1. Introduction
Currently, the optimization of CO
2 uptake has become a crucial focus in the pursuit of sustainable solutions to mitigate climate change. The atmospheric CO
2 concentration has been rapidly increasing at an exceptional rate of 2.2 ppm/year [
1]. It is estimated that by approximately 2050, the atmospheric CO
2 concentration may reach around 450 ppm. Consequently, this increase in CO
2 levels is projected to cause a temperature rise of 2 °C to 3 °C on the Earth’s surface due to the greenhouse effect [
2]. To tackle these challenges, numerous methods and techniques have been developed to capture and store CO
2, each offering its own advantages and potential applications [
3,
4,
5]. Carbon capture and storage is a method that captures CO
2 emissions generated from industrial processes and securely stores them underground, ensuring that they do not escape into the atmosphere [
6,
7]. Carbon capture and utilization refers to the process of capturing CO
2 emissions and converting them into valuable commodities, such as chemicals, plastics, or fuels [
8,
9]. Biological carbon sequestration exploits the natural capacity of plants and trees to absorb CO
2 through photosynthesis [
10]. Enhanced weathering accelerates natural processes to capture CO
2 by applying crushed minerals like basalt or olivine, which react with CO
2 to form stable carbonates [
11]. Direct air capture uses specialized technologies to extract CO
2 directly from the ambient air [
12]. The choice of approach relies on various factors, including the CO
2 emission source, implementation scale, economic feasibility, and environmental considerations.
The continuous growth of the construction industry has led to a rise in waste generation, particularly in the form of concrete by-products. Traditionally, waste concrete powder has been viewed as an environmental concern, typically leading to landfill disposal or pollution if not handled properly [
13]. In the European Union, construction and demolition waste comprise approximately 35–46% of the total waste stream in the European Union [
14]. Notably, end-of-life concrete makes up a significant portion of this construction and demolition waste, contributing around 12–40% to the overall waste volume [
15]; however, recent studies highlighted the potential of waste concrete powder as a valuable resource for CO
2 sequestration [
16]. By utilizing waste concrete powder in a controlled environment, we can enhance its ability to capture and store CO
2, turning it into a beneficial asset rather than a liability [
17]. Studies have specifically explored the recycling of alkaline-rich waste, including filter-pressed concrete slurry waste, through accelerated carbonation techniques [
18,
19].
Waste concrete powder (WCP) exhibits promising potential as an option for carbon dioxide (CO
2) sequestration, owing to its capability to undergo chemical reactions with CO
2 and effectively store it within its structure. The CO
2 uptake capacity refers to the amount of carbon dioxide that can be absorbed and stored by WCP through a process known as mineral carbonation [
20]. Mineral carbonation is a process that converts CO
2 into solid mineral forms, thereby effectively sequestering the carbon and preventing its release into the atmosphere [
21]. The CO
2 uptake capacity of WCP is influenced by various factors such as the carbonation time (CT) (duration of the carbonation process) and the water-to-solid ratio (WSR), which evaluates the amount of water used in relation to the quantity of WCP [
22]. Optimizing these variables is crucial for enhancing the CO
2 uptake capacity of WCP and establishing it as a sustainable solution for carbon capture. This entails selecting the appropriate WCP composition, identifying optimal CO
2 absorption conditions, and developing efficient implementation techniques [
23]. Studies have shown that the ability of WCP to absorb CO
2 can be significantly enhanced by optimizing these factors [
24]. For example, enhancing the fineness of the crushed concrete particles, which refers to reducing the particle size of WCP, can create more surface area for the chemical reaction to occur, leading to a higher CO
2 uptake capacity [
25]. Kaliyavaradhan et al. [
26] explored the dual functionality of concrete slurry waste as a CO
2 capture agent and a supplementary cementitious material. The study investigated the influence of WSR and reaction time on the CO
2 uptake capacity of concrete slurry waste using response surface methodology (RSM). The optimal conditions were found to be a w/s ratio of 0.25 and a reaction time of 72 h, resulting in a remarkable maximum CO
2 uptake of 20.4%.
Accelerated mineral carbonation has gained considerable attention as a promising technique in carbon capture and storage. This process allows for the long-term storage of CO
2 by employing a controlled chemical reaction between CO
2 and alkaline oxides, like calcium oxide (CaO) and magnesium oxide (MgO), commonly present in natural silicate rocks or industrial by-products. The outcome of this reaction is the formation of stable carbonate compounds, such as calcium carbonate (CaCO
3) and magnesium carbonate (MgCO
3), which find diverse practical applications. Pan et al. [
27] estimated the global CO
2 mitigation potential of applying accelerated carbonation to various alkaline solid wastes. The results indicate that CO
2 mineralization and utilization can significantly reduce CO
2 emissions, achieving a 12.5% global reduction equivalent to 4.02 Gt per year.
A significant number of experimental investigations are dedicated to evaluating and optimizing CO
2 storage through accelerated carbonation processes. These studies aim to optimize operational parameters, encompassing temperature, pressure, gas humidity, liquid and gas flow rates, liquid-to-solid ratio, solid pre-treatment, and particle size [
28,
29,
30]. Based on experimental results, the filter-pressed concrete slurry waste has exhibited an impressive CO
2 sequestration capacity, reaching up to 75% of the total CO
2 uptake in just a few hours. Over a duration of 144 h of carbonation, concrete slurry waste demonstrated the capability to capture 110 g of CO
2/kg of dry concrete slurry waste [
31].
Due to the costly, limited, and time-intensive nature of conducting experimental studies on CO
2 uptake from waste concrete powder across different operational conditions to determine the highest CO
2 uptake, there is a need for alternative effective approaches to assess characteristics effectively. Employing optimization methods offers an advantageous approach to minimize the necessity for extensive experimental trials [
32,
33,
34]. This study aims to bridge the existing gap in the application of AI techniques and the recent optimization method, specifically the Marine Predators Algorithm (MPA), for maximizing CO
2 absorption from WCP. Although WCP has shown promise as a potential CO
2 sequestration agent, there is a lack of comprehensive studies that explore the optimization of CO
2 uptake from WCP using advanced AI algorithms. Therefore, this study aims to bridge this gap by integrating the ANFIS modeling approach and the MPA to identify optimal values for CT and WSR, thus, enhancing CO
2 absorption from WCP. The primary point of this research is to enhance CO
2 uptake through the utilization of the MPA in combination with ANFIS modeling. The initial step involves the development of an ANFIS model based on experimental datasets to simulate the CO
2 uptake, considering the CT and WST as variables. Subsequently, the MPA is employed to identify the optimal values for the CT and WSR, aiming to maximize the CO
2 uptake. Furthermore, this study includes a comparative analysis between ANFIS modeling and the traditional ANOVA method in terms of accuracy and prediction capabilities for CO
2 uptake from WCP. This analysis provides insights into the superiority of ANFIS modeling, which exhibits a substantial increase in the R-squared and a significant reduction in the RMSE.
The contribution aspect of this study is observed in the integration of AI techniques, particularly ANFIS modeling and the MPA, to optimize CO2 absorption from waste concrete powder. While previous studies have primarily focused on exploring the CO2 sequestration potential of WCP, a limited number have employed advanced AI algorithms to enhance the absorption process. By utilizing ANFIS modeling, this research improves the accuracy and prediction capabilities for CO2 uptake, leading to significant advancements in modeling accuracy; moreover, the integration of ANFIS and the MPA further enhances the percentage value of CO2 uptake, demonstrating the effectiveness of this novel approach. The findings of this study contribute to sustainable waste management practices in the construction industry by providing valuable insights into optimizing CO2 absorption from waste concrete powder using AI techniques.
The key contributions of the paper can be outlined as follows:
Development of a robust ANFIS model for CO2 uptake, considering carbonation time and water-to-solid ratio;
Introduction of a novel implementation of MPA to enhance CO2 uptake;
Conducting a thorough analysis and comparison between the proposed Marine Predators Algorithm (MPA) and various featured algorithms such as Particle Swarm Optimization (PSO), Genetic Algorithm (GA), Harris Hawks Optimization (HHO), and Cuckoo Search (CS) to validate the efficacy of the MPA method; furthermore, employing statistical tests to ensure an equitable and objective assessment of the different metaheuristic algorithms performed.
3. Results and Discussion
This study examined the CO2 absorption from WCP, with a focus on assessing the impacts of two critical factors: carbonation time (CT) and water-to-solid ratio (WSR). The investigation harnessed the power of artificial intelligence (AI) and the Marine Predators Algorithm (MPA) to pinpoint optimal values for these parameters.
The impact of carbonation time (CT) on CO
2 uptake from waste concrete powder is a pivotal aspect in assessing the carbon capture potential of this material. Carbonation time refers to the duration of exposure of waste concrete powder to CO
2-rich environments, initiating chemical reactions between CO
2 and the calcium-containing compounds within the concrete. According to
Table 1, longer CTs are associated with several notable effects on CO
2 uptake. With an increase in the CT, there is a tendency for enhanced CO
2 uptake driven due to enhanced penetration, leading to more extensive chemical reactions and calcium carbonate formation; however, identifying the optimal carbonation duration becomes crucial, as beyond a certain point, additional CO
2 exposure might yield diminishing returns in CO
2 absorption. Moreover, prolonged carbonation can induce physical changes in waste concrete powder, influencing properties like porosity, density, and strength, which impact its applicability.
Exploring the impact of the water-to-solid ratio (WSR) on CO
2 uptake from WCP constitutes a vital aspect of this study. The water-to-solid ratio denotes the proportion of water used in the carbonation process relative to the quantity of waste concrete powder. The changes in the WSR have the potential to influence CO
2 absorption capacity, potentially leading to enhancement due to improved interaction between CO
2 and the WCP. Additionally, at the beginning of the process, with a decrease in WSR or with an increase in the solid–liquid ratio, there is a corresponding elevation in the amount of CO
2 uptake. This implies a greater availability of calcium (Ca) to engage in CO
2 capture and the subsequent formation of calcium carbonate (CaCO
3). Additionally, the CO
2 uptake efficiency (measured as g-CO
2/g-concrete fines) remains relatively consistent, showing a slight increase with higher solid–liquid ratios, as displayed in
Table 1. Furthermore, water distribution within the material plays a role in the uniformity of carbonation, and variations in the ratio can also lead to changes in physical properties. Therefore, determining the optimal WSR becomes crucial, similar to CT, in order to attain effective CO
2 capture.
3.1. Modeling Phase
The ANFIS framework was constructed based on a dataset of 23 experiments, with the dataset divided into two groups for training and testing purposes. The first group, comprising 18 points, was used for training the model, and the remaining points were designated for testing the model’s performance. The hybrid training method integrated least squares estimation in the forward path and utilized backpropagation for the backward direction to achieve an effective training process. A set of 10 fuzzy rules for the system was derived using the subtractive clustering method. The model was trained iteratively until a reduced RMSE was achieved.
Table 2 presents the statistical metrics obtained from the ANFIS model.
The ANFIS model for CO
2 uptake exhibited RMSE values of 3.3124 × 10
−6 and 0.022 for the training and testing datasets, respectively, as displayed in
Table 2. The R-Square for the training and testing stages are 1.0 and 0.9994, respectively, as presented in
Table 2. Comparing these results with ANOVA [
23], the R-Square has increased by approximately 19% from 0.84 using ANOVA to 0.9999 using ANFIS. Additionally, the RMSE has significantly decreased from 1.96 using ANOVA to 0.0102 using ANFIS, indicating the effectiveness of the ANFIS modeling phase.
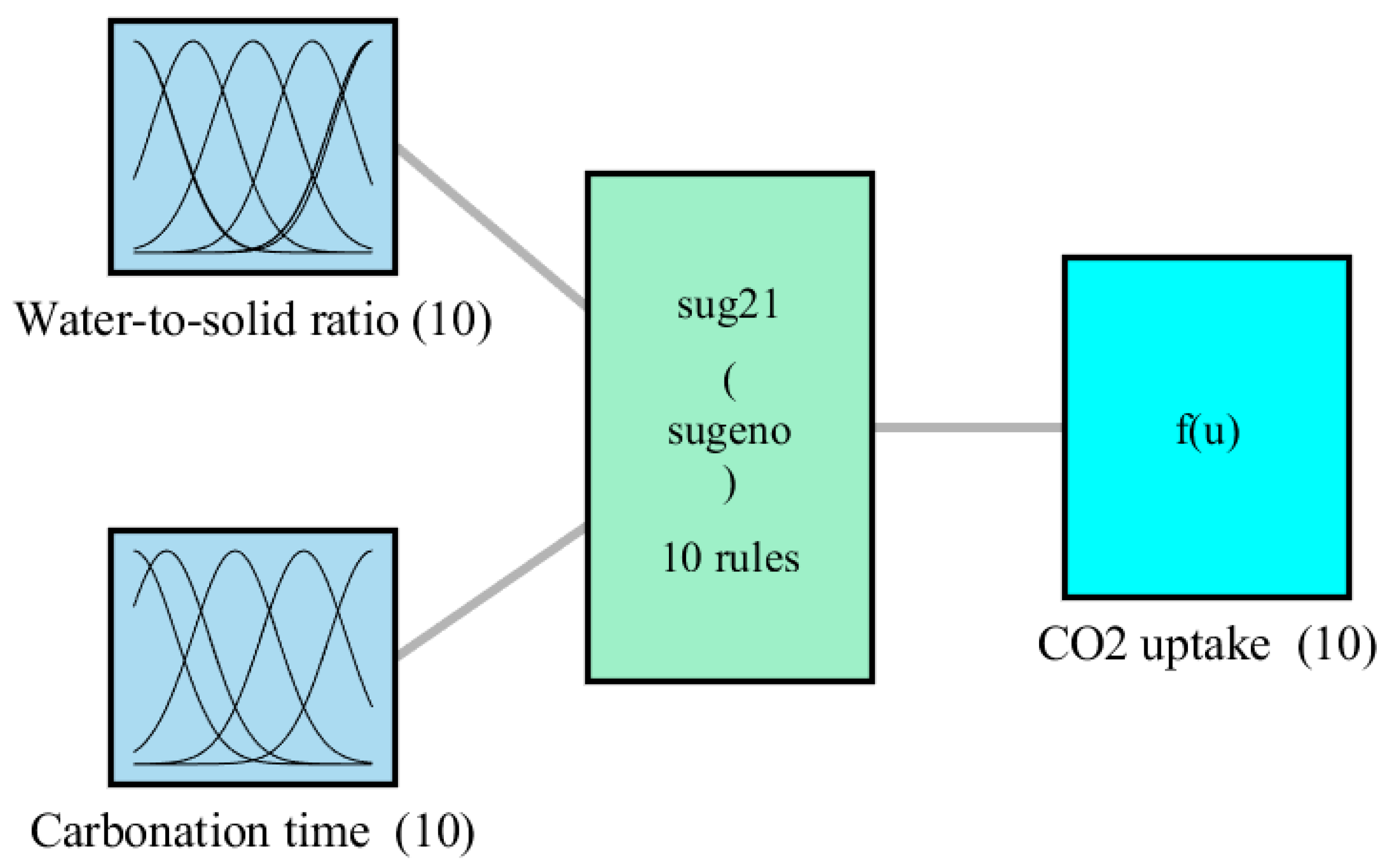
Figure 3 demonstrates the architecture of the ANFIS model with two inputs and one output, while
Figure 4 displays the general contours of the Gaussian-form MFs.
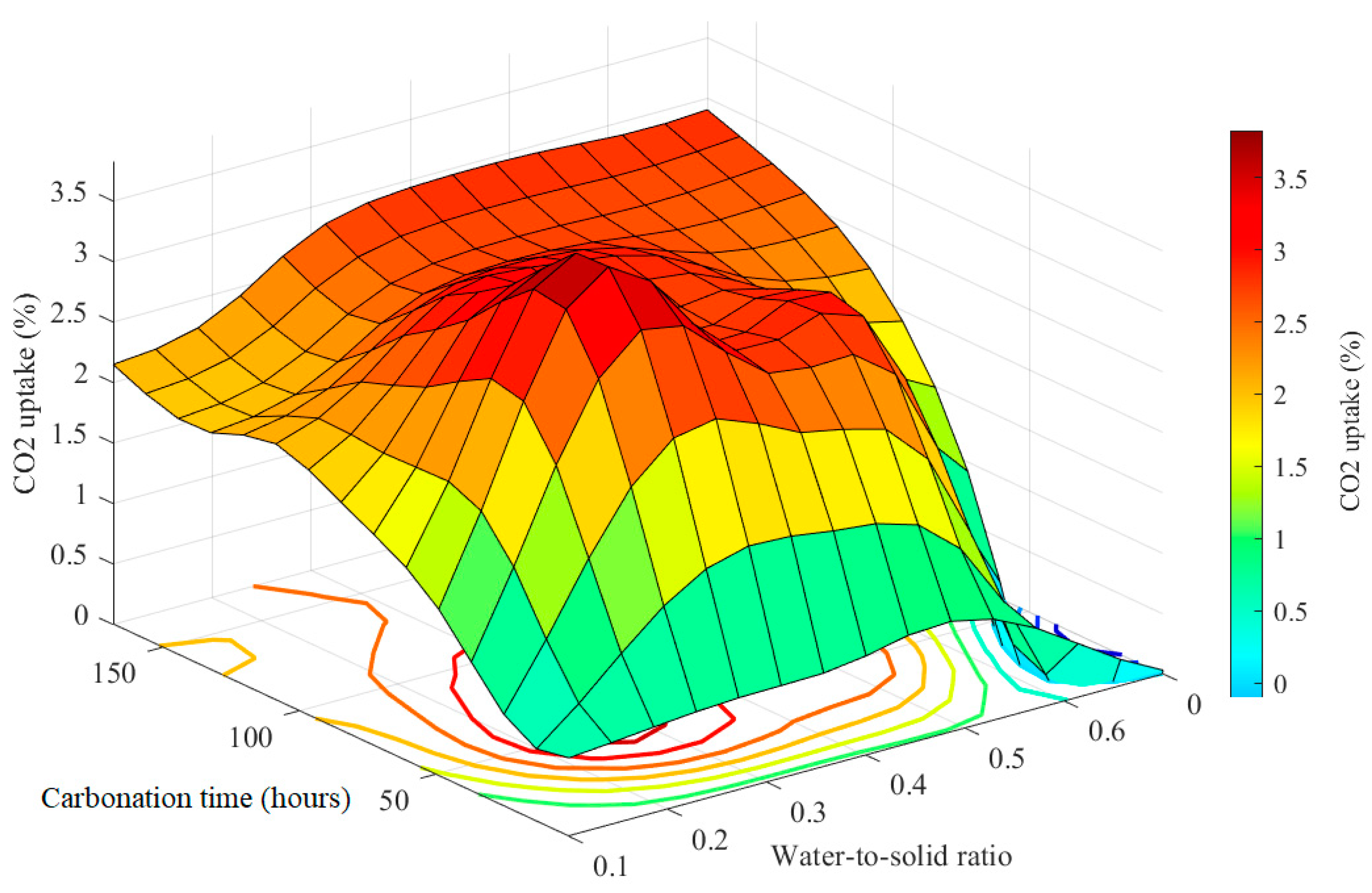
Figure 5 presents a spatial representation from a 3-D perspective, illustrating the contours of the input–output function for each combination of inputs. The color gradient ranges from dark red, representing the highest output value of CO
2 uptake, to blue, indicating the lowest. The figure demonstrates the integration of ANFIS and MPA, highlighting the accomplishment of a remarkable CO
2 uptake rate of 3.86%. This optimization is achieved through the identification of the optimal carbonation time (CT) of 54.3 h and the selection of a suitable water-to-solid ratio (WSR) of 0.327.
Figure 6 displays the comparison between the predicted and measured data of the ANFIS model for CO
2 uptake. The plot demonstrates a strong alignment between the estimated and measured values. This alignment between the estimated and measured values is particularly robust, indicating a high level of accuracy and reliability in the ANFIS model’s predictive capabilities; furthermore,
Figure 7 exhibits the prediction results for both the training and testing phases, depicting a close approximation to the line of 100% accuracy.
Table 3 presents the validation results of the ANFIS model for CO
2 uptake in comparison to ANOVA. The absolute error values for CO
2 uptake are 0.25 and 0.14, respectively, when compared to the experimental data for ANOVA and ANFIS. The ANFIS model exhibits a 44% reduction in absolute error for CO
2 uptake compared to ANOVA. Furthermore, the percentage error values are 8.83% and 4.95% for ANOVA and ANFIS, respectively. These findings highlight the superior performance of the ANFIS model when compared to the ANOVA model.
3.2. Optimization Phase
In this section, the objective was to determine the optimal values of the water-to-solid ratio (WSR) and carbonation time (CT) that would result in a high CO
2 uptake percentage. To achieve this, the Marine Predators Algorithm (MPA) was utilized in combination with reliable ANFIS models of CO
2 uptake. In the process of optimization, the two parameters CT and WSR are utilized as decision variables, aiming to maximize the CO
2 uptake, which functions as the objective function. The optimized results obtained using the experimental, RSM, and recommended methods are displayed in
Table 4. The integration of ANFIS and MPA led to a significant increase in the CO
2 uptake percentage by approximately 30% compared to the experimental data and RSM. The optimal values under these conditions were found to be 0.27 for the WSR and 54.3 h for the CT.
Figure 8 provides a visual representation of the convergence process of particles concerning three key aspects: the objective function, WSR, and CT. The figure offers a clear trajectory of each particle’s iterative approach toward the optimal values of these parameters. It can be observed that all particles converged to the optimal values within 20 iterations. This concise and efficient convergence underscores the efficacy of the method employed, instilling confidence in the precision and reliability of the obtained optimal parameter values.
The Particle Swarm Optimization (PSO), Genetic Algorithm (GA), Harris Hawks Optimization (HHO), and Cuckoo Search (CS) outcomes are compared to validate the MPA suppository. To ensure consistent outcomes, each optimizer is executed 30 times, and a comprehensive statistical analysis is performed.
Table 5 and
Figure 9 present the detailed results of 30 runs utilizing different optimizers. Statistical indicators such as the highest value, lowest value, average value, and standard deviation are computed and presented in
Table 6. The average values of the cost function (based on the ANFIS model of CO
2 uptake) range from 3.762 to 3.857. The MPA achieved the highest average value (3.857), followed by PSO (3.836), and then GA (3.762) with the lowest average value. The standard deviation values range from 0.002 to 0.184, where MPA demonstrates the best standard deviation (STD) value of 0.002, followed by HHO (0.055), and CS (0.184). These results highlight the effectiveness of MPA in identifying optimal values that lead to the highest CO
2 uptake.