Mathematical Modeling of Drying Kinetics and Technological and Chemical Properties of Pereskia sp. Leaf Powders
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material
2.2. Convective Drying and Experimental Design
2.3. Moisture Ratio
2.4. Mathematical Models
2.5. Effective Diffusivity
2.6. Thermodynamic Properties
2.7. Physical and Physicochemical Characteristics of Powders
2.8. Statistical Analysis
3. Results and Discussion
3.1. Drying Kinetics
3.2. Effective Diffusivity
3.3. Thermodynamic Properties
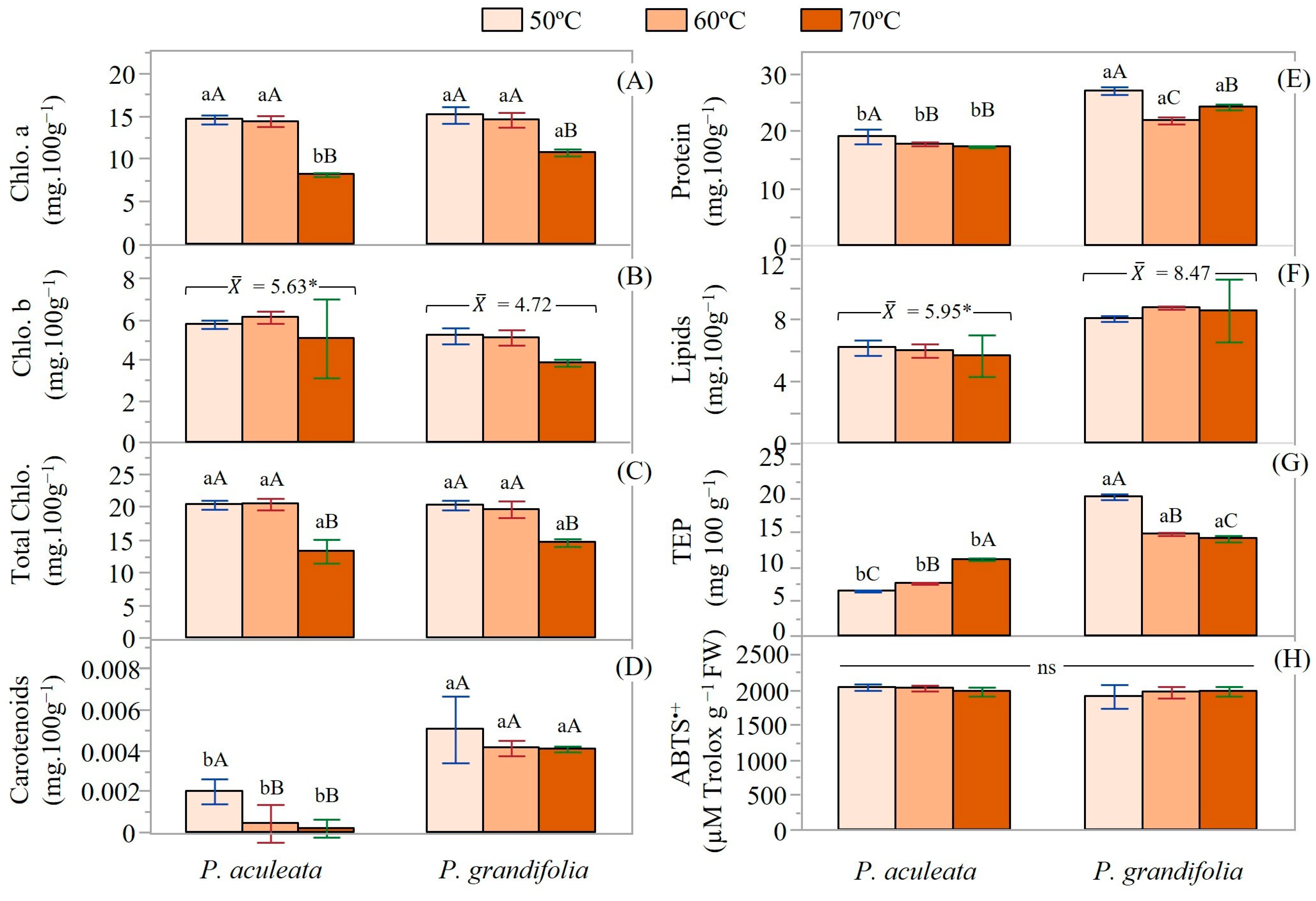
3.4. Technological and Chemical Properties of Pereskia sp. Powders
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Venter, N.; Cowie, B.W.; Paterson, I.D.; Witkowski, E.T.F.; Byrne, M.J. The Interactive Effects of CO2 and Water on the Growth and Physiology of the Invasive Alien Vine Pereskia aculeata (Cactaceae): Implications for Its Future Invasion and Management. Environ. Exp. Bot. 2022, 194, 104737. [Google Scholar] [CrossRef]
- Souza, L.F.; Gasparetto, B.F.; Lopes, R.R.; Barros, I.B.I. Temperature Requirements for Seed Germination of Pereskia aculeata and Pereskia grandifolia. J. Therm. Biol. 2016, 57, 6–10. [Google Scholar] [CrossRef] [PubMed]
- Martin, A.A.; de Freitas, R.A.; Sassaki, G.L.; Evangelista, P.H.L.; Sierakowski, M.R. Chemical Structure and Physical-Chemical Properties of Mucilage from the Leaves of Pereskia aculeata. Food Hydrocoll. 2017, 70, 20–28. [Google Scholar] [CrossRef]
- Maciel, V.B.V.; Yoshida, C.M.P.; Boesch, C.; Goycoolea, F.M.; Carvalho, R.A. Iron-Rich Chitosan-Pectin Colloidal Microparticles Laden with Ora-pro-Nobis (Pereskia aculeata Miller) Extract. Food Hydrocoll. 2020, 98, 105313. [Google Scholar] [CrossRef]
- Silva, S.H.; Neves, I.C.O.; de Meira, A.C.F.O.; Alexandre, A.C.S.; Oliveira, N.L.; Resende, J.V. de Freeze-Dried Petit Suisse Cheese Produced with Ora-pro-Nóbis (Pereskia aculeata Miller) Biopolymer and Carrageenan Mix. LWT 2021, 149, 111764. [Google Scholar] [CrossRef]
- Teixeira, V.M.C.; de Oliveira, A.; Backes, E.; de Souza, C.G.M.; Castoldi, R.; de Sá-Nakanishi, A.B.; Bracht, L.; Comar, J.F.; Corrêa, R.C.G.; Leimann, F.V.; et al. A Critical Appraisal of the Most Recent Investigations on Ora-Pro-Nobis (Pereskia sp.): Economical, Botanical, Phytochemical, Nutritional, and Ethnopharmacological Aspects. Plants 2023, 12, 3874. [Google Scholar] [CrossRef]
- Garcia, J.A.A.; Corrêa, R.C.G.; Barros, L.; Pereira, C.; Abreu, R.M.V.; Alves, M.J.; Calhelha, R.C.; Bracht, A.; Peralta, R.M.; Ferreira, I.C.F.R. Phytochemical Profile and Biological Activities of “Ora-pro-Nobis” Leaves (Pereskia aculeata Miller), an Underexploited Superfood from the Brazilian Atlantic Forest. Food Chem. 2019, 294, 302–308. [Google Scholar] [CrossRef]
- Mahanti, N.K.; Chakraborty, S.K.; Sudhakar, A.; Verma, D.K.; Shankar, S.; Thakur, M.; Singh, S.; Tripathy, S.; Gupta, A.K.; Srivastav, P.P. Refractance WindowTM-Drying vs. Other Drying Methods and Effect of Different Process Parameters on Quality of Foods: A Comprehensive Review of Trends and Technological Developments. Future Foods 2021, 3, 100024. [Google Scholar] [CrossRef]
- Salhi, M.; Chaatouf, D.; Raillani, B.; Amraqui, S.; Mezrhab, A. Investigating the Effect of Food Trays Porosity on the Drying Process. Innov. Food Sci. Emerg. Technol. 2022, 76, 102939. [Google Scholar] [CrossRef]
- Bassani, A.; Carullo, D.; Rossi, F.; Fiorentini, C.; Garrido, G.D.; Reklaitis, G.V.R.; Bonadies, I.; Spigno, G. Modeling of a Spray-Drying Process for the Encapsulation of High-Added Value Extracts from Food by-Products. Comput. Chem. Eng. 2022, 161, 107772. [Google Scholar] [CrossRef]
- Cavalcanti-Mata, M.E.; Duarte, M.E.; Tolentino, M.; Mendes, F.A.; Batista, L.; de Lima, J.M.; Lúcio, A.; Nascimento, A.P.; Almeida, R.D.; Lisboa, H.M. Drying Kinetics of Industrial Pineapple Waste: Effective Diffusivity and Thermodynamic Properties Resulting from New Mathematical Models Derived from the Fick Equation. Processes 2024, 12, 1198. [Google Scholar] [CrossRef]
- Instituto Adolfo Lutz (IAL). Normas Analíticas Do Instituto Adolfo Lutz: Métodos Químicos e Físicos Para Análise de Alimentos, 4th ed.; IAL: São Paulo, Brazil, 2008. [Google Scholar]
- Moura, H.V.; de Figueirêdo, R.M.F.; de Melo Queiroz, A.J.; de Vilela Silva, E.T.; Esmero, J.A.D.; Lisbôa, J.F. Mathematical Modeling and Thermodynamic Properties of the Drying Kinetics of Trapiá Residues. J. Food Process Eng. 2021, 44, e13768. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; Foundation for Statistical Computing: Vienna, Austria, 2018. [Google Scholar]
- Reddy Mugi, V.; Chandramohan, V.P. Energy, Exergy and Economic Analysis of an Indirect Type Solar Dryer Using Green Chilli: A Comparative Assessment of Forced and Natural Convection. Therm. Sci. Eng. Prog. 2021, 24, 100950. [Google Scholar] [CrossRef]
- Buera, M.P.; Lozano, R.D.; Petriella, C. Definition of Color in the Non-Enzymatic Browning Process. Die Farbe 1985, 32, 316–326. [Google Scholar]
- de Souza, R.L.; de Oliveira, L.D.S.; da Silva, F.L.H.; Amorim, B.C. Caracterização Da Poligalacturonase Produzida Por Fermentação Semi-Sólida Utilizando-Se Resíduo Do Maracujá Como Substrato. Rev. Bras. Eng. Agríc. Ambient. 2010, 14, 987–992. [Google Scholar] [CrossRef]
- Tang, J.; Zhao, Y.; Cheng, W.; Liu, T.; Yang, G.; Chen, M.; Lei, G.; Xu, J.; Huang, Y. Evaluation of Thermal and Mechanical Properties of Foamed Phosphogypsum-Based Cementitious Materials for Well Cementing in Hydrate Reservoirs. J. Mar. Sci. Eng. 2024, 12, 1056. [Google Scholar] [CrossRef]
- Tonon, R.V.; Brabet, C.; Hubinger, M.D. Aplicação Da Secagem Por Atomização Para a Obtenção de Produtos Funcionais Com Alto Valor Agregado a Partir Do Açaí. Inclusão Soc. 2013, 6, 70–76. [Google Scholar]
- Decker, B.L.A.; de Miguel, E.C.; Fonteles, T.V.; Fernandes, F.A.N.; Rodrigues, S. Impact of Spray Drying on the Properties of Grape Pomace Extract Powder. Processes 2024, 12, 1390. [Google Scholar] [CrossRef]
- Hausner, H.H. Friction Conditions in a Mass of Metal Powder. Int. J. Powder Metal 1967, 3, 7–13. [Google Scholar]
- Bu, Y.; Addo, A.; Amyotte, P.; Yuan, C. Classification of Dispersibility for Combustible Dust Based on Hausner Ratio. J. Loss Prev. Process Ind. 2023, 81, 104925. [Google Scholar] [CrossRef]
- Eastman, J.E.; Moore, C.O. Cold Water Soluble Granular Starch for Gelled Food Composition. U.S. Patent No. 4,465,702, 14 August 1984. [Google Scholar]
- Cano-Chauca, M.; Stringheta, P.C.; Ramos, A.M.; Cal-Vidal, J. Effect of the Carriers on the Microstructure of Mango Powder Obtained by Spray Drying and Its Functional Characterization. Innov. Food Sci. Emerg. Technol. 2005, 6, 420–428. [Google Scholar] [CrossRef]
- Ceballos, A.M.; Giraldo, G.I.; Orrego, C.E. Effect of Freezing Rate on Quality Parameters of Freeze Dried Soursop Fruit Pulp. J. Food Eng. 2012, 111, 360–365. [Google Scholar] [CrossRef]
- Lichtenthaler, H.K. Chlorophylls and Carotenoids: Pigments of Photosynthetic Biomembranes. In Methods in Enzymology; Academic Press: Cambridge, MA, USA, 1987; pp. 350–382. [Google Scholar]
- Association of Official Analytical Chemists (AOAC). Official Methods of Analysis of the Association of Official Analytical Chemists, 18th ed.; AOAC International: Gaithersburg, MD, USA, 2006. [Google Scholar]
- Waterhouse, A. Folin-Ciocalteau Micro Method for Total Phenol in Wine. Am. J. Enol. Vitic. 2006, 48, 357–363. [Google Scholar]
- dos Lima, M.S.; Silani, I.D.S.V.; Toaldo, I.M.; Corrêa, L.C.; Biasoto, A.C.T.; Pereira, G.E.; Bordignon-Luiz, M.T.; Ninow, J.L. Phenolic Compounds, Organic Acids and Antioxidant Activity of Grape Juices Produced from New Brazilian Varieties Planted in the Northeast Region of Brazil. Food Chem. 2014, 161, 94–103. [Google Scholar] [CrossRef] [PubMed]
- Patel, M.R.; Panwar, N.L. Drying Kinetics, Quality Assessment and Socio Environmental Evaluation of Solar Dried Underutilized Arid Vegetable Cucumis Callosus. Energy Nexus 2022, 7, 100128. [Google Scholar] [CrossRef]
- del Razola-Díaz, M.C.; Guerra-Hernández, E.J.; Gómez-Caravaca, A.M.; García-Villanova, B.; Verardo, V. Mathematical Modelling of Drying Kinetics of Avocado Peels and Its Influence on Flavan-3-Ols Content and Antioxidant Activity. LWT 2023, 176, 114552. [Google Scholar] [CrossRef]
- Getahun, E.; Delele, M.A.; Gabbiye, N.; Fanta, S.W.; Demissie, P.; Vanierschot, M. Importance of Integrated CFD and Product Quality Modeling of Solar Dryers for Fruits and Vegetables: A Review. Solar Energy 2021, 220, 88–110. [Google Scholar] [CrossRef]
- de Gondim, C.M.; de Figueirêdo, R.M.F.; de Queiroz, A.J.M.; Esmero, J.A.D.; Moura, H.V.; dos Moreira, I.S. Thermodynamic Properties and Bioactive Compounds of Mandacaru Dried Waste. J. Food Process Preserv. 2022, 46, e17230. [Google Scholar] [CrossRef]
- Masud, M.H.; Himel, H.H.; Arefin, A.M.E.; Ananno, A.A.; Rashid, M.; Dabnichki, P. Mathematical Modelling and Exergo-Environmental Analysis of Drying Potato Samples in a Waste Heat-Based Convective Dryer. Environ. Chall. 2021, 5, 100372. [Google Scholar] [CrossRef]
- Jin, X.; van der Sman, R.G.M. Interaction between Large Deformation and Moisture Transport during Dehydration of Vegetables. Food Struct. 2022, 32, 100269. [Google Scholar] [CrossRef]
- Martins, J.J.A.; Marques, J.I.; Santos, D.C.; Rocha, A.P.T. Modelagem MatemÁtica Da Secagem de Cascas de Mulungu Mathematical Modeling of the Drying of Husks Mulungu. Biosci. J. 2014, 30, 1652–1660. [Google Scholar]
- Almeida, M.E.F.; Simão, A.A.; Corrêa, A.D.; Fernandes, R.V. de B. Improvement of Physiological Parameters of Rats Subjected to Hypercaloric Diet, with the Use of Pereskia grandifolia (Cactaceae) Leaf Flour. Obes. Res. Clin. Pract. 2016, 10, 701–709. [Google Scholar] [CrossRef]
- Xu, Y.; Xiao, Y.; Lagnika, C.; Li, D.; Liu, C.; Jiang, N.; Song, J.; Zhang, M. A Comparative Evaluation of Nutritional Properties, Antioxidant Capacity and Physical Characteristics of Cabbage (Brassica Oleracea Var. Capitate Var L.) Subjected to Different Drying Methods. Food Chem. 2020, 309, 124935. [Google Scholar] [CrossRef]
- Santos, L.G.; Martins, V.G. Functional, Thermal, Bioactive and Antihypertensive Properties of Hot Trub Derived from Brewing Waste as an Alternative Source of Protein. Food Hydrocoll. 2024, 146, 109292. [Google Scholar] [CrossRef]
- Böhm, W.; Stegmann, R.; Gulbis, O.; Henle, T. Amino Acids and Glycation Compounds in Hot Trub Formed during Wort Boiling. Eur. Food Res. Technol. 2023, 249, 119–131. [Google Scholar] [CrossRef]
- Fan, Y.; Zhang, L.; Zheng, C.; Wang, X.; Wang, K.; Zhu, J. Motion Behavior of Non-Newtonian Fluid-Solid Interaction Foods. J. Food Eng. 2023, 347, 111448. [Google Scholar] [CrossRef]
- Shen, C.; Li, Y.; Xu, X. Analysis of the Compressibility of Edible Powders under Low Pressure. J. Food Eng. 2022, 316, 110828. [Google Scholar] [CrossRef]
- Ebert, S.; Gibis, M.; Terjung, N.; Weiss, J. Survey of Aqueous Solubility, Appearance, and PH of Plant Protein Powders from Carbohydrate and Vegetable Oil Production. LWT 2020, 133, 110078. [Google Scholar] [CrossRef]
- Shahbal, N.; Jing, X.; Bhandari, B.; Dayananda, B.; Prakash, S. Effect of Enzymatic Hydrolysis on Solubility and Surface Properties of Pea, Rice, Hemp, and Oat Proteins: Implication on High Protein Concentrations. Food Biosci. 2023, 53, 102515. [Google Scholar] [CrossRef]
- Fournaise, T.; Gaiani, C.; Petit, J. Descriptive Modelling of Food Powders Reconstitution Kinetics Followed by Laser Granulometry. Chem. Eng. Sci. 2022, 252, 117440. [Google Scholar] [CrossRef]
- Pravallika, K.; Chakraborty, S.; Singhal, R.S. Supercritical Drying of Food Products: An Insightful Review. J. Food Eng. 2023, 343, 111375. [Google Scholar] [CrossRef]
- Welsh, Z.G.; Simpson, M.J.; Khan, M.I.H.; Karim, M.A. Generalized Moisture Diffusivity for Food Drying through Multiscale Modeling. J. Food Eng. 2023, 340, 111309. [Google Scholar] [CrossRef]
- Stępień, A.; Grzyb, K. Comparison of Critical Storage Parameters of the Powders Containing Soy Protein Isolate and Inulin, Based on the Concepts: Water Activity and Temperature of Glass Transition. Int. J. Biol. Macromol. 2023, 230, 123174. [Google Scholar] [CrossRef]
- Anju, T.; Rai, N.K.S.R.; Uthirchamkavu, I.; Sreedharan, S.; Ndhlala, A.R.; Singh, P.; Kumar, A. Analysis of Nutritional and Antioxidant Potential of Three Traditional Leafy Vegetables for Food Security and Human Wellbeing. S. Afr. J. Bot. 2022, 145, 99–110. [Google Scholar] [CrossRef]
- Derardja, A.E.; Pretzler, M.; Kampatsikas, I.; Radovic, M.; Fabisikova, A.; Zehl, M.; Barkat, M.; Rompel, A. Polyphenol Oxidase and Enzymatic Browning in Apricot (Prunus armeniaca L.): Effect on Phenolic Composition and Deduction of Main Substrates. Curr. Res. Food Sci. 2022, 5, 196–206. [Google Scholar] [CrossRef]
- Lefebvre, T.; Talbi, A.; Atwi-Ghaddar, S.; Destandau, E.; Lesellier, E. Development of an Analytical Method for Chlorophyll Pigments Separation by Reversed-Phase Supercritical Fluid Chromatography. J. Chromatogr. 2020, 1612, 460643. [Google Scholar] [CrossRef]
- Mileti, O.; Baldino, N.; Lupi, F.R.; Gabriele, D. Interfacial Behavior of Vegetable Protein Isolates at Sunflower Oil/Water Interface. Colloids Surf. Biointerfaces 2023, 221, 113035. [Google Scholar] [CrossRef]
- Masiero, J.F.; Barbosa, E.J.; de Macedo, L.O.; Souza, A.; Yukuyama, M.N.; Arantes, G.J.; Bou-Chacra, N.A. Vegetable Oils in Pharmaceutical and Cosmetic Lipid-Based Nanocarriers Preparations. Ind. Crops Prod. 2021, 170, 113838. [Google Scholar] [CrossRef]
- Sousa, A.S.B.; Silva, M.C.A.; Lima, R.P.; de Albuquerque Meireles, B.R.L.; Cordeiro, A.T.M.; Santos, E.F.S.; Amaro, A.L.; Pintado, M.M.E.; Silva, S.M. Phenolic compounds and antioxidant activity as discriminating markers and adding value of mango varieties. Sci. Hortic. 2021, 287, 110259. [Google Scholar] [CrossRef]
- Ouyang, M.; Huang, Y.; Wang, Y.; Luo, F.; Liao, L. Stability of Carotenoids and Carotenoid Esters in Pumpkin (Cucurbita maxima) Slices during Hot Air Drying. Food Chem. 2022, 367, 130710. [Google Scholar] [CrossRef]
- Lee, S.Y.; Liang, Y.N.; Stuckey, D.C.; Hu, X. Single-Step Extraction of Bioactive Compounds from Cruciferous Vegetable (Kale) Waste Using Natural Deep Eutectic Solvents. Sep. Purif. Technol. 2023, 317, 123677. [Google Scholar] [CrossRef]
- Márquez-Cardozo, C.J.; Caballero-Gutiérrez, B.L.; Ciro-Velázquez, H.J.; Restrepo-Molina, D.A. Effect of Pretreatment and Temperature on the Drying Kinetics and Physicochemical and Techno-Functional Characteristics of Pumpkin (Cucurbita maxima). Heliyon 2021, 7, e06802. [Google Scholar] [CrossRef] [PubMed]
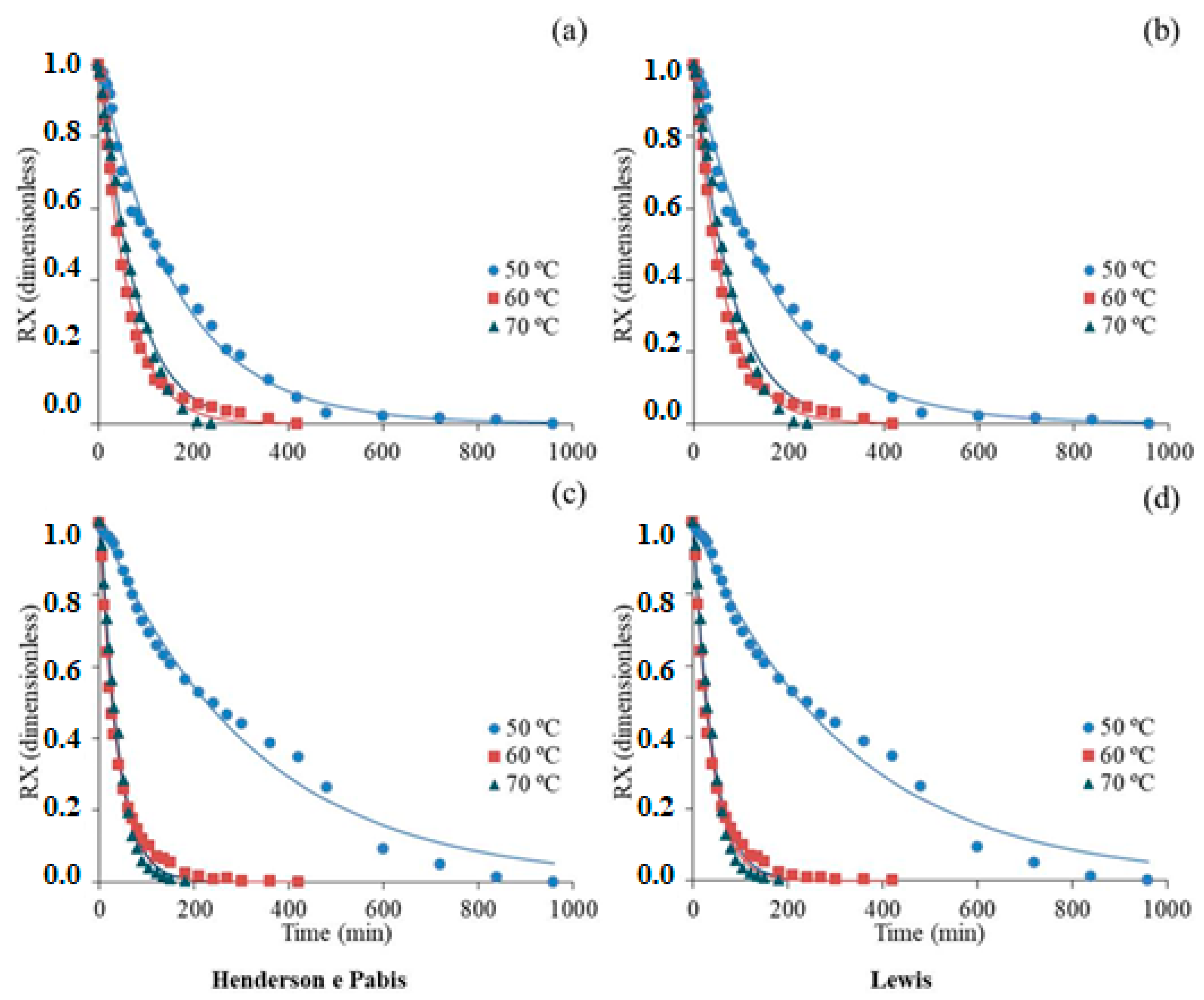
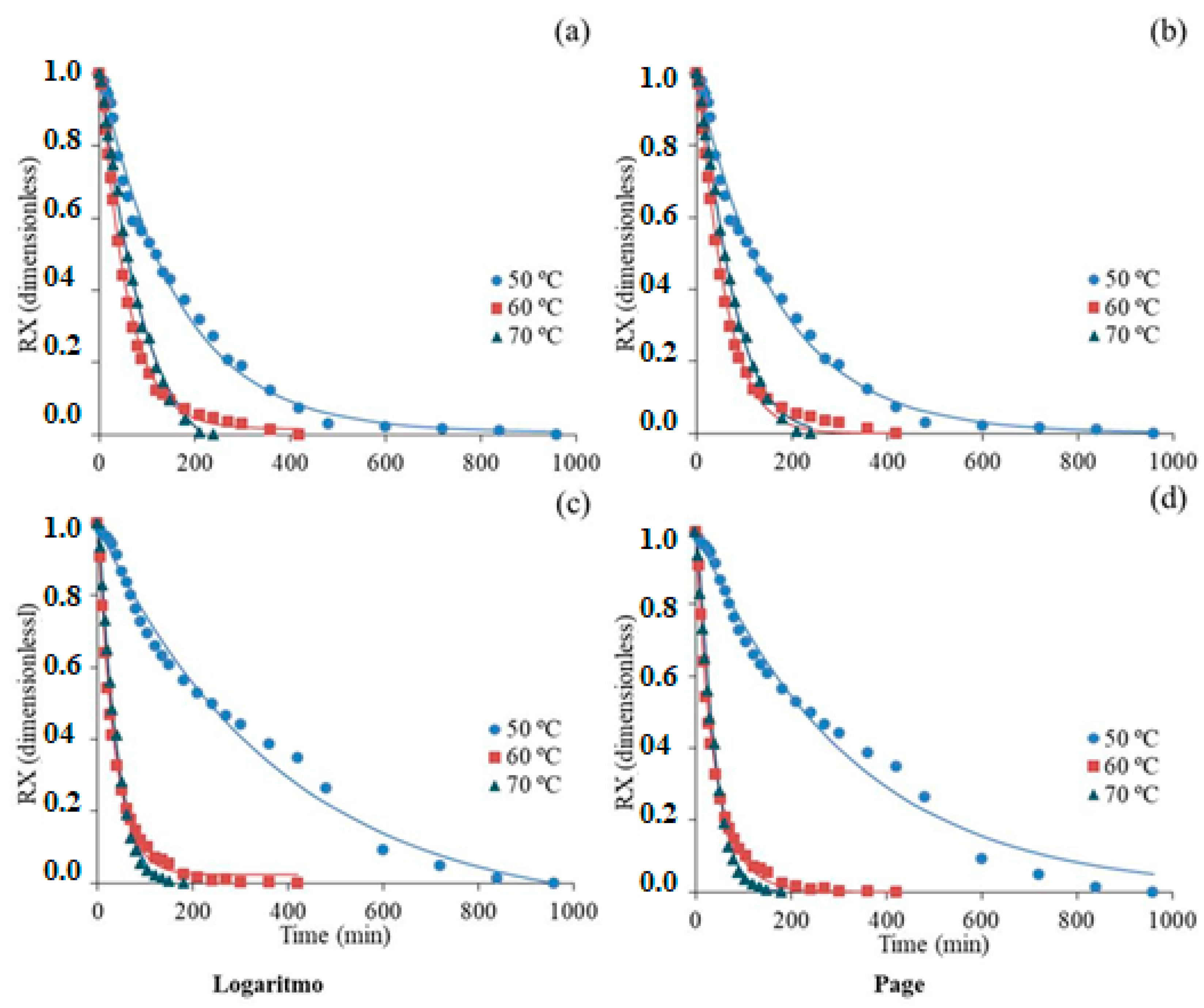


| Models | Equations |
|---|---|
| Henderson and Pabis | RX = |
| Lewis | RX = |
| Logaritmo | RX = + c |
| Page | RX = |
| Model | Species | T (°C) | a | c | k | n | R2 | MSE | χ2 |
|---|---|---|---|---|---|---|---|---|---|
| Henderson and Pabis | P. grandifolia | 50 | 1.0053 | - | 0.0031 | - | 0.9876 | 0.036 | 0.0281 |
| 60 | 0.9883 | - | 0.0267 | - | 0.993 | 0.0261 | 0.0207 | ||
| 70 | 1.0596 | - | 0.0268 | - | 0.9931 | 0.03 | 0.0241 | ||
| P. aculeata | 50 | 1.0156 | - | 0.006 | - | 0.9928 | 0.03 | 0.0228 | |
| 60 | 1.0561 | - | 0.017 | - | 0.9959 | 0.0226 | 0.0182 | ||
| 70 | 1.0633 | - | 0.0135 | - | 0.9904 | 0.035 | 0.0277 | ||
| Lewis | P. grandifolia | 50 | - | - | 0.0031 | - | 0.9875 | 0.0354 | 0.028 |
| 60 | - | - | 0.0271 | - | 0.9929 | 0.0257 | 0.0204 | ||
| 70 | - | - | 0.025 | - | 0.9891 | 0.0366 | 0.0314 | ||
| P. aculeata | 50 | - | - | 0.0059 | - | 0.9924 | 0.0303 | 0.023 | |
| 60 | - | - | 0.0159 | - | 0.9924 | 0.0301 | 0.0243 | ||
| 70 | - | - | 0.0125 | - | 0.9839 | 0.044 | 0.0372 | ||
| Logarítmico | P. grandifolia | 50 | 1.0846 | 0.0919 | 0.0026 | - | 0.9902 | 0.0326 | 0.0254 |
| 60 | 0.9776 | −0.0234 | 0.0289 | - | 0.9953 | 0.022 | 0.0184 | ||
| 70 | 1.0875 | 0.0415 | 0.0242 | - | 0.9956 | 0.0247 | 0.019 | ||
| P. aculeata | 50 | 1.0117 | −0.0051 | 0.0061 | - | 0.9928 | 0.0305 | 0.023 | |
| 60 | 1.0465 | −0.0149 | 0.0177 | - | 0.9964 | 0.0217 | 0.0161 | ||
| 70 | 1.1469 | 0.1046 | 0.011 | - | 0.9962 | 0.0225 | 0.0158 | ||
| Page | P. grandifolia | 50 | - | - | 0.0028 | 1.016 | 0.9876 | 0.036 | 0.028 |
| 60 | - | - | 0.0394 | 0.896 | 0.9956 | 0.0208 | 0.0142 | ||
| 70 | - | - | 0.0109 | 1.2242 | 0.9986 | 0.0082 | 0.0135 | ||
| P. aculeata | 50 | - | - | 0.0061 | 0.9909 | 0.9924 | 0.034 | 0.0229 | |
| 60 | - | - | 0.0089 | 1.1437 | 0.996 | 0.0224 | 0.0181 | ||
| 70 | - | - | 0.004 | 1.2647 | 0.9984 | 0.014 | 0.0103 |
| Specie | T (°C) | Effective Diffusivity (m2 s−1) | R2 |
|---|---|---|---|
| P. aculeata | 50 | 3.51 × 10−11 | 0.9424 |
| 60 | 3.26 × 10−10 | 0.9753 | |
| 70 | 3.57 × 10−10 | 0.9535 | |
| P. grandifolia | 50 | 5.72 × 10−11 | 0.9694 |
| 60 | 3.97 × 10−10 | 0.9839 | |
| 70 | 5.33 × 10−10 | 0.9713 |
| Specie | T (°C) | ΔH (kJ mol−1) | ΔS (kJ mol−1 K−1) | ΔG (kJ mol−1) |
|---|---|---|---|---|
| P. aculeata | 50 | 105.21 | −0.1090 | 140.4493 |
| 60 | 105.13 | −0.1093 | 141.5409 | |
| 70 | 105.05 | −0.1095 | 142.6351 | |
| P. grandifolia | 50 | 100.90 | −0.1190 | 139.35 |
| 60 | 100.81 | −0.1192 | 140.54 | |
| 70 | 100.73 | −0.1195 | 141.74 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
de Alcântara, C.M.; Moreira, I.d.S.; Cavalcanti, M.T.; Lima, R.P.; Moura, H.V.; da Silva Neves, R.; Cassimiro, C.A.L.; Martins, J.J.A.; da Costa Batista, F.R.; Pereira, E.M. Mathematical Modeling of Drying Kinetics and Technological and Chemical Properties of Pereskia sp. Leaf Powders. Processes 2024, 12, 2077. https://doi.org/10.3390/pr12102077
de Alcântara CM, Moreira IdS, Cavalcanti MT, Lima RP, Moura HV, da Silva Neves R, Cassimiro CAL, Martins JJA, da Costa Batista FR, Pereira EM. Mathematical Modeling of Drying Kinetics and Technological and Chemical Properties of Pereskia sp. Leaf Powders. Processes. 2024; 12(10):2077. https://doi.org/10.3390/pr12102077
Chicago/Turabian Stylede Alcântara, Charlene Maria, Inacia dos Santos Moreira, Mônica Tejo Cavalcanti, Renato Pereira Lima, Henrique Valentim Moura, Romildo da Silva Neves, Carlos Alberto Lins Cassimiro, Jorge Jacó Alves Martins, Fabiane Rabelo da Costa Batista, and Emmanuel Moreira Pereira. 2024. "Mathematical Modeling of Drying Kinetics and Technological and Chemical Properties of Pereskia sp. Leaf Powders" Processes 12, no. 10: 2077. https://doi.org/10.3390/pr12102077
APA Stylede Alcântara, C. M., Moreira, I. d. S., Cavalcanti, M. T., Lima, R. P., Moura, H. V., da Silva Neves, R., Cassimiro, C. A. L., Martins, J. J. A., da Costa Batista, F. R., & Pereira, E. M. (2024). Mathematical Modeling of Drying Kinetics and Technological and Chemical Properties of Pereskia sp. Leaf Powders. Processes, 12(10), 2077. https://doi.org/10.3390/pr12102077







