Characteristics of Mucosa-Associated Microbiota in Ulcerative Colitis Patients with 5-Aminosalicylic Acid Intolerance
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics
2.2. Patients and Sample Collection
2.3. Definition of 5-ASA Intolerance
2.4. DNA Extraction, PCR Amplification, Library Preparation, and 16SrRNA Sequencing
2.5. Bioinformatics Analysis
2.6. Alpha-Diversity
2.7. Statistical Analysis
3. Results
3.1. Patient Background
3.2. Analysis of Mucosa Samples
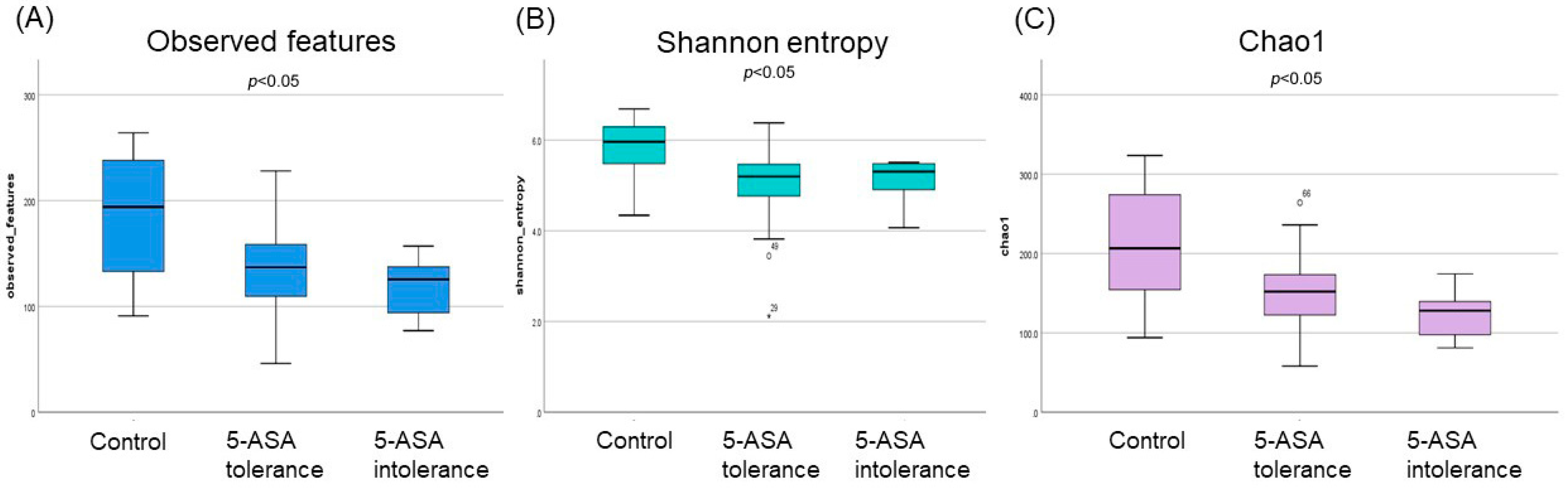
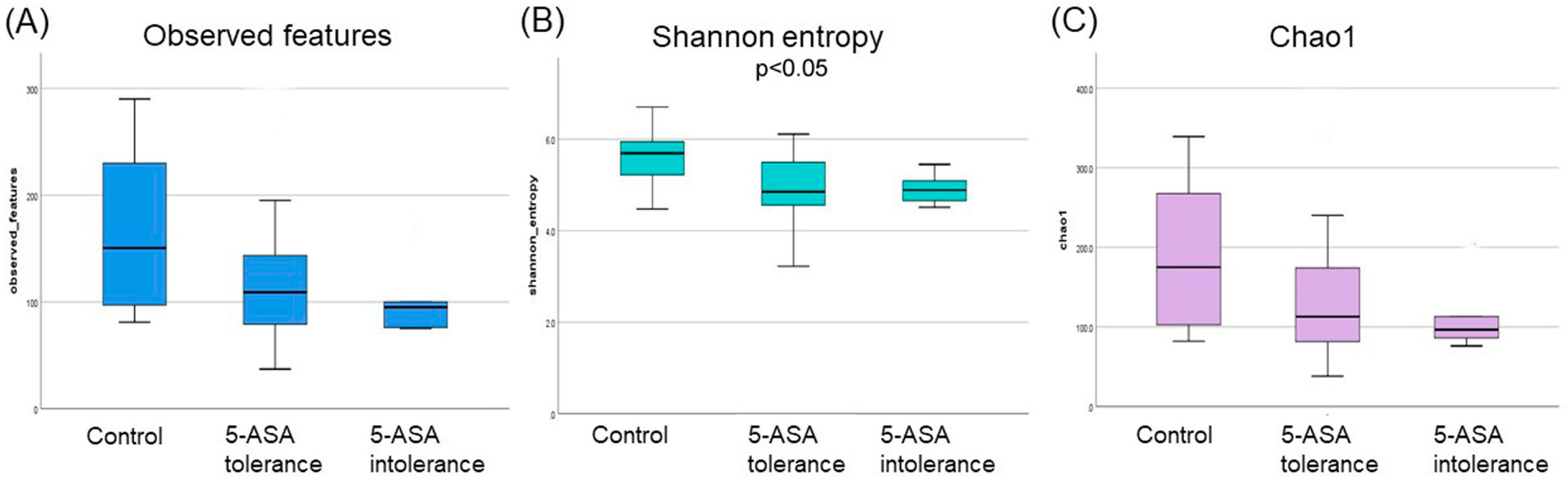
3.2.1. The α-Diversity Indices among the Three Groups Using Ileum Mucus
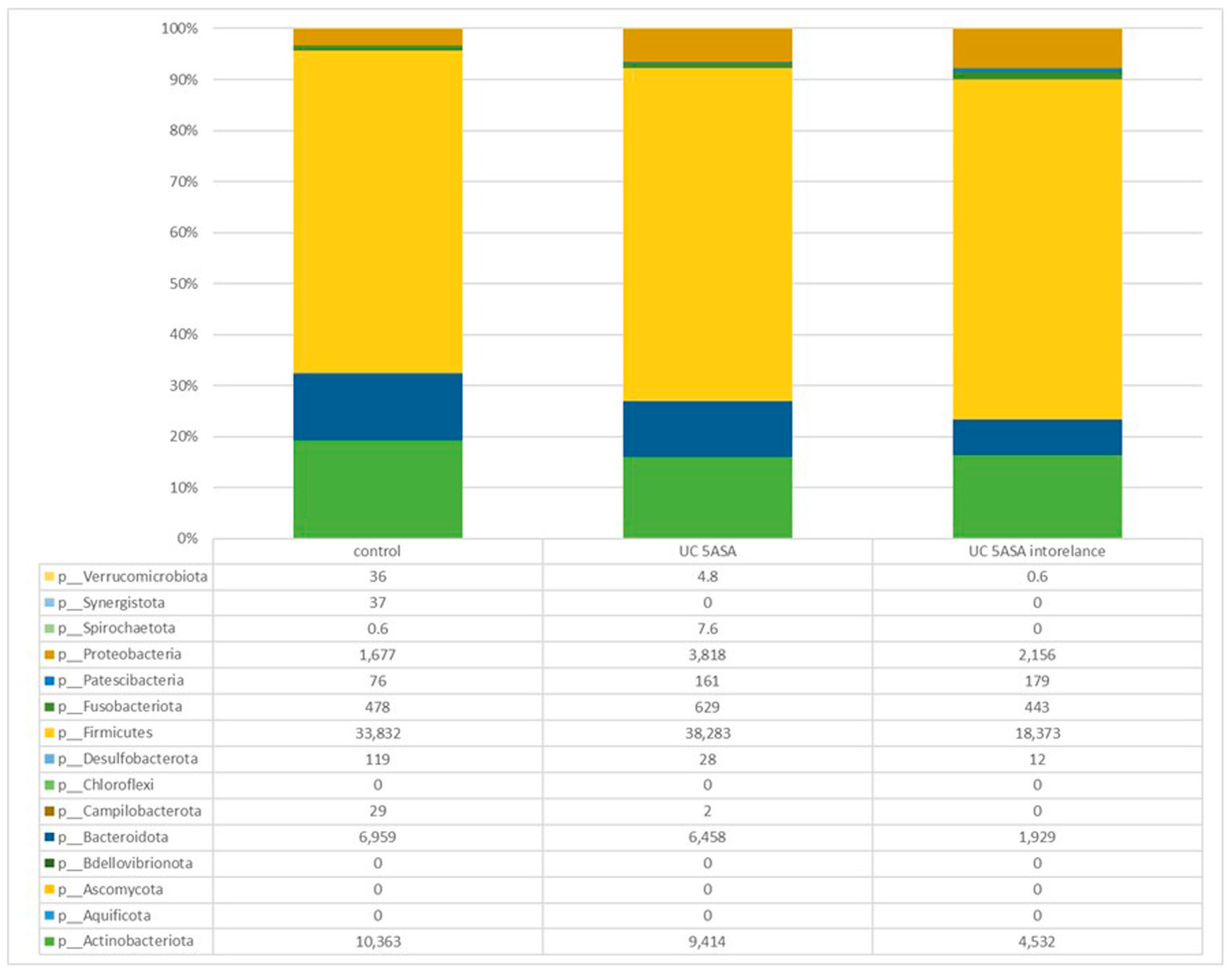
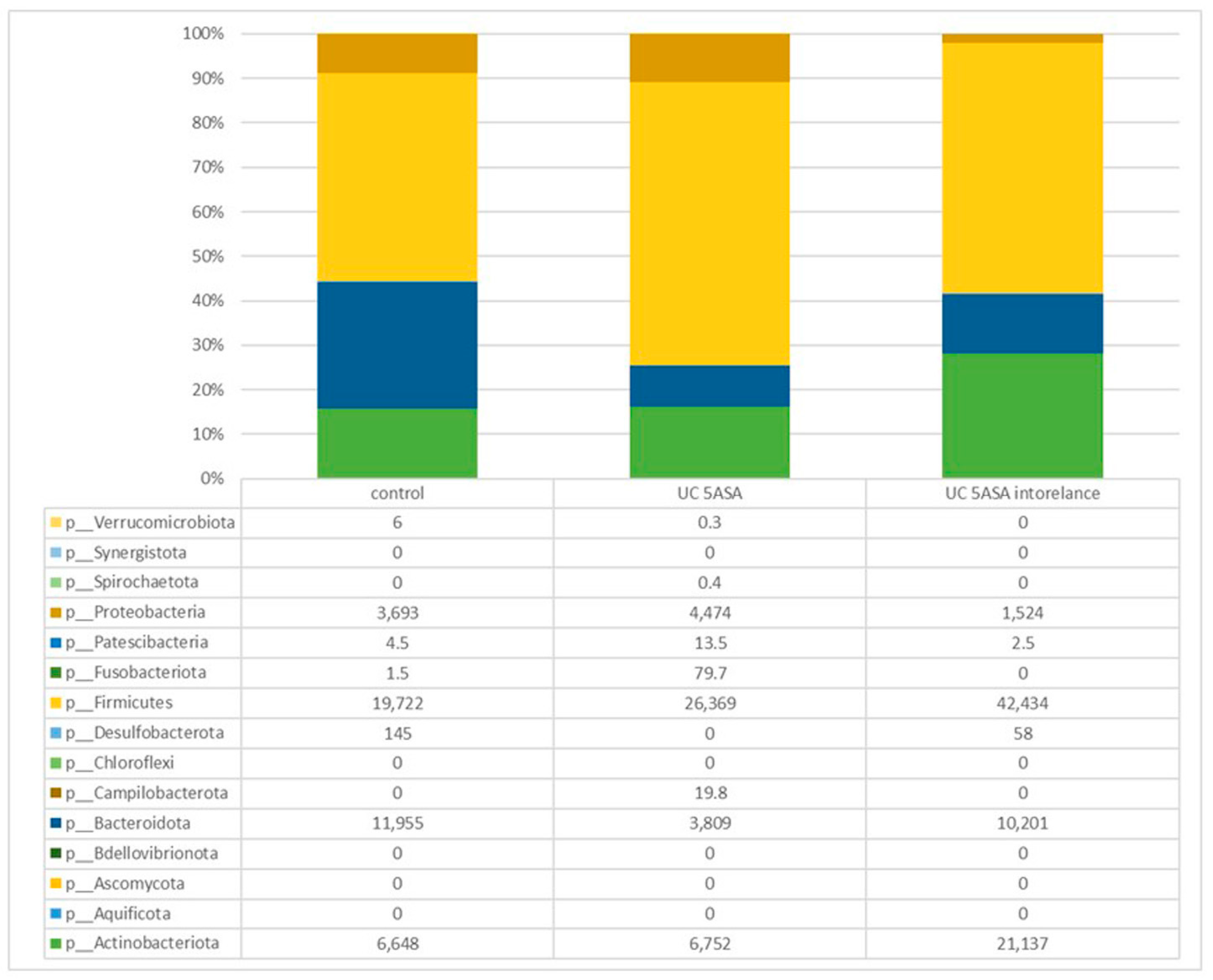
3.2.2. Comparative Analyses of the Taxonomic Composition of the Ileum Microbial Community
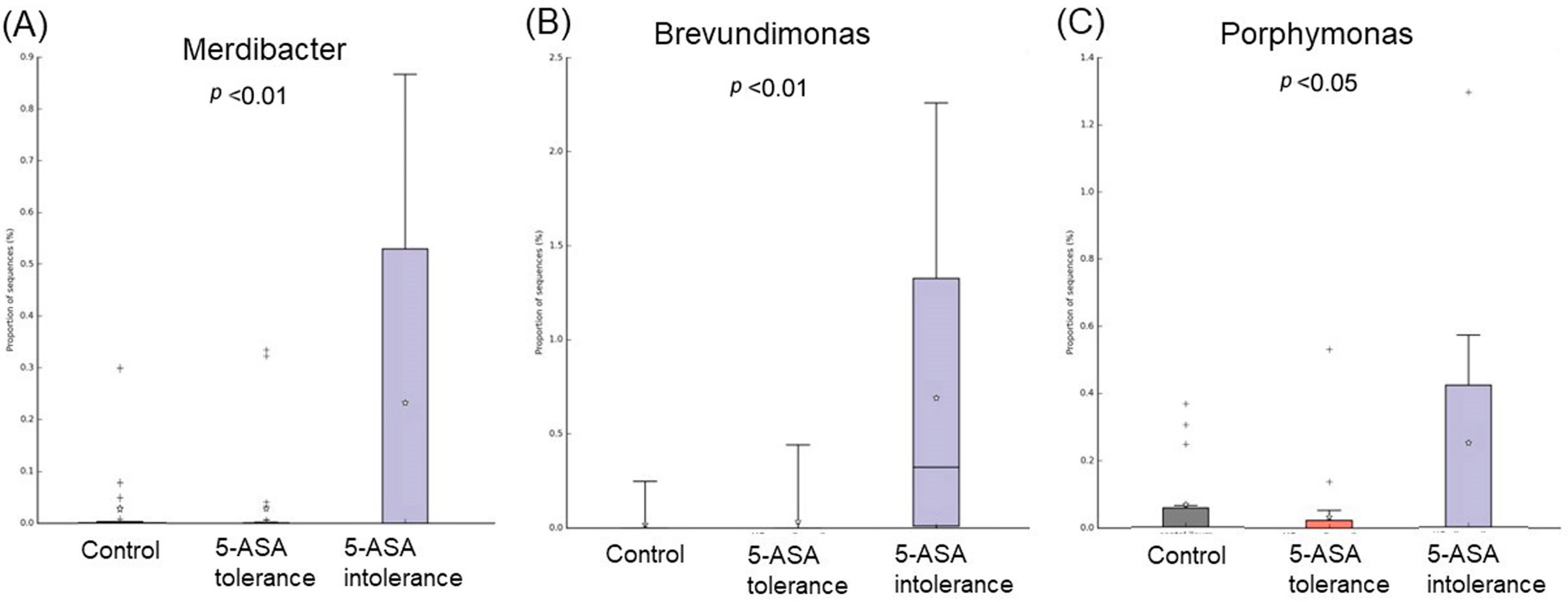
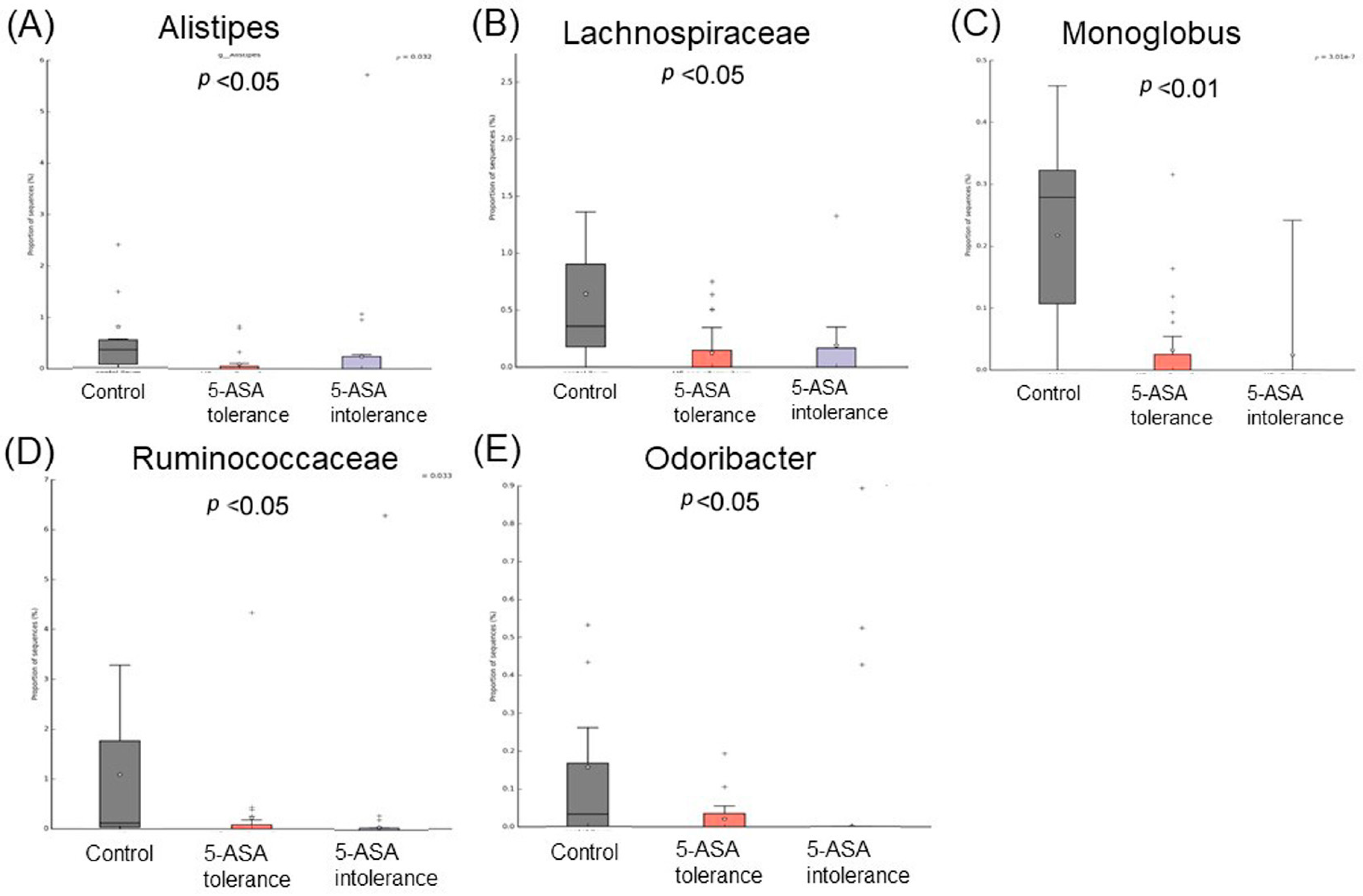
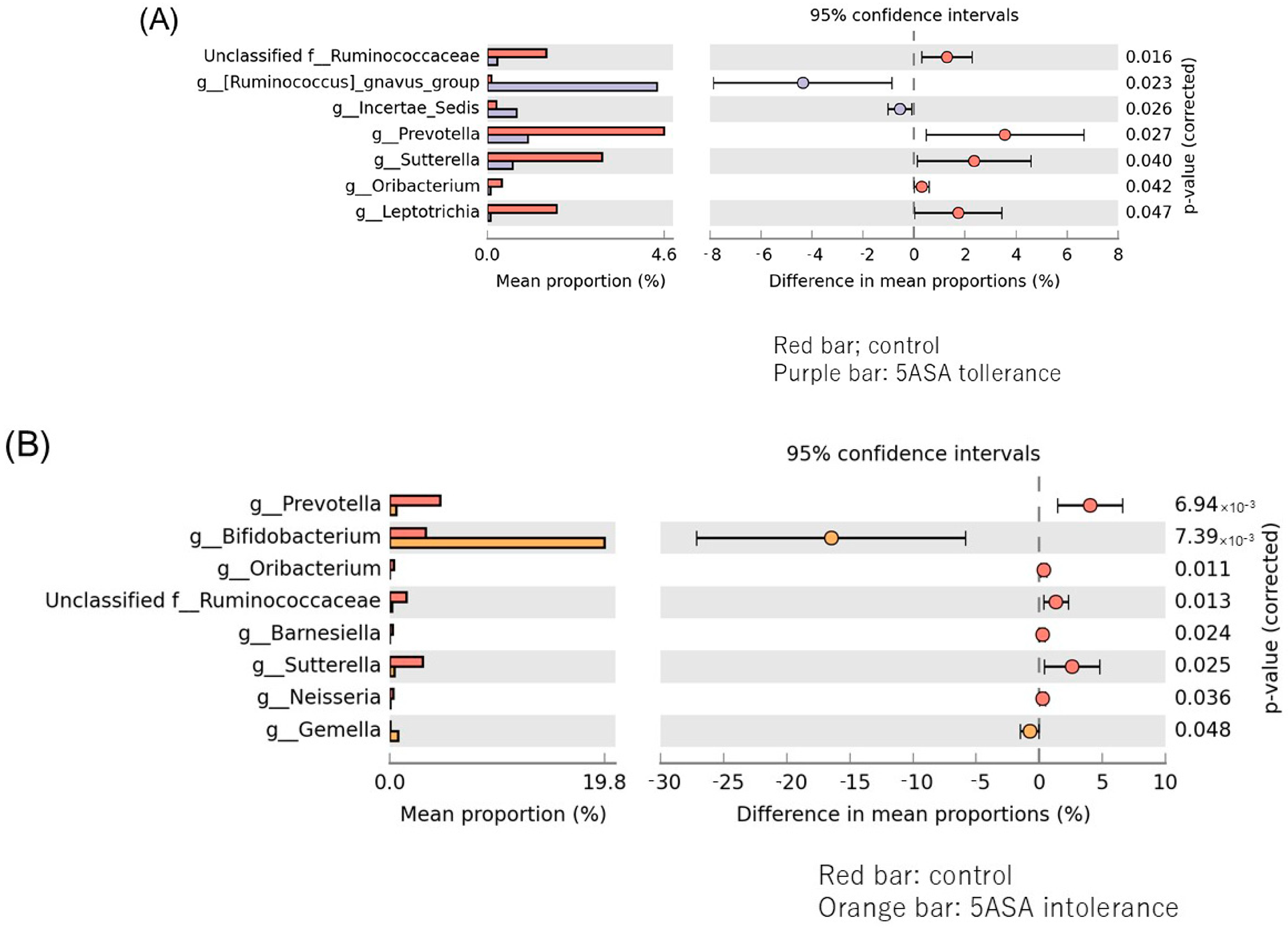
3.2.3. Comparison between Two Groups of Ileal MAM (Figure 5)
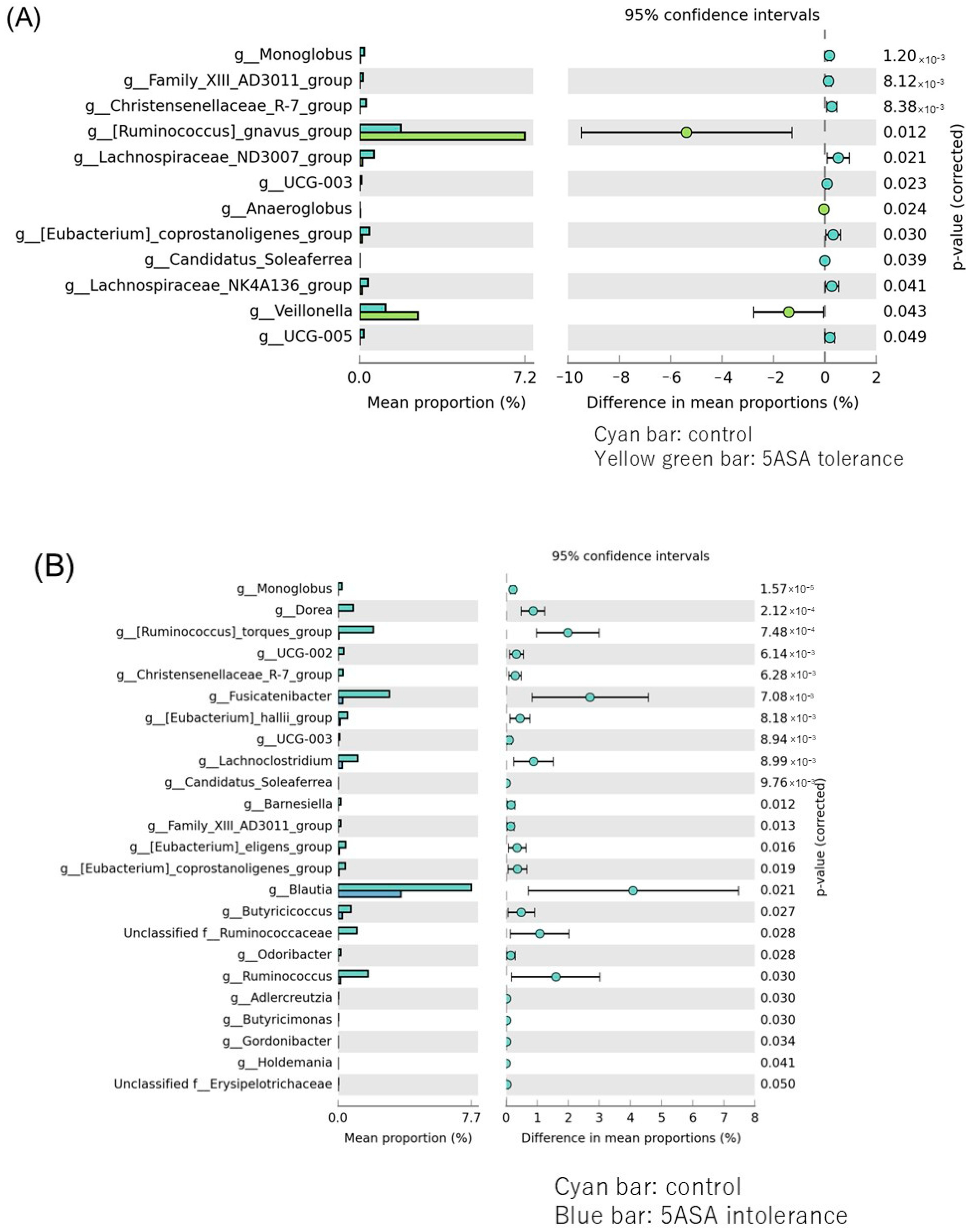
3.2.4. The α-Diversity Indices among the Three Groups Using Sigmoid Colonic Mucus
3.2.5. Comparative Analyses of the Taxonomic Composition of the Microbial Community
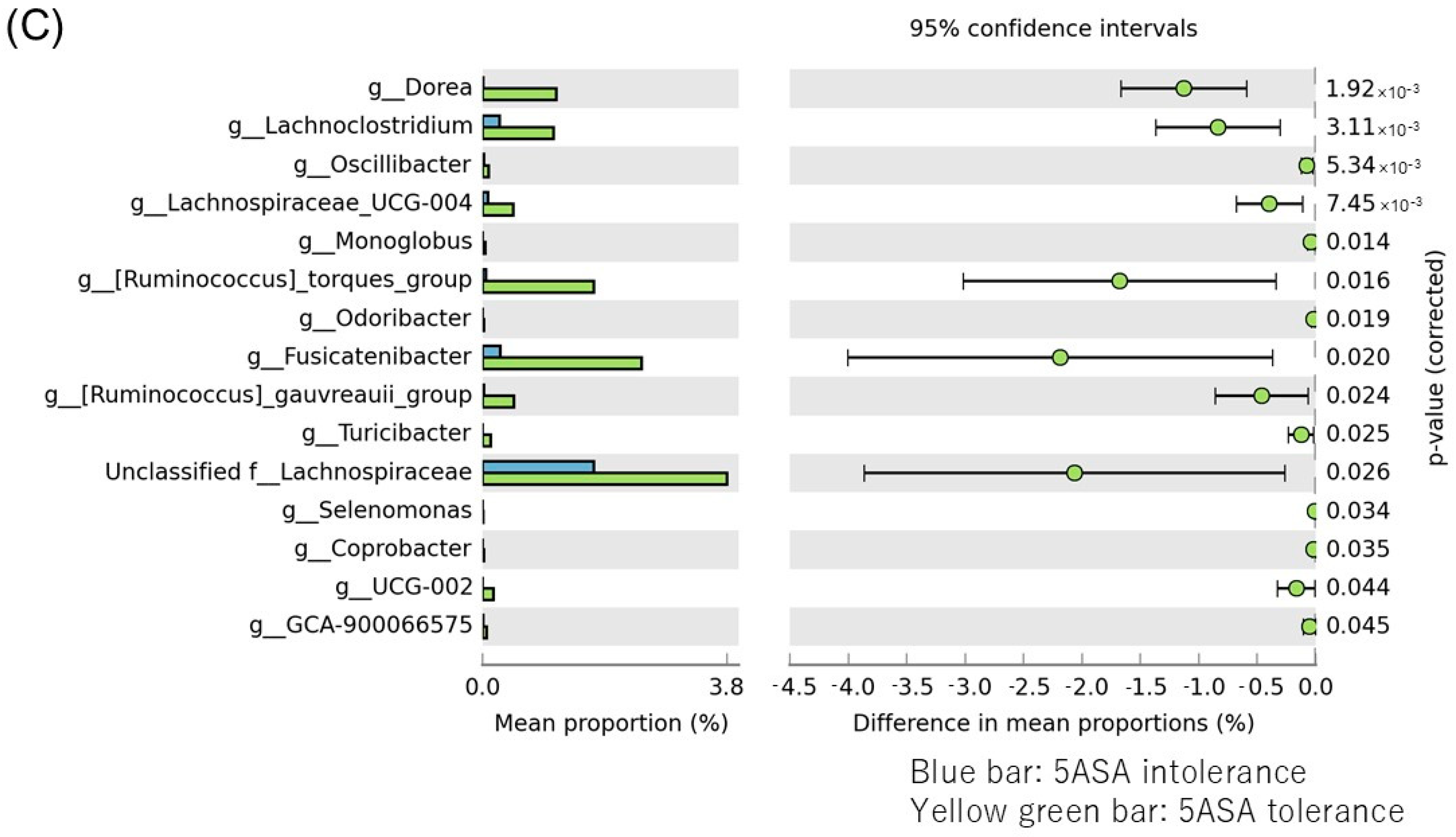
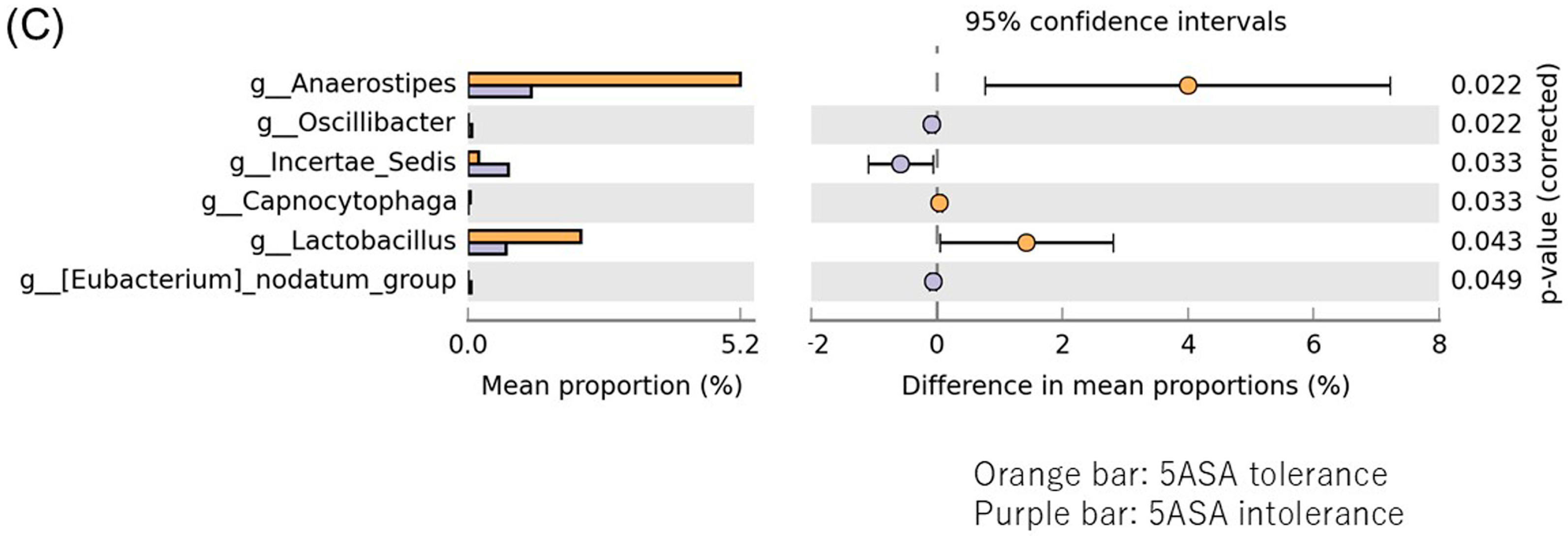
3.2.6. Comparison between Two Groups of Sigmoid Colonic MAM
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kobayashi, T.; Siegmund, B.; Le Berre, C.; Wei, S.C.; Ferrante, M.; Shen, B.; Bernstein, C.N.; Danese, S.; Peyrin-Biroulet, L.; Hibi, T. Ulcerative Colitis. Nat. Rev. Dis. Primers 2020, 6, 74. [Google Scholar] [CrossRef] [PubMed]
- Chu, H.; Khosravi, A.; Kusumawardhani, I.P.; Kwon, A.H.K.; Vasconcelos, A.C.; Cunha, L.D.; Mayer, A.E.; Shen, Y.; Wu, W.-L.; Kambal, A.; et al. Gene-Microbiota Interactions Contribute to the Pathogenesis of Inflammatory Bowel Disease. Science 2016, 352, 1116–1120. [Google Scholar] [CrossRef] [PubMed]
- Hu, S.; Vich Vila, A.; Gacesa, R.; Collij, V.; Stevens, C.; Fu, J.M.; Wong, I.; Talkowski, M.E.; Rivas, M.A.; Imhann, F.; et al. Whole Exome Sequencing Analyses Reveal Gene-Microbiota Interactions in the Context of IBD. Gut 2021, 70, 285–296. [Google Scholar] [CrossRef] [PubMed]
- Qiu, P.; Ishimoto, T.; Fu, L.; Zhang, J.; Zhang, Z.; Liu, Y. The Gut Microbiota in Inflammatory Bowel Disease. Front. Cell. Infect. Microbiol. 2022, 12, 733992. [Google Scholar] [CrossRef] [PubMed]
- Andoh, A.; Nishida, A. Alteration of the Gut Microbiome in Inflammatory Bowel Disease. Digestion 2023, 104, 16–23. [Google Scholar] [CrossRef]
- Meade, S.; Liu Chen Kiow, J.; Massaro, C.; Kaur, G.; Squirell, E.; Bressler, B.; Lunken, G. Gut Microbiome-Associated Predictors as Biomarkers of Response to Advanced Therapies in Inflammatory Bowel Disease: A Systematic Review. Gut Microbes 2023, 15, 2287073. [Google Scholar] [CrossRef]
- Shan, Y.; Lee, M.; Chang, E.B. The Gut Microbiome and Inflammatory Bowel Diseases. Annu. Rev. Med. 2022, 73, 455–468. [Google Scholar] [CrossRef]
- Browne, A.S.; Kelly, C.R. Fecal Transplant in Inflammatory Bowel Disease. Gastroenterol. Clin. N. Am. 2017, 46, 825–837. [Google Scholar] [CrossRef]
- Matsuoka, K.; Kobayashi, T.; Ueno, F.; Matsui, T.; Hirai, F.; Inoue, N.; Kato, J.; Kobayashi, K.; Kobayashi, K.; Koganei, K.; et al. Evidence-Based Clinical Practice Guidelines for Inflammatory Bowel Disease. J. Gastroenterol. 2018, 53, 305–353. [Google Scholar] [CrossRef]
- Harbord, M.; Eliakim, R.; Bettenworth, D.; Karmiris, K.; Katsanos, K.; Kopylov, U.; Kucharzik, T.; Molnár, T.; Raine, T.; Sebastian, S.; et al. Third European Evidence-Based Consensus on Diagnosis and Management of Ulcerative Colitis. Part 2: Current Management. J. Crohns Colitis 2017, 11, 769–784. [Google Scholar] [CrossRef]
- Peppercorn, M.A.; Goldman, P. The Role of Intestinal Bacteria in the Metabolism of Salicylazosulfapyridine. J. Pharmacol. Exp. Ther. 1972, 181, 555–562. [Google Scholar] [PubMed]
- Ko, C.W.; Singh, S.; Feuerstein, J.D.; Falck-Ytter, C.; Falck-Ytter, Y.; Cross, R.K.; American Gastroenterological Association Institute Clinical Guidelines Committee. AGA Clinical Practice Guidelines on the Management of Mild-to-Moderate Ulcerative Colitis. Gastroenterology 2019, 156, 748–764. [Google Scholar] [CrossRef] [PubMed]
- Nakase, H.; Uchino, M.; Shinzaki, S.; Matsuura, M.; Matsuoka, K.; Kobayashi, T.; Saruta, M.; Hirai, F.; Hata, K.; Hiraoka, S.; et al. Evidence-Based Clinical Practice Guidelines for Inflammatory Bowel Disease 2020. J. Gastroenterol. 2021, 56, 489–526. [Google Scholar] [CrossRef] [PubMed]
- Javdan, B.; Lopez, J.G.; Chankhamjon, P.; Lee, Y.-C.J.; Hull, R.; Wu, Q.; Wang, X.; Chatterjee, S.; Donia, M.S. Personalized Mapping of Drug Metabolism by the Human Gut Microbiome. Cell 2020, 181, 1661–1679.e22. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, M.; Zimmermann-Kogadeeva, M.; Wegmann, R.; Goodman, A.L. Mapping Human Microbiome Drug Metabolism by Gut Bacteria and Their Genes. Nature 2019, 570, 462–467. [Google Scholar] [CrossRef]
- Koppel, N.; Maini Rekdal, V.; Balskus, E.P. Chemical Transformation of Xenobiotics by the Human Gut Microbiota. Science 2017, 356, eaag2770. [Google Scholar] [CrossRef]
- Lee, J.W.J.; Plichta, D.; Hogstrom, L.; Borren, N.Z.; Lau, H.; Gregory, S.M.; Tan, W.; Khalili, H.; Clish, C.; Vlamakis, H.; et al. Multi-Omics Reveal Microbial Determinants Impacting Responses to Biologic Therapies in Inflammatory Bowel Disease. Cell Host Microbe 2021, 29, 1294–1304.e4. [Google Scholar] [CrossRef]
- Forslund, S.K.; Chakaroun, R.; Zimmermann-Kogadeeva, M.; Markó, L.; Aron-Wisnewsky, J.; Nielsen, T.; Moitinho-Silva, L.; Schmidt, T.S.B.; Falony, G.; Vieira-Silva, S.; et al. Combinatorial, Additive and Dose-Dependent Drug-Microbiome Associations. Nature 2021, 600, 500–505. [Google Scholar] [CrossRef]
- Gopalakrishnan, V.; Spencer, C.N.; Nezi, L.; Reuben, A.; Andrews, M.C.; Karpinets, T.V.; Prieto, P.A.; Vicente, D.; Hoffman, K.; Wei, S.C.; et al. Gut Microbiome Modulates Response to Anti-PD-1 Immunotherapy in Melanoma Patients. Science 2018, 359, 97–103. [Google Scholar] [CrossRef]
- Vétizou, M.; Pitt, J.M.; Daillère, R.; Lepage, P.; Waldschmitt, N.; Flament, C.; Rusakiewicz, S.; Routy, B.; Roberti, M.P.; Duong, C.P.M.; et al. Anticancer Immunotherapy by CTLA-4 Blockade Relies on the Gut Microbiota. Science 2015, 350, 1079–1084. [Google Scholar] [CrossRef]
- Donald, I.P.; Wilkinson, S.P. The Value of 5-Aminosalicylic Acid in Inflammatory Bowel Disease for Patients Intolerant or Allergic to Sulphasalazine. Postgrad. Med. J. 1985, 61, 1047–1048. [CrossRef] [PubMed]
- Mikami, Y.; Tsunoda, J.; Suzuki, S.; Mizushima, I.; Kiyohara, H.; Kanai, T. Significance of 5-Aminosalicylic Acid Intolerance in the Clinical Management of Ulcerative Colitis. Digestion 2023, 104, 58–65. [Google Scholar] [CrossRef] [PubMed]
- Di Paolo, M.C.; Paoluzi, O.A.; Pica, R.; Iacopini, F.; Crispino, P.; Rivera, M.; Spera, G.; Paoluzi, P. Sulphasalazine and 5-Aminosalicylic Acid in Long-Term Treatment of Ulcerative Colitis: Report on Tolerance and Side-Effects. Dig. Liver Dis. 2001, 33, 563–569. [Google Scholar] [CrossRef] [PubMed]
- Ransford, R.A.J.; Langman, M.J.S. Sulphasalazine and Mesalazine: Serious Adverse Reactions Re-Evaluated on the Basis of Suspected Adverse Reaction Reports to the Committee on Safety of Medicines. Gut 2002, 51, 536–539. [Google Scholar] [CrossRef]
- Dignass, A.; Van Assche, G.; Lindsay, J.O.; Lémann, M.; Söderholm, J.; Colombel, J.F.; Danese, S.; D’Hoore, A.; Gassull, M.; Gomollón, F.; et al. The Second European Evidence-Based Consensus on the Diagnosis and Management of Crohn’s Disease: Current Management. J. Crohns Colitis 2010, 4, 28–62. [Google Scholar] [CrossRef]
- Wang, Y.; Parker, C.E.; Feagan, B.G.; MacDonald, J.K. Oral 5-Aminosalicylic Acid for Maintenance of Remission in Ulcerative Colitis. Cochrane Database Syst. Rev. 2016, 2016, CD000544. [Google Scholar] [CrossRef]
- Miyoshi, J.; Matsuoka, K.; Yoshida, A.; Naganuma, M.; Hisamatsu, T.; Yajima, T.; Inoue, N.; Okamoto, S.; Iwao, Y.; Ogata, H.; et al. 5-Aminosalicylic Acid Aggravates Colitis Mimicking Exacerbation of Ulcerative Colitis. Intest Res 2018, 16, 635–640. [Google Scholar] [CrossRef]
- Bernstein, C.N.; Forbes, J.D. Gut Microbiome in Inflammatory Bowel Disease and Other Chronic Immune-Mediated Inflammatory Diseases. Inflamm. Intest. Dis. 2017, 2, 116–123. [Google Scholar] [CrossRef]
- Zhang, M.; Sun, K.; Wu, Y.; Yang, Y.; Tso, P.; Wu, Z. Interactions between Intestinal Microbiota and Host Immune Response in Inflammatory Bowel Disease. Front. Immunol. 2017, 8, 942. [Google Scholar] [CrossRef]
- Mizuno, S.; Ono, K.; Mikami, Y.; Naganuma, M.; Fukuda, T.; Minami, K.; Masaoka, T.; Terada, S.; Yoshida, T.; Saigusa, K.; et al. 5-Aminosalicylic Acid Intolerance Is Associated with a Risk of Adverse Clinical Outcomes and Dysbiosis in Patients with Ulcerative Colitis. Intest. Res. 2020, 18, 69–78. [Google Scholar] [CrossRef]
- Carstens, A.; Annika, R.; Anna, A.; Anders, M.; Lars, A.; Jonas, H.; Lars, E. Differential Clustering of Fecal and Mucosa-Associated Microbiota in ‘healthy’ Individuals. J. Dig. Dis. 2018, 19, 745–752. [Google Scholar] [CrossRef] [PubMed]
- Juge, N. Relationship between Mucosa-Associated Gut Microbiota and Human Diseases. Biochem. Soc. Trans. 2020, 50, 1225–1236. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, H.; Kuroki, Y.; Higashi, S.; Goda, K.; Fukushima, S.; Katsumoto, R.; Oosawa, M.; Murao, T.; Ishii, M.; Oka, K.; et al. Analysis of the colonic mucosa associated microbiota(MAM) using brushing samples during colonic endoscopic procedures. J. Clin. Biochem. Nutr. 2019, 65, 132–137. [Google Scholar] [CrossRef] [PubMed]
- Osawa, M.; Handa, O.; Fukushima, S.; Matsumoto, H.; Umegaki, E.; Inoue, R.; Naito, Y.; Shiotani, A. Reduced Abundance of Butyric Acid-Producing Bacteria in the Ileal Mucosa-Associated Microbiota of Ulcerative Colitis Patients. J. Clin. Biochem. Nutr. 2023, 73, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Maaser, C.; Sturm, A.; Vavricka, S.R.; Kucharzik, T.; Fiorino, G.; Annese, V.; Calabrese, E.; Baumgart, D.C.; Bettenworth, D.; Borralho Nunes, P.; et al. ECCO-ESGAR Guideline for Diagnostic Assessment in IBD Part 1: Initinal diagnosis, monitoring of known IBD, detection of complication. J. Crohns Colitis 2019, 13, 144–164. [Google Scholar] [CrossRef]
- Shimizu, H.; Arai, K.; Tang, J.; Hosoi, K.; Funayama, R. 5-Aminosalicylate Intolerance Causing Exacerbation in Pediatric Ulcerative Colitis. Pediatr. Int. 2017, 59, 583–587. [Google Scholar] [CrossRef]
- Hayashi, A.; Mikami, Y.; Miyamoto, K.; Kamada, N.; Sato, T.; Mizuno, S.; Naganuma, M.; Teratani, T.; Aoki, R.; Fukuda, S.; et al. Intestinal Dysbiosis and Biotin Deprivation Induce Alopecia through Overgrowth of Lactobacillus Murinus in Mice. Cell Rep. 2017, 20, 1513–1524. [Google Scholar] [CrossRef]
- Klindworth, A.; Pruesse, E.; Schweer, T.; Peplies, J.; Quast, C.; Horn, M.; Glöckner, F.O. Evaluation of General 16S Ribosomal RNA Gene PCR Primers for Classical and next-Generation Sequencing-Based Diversity Studies. Nucleic Acids Res. 2013, 41, e1. [Google Scholar] [CrossRef]
- Parks, D.H.; Tyson, G.W.; Hugenholtz, P.; Beiko, R.G. STAMP: Statistical analysis of taxonomic and functional profiles. Bioinformatics 2014, 30, 3123–3124. [Google Scholar] [CrossRef]
- Herrera-deGuise, C.; Varela, E.; Sarrabayrouse, G.; Pozuelo del Río, M.; Alonso, V.R.; Sainz, N.B.; Casellas, F.; Mayorga, L.F.; Manichanh, C.; Vidaur, F.A.; et al. Gut Microbiota Composition in Long-Remission Ulcerative Colitis in Close to Healthy Gut Microbiota. Inflamm. Bowel Dis. 2023, 29, 1362–1369. [Google Scholar] [CrossRef]
- Basha, O.M.; Hafez, A.H.; Salem, S.M.; Anis, R.H.; Hanafy, A.S. Impact of gut Microbiome alteration in Ulcerative Colitis patients on disease severity and outcom. Clin. Exp. Med. 2023, 23, 1763–1772. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Zhang, H.; Chen, T.; Shi, L.; Wang, D.; Tang, D. Regulatory Role of Short-Chain Fatty Acids in Inflammatory Bowel Disease. Cell Commun. Signal. 2022, 20, 64. [Google Scholar] [CrossRef] [PubMed]
- Lima, S.F.; Gogokhia, L.; Viladomiu, M.; Chou, L.; Putzel, G.; Jin, W.-B.; Pires, S.; Guo, C.-J.; Gerardin, Y.; Crawford, C.V.; et al. Transferable Immunoglobulin A–Coated Odoribacter Splanchnicus in Responders to Fecal Microbiota Transplantation for Ulcerative Colitis Limits Colonic Inflammation. Gastroenterology 2022, 162, 166–178. [Google Scholar] [CrossRef] [PubMed]
- Parker, B.J.; Wearsch, P.A.; Veloo, A.C.M.; Rodriguez-Palacios, A. The Genus Alistipes: Gut Bacteria with Emerging Implications to Inflammation, Cancer, and Mental Health. Front. Immunol. 2020, 11, 906. [Google Scholar] [CrossRef] [PubMed]
- Dziarski, R.; Park, S.Y.; Kashyap, D.R.; Dowd, S.E.; Gupta, D. Pglyrp-Regulated Gut Microflora Prevotella falsenii, Parabacteroides distasonis and Bacteroides eggerthii Enhance and Alistipes finegoldii Attenuates Colitis in Mice. PLoS ONE 2016, 11, e0146162. [Google Scholar] [CrossRef]
- Frank, D.N.; St Amand, A.L.; Feldman, R.A.; Boedeker, E.C.; Harpaz, N.; Pace, N.R. Molecular-Phylogenetic Characterization of Microbial Community Imbalances in Human Inflammatory Bowel Diseases. Proc. Natl. Acad. Sci. USA 2007, 104, 13780–13785. [Google Scholar] [CrossRef]
- Gupta, A.; Khanna, S. Fecal Microbiota Transplantation. JAMA 2017, 318, 102. [Google Scholar] [CrossRef]
- Moayyedi, P.; Surette, M.G.; Kim, P.T.; Libertucci, J.; Wolfe, M.; Onischi, C.; Armstrong, D.; Marshall, J.K.; Kassam, Z.; Reinisch, W.; et al. Fecal Microbiota Transplantation Induces Remission in Patients With Active Ulcerative Colitis in a Randomized Controlled Trial. Gastroenterology 2015, 149, 102–109.e6. [Google Scholar] [CrossRef]
- Paramsothy, S.; Kamm, M.A.; Kaakoush, N.O.; Walsh, A.J.; van den Bogaerde, J.; Samuel, D.; Leong, R.W.L.; Connor, S.; Ng, W.; Paramsothy, R.; et al. Multidonor Intensive Faecal Microbiota Transplantation for Active Ulcerative Colitis: A Randomised Placebo-Controlled Trial. Lancet 2017, 389, 1218–1228. [Google Scholar] [CrossRef]
- Costello, S.P.; Hughes, P.A.; Waters, O.; Bryant, R.V.; Vincent, A.D.; Blatchford, P.; Katsikeros, R.; Makanyanga, J.; Campaniello, M.A.; Mavrangelos, C.; et al. Effect of Fecal Microbiota Transplantation on 8-Week Remission in Patients with Ulcerative Colitis: A Randomized Clinical Trial. JAMA 2019, 321, 156–164. [Google Scholar] [CrossRef]
- Paramsothy, S.; Paramsothy, R.; Rubin, D.T.; Kamm, M.A.; Kaakoush, N.O.; Mitchell, H.M.; Castaño-Rodríguez, N. Faecal Microbiota Transplantation for Inflammatory Bowel Disease: A Systematic Review and Meta-Analysis. J. Crohns Colitis 2017, 11, 1180–1199. [Google Scholar] [CrossRef] [PubMed]
- Costello, S.P.; Soo, W.; Bryant, R.V.; Jairath, V.; Hart, A.L.; Andrews, J.M. Systematic Review with Meta-Analysis: Faecal Microbiota Transplantation for the Induction of Remission for Active Ulcerative Colitis. Aliment. Pharmacol. Ther. 2017, 46, 213–224. [Google Scholar] [CrossRef] [PubMed]
- Allegretti, J.; Eysenbach, L.M.; El-Nachef, N.; Fischer, M.; Kelly, C.; Kassam, Z. The Current Landscape and Lessons from Fecal Microbiota Transplantation for Inflammatory Bowel Disease: Past, Present, and Future. Inflamm. Bowel Dis. 2017, 23, 1710–1717. [Google Scholar] [CrossRef] [PubMed]
- Nagata, N.; Nishijima, S.; Miyoshi-Akiyama, T.; Kojima, Y.; Kimura, M.; Aoki, R.; Ohsugi, M.; Ueki, K.; Miki, K.; Iwata, E.; et al. Population-Level Metagenomics Uncovers Distinct Effects of Multiple Medications on the Human Gut Microbiome. Gastroenterology 2022, 163, 1038–1052. [Google Scholar] [CrossRef]


| Control | 5-ASA Tolerance Group | 5-ASA Intolerance Group | p | |
|---|---|---|---|---|
| 19 | 38 | 15 | ||
| Age [mean ± SD] | 56.2 ± 13.7 | 44.5 ± 16.1 | 48.1 ± 13.4 | 0.018 |
| Gender (Male/Female) | 7/12 | 16/22 | 6/9 | NS |
| Disease duration (yr) | - | 14.0 ± 9.1 | 14.1 ± 16.6 | NS |
| Disease type | - | 18/16/4 | 11/4/0 | NS |
| Pan-colitis/Left side colitis/proctitis | ||||
| Type of 5ASA | - | 7/11/18/2 | 4/3/5/3 | NS |
| Time-dependant/pH-dependant/MMX/SASP | ||||
| Biologics | - | 7(18.4%) [3/1/3/0] | 11(100%) [1/0/1/9] | <0.01 |
| TNFα/IL12-23/VED/JAK | ||||
| Endoscopic activity | - | 29/7(95%)/2/0 | 7/2(82%)/2/0 | NS |
| Mayo 0/1/2/3 | ||||
| DLST | - | - | 0/6/5 | |
| Positive/negative/not inspected |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Matsumoto, H.; Sasahira, M.; Go, T.T.; Yo, S.; Ninomiya, T.; Osawa, M.; Handa, O.; Umegami, E.; Inoue, R.; Shiotani, A. Characteristics of Mucosa-Associated Microbiota in Ulcerative Colitis Patients with 5-Aminosalicylic Acid Intolerance. Biomedicines 2024, 12, 2125. https://doi.org/10.3390/biomedicines12092125
Matsumoto H, Sasahira M, Go TT, Yo S, Ninomiya T, Osawa M, Handa O, Umegami E, Inoue R, Shiotani A. Characteristics of Mucosa-Associated Microbiota in Ulcerative Colitis Patients with 5-Aminosalicylic Acid Intolerance. Biomedicines. 2024; 12(9):2125. https://doi.org/10.3390/biomedicines12092125
Chicago/Turabian StyleMatsumoto, Hiroshi, Momoyo Sasahira, Tei Tei Go, Shogen Yo, Takehiro Ninomiya, Motoyasu Osawa, Osamu Handa, Eiji Umegami, Ryo Inoue, and Akiko Shiotani. 2024. "Characteristics of Mucosa-Associated Microbiota in Ulcerative Colitis Patients with 5-Aminosalicylic Acid Intolerance" Biomedicines 12, no. 9: 2125. https://doi.org/10.3390/biomedicines12092125
APA StyleMatsumoto, H., Sasahira, M., Go, T. T., Yo, S., Ninomiya, T., Osawa, M., Handa, O., Umegami, E., Inoue, R., & Shiotani, A. (2024). Characteristics of Mucosa-Associated Microbiota in Ulcerative Colitis Patients with 5-Aminosalicylic Acid Intolerance. Biomedicines, 12(9), 2125. https://doi.org/10.3390/biomedicines12092125





