Association between Neutrophil-Lymphocyte Ratio and Herpes Zoster Infection in 1688 Living Donor Liver Transplantation Recipients at a Large Single Center
Abstract
:1. Introduction
2. Materials and Methods
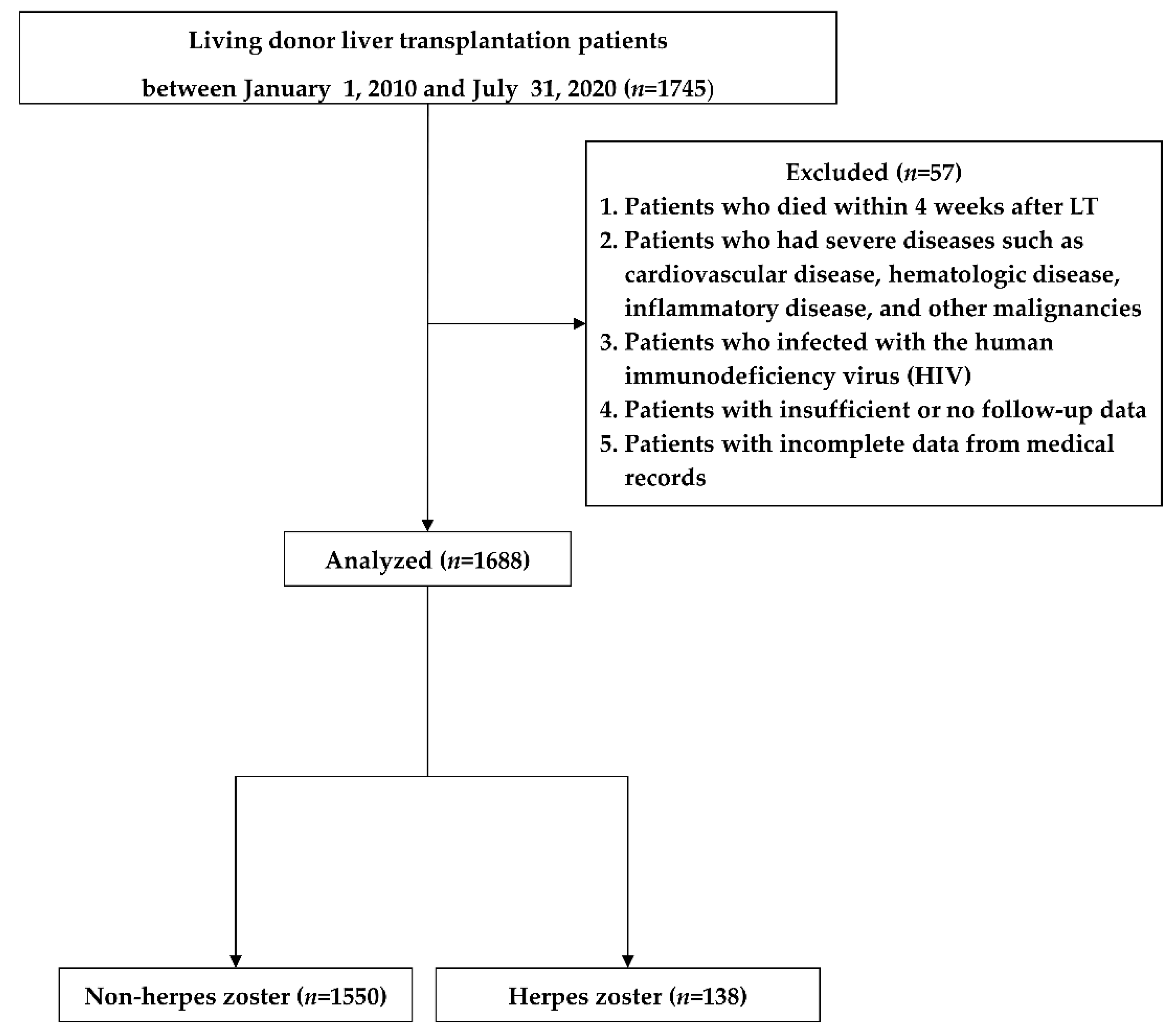
2.1. Study Design and Patients
2.2. Clinical Data Collection and Outcome Assessments
2.3. Identification of HZ and Immunosuppression
2.4. Primary and Secondary Outcomes
2.5. Statistical Analyses
3. Results
3.1. Incidence of HZ
3.2. Patient Characteristics
3.3. Primary Outcomes
3.4. Secondary Outcomes
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Jih, J.S.; Chen, Y.J.; Lin, M.W.; Chen, Y.C.; Chen, T.J.; Huang, Y.L.; Chen, C.C.; Lee, D.D.; Chang, Y.T.; Wang, W.J.; et al. Epidemiological features and costs of herpes zoster in Taiwan: A national study 2000 to 2006. Acta Derm. Venereol. 2009, 89, 612–616. [Google Scholar] [CrossRef] [Green Version]
- McDonald, J.R.; Zeringue, A.L.; Caplan, L.; Ranganathan, P.; Xian, H.; Burroughs, T.E.; Fraser, V.J.; Cunningham, F.; Eisen, S.A. Herpes zoster risk factors in a national cohort of veterans with rheumatoid arthritis. Clin. Infect. Dis. 2009, 48, 1364–1371. [Google Scholar] [CrossRef]
- Engels, E.A.; Rosenberg, P.S.; Biggar, R.J. Zoster incidence in human immunodeficiency virus-infected hemophiliacs and homosexual men, 1984–1997. J. Infect. Dis. 1999, 180, 1784–1789. [Google Scholar] [CrossRef] [Green Version]
- Thomas, S.L.; Hall, A.J. What does epidemiology tell us about risk factors for herpes zoster? Lancet Infect. Dis. 2004, 4, 26–33. [Google Scholar] [CrossRef]
- Nagel, M.A.; Gilden, D. Update on varicella zoster virus vasculopathy. Curr. Infect. Dis. Rep. 2014, 16, 407. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Donahue, J.G.; Choo, P.W.; Manson, J.E.; Platt, R. The incidence of herpes zoster. Arch. Intern. Med. 1995, 155, 1605–1609. [Google Scholar] [CrossRef] [PubMed]
- Yoon, K.J.; Kim, S.H.; Lee, E.H.; Choi, J.H. Disseminated herpes zoster in an immunocompetent elderly patient. Korean J. Pain 2013, 26, 195–198. [Google Scholar] [CrossRef] [PubMed]
- Levitsky, J.; Kalil, A.; Meza, J.L.; Hurst, G.E.; Freifeld, A. Herpes zoster infection after liver transplantation: A case-control study. Liver Transpl. 2005, 11, 320–325. [Google Scholar] [CrossRef] [PubMed]
- Koo, S.; Gagne, L.S.; Lee, P.; Pratibhu, P.P.; James, L.M.; Givertz, M.M.; Marty, F.M. Incidence and risk factors for herpes zoster following heart transplantation. Transpl. Infect. Dis. 2014, 16, 17–25. [Google Scholar] [CrossRef] [Green Version]
- Miller, G.G.; Dummer, J.S. Herpes simplex and varicella zoster viruses: Forgotten but not gone. Am. J. Transpl. 2007, 7, 741–747. [Google Scholar] [CrossRef]
- Pergam, S.A.; Forsberg, C.W.; Boeckh, M.J.; Maynard, C.; Limaye, A.P.; Wald, A.; Smith, N.L.; Young, B.A. Herpes zoster incidence in a multicenter cohort of solid organ transplant recipients. Transpl. Infect. Dis. 2011, 13, 15–23. [Google Scholar] [CrossRef] [Green Version]
- Hamaguchi, Y.; Mori, A.; Uemura, T.; Ogawa, K.; Fujimoto, Y.; Okajima, H.; Kaido, T.; Uemoto, S. Incidence and risk factors for herpes zoster in patients undergoing liver transplantation. Transpl. Infect. Dis. 2015, 17, 671–678. [Google Scholar] [CrossRef] [PubMed]
- Kim, W.; Kim, S.; Oh, J.; Jeong, Y.J.; Rhu, J.; Kim, K.S.; Lee, J.; Choi, G.S.; Kim, J.M.; Joh, J.W. Incidence and risk factors for herpes zoster after adult liver transplantation. Ann. Surg. Treat. Res. 2019, 96, 95–99. [Google Scholar] [CrossRef] [PubMed]
- Herrero, J.I.; Quiroga, J.; Sangro, B.; Pardo, F.; Rotellar, F.; Alvarez-Cienfuegos, J.; Prieto, J. Herpes zoster after liver transplantation: Incidence, risk factors, and complications. Liver Transpl. 2004, 10, 1140–1143. [Google Scholar] [CrossRef]
- Gourishankar, S.; McDermid, J.C.; Jhangri, G.S.; Preiksaitis, J.K. Herpes zoster infection following solid organ transplantation: Incidence, risk factors and outcomes in the current immunosuppressive era. Am. J. Transpl. 2004, 4, 108–115. [Google Scholar] [CrossRef] [PubMed]
- Song, M.; Graubard, B.I.; Rabkin, C.S.; Engels, E.A. Neutrophil-to-lymphocyte ratio and mortality in the United States general population. Sci. Rep. 2021, 11, 464. [Google Scholar] [CrossRef] [PubMed]
- Tamhane, U.U.; Aneja, S.; Montgomery, D.; Rogers, E.K.; Eagle, K.A.; Gurm, H.S. Association between admission neutrophil to lymphocyte ratio and outcomes in patients with acute coronary syndrome. Am. J. Cardiol. 2008, 102, 653–657. [Google Scholar] [CrossRef]
- Templeton, A.J.; McNamara, M.G.; Šeruga, B.; Vera-Badillo, F.E.; Aneja, P.; Ocaña, A.; Leibowitz-Amit, R.; Sonpavde, G.; Knox, J.J.; Tran, B.; et al. Prognostic role of neutrophil-to-lymphocyte ratio in solid tumors: A systematic review and meta-analysis. J. Natl. Cancer Inst. 2014, 106, dju124. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Onodera, T.; Goseki, N.; Kosaki, G. [Prognostic nutritional index in gastrointestinal surgery of malnourished cancer patients]. Nihon Geka Gakkai Zasshi 1984, 85, 1001–1005. [Google Scholar]
- Hwang, S.; Ahn, C.S.; Kim, K.H.; Moon, D.B.; Ha, T.Y.; Song, G.W.; Jung, D.H.; Park, G.C.; Lee, S.G. A cross-sectional analysis of long-term immunosuppressive regimens after liver transplantation at Asan Medical Center: Increased preference for mycophenolate mofetil. Ann. Hepatobiliary Pancreat. Surg. 2018, 22, 19–26. [Google Scholar] [CrossRef] [Green Version]
- Salah, A.; Fujimoto, M.; Yoshizawa, A.; Yurugi, K.; Miyagawa-Hayashino, A.; Sumiyoshi, S.; Minamiguchi, S.; Uemoto, S.; Maekawa, T.; Haga, H. Application of complement component 4d immunohistochemistry to ABO-compatible and ABO-incompatible liver transplantation. Liver Transpl. 2014, 20, 200–209. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Demetris, A.; Adams, D.; Bellamy, C.; Blakolmer, K.; Clouston, A.; Dhillon, A.P.; Fung, J.; Gouw, A.; Gustafsson, B.; Haga, H.; et al. Update of the international banff schema for liver allograft rejection: Working recommendations for the histopathologic staging and reporting of chronic rejection. Hepatology 2000, 31, 792–799. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.S.; Lin, Y.C.; Lao, H.C.; Chen, C.C. Interventional treatments for postherpetic neuralgia: A systematic review. Pain Physician 2019, 22, 209–228. [Google Scholar] [CrossRef] [PubMed]
- Yawn, B.P.; Gilden, D. The global epidemiology of herpes zoster. Neurology 2013, 81, 928–930. [Google Scholar] [CrossRef] [Green Version]
- Kim, Y.J.; Lee, C.N.; Lim, C.Y.; Jeon, W.S.; Park, Y.M. Population-based study of the epidemiology of herpes zoster in Korea. J. Korean Med. Sci. 2014, 29, 1706–1710. [Google Scholar] [CrossRef] [Green Version]
- van Wijck, A.J.M.; Aerssens, Y.R. Pain, itch, quality of life, and costs after herpes zoster. Pain Pract. 2017, 17, 738–746. [Google Scholar] [CrossRef]
- Johnson, R.W.; Bouhassira, D.; Kassianos, G.; Leplège, A.; Schmader, K.E.; Weinke, T. The impact of herpes zoster and post-herpetic neuralgia on quality-of-life. BMC Med. 2010, 8, 37. [Google Scholar] [CrossRef] [Green Version]
- Meyers, J.L.; Candrilli, S.D.; Rausch, D.A.; Yan, S.; Patterson, B.J.; Levin, M.J. Costs of herpes zoster complications in older adults: A cohort study of US claims database. Vaccine 2019, 37, 1235–1244. [Google Scholar] [CrossRef]
- Gialloreti, L.E.; Merito, M.; Pezzotti, P.; Naldi, L.; Gatti, A.; Beillat, M.; Serradell, L.; di Marzo, R.; Volpi, A. Epidemiology and economic burden of herpes zoster and post-herpetic neuralgia in Italy: A retrospective, population-based study. BMC Infect. Dis. 2010, 10, 230. [Google Scholar] [CrossRef] [Green Version]
- Fleming, D.M.; Cross, K.W.; Cobb, W.A.; Chapman, R.S. Gender difference in the incidence of shingles. Epidemiol. Infect. 2004, 132, 1–5. [Google Scholar] [CrossRef] [Green Version]
- Ko, G.B.; Kim, T.; Kim, S.H.; Choi, S.H.; Kim, Y.S.; Woo, J.H.; Kim, Y.H.; Park, J.B.; Lee, S.K.; Park, S.K.; et al. Increased incidence of herpes zoster in the setting of cytomegalovirus preemptive therapy after kidney transplantation. Transpl. Infect. Dis. 2013, 15, 416–423. [Google Scholar] [CrossRef]
- Cheong, H.; Kim, B.; Jeong Joo, E.; Chang, Y.; Yoo, S. 2303. Chronic hepatitis B virus infection and risk of herpes zoster: A cohort study. Open Forum Infect. Dis. 2019, 6, S789. [Google Scholar] [CrossRef]
- Lauzurica, R.; Bayés, B.; Frías, C.; Fontseré, N.; Hernandez, A.; Matas, L.; Jimenez, A.; Bonet, J.; Romero, R. Disseminated varicella infection in adult renal allograft recipients: Role of mycophenolate mofetil. Transpl. Proc. 2003, 35, 1758–1759. [Google Scholar] [CrossRef]
- Satoh, S.; Tada, H.; Murakami, M.; Tsuchiya, N.; Inoue, T.; Togashi, H.; Matsuura, S.; Hayase, Y.; Suzuki, T.; Habuchi, T. The influence of mycophenolate mofetil versus azathioprine and mycophenolic acid pharmacokinetics on the incidence of acute rejection and infectious complications after renal transplantation. Transpl. Proc. 2005, 37, 1751–1753. [Google Scholar] [CrossRef]
- Moore, J.; Middleton, L.; Cockwell, P.; Adu, D.; Ball, S.; Little, M.A.; Ready, A.; Wheatley, K.; Borrows, R. Calcineurin inhibitor sparing with mycophenolate in kidney transplantation: A systematic review and meta-analysis. Transplantation 2009, 87, 591–605. [Google Scholar] [CrossRef] [Green Version]
- Chapman, J.R. Chronic calcineurin inhibitor nephrotoxicity-lest we forget. Am. J. Transpl. 2011, 11, 693–697. [Google Scholar] [CrossRef] [PubMed]
- Sintchak, M.D.; Fleming, M.A.; Futer, O.; Raybuck, S.A.; Chambers, S.P.; Caron, P.R.; Murcko, M.A.; Wilson, K.P. Structure and mechanism of inosine monophosphate dehydrogenase in complex with the immunosuppressant mycophenolic acid. Cell 1996, 85, 921–930. [Google Scholar] [CrossRef] [Green Version]
- Allison, A.C.; Eugui, E.M. Preferential suppression of lymphocyte proliferation by mycophenolic acid and predicted long-term effects of mycophenolate mofetil in transplantation. Transpl. Proc. 1994, 26, 3205–3210. [Google Scholar]
- Levin, M.J.; Smith, J.G.; Kaufhold, R.M.; Barber, D.; Hayward, A.R.; Chan, C.Y.; Chan, I.S.; Li, D.J.; Wang, W.; Keller, P.M.; et al. Decline in varicella-zoster virus (VZV)-specific cell-mediated immunity with increasing age and boosting with a high-dose VZV vaccine. J. Infect. Dis. 2003, 188, 1336–1344. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Glowacka, P.; Rudnicka, L.; Warszawik-Hendzel, O.; Sikora, M.; Goldust, M.; Gajda, P.; Stochmal, A.; Blicharz, L.; Rakowska, A.; Olszewska, M. The antiviral properties of cyclosporine. Focus on coronavirus, hepatitis C virus, influenza virus, and human immunodeficiency virus infections. Biology 2020, 9, 192. [Google Scholar] [CrossRef]
- Grivennikov, S.I.; Greten, F.R.; Karin, M. Immunity, inflammation, and cancer. Cell 2010, 140, 883–899. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bowers, N.L.; Helton, E.S.; Huijbregts, R.P.H.; Goepfert, P.A.; Heath, S.L.; Hel, Z. Immune suppression by neutrophils in HIV-1 infection: Role of PD-L1/PD-1 pathway. PLoS Pathog. 2014, 10, e1003993. [Google Scholar] [CrossRef] [Green Version]
- Ohki, S.; Shibata, M.; Gonda, K.; Machida, T.; Shimura, T.; Nakamura, I.; Ohtake, T.; Koyama, Y.; Suzuki, S.; Ohto, H.; et al. Circulating myeloid-derived suppressor cells are increased and correlate to immune suppression, inflammation and hypoproteinemia in patients with cancer. Oncol. Rep. 2012, 28, 453–458. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Soh, Y.; Lee, J.H.; Jung, J.; Dong, S.H.; Byun, J.Y.; Park, M.S.; Kim, S.H.; Yeo, S.G. A high neutrophil-to-lymphocyte ratio is associated with recovery from ramsay hunt syndrome. ORL J. Otorhinolaryngol. Relat. Spec. 2019, 81, 130–137. [Google Scholar] [CrossRef] [PubMed]
- Moon, D.B.; Lee, S.G.; Hwang, S.; Kim, K.H.; Ahn, C.S.; Ha, T.Y.; Song, G.W.; Jung, D.H.; Park, G.C.; Namkoong, J.M.; et al. More than 300 consecutive living donor liver transplants a year at a single center. Transpl. Proc. 2013, 45, 1942–1947. [Google Scholar] [CrossRef]

| Study Population | ||||
|---|---|---|---|---|
| Total (n = 1688) | Non-HZ (n = 1550) | HZ (n = 138) | p-Value | |
| Demographic variables | ||||
| Age, years | 53.04 ± 8.03 | 52.82 ± 8.02 | 55.56 ± 7.68 | <0.001 |
| Sex, male | 1276 (69.68) | 1180 (76.13) | 96 (66.57) | 0.106 |
| BMI, kg/m2 | 24.13 ± 3.21 | 24.13 ± 3.16 | 24.21 ± 3.78 | 0.786 |
| DM | 410 (24.29) | 374 (24.13) | 36 (26.09) | 0.682 |
| HTN | 274 (16.23) | 247 (15.94) | 27 (19.57) | 0.323 |
| CAD | 118 (6.99) | 111 (7.16) | 7 (5.07) | 0.467 |
| MELD score | 15.17 ± 8.48 | 15.11 ± 8.46 | 15.79 ± 8.62 | 0.369 |
| Child–Pugh score | 7.92 ± 2.19 | 7.90 ± 2.20 | 8.10 ± 2.18 | 0.306 |
| Child–Pugh classification | 0.473 | |||
| A | 562 (33.29) | 520 (33.55) | 42 (30.43) | |
| B | 696 (41.23) | 641 (41.35) | 55 (39.86) | |
| C | 430 (25.47) | 389 (25.10) | 41 (29.71) | |
| ABO incompatibility | 333 (19.73) | 301 (19.42) | 32 (23.19) | 0.340 |
| Retransplantation | 10 (0.59) | 9 (0.58) | 1 (0.72) | 0.575 |
| Etiology | ||||
| HBV | 1085 (64.28) | 1003 (64.71) | 82 (59.42) | 0.250 |
| HCV | 132 (7.82) | 121 (7.81) | 11 (7.97) | 0.923 |
| Alcoholic liver cirrhosis | 290 (17.18) | 267 (17.23) | 23 (16.67) | 0.961 |
| HCC | 852 (50.47) | 786 (50.71) | 66 (47.83) | 0.575 |
| Toxic hepatitis | 25 (1.48) | 24 (1.55) | 1 (0.72) | 0.716 |
| Other | 1 (0.06) | 1 (0.06) | 0 (0.00) | 1.000 |
| Donor-related variables | ||||
| Age, years | 27.32 ± 7.68 | 27.35 ± 7.68 | 26.99 ± 7.71 | 0.598 |
| Sex, male | 1229 (72.81) | 1128 (72.77) | 101 (73.19) | 0.996 |
| BMI, kg/m2 | 22.92 ± 2.91 | 22.89 ± 2.90 | 23.28 ± 3.02 | 0.130 |
| Laboratory variables | ||||
| WBC | 3.81 ± 2.73 | 3.81 ± 2.76 | 3.78 ± 2.47 | 0.899 |
| Hemoglobin | 10.79 ± 2.21 | 10.81 ± 2.22 | 10.54 ± 2.11 | 0.169 |
| INR | 1.48 ± 0.52 | 1.48 ± 0.53 | 1.51 ± 0.48 | 0.496 |
| Fibrinogen | 176.53 ± 76.70 | 176.98 ± 77.46 | 171.54 ± 67.68 | 0.425 |
| Albumin | 3.16 ± 0.59 | 3.16 ± 0.59 | 3.12 ± 0.55 | 0.437 |
| Total bilirubin | 5.15 ± 8.61 | 5.12 ± 8.53 | 5.57 ± 9.46 | 0.555 |
| Sodium | 137.81 ± 5.14 | 137.79 ± 5.17 | 137.99 ± 4.87 | 0.656 |
| Creatinine | 0.93 ± 0.85 | 0.93 ± 0.87 | 0.91 ± 0.68 | 0.814 |
| NLR | 3.36 ± 3.72 | 3.29 ± 3.60 | 4.13 ± 4.88 | 0.011 |
| PLR | 95.17 ± 67.03 | 95.13 ± 67.40 | 95.67 ± 63.05 | 0.927 |
| PNI | 36.07 ± 6.50 | 36.13 ± 6.55 | 35.44 ± 5.92 | 0.233 |
| CRP/albumin | 0.25 ± 0.52 | 0.24 ± 0.53 | 0.26 ± 0.44 | 0.168 |
| Intraoperative variables | ||||
| Total ischemic time, min | 127.08 ± 29.06 | 127.23 ± 29.26 | 125.44 ± 26.66 | 0.487 |
| Post-reperfusion syndrome | 894 (52.96) | 815 (52.58) | 79 (57.25) | 0.335 |
| GRWR | 1.13 ± 0.24 | 1.12 ± 0.24 | 1.16 ± 0.27 | |
| Intraoperative transfusion | 1359 (80.51) | 1239 (79.94) | 120 (86.96) | 0.060 |
| Immunosuppressant variables | ||||
| Steroid use | 1672 (99.05) | 1537 (99.16) | 135 (97.83) | 0.137 |
| MMF use | 1322 (78.32) | 1203 (77.61) | 119 (86.23) | 0.025 |
| Cyclosporine use | 169 (10.01) | 160 (10.32) | 9 (6.52) | 0.201 |
| Tacrolimus use | 1519 (89.99) | 1390 (89.68) | 129 (93.48) | 0.201 |
| Acute cellular rejection | 159 (9.42) | 143 (9.23) | 16 (11.59) | 0.447 |
| Univariate | Multivariate | |||||
|---|---|---|---|---|---|---|
| HR | 95% CI | p-Value | HR | 95% CI | p-Value | |
| NLR | 1.05 | 1.02–1.09 | 0.003 | 1.05 | 1.02–1.09 | 0.005 |
| Age, years | 1.06 | 1.04–1.09 | <0.001 | 1.06 | 1.04–1.09 | <0.001 |
| Sex, male | 0.69 | 0.48–0.99 | 0.047 | 0.66 | 0.46–0.95 | 0.026 |
| BMI, kg/m2 | 1.00 | 0.95–1.06 | 0.941 | |||
| DM | 1.06 | 0.73–1.55 | 0.748 | |||
| HTN | 1.35 | 0.89–2.06 | 0.160 | |||
| CAD | 0.83 | 0.39–1.76 | 0.623 | |||
| MELD score | 1.00 | 0.98–1.02 | 0.778 | |||
| Child–Pugh score | 1.02 | 0.95–1.10 | 0.576 | |||
| Child–Pugh classification | 0.753 | |||||
| A | ||||||
| B | 1.05 | 0.70–1.56 | 0.827 | |||
| C | 1.17 | 0.76–1.80 | 0.465 | |||
| HBV | 0.74 | 0.53–1.04 | 0.085 | 0.68 | 0.48–0.96 | 0.030 |
| HCV | 1.05 | 0.57–1.93 | 0.887 | |||
| Alcoholic liver cirrhosis | 1.10 | 0.70–1.71 | 0.690 | |||
| HCC | 0.86 | 0.62–1.20 | 0.368 | |||
| Toxic hepatitis | 0.61 | 0.09–4.32 | 0.623 | |||
| Retransplantation | 1.41 | 0.20–9.99 | 0.734 | |||
| Calcineurin inhibitor | ||||||
| Tacrolimus | 1.00 (Ref.) | 1.00 (Ref.) | ||||
| Cyclosporine | 0.53 | 0.27–1.03 | 0.063 | 0.54 | 0.27–1.08 | 0.082 |
| MMF | 1.77 | 1.09–2.87 | 0.021 | 1.81 | 1.11–2.97 | 0.019 |
| Steroid | 0.68 | 0.22–2.14 | 0.515 | |||
| ACR | 1.39 | 0.83–2.33 | 0.219 | |||
| GRWR | 1.53 | 0.80–2.94 | 0.203 | |||
| PLR | 1.00 | 1.00–1.00 | 0.536 | |||
| PNI | 0.98 | 0.96–1.01 | 0.235 | |||
| CRP/albumin | 1.14 | 0.93–1.39 | 0.219 | |||
| Total ischemic time, hour | 1.00 | 0.99–1.00 | 0.564 | |||
| Transfusion | 1.43 | 0.88–2.35 | 0.155 | |||
| Univariate | Multivariate | |||||
|---|---|---|---|---|---|---|
| HR | 95% CI | p-Value | HR | 95% CI | p-Value | |
| NLR | 1.08 | 1.03–1.13 | <0.001 | 1.08 | 1.03–1.13 | 0.001 |
| Age, years | 1.11 | 1.07–1.16 | <0.001 | 1.11 | 1.07–1.16 | <0.001 |
| Sex, male | 0.40 | 0.23–0.69 | <0.001 | 0.39 | 0.22–0.67 | <0.001 |
| BMI, kg/m2 | 0.97 | 0.89–1.05 | 0.446 | |||
| DM | 1.30 | 0.73–2.34 | 0.377 | |||
| HTN | 1.11 | 0.55–2.27 | 0.768 | |||
| CAD | 1.53 | 0.61–3.83 | 0.366 | |||
| MELD score | 1.00 | 0.97–1.03 | 0.853 | |||
| Child–Pugh score | 1.02 | 0.91–1.15 | 0.692 | |||
| Child–Pugh classification | 0.915 | |||||
| A | ||||||
| B | 1.11 | 0.59–2.11 | 0.750 | |||
| C | 1.15 | 0.57–2.33 | 0.689 | |||
| HBV | 0.72 | 0.42–1.24 | 0.241 | 0.81 | 0.46–1.41 | 0.454 |
| HCV | 1.57 | 0.67–3.65 | 0.302 | |||
| Alcoholic liver cirrhosis | 0.55 | 0.22–1.38 | 0.207 | |||
| HCC | 0.78 | 0.46–1.34 | 0.374 | |||
| Toxic hepatitis | 1.59 | 0.22–11.44 | 0.646 | |||
| Retransplantation | 0.0000 | 0.962 | ||||
| Calcineurin inhibitor | ||||||
| Tacrolimus | 1.00 (Ref.) | |||||
| Cyclosporine | 0.48 | 0.15–1.52 | 0.213 | |||
| MMF | 1.60 | 0.76–3.38 | 0.221 | 1.87 | 0.88–3.98 | 0.106 |
| Steroid | 0.37 | 0.09–1.53 | 0.173 | |||
| ACR | 1.59 | 0.72–3.50 | 0.255 | |||
| GRWR | 1.55 | 0.54–4.46 | 0.417 | |||
| PLR | 1.00 | 1.00–1.01 | 0.444 | |||
| PNI | 0.98 | 0.95–1.02 | 0.365 | |||
| CRP/albumin | 1.12 | 0.81–1.55 | 0.500 | |||
| Total ischemic time, hour | 1.00 | 0.99–1.00 | 0.327 | |||
| Transfusion | 2.76 | 1.00–7.60 | 0.051 | 2.28 | 0.82–6.35 | 0.116 |
| C-Index (95%CI) | C-Index Difference (95% CI) | p-Value | IDI (95% CI) | p-Value | ||
|---|---|---|---|---|---|---|
| HZ | Model 1 * | 0.678 | ||||
| Model 2 † | 0.676 | −0.002 (-−0.014–0.010) | 0.712 | 0.008 (0.001–0.029) | 0.020 | |
| PHN | Model 1 * | 0.766 | ||||
| Model 2 † | 0.772 | 0.006 (-−0.012–0.024) | 0.523 | 0.015 (0.000–0.059) | 0.047 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sim, J.-H.; Moon, Y.-J.; Kim, S.-H.; Kim, K.-S.; Lee, J.-S.; Song, J.-G.; Hwang, G.-S. Association between Neutrophil-Lymphocyte Ratio and Herpes Zoster Infection in 1688 Living Donor Liver Transplantation Recipients at a Large Single Center. Biomedicines 2021, 9, 963. https://doi.org/10.3390/biomedicines9080963
Sim J-H, Moon Y-J, Kim S-H, Kim K-S, Lee J-S, Song J-G, Hwang G-S. Association between Neutrophil-Lymphocyte Ratio and Herpes Zoster Infection in 1688 Living Donor Liver Transplantation Recipients at a Large Single Center. Biomedicines. 2021; 9(8):963. https://doi.org/10.3390/biomedicines9080963
Chicago/Turabian StyleSim, Ji-Hoon, Young-Jin Moon, Sung-Hoon Kim, Kyoung-Sun Kim, Ju-Seung Lee, Jun-Gol Song, and Gyu-Sam Hwang. 2021. "Association between Neutrophil-Lymphocyte Ratio and Herpes Zoster Infection in 1688 Living Donor Liver Transplantation Recipients at a Large Single Center" Biomedicines 9, no. 8: 963. https://doi.org/10.3390/biomedicines9080963






