R.; Mineo, J.R.; Silva, N.M.; et al. Behavioral alterations in long-term Toxoplasma gondii infection of C57BL/6 mice are associated with neuroinflammation and disruption of the blood brain barrier. PLoS ONE 2021, 16, e0258199. [Google Scholar] [CrossRef]
- Dos Santos, S.E.; Medeiros, M.; Porfirio, J.; Tavares, W.; Pessôa, L.; Grinberg, L.; Leite, R.E.; Ferretti-Rebustini, R.E.; Suemoto, C.K.; Jacob Filho, W.; et al. Similar Microglial Cell Densities across Brain Structures and Mammalian Species: Implications for Brain Tissue Function. J. Neurosci. 2020, 40, 4622–4643. [Google Scholar] [CrossRef]
- Masuda, T.; Sankowski, R.; Staszewski, O.; Prinz, M. Microglia Heterogeneity in the Single-Cell Era. Cell Rep. 2020, 30, 1271–1281. [Google Scholar] [CrossRef]
- EHirbec, H.; Noristani, H.N.; Perrin, F.E. Microglia Responses in Acute and Chronic Neurological Diseases: What Microglia-Specific Transcriptomic Studies Taught (and did Not Teach) Us. Front. Aging Neurosci. 2017, 9, 227. [Google Scholar]
- Bollinger, J.L.; Collins, K.E.; Patel, R.; Wellman, C.L. Behavioral stress alters corticolimbic microglia in a sex- and brain region-specific manner. PLoS ONE 2017, 12, e0187631. [Google Scholar] [CrossRef]
- Sierra, A.; Paolicelli, R.C.; Kettenmann, H. Cien Años de Microglía: Milestones in a Century of Microglial Research. Trends Neurosci. 2019, 42, 778–792. [Google Scholar] [CrossRef]
- Nimmerjahn, A.; Kirchhoff, F.; Helmchen, F. Resting microglial cells are highly dynamic surveillants of brain parenchyma in vivo. Science 2005, 308, 1314–1318. [Google Scholar] [CrossRef]
- Hart, A.D.; Wyttenbach, A.; Perry, V.H.; Teeling, J.L. Age related changes in microglial phenotype vary between CNS regions: Grey versus white matter differences. Brain Behav. Immun. 2012, 26, 754–765. [Google Scholar] [CrossRef]
- Lawson, L.J.; Perry, V.H.; Dri, P.; Gordon, S. Heterogeneity in the distribution and morphology of microglia in the normal adult mouse brain. Neuroscience 1990, 39, 151–170. [Google Scholar] [CrossRef]
- Gorse, K.M.; Lafrenaye, A.D. The Importance of Inter-Species Variation in Traumatic Brain Injury-Induced Alterations of Microglial-Axonal Interactions. Front. Neurol. 2018, 9, 778. [Google Scholar] [CrossRef]
- Geirsdottir, L.; David, E.; Keren-Shaul, H.; Weiner, A.; Bohlen, S.C.; Neuber, J.; Balic, A.; Giladi, A.; Sheban, F.; Dutertre, C.A.; et al. Cross-Species Single-Cell Analysis Reveals Divergence of the Primate Microglia Program. Cell 2019, 179, 1609–1622.e16. [Google Scholar] [CrossRef]
- Wolf, S.A.; Boddeke, H.W.; Kettenmann, H. Microglia in Physiology and Disease. Annu. Rev. Physiol. 2017, 79, 619–643. [Google Scholar] [CrossRef]
- Stratoulias, V.; Venero, J.L.; Tremblay, M.; Joseph, B. Microglial subtypes: Diversity within the microglial community. EMBO J. 2019, 38, e101997. [Google Scholar] [CrossRef]
- Savage, J.C.; Carrier, M.; Tremblay, M. Morphology of Microglia Across Contexts of Health and Disease. Methods Mol. Biol. 2019, 2034, 13–26. [Google Scholar]
- Fernández-Arjona, M.D.M.; Grondona, J.M.; Fernández-Llebrez, P.; López-Ávalos, M.D. Microglial Morphometric Parameters Correlate With the Expression Level of IL-1β, and Allow Identifying Different Activated Morphotypes. Front. Cell Neurosci. 2019, 13, 472. [Google Scholar] [CrossRef]
- Fernández-Arjona, M.D.M.; Grondona, J.M.; Granados-Durán, P.; Fernández-Llebrez, P.; López-Ávalos, M.D. Microglia Morphological Categorization in a Rat Model of Neuroinflammation by Hierarchical Cluster and Principal Components Analysis. Front. Cell Neurosci. 2017, 11, 235. [Google Scholar] [CrossRef]
- Savage, J.C.; St-Pierre, M.K.; Hui, C.W.; Tremblay, M.E. Microglial Ultrastructure in the Hippocampus of a Lipopolysaccharide-Induced Sickness Mouse Model. Front. Neurosci. 2019, 13, 1340. [Google Scholar] [CrossRef]
- Paolicelli, R.C.; Sierra, A.; Stevens, B.; Tremblay, M.E.; Aguzzi, A.; Ajami, B.; Amit, I.; Audinat, E.; Bechmann, I.; Bennett, M.; et al. Microglia states and nomenclature: A field at its crossroads. Neuron 2022, 110, 3458–3483. [Google Scholar] [CrossRef]
- Johnson, S.K.; Johnson, P.T.J. Toxoplasmosis: Recent Advances in Understanding the Link Between Infection and Host Behavior. Annu. Rev. Anim. Biosci. 2021, 9, 249–264. [Google Scholar] [CrossRef]
- Sasai, M.; Pradipta, A.; Yamamoto, M. Host immune responses to Toxoplasma gondii. Int. Immunol. 2018, 30, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, Y.; Orellana, M.A.; Schreiber, R.D.; Remington, J.S. Interferon-gamma: The major mediator of resistance against Toxoplasma gondii. Science 1988, 240, 516–518. [Google Scholar] [CrossRef] [PubMed]
- Jeffers, V.; Tampaki, Z.; Kim, K.; Sullivan, W.J. A latent ability to persist: Differentiation in Toxoplasma gondii. Cell Mol. Life Sci. 2018, 75, 2355–2373. [Google Scholar] [CrossRef] [PubMed]
- Xiao, J.; Li, Y.; Prandovszky, E.; Kannan, G.; Viscidi, R.P.; Pletnikov, M.V.; Yolken, R.H. Behavioral Abnormalities in a Mouse Model of Chronic Toxoplasmosis Are Associated with MAG1 Antibody Levels and Cyst Burden. PLoS Negl. Trop. Dis. 2016, 10, e0004674. [Google Scholar] [CrossRef] [PubMed]
- Afonso, C.; Paixão, V.B.; Costa, R.M. Chronic Toxoplasma infection modifies the structure and the risk of host behavior. PLoS ONE 2012, 7, e32489. [Google Scholar] [CrossRef] [PubMed]
- Shen, B.; Yuan, Y.; Cheng, J.; Pan, M.; Xia, N.; Zhang, W.; Wang, Y.; Zhou, Y.; Zhao, J. Activation of chronic toxoplasmosis by transportation stress in a mouse model. Oncotarget 2016, 7, 87351–87360. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Sullivan, W.J.; Jeffers, V. Mechanisms of Toxoplasma gondii persistence and latency. FEMS Microbiol. Rev. 2012, 36, 717–733. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, Y.; Claflin, J.; Wang, X.; Lengi, A.; Kikuchi, T. Microglia and macrophages as innate producers of interferon-gamma in the brain following infection with Toxoplasma gondii. Int. J. Parasitol. 2005, 35, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Soares, G.L.D.S.; Leão, E.R.L.P.D.; Freitas, S.F.; Alves, R.M.C.; Tavares, N.D.P.; Costa, M.V.N.; Menezes, G.C.D.; Oliveira, J.H.P.D.; Guerreiro, L.C.F.; Assis, A.C.L.D.; et al. Behavioral and Neuropathological Changes after Toxoplasma gondii Ocular Conjunctival Infection in BALB/c Mice. Front. Cell Infect. Microbiol. 2022, 12, 812152. [Google Scholar] [CrossRef] [PubMed]
- de Sousa, A.A.; Dos Reis, R.R.; de Lima, C.M.; de Oliveira, M.A.; Fernandes, T.N.; Gomes, G.F.; Diniz, D.G.; Magalhaes, N.M.; Diniz, C.G.; Sosthenes, M.C.; et al. Three-dimensional morphometric analysis of microglial changes in a mouse model of virus encephalitis: Age and environmental influences. Eur. J. Neurosci. 2015, 42, 2036–2050. [Google Scholar] [CrossRef]
- Rosso, M.; Wirz, R.; Loretan, A.V.; Sutter, N.A.; da Cunha, C.T.P.; Jaric, I.; Würbel, H.; Voelkl, B. Reliability of common mouse behavioural tests of anxiety: A systematic review and meta-analysis on the effects of anxiolytics. Neurosci. Biobehav. Rev. 2022, 143, 104928. [Google Scholar] [CrossRef] [PubMed]
- Ennaceur, A. Tests of unconditioned anxiety—Pitfalls and disappointments. Physiol. Behav. 2014, 135, 55–71. [Google Scholar] [CrossRef] [PubMed]
- Shu, S.; Ju, G.; Fan, L. The glucose oxidase-DAB-nickel method in peroxidase histochemistry of the nervous system. Neurosci. Lett. 1988, 85, 169–171. [Google Scholar] [CrossRef] [PubMed]
- Saper, C.B.; Sawchenko, P.E. Magic peptides, magic antibodies: Guidelines for appropriate controls for immunohistochemistry. J. Comp. Neurol. 2003, 465, 161–163. [Google Scholar] [CrossRef] [PubMed]
- West, M.J. Design-based stereological methods for counting neurons. Prog. Brain Res. 2002, 135, 43–51. [Google Scholar] [PubMed]
- West, M.J.; Slomianka, L.; Gundersen, H.J. Unbiased stereological estimation of the total number of neurons in thesubdivisions of the rat hippocampus using the optical fractionator. Anat. Rec. 1991, 231, 482–497. [Google Scholar] [CrossRef] [PubMed]
- Gundersen, H.; Jensen, E. The efficiency of systematic sampling in stereology and its prediction. J. Microsc. 1987, 147, 229–263. [Google Scholar] [CrossRef] [PubMed]
- Glaser, E.M.; Wilson, P.D. The coefficient of error of optical fractionator population size estimates: A computer simulation comparing three estimators. J. Microsc. 1998, 192, 163. [Google Scholar] [CrossRef] [PubMed]
- West, M.J. Stereological methods for estimating the total number of neurons and synapses: Issues of precision and bias. Trends Neurosci. 1999, 22, 51–61. [Google Scholar] [CrossRef]
- Slomianka, L.; West, M. Estimators of the precision of stereological estimates: An example based on the CA1 pyramidal cell layer of rats. Neuroscience 2005, 136, 757–767. [Google Scholar] [CrossRef]
- Tan, Y.L.; Yuan, Y.; Tian, L. Microglial regional heterogeneity and its role in the brain. Mol. Psychiatry 2020, 25, 351–367. [Google Scholar] [CrossRef] [PubMed]
- Bernier, L.P.; Bohlen, C.J.; York, E.M.; Choi, H.B.; Kamyabi, A.; Dissing-Olesen, L.; Hefendehl, J.K.; Collins, H.Y.; Stevens, B.; Barres, B.A.; et al. Nanoscale Surveillance of the Brain by Microglia via cAMP-Regulated Filopodia. Cell Rep. 2019, 27, 2895–2908.e4. [Google Scholar] [CrossRef] [PubMed]
- Illes, P.; Rubini, P.; Ulrich, H.; Zhao, Y.; Tang, Y. Regulation of Microglial Functions by Purinergic Mechanisms in the Healthy and Diseased CNS. Cells 2020, 9, 1108. [Google Scholar] [CrossRef] [PubMed]
- da Silva Creão, L.S.; Neto, J.B.T.; de Lima, C.M.; Dos Reis, R.R.; de Sousa, A.A.; Dos Santos, Z.A.; Diniz, J.A.P.; Diniz, D.G.; Diniz, C.W.P. Microglial Metamorphosis in Three Dimensions in Virus Limbic Encephalitis: An Unbiased Pictorial Representation Based on a Stereological Sampling Approach of Surveillant and Reactive Microglia. Brain Sci. 2021, 11, 1009. [Google Scholar] [CrossRef] [PubMed]
- Carvalho-Paulo, D.; Bento Torres Neto, J.; Filho, C.S.; de Oliveira, T.C.G.; de Sousa, A.A.; Dos Reis, R.R.; Dos Santos, Z.A.; de Lima, C.M.; de Oliveira, M.A.; Said, N.M.; et al. Microglial morphology across distantly related species: Phylogenetic, environmental and age influences on microglia reactive and surveillance states. Front. Immunol. 2021, 12, 683026. [Google Scholar] [CrossRef] [PubMed]
- Torres-Platas, S.G.; Comeau, S.; Rachalski, A.; Bo, G.D.; Cruceanu, C.; Turecki, G.; Giros, B.; Mechawar, N. Morphometric characterization of microglial phenotypes in human cerebral cortex. J. Neuroinflamm. 2014, 11, 12. [Google Scholar] [CrossRef] [PubMed]
- Papageorgiou, I.E.; Fetani, A.F.; Lewen, A.; Heinemann, U.; Kann, O. Widespread activation of microglial cells in the hippocampus of chronic epileptic rats correlates only partially with neurodegeneration. Brain Struct. Funct. 2014, 220, 2423–2439. [Google Scholar] [CrossRef] [PubMed]
- Yamada, J.; Jinno, S. Novel objective classification of reactive microglia following hypoglossal axotomy using hierarchical cluster analysis. J. Comp. Neurol. 2013, 521, 1184–1201. [Google Scholar] [CrossRef]
- Schweitzer, L.; Renehan, W.E. The use of cluster analysis for cell typing. Brain Res. Brain Res. Protoc. 1997, 1, 100–108. [Google Scholar] [CrossRef]
- Diniz, D.G.; de Oliveira, M.A.; de Lima, C.M.; Fôro, C.A.R.; Sosthenes, M.C.K.; Bento-Torres, J.; da Costa Vasconcelos, P.F.; Anthony, D.C.; Diniz, C.W.P. Age, environment, object recognition and morphological diversity of GFAP-immunolabeled astrocytes. Behav. Brain Funct. 2016, 12, 28. [Google Scholar] [CrossRef]
- Kolb, H.; Fernandez, E.; Schouten, J.; Ahnelt, P.; Linberg, K.A.; Fisher, S.K. Are there three types of horizontal cell in the human retina? J. Comp. Neurol. 1994, 343, 370–386. [Google Scholar] [CrossRef] [PubMed]
- Råberg, L.; Graham, A.L.; Read, A.F. Decomposing health: Tolerance and resistance to parasites in animals. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2009, 364, 37–49. [Google Scholar] [CrossRef]
- Zhang, Y.; He, J.; Zheng, H.; Huang, S.; Lu, F. Association of TREM-1, IL-1β, IL-33/ST2, and TLR Expressions With the Pathogenesis of Ocular Toxoplasmosis in Mouse Models on Different Genetic Backgrounds. Front. Microbiol. 2019, 10, 2264. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Yuk, J.M.; Cha, G.H.; Lee, Y.H. Expression of cytokines and co-stimulatory molecules in the Toxoplasma gondii-infected dendritic cells of C57BL/6 and BALB/c mice. Parasites Hosts Dis. 2023, 61, 138–146. [Google Scholar] [CrossRef]
- Bergersen, K.V.; Barnes, A.; Worth, D.; David, C.; Wilson, E.H. Targeted Transcriptomic Analysis of C57BL/6 and BALB/c Mice During Progressive Chronic. Front. Cell Infect. Microbiol. 2021, 11, 645778. [Google Scholar] [CrossRef]
- Biswas, A.; Bruder, D.; Wolf, S.A.; Jeron, A.; Mack, M.; Heimesaat, M.M.; Dunay, I.R. Ly6C(high) monocytes control cerebral toxoplasmosis. J. Immunol. 2015, 194, 3223–3235. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Reyes, J.; Rovira-Diaz, E.; Fox, B.A.; Bzik, D.J.; Yap, G.S. Cutting Edge: CD36 Mediates Phagocyte Tropism and Avirulence of. J. Immunol. 2021, 207, 1507–1512. [Google Scholar] [CrossRef] [PubMed]
- Hwang, Y.S.; Shin, J.H.; Yang, J.P.; Jung, B.K.; Lee, S.H.; Shin, E.H. Characteristics of Infection Immunity Regulated by. Front. Immunol. 2018, 9, 158. [Google Scholar] [CrossRef] [PubMed]
- Blanchard, N.; Dunay, I.R.; Schlüter, D. Persistence of Toxoplasma gondii in the central nervous system: A fine-tuned balance between the parasite, the brain and the immune system. Parasite Immunol. 2015, 37, 150–158. [Google Scholar] [CrossRef]
- Bezerra, E.C.M.; Dos Santos, S.V.; Dos Santos, T.C.C.; de Andrade, H.F.; Meireles, L.R. Behavioral evaluation of BALB/c (Mus musculus) mice infected with genetically distinct strains of Toxoplasma gondii. Microb. Pathog. 2019, 126, 279–286. [Google Scholar] [CrossRef]
- Dubey, J.P.; Ferreira, L.R.; Martins, J.; McLeod, R. Oral oocyst-induced mouse model of toxoplasmosis: Effect of infection with Toxoplasma gondii strains of different genotypes, dose, and mouse strains (transgenic, out-bred, in-bred) on pathogenesis and mortality. Parasitology 2012, 139, 1–13. [Google Scholar] [CrossRef]
- Saeij, J.P.; Boyle, J.P.; Boothroyd, J.C. Differences among the three major strains of Toxoplasma gondii and their specific interactions with the infected host. Trends Parasitol. 2005, 21, 476–481. [Google Scholar] [CrossRef] [PubMed]
- Calero-Bernal, R.; Fernández-Escobar, M.; Katzer, F.; Su, C.; Ortega-Mora, L.M. Unifying Virulence Evaluation in Toxoplasma gondii: A Timely Task. Front. Cell Infect. Microbiol. 2022, 12, 868727. [Google Scholar] [CrossRef] [PubMed]
- Desmettre, T. Toxoplasmosis and behavioural changes. J. Fr. Ophtalmol. 2020, 43, e89–e93. [Google Scholar] [CrossRef]
- Pittman, K.J.; Aliota, M.T.; Knoll, L.J. Dual transcriptional profiling of mice and Toxoplasma gondii during acute and chronic infection. BMC Genom. 2014, 15, 806. [Google Scholar] [CrossRef] [PubMed]
- Radke, J.B.; Worth, D.; Hong, D.; Huang, S.; Sullivan, W.J., Jr.; Wilson, E.H.; White, M.W. Transcriptional repression by ApiAP2 factors is central to chronic toxoplasmosis. PLoS Pathog. 2018, 14, e1007035. [Google Scholar] [CrossRef]
- Bergersen, K.V.; Ramirez, A.D.; Kavvathas, B.; Mercer, F.; Wilson, E.H. Human neutrophil-like cells demonstrate antimicrobial responses to the chronic cyst form of Toxoplasma gondii. Parasite Immunol. 2023, 45, e13011. [Google Scholar] [CrossRef]
- Suzuki, Y.; Lutshumba, J.; Chen, K.C.; Abdelaziz, M.H.; Sa, Q.; Ochiai, E. IFN-γ production by brain-resident cells activates cerebral mRNA expression of a wide spectrum of molecules critical for both innate and T cell-mediated protective immunity to control reactivation of chronic infection with Toxoplasma gondii. Front. Cell Infect. Microbiol. 2023, 13, 1110508. [Google Scholar] [CrossRef]
- Carlo, C.N.; Stevens, C.F. Analysis of differential shrinkage in frozen brain sections and its implications for the use of guard zones in stereology. J. Comp. Neurol. 2011, 519, 2803–2810. [Google Scholar] [CrossRef]







| Animal | Total Number | Thickness (µm) | Scheaffer CE |
|---|---|---|---|
| (Uninfected Swiss albino) | |||
| Animal 6 | 4448 | 41.20 | 0.042 |
| Animal 7 | 4243 | 40.50 | 0.043 |
| Animal 12M | 6018 | 34.80 | 0.046 |
| Animal 6M | 5688 | 35.20 | 0.048 |
| Animal 7M | 3913 | 22.50 | 0.043 |
| Mean | 4862 | 34.84 | 0.043 |
| SD | 931.9 | 7.498 | 0.044 |
| CV | 0.191 | 0.215 | |
| CV² | 0.036 | 0.046 | |
| CE2 | 0.001 | 0.001 | |
| CE² / CV² | 0.027 | 0.021 | |
| CV²—CE² | 0.035 | 0.045 | |
| CVB² (%) | 97.22 | 97.82 | |
| (12 DPI Infected Swiss albino) | |||
| Animal 18 | 6392 | 34.80 | 0.032 |
| Animal 22 | 7355 | 41.00 | 0.033 |
| Animal 37 | 8793 | 51.50 | 0.033 |
| Animal 41 | 8640 | 37.40 | 0.029 |
| Animal 42 | 8347 | 38.10 | 0.029 |
| Animal 43 | 8229 | 51.00 | 0.037 |
| Mean | 7959 | 42.30 | 0.032 |
| SD | 916.8 | ||
| CV | 0.115 | 0.170 | |
| CV2 | 0.013 | 0.020 | |
| CE2 | 0.001 | 0.001 | |
| CE2/CV2 | 0.076 | 0.050 | |
| CV2−CE2 | 0.012 | 0.019 | |
| CVB2 (%) | 92.30 | 95.00 | |
| Animal | Thickness (µm) | No. of Sections | No. of Probes | Probe (µm) | Grid (µm) | Dissector Height (µm) | Interval |
|---|---|---|---|---|---|---|---|
| Control | |||||||
| Animal 6 | 41.2 | 6 | 721 | 50 × 50 | 70 × 70 | 15 | 1/3 |
| Animal 7 | 40.5 | 6 | 706 | 50 × 50 | 70 × 70 | 15 | 1/3 |
| Animal 12M | 34.8 | 5 | 492 | 50 × 50 | 70 × 70 | 15 | 1/5 |
| Animal 6M | 35.2 | 5 | 438 | 50 × 50 | 70 × 70 | 15 | 1/5 |
| Animal 7M | 22.5 | 5 | 420 | 50 × 50 | 70 × 70 | 15 | 1/5 |
| 12 DPI | |||||||
| Animal 18 | 34.8 | 5 | 638 | 50 × 50 | 70 × 70 | 15 | 1/3 |
| Animal 22 | 41.0 | 6 | 810 | 50 × 50 | 70 × 70 | 15 | 1/3 |
| Animal 37 | 51.5 | 6 | 797 | 50 × 50 | 70 × 70 | 15 | 1/3 |
| Animal 41 | 37.4 | 6 | 907 | 50 × 50 | 70 × 70 | 15 | 1/3 |
| Animal 42 | 38.1 | 5 | 808 | 50 × 50 | 70 × 70 | 15 | 1/3 |
| Animal 43 | 51.0 | 6 | 661 | 50 × 50 | 70 × 70 | 15 | 1/3 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Diniz, D.G.; de Oliveira, J.H.P.; Guerreiro, L.C.F.; de Menezes, G.C.; de Assis, A.C.L.; Duarte, T.Q.; dos Santos, I.B.D.; Maciel, F.D.; Soares, G.L.d.S.; Araújo, S.C.; et al. Contrasting Disease Progression, Microglia Reactivity, Tolerance, and Resistance to Toxoplasma gondii Infection in Two Mouse Strains. Biomedicines 2024, 12, 1420. https://doi.org/10.3390/biomedicines12071420
Diniz DG, de Oliveira JHP, Guerreiro LCF, de Menezes GC, de Assis ACL, Duarte TQ, dos Santos IBD, Maciel FD, Soares GLdS, Araújo SC, et al. Contrasting Disease Progression, Microglia Reactivity, Tolerance, and Resistance to Toxoplasma gondii Infection in Two Mouse Strains. Biomedicines. 2024; 12(7):1420. https://doi.org/10.3390/biomedicines12071420
Chicago/Turabian StyleDiniz, Daniel G., Jhonnathan H. P. de Oliveira, Luma C. F. Guerreiro, Gabriel C. de Menezes, Alexa C. L. de Assis, Tainá Q. Duarte, Izabelly B. D. dos Santos, Flávia D. Maciel, Gabrielly L. da S. Soares, Sanderson C. Araújo, and et al. 2024. "Contrasting Disease Progression, Microglia Reactivity, Tolerance, and Resistance to Toxoplasma gondii Infection in Two Mouse Strains" Biomedicines 12, no. 7: 1420. https://doi.org/10.3390/biomedicines12071420
APA StyleDiniz, D. G., de Oliveira, J. H. P., Guerreiro, L. C. F., de Menezes, G. C., de Assis, A. C. L., Duarte, T. Q., dos Santos, I. B. D., Maciel, F. D., Soares, G. L. d. S., Araújo, S. C., Franco, F. T. d. C., do Carmo, E. L., Morais, R. d. A. B., de Lima, C. M., Brites, D., Anthony, D. C., Diniz, J. A. P., & Diniz, C. W. P. (2024). Contrasting Disease Progression, Microglia Reactivity, Tolerance, and Resistance to Toxoplasma gondii Infection in Two Mouse Strains. Biomedicines, 12(7), 1420. https://doi.org/10.3390/biomedicines12071420








