1. Introduction
Despite the expected increase in the share of renewable energy (wind, solar, geothermal, and tidal energy) from 2.7% in 2022 to 11.7% in 2045 in the world demand by fuel type, petroleum is (31.2% in 2022) and is expected to remain (29.5% in 2045) the major fuel satisfying mankind energy demand [
1]. The petroleum quest is expected to increase from 99.6 in 2022 to 116 million barrels per day in 2045 with the anticipated enhancement of petroleum-based fuel consumption in the transportation sector by 8.7 million barrels per day in 2045 relative to 2022 [
1]. Therefore, petroleum-based fuels which are the main source driving our vehicles today are also expected to do this during the next couple of decades, regardless of developments and innovations in the electric vehicle sector and biofuel technologies [
2,
3,
4,
5]. The efficiency of modern petroleum refining, the major fuel supplier, is strongly dependent on the performance of bottom-of-the-barrel conversion processes and the utilization of their products [
6]. The transformation of low-value petroleum residue into high-value automotive fuels provides a high refining margin due to the downward trend in heavy oil product demand and the high demand for aviation and diesel engine fuels [
7]. Among the residue conversion technologies, slurry hydrocracking and ebullated bed hydrocracking attain the highest conversion levels [
8,
9,
10]. The residue hydrocracking decreases the manufacturing of heavy oil products at the expense of magnification of the production of naphtha, middle distillate, and gas oils. While the middle distillates from the residue hydrocracking unit after hydrotreatment are directly blended in the finished automotive diesel pool, the gas oils are mostly catalytically cracked, and the naphtha is difficult to market due to their low octane, high nitrogen content, and low chemical stability. After the commissioning of H-Oil ebullated bed vacuum residue hydrocracking in the “LUKOIL Neftohim Burgas” (LNB) refinery in 2015, the utilization of H-Oil naphtha became an issue. Although its sulphur content was not very high (between 200 and 700 ppm), its lower Saybolt Color Numbers according to ASTM D 156 standard [
11] (lower than 20), made it difficult to market. For that reason, it was hydrotreated before being marketed. The possible options for H-Oil naphtha utilization are: (1) using it as a feedstock to produce low molecular weight alkenes—pyrolysis; (2) as catalytic reforming feedstock; (3) as steam reformer raw material. Pang et al. [
12] reported that during the hydrocracking of deasphalted oils from Arabian crude the major compounds in hydrocracked naphtha, whose yield varied between 0 and 17.1% depending on reaction severity, were mainly C
6–C
10 alkanes and alkenes and some aromatic hydrocarbons. The predominance of alkane hydrocarbons in the residual hydrocracked naphtha suggests that it could be suitable for use as a feedstock for steam cracking [
13]. The application of H-Oil naphtha as reformer feedstock is hampered due to the high nitrogen content that is a poison for reformer catalysts, and its removal requires specially dedicated hydrotreating units having higher hydrodenitrogenation (HDN) capabilities [
14]. The use of H-Oil naphtha as a steam reformer feedstock needs the reformer to have a pre-reformer unit to avoid excessive coking in reformer furnace tubes [
15]. However, the alkenes it contains can accelerate pre-reformer catalyst deactivation due to increased coke accumulation on the catalyst [
16]. Our literature search for the utilization of residue hydrocracking naphtha showed very scarce data. Ershov et al. [
17] proposed a technology to produce high-octane gasoline by blending low-octane hydrocracked naphtha with 30% bioethanol. Most published research to utilize residue conversion naphtha is related to the treatment of visbreaker and coker naphthas [
18,
19,
20,
21,
22,
23,
24,
25,
26,
27,
28]. In the process of searching for alternative more profitable options for H-Oil naphtha utilization commercial tests were carried out at the fluid catalytic cracking unit, and the Prime-G+ catalytic cracking gasoline hydrotreatment unit in the LNB refinery. The results of these tests are discussed in this research.
This paper aims to summarize the experience gained in the LNB refinery with the processing of H-Oil naphtha in naphtha hydrotreater, Prime-G+ FCC gasoline desulphurizer, and fluid catalytic cracker, and discern the most efficient way of H-Oil naphtha utilization.
2. Materials and Methods
The bulk properties of H-Oil naphtha employed in this study are summarized in
Table 1.
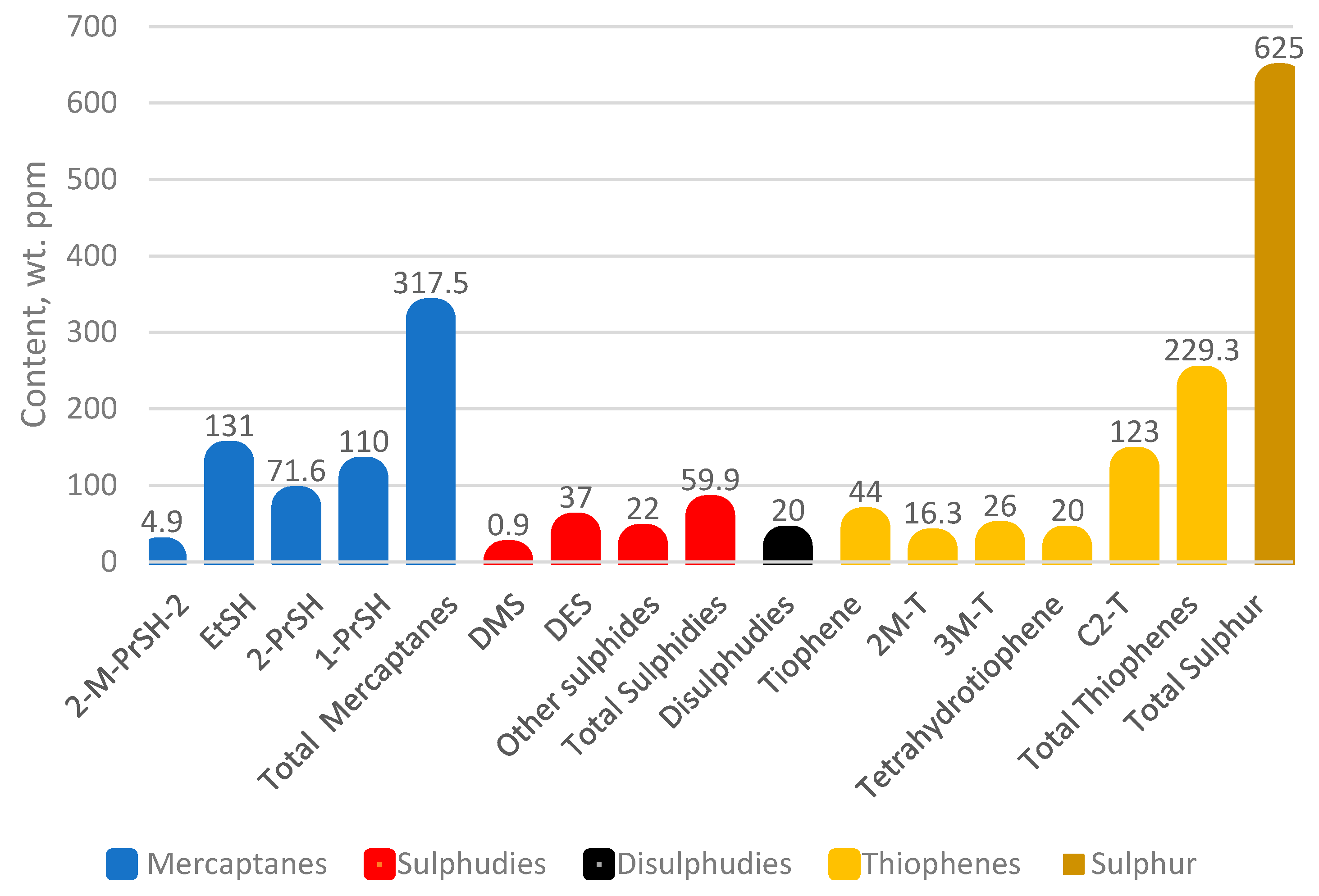
Figure 1 presents a graph of the distribution of sulphur compounds in H-Oil naphtha.
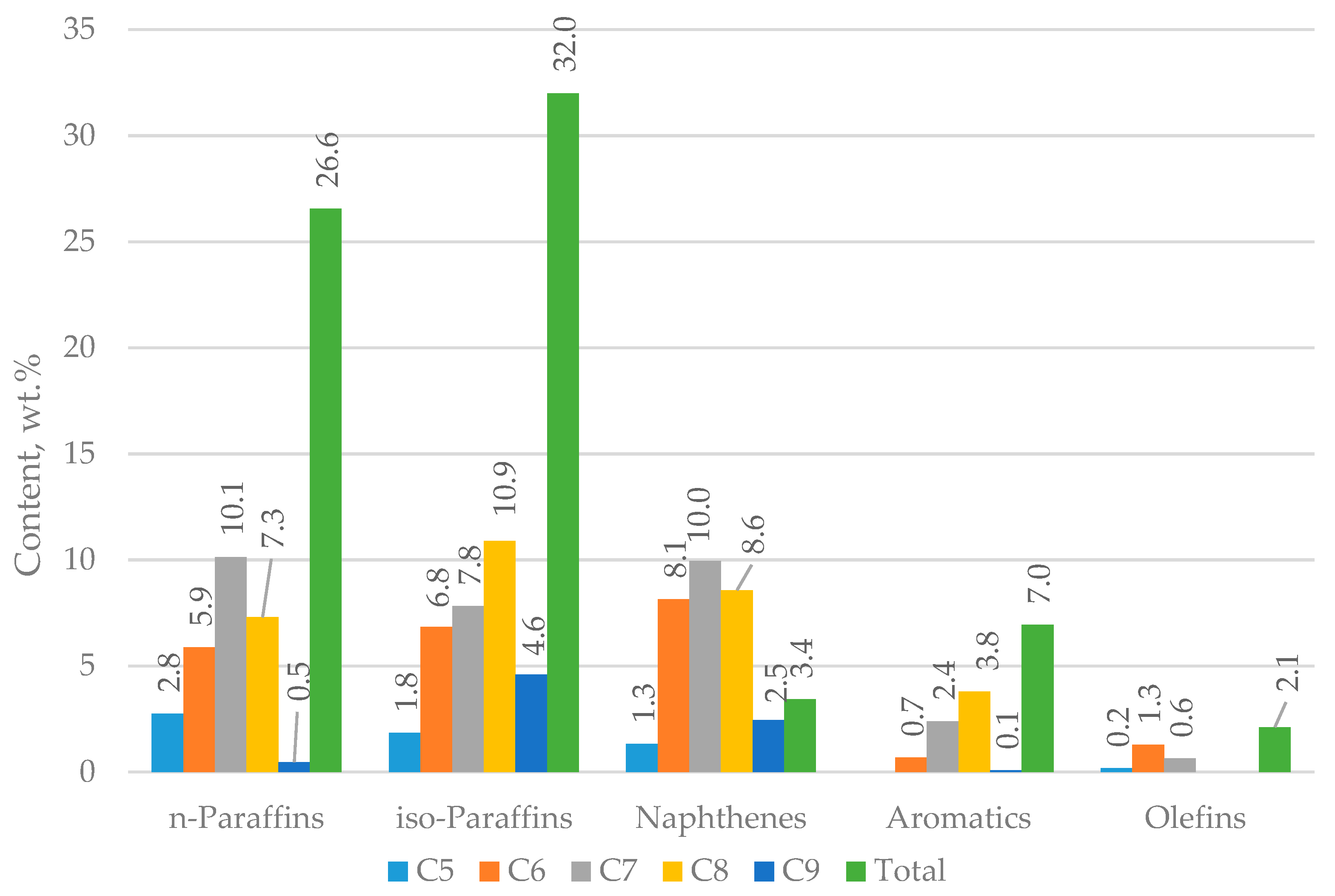
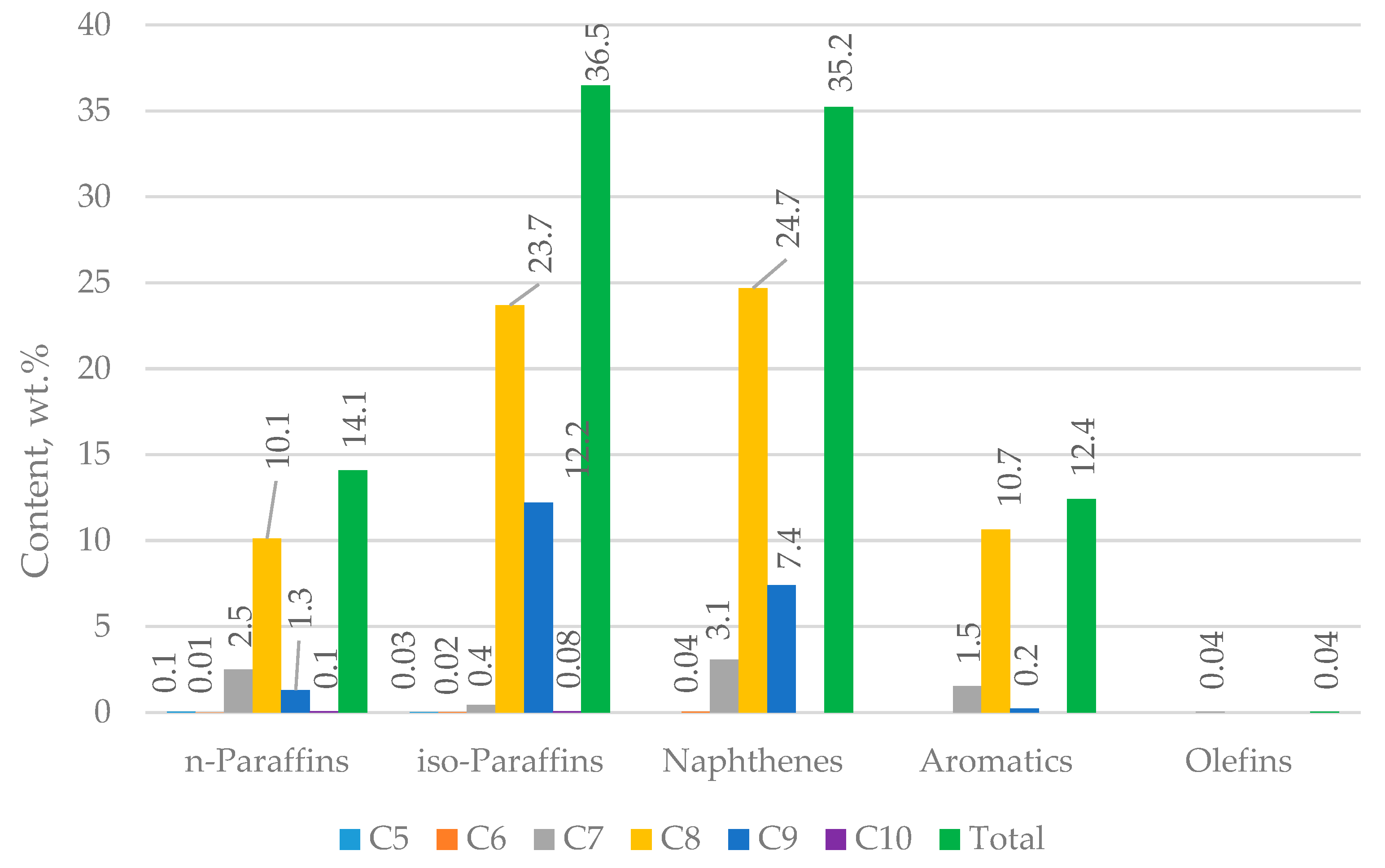
Figure 2 shows the distribution of hydrocarbons in H-Oil naphtha measured by PIANO (n-paraffins, iso-paraffins, aromatics, naphtenes, olefins) analysis.
The density of H-Oil naphtha was measured following the requirements of the standard ASTM D 4052 [
30]. The sulphur content of H-Oil naphtha was determined by an energy dispersive X-ray fluorescent spectrometer (EDXRF) analyzer following the requirements of the standard ASTM D 4294 [
31]. Maleic anhydride value (MAV) expressed in milligrams of maleic anhydride reacted per gram of sample was determined using the standard IFP 9407 method. It determines the conjugated diolefins content based on the reaction of conjugated dienes with maleic anhydride in accordance with the Diels-Alder reaction. The IFP 9407 method is similar to the standardized 326-82UOP [
32] method, which provides the DV (diene value), the two values being linked by the relationship MAV = 3.86 DV [
33].
Standard method ASTM D5134 [
34] was applied to determine H-Oil naphtha hydrocarbon composition. The gas chromatographic system consists of a gas chromatograph Hewlett Packard 5890 Series II equipped with a flame ionization detector (Agilent Technologies, Inc., Santa Clara, CA, USA). A PIONA capillary column with 50 m length, 0.20 mm diameter, and 0.5 μm coating thickness (100% dimethylpolysiloxane) (Agilent J&W, Santa Clara, CA, USA) was employed. The injector and the detector temperatures were 200 °C and 250 °C, respectively. The oven temperature program was as follows: initial temperature 35 °C, without isotherm, a ramp at rate 2 °C/min to final temperature 200 °C, and final time 60 min. As a carrier gas hydrogen was used with a flow rate of 1.1 mL/min. Column inlet pressure and a split ratio were 150 kPa and 100:1, respectively.
Sulphur species distribution in the H-Oil naphtha was measured following the requirements of the standard ASTM D5623 [
35]. The gas chromatographic system model 7890A employed was equipped with 355 sulfur chemiluminescence (Agilent Technologies, Inc., USA) and HP-1 capillary column with 30 m length, 0.32 mm diameter and 4.0 μm coating thickness (100% dimethylpolysiloxane) (Agilent J&W). The injector temperature was 240 °C. The oven temperature program was as follows: initial temperature 50 °C, 4 min isotherm, first ramp at rate 20 °C/min to final temperature 120 °C, and final time 4 min, second ramp at rate 10 °C/min to final temperature 220 °C, and final time 30 min. Helium was used as a carrier gas with a flow rate of 2.6 mL/min. Column inlet split ratio was 50:1. The 355 sulfur chemiluminescence detector parameters were set as follows: burner temperature 800 °C, vacuum of burner 370 torr, vacuum of reaction cell 7 torr, hydrogen 40 mL/min, air 60 mL/min.
The research octane number (RON) of H-Oil naphtha, and gasoline samples studied in this work was measured according to the requirements of ASTM D 2699 [
36], while the motor octane number (MON) was measured according to the requirements of ASTM D 2700 [
37].
The processing of H-Oil naphtha during the study was carried out at the LNB FCCU and Prime G FCC gasoline hydrotreatment. A process scheme of the LNB FCC unit is presented in [
38]. The catalysts used in the FCC unit during the study were octane barrel main catalyst and ZSM-5 containing catalyst additive, whose properties are summarized in
Table 2.
The conversion of typical FCC feedstock—vacuum gas oil is determined using Equation (1).
where,
FCC feed conversion = conversion of feed material boiling above 210 °C into material boiling below 210 °C, wt.%;
FCC feed210°C+ = Flow rate of FCC feed material boiling above 210 °C+, t/h. (This is the flow rate of vacuum gas oil feedstock);
FCC product210°C+ = Flow rate of FCC product material boiling above 210 °C+, t/h. (This is the sum of flow rates of products light cycle oil (LCO), heavy cycle oil (HCO), and slurry oil (SLO)).
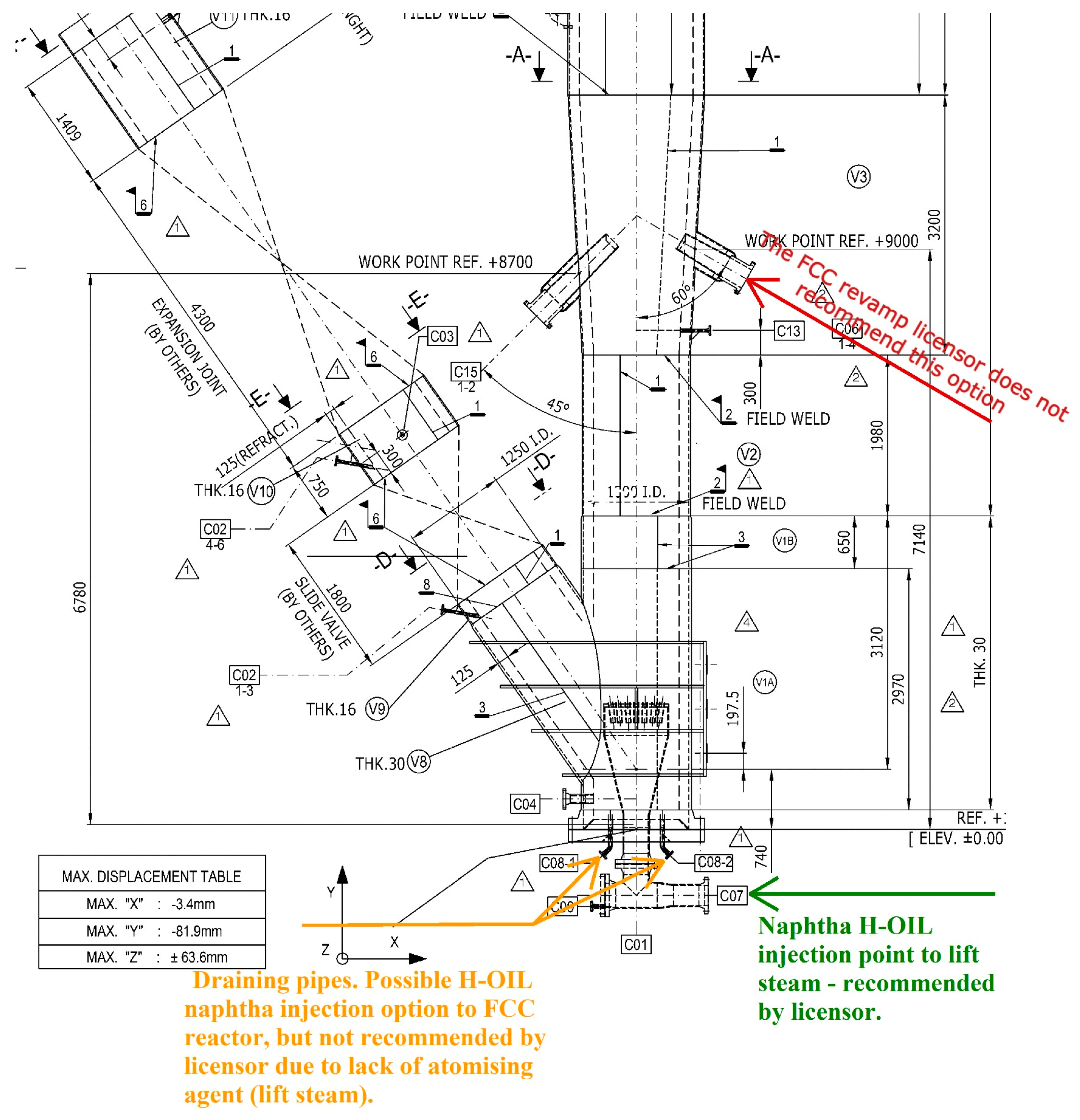
The H-Oil naphtha after consideration was decided to feed the FCC riser not in the feed nozzles because of the risk their damage but in the line of lift steam as shown in
Figure 3 and
Figure S1.
The conversion of H-Oil naphtha in the FCC process was calculated using Equation (2).
where,
FCC HOil Naphtha conversion = Conversion of H-Oil naphtha in dry gas, C3 fraction, and C4 fraction, wt.% of H-Oil naphtha processed in FCC unit;
ΔDry gas = A difference between the amount of dry gas produced from the vacuum gas oil catalytic cracking and that produced from the mixture vacuum gas oil/H-Oil naphtha at the same quantity of vacuum gas oil, t/h.
ΔC3 = A difference between the amount of C3 fraction produced from the vacuum gas oil catalytic cracking and that produced from the mixture of vacuum gas oil/H-Oil naphtha at the same quantity of vacuum gas oil, t/h;
ΔC4 = A difference between the amount of C4 fraction produced from the vacuum gas oil catalytic cracking and that produced from the mixture of vacuum gas oil/H-Oil naphtha at the same quantity of vacuum gas oil, t/h.
HOil Naphtha = Amount of H-Oil naphtha cracked in the FCC unit along with the vacuum gas oil, t/h
A diagram of the Prime-G+ process unit of the LNB refinery is presented in [
39], while the different processing schemes of Prime-G+ technology are exemplified in [
40]. This is a technology licensed by Axens and it is broadly employed to desulphurize FCC gasoline, with over 300 licensed units all over the world [
40,
41]. For more information about the Prime-G+ process unit and its operation, the reader can refer to [
39,
40,
41]. The catalysts used in the selective hydrogenation reactor (SHU), and in the hydrotreatment reactor are part of a license agreement and their properties cannot be shown due to confidential reasons.
3. Results
The H-Oil naphtha is a chemically unstable product and its appearance as evident from
Figure 4 changes with time. Our attempt to stabilize it by using the synthetic antioxidant additive—BHT (2,6-bis(1,1-dimethylethyl)-4-methylphenol), typically employed to improve catalytic cracking gasoline oxidation stability as reported in [
42] failed. For that reason, to make this product marketable, it was hydrotreated along with straight-run naphtha in a naphtha hydrotreater in the LUKOIL Neftohim Burgas (LNB) refinery. After hydrotreatment of the blend of 27% H-Oil naphtha/73% straight run naphtha in HDS-1 LNB hydrotreating unit on Co-Mo commercial catalyst (TK 576 Brim) at a reaction temperature of 305 °C, a total pressure of 2.4 MPa, hydrogen/oil ratio of 22.5 Nm
3/m
3, and liquid hourly space velocity (LHSV) of 2.5 h
−1 the bromine number of the feed mixture dropped from 2.0 to 0.05 g Br
2/100 g oil, and the hydrotreated product remained chemically stable for a long period. The sulphur level of the hydrotreated product was decreased from 270 ppm in the blend naphtha feed to 3 ppm (98.9% hydrodesulphurization extent). Although the octane characteristics of the hydrotreated blend naphtha product were low (RON of 64, and MON of 62) it was used not only as a finished product to export but also as a blending component for automotive gasoline production. To explore the feasibility of H-Oil naphtha to be used as a feed component for a naphtha catalytic reformer it was fractionated in a laboratory TBP apparatus operating under requirements of ASTM D 2892 standard [
43] into light and heavy H-Oil naphtha fractions. As evident from the data in
Figure 1 the characteristic number N + 2A (N is naphthene content, while A is aromatic content in naphtha) used to qualify the naphtha catalytic reformer feed [
44] of the whole H-Oil naphtha is very low (17.4) that makes it unsuitable for catalytic reforming. The fractionation of H-Oil naphtha into light (initial boiling point (IBP)—110 °C,
Figure 5), and heavy (110 °C—final boiling point (FBP),
Figure 6) products, however, as obvious from the data in
Figure 6 provides a good stream for catalytic reformer feed from a hydrocarbon composition point of view (N + 2A = 59.4). The nitrogen content of the heavy H-Oil naphtha, however, is very high (188 ppm) which makes it an inappropriate feed for reforming.
For comparison purposes only the straight-run heavy naphtha used as a feedstock for the LNB catalytic reformer unit has a nitrogen content of 0.3 ppm. The maximum nitrogen content in naphtha feed that can be tolerated by the reformer catalyst is 3 ppm, which means that not more than 1.5% heavy H-Oil naphtha in the reformer feed is possible. The actual heavy H-Oil naphtha quantity amounts to about 10% of reformer feed, and thus the greater amount of heavy H-Oil naphtha will remain untreated.
The other option not investigated yet was to process the H-Oil naphtha along with the FCC gasoline in the Prime-G+ FCC gasoline post-treatment unit. The Prime-G+ technology selectively removes FCC full-range gasoline sulphur while assuring minimal octane loss [
39,
40,
41].
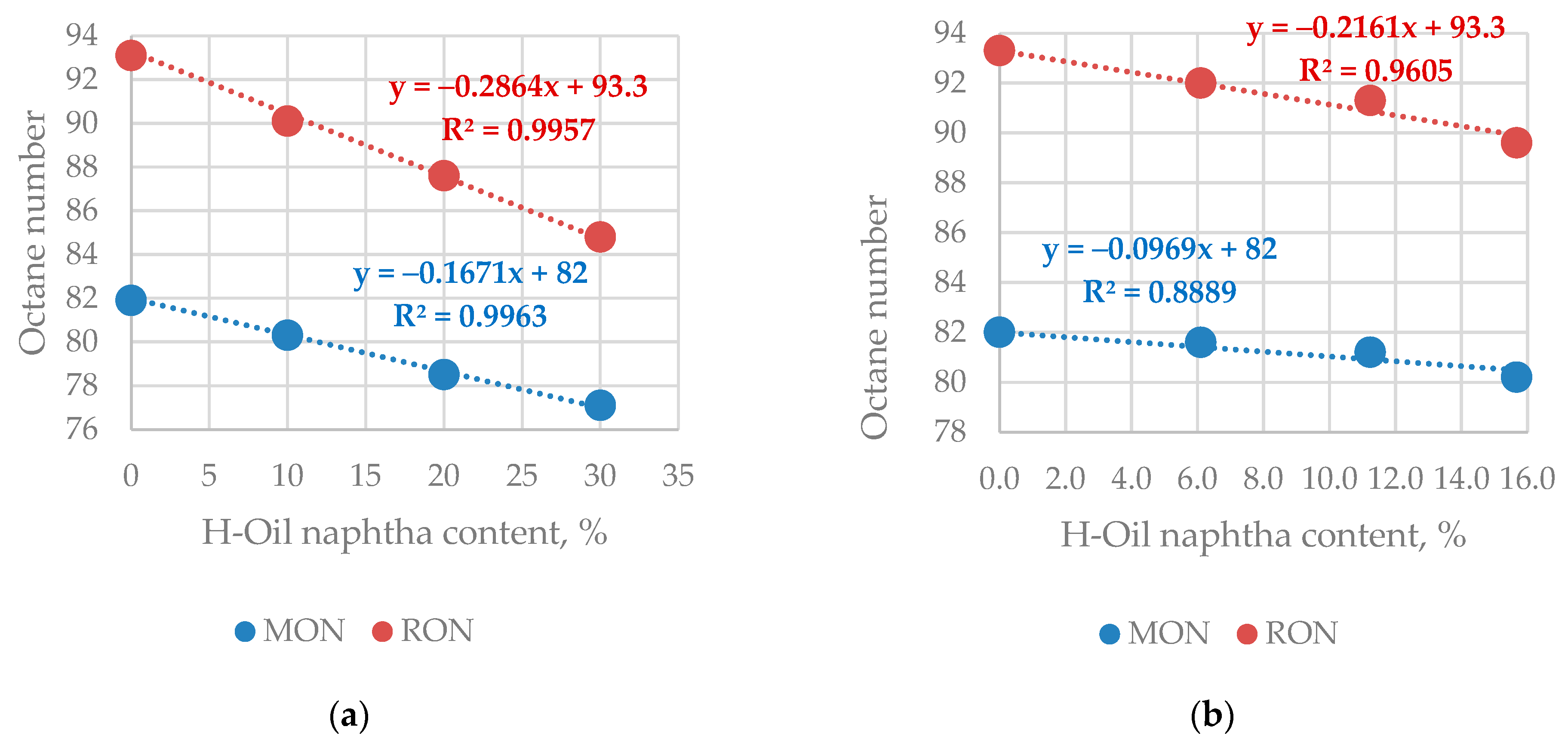
Figure 7 presents the dependence of RON, and MON of the blends Prime-G+ hydrotreated FCC gasoline—H-Oil naphtha (
Figure 7a), and Prime-G+ hydrotreated mixture FCC gasoline/H-Oil naphtha (
Figure 7b).
The interesting observation, in this case, is that the octane numbers of the blend FCC gasoline—H-Oil naphtha fall steeper than those treated in the Prime-G+ FCC gasoline—H-Oil naphtha mixtures. This finding suggests that the co-processing of H-Oil naphtha with FCC gasoline in the Prime-G+ unit enhances the blending octane number of H-Oil naphtha. The blending octane numbers of the H-Oil naphtha calculated by Equation (3) [
45] for the blends FCC gasoline—H-Oil naphtha was 64.7 RON, and 65.6 MON, while for those treated in the Prime-G+ FCC gasoline—H-Oil naphtha mixtures they were 72.4 RON, and 73.6 MON. The reason for the higher blending number of H-Oil naphtha co-processed with FCC gasoline in the Prime-G+ hydrotreater is difficult to find with the available in this study data. Additional investigation is needed to explain this phenomenon.
This data clearly indicates that the treatment of H-Oil naphtha along with FCC gasoline in the Prime-G+ unit improves its blending octane numbers by 8 points.
Table 3 summarizes the results from the commercial test performed at the LNB FCC unit with and without processing of H-Oil naphtha in the absence, and presence of ZSM-5 containing catalyst additive.
The data in
Table 3 (Cases 1, and 2) indicates that in processing of H-Oil naphtha during the first stage of the commercial test the VGO conversion incremented by 1.2%. Bearing in mind that the difference in VGO conversion at constant operating conditions observed at the commercial FCC unit in LNB refinery can vary by 0.8 wt.% [
8], and the higher content of H-Oil VGO in Case 2 (2 wt.% higher), which is more refractive than the straight run VGO [
8] it is very difficult to categorically conclude that H-Oil naphtha has any appreciable effect on VGO conversion. The estimated H-Oil naphtha conversion by Equation (2) shows the figure of 50.2 wt.%. Thus, half of the H-Oil naphtha is converted to dry gas, C3, and C4 fractions, and the other half remains in the FCC gasoline. As a result, the RON of FCC gasoline decreased by 0.4 points (see
Table 3), while MON remained unchanged. Considering that in the case of H-Oil naphtha (Case 2 from
Table 3), 50% of it is converted to dry gas and liquid petroleum gas (LPG), and 50% left in FCC gasoline, the content of H-Oil naphtha in FCC gasoline should be 2.4%. Using equation 3 the blending RON and MON of H-Oil naphtha treated in FCC unit equals 77.9, and 82.2, respectively. These findings show that the blending octane numbers of H-Oil naphtha processed in the FCC unit are the highest among those of its direct blending with Prime-G+ FCC hydrotreated gasoline, and its co-processing with FCC gasoline in Prime-G+ (see
Table 4).
The data in
Table 3 (Cases 3, and 4) displays that the use of ZSM-5 containing catalyst additive in the amount of 3% of catalyst inventory decreased the yield of gasoline by 1.6% at the expense of higher yields of C4 (+0.8 wt.%), C3 (+0.7 wt.%), and dry gas (+0.2 wt.%). The RON of gasoline grew by 0.5 points, and MON remained unchanged (Cases 3, and 4 from
Table 3). The data in
Table 3 also shows that an interruption of H-Oil naphtha processing in the FCC unit during employment of ZSM-5 containing catalyst additive when H-Oil VGO content in the FCC feed is the same (17 wt.%) the registered difference in VGO conversion of 0.3 wt.% which is within the already discussed earlier limit of 0.8 wt.% proves that the H-Oil naphtha has no impact on VGO conversion (Cases 4, and 5 from
Table 3).
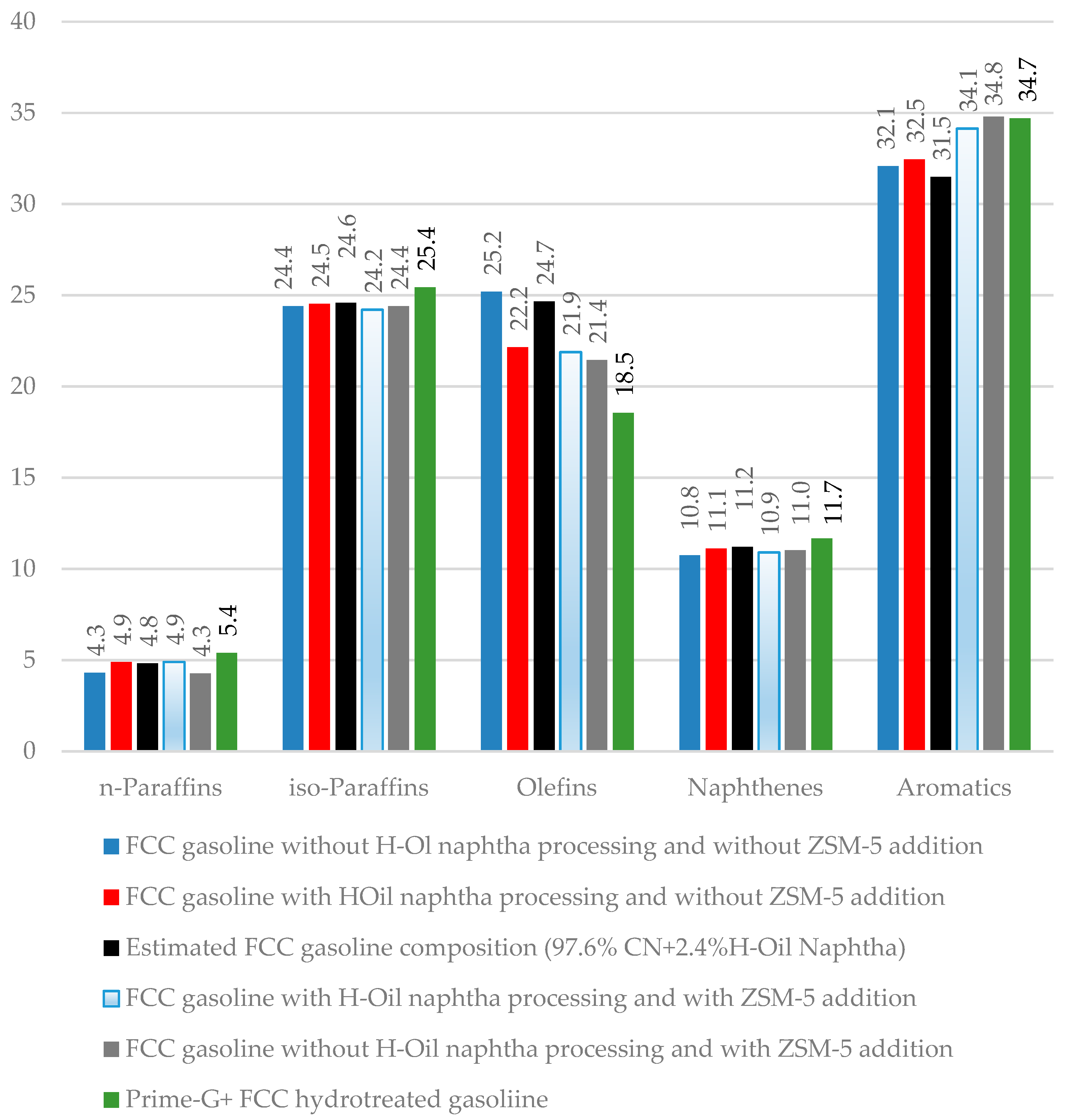
Figure 8 presents data about the hydrocarbon group composition of samples of gasoline taken from the FCC unit, and Prime-G+ unit during the commercial test with and without processing of H-Oil naphtha in the FCC unit with and without using ZSM-5 containing catalyst additive.
Figure 8 contains data of estimated group hydrocarbon composition of gasoline that consists of 97.6% FCC gasoline, and 2.4% H-Oil naphtha (black cluster in
Figure 8) using the data for FCC gasoline composition without processing of H-Oil naphtha (blue cluster in
Figure 8) and the data for H-Oil naphtha composition (
Figure 2). Comparing the measured and estimated group hydrocarbon composition of FCC gasoline sampled during processing H-Oil naphtha (red and black clusters in
Figure 8) one can see the existence of a significant difference in olefin content (2.5 wt.% lower olefins in measured versus estimated). The H-Oil naphtha conversion of 50.2% almost coincides with the number obtained by multiplication of 2.5 wt.% by the yield of FCC gasoline (47.8 wt.%, see
Table 3) and division of it by the percent of H-Oil naphtha in FCC feed (2.3%), that is 50.9%. Thus, we may suggest that the gasoline olefin cracking is the main contributor to H-Oil naphtha conversion.
Comparing the data of hydrocarbon composition of FCC gasoline samples taken with and without employment of the ZSM 5 containing catalyst additive with and without processing of H-Oil naphtha in FCC unit (light blue with red clusters, and grey with blue clusters in
Figure 8) one can see that the only difference for the case with H-Oil naphtha processing (red and light blue clusters) is the content of aromatics being 1.6% higher when ZSM-5 additive was used. Not the same difference in hydrocarbon composition of the gasoline samples taken when no H-Oil naphtha was processed in the FCC unit with and without using the ZSM-5 containing catalyst additive (grey and blue clusters in
Figure 8) was observed. In that case, a significant reduction in olefin content (−3.8 wt.%) at the expense of aromatic content enhancement (+2.7 wt.%) was registered. The decrease in olefin content implies that mainly gasoline olefins are cracked in the presence of 3% ZSM-5 containing additive in catalyst inventory. The data in
Figures S2–S6 shows that during the use of ZSM-5 containing additive without processing H-Oil naphtha the content of C
5 paraffin in FCC gasoline without processing of H-Oil naphtha increases, that of C
6 paraffin remains unaltered, while the content of C
7, and C
8 paraffin drops. The content of C
9+ paraffin does not change. As far as olefin content is concerned, for the case without H-Oil naphtha processing during employment of the ZSM-5 containing catalyst additive the content of all C
5–C
9 olefins drops. This means that all C
5–C
9 olefins are cracked in the presence of 3% ZSM-5 containing catalyst additive. For the case of H-Oil naphtha processing the data in
Figures S2–S6 indicates that the use of ZSM-5 additive leads to a slight increase of C
5, and C
6 paraffin at the expense of slight decrease of C
7, and C
8 paraffin contents. In contrast to the case without processing of H-Oil naphtha with H-Oil naphtha treatment in the FCC unit shows an increment of C
5 olefins.
The data in
Figure 8 indicates that the hydrotreatment of FCC gasoline (green cluster in
Figure 8) in comparison with unhydrotreated FCC gasoline (grey cluster in
Figure 8) leads to an augmentation of paraffin (normal + iso -) by 2.1 wt.%, and naphthenes by 0.7 wt.%, and reduction of olefins by 2.9 wt.%. As evident from the data in
Table 3 this hydrocarbon composition change is accompanied by about 0.5 points decrease of RON and no change in MON.
To quantify the effect of H-Oil naphtha processing in the FCC unit along with hydrotreated VGO with and without using ZSM-5 containing additive on finished gasoline octane numbers a recipe for production of commodity gasoline meeting EN 228 requirements [
46] applied in the LNB refinery was employed.
Table 5 summarizes the results of octane number tests performed on gasoline blends with the same component composition but using four different FCC gasoline samples. It is evident from the data in
Table 5 that the processing of H-Oil naphtha in the amount of 2.3 wt.% of feed in the FCC unit does not affect the blending octane numbers of FCC gasoline. The blending RON and MON of FCC gasoline for the first two cases (Cases 1, and 2
Table 5), if the mixture of remaining gasoline components (reformate, alkylate, MTBE, and naphtha) obey the linear-by volume (LBV) blending rule [
45], were calculated by the use of equation 3 to equal to 97.0, and 82.2, respectively. Interestingly, the same gasoline recipe mixture showed about 1-point higher RON, and 0.7 point higher MON when the ZSM-5 containing catalyst additive was used. The calculated blending RON, and MON for the cases using ZSM-5 are equal to 99.0, and 83.8, respectively. Thus, the use of the ZSM-5 additive allows to increase in the FCC gasoline blending RON, and MON by 2.0, and 1.6 points, respectively. The higher blending RON, and MON of the FCC gasoline obtained when the ZSM-5 additive is used may enable to increase in the share of low octane number naphtha in the commodity gasoline. Moreover, the difference in prices between naphtha and EN 228 gasoline for the time of the study amounted to 238.8 USD/ton which was an incentive to magnify low octane number naphtha content in the finished EN 228 gasoline.
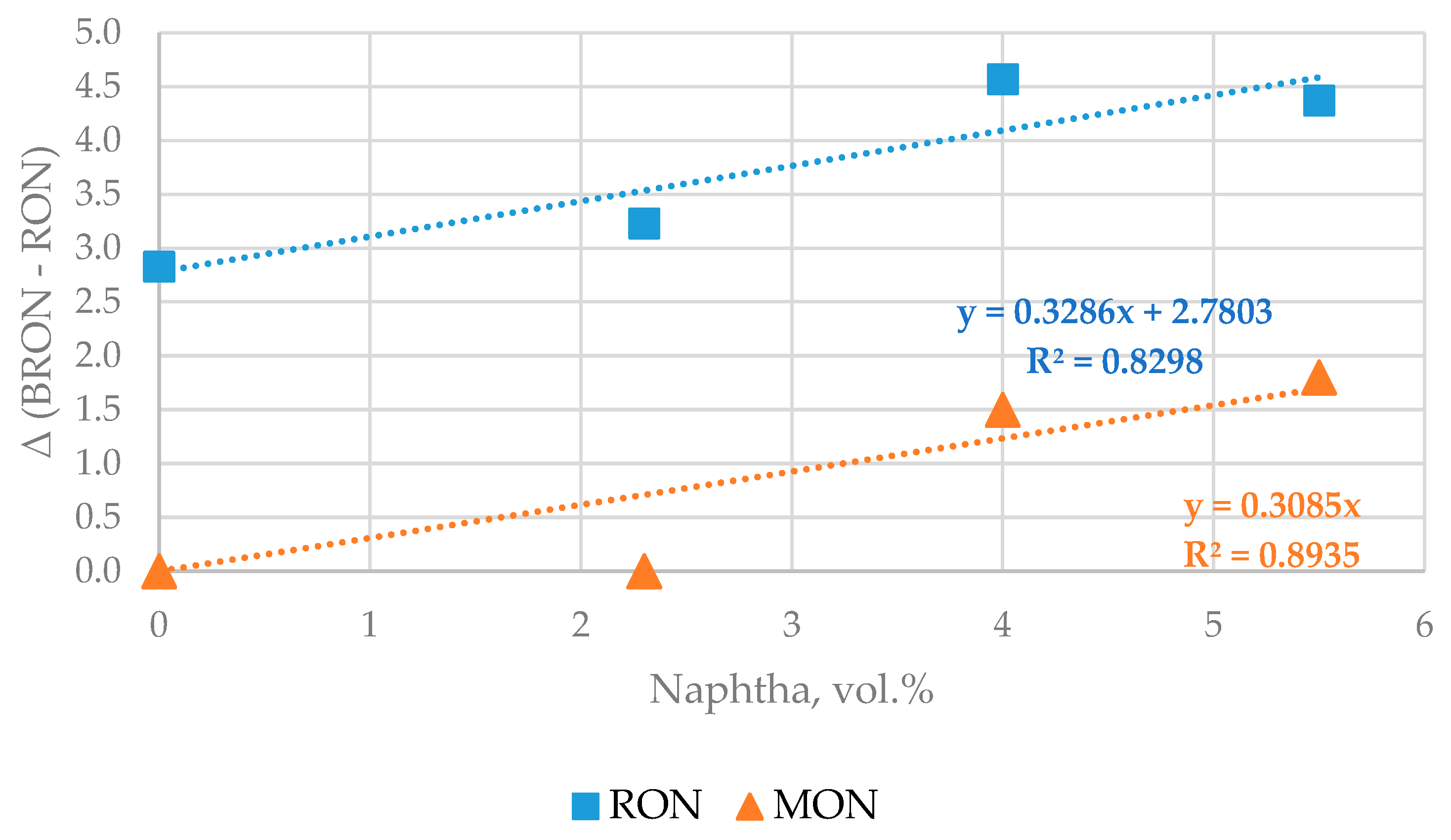
Table 6 presents data of octane numbers of blends of the gasoline components FCC gasoline, reformate, alkylate, and MTBE in the same ratio, while the content of low octane naphtha was 4, and 5.5 vol.% in the four mixtures.
The data in
Table 6 indicates that the increase of low octane naphtha from 4 to 5.5 vol.% has no effect on the octane number of finished gasoline and that it meets the requirements of EN 228. Therefore, the use of ZSM 5 containing catalyst additive in the FCC unit seems to enable increasing the content of naphtha in the finished EN 228 gasoline. Based on the data generated in this study it was observed that in the processing of H-Oil naphtha in the FCC unit and using ZSM 5 containing catalyst additive the difference between FCC gasoline octane numbers and its blending octane numbers seems to go up with an increment of naphtha content in the gasoline mixture (see
Figure 9). This suggests that further enhancement of low octane number naphtha in finished EN gasoline could be possible.
To test this hypothesis the content of H-Oil naphtha in FCC feedstock was increased from 2.3 wt.% (Case 1 of
Table 3) to 4.4% (Case 6 from
Table 3). The use of ZSM 5 containing catalyst additive in the amount of 3% of FCC catalyst inventory leads to 3.8 wt.% conversions calculated by equation 4 and using the data in Case 3 and Case 4 from
Table 3.
where,
FCC gasoline conversion = Conversion of FCC gasoline to dry gas, and LPG resulting from the use of the ZSM5 containing catalyst additive, wt.%;
FCC gasoline = Amount of gasoline produced in the FCC unit when no ZSM5-containing catalyst additive is used, t/h;
FCC gasoline (ZSM5) = Amount of gasoline produced in the FCC unit when ZSM5 containing catalyst additive is used, t/h.
It is difficult to define which part of FCC gasoline is converted by the ZSM5 containing catalyst additive, that obtained from the VGO cracking, or the one coming from the H-Oil naphtha. The processing of H-Oil naphtha in the amount of 4.4% in FCC unit feedstock and taking into account that 50% of the H-Oil naphtha is converted to dry gas, and LPG one can calculate that the H-Oil naphtha part in the FCC gasoline would equal to 4.7%. Thus, the reduction of FCC yields as a result of ZSM 5 containing additive usage is compensated by the H-Oil naphtha processing. The FCC gasoline with 4.7% of it coming from the H-Oil naphtha (Case 6 from
Table 3) was used to prepare a blend with reformate, alkylate, and MTBE in the ratio shown in
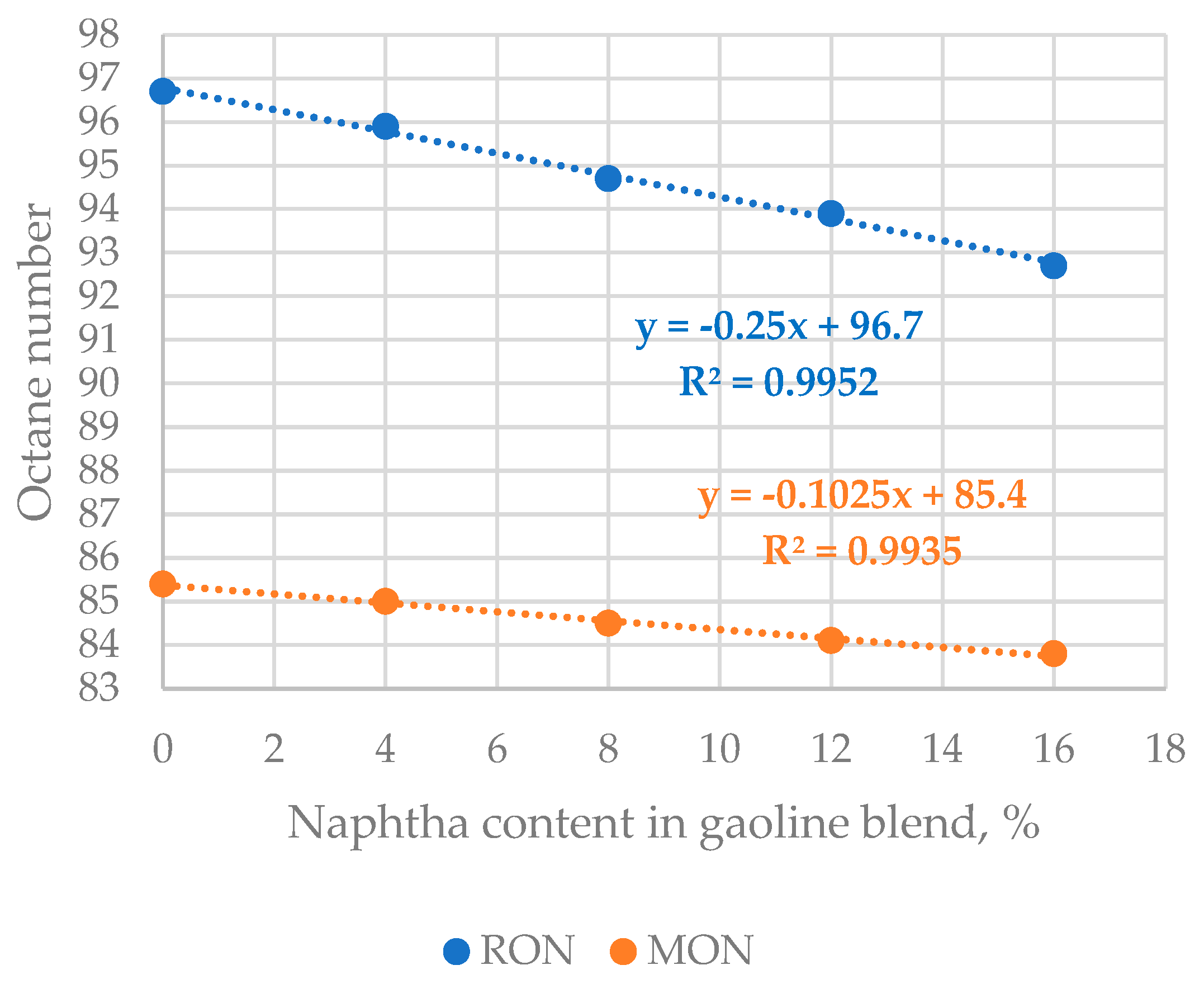
Table 5, and then this blend was mixed with 0, 4, 8, 12, and 16 vol.% low octane number naphtha (see
Table S1). RON and MON of the five gasoline composites were determined and
Figure 10 indicates the dependence of gasoline blend octane numbers on naphtha content in it. The data in
Figure 10 displays that the gasoline mixture octane numbers linearly fall with the enhancement of naphtha content. The slope of RON reduction is 2.5 times as high as that of MON reduction. The data in
Figure 10 also exhibits that EN 228 gasoline can be produced with 4 vol.% naphtha in the gasoline blend.
Considering that the content of FCC gasoline in the blend meeting EN 228 requirements is 51.5 vol.% (see the data in
Table S1, mixture No 2), and that 4.7% of it comes from the H-Oil naphtha the naphtha contribution would equal 6.4%. In other words, the processing of H-Oil naphtha in FCC unit in the amount of 4.4 wt.% of feed, and the use of ZSM 5 containing catalyst additive enable increasing the involvement of naphtha in automotive EN 228 gasoline production from 4 to 6.4%. Estimation of margin improvement using a refinery LP model availing Aspen Unified PIMS software with the data generated in this research showed an effect of a seven-digit figure USD/year. The blending RON of naphtha computed from the data in
Table S1 equals 73.4, while that of MON equals 74.9. Comparing this data with that in
Table 5 it could be seen that the naphtha blending RON is higher by 9.4 points, while the blending MON is higher by 12.9 points from the stand-alone naphtha.
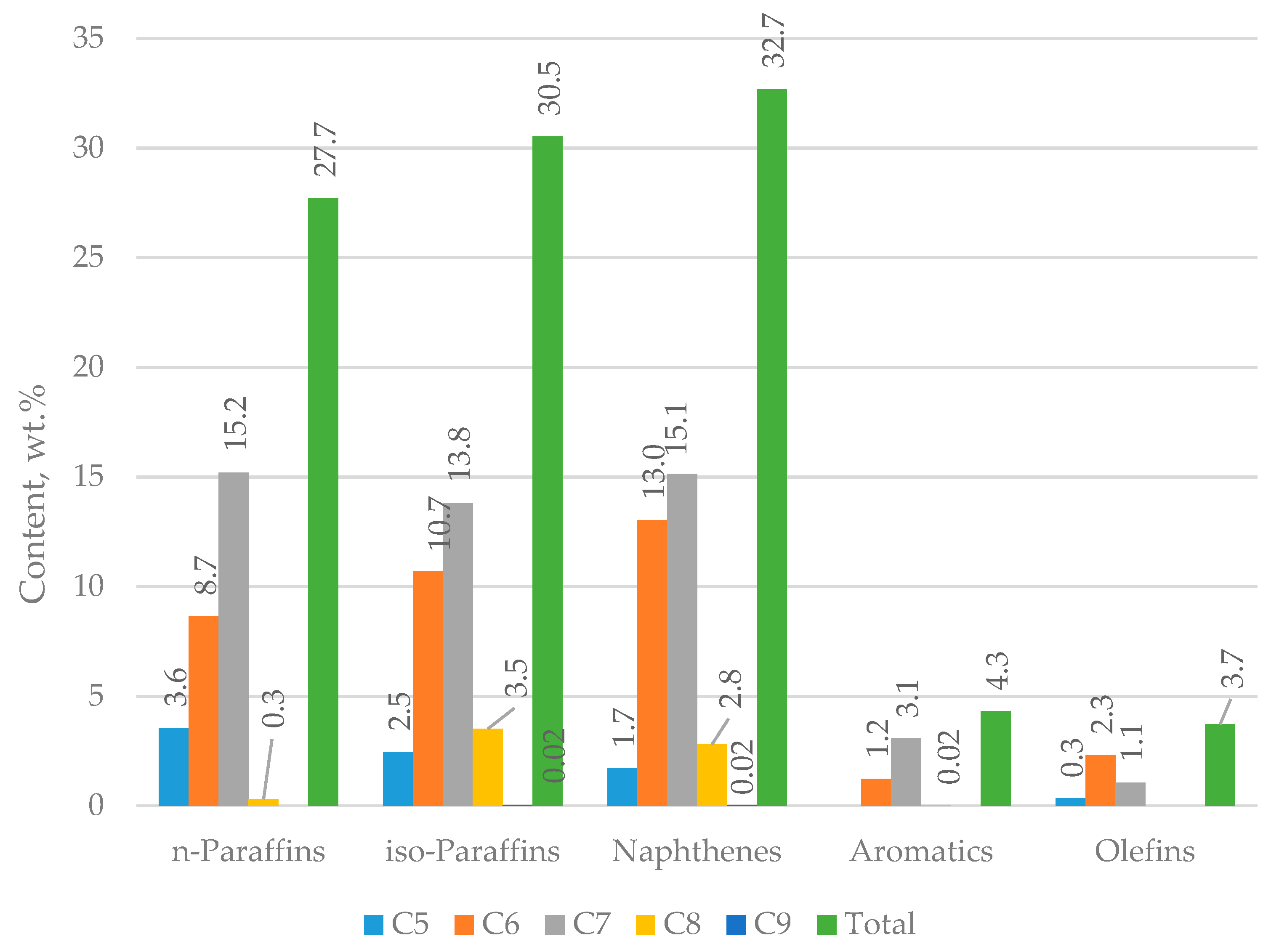
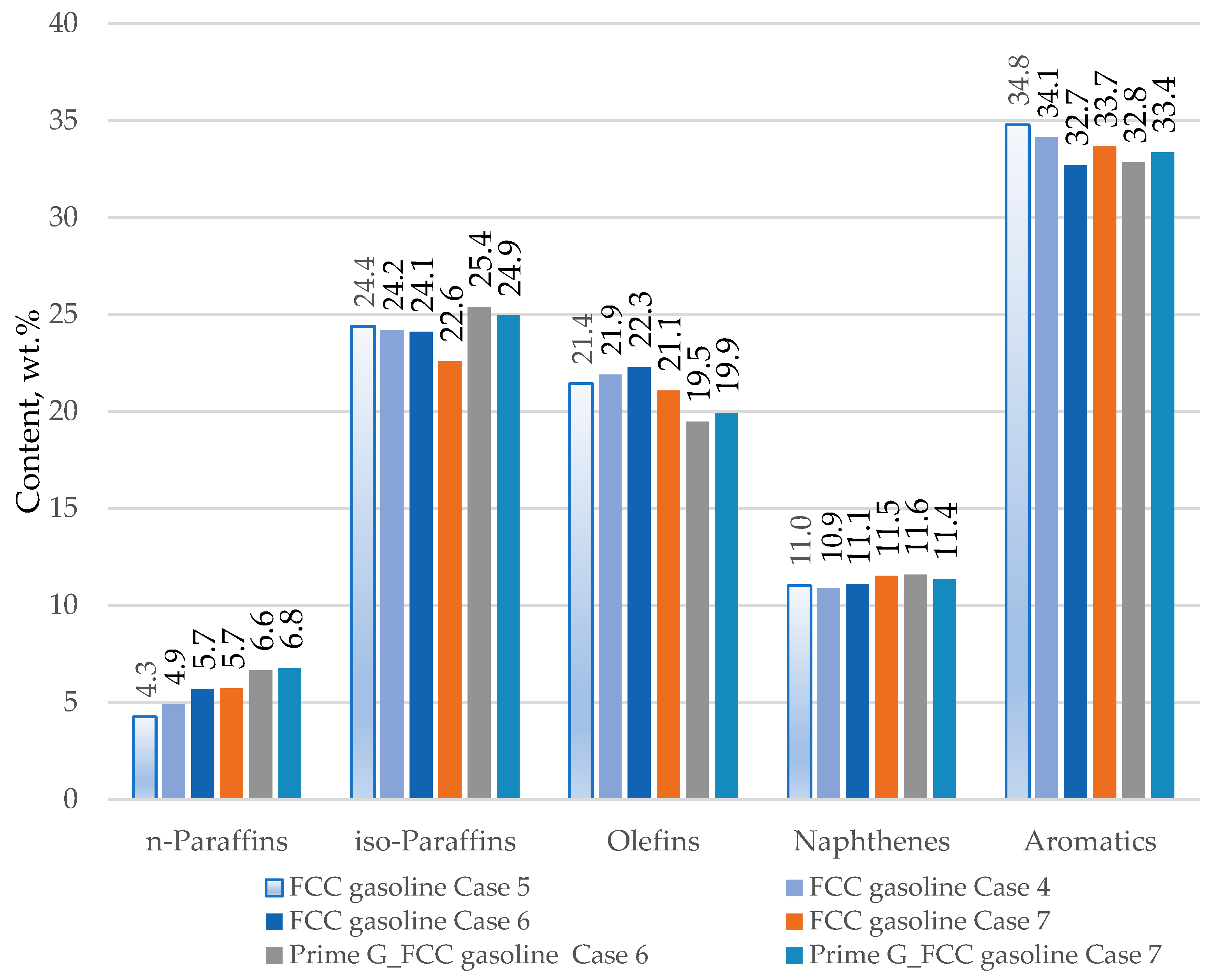
Figure 11 presents data on the hydrocarbon group composition of FCC gasoline samples taken from the FCC unit during the usage of ZSM 5 containing catalyst additive in the amount of 3% of catalyst inventory with different percentages of H-Oil naphtha in the total FCC feed. Case 5: 0% H-Oil naphtha; Case 4 2.7% H-Oil naphtha; Case 6: 4.4% H-Oil naphtha; and Case 7: 5.7% H-Oil naphtha. It can be seen from this data that n-paraffin content goes up with the enhancement of H-Oil naphtha in combined FCC feed. The hydrotreatment of FCC gasoline in the Prime-G+ process additionally magnifies the n-paraffin content. Iso-paraffin content slightly goes down with H-Oil naphtha amplification, while the Prime-G+ process treatment increments iso-paraffin content. Olefin content seems to go through a maximum with H-Oil naphtha enlargement from 0 to 4.4% and then falls at 5.7% H-Oil naphtha content in the combined FCC feed. The Prime-G+ desulphurization process decreases the olefin content in studied FCC gasoline samples. Naphthene content exhibits slight augmentation with both H-Oil naphtha increment and Prime-G process hydrotreatment. Aromatic content in opposite olefin content displays a reduction with H-Oil naphtha increase to 4.4% H-Oil naphtha in the combined FCC feed and then goes up. The Prime-G+ hydrodesulphurization process does not affect the aromatic content in FCC gasoline obtained during H-Oil naphtha treatment in the FCC unit.
4. Discussion
Oxidation stability of gasoline was found by researchers to deteriorate when olefins, and dienes present therein [
47,
48,
49,
50,
51,
52,
53,
54]. The data for olefin content (2.1 wt.%) and MAV (1.7 mg/g) of H-Oil naphtha (
Figure 2 and
Table 1) is an order of magnitude lower than that of FCC gasoline (
Figure 11, olefin content of 25.2 wt.%; MAV of 10 mg/g). Irrespective of this when adding 50 ppm of synthetic antioxidant BHT to the FCC gasoline its oxidation stability is improved and the storage period reaches 2 years [
55], while the addition of the same synthetic antioxidant even at higher treating rates does not inhibit the gum formation in H-Oil naphtha and it forms sediments after a week and change its colour as shown in
Figure 7. Uzcategui et al. [
56] reported that thermally cracked naphtha encompassed molecules, which can commence free radical reactions, and oxidation is known to follow the free radical mechanism [
57]. The H-Oil naphtha, which is a product of the thermal conversion of vacuum residue [
58] may contain some compounds in an amount sufficient to initiate the chain free radical reactions of oxidation leading to gum formation, and these components differ from those contained in FCC gasoline. The hydrotreatment of H-Oil naphtha along with straight-run naphtha, however, seems to remove these species and enable the achievement of a long period of storage of the hydrotreated product. The composition of H-Oil naphtha shown in
Table 1, and
Figure 1 and
Figure 2 could be considered suitable to use this stream as a steam cracking feed without the need for hydrotreatment because the studies of Gholami et al. [
59], and Karaba et al. [
13] reported that C
5–C
8 paraffin and olefins, which are the predominant components in H-Oil naphtha do not inhibit the cracking reaction. Thus, its hydrotreatment improves only its storage period and reduces sulphur content which allows to use of this secondary naphtha stream as a component for the production of near-zero sulphur automotive gasoline.
The conceivable employment of H-Oil naphtha as a reformer feed as indicated in
Figure 5 and
Figure 6 may be possible if it is separated into light and heavy fractions. The light fraction is not an appropriate feed for catalytic reforming because its characteristic number N + 2A is very low (17.4) and is therefore only suitable for steam cracking [
60]. The heavy fraction presenting about 40% of total H-Oil naphtha has a very good characteristic number N + 2A of 59.4. Unfortunately, its extremely high nitrogen content (188 ppm) relative to the typical reformer feed—the straight-run heavy naphtha (nitrogen content of 0.3 ppm) requires the use of a dedicated hydrotreatment unit operated at relatively high pressure to be capable of removing its high nitrogen content [
61].
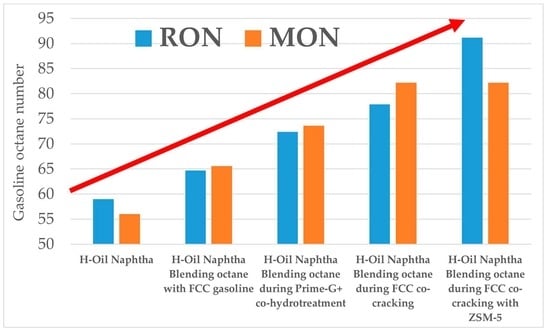
The utilization of H-Oil naphtha as a component for premium automotive EN 228 gasoline production as illustrated in
Figure 10 and
Table 4 shows that its octane ratings go up when co-processed in the Prime-G+ unit along with FCC gasoline, and in the FCC unit co-processed with VGO. The most efficient way of H-Oil naphtha utilization seems to be that of its catalytic cracking achieving 50% conversion to dry gas and LPG (see
Table 3) and not altering the blending octane numbers of FCC gasoline as shown in
Table 5 and registering the highest H-Oil naphtha blending octane number case as shown in
Table 4. The refinery LP model estimation revealed a potential for margin improvement in the amount of six-digit figure USD/year for the case of H-Oil naphtha processing in the FCC unit. A further improvement in the utilization of H-Oil naphtha as FCC unit feedstock was registered during the use of the ZSM 5-containing catalyst additive. The blending octane number of FCC gasoline increased from 97 to 99 RON, and from 82.2 to 83.8 MON when the ZSM 5-containing additive was employed. This enabled increasing the low octane number naphtha contribution to the premium automotive EN 228 gasoline grade production from 4 to 6.4 vol.%. The refinery LP model estimation disclosed another considerable improvement in refining margin, a result of H-Oil naphtha processing in the FCC unit in the presence of ZSM-5 containing additive.
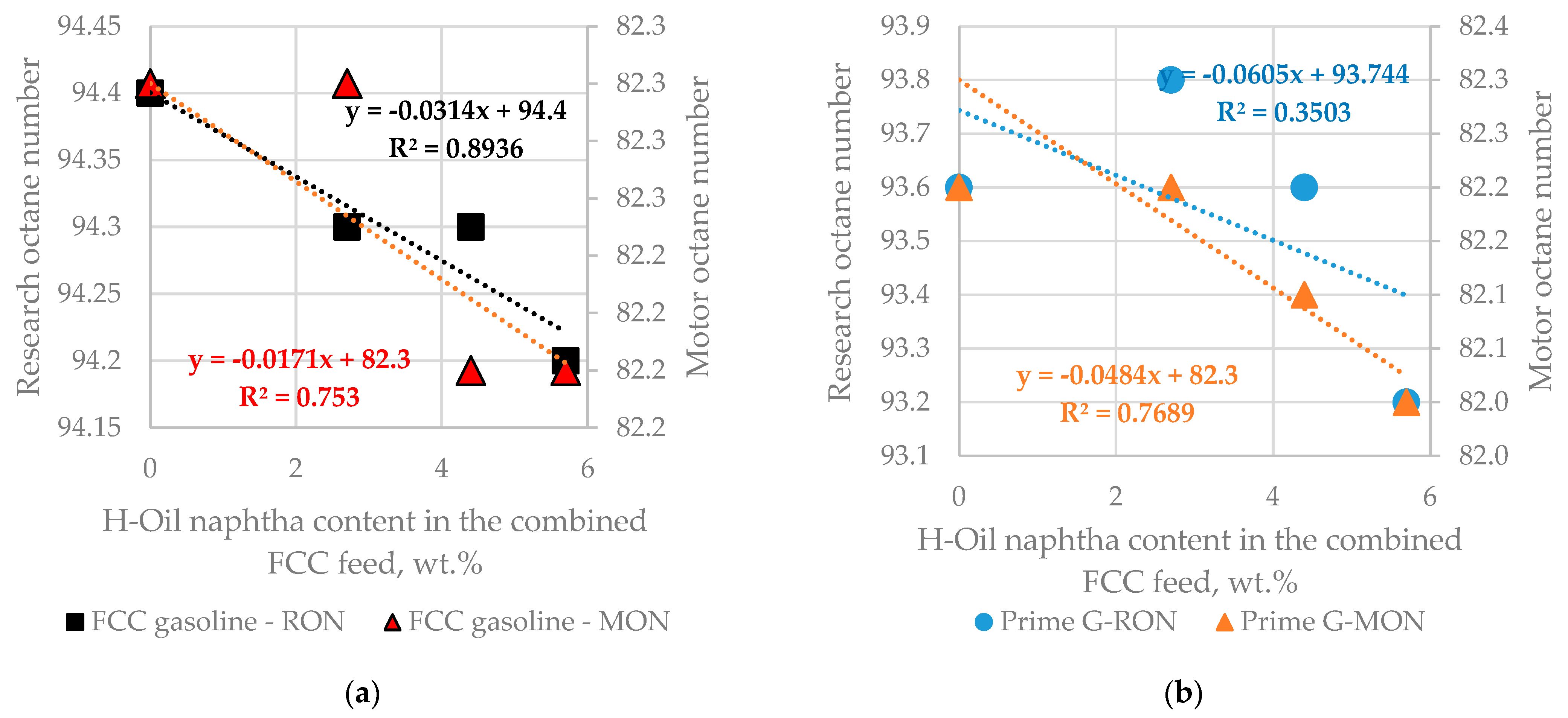
Summarization of results about FCC gasoline octane number variation as a function of H-Oil naphtha content in the combined FCC feed during the addition of 3% ZSM 5 catalyst additive to the total catalyst inventory shown in
Table 3 (Cases 4, 5, 6, and 7) is presented in
Figure 12. Although the slope of octane dependence on H-Oil naphtha content is negative its value is too low and for 10% H-Oil naphtha content alteration the RON decrease is 0.3 point, and MON is 0.2 which is within reproducibility of octane number measurement (reproducibility of 0.7 RON, and 0.9 MON according to the standards ASTM D 2699, and ASTM D 2700 [
36,
37].
Figure 12 b indicates that the negative slope of Prime-G+ hydrotreated FCC gasoline is twice as high as that of the non-post-treated FCC gasoline.
Another important aspect of H-Oil naphtha processing in the FCC unit in the way it is introduced in the FCC reactor (see
Figure 3 and
Figure S1) is the reduction of lift steam. The decrease in the amount of lift steam equals the ratio of H-Oil naphtha quantity processed in FCC unit and the molecular weight of H-Oil naphtha (109 g/mol, see
Table 1) divided by the molecular weight of water (18 g/mol). For Case 7, for example, the processing of 10.9 t/h H-Oil naphtha leads to a decrease in consumption of lift steam by 1.8 t/h.
Additional improvement in the utilization of H-Oil naphtha could be achieved in future studies directed to the use of specialized additives and co-processing of bio-components as discussed in [
62,
63,
64,
65,
66,
67,
68].