Ideal Feedstock and Fermentation Process Improvements for the Production of Lignocellulolytic Enzymes
Abstract
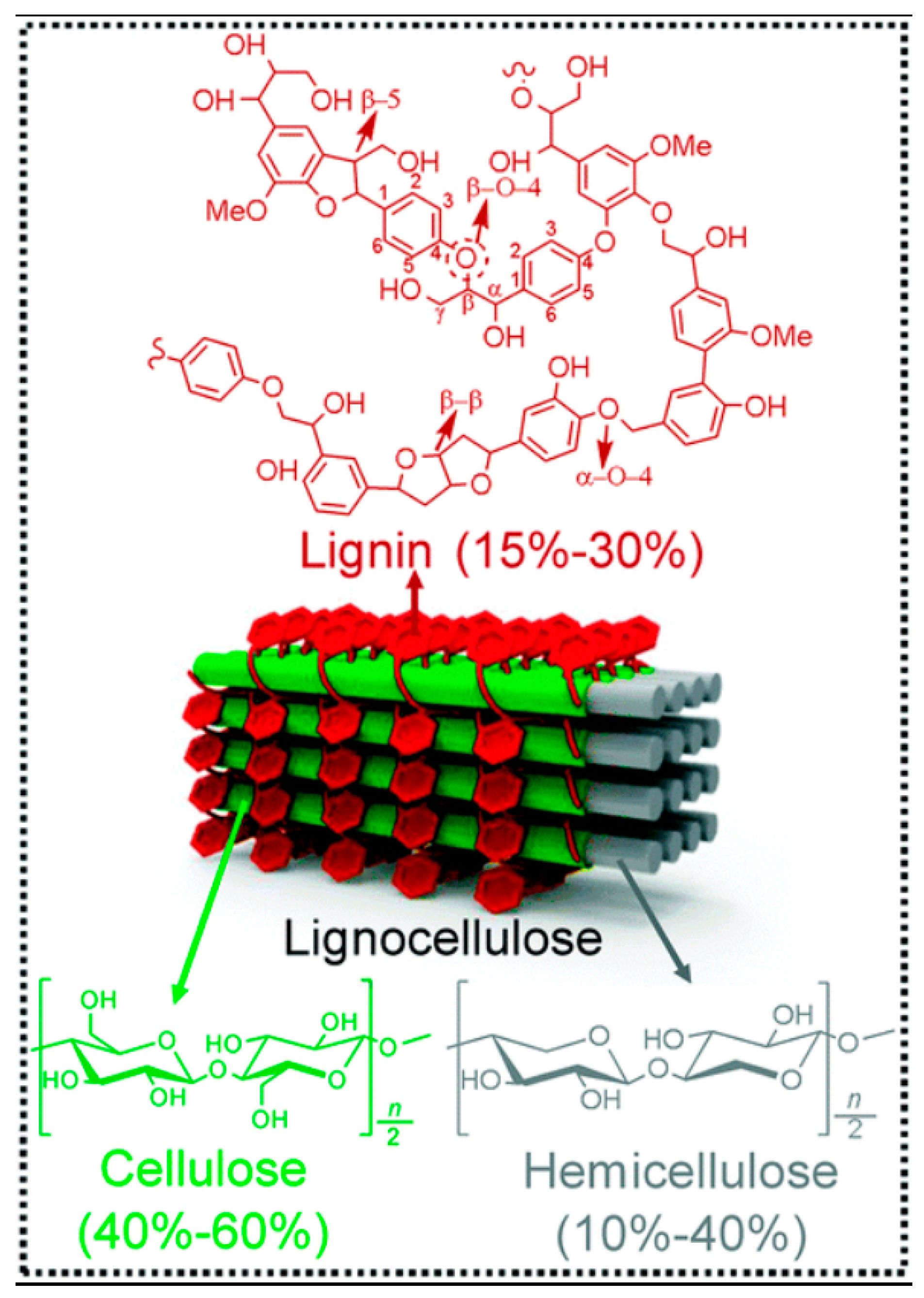
:1. Introduction
2. Lignocellulosic Hydrolytic Enzymes
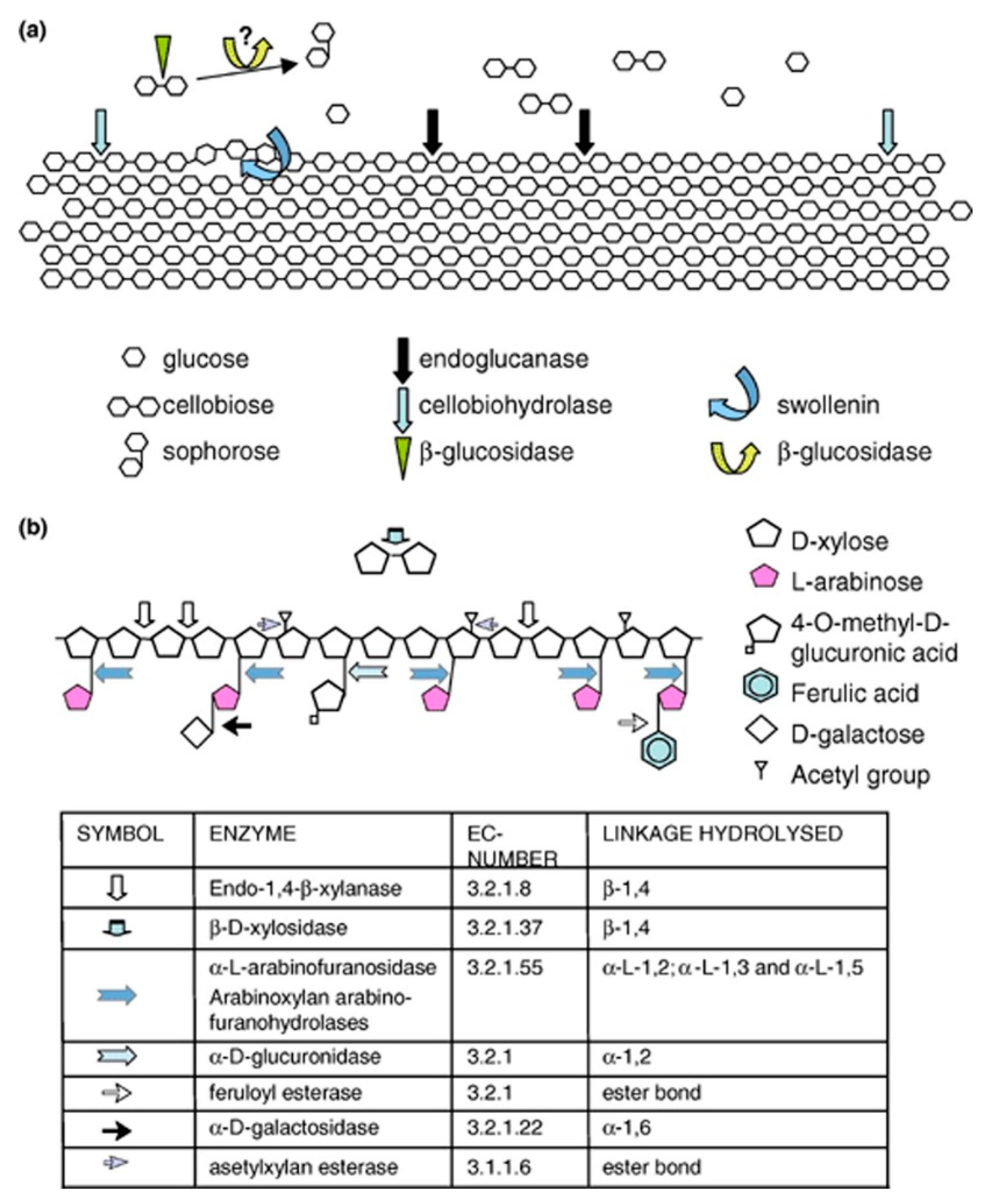
2.1. Cellulases
2.2. Hemicellulases
2.3. Lignases
3. Applications of Lignocellulolytic Enzymes
4. Characteristics of an Ideal Feedstock
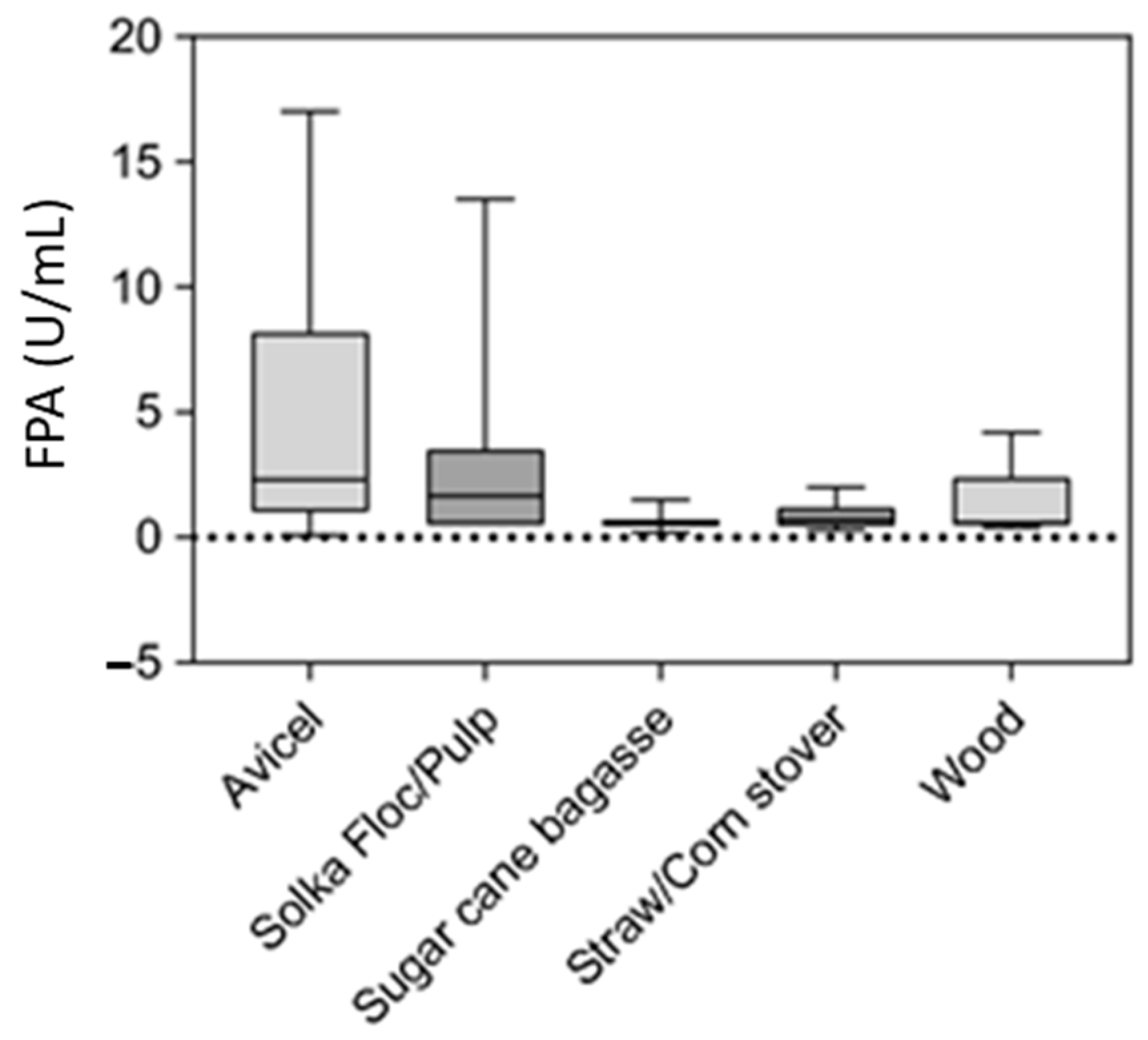
5. Feedstocks for Lignocellulolytic Enzyme Production
6. Pretreatment of Feedstock for Lignocellulolytic Enzyme Production
- The pretreatment method should produce higher amounts of molecular entities for any specific product. For example, in the case of bioethanol production, the main ingredient required for microbial species is glucose or other monosaccharides which are to be converted into ethanol. Acid hydrolysis is one of the chemical methods that can produce higher amounts of simple sugars as compared to other methods [100]. On the other hand, if the final output is the production of lignocellulolytic enzymes such as cellulase, then the presence of reactive cellulosic fibers is required [101]. The physical pretreatment methods are usually employed to remove the lignin barrier so that cellulose and hemicellulose are available for subsequent biochemical reactions.
- The pretreatment method should not degrade the monosaccharides if they are the final product of the pretreatment. Some methods such as acid hydrolysis can degrade the pentoses and hexoses further into furfurals and Trihalomethanes (THMs), which may have an inhibitory effect on fungal activity [100].
- The pretreatment method should also not release any other type of compounds that can inhibit the growth of the microbial species, which are to be employed for the end-use of the lignocellulosic biomass. In this regard, all pretreatment methods should be checked and optimized according to the end-use of the products [102].
- The size and specifications of the pretreatment reactors should also be considered before employing a specific pretreatment method. For example, most of the acid hydrolysis reactions require high temperature and pressure (such as in an autoclave).
- The physical state of the pretreatment output also plays an important role in the determination of a suitable method. For example, if the liquid medium is required for the microbial or any other biochemical reaction, then the presence of solid residues at the end of the pretreatment procedure should be minimal.
- Simplicity of the pretreatment procedure is also required for setting up the reaction at different scales.
- The characteristics of the pretreatment feedstocks should also be taken into account before selecting a specific type of feedstock. For example, in the case of the sugarcane bagasse, chemical pretreatment methods along with steam or liquid hot water can be employed because it does not have high amounts of proteins or lipids. However, if the feedstock has high contents of proteins and lipids, such as distillers dried grains with solubles (DDGS), there is a high chance that such molecular components will also be degraded by the severe pretreatment conditions.
7. Microbial Production of Lignocellulolytic Enzymes
7.1. Modes of Fermentation for Lignocellulolytic Enzyme Production
7.2. Fermentation Enhancement Strategies
8. Challenges of Enzyme Production
9. Conclusions and Future Perspective
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wu, X.; Luo, N.; Xie, S.; Zhang, H.; Zhang, Q.; Wang, F.; Wang, Y. Photocatalytic transformations of lignocellulosic biomass into chemicals. Chem. Soc. Rev. 2020, 49, 6198–6223. [Google Scholar] [CrossRef] [PubMed]
- Cai, J.; He, Y.; Yu, X.; Banks, S.W.; Yang, Y.; Zhang, X.; Yu, Y.; Liu, R.; Bridgwater, A.V. Review of physicochemical properties and analytical characterization of lignocellulosic biomass. Renew. Sustain. Energy Rev. 2017, 76, 309–322. [Google Scholar] [CrossRef] [Green Version]
- Wang, S.; Dai, G.; Yang, H.; Luo, Z. Lignocellulosic biomass pyrolysis mechanism: A state-of-the-art review. Prog. Energy Combust. Sci. 2017, 62, 33–86. [Google Scholar] [CrossRef]
- Sindhu, R.; Binod, P.; Pandey, A. Biological pretreatment of lignocellulosic biomass—An overview. Bioresour. Technol. 2016, 199, 76–82. [Google Scholar] [CrossRef] [PubMed]
- Valdés, G.; Mendonça, R.T.; Aggelis, G. Lignocellulosic biomass as a substrate for oleaginous microorganisms: A review. Appl. Sci. 2020, 10, 7698. [Google Scholar] [CrossRef]
- Iram, A.; Cekmecelioglu, D.; Demirci, A. Distillers’ dried grains with solubles (DDGS) and its potential as the fermentation feedstock. Appl. Microbiol. Biotechnol. 2020, 104, 6115–6128. [Google Scholar] [CrossRef]
- Dos Santos, A.C.; Ximenes, E.; Kim, Y.; Ladisch, M.R. Lignin–enzyme interactions in the hydrolysis of lignocellulosic biomass. Trends Biotechnol. 2019, 37, 518–531. [Google Scholar] [CrossRef]
- Mandels, M.; Reese, E.T. Induction of cellulase in Trichoderma viride as influenced by carbon sources and metals. J. Bacteriol. 1957, 73, 269. [Google Scholar] [CrossRef] [Green Version]
- Kim, Y.; Hendrickson, R.; Mosier, N.S.; Ladisch, M.R.; Bals, B.; Balan, V.; Dale, B.E. Enzyme hydrolysis and ethanol fermentation of liquid hot water and AFEX pretreated distillers’ grains at high-solids loadings. Bioresour. Technol. 2008, 99, 5206–5215. [Google Scholar] [CrossRef]
- Alvira, P.; Tomás-Pejó, E.; Ballesteros, M.; Negro, M.J. Pretreatment technologies for an efficient bioethanol production process based on enzymatic hydrolysis: A review. Bioresour. Technol. 2010, 101, 4851–4861. [Google Scholar] [CrossRef]
- Juhasz, T.; Szengyel, Z.; Reczey, K.; Siika-Aho, M.; Viikari, L. Characterization of cellulases and hemicellulases produced by Trichoderma reesei on various carbon sources. Process Biochem. 2005, 40, 3519–3525. [Google Scholar] [CrossRef]
- Knowles, J.; Lehtovaara, P.; Teeri, T. Cellulase families and their genes. Trends Biotechnol. 1987, 5, 255–261. [Google Scholar] [CrossRef]
- Habte-Tsion, H.-M.; Kumar, V. Nonstarch polysaccharide enzymes—General aspects. In Enzymes in Human and Animal Nutrition; Nunes, C., Kumar, V., Eds.; Elsevier: Amsterdam, The Netherlands, 2018; pp. 183–209. [Google Scholar]
- Ahmed, A.; Batool, K.; Bibi, A. Microbial β-glucosidase: Sources, production and applications. J. Appl. Environ. Microbiol. 2017, 5, 31–46. [Google Scholar] [CrossRef] [Green Version]
- Eriksson, K.-E. Biotechnology in the pulp and paper industry. Wood Sci. Technol. 1990, 24, 79–101. [Google Scholar] [CrossRef]
- Godoy, M.G.; Amorim, G.M.; Barreto, M.S.; Freire, D.M.G. Agricultural residues as animal feed: Protein enrichment and detoxification using solid-state fermentation. In Current Developments in Biotechnology and Bioengineering; Elsevier: Amsterdam, The Netherlands, 2018; pp. 235–256. [Google Scholar]
- Kunamneni, A.; Plou, F.J.; Alcalde, M.; Ballesteros, A. Trichoderma enzymes for food industries. In Biotechnology and Biology of Trichoderma; Elsevier: Amsterdam, The Netherlands, 2014; pp. 339–344. [Google Scholar]
- Álvarez, C.; Reyes-Sosa, F.M.; Díez, B. Enzymatic hydrolysis of biomass from wood. Microb. Biotechnol. 2016, 9, 149–156. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Songsiriritthigul, C.; Buranabanyat, B.; Haltrich, D.; Yamabhai, M. Efficient recombinant expression and secretion of a thermostable GH26 mannan endo-1, 4-β-mannosidase from Bacillus licheniformis in Escherichia coli. Microb. Cell Fact. 2010, 9, 20. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yoshida, T.; Nakajima, T.; Ichishima, E. Overproduction of 1, 2-α-mannosidase, a glycochain processing enzyme, by Aspergillus oryzae. Biosci. Biotechnol. Biochem. 1998, 62, 309–315. [Google Scholar] [CrossRef] [Green Version]
- Ademark, P.; Lundqvist, J.; Hägglund, P.; Tenkanen, M.; Torto, N.; Tjerneld, F.; Stålbrand, H. Hydrolytic properties of a β-mannosidase purified from Aspergillus niger. J. Biotechnol. 1999, 75, 281–289. [Google Scholar] [CrossRef]
- Novy, V.; Nielsen, F.; Seiboth, B.; Nidetzky, B. The influence of feedstock characteristics on enzyme production in Trichoderma reesei: A review on productivity, gene regulation and secretion profiles. Biotechnol. Biofuels 2019, 12, 238. [Google Scholar] [CrossRef]
- Waqasi, M.; Mehmood, Z.; Mahmood, K.; Azam, M.; Khan, G.M.; Ibrahim, M.; Rasool, A.; Ahmed, S. Immobilization of b-glucuronidase on the biomaterials/nano particles and its industrial applications. Indo Am. J. Pharm. Sci. 2018, 5, 2287–2291. [Google Scholar]
- Wang, H.; Lu, F.; Sun, Y.; Du, L. Heterologous expression of lignin peroxidase of Phanerochaete chrysosporium in Pichia methanolica. Biotechnol. Lett. 2004, 26, 1569–1573. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Zhang, J.; Zhang, X.; Geng, A. Purification and characterization of a novel manganese peroxidase from white-rot fungus Cerrena unicolor BBP6 and its application in dye decolorization and denim bleaching. Process Biochem. 2018, 66, 222–229. [Google Scholar] [CrossRef]
- Mónica, M.-G.A.; Jaime, M.Q. Phenoloxidases of fungi and bioremediation. Fungal Bioremediation Fundam. Appl. 2019, 3, 62–90. [Google Scholar]
- Siddiqui, K.S.; Ertan, H.; Charlton, T.; Poljak, A.; Khaled, A.K.D.; Yang, X.; Marshall, G.; Cavicchioli, R. Versatile peroxidase degradation of humic substances: Use of isothermal titration calorimetry to assess kinetics, and applications to industrial wastes. J. Biotechnol. 2014, 178, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Bhat, M.K. Cellulases and related enzymes in biotechnology. Biotechnol. Adv. 2000, 18, 355–383. [Google Scholar] [CrossRef]
- Ghose, T.K.; Panda, T.; Bisaria, V.S. Effect of culture phasing and mannanase on production of cellulase and hemicellulase by mixed culture of Trichoderma reesei D 1–6 and Aspergillus wentii pt 2804. Biotechnol. Bioeng. 1985, 27, 1353–1361. [Google Scholar] [CrossRef]
- Dashtban, M.; Schraft, H.; Qin, W. Fungal bioconversion of lignocellulosic residues; opportunities & perspectives. Int. J. Biol. Sci. 2009, 5, 578. [Google Scholar]
- Cai, Y.J.; Chapman, S.J.; Buswell, J.A.; Chang, S. Production and distribution of endoglucanase, cellobiohydrolase, and β-glucosidase components of the cellulolytic system of Volvariella volvacea, the edible straw mushroom. Appl. Environ. Microbiol. 1999, 65, 553–559. [Google Scholar] [CrossRef] [Green Version]
- Aro, N.; Pakula, T.; Penttilä, M. Transcriptional regulation of plant cell wall degradation by filamentous fungi. FEMS Microbiol. Rev. 2005, 29, 719–739. [Google Scholar] [CrossRef] [Green Version]
- Elisashvili, V.; Kachlishvili, E.; Penninckx, M. Effect of growth substrate, method of fermentation, and nitrogen source on lignocellulose-degrading enzymes production by white-rot basidiomycetes. J. Ind. Microbiol. Biotechnol. 2008, 35, 1531–1538. [Google Scholar] [CrossRef]
- Shallom, D.; Shoham, Y. Microbial hemicellulases. Curr. Opin. Microbiol. 2003, 6, 219–228. [Google Scholar] [CrossRef]
- Hofrichter, M. Review: Lignin conversion by manganese peroxidase (MnP). Enzyme Microb. Technol. 2002, 30, 454–466. [Google Scholar] [CrossRef]
- Kuwahara, M.; Glenn, J.K.; Morgan, M.A.; Gold, M.H. Separation and characterization of two extracelluar H2O2-dependent oxidases from ligninolytic cultures of Phanerochaete chrysosporium. FEBS Lett. 1984, 169, 247–250. [Google Scholar] [CrossRef] [Green Version]
- Tuomela, M.; Vikman, M.; Hatakka, A.; Itävaara, M. Biodegradation of lignin in a compost environment: A review. Bioresour. Technol. 2000, 72, 169–183. [Google Scholar] [CrossRef]
- Bugg, T.D.H.; Ahmad, M.; Hardiman, E.M.; Singh, R. The emerging role for bacteria in lignin degradation and bio-product formation. Curr. Opin. Biotechnol. 2011, 22, 394–400. [Google Scholar] [CrossRef]
- Gupta, V.G.; Schmoll, M.; Herrera-Estrella, A.; Upadhyay, R.S.; Druzhinina, I.; Tuohy, M. Biotechnology and Biology of Trichoderma; Newnes: Boston, MA, USA, 2014; ISBN 0444595945. [Google Scholar]
- Singh, A.; Kuhad, R.C.; Ward, O.P. Industrial application of microbial cellulases. In Lignocellulose Biotechnology Future Prospects; I.K. International Publishing House Pvt. Ltd.: New Delhi, India, 2007; pp. 345–358. [Google Scholar]
- Raveendran, S.; Parameswaran, B.; Beevi Ummalyma, S.; Abraham, A.; Kuruvilla Mathew, A.; Madhavan, A.; Rebello, S.; Pandey, A. Applications of microbial enzymes in food industry. Food Technol. Biotechnol. 2018, 56, 16–30. [Google Scholar] [CrossRef]
- Juturu, V.; Wu, J.C. Insight into microbial hemicellulases other than xylanases: A review. J. Chem. Technol. Biotechnol. 2013, 88, 353–363. [Google Scholar] [CrossRef]
- Ravindran, R.; Jaiswal, A.K. Microbial enzyme production using lignocellulosic food industry wastes as feedstock: A review. Bioengineering 2016, 3, 30. [Google Scholar] [CrossRef] [Green Version]
- Chandel, A.K.; Singh, O. V Weedy lignocellulosic feedstock and microbial metabolic engineering: Advancing the generation of ‘Biofuel’. Appl. Microbiol. Biotechnol. 2011, 89, 1289–1303. [Google Scholar] [CrossRef]
- Taha, M.; Foda, M.; Shahsavari, E.; Aburto-Medina, A.; Adetutu, E.; Ball, A. Commercial feasibility of lignocellulose biodegradation: Possibilities and challenges. Curr. Opin. Biotechnol. 2016, 38, 190–197. [Google Scholar] [CrossRef]
- Koutinas, A.A.; Wang, R.; Webb, C. Restructuring upstream bioprocessing: Technological and economical aspects for production of a generic microbial feedstock from wheat. Biotechnol. Bioeng. 2004, 85, 524–538. [Google Scholar] [CrossRef] [PubMed]
- Saini, J.K.; Saini, R.; Tewari, L. Lignocellulosic agriculture wastes as biomass feedstocks for second-generation bioethanol production: Concepts and recent developments. 3 Biotech 2015, 5, 337–353. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mosier, N.; Wyman, C.; Dale, B.; Elander, R.; Lee, Y.Y.; Holtzapple, M.; Ladisch, M. Features of promising technologies for pretreatment of lignocellulosic biomass. Bioresour. Technol. 2005, 96, 673–686. [Google Scholar] [CrossRef] [PubMed]
- Narasimha, G.; Sridevi, A.; Buddolla, V.; Subhosh, C.M.; Rajasekhar, R.B. Nutrient effects on production of cellulolytic enzymes by Aspergillus niger. Afr. J. Biotechnol. 2006, 5, 472–476. [Google Scholar]
- Fuentes, M.E.; Quiñones, R.A.; Gutiérrez, M.H.; Pantoja, S. Effects of temperature and glucose concentration on the growth and respiration of fungal species isolated from a highly productive coastal upwelling ecosystem. Fungal Ecol. 2015, 13, 135–149. [Google Scholar] [CrossRef]
- Van den Brink, J.; de Vries, R.P. Fungal enzyme sets for plant polysaccharide degradation. Appl. Microbiol. Biotechnol. 2011, 91, 1477. [Google Scholar] [CrossRef] [Green Version]
- Hankin, L.; Anagnostakis, S.L. Solid media containing carboxymethylcellulose to detect Cx cellulase activity of micro-organisms. Microbiology 1977, 98, 109–115. [Google Scholar] [CrossRef] [Green Version]
- Binder, J.B.; Raines, R.T. Simple chemical transformation of lignocellulosic biomass into furans for fuels and chemicals. J. Am. Chem. Soc. 2009, 131, 1979–1985. [Google Scholar] [CrossRef]
- Ahamed, A.; Vermette, P. Culture-based strategies to enhance cellulase enzyme production from Trichoderma reesei RUT-C30 in bioreactor culture conditions. Biochem. Eng. J. 2008, 40, 399–407. [Google Scholar] [CrossRef]
- Griffin, H.L.; Sloneker, J.H.; Inglett, G.E. Cellulase production by Trichoderma viride on feedlot waste. Appl. Microbiol. 1974, 27, 1061–1066. [Google Scholar] [CrossRef]
- Hulme, M.A.; Stranks, D.W. Influence of carbohydrate accessibility on cellulase production by Lenzites trabea and Polyporus versicolor. Aust. J. Biol. Sci. 1974, 27, 457–464. [Google Scholar] [CrossRef] [Green Version]
- Pyč, R.; Fiechter, A.; Galas, E. The production of cellulolytic enzymes by fungal cultures. Eur. J. Appl. Microbiol. Biotechnol. 1977, 4, 151–158. [Google Scholar] [CrossRef]
- Dhawan, S.; Gupta, J.K. Enzymic hydrolysis of common cellulosic wastes by cellulase. J. Gen. Appl. Microbiol. 1977, 23, 155–161. [Google Scholar] [CrossRef]
- Ghosh, B.S.; Kundu, A.B. Induction of cellulases and hemicellulases by Tamarind “Tamarindus indica”kernel polysaccharides. J. Ferment. Technol. 1980, 58, 135–141. [Google Scholar]
- Rajoka, M.I.; Malik, K.A. Cellulase and Hemicellulase production by Cellulomonas flavigena NIAB 441. Biotechnol. Lett. 1984, 6, 597–600. [Google Scholar] [CrossRef]
- Dekker, R.F.H. Bioconversion of hemicellulose: Aspects of hemicellulase production by Trichoderma reesei QM 9414 and enzymic saccharification of hemicellulose. Biotechnol. Bioeng. 1983, 25, 1127–1146. [Google Scholar] [CrossRef]
- Shamala, T.R.; Sreekantiah, K.R. Production of cellulases and D-xylanase by some selected fungal isolates. Enzyme Microb. Technol. 1986, 8, 178–182. [Google Scholar] [CrossRef]
- Rajoka, M.I.; Malik, K.A. Comparison of different strains of Cellulomonas for production of cellulolytic and xylanolytic enzymes from biomass produced on saline lands. Biotechnol. Lett. 1986, 8, 753–756. [Google Scholar] [CrossRef]
- Okeke, B.C.; Obi, S.K.C. Saccharification of agro-waste materials by fungal cellulases and hemicellulases. Bioresour. Technol. 1995, 51, 23–27. [Google Scholar] [CrossRef]
- Elshafei, A.M.; Vega, J.L.; Klasson, K.T.; Clausen, E.C.; Gaddy, J.L. Cellulase and hemicellulase formation by fungi using corn stover as the substrate. Biol. Wastes 1990, 32, 209–218. [Google Scholar] [CrossRef]
- Kim, S.W.; Kang, S.W.; Lee, J.S. Cellulase and xylanase production by Aspergillus niger KKS in various bioreactors. Bioresour. Technol. 1997, 59, 63–67. [Google Scholar] [CrossRef]
- Castillo, M.R.; Gutierrez-Correa, M.; Linden, J.C.; Tengerdy, R.P. Mixed culture solid substrate fermentation for cellulolytic enzyme production. Biotechnol. Lett. 1994, 16, 967–972. [Google Scholar] [CrossRef]
- Vlaev, S.D.; Djejeva, G.; Raykovska, V.; Schügerl, K. Cellulase production by Trichoderma sp. grown on corn fibre substrate. Process Biochem. 1997, 32, 561–565. [Google Scholar] [CrossRef]
- Duenas, R.; Tengerdy, R.P.; Gutierrez-Correa, M. Cellulase production by mixed fungi in solid-substrate fermentation of bagasse. World J. Microbiol. Biotechnol. 1995, 11, 333–337. [Google Scholar] [CrossRef]
- Esterbauer, H.; Steiner, W.; Labudova, I.; Hermann, A.; Hayn, M. Production of Trichoderma cellulase in laboratory and pilot scale. Bioresour. Technol. 1991, 36, 51–65. [Google Scholar] [CrossRef]
- Sugden, C.; Bhat, M.K. Cereal straw and pure cellulose as carbon sources for growth and production of plant cell-wall degrading enzymes by Sporotrichum thermophile. World J. Microbiol. Biotechnol. 1994, 10, 444–451. [Google Scholar] [CrossRef]
- Ismail, A.-M.S. Utilization of orange peels for the production of multienzyme complexes by some fungal strains. Process Biochem. 1996, 31, 645–650. [Google Scholar] [CrossRef]
- Kang, S.W.; Park, Y.S.; Lee, J.S.; Hong, S.I.; Kim, S. Production of cellulases and hemicellulases by Aspergillus niger KK2 from lignocellulosic biomass. Bioresour. Technol. 2004, 91, 153–156. [Google Scholar] [CrossRef]
- Olsson, L.; Christensen, T.M.I.E.; Hansen, K.P.; Palmqvist, E.A. Influence of the carbon source on production of cellulases, hemicellulases and pectinases by Trichoderma reesei Rut C-30. Enzyme Microb. Technol. 2003, 33, 612–619. [Google Scholar] [CrossRef]
- Camassola, M.; Dillon, A.J.P. Biological pretreatment of sugar cane bagasse for the production of cellulases and xylanases by Penicillium echinulatum. Ind. Crops Prod. 2009, 29, 642–647. [Google Scholar] [CrossRef]
- Leite, R.S.R.; Bocchini, D.A.; Martins, E.D.S.; Silva, D.; Gomes, E.; Da Silva, R. Production of cellulolytic and hemicellulolytic enzymes from Aureobasidium pulluans on solid state fermentation. Appl. Biochem. Biotechnol. 2007, 137, 281–288. [Google Scholar] [PubMed]
- Camassola, M.; Dillon, A.J.P. Production of cellulases and hemicellulases by Penicillium echinulatum grown on pretreated sugar cane bagasse and wheat bran in solid-state fermentation. J. Appl. Microbiol. 2007, 103, 2196–2204. [Google Scholar] [CrossRef] [PubMed]
- Panagiotou, G.; Kekos, D.; Macris, B.J.; Christakopoulos, P. Production of cellulolytic and xylanolytic enzymes by Fusarium oxysporum grown on corn stover in solid state fermentation. Ind. Crops Prod. 2003, 18, 37–45. [Google Scholar] [CrossRef]
- Dogaris, I.; Vakontios, G.; Kalogeris, E.; Mamma, D.; Kekos, D. Induction of cellulases and hemicellulases from Neurospora crassa under solid-state cultivation for bioconversion of sorghum bagasse into ethanol. Ind. Crops Prod. 2009, 29, 404–411. [Google Scholar] [CrossRef]
- Singh, A.; Singh, N.; Bishnoi, N.R. Production of cellulases by Aspergillus heteromorphus from wheat straw under submerged fermentation. Int. J. Civ. Environ. Eng. 2009, 1, 23–26. [Google Scholar]
- Pothiraj, C.; Balaji, P.; Eyini, M. Enhanced production of cellulases by various fungal cultures in solid state fermentation of cassava waste. Afr. J. Biotechnol. 2006, 5, 1882–1885. [Google Scholar]
- Jatinder, K.; Chadha, B.S.; Saini, H.S. Optimization of culture conditions for production of cellulases and xylanases by Scytalidium thermophilum using response surface methodology. World J. Microbiol. Biotechnol. 2006, 22, 169. [Google Scholar] [CrossRef]
- Xin, F.; Geng, A. Horticultural waste as the substrate for cellulase and hemicellulase production by Trichoderma reesei under solid-state fermentation. Appl. Biochem. Biotechnol. 2010, 162, 295–306. [Google Scholar] [CrossRef]
- Dhillon, G.S.; Kaur, S.; Brar, S.K.; Verma, M. Potential of apple pomace as a solid substrate for fungal cellulase and hemicellulase bioproduction through solid-state fermentation. Ind. Crops Prod. 2012, 38, 6–13. [Google Scholar] [CrossRef]
- Dhillon, G.S.; Oberoi, H.S.; Kaur, S.; Bansal, S.; Brar, S.K. Value-addition of agricultural wastes for augmented cellulase and xylanase production through solid-state tray fermentation employing mixed-culture of fungi. Ind. Crops Prod. 2011, 34, 1160–1167. [Google Scholar] [CrossRef]
- Saratale, G.D.; Kshirsagar, S.D.; Sampange, V.T.; Saratale, R.G.; Oh, S.-E.; Govindwar, S.P.; Oh, M.-K. Cellulolytic enzymes production by utilizing agricultural wastes under solid state fermentation and its application for biohydrogen production. Appl. Biochem. Biotechnol. 2014, 174, 2801–2817. [Google Scholar] [CrossRef] [PubMed]
- Dhillon, G.S.; Brar, S.K.; Kaur, S.; Metahni, S.; M’hamdi, N. Lactoserum as a moistening medium and crude inducer for fungal cellulase and hemicellulase induction through solid-state fermentation of apple pomace. Biomass Bioenergy 2012, 41, 165–174. [Google Scholar] [CrossRef]
- Dias, L.M.; Dos Santos, B.V.; Albuquerque, C.J.B.; Baeta, B.E.L.; Pasquini, D.; Baffi, M.A. Biomass sorghum as a novel substrate in solid-state fermentation for the production of hemicellulases and cellulases by Aspergillus niger and A. fumigatus. J. Appl. Microbiol. 2018, 124, 708–718. [Google Scholar] [CrossRef] [PubMed]
- Iram, A.; Cekmecelioglu, D.; Demirci, A. Screening of bacterial and fungal strains for cellulase and xylanase production using distillers’ dried grains with solubles (DDGS) as the main feedstock. Biomass Convers. Biorefinery 2020. [Google Scholar] [CrossRef]
- Motta, F.L.; Andrade, C.C.P.; Santana, M.H.A. A review of xylanase production by the fermentation of xylan: Classification, characterization and applications. In Sustainable Degradation of Lignocellulosic Biomass: Techniques, Applications and Commercialization; InTech: Rijeka, Croatia, 2013; Volume 1. [Google Scholar]
- Goyal, M.; Kalra, K.L.; Sareen, V.K.; Soni, G. Xylanase production with xylan rich lignocellulosic wastes by a local soil isolate of Trichoderma viride. Braz. J. Microbiol. 2008, 39, 535–541. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Klein-Marcuschamer, D.; Oleskowicz-Popiel, P.; Simmons, B.A.; Blanch, H.W. The challenge of enzyme cost in the production of lignocellulosic biofuels. Biotechnol. Bioeng. 2012, 109, 1083–1087. [Google Scholar] [CrossRef] [PubMed]
- Baldrian, P.; Šnajdr, J. Production of ligninolytic enzymes by litter-decomposing fungi and their ability to decolorize synthetic dyes. Enzyme Microb. Technol. 2006, 39, 1023–1029. [Google Scholar] [CrossRef]
- Pant, D.; Adholeya, A. Enhanced production of ligninolytic enzymes and decolorization of molasses distillery wastewater by fungi under solid state fermentation. Biodegradation 2007, 18, 647–659. [Google Scholar] [CrossRef]
- Lang, E.; Eller, G.; Zadrazil, F. Lignocellulose decomposition and production of ligninolytic enzymes during interaction of white rot fungi with soil microorganisms. Microb. Ecol. 1997, 34, 1–10. [Google Scholar] [CrossRef]
- Verma, P.; Madamwar, D. Production of ligninolytic enzymes for dye decolorization by cocultivation of white-rot fungi Pleurotus ostreatus and Phanerochaete chrysosporium under solid-state fermentation. Appl. Biochem. Biotechnol. 2002, 102, 109–118. [Google Scholar] [CrossRef]
- Gassara, F.; Brar, S.K.; Tyagi, R.D.; John, R.P.; Verma, M.; Valero, J.R. Parameter optimization for production of ligninolytic enzymes using agro-industrial wastes by response surface method. Biotechnol. Bioprocess Eng. 2011, 16, 343–351. [Google Scholar] [CrossRef]
- Pickard, M.A.; Vandertol, H.; Roman, R.; Vazquez-Duhalt, R. High production of ligninolytic enzymes from white rot fungi in cereal bran liquid medium. Can. J. Microbiol. 1999, 45, 627–631. [Google Scholar] [CrossRef]
- Adsul, M.G.; Ghule, J.E.; Singh, R.; Shaikh, H.; Bastawde, K.B.; Gokhale, D.V.; Varma, A.J. Polysaccharides from bagasse: Applications in cellulase and xylanase production. Carbohydr. Polym. 2004, 57, 67–72. [Google Scholar] [CrossRef]
- Iram, A.; Cekmecelioglu, D.; Demirci, A. Optimization of dilute sulfuric acid, aqueous ammonia, and steam explosion as the pretreatments steps for distillers’ dried grains with solubles as a potential fermentation feedstock. Bioresour. Technol. 2019, 282, 475–481. [Google Scholar] [CrossRef] [PubMed]
- De Almeida, M.N.; Guimarães, V.M.; Bischoff, K.M.; Falkoski, D.L.; Pereira, O.L.; Gonçalves, D.S.P.O.; de Rezende, S.T. Cellulases and hemicellulases from endophytic acremonium species and its application on sugarcane bagasse hydrolysis. Appl. Biochem. Biotechnol. 2011, 165, 594–610. [Google Scholar] [CrossRef] [PubMed]
- Ben Taher, I.; Bennour, H.; Fickers, P.; Hassouna, M. Valorization of potato peels residues on cellulase production using a mixed culture of Aspergillus niger ATCC 16404 and trichoderma reesei DSMZ 970. Waste Biomass Valoriz. 2017, 8, 183–192. [Google Scholar] [CrossRef]
- McMillan, J.D. Pretreatment of lignocellulosic biomass. In Enzymatic Conversion of Biomass for Fuels Production; ACS Publications: Washington, DC, USA, 1994; ISBN 1947-5918. [Google Scholar]
- Zhao, X.; Cheng, K.; Liu, D. Organosolv pretreatment of lignocellulosic biomass for enzymatic hydrolysis. Appl. Microbiol. Biotechnol. 2009, 82, 815. [Google Scholar] [CrossRef]
- Kumar, P.; Barrett, D.M.; Delwiche, M.J.; Stroeve, P. Methods for pretreatment of lignocellulosic biomass for efficient hydrolysis and biofuel production. Ind. Eng. Chem. Res. 2009, 48, 3713–3729. [Google Scholar] [CrossRef]
- Zheng, Y.; Zhao, J.; Xu, F.; Li, Y. Pretreatment of lignocellulosic biomass for enhanced biogas production. Prog. Energy Combust. Sci. 2014, 42, 35–53. [Google Scholar] [CrossRef]
- Michelin, M.; Mota, A.M.O.; Silva, D.P.; Ruzene, D.S.; Vicente, A.A.; Teixeira, J.A. Production of biomass-degrading enzymes by Trichoderma reesei using liquid hot water-pretreated corncob in different conditions of oxygen transfer. BioEnergy Res. 2019, 12, 583–592. [Google Scholar] [CrossRef] [Green Version]
- Singh, A.; Tuteja, S.; Singh, N.; Bishnoi, N.R. Enhanced saccharification of rice straw and hull by microwave–alkali pretreatment and lignocellulolytic enzyme production. Bioresour. Technol. 2011, 102, 1773–1782. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Zhang, H.; Lu, Y.; Xue, H. Solid state fermentation process coupled biological pretreatment with cellulase production by Piptoporus betulinus for enhanced cellulose hydrolysis. Cellulose 2019, 26, 3815–3824. [Google Scholar] [CrossRef]
- Leite, P.; Salgado, J.M.; Venâncio, A.; Domínguez, J.M.; Belo, I. Ultrasounds pretreatment of olive pomace to improve xylanase and cellulase production by solid-state fermentation. Bioresour. Technol. 2016, 214, 737–746. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, K.; Pei, Z.; Wang, D. Organic solvent pretreatment of lignocellulosic biomass for biofuels and biochemicals: A review. Bioresour. Technol. 2016, 199, 21–33. [Google Scholar] [CrossRef] [PubMed]
- Gupta, R.; Aswal, V.K.; Saini, J.K. Sequential dilute acid and alkali deconstruction of sugarcane bagasse for improved hydrolysis: Insight from small angle neutron scattering (SANS). Renew. Energy 2020, 147, 2091–2101. [Google Scholar]
- Kim, S.; Kim, C.H. Bioethanol production using the sequential acid/alkali-pretreated empty palm fruit bunch fiber. Renew. Energy 2013, 54, 150–155. [Google Scholar] [CrossRef]
- Wang, Y.; Guo, W.; Cheng, C.-L.; Ho, S.-H.; Chang, J.-S.; Ren, N. Enhancing bio-butanol production from biomass of Chlorella vulgaris JSC-6 with sequential alkali pretreatment and acid hydrolysis. Bioresour. Technol. 2016, 200, 557–564. [Google Scholar] [CrossRef]
- Obeng, E.M.; Adam, S.N.N.; Budiman, C.; Ongkudon, C.M.; Maas, R.; Jose, J. Lignocellulases: A review of emerging and developing enzymes, systems, and practices. Bioresour. Bioprocess. 2017, 4, 16. [Google Scholar] [CrossRef]
- Gomes, E.; Aguiar, A.P.; Carvalho, C.C.; Bonfá, M.R.B.; da Silva, R.; Boscolo, M. Ligninases production by Basidiomycetes strains on lignocellulosic agricultural residues and their application in the decolorization of synthetic dyes. Braz. J. Microbiol. 2009, 40, 31–39. [Google Scholar] [CrossRef]
- Sukumaran, R.K.; Singhania, R.R.; Pandey, A. Microbial cellulases-production, applications and challenges. J. Sci. Ind. Res. 2005, 64, 890–896. [Google Scholar]
- Baig, M.M.V. Cellulolytic enzymes of Trichoderma lignorum produced on banana agro-waste: Optimisation of culture medium and conditions. J. Sci. Ind. Res. 2005, 64, 57–60. [Google Scholar]
- Kovács, K.; Szakacs, G.; Zacchi, G. Comparative enzymatic hydrolysis of pretreated spruce by supernatants, whole fermentation broths and washed mycelia of Trichoderma reesei and Trichoderma atroviride. Bioresour. Technol. 2009, 100, 1350–1357. [Google Scholar] [CrossRef] [PubMed]
- Bailey, M.J.; Buchert, J.; Viikari, L. Effect of pH on production of xylanase by Trichoderma reesei on xylan-and cellulose-based media. Appl. Microbiol. Biotechnol. 1993, 40, 224–229. [Google Scholar] [CrossRef]
- Peterson, R.; Nevalainen, H. Trichoderma reesei RUT-C30–thirty years of strain improvement. Microbiology 2012, 158, 58–68. [Google Scholar] [CrossRef] [Green Version]
- Simmons, E.G. Classification of some cellulase-producing Trichoderma species. In Proceedings of the Second International Mycological Congress, Tampa, FL, USA, 27 August–3 September 1977; Taylor & Francis: Tampa, FL, USA, 1977; Volume 2, p. 618. [Google Scholar]
- Park, Y.; Kang, S.; Lee, J.; Hong, S.L.; Kim, S. Xylanase production in solid state fermentation by Aspergillus niger mutant using statistical experimental designs. Appl. Microbiol. Biotechnol. 2002, 58, 761–766. [Google Scholar]
- Prajapati, B.P.; Suryawanshi, R.K.; Agrawal, S.; Ghosh, M.; Kango, N. Characterization of cellulase from Aspergillus tubingensis NKBP-55 for generation of fermentable sugars from agricultural residues. Bioresour. Technol. 2018, 250, 733–740. [Google Scholar] [CrossRef]
- Ang, S.K.; Shaza, E.M.; Adibah, Y.; Suraini, A.A.; Madihah, M.S. Production of cellulases and xylanase by Aspergillus fumigatus SK1 using untreated oil palm trunk through solid state fermentation. Process Biochem. 2013, 48, 1293–1302. [Google Scholar] [CrossRef] [Green Version]
- Watanabe, J.; Tanaka, H.; Mogi, Y.; Yamazaki, T.; Suzuki, K.; Watanabe, T.; Yamada, O.; Akita, O. Loss of Aspergillus oryzae amyR function indirectly affects hemicellulolytic and cellulolytic enzyme production. J. Biosci. Bioeng. 2011, 111, 408–413. [Google Scholar] [CrossRef]
- Stricker, A.R.; Mach, R.L.; De Graaff, L.H. Regulation of transcription of cellulases-and hemicellulases-encoding genes in Aspergillus niger and Hypocrea jecorina (Trichoderma reesei). Appl. Microbiol. Biotechnol. 2008, 78, 211. [Google Scholar] [CrossRef]
- Adsul, M.G.; Terwadkar, A.P.; Varma, A.J.; Gokhale, D. Cellulases from Penicillium janthinellum mutants: Solid-state production and their stability in ionic liquids. Bioresources 2009, 4, 1670–1681. [Google Scholar]
- Bailey, M.J.; Nevalainen, K.M.H. Induction, isolation and testing of stable Trichoderma reesei mutants with improved production of solubilizing cellulase. Enzyme Microb. Technol. 1981, 3, 153–157. [Google Scholar] [CrossRef]
- Yoon, L.W.; Ang, T.N.; Ngoh, G.C.; Chua, A.S.M. Fungal solid-state fermentation and various methods of enhancement in cellulase production. Biomass Bioenergy 2014, 67, 319–338. [Google Scholar] [CrossRef]
- Hölker, U.; Lenz, J. Solid-state fermentation—Are there any biotechnological advantages? Curr. Opin. Microbiol. 2005, 8, 301–306. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Lao, G.; Wilson, D.B. Characterization of a Thermomonospora fusca exocellulase. Biochemistry 1995, 34, 3386–3395. [Google Scholar] [CrossRef]
- Hansen, G.H.; Lübeck, M.; Frisvad, J.C.; Lübeck, P.S.; Andersen, B. Production of cellulolytic enzymes from ascomycetes: Comparison of solid state and submerged fermentation. Process Biochem. 2015, 50, 1327–1341. [Google Scholar] [CrossRef]
- Haltrich, D.; Nidetzky, B.; Kulbe, K.D.; Steiner, W.; Župančič, S. Production of fungal xylanases. Bioresour. Technol. 1996, 58, 137–161. [Google Scholar] [CrossRef]
- Machida, M.; Yamada, O.; Gomi, K. Genomics of Aspergillus oryzae: Learning from the history of Koji Mold and exploration of its future. DNA Res. 2008, 15, 173–183. [Google Scholar] [CrossRef] [Green Version]
- Te Biesebeke, R.; Ruijter, G.; Rahardjo, Y.S.P.; Hoogschagen, M.J.; Heerikhuisen, M.; Levin, A.; van Driel, K.G.A.; Schutyser, M.A.I.; Dijksterhuis, J.; Zhu, Y.; et al. Aspergillus oryzae in solid-state and submerged fermentations: Progress report on a multi-disciplinary project. FEMS Yeast Res. 2002, 2, 245–248. [Google Scholar] [CrossRef] [Green Version]
- Chen, H.; He, Q. Value-added bioconversion of biomass by solid-state fermentation. J. Chem. Technol. Biotechnol. 2012, 87, 1619–1625. [Google Scholar] [CrossRef]
- Deka, D.; Das, S.P.; Sahoo, N.; Das, D.; Jawed, M.; Goyal, D.; Goyal, A. Enhanced cellulase production from Bacillus subtilis by optimizing physical parameters for bioethanol production. Isrn Biotechnol. 2013, 2013, 965310. [Google Scholar] [CrossRef] [Green Version]
- Zhang, B.; Weng, Y.; Xu, H.; Mao, Z. Enzyme immobilization for biodiesel production. Appl. Microbiol. Biotechnol. 2012, 93, 61–70. [Google Scholar] [CrossRef] [PubMed]
- Ahamed, A.; Vermette, P. Enhanced enzyme production from mixed cultures of Trichoderma reesei RUT-C30 and Aspergillus niger LMA grown as fed batch in a stirred tank bioreactor. Biochem. Eng. J. 2008, 42, 41–46. [Google Scholar] [CrossRef]
- Haltrich, D.; Laussamayer, B.; Steiner, W.; Nidetzky, B.; Kulbe, K.D. Cellulolytic and hemicellulolytic enzymes of Sclerotium rolfsii: Optimization of the culture medium and enzymatic hydrolysis of lignocellulosic material. Bioresour. Technol. 1994, 50, 43–50. [Google Scholar] [CrossRef]
- Xue, Y.; Han, J.; Li, Y.; Liu, J.; Gan, L.; Long, M. Promoting cellulase and hemicellulase production from Trichoderma orientalis EU7-22 by overexpression of transcription factors Xyr1 and Ace3. Bioresour. Technol. 2020, 296, 122355. [Google Scholar] [CrossRef] [PubMed]
- Fujii, T.; Inoue, H.; Ishikawa, K. Enhancing cellulase and hemicellulase production by genetic modification of the carbon catabolite repressor gene, creA, in Acremonium cellulolyticus. AMB Express 2013, 3, 73. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nakari-Setälä, T.; Paloheimo, M.; Kallio, J.; Vehmaanperä, J.; Penttilä, M.; Saloheimo, M. Genetic modification of carbon catabolite repression in Trichoderma reesei for improved protein production. Appl. Environ. Microbiol. 2009, 75, 4853–4860. [Google Scholar] [CrossRef] [Green Version]
- Häkkinen, M.; Valkonen, M.J.; Westerholm-Parvinen, A.; Aro, N.; Arvas, M.; Vitikainen, M.; Penttilä, M.; Saloheimo, M.; Pakula, T.M. Screening of candidate regulators for cellulase and hemicellulase production in Trichoderma reesei and identification of a factor essential for cellulase production. Biotechnol. Biofuels 2014, 7, 14. [Google Scholar] [CrossRef] [Green Version]
- Juturu, V.; Wu, J.C. Microbial cellulases: Engineering, production and applications. Renew. Sustain. Energy Rev. 2014, 33, 188–203. [Google Scholar] [CrossRef]
- Sohail, M.; Siddiqi, R.; Ahmad, A.; Khan, S.A. Cellulase production from Aspergillus niger MS82: Effect of temperature and pH. N. Biotechnol. 2009, 25, 437–441. [Google Scholar] [CrossRef]
- Shankar, T.; Isaiarasu, L. Cellulase production by Bacillus pumilus EWBCM1 under varying cultural conditions. Middle-East J. Sci. Res. 2011, 8, 40–45. [Google Scholar]
- Bergquist, P.L.; Gibbs, M.D.; Morris, D.D.; Te’o, V.S.J.; Saul, D.J.; Morgan, H.W. Molecular diversity of thermophilic cellulolytic and hemicellulolytic bacteria. FEMS Microbiol. Ecol. 1999, 28, 99–110. [Google Scholar] [CrossRef]
- Potprommanee, L.; Wang, X.-Q.; Han, Y.-J.; Nyobe, D.; Peng, Y.-P.; Huang, Q.; Liu, J.; Liao, Y.-L.; Chang, K.-L. Characterization of a thermophilic cellulase from Geobacillus sp. HTA426, an efficient cellulase-producer on alkali pretreated of lignocellulosic biomass. PLoS ONE 2017, 12, e0175004. [Google Scholar] [CrossRef] [PubMed]
- Asha, B.M.; Revathi, M.; Yadav, A.; Sakthivel, N. Purification and characterization of a thermophilic cellulase from a novel cellulolytic strain, Paenibacillus barcinonensis. J. Microbiol. Biotechnol. 2012, 22, 1501–1509. [Google Scholar] [CrossRef]
- Acharya, P.B.; Acharya, D.K.; Modi, H.A. Optimization for cellulase production by Aspergillus niger using saw dust as substrate. Afr. J. Biotechnol. 2008, 7, 4147–4152. [Google Scholar]
- Shaikh, N.M.; Patel, A.A.; Mehta, S.A.; Patel, N.D. Isolation and screening of cellulolytic bacteria inhabiting different environment and optimization of cellulase production. Univers. J. Environ. Res. Technol. 2013, 3, 39–49. [Google Scholar]
- Gomes, I.; Gomes, J.; Steiner, W.; Esterbauer, H. Production of cellulase and xylanase by a wild strain of Trichoderma viride. Appl. Microbiol. Biotechnol. 1992, 36, 701–707. [Google Scholar] [CrossRef]
- Soni, R.; Nazir, A.; Chadha, B.S. Optimization of cellulase production by a versatile Aspergillus fumigatus fresenius strain (AMA) capable of efficient deinking and enzymatic hydrolysis of Solka floc and bagasse. Ind. Crops Prod. 2010, 31, 277–283. [Google Scholar] [CrossRef]
- Deswal, D.; Khasa, Y.P.; Kuhad, R.C. Optimization of cellulase production by a brown rot fungus Fomitopsis sp. RCK2010 under solid state fermentation. Bioresour. Technol. 2011, 102, 6065–6072. [Google Scholar] [CrossRef]
- Liang, Y.-L.; Zhang, Z.; Wu, M.; Wu, Y.; Feng, J.-X. Isolation, screening, and identification of cellulolytic bacteria from natural reserves in the subtropical region of China and optimization of cellulase production by Paenibacillus terrae ME27-1. Biomed Res. Int. 2014, 2014, 512497. [Google Scholar] [CrossRef] [Green Version]
- Haltrich, D.; Preiss, M.; Steiner, W. Optimization of a culture medium for increased xylanase production by a wild strain of Schizophyllum commune. Enzyme Microb. Technol. 1993, 15, 854–860. [Google Scholar] [CrossRef]
- Chaabouni, S.E.; Belguith, H.; Hassairi, I.; M’rad, K.; Ellouz, R. Optimization of cellulase production by Penicillium occitanis. Appl. Microbiol. Biotechnol. 1995, 43, 267–269. [Google Scholar] [CrossRef]
- Lee, J. Biological conversion of lignocellulosic biomass to ethanol. J. Biotechnol. 1997, 56, 1–24. [Google Scholar] [CrossRef]
- Kuhad, R.C.; Deswal, D.; Sharma, S.; Bhattacharya, A.; Jain, K.K.; Kaur, A.; Pletschke, B.I.; Singh, A.; Karp, M. Revisiting cellulase production and redefining current strategies based on major challenges. Renew. Sustain. Energy Rev. 2016, 55, 249–272. [Google Scholar] [CrossRef]
- Weber, J.; Agblevor, F.A. Microbubble fermentation of Trichoderma reesei for cellulase production. Process Biochem. 2005, 40, 669–676. [Google Scholar] [CrossRef]

| Enzyme | Type | Enzyme Commission (EC) Number | Activity-Characteristics | Application Areas | References |
|---|---|---|---|---|---|
| Cellulases | Endo-β-glucanase | 3.2.1.4 | Hydrolysis of 1–3 or 1–4 bonds in the beta-D-glucans within the chain | Cereal grains; polishing; feed supplements | [12,13] |
| β-Glucosidase | 3.2.1.21 | Hydrolysis from non-reducing end | Flavor enhancement; biofuel industry | [12,14] | |
| Exoglucanases | 3.2.1.91 | reducing or non-reducing end creating cellobiose | Food, pulp and paper industry | [12,15] | |
| Hemicellulases | Endo-β-1,4-xylanase | 3.2.1.8 | β-1,4 bonds within Xylan chains | Food industry | [16,17] |
| 1,4-β-Xylosidase | 3.2.1.37 | Hydrolysis from non-reducing end in β-D-Xylan | Food industry | [17,18] | |
| Endo-1,4-β-mannosidase | 3.2.1.78 | β-1,4 bonds within mannan chains | Delignification in pulp industry | [17,19] | |
| 1,2-α-Mannosidase | 3.2.1.113 | Removal of terminal alpha-D-mannose residues | Delignification in pulp industry | [17,20] | |
| β-Mannosidase | 3.2.1.25 | Hydrolysis from nonreducing end to form d-mannose residues | Delignification in pulp industry | [17,21] | |
| α-Galactosidase | 3.2.1.22 | Hydrolysis of α-galactoglucomannan | Guar gum digestion | [17,22] | |
| β-Galactosidase | 3.2.1.23 | Hydrolysis of β-galactoglucomannan | Medicine; guar gum digestion | [17,22] | |
| α-L-Arabinofuranosidase | 3.2.1.55 | Hydrolysis of arabinoxylan and arabinoglucoronoxylan | Feed industry and baking | [17,22] | |
| α-Glucuronidase | 3.2.1.139 | Hydrolysis of arabinoglucoronoxylan | Food industry | [22,23] | |
| Acetyl esterase | 3.1.1.6 | Hydrolysis of the ester bond between arabinose and ferulic acid (Lignin) | Cider clarification | [17,22] | |
| Acetyl xylan esterase | 3.1.1.72 | Cleaving of Acetyl groups in hemicellulose | Cider clarification | [17,22] | |
| Lignin modifying enzymes (LMEs) | Lignin peroxidase | 1.11.1.14 | Oxidoreductase | Waste treatment | [24] |
| Manganese peroxidase | 1.11.1.13 | Oxidoreductase | Wastewater treatment in the production of synthetic dyes | [25] | |
| Phenoloxidases | 1.10.3.2 | Multicopper oxidases | Bioremediation | [26] | |
| Hybrid peroxidase | 1.11.1.16 | Oxidoreductase | Industrial waste treatment | [27] |
| Year | Feedstock Used | Composition of Enzyme | Maximum Enzyme Produced | Units | References |
|---|---|---|---|---|---|
| 1970–1979 | Feedlot waste | Cellulase and hemicellulase complexes | 0.4 enzyme cocktail | FPU/g/mL | [55] |
| Ball-milled Populus tremuloides | Cellulase complexes | 1.5 enzyme cocktail | U/mL | [56] | |
| Wheat straw, sprouts, malt and corn cobs | Cellulase and hemicellulase complexes | 29.69 enzyme cocktail | mg sugar/mL media | [57] | |
| Sugar cane bagasse | Cellulases | 48.1 enzyme cocktail | % degradation of feedstock | [58] | |
| 1980–1989 | Tamarind kernel polysaccharide (TKP) | Cellulases, hemicellulases, β-glucosidase and β -xylosidase | N/A | N/A | [59] |
| Kallar grass | CMCase and Xylanase | 3.8 CMCase 16.0 Xylanase | IU/mL | [60] | |
| Hemicellulose substrates and bagasse | Xylanase and xylosidase | 1.5 xylanase 0.08 β-xylosidase | U/mL | [61] | |
| Wheat bran, rice straw | β-d-glucosidase, d-xylanase | 60 Β-d-glucosidase, 740 d-xylanase | U/g | [62] | |
| Kallar grass | CMCase, avicelase, xylanase, β -glucosidase and β -xylosidase | 3.2 CMCase | IU/mL | [63] | |
| 1990–1999 | Corn cobs, rice hulls and melonseed shells | Cellulase and hemicellulase cocktails | 20 enzyme cocktail | % yield | [64] |
| Corn Stover | Cellulase and hemicellulase cocktails | 0.7 FPA | nmol/mL/s | [65] | |
| Rice straw | Cellulases and xylanases | 9.7 FPA cellulase 9100 Xylanase | IU/l/h U/g | [66] | |
| Sweet sorghum silage | Cellulases and xylanases | 4 Cellulase 180 xylanase | IU/g | [67] | |
| Corn fiber | Cellulase and Xylanase | 3.4 cellulase 3.7 Xylanase | U/cm3 | [68] | |
| Bagasse | Cellulase and β-glucosidase | 18.7 cellulase 38.6 β-glucosidase | IU/g | [69] | |
| Wood, straw | Cellulase | 7–18 | FPU/mL | [70] | |
| Barley and Wheat Straw | Xylanase, glucosidase, xylosidase, esterase and arabinofuranosidase | 0.16 cellobiase 1.4 xylanase | µmol/mL/min | [71] | |
| Orange peels | Cellulase, xylanase, pectinase | 3.39 cellulase 3.33 xylanase | U/mL | [72] | |
| 2000–2009 | Corn stover | Cellulases and hemicellulases | 1.2 Filter paper activity | FPU/mL | [11] |
| Rice straw and wheat bran | Cellulases and hemicellulases | 129 CMCase 100 β-glucosidase 5070 Xylanase | IU/g | [73] | |
| Sugar beet pulp | Endoglucanase, arabinosidase | 0.19 Endoglucanase 0.009 arabinosidase | U/mL | [74] | |
| Sugar cane bagasse | Cellulases and xylanases | 0.13 FPA 0.33 Xylanases | U/mL | [75] | |
| Wheat bran | Cellulolytic and hemicellulolytic enzymes | 1.05 endoglucanase 1.3 β-glucosidase 5.0 xylanase | U/mL | [76] | |
| Wheat bran and sugar cane bagasse | Cellulases and xylanases | 32·89 FPA 10 Xylanase | U/g | [77] | |
| Corn stover | Cellulolytic and xylanolytic enzymes | 304 endoglucanase 1840 Xylanases | U/g | [78] | |
| Sorghum Bagasse | Cellulases and xylanases | 492.8 endoglucanase 297.8 Xylanases | U/g | [79] | |
| Wheat straw | Cellulases | 3.2 FPA83 CMCase | IU/mL | [80] | |
| Cassava waste | Cellulases | 0.46 CMCase 0.28 FPase | IU/mL | [81] | |
| Wheat bran and rice straw | Cellulases | 62.5 endoglucanase 3.0 FPase 196 Xylanase | units/g substrate | [82] | |
| 2010–2020 | Horticulture waste | Cellulase and hemicellulase | 15 FPase 52.1 Xylanase | U/g | [83] |
| Apple pomace | Cellulase and hemicellulase | 133.68 FPase 1412.58 Xylanase | IU/g | [84] | |
| Agricultural wastes | Cellulase and xylanase | 13.57 Cellulase 3106.34 Xylanase | IU/g | [85] | |
| Agricultural Wastes | Cellulase and xylanase | 30.22 FPase 427.0 Xylanase | U/g | [86] | |
| Apple pomace | Cellulase and hemicellulase | 383.7 FPase 4868 Xylanase | IU/g | [87] | |
| Sorghum and wheat bran | Cellulase and hemicellulase | 30.64 Cellulase 300.07 Xylanase | U/g | [88] | |
| DDGS | Cellulase and hemicellulase | 0.592 Cellulase 34.8 Xylanase | IU/mL | [89] |
| Type | Pretreatment Method | Microorganisms | Example of Pretreatment Method | Feedstock | Enzyme Production | References |
|---|---|---|---|---|---|---|
| Physical | Liquid hot water | Trichoderma reesei | 200 °C for 30 min | Corn Cob | 3.5 FPU/mL | [107] |
| Steam | Trichoderma reesei | 121 °C for 2 h | Horticultural Waste | 72 U/g | [83] | |
| Milling | Trichoderma reesei | Milled to 200 to 500 µm particle sizes | Horticultural Waste | 6.6 U/g | [83] | |
| Microwave | Aspergillus heteromorphus | 22.5 min irradiation time at 30 g/L substrate concentration | Rice Straw and Hulls | 14.1 U/g | [108] | |
| Chemical | Dilute acid hydrolysis | 11 different bacterial and fungal strains | 5% sulfuric acid at 120 °C for 30 mins with 20% solid load | Distillers’ Dried Grains with Solubles | 0.592 IU/mL | [89] |
| Alkaline treatment | Endophytic Acremonium Species | 1% NaOH at 10% solid load | Sugarcane Bagasse | 0.14 U/mL | [101] | |
| Biological | Fungal treatment | Piptoporus betulinus | 7 mm diameter mycelial discs | Rice Straw | 7.43 U/g | [109] |
| - | Optimized Conditions | - | |||||
|---|---|---|---|---|---|---|---|
| Enzyme | pH | Temperature (°C) | Agitation (RPM) | Time (h) | Microorganism (s) | Increase in Production | References |
| Cellulase | 4–4.5 | 28 | 120 | 96 | Aspergillus niger | 0.02 to 0.1813 IU/mL | [152] |
| CMCase | 7.2 | 39.11 | 121 | NO | Bacillus subtilis | 0.43 to 0.56 U/mL | [138] |
| Cellulase | 7.5 | 40 | NO | 96 | Different Pseudomonas and Bacillus species | 0.98–3.4 U/mL | [153] |
| Cellulase | 4 | 35 | NO | 54 | Aspergillus niger | 0–0.37 IU/mL | [147] |
| FPase | NO | 32.8 | NO | 144 | Trichoderma viride | 0.12 to 0.55 U/mL | [154] |
| Xylanase | NO | 34.7 | NO | 158 | Trichoderma viride | 30 to 145 U/mL | [154] |
| FPase | NO | 37 | NO | NO | Aspergillus fumigatus | 0–9.73 U/g | [155] |
| CMCase | 5.5 | 30 | NO | 264 | Fomitopsis sp. | 0–71.7 IU/g | [156] |
| CMCase | 5.5 | 50 | NO | NO | Paenibacillus terrae | 0.1–2.08 U/mL | [157] |
| Xylanase | NO | NO | NO | 264 | Schizophyllum commune | 0.08–5,740 IU/mL | [158] |
| Enzyme | Nitrogen Source Optimization | Microorganism (s) | Increase in Enzyme Activity | References |
|---|---|---|---|---|
| Cellulase | 0.125% peptone | Aspergillus niger | 0.05–0.1813 IU/mL | [152] |
| Cellulase | 4 g/L NaNO3 | Penicillium occitanis | 0.5–13 U/mL | [159] |
| FPase | 3% sulfite pulp | Trichoderma viride | 0.12–0.39 U/mL | [154] |
| Xylanase | 3% sulfite pulp | Trichoderma viride | 30–70.25 U/mL | [154] |
| FPase | 0.25% beef extract | Aspergillus fumigatus | 0–9.73 U/g | [155] |
| FPase | 80.2 g/L Peptone | Sclerotium rolfsii | 0–5.72 FPU/mL | [141] |
| Xylanase | 55.4 g/L yeast extract | Schizophyllum commune | 0.08–5.74 IU/mL | [158] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Iram, A.; Cekmecelioglu, D.; Demirci, A. Ideal Feedstock and Fermentation Process Improvements for the Production of Lignocellulolytic Enzymes. Processes 2021, 9, 38. https://doi.org/10.3390/pr9010038
Iram A, Cekmecelioglu D, Demirci A. Ideal Feedstock and Fermentation Process Improvements for the Production of Lignocellulolytic Enzymes. Processes. 2021; 9(1):38. https://doi.org/10.3390/pr9010038
Chicago/Turabian StyleIram, Attia, Deniz Cekmecelioglu, and Ali Demirci. 2021. "Ideal Feedstock and Fermentation Process Improvements for the Production of Lignocellulolytic Enzymes" Processes 9, no. 1: 38. https://doi.org/10.3390/pr9010038
APA StyleIram, A., Cekmecelioglu, D., & Demirci, A. (2021). Ideal Feedstock and Fermentation Process Improvements for the Production of Lignocellulolytic Enzymes. Processes, 9(1), 38. https://doi.org/10.3390/pr9010038





