1. Introduction
The methanol-to-olefin (MTO) process [
1] transfers methanol to ethylene and propylene, and is an important way to produce olefin independently of petroleum. It can help to improve the stable supply of olefin in China [
2], as methanol can be derived from coal or natural gas. The MTO reaction was first proposed in the 1970s, with ZSM-5 as the catalyst [
3]. Currently, the primary catalyst used in the MTO process is SAPO-34, the total selectivity of ethylene and propylene can reach 80%, and their ratio is adjustable in the interval 0.5~1.5 [
4]. Three leading MTO technologies which have been successfully applied in industries [
5] are the DMTO developed by Dalian Institute of Chemical Physics (DICP), the SMTO by Sinopec Shanghai Research Institute of Petrochemical Technology, and the MTO technology developed by UOP/Norsk Hydro. The reaction and separation sections are the keys to these processes.
The conversion of methanol and the selectivity to olefin are two key parameters to evaluate the MTO reactor’s performance, and are expected to be as high as possible. However, the reactor parameters might have different influences on them. For example, multiple reactions are in progress, and their reaction rates increase along with the reactor’s temperature as does the conversion. In comparison, the variation trend of selectivity might differ depending on all reaction rates and their ratios. For the distillation columns of the separation section, lower energy consumption and capital cost are expected while ensuring the purification of olefin products. Many parameters, such as pressure, reflux ratio, and the number of stages, affect these costs. In addition, the utilities selected for condensers and reboilers directly affect the heat exchange areas and energy costs. The costs of heat exchangers, pumps, and compressors arranged between two adjacent columns are related to the neighboring columns’ feeds, products, and operating parameters. All of these columns and auxiliary devices are affected by the separation section’s feed and, thus, the reactor parameters. For the optimization of the MTO process, the reaction and separation sections should be considered together with the primary parameters optimized.
For the MTO reactors, the cracking of C
4+ was promoted in order to increase the production of ethylene and propylene by improving the reactor and optimizing the coke distribution on the catalyst [
6]. Moreover, much research on the MTO reaction’s mechanism has been carried out [
7], including the MTO reactions with the conversions between alkanes and alkenes [
8], the kinetic parameters, the relationships between temperature and reaction rates [
9], an eight-lumped kinetic model [
10] and a seven-lumped kinetic model [
11]. The kinetic mechanism models are complex, require much calculation for simulation and optimization, and sometimes cannot converge.
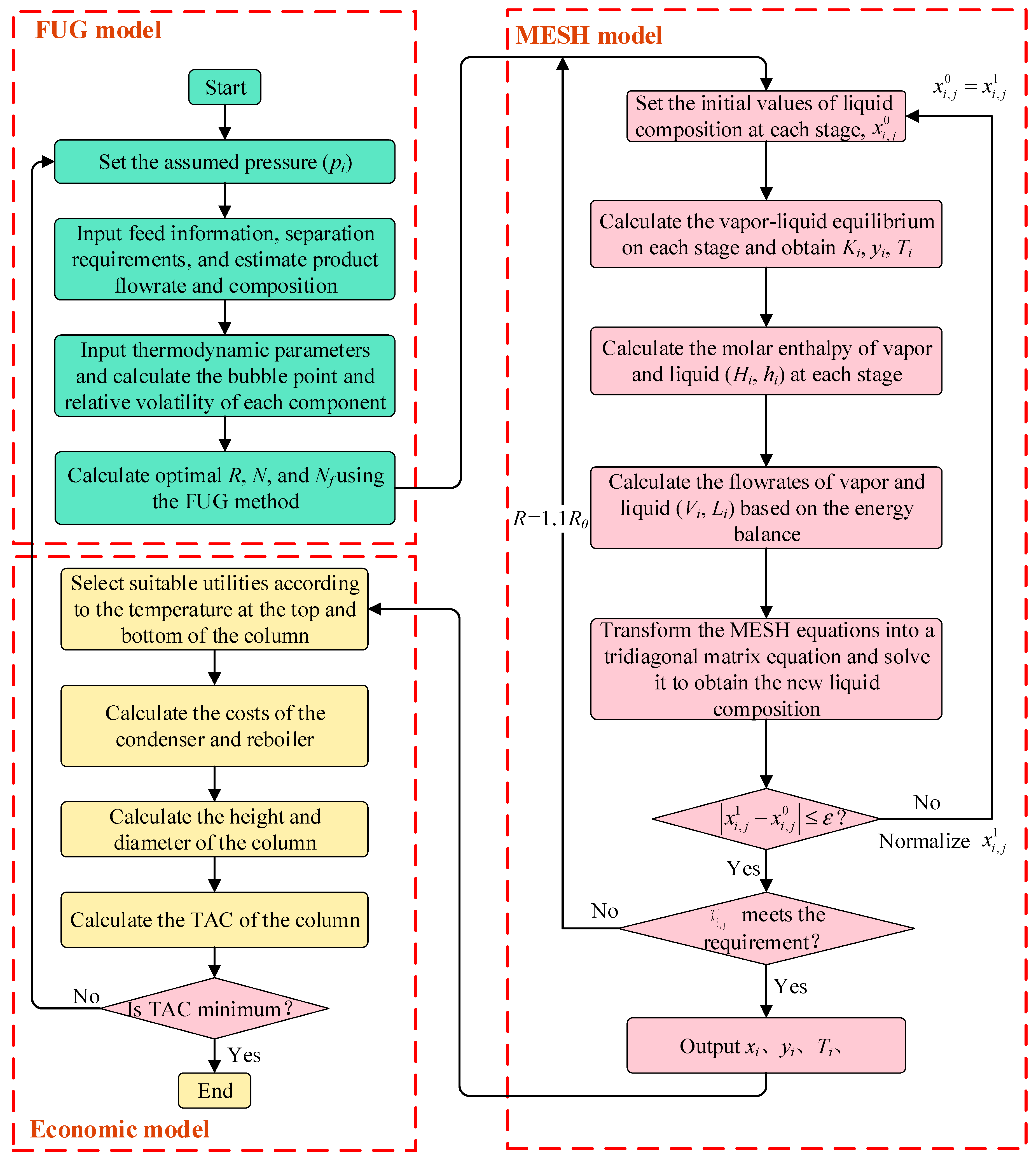
In the separation section, the optimization of distillation is of great significance for energy savings and pollution reductions, and can be achieved based on shortcut and rigorous methods. The Fenske–Underwood–Gilliland (FUG) method is well-known for its simplification and efficiency [
12], and is widely applied. Ye et al. [
13] extended the Underwood equation to columns with side streams and optimized the distillation sequence considering the condensers and reboilers. Cui [
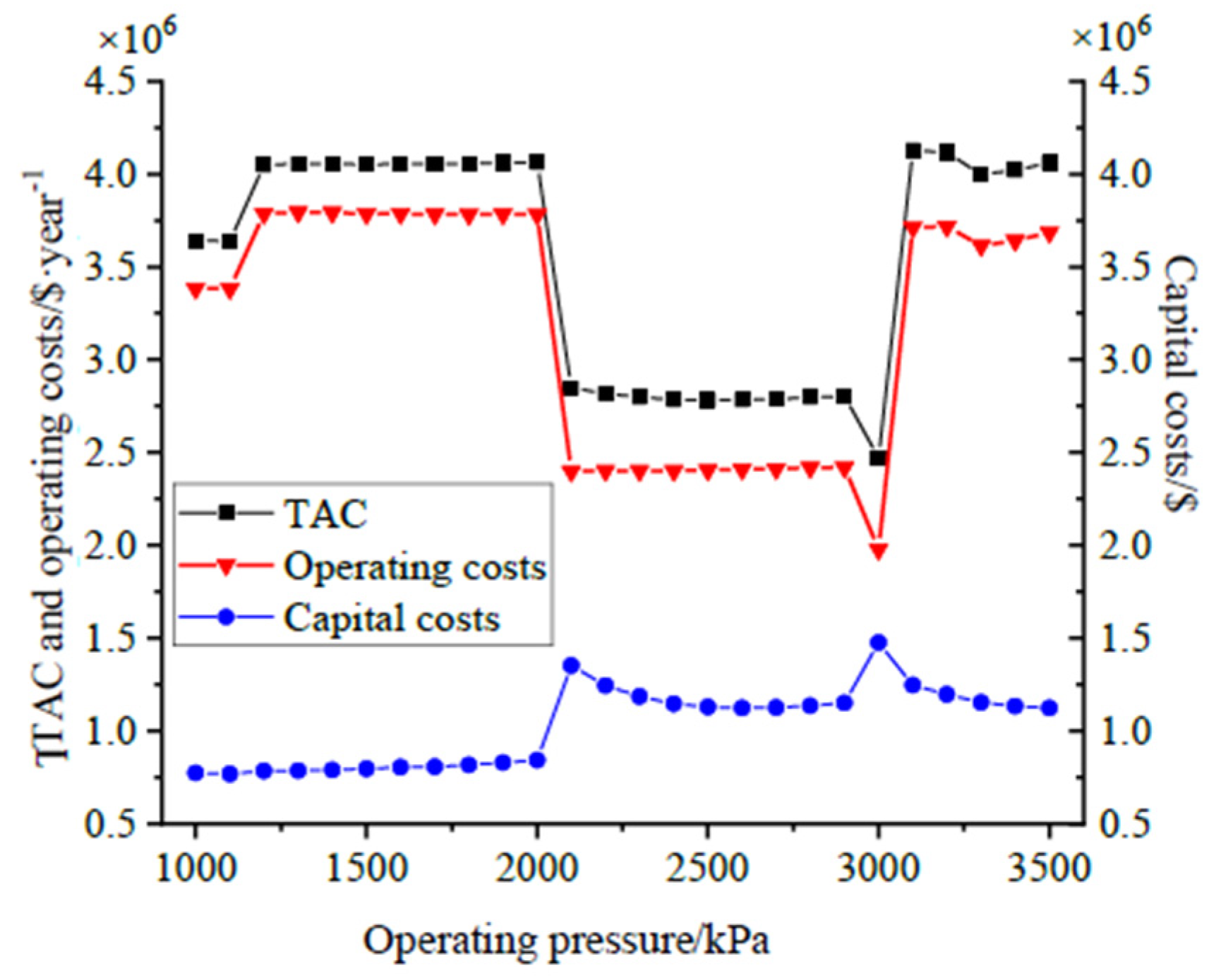
14] established the optimization procedure based on the FUG model and determined the operating pressure according to the minimum annual cost. For the thermally coupled reactive distillation, Gomez-Castro [
15] proposed a method to minimize the heat load of columns and illustrated its performance for targeting the optimal design. The boundary value method (BVM) was proposed by Fidkowski [
16] to design a distillation column and check the feasibility of the design. Lucia et al. [
17] proposed the shortest stripping line approach to target the minimum energy demand. However, shortcut models are usually based on the assumptions of constant molar overflow and constant relative volatilities, which might cause significant errors.
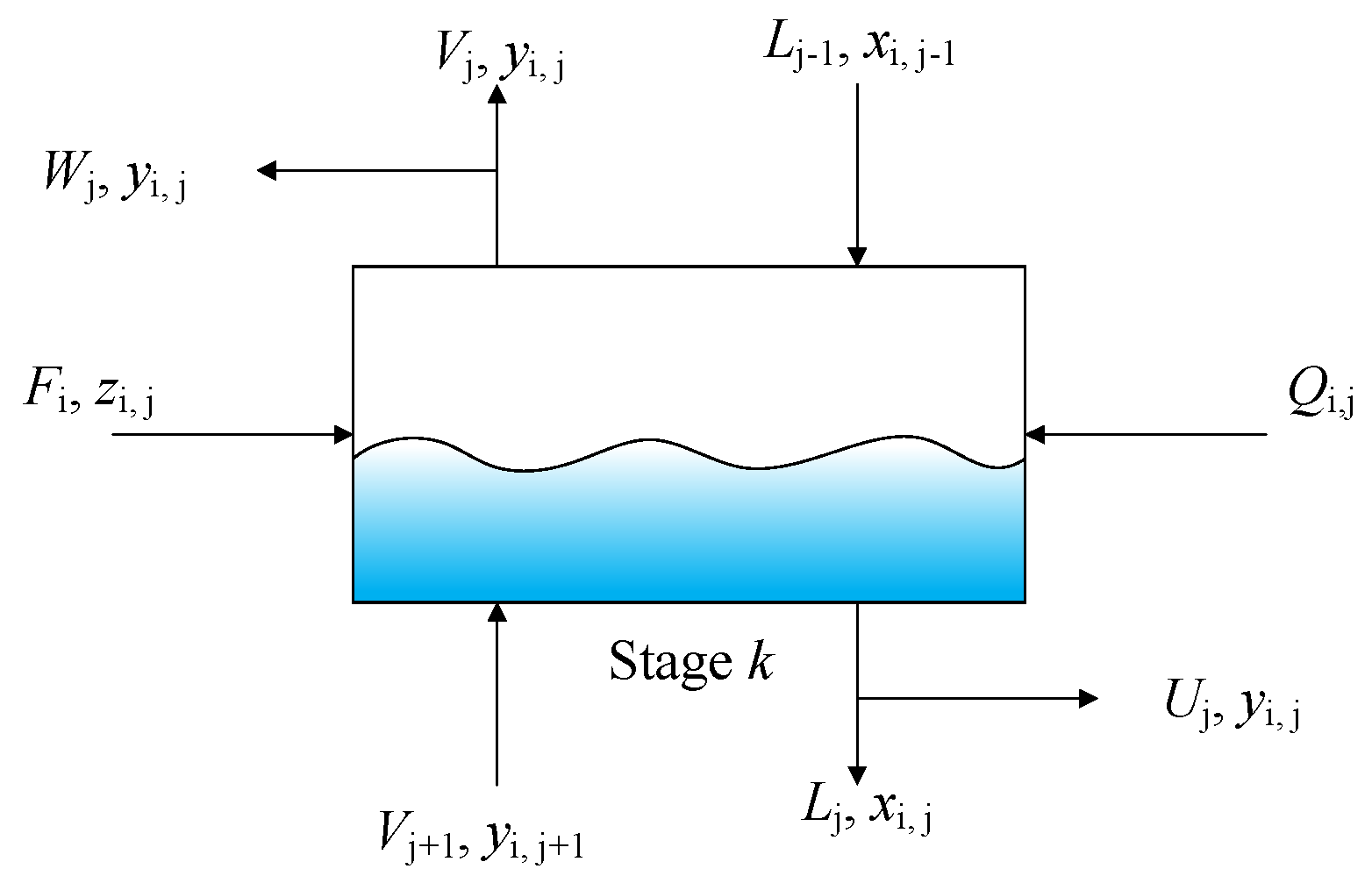
Rigorous models can identify accurate and detailed results for the distillation columns [
18]. They are based on material balances (M), equilibrium relationships (E), summation of compositions (S), and enthalpy balances (H), and are known as MESH equations. These equations have been embedded in commercial software, such as Aspen Plus and Unisim, for the design and sensitive analysis of distillation columns [
19]. However, distillation sequences with multiple columns can only be designed sequentially, and poor results might be obtained [
20].
All columns should be optimized simultaneously, considering their interactions. Viswanathan et al. [
21] used the mixed integer nonlinear programming (MINLP) method to optimize a distillation column with the number of theoretical stages as the decision variable. The MESH equations are used in the optimization model as the constraints, and other process parameters are taken as decision variables [
22]. The model includes thousands of equations and variables or more, and the challenges are concentrated on the initialization and convergence of the model. In some methods, the MESH equations are pre-solved to establish the mappings among different variables. Seidel et al. [
23] proposed an approach for infeasible path optimization of distillation-based flowsheets. Although this method can significantly reduce the number of decision variables, the difficulty lies in establishing an appropriate solution model.
Some research has been carried out for the optimization of the MTO process. Based on Aspen Plus software, Yu [
24] simulated the process, proposed an improved design with lower cost, and compared different methods of separating propane and propylene [
25]. Dimian [
26] studied the heat integration of the MTO process and provided an energy-efficient design. Chen et al. [
27] optimized the refrigeration cycle of the MTO process and proposed an alternative arrangement with better economic performance. Although these studies optimized the MTO processes to some extent, the interactions between the reaction and separation sections were left out of consideration.
Different methods and models have been developed to integrate the reaction and separation systems. Yin et al. [
28] proposed a method to automatically identify the optimal distillation sequences and generate the appropriate solutions according to different reaction parameters. Hentschel et al. [
29] combined the kinetic reaction model with the FUG model to minimize the total cost of the reaction–distillation process. Both methods can analyze and sort multiple alternative schemes efficiently. Kong et al. [
30] proposed an optimization framework for biofuel production based on the superstructure model for synthesizing the process and allocating utilities simultaneously. Based on the generalized model description, critical characteristic identification, and model integration, Ryu et al. [
31] proposed a general model for simultaneously optimizing the reaction, separation, and heat exchanger network. Although the MINLP model was generally used to optimize the reaction–separation system [
32], it is not easy to solve and target its global optimum. In addition, stochastic optimization algorithms can be used to solve large-scale MINLP problems efficiently, such as genetic algorithm [
33], simulated annealing algorithm [
34], and particle swarm optimization [
35]. Among these, the genetic algorithm is widely used for optimizing complex systems.
Although some researchers have studied the optimization of the MTO process and the integration of the reaction and separation systems, there are no reports about the systematic integration of the reaction and separation sections in the available literature. In the MTO reactor, coke is formed at high temperatures. It attaches to the catalyst’s surface and affects its activity, hence the reactor’s conversion and selectivity, the separation section, and the energy consumption. Among the parameters affecting the total cost of distillation columns, the pressure is significantly important, as it affects components’ activity, the reflux ratio, and the number of stages.
This work develops an efficient method for integrating the reaction and separation sections considering the auxiliary devices. The reactor model will be established based on the lumped model, and the column will be modeled with the combined shortcut and the rigorous model. A procedure based on the genetic algorithm will be proposed to solve the model efficiently. This manuscript is organized as follows:
Section 2 will analyze the interaction between the reaction and distillation sections of the MTO process; in
Section 3, the building of the optimization model is described, considering the optimization of the catalyst’s coke content, reaction temperature, each column’s operating pressure, and the allocation of utilities, etc. In
Section 4, a case is studied to illustrate the application of the proposed method. The proposed method is summarized in the Conclusions section.
2. Interaction between the Reaction and Distillation Sections
MTO Process
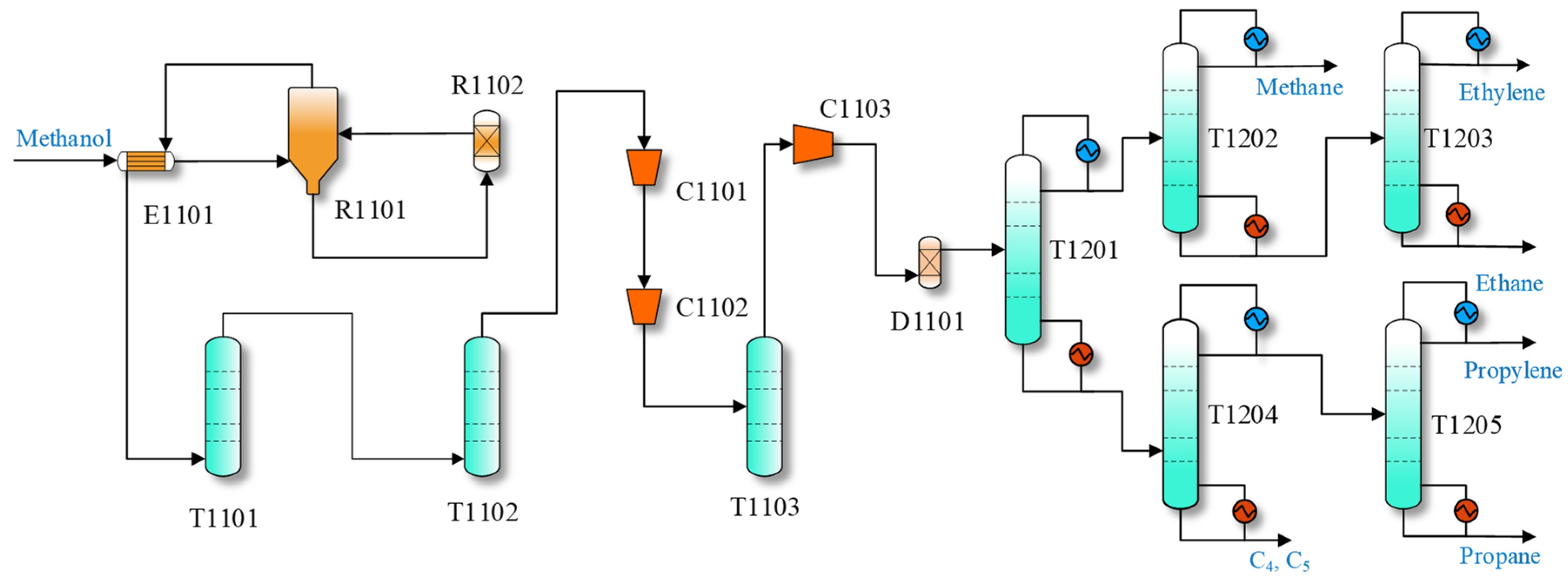
The general MTO process with a front-end dethanizer is shown in
Figure 1. In this process, the fluidized bed reactor (R1101) is the core of the entire process. The methanol, preheated to 300 °C, reacts quickly in the reactor, generating a significant amount of heat. Some coke will form and deposit on the catalysts’ surface and cause the catalyst’s deactivation. The deactivated catalyst is regenerated in regenerator R1102 to remove the coke. After preheating the methanol, the reactor effluent is sent to a quench column (T1101) and scrubber (T1102) to decrease its temperature and remove the water and catalyst. The high-temperature gas countercurrent makes contact with the water in column T1101. The gas product exiting the scrubber (T1102) mainly consists of low-carbon olefins (C
1–C
5).
The gas is compressed by compressors (C1101, C1102) in the separation section and then fed to the alkali column (T1103) to remove the oxides and acids. Then, it passes through the compressor (C1103), dryer (D1101), and inlets into the distillation sequence with five columns. The dethanizer (T1201) performs the split between C2 and C3. Its top product is separated in the demethanizer (T1202) and ethylene column (T1203) to remove C1 and ethane, respectively, and pure ethylene is obtained at the top of T1203. The bottom product of T1201 is fed to the depropanizer (T1204) to separate C3 from the top, which is further separated by the propylene column (T1205) to obtain pure propylene. The bottom product of T1204 consists of C4 and C5, and is sent out of the separating section.
In this process, the reactor effluent is separated in the separation section. The reactor parameters, such as feed composition, temperature, catalyst, etc., affect the product, operation, and energy consumption of distillation columns. Optimizing the reaction and separation sections together is necessary to reduce energy consumption.
In the reactor, methanol conversion and the selectivity to olefin are expected to be as high as possible. However, different parameters might have different influences on them. For example, the reaction rates and conversion increase along with the reactor’s temperature. The selectivity to olefin depends on each reaction rate and their ratios, and its variation might be different. Furthermore, coke is formed at high temperatures, attaches to the catalyst’s surface, and affects the catalyst’s activity as well as the selectivity of desired products. Therefore, the reactor temperature (T) and coke content of the catalyst (CC) are the key parameters influencing the products’ flowrates and compositions. In the separation section, parameters such as the operating pressure, reflux ratio, and number of stages affect the total cost. The utilities selected for condensers and reboilers directly affect the heat exchange area and energy costs. For units arranged between two adjacent columns, such as heat exchangers, pumps, and valves, their expenses are related to the neighboring columns. These columns and auxiliary devices are affected by the feed of the separation section and, thus, the reactor parameters.
In this work, an efficient method is developed to integrate the reaction and separation sections. The reactor temperature (T) and the coke content of the catalyst (CC) will be optimized together with the parameters of all distillation columns, considering the auxiliary devices arranged between two adjacent columns, such as heat exchangers, pumps, and valves. Both the energy cost and capital cost will be considered in the optimization. An MTO process is optimized based on the proposed method.