Production of Butyric Anhydride Using Single Reactive Distillation Column with Internal Material Circulation
Abstract
:1. Introduction
2. Reaction System
2.1. Synthesis of Butyric Anhydride
2.2. Reaction Kinetics
2.3. Thermodynamics Data
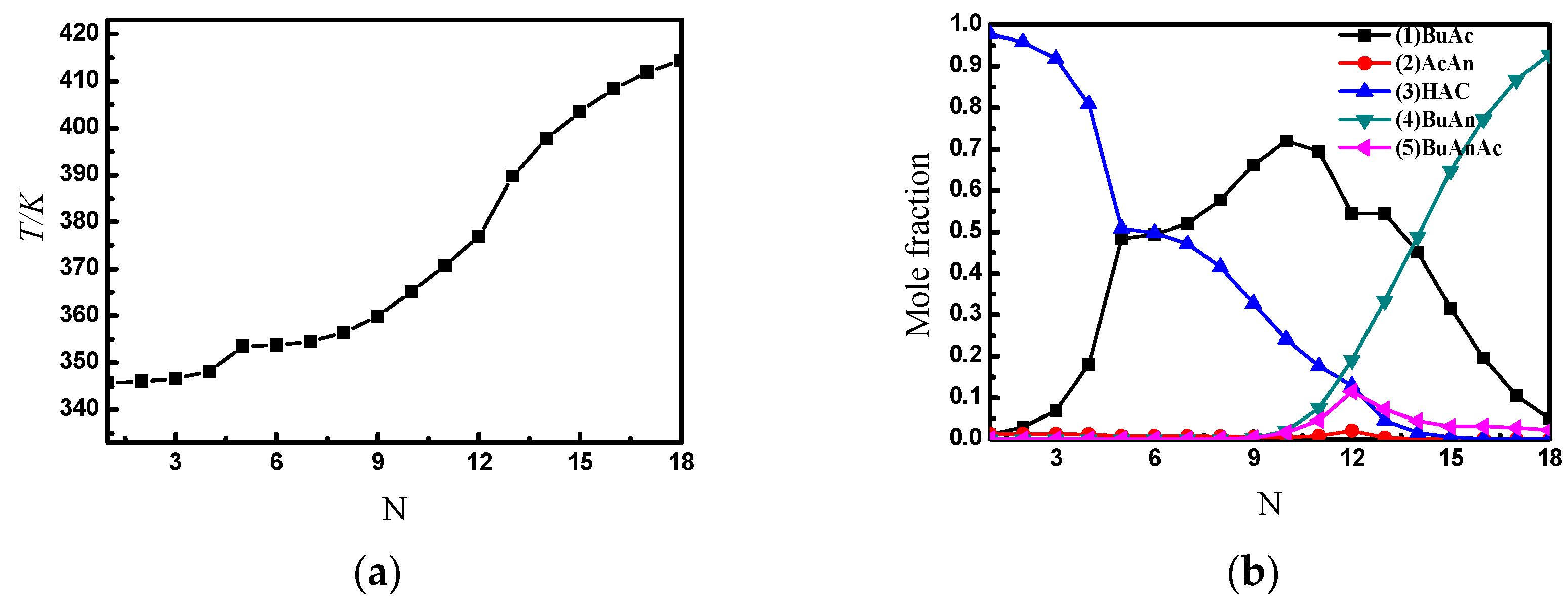
3. Simulation of Two-Column RD Process for the Production of Butyric Anhydride
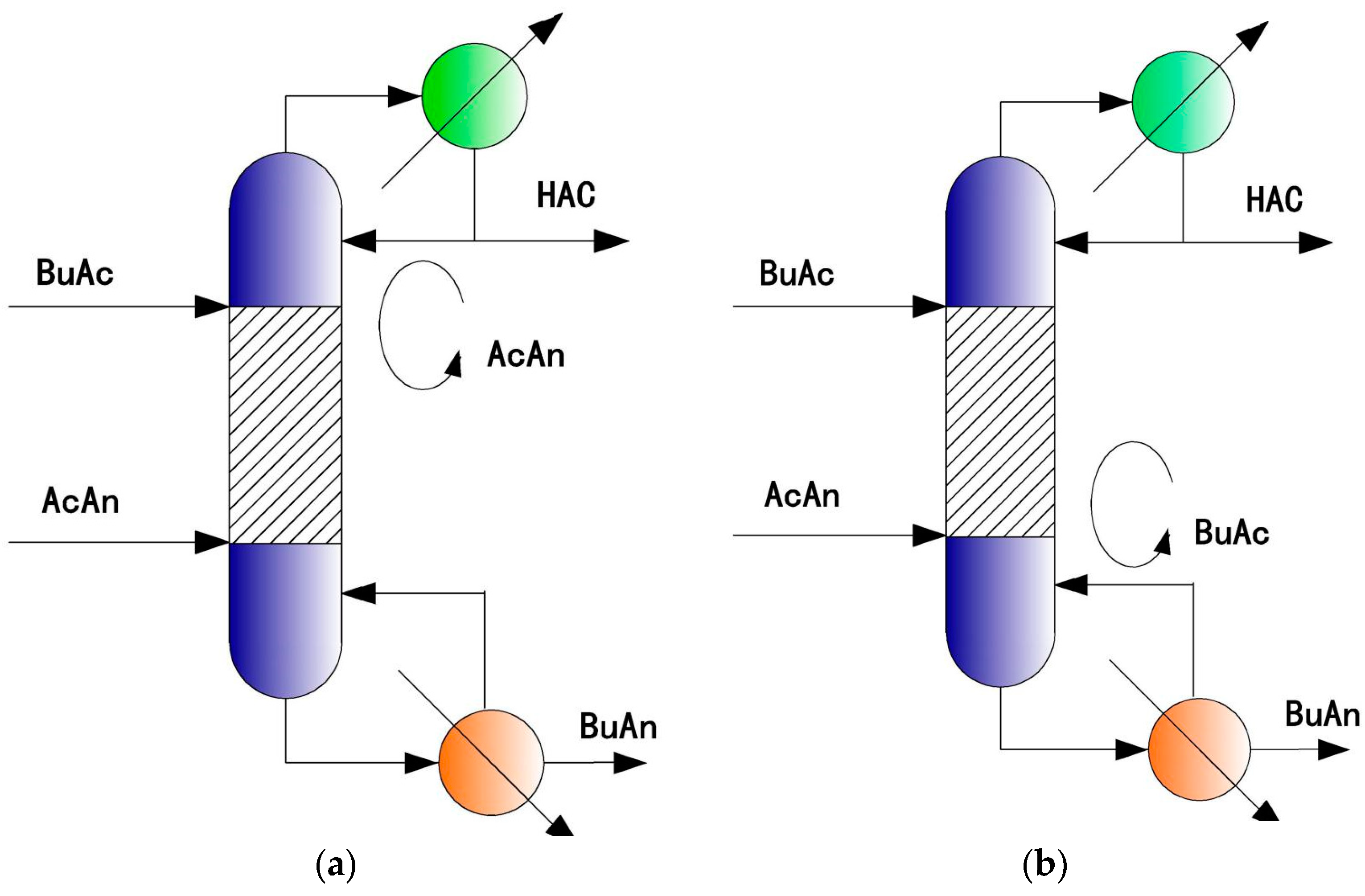
4. Single RD Column with Internal Material Circulation
4.1. Principle and Two Applications
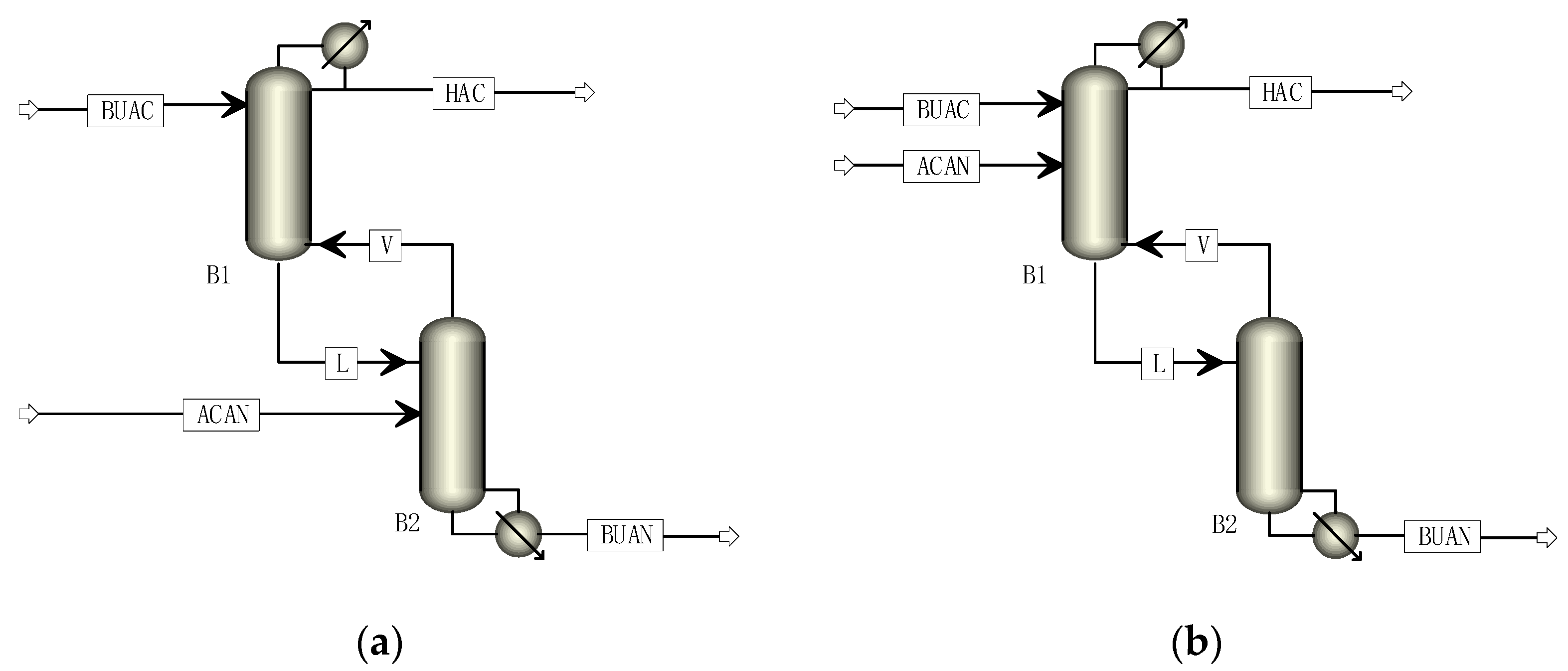
4.2. Simulation of the SRDC Process
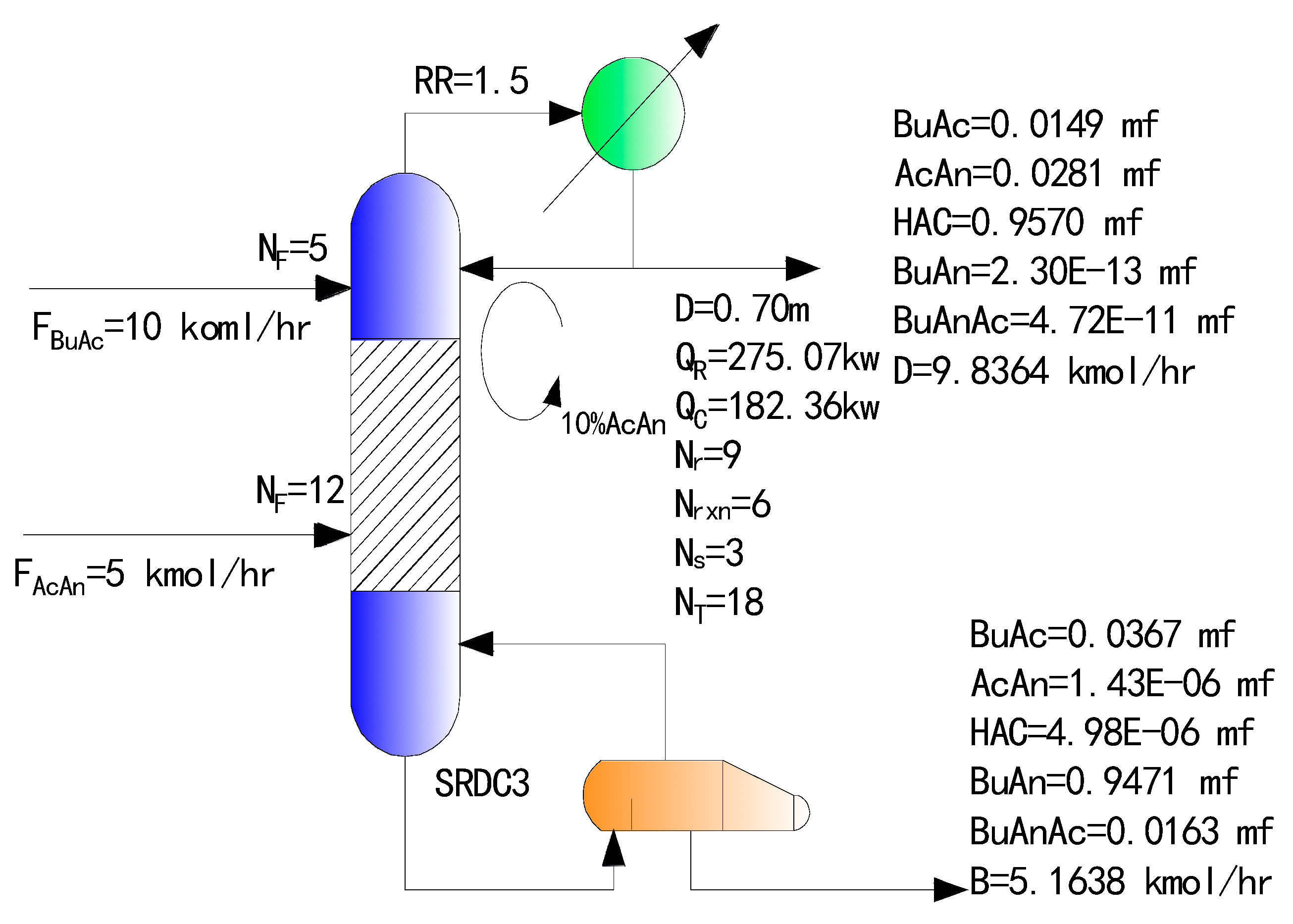
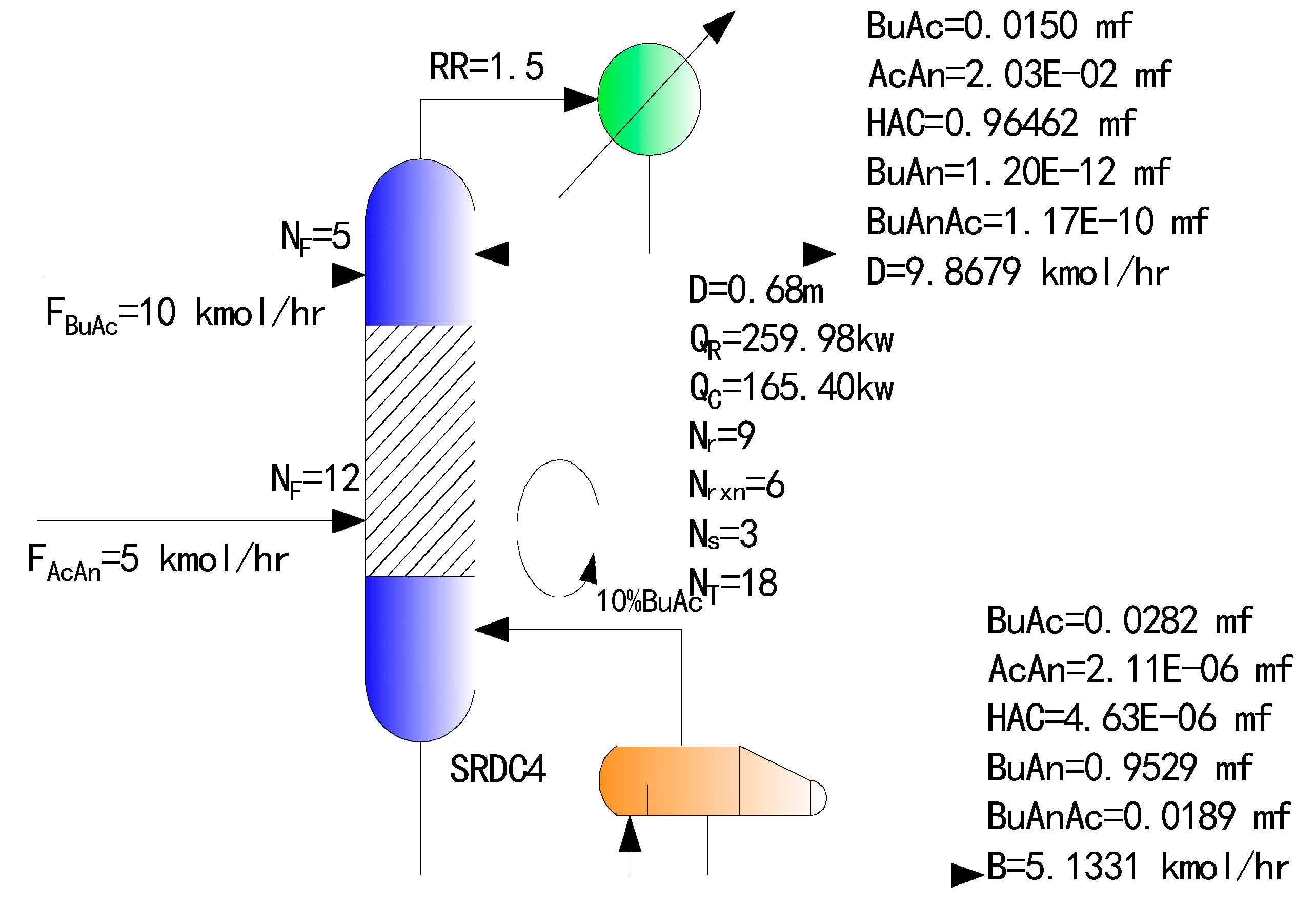
4.3. Simulation Results of the Optimized SRDC
4.4. Economic Comparison
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Nomenclature
| AcAn | Acetic Anhydride |
| AC | the heat transfer area of condenser |
| AR | the heat transfer area of reboiler |
| B | bottom rate |
| BuAc | butyric acid |
| BuAn | butyric anhydride |
| BuAnAc | butyric anhydride acetate |
| D | distillate rate |
| F | the fresh feed of the reactive distillation column |
| HAC | acetic acid |
| HX | heater exchanger |
| IDr | the internal diameter of the rectifying section |
| IDrxn | the internal diameter of the reaction section |
| IDs | the internal diameter of the stripping section |
| kn + | the forward reaction rate constants, dm3·mol−1·min−1 |
| kn− | the backward reaction rate constants, dm3·mol−1·min−1 |
| N | the number of trays |
| NT | the total number of trays |
| NF | the feed stage of the fresh feed |
| Nr | the number of trays in rectifying section |
| Nrxn | the number of trays in reactive section |
| Ns | the number of trays in stripping section |
| NT | the total number of trays |
| QR | reboiler duty |
| QC | condenser duty |
| RR | the reflux ratio |
| RF | the recycled flow of BuAc |
| RD | reactive distillation |
| SRDC | single reactive distillation column |
| TAC | total annual cost |
| Tb | boiling point |
References
- Gao, F. Development of Continuous Reactive Distillation Process for Production of Butyric Anhydride. Master’s Thesis, Qingdao University of Science and Technology, Qingdao, China, 2007. [Google Scholar]
- Sahlodin, A.-M.; Barton, P.-I. Optimal Campaign Continuous Manufacturing. Ind. Eng. Chem. Res. 2015, 54, 11344–11359. [Google Scholar] [CrossRef]
- Tian, H.; Zhao, S.; Zheng, H.; Haung, Z. Optimization of coproduction of ethyl acetate and butyl acetate by reactive distillation. Chin. J. Chem. Eng. 2015, 23, 667–674. [Google Scholar] [CrossRef]
- Mallaiah, M.; Kishore, K.-A.; Reddy, G.-V. Catalytic Reactive Distillation for the Esterification Process: Experimental and Simulation. Chem. Biochem. Eng. Q. 2017, 31, 293–302. [Google Scholar] [CrossRef]
- Xu, H.; Ye, Q.; Zhang, H.; Qin, J.-W.; Li, N. Design and control of reactive distillation–recovery distillation flowsheet with a decanter for synthesis of N-propyl propionate. Chem. Eng. Process. 2014, 85, 38–47. [Google Scholar] [CrossRef]
- Gonzalez, D.-R.; Bastidas, P.; Rodriguez, G.; Gil, I. Design alternatives and control performance in the pilot scale production of isoamyl acetate via reactive distillation. Chem. Eng. Res. Des. 2017, 123, 347–359. [Google Scholar] [CrossRef]
- Chung, Y.-H.; Peng, T.-H.; Lee, H.-Y.; Chen, C.-L.; Chien, I.-L. Design and control of reactive distillation system for esterification of levulinic Acid and n-Butanol. Ind. Eng. Chem. Res. 2015, 54, 3341–3354. [Google Scholar] [CrossRef]
- Chua, W.-J.; Rangaiah, G.-P.; Hidajat, K. Design and optimization of isopropanol process based on two alternatives for reactive distillation. Chem. Eng. Process. 2017, 118, 108–116. [Google Scholar] [CrossRef]
- Li, B.-C.; Han, X.-P.; Zhang, W.-L.; Geng, C.-X. Simulation and experimental research on butyric anhydride synthesis by reactive distillation. J. Chem. Ind. Eng. 2017, 45, 72–78. [Google Scholar]
- Li, B.-C.; Xue, X.-X.; Zhang, W.-L.; Geng, C.-X. Study on the kinetics of preparation of butyric anhydride by acylation. J. Chem. Ind. Eng. 2017, 45, 49–53. [Google Scholar]
- Renon, H.; Prausnitz, J.-M. Local compositions in thermodynamic excess functions for liquid mixtures. AI ChE J. 1968, 14, 135–144. [Google Scholar]
- Luyben, W.-L. Design and control of a methanol reactor/column process. Ind. Eng. Chem. Res. 2010, 49, 6150–6163. [Google Scholar] [CrossRef]


| Component ID | Component Name | Formula | Tb/K |
|---|---|---|---|
| BuAc | butyric acid | C3H7COOH | 389.15 |
| AcAn | acetic anhydride | (CH3CO)2O | 364.15 |
| HAC | acetic acid | CH3COOH | 348.15 |
| BuAn | butyric anhydride | (C3H7CO)2O | 417.15 |
| BuAnAc | Butyric anhydride acetate | C3H7(CO3C)CH3 | 413.15 |
| Component j | AcAn | HAC | HAC |
|---|---|---|---|
| Component i | BuAc | BuAc | AcAn |
| source | VLE-HOC | VLE-HOC | VLE-HOC |
| temp. unit | K | K | K |
| Aij | 0 | −0.564 | 0.8146 |
| Aji | 0 | 1.7591 | −0.5218 |
| Bij | 20.9697 | 0 | −147.1604 |
| Bji | 608.8256 | 0 | 332.2892 |
| Cij | 0.3 | 0.3 | 0.3 |
| Parameter | Traditional Two-Column RD Process | SRDC3 | SRDC4 | ||
|---|---|---|---|---|---|
| Column C1 | Column C2 | ||||
| Conversion of BuAc/% | 95.37 | 95.69 | 96.40 | ||
| Conversion of AcAn/% | 96.40 | 96.70 | 97.60 | ||
| Mole fraction of bottom/% | BuAc | 3.97 | 3.67 | 2.80 | |
| AcAn | 2.11 × 10−6 | 1.43 × 10−4 | 2.11 × 10−4 | ||
| HAC | 4.87 × 10−6 | 4.98 × 10−4 | 4.63 × 10−4 | ||
| BuAn | 94.32 | 94.71 | 95.29 | ||
| BuAnAc | 1.71 | 1.63 | 1.89 | ||
| Mole fraction of top/% | BuAc | 1.57 | 1.49 | 1.50 | |
| AcAn | 3.05 | 2.81 | 2.03 | ||
| HAC | 95.37 | 95.70 | 96.46 | ||
| BuAn | 1.59 × 10−5 | 2.3 × 10−11 | 1.20 × 10−10 | ||
| BuAnAc | 2.27 × 10−5 | 4.72 × 10−9 | 1.17 × 10−8 | ||
| Temperature/K | Bottom | 408.24 | 413.55 | 414.11 | 414.38 |
| Top | 345.77 | 391.50 | 345.76 | 345.76 | |
| Parameter | Traditional Two-Column RD Process | SRDC3 | SRDC4 | |
|---|---|---|---|---|
| Column C1 | Column C2 | |||
| IDr (m) | 0.79 | 0.53 | 0.79 | 0.68 |
| IDrxn (m) | 0.68 | - | 0.70 | 0.67 |
| IDs (m) | 0.90 | - | 0.89 | 0.95 |
| Total number of trays (NT) | 18 | 8 | 18 | 18 |
| Condenser duty (QC) (km) | 165.44 | 78.49 | 182.36 | 165.40 |
| Heat transfer area of condenser (AC) (m2) | 5.49 | 1.13 | 5.49 | 5.49 |
| Reboiler duty (QR) (km) | 256.10 | 80.28 | 275.07 | 256.98 |
| Heat transfer area of reboiler (AR) (m2) | 18.10 | 7.21 | 23.39 | 23.84 |
| Shell cost (106 $) | 0.096 | 0.027 | 0.097 | 0.090 |
| Heater exchanger (HX) cost (106 $) | 0.070 | 0.034 | 0.085 | 0.085 |
| Energy cost (106 $/year) | 0.058 | 0.018 | 0.062 | 0.058 |
| Capital (106 $) | 0.166 | 0.061 | 0.182 | 0.175 |
| Total annual cost (TAC) (106 $/year) | 0.152 | 0.123 | 0.116 | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, G.; Jin, F.; Guo, X.; Xiang, S.; Tao, S. Production of Butyric Anhydride Using Single Reactive Distillation Column with Internal Material Circulation. Processes 2020, 8, 1. https://doi.org/10.3390/pr8010001
Chen G, Jin F, Guo X, Xiang S, Tao S. Production of Butyric Anhydride Using Single Reactive Distillation Column with Internal Material Circulation. Processes. 2020; 8(1):1. https://doi.org/10.3390/pr8010001
Chicago/Turabian StyleChen, Guanghui, Fushuang Jin, Xiaokai Guo, Shuguang Xiang, and Shaohui Tao. 2020. "Production of Butyric Anhydride Using Single Reactive Distillation Column with Internal Material Circulation" Processes 8, no. 1: 1. https://doi.org/10.3390/pr8010001
APA StyleChen, G., Jin, F., Guo, X., Xiang, S., & Tao, S. (2020). Production of Butyric Anhydride Using Single Reactive Distillation Column with Internal Material Circulation. Processes, 8(1), 1. https://doi.org/10.3390/pr8010001





