Selective Complexation and Leaching of Cobalt Using Histidine in an Alkaline Medium
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Leaching Experiment
2.3. Analytical Method
3. Results and Discussion
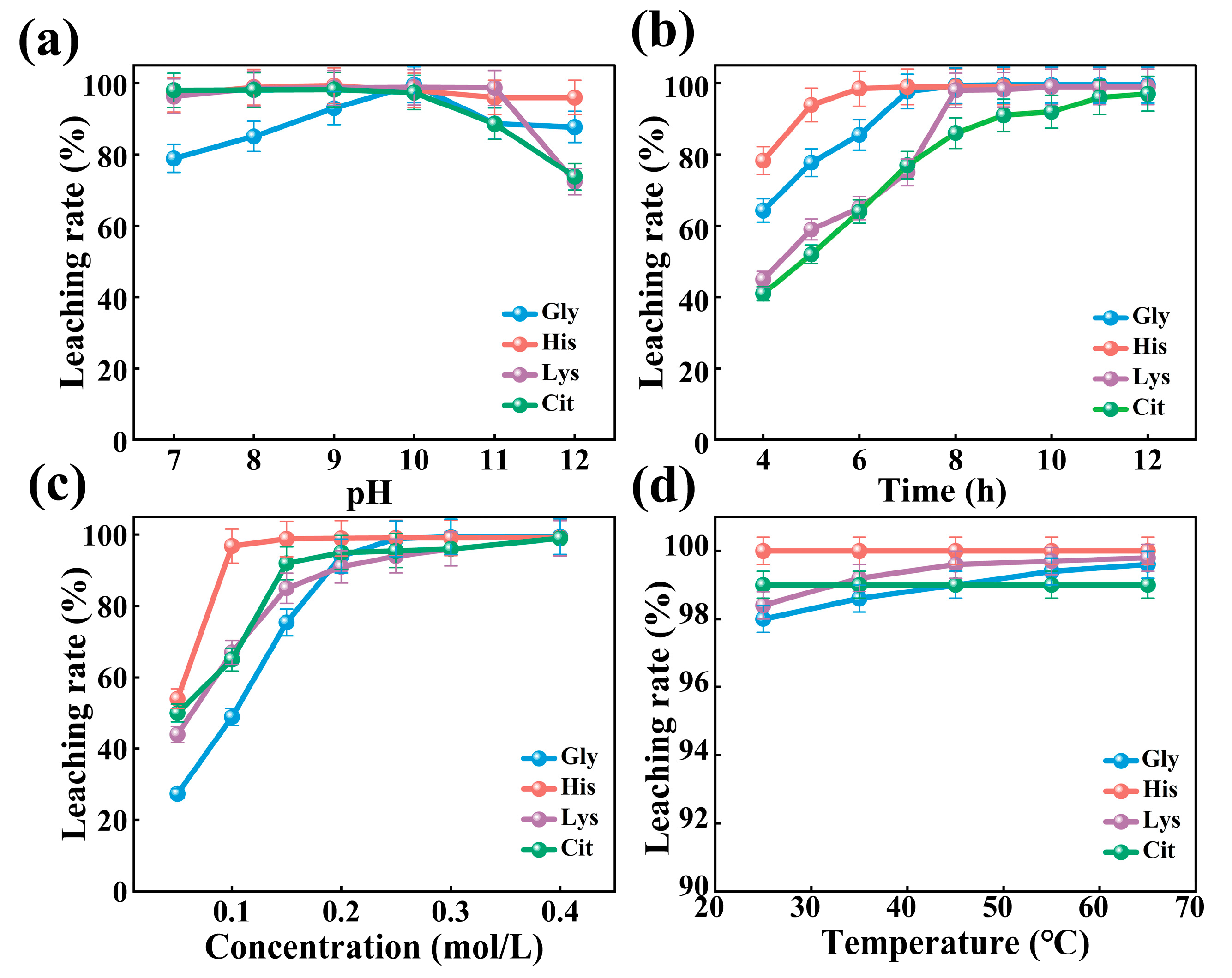
3.1. Comparison of the Leaching Effects of Different Amino Acids on Cobalt
3.2. Parameter Optimization
3.2.1. Effect of Temperature
3.2.2. Effect of pH
3.2.3. Effect of Stirring Speed
3.2.4. Effect of Histidine Concentration
3.2.5. Effect of His/Co Molar Ratio
3.2.6. Selective Leaching of Cu-Co Alloy Slag
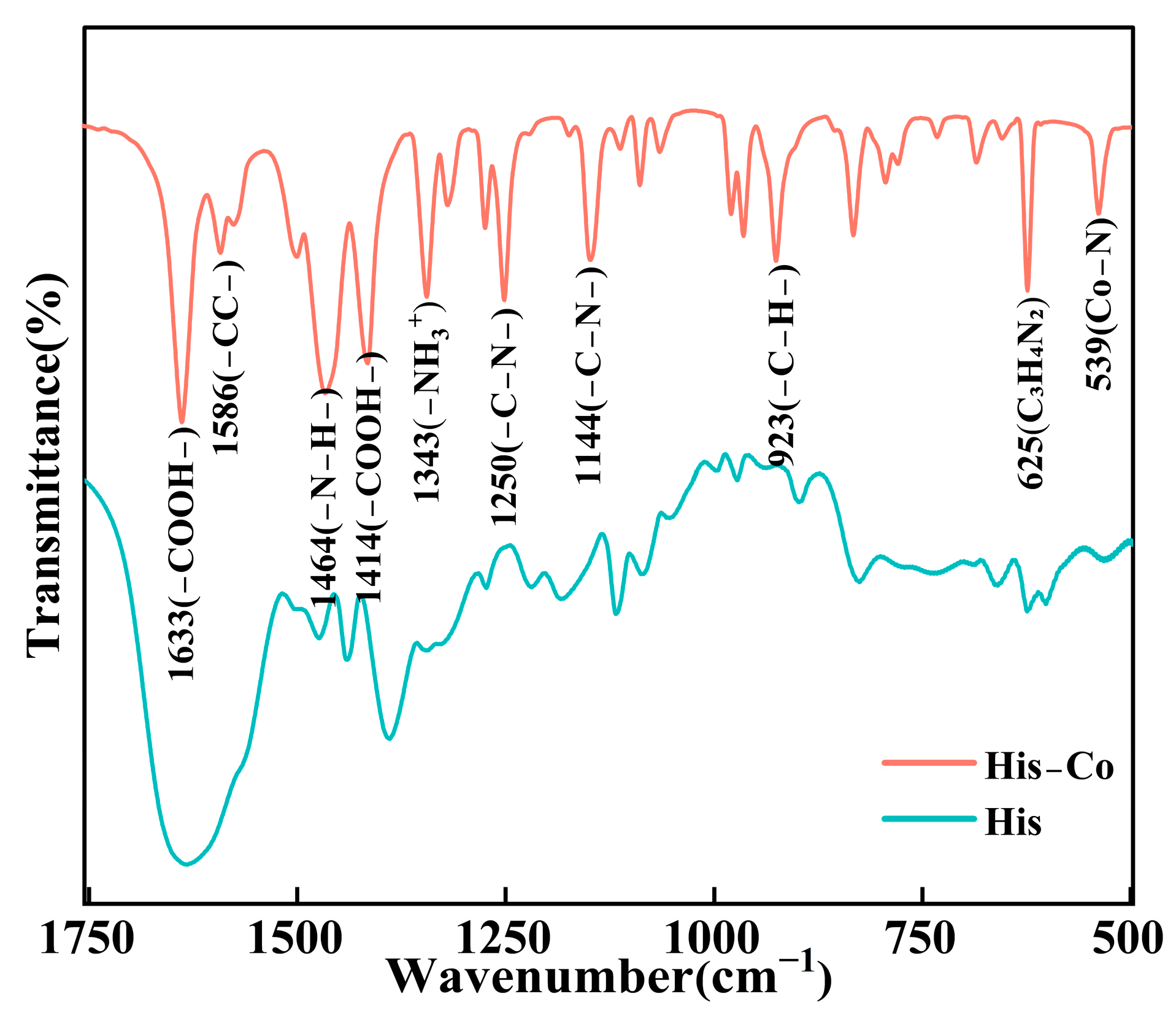
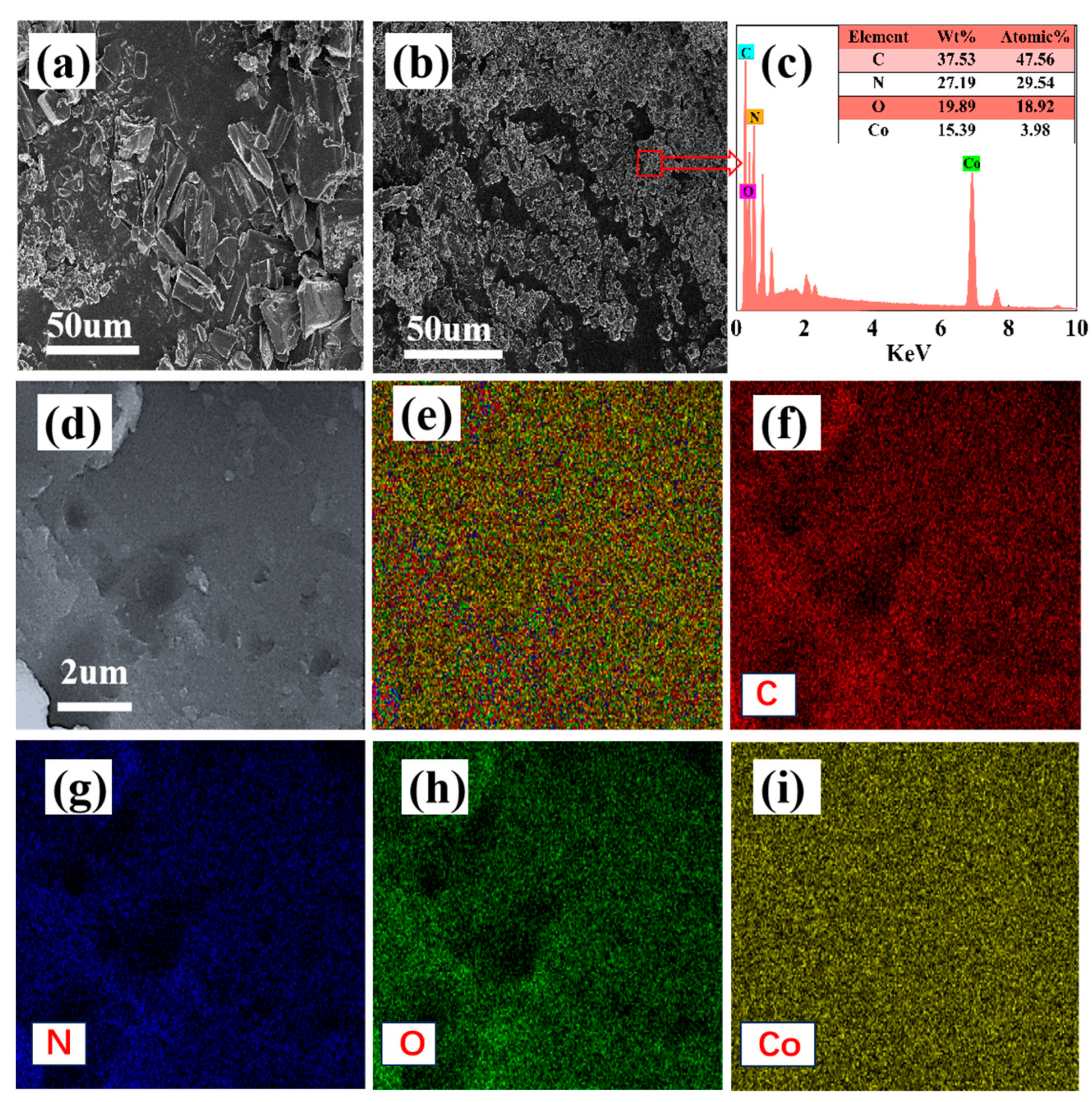
3.3. Characterization Analysis
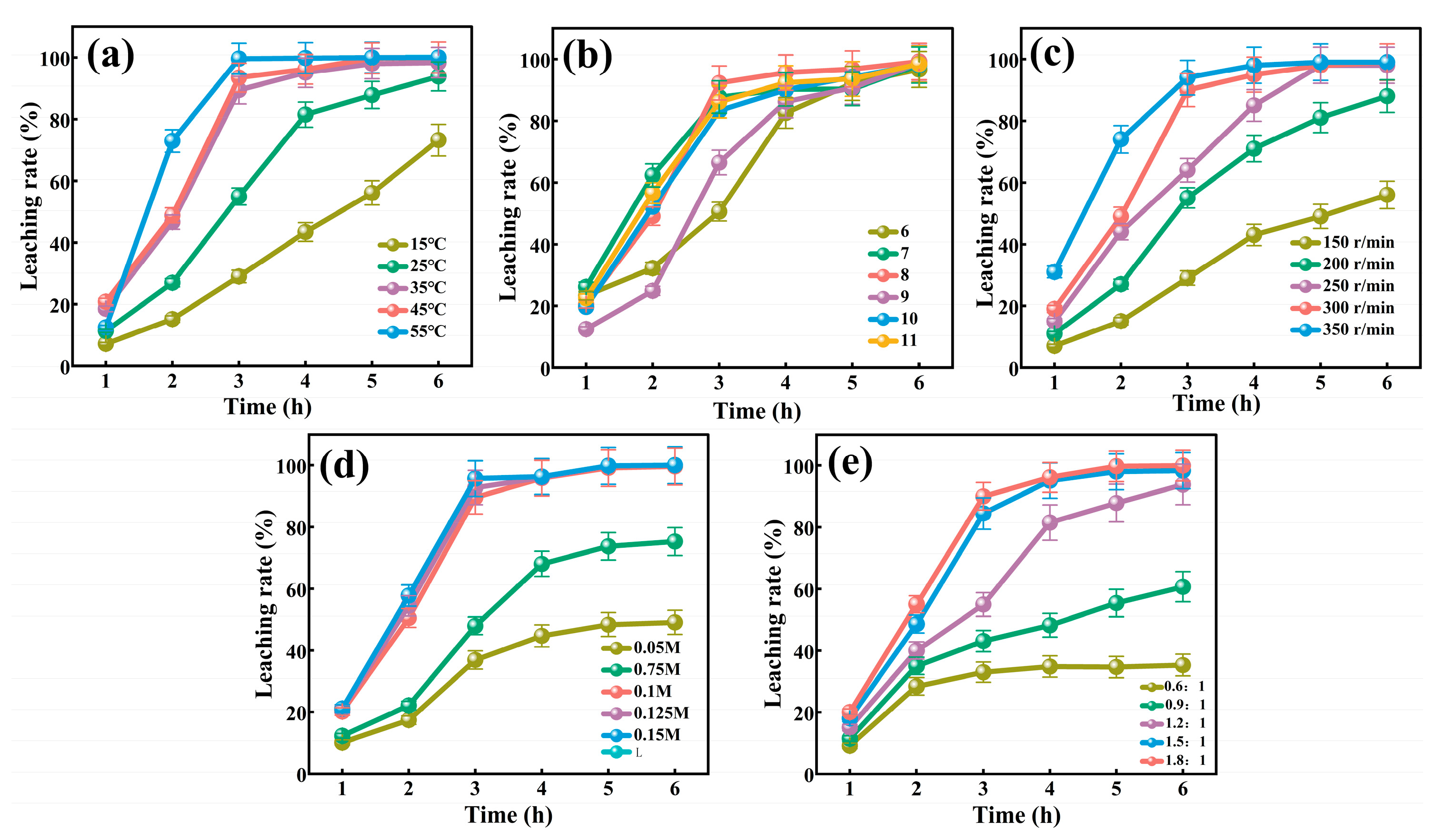
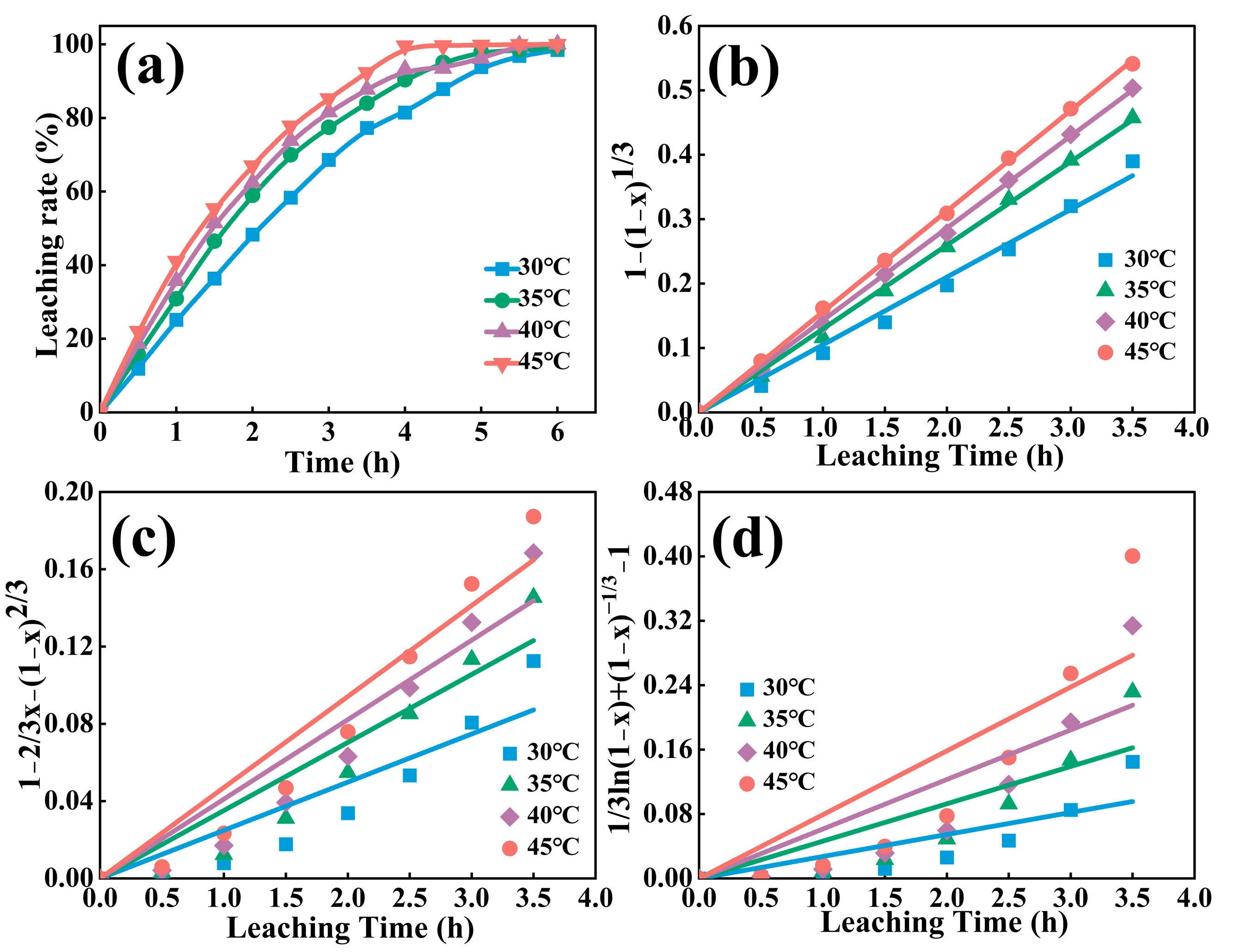
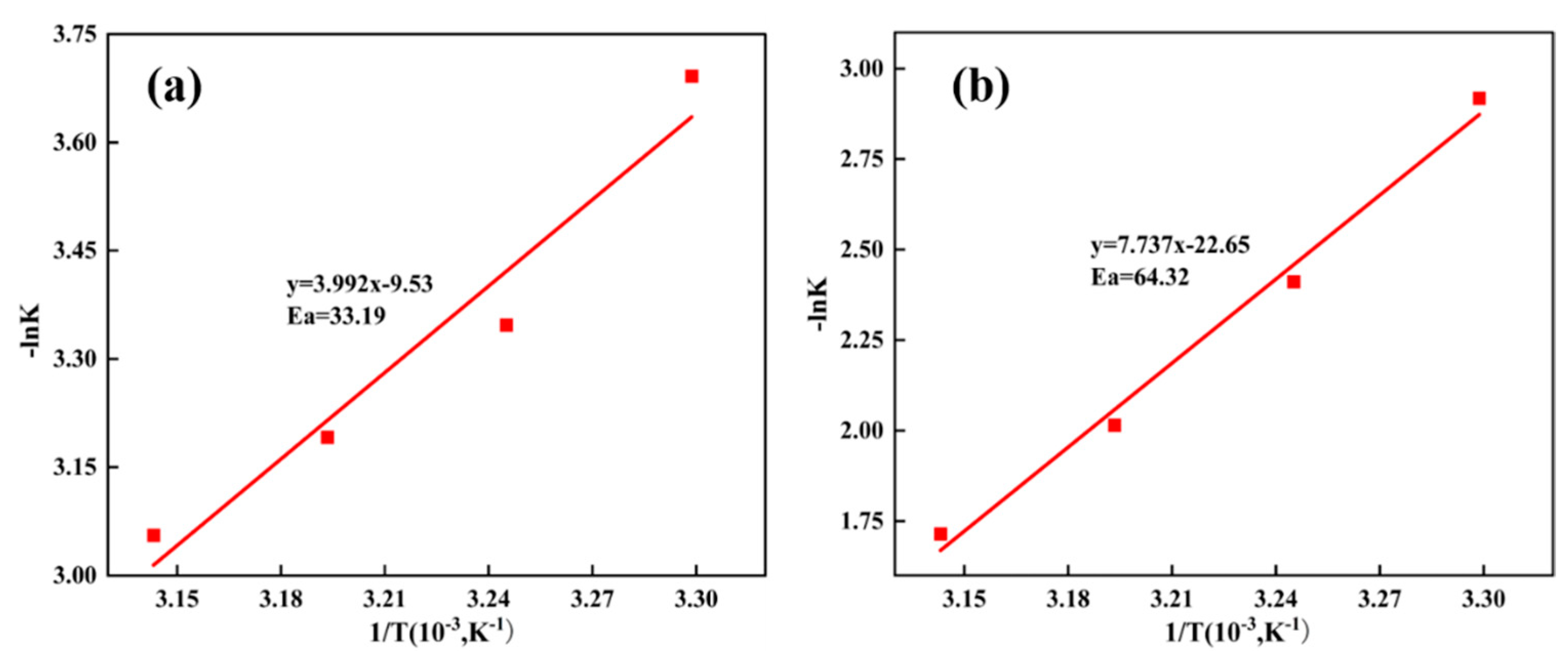
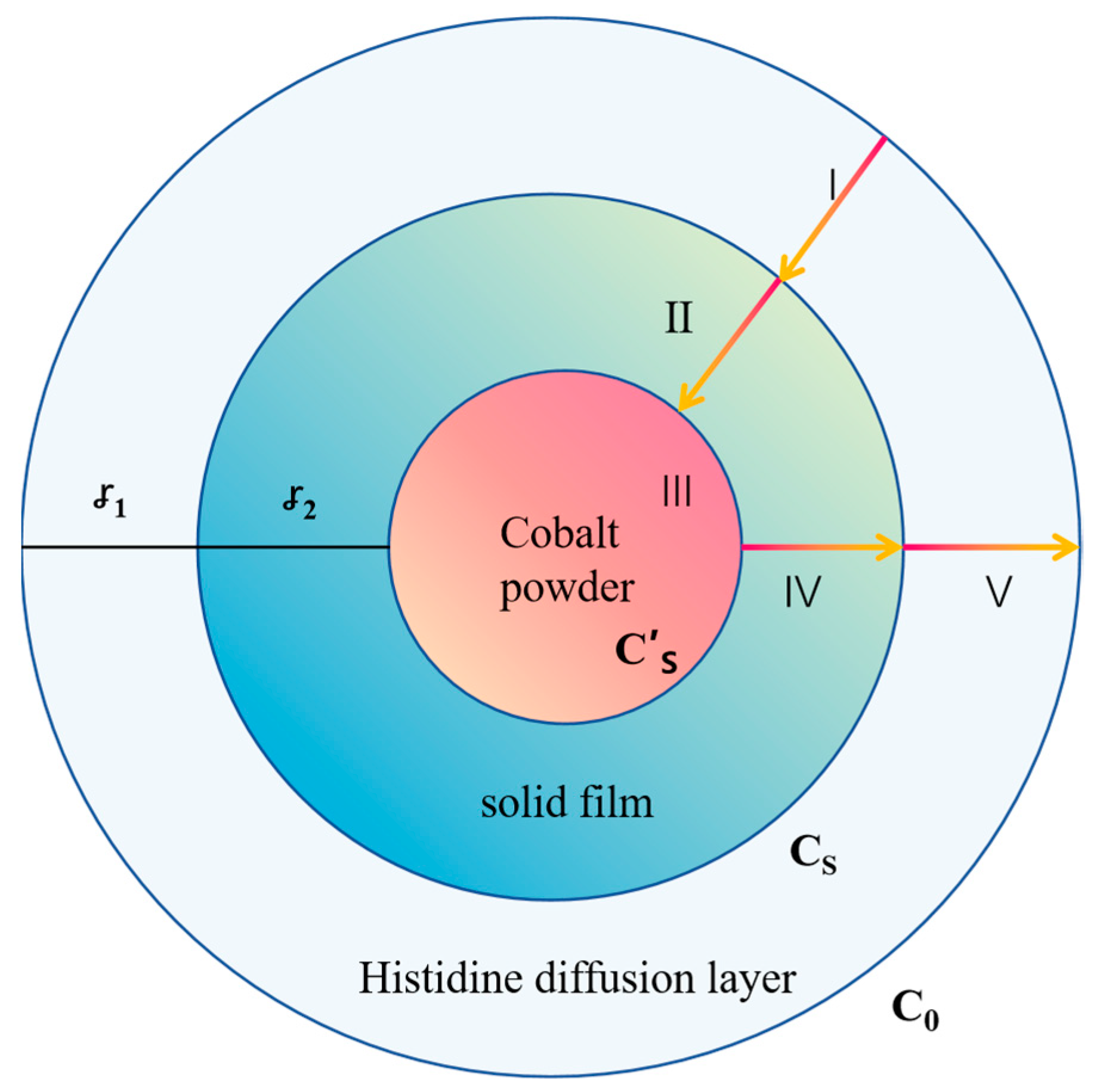
3.4. Leaching Kinetics
3.5. Future Research Prospects
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Oke, E.A.; Potgieter, H.; Mondlane, F.; Skosana, N.P.; Teimouri, S.; Nyembwe, J.K. Concurrent leaching of copper and cobalt from a copper-cobalt ore using sulfuric and organic acids. Miner. Eng. 2024, 216, 108853. [Google Scholar] [CrossRef]
- Zhang, L.; Xiao, W.; Li, G.; Wang, D.; Wu, J.; Du, H.; Lin, Y.; Ye, C.; Qi, T.; Wang, Z. A cleaner and sustainable method for recovering rare earth and cobalt from NdFeB leaching residues. J. Clean. Prod. 2023, 422, 138576. [Google Scholar]
- Huang, Y.; Chen, P.; Shu, X.; Fu, B.; Peng, W.; Liu, J.; Cao, Y.; Zhu, X. Extraction and recycling technologies of cobalt from primary and secondary resources: A comprehensive review. Int. J. Miner. Metall. Mater. 2024, 31, 628–649. [Google Scholar]
- Qin, H.; Makuza, B.; Zhao, J.; Yu, D.; Guo, X.; Tian, Q. Hydrochlorination of Copper-Cobalt Alloy for Efficient Separation of Valuable Metals. J. Sustain. Metall. 2022, 8, 795–805. [Google Scholar]
- Jung, H.; Inaba, Y.; Jiang, V.; West, A.C.; Banta, S. Engineering Polyhistidine Tags on Surface Proteins of Acidithiobacillus ferrooxidans: Impact of Localization on the Binding and Recovery of Divalent Metal Cations. ACS Appl. Mater. Interfaces 2022, 14, 10125–10133. [Google Scholar] [CrossRef]
- Cai, C.; Fajar, A.T.; Hanada, T.; Wakabayashi, R.; Goto, M. Amino Acid Leaching of Critical Metals from Spent Lithium-Ion Batteries Followed by Selective Recovery of Cobalt Using Aqueous Biphasic System. ACS Omega 2023, 8, 3198–3206. [Google Scholar] [PubMed]
- Lin, L.; Lu, Z.; Zhang, W. Recovery of lithium and cobalt from spent Lithium- Ion batteries using organic aqua regia (OAR): Assessment of leaching kinetics and global warming potentials. Resour. Conserv. Recycl. 2021, 167, 105416. [Google Scholar]
- Tian, L.; Gong, A.; Wu, X.; Yu, X.; Xu, Z.; Chen, L. Process and kinetics of the selective extraction of cobalt from high-silicon low-grade cobalt ores using ammonia leaching. Int. J. Miner. Metall. Mater. 2022, 29, 218–227. [Google Scholar]
- Biswal, B.K.; Jadhav, U.U.; Madhaiyan, M.; Ji, L.; Yang, E.H.; Cao, B. Biological Leaching and Chemical Precipitation Methods for Recovery of Co and Li from Spent Lithium-Ion Batteries. ACS Sustain. Chem. Eng. 2018, 6, 12343–12352. [Google Scholar]
- Li, L.; Bian, Y.; Zhang, X.; Guan, Y.; Fan, E.; Wu, F.; Chen, R. Process for recycling mixed-cathode materials from spent lithium-ion batteries and kinetics of leaching. Waste Manag. 2018, 71, 362–371. [Google Scholar] [CrossRef]
- Oraby, E.; Deng, Z.; Li, H.; Eksteen, J. Selective extraction of nickel and cobalt from disseminated sulfide flotation cleaner tailings using alkaline glycine-ammonia leaching solutions. Miner. Eng. 2023, 204, 108418. [Google Scholar] [CrossRef]
- Shi, S.X.; Jiang, S.Q.; Nie, C.C.; Li, B.; Chang, H.H.; Zhu, X.N. Kinetic characteristics and mechanism of copper leaching from waste printed circuit boards by environmental friendly leaching system. Process Saf. Environ. Prot. 2022, 166, 123–132. [Google Scholar] [CrossRef]
- Oraby, E.; Li, H.; Deng, Z.; Eksteen, J. Selective extraction of Ni and Co from a pyrrhotite-rich flotation slime using an alkaline glycine-based leach system. Miner. Eng. 2023, 203, 108330. [Google Scholar] [CrossRef]
- Oraby, E.; Eksteen, J. The selective leaching of copper from a gold–copper concentrate in glycine solutions. Hydrometallurgy 2014, 150, 14–19. [Google Scholar] [CrossRef]
- Chen, M.; Wang, R.; Qi, Y.; Han, Y.; Wang, R.; Fu, J.; Meng, F.; Yi, X.; Huang, J.; Shu, J. Cobalt and lithium leaching from waste lithium ion batteries by glycine. J. Power Sources 2021, 482, 228942. [Google Scholar] [CrossRef]
- Sethurajan, M.; Shirodker, M.G.P.; Rene, E.R.; van Hullebusch, E.D. Hydrometallurgical leaching and recovery of cobalt from lithium ion battery. Environ. Technol. Innov. 2022, 28, 102915. [Google Scholar] [CrossRef]
- Eksteen, J.; Oraby, E.; Nguyen, V. Leaching and ion exchange based recovery of nickel and cobalt from a low grade, serpentine-rich sulfide ore using an alkaline glycine lixiviant system. Miner. Eng. 2020, 145, 106073. [Google Scholar] [CrossRef]
- Li, H.; Deng, Z.; Oraby, E.; Eksteen, J. Amino acids as lixiviants for metals extraction from natural and secondary resources with emphasis on glycine: A literature review. Hydrometallurgy 2023, 216, 106008. [Google Scholar] [CrossRef]
- Sun, Y.; Shi, Y.; Li, C.; Shi, H. Histidine Protonation Behaviors on Structural Properties and Aggregation Properties of Aβ(1-42) Mature Fibril: Approaching by Edge Effects. J. Phys. Chem. 2024, 128, 7341–7349. [Google Scholar] [CrossRef]
- Essential Amino Acids. Findings from University Erlangen-Nurnberg Has Provided New Data on Essential Amino Acids (A facile UV-light mediated synthesis of L-histidine stabilized silver nanocluster for efficient photodegradation of methylene blue). Sci. Lett. 2015, 404, 27–35. [Google Scholar]
- Li, H.; Oraby, E.; Eksteen, J. An alternative amino acid leaching of base metals from waste printed circuit boards using alkaline glutamate solutions: A comparative study with glycine. Sep. Purif. Technol. 2025, 356, 129953. [Google Scholar]
- Liu, Z.; Zu, Y.; Fu, Y.; Meng, R.; Guo, S.; Xing, Z.; Tan, S. Hydrothermal synthesis of histidine-functionalized single-crystalline gold nanoparticles and their pH-dependent UV absorption characteristic. Colloids Surf. B Biointerfaces 2009, 76, 311–316. [Google Scholar] [CrossRef] [PubMed]
- Nidya, M.; Umadevi, M.; Rajkumar, B.J. Optical and morphological studies of L-histidine functionalised silver nanoparticles synthesised by two different methods. J. Exp. Nanosci. 2015, 10, 167–180. [Google Scholar]
- Lin, S.M.; Geng, S.; Li, N.; Liu, S.G.; Li, N.B.; Luo, H.Q. l-Histidine-protected copper nanoparticles as a fluorescent probe for sensing ferric ions. Sens. Actuators B. Chem. 2017, 252, 912–918. [Google Scholar]
- Eksteen, J.; Oraby, E. The leaching and adsorption of gold using low concentration amino acids and hydrogen peroxide: Effect of catalytic ions, sulphide minerals and amino acid type. Miner. Eng. 2015, 70, 36–42. [Google Scholar] [CrossRef]
- Armentrout, P.B.; Stevenson, B.C.; Ghiassee, M.; Boles, G.C.; Berden, G.; Oomens, J. Infrared multiple-photon dissociation spectroscopy of cationized glycine: Effects of alkali metal cation size on gas-phase conformation. Phys. Chem. Chem. Phys. 2022, 24, 37. [Google Scholar]
- Lei, T.; Shu, J.; Deng, Y.; Hu, L.; Chen, S.; Chen, M.; Huang, W. Enhanced recovery of copper from reclaimed copper smelting fly ash via leaching and electrowinning processes. Sep. Purif. Technol. 2021, 273, 118943. [Google Scholar]
- Boukaoud, A.; Chiba, Y.; Sebbar, D. A periodic DFT study of IR spectra of amino acids: An approach toward a better understanding of the N-H and O-H stretching regions. Vib. Spectrosc. 2021, 116, 103280. [Google Scholar]
- Pyreu, D.; Gridchin, S. Mixed-Ligand Nickel(II) Complexes with Histidine and Cysteine in Aqueous Solution: Thermodynamic Approach. J. Solut. Chem. 2023, 53, 372–385. [Google Scholar]
- Tian, H.; Guo, Z.; Pan, J.; Zhu, D.; Yang, C.; Xue, Y.; Li, S.; Wang, D. Comprehensive review on metallurgical recycling and cleaning of copper slag. Resour. Conserv. Recycl. 2021, 168, 105366. [Google Scholar] [CrossRef]
- Paiva, F.; Batista, J.; Rêgo, F.; Lima, J.; Freire, P.; Melo, F.; Filho, J.M.; de Menezes, A.; Nogueira, C. Infrared and Raman spectroscopy and DFT calculations of DL amino acids: Valine and lysine hydrochloride. J. Mol. Struct. 2017, 1127, 419–426. [Google Scholar]
- Haripriya, M.; Manimekala, T.; Dharmalingam, G.; Minakshi, M.; Sivasubramanian, R.J.C.A.A.J. Asymmetric Supercapacitors Based on ZnCo2O4 Nanohexagons and Orange Peel Derived Porous Carbon Electrodes. Chem. Asian J. 2024, 19, e202400202. [Google Scholar] [PubMed]
- Ayyanusamy, P.; Alphonse, R.; Minakshi, M.; Sivasubramanian, R. Synthesis of amorphous nickel-cobalt hydroxides for Ni-Zn rechargeable aqueous battery. Chemistry 2024, 30, e202402325. [Google Scholar] [CrossRef]
- Amino Acids-Histidine. New Histidine Findings from University of Tanta Reported (Nano-sized metal complexes of azo L-histidine: Synthesis, characterization and their application for catalytic oxidation of 2-amino phenol). Chem. Chem. 2018, 32, 4229. [Google Scholar]
- Amino Acids-Histidine. Findings from Y. He and Colleagues Update Understanding of Histidine (Histidine and histidine dimer as green inhibitors for carbon steel in 3wt% sodium chloride solution; Electrochemical, XPS and Quantum chemical calculation studies). J. Eng. 2018, 13, 2136–2153. [Google Scholar]
- Technology—Vacuum Technology. Study Findings from D. Moszynski et al. Broaden Understanding of Vacuum Technology (XPS study of cobalt-ceria catalysts for ammonia synthesis—The reduction process). Chem. Chem. 2018, 155, 434–438. [Google Scholar]
- Han, Y.; Yi, X.; Wang, R.; Huang, J.; Chen, M.; Sun, Z.; Sun, S.; Shu, J. Copper extraction from waste printed circuit boards by glycine. Sep. Purif. Technol. 2020, 253, 117463. [Google Scholar]
- Hosseinzadeh, M.; Entezari Zarandi, A.; Pasquier, L.C.; Azizi, A. Kinetic Investigation on Leaching of Copper from a Low-Grade Copper Oxide Deposit in Sulfuric Acid Solution: A Case Study of the Crushing Circuit Reject of a Copper Heap Leaching Plant. J. Sustain. Metall. 2021, 7, 1154–1168. [Google Scholar]
- Bidari, E.; Aghazadeh, V. Investigation of Copper Ammonia Leaching from Smelter Slags: Characterization, Leaching and Kinetics. Metall. Mater. Trans. 2015, 46, 2305–2314. [Google Scholar]
- Tanda, B.C.; Oraby, E.A.; Eksteen, J.J. Kinetics of malachite leaching in alkaline glycine solutions. Miner. Process. Extr. Metall. 2021, 130, 16–24. [Google Scholar]
- Apua, M.C.; Madiba, M.S. Leaching kinetics and predictive models for elements extraction from copper oxide ore in sulphuric acid. J. Taiwan Inst. Chem. Eng. 2021, 121, 313–320. [Google Scholar] [CrossRef]



| Elements | Co | Cu | Fe | Pb | Ni |
|---|---|---|---|---|---|
| Wt.% | 6.2 | 30.7 | 61.2 | 1.4 | 0.42 |
| Temperature (°C) | Internal Diffusion Control 1 − (2/3)x − (1 − x)2/3 | Chemical Reaction Control 1 − (1 − x)1/3 | Mixed Control 1/3ln(1 − x) + (1 − x)−1/3 − 1 | |||
|---|---|---|---|---|---|---|
| k1 (min−1) | R2 | k2 (min−1) | R2 | k2 (min−1) | R2 | |
| 30 | 0.02493 | 0.846 | 0.05406 | 0.989 | 0.02732 | 0.716 |
| 35 | 0.03519 | 0.902 | 0.08991 | 0.998 | 0.04637 | 0.765 |
| 40 | 0.04110 | 0.911 | 0.13334 | 0.999 | 0.06148 | 0.747 |
| 45 | 0.04709 | 0.929 | 0.17980 | 0.999 | 0.07924 | 0.752 |
| Control Model | Slope k′ | Correlation Coefficient (R2) | Ea (kJ/mol) |
|---|---|---|---|
| Chemical reaction control | 7.74 | 0.99 | 64.32 |
| Internal diffusion control | 3.99 | 0.95 | 33.19 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, M.; Wang, Q.; Guo, W.; Zhao, X.; Zhang, Y.; Zhou, X.; Lei, Z.; Zhang, Y. Selective Complexation and Leaching of Cobalt Using Histidine in an Alkaline Medium. Processes 2025, 13, 1039. https://doi.org/10.3390/pr13041039
Li M, Wang Q, Guo W, Zhao X, Zhang Y, Zhou X, Lei Z, Zhang Y. Selective Complexation and Leaching of Cobalt Using Histidine in an Alkaline Medium. Processes. 2025; 13(4):1039. https://doi.org/10.3390/pr13041039
Chicago/Turabian StyleLi, Mengying, Qingliang Wang, Weiduo Guo, Xu Zhao, Yaolong Zhang, Xiankun Zhou, Zhiwu Lei, and Yahui Zhang. 2025. "Selective Complexation and Leaching of Cobalt Using Histidine in an Alkaline Medium" Processes 13, no. 4: 1039. https://doi.org/10.3390/pr13041039
APA StyleLi, M., Wang, Q., Guo, W., Zhao, X., Zhang, Y., Zhou, X., Lei, Z., & Zhang, Y. (2025). Selective Complexation and Leaching of Cobalt Using Histidine in an Alkaline Medium. Processes, 13(4), 1039. https://doi.org/10.3390/pr13041039







