Reactive Hyperemia and Cardiovascular Autonomic Neuropathy in Type 2 Diabetic Patients: A Systematic Review of Randomized and Nonrandomized Clinical Trials
Abstract
:1. Introduction
1.1. Diabetic Autonomic Neuropathy
1.2. Diagnostic Tests for CAN
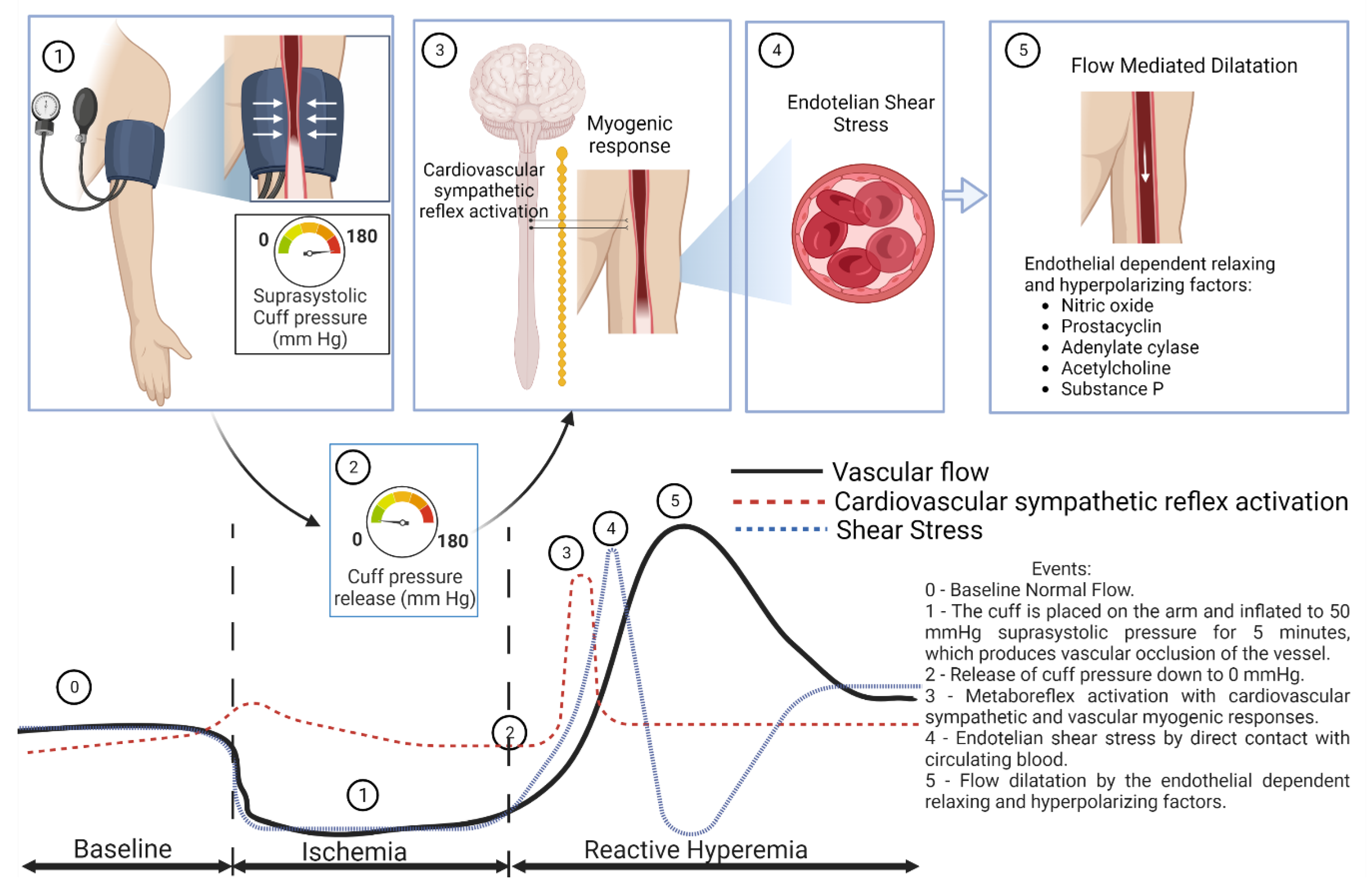
1.3. Reactive Hyperemia
1.4. Reactive Hyperemia and Diabetes
| Parameter | Description | Physiological Significance |
|---|---|---|
| Linear Methods | ||
| SDNN | Standard deviation of all RR intervals in the measurement period. | Independent indicator of frequencies, allowing the calculation of total variability [56]. |
| RMSSD | Square root of the mean value of the sum of the squared differences of all successive RR intervals. | Reflects short-term variations in RR intervals and is used to determine the influence of the parasympathetic nervous system (PNS) on the cardiovascular system. RMSSD is directly associated with short-term variability [56]. |
| CV | Coefficient of variation; determined by dividing the standard deviation by the mean of the RR intervals. | An indicator allowing the calculation of total variability [57,58]. |
| Frequency methods | ||
| HF | High frequency | These metrics are located between 0.15 and 0.4 Hz. HF is clearly related to PNS activity and has an HR-relaxant effect [56]. |
| nHFP | Normalized HF power | |
| LF | Low frequency | Located between 0.04 and 0.15 Hz. These metrics are the most controversial in terms of interpretation because they can be influenced by the sympathetic nervous system (SNS) and/or PNS. In any case, it seems that long-term records provide more information about SNS activity [56]. |
| nLFP | Normalized LF power | |
| LHR | LF/HF ratio | From this ratio between low and high frequencies, the sympatho-vagal balance can be evaluated. Due to the controversy in the interpretation of LF in isolation, the LF/HF ratio is used to more accurately estimate SNS activity [56]. |
| Non-linear methods (Poincaré diagram: consecutive RR intervals are transformed into a two-dimensional scatterplot) | ||
| SD1 | Standard deviation 1 | Transverse diameter of the ellipse; reflects short-term variability. |
| SD2 | Standard deviation 2 | Longitudinal diameter of the ellipse; reflects long-term variability. |
| SSR | SD1/SD2 ratio | Reflects the activity of the autonomic nervous system (ANS) [33]. |
2. Methodology
2.1. Information Sources
2.2. Search Strategy
2.3. Article Selection Algorithm and Search Results
2.4. Data Analysis
3. Results
Outlook and Limitations
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ANS | Autonomic nervous system |
| CAN | Cardiovascular autonomic neuropathy |
| CARTs | Cardiovascular autonomic reflex tests |
| CV | Coefficient of variation |
| DAN | Diabetic autonomic neuropathy |
| DM | Diabetes mellitus |
| DM2 | Diabetes mellitus type 2 |
| DN | Diabetic neuropathy |
| ECG | Electrocardiogram |
| ED | Erectile dysfunction |
| FMD | Flow-mediated dilation |
| HDL-c | High-density lipoprotein |
| HF | High frequency |
| HR | Heart rate |
| HRV | Heart rate variability |
| LDL-c | Low-density lipoprotein |
| LF | Low frequency |
| LHR | Low frequency–high frequency ratio |
| MSNA | Muscle sympathetic nerve activity |
| nHFP | Normalized high-frequency power |
| nLFP | Normalized low-frequency power |
| PAT | Peripheral arterial tonometry |
| PET | Positron emission tomography |
| PNS | Parasympathetic nervous system |
| PPG | Photoplethysmography |
| PSRH | Post-stretch reactive hyperemia |
| RH | Reactive hyperemia |
| RMSSD | Root mean square of the RR intervals |
| SBP | Systolic blood pressure |
| SD1 | Standard deviation 1 |
| SD2 | Standard deviation 2 |
| SDNN | Standard deviation of RR intervals |
| SNS | Sympathetic nervous system |
| SSR | SD1/SD2 ratio |
| TtP | Pulse transit time |
References
- International Diabetes Federation—Home. Available online: https://idf.org/atlas-idf-2019/ (accessed on 31 March 2021).
- Shlomo, M.; Polonsky, K.S.; Kronenberg, H.M.; Reed Larsen, P. Williams Tratado de Endocrinología, 13th ed.; Saunders Elsevier Inc.: Philadelphia, PA, USA, 2017; pp. 1386–1435, 1526–1557, 1633–1642. [Google Scholar]
- Hassan, G.S. Ewing Battery Used for Diagnosis of Autonomic Neuropathy in Type 2 Diabetic Patients in Babylon Governorate. Med. J. Babylon 2006, 3. Available online: https://www.iasj.net/iasj/download/fc44f1ae161432e8 (accessed on 23 February 2022).
- Hahad, O.; Wild, P.S.; Prochaska, J.H.; Schulz, A.; Hermanns, I.; Lackner, K.J.; Pfeiffer, N.; Schmidtmann, I.; Beutel, M.; Gori, T.; et al. Endothelial Function Assessed by Digital Volume Plethysmography Predicts the Development and Progression of Type 2 Diabetes Mellitus. J. Am. Heart Assoc. Cardiovasc. Cerebrovasc. Dis. 2019, 8, e012509. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6818038/ (accessed on 23 February 2022). [CrossRef]
- Bhadada, S.K.; Jyotsna, V.P.; Agarwal, J.K. Diabetic neuropathy: Current concepts. Indian Acad. Clin. Med. 2001, 2, 305–318. Available online: https://www.semanticscholar.org/paper/Diabetic-Neuropathy%3A-Current-Concepts-Bhadada-Sahay/7e46683dcfa2463ad554826b21894f74f0cd3ff0 (accessed on 23 February 2022).
- Duby, J.J.; Setter, S.M.; White, J.R.; Rasmussen, K.A. Diabetic neuropathy intensive review. Am. Soc. Health-Syst. Pharm. 2004, 61, 160–176. Available online: https://academic.oup.com/ajhp/article-abstract/61/2/160/5143172?redirectedFrom=fulltext&login=true#no-access-message (accessed on 23 February 2022). [CrossRef] [PubMed]
- Ravindranathan, D. Photoplethysmography for the Evaluation of Diabetic Autonomic Neuropathy; Cardiff University: Cardiff, UK, 2009; Available online: https://orca.cardiff.ac.uk/54981/1/U585370.pdf (accessed on 23 February 2022).
- Wei, H.-C.; Ta, N.; Hu, W.-R.; Xiao, M.-X.; Tang, X.-J.; Haryadi, B.; Liou, J.J.; Wu, H.-T. Digital Volume Pulse Measured at the Fingertip as an Indicator of Diabetic Peripheral Neuropathy in the Aged and Diabetic. Entropy 2019, 21, 1229. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7514575/ (accessed on 31 March 2021). [CrossRef]
- Ono, Y.; Esaki, K.; Takahashi, Y.; Nakabayashi, M.; Ichinose, M.; Lee, K. Muscular blood flow responses as an early predictor of the severity of diabetic neuropathy at a later stage in streptozotocin-induced type I diabetic rats: A diffuse correlation spectroscopy study. Biomed. Opt. Express 2018, 9, 4539–4551. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6157794/ (accessed on 23 February 2022). [CrossRef] [PubMed]
- Wei, H.-C.; Ta, N.; Hu, W.-R.; Wang, S.-Y.; Xiao, M.-X.; Tang, X.-J.; Chen, J.-J.; Wu, H.-T. Percussion Entropy Analysis of Synchronized ECG and PPG Signals as a Prognostic Indicator for Future Peripheral Neuropathy in Type 2 Diabetic Subjects. Diagnostics 2020, 10, 32. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7168256/ (accessed on 31 March 2021). [CrossRef] [PubMed]
- Lewis, J.E.; Lantigua, L.; Atlas, S.E.; Lopez, J.; Mendez, A.; Goldberg, S.; Medici, S.; Konefal, J.; Woolger, J.M.; Tiozzo, E.; et al. A cross-sectional assessment to detect type 2 diabetes with endothelial and autonomic nervous system markers using a novel system. J. Diabetes Metab. Disord. 2014, 13, 118. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4271477/ (accessed on 23 February 2022). [CrossRef]
- Vinik, A.I. Neuropathy: New concepts in evaluation and treatment. South. Med. Assoc. 2002, 95, 21–23. [Google Scholar] [CrossRef]
- Vinik, A.I.; Erbas, T. Recognizing and treating diabetic autonomic neuropathy. Clevel. Clin. J. Med. 2001, 68, 928–944. Available online: http://cairdtech.com/PCD/PCD%20Version%2010.87/WinRoot/StarScanHolter/Documentation/Recognizing%20and%20treating%20diabetic%20autonomic%20neuropathy.pdf (accessed on 23 February 2022). [CrossRef] [PubMed]
- Serhiyenko, V.A.; Serhiyenko, A.A. Cardiac autonomic neuropathy: Risk factors, diagnosis and treatment. World J. Diabetes 2018, 9, 1–24. Available online: http://www.wjgnet.com/1948-9358/full/v9/i1/1.htm (accessed on 23 February 2022). [CrossRef]
- Baqar, S.; Straznicky, N.E.; Lambert, G.; Kong, Y.W.; Dixon, J.B.; Jerums, G.; Ekinci, E.I.; Lambert, E. Comparison of endothelial function and sympathetic nervous system activity along the glucose continuum in individuals with differing metabolic risk profiles and low dietary sodium intake. BMJ Open Diabetes Res. Care 2019, 7, e000606. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6501854/ (accessed on 23 February 2022). [CrossRef]
- Tan, J.S.; Lin, C.C.; Chen, G.S. Vasomodulation of peripheral blood flow by focused ultrasound potentiates improvement of diabetic neuropathy. BMJ Open Diabetes Res. Care 2020, 8, e001004. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7078690/ (accessed on 23 February 2022). [CrossRef]
- Maiuolo, J.; Gliozzi, M.; Musolino, V.; Carresi, C.; Nucera, S.; Macrì, R.; Scicchitano, M.; Bosco, F.; Scarano, F.; Ruga, S.; et al. The Role of Endothelial Dysfunction in Peripheral Blood Nerve Barrier: Molecular Mechanisms and Pathophysiological Implications. Int. J. Mol. Sci. 2019, 20, 3022. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6628074/ (accessed on 23 February 2022). [CrossRef] [PubMed]
- Fisher, V.L.; Tahrani, A.A. Cardiac autonomic neuropathy in patients with diabetes mellitus: Current perspectives. Diabetes Metab. Syndr. Obes. Targets Ther. 2017, 10, 419–434. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5638575/ (accessed on 23 February 2022). [CrossRef] [PubMed]
- Vinik, A.; Maser, R.; Braxton, M.; Freeman, R. Diabetic Autonomic Neuropathy. Diabetes Care 2003, 26, 1553–1579. Available online: https://diabetesjournals.org/care/article/26/5/1553/24595/Diabetic-Autonomic-Neuropathy (accessed on 23 February 2022). [CrossRef] [PubMed]
- Barwick, A.L.; Tessier, J.W.; Janse de Jonge, X.; Ivers, J.R.; Chuter, V.H. Peripheral sensory neuropathy is associated with altered postocclusive reactive hyperemia in the diabetic foot. BMJ Open Diabetes Res. Care 2016, 4, e000235. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4947724/ (accessed on 31 March 2021). [CrossRef] [PubMed]
- Stirban, A. Microvascular Dysfunction in the Context of Diabetic Neuropathy. Curr. Diabetes Rep. 2014, 14, 541. [Google Scholar] [CrossRef]
- Malik, R.A.; Tesfaye, S.; Thompson, S.D.; Veves, A.; Sharma, A.K.; Boulton, A.J.; Ward, J.D. Endoneuriallocalisation of microvascular damage in human diabetic neuropathy. Diabetologia 1993, 36, 454–459. Available online: https://link.springer.com/content/pdf/10.1007/BF00402283.pdf (accessed on 23 February 2022). [CrossRef]
- Tesfaye, S.; Selvarajah, D. Advances in the epidemiology, pathogenesis and management of diabetic peripheral neuropathy. Diabetes Metab. Res. Rev. 2012, 28, 8–14. Available online: https://onlinelibrary.wiley.com/doi/abs/10.1002/dmrr.2239 (accessed on 23 February 2022). [CrossRef]
- Spallone, V.; Ziegler, D.; Freeman, R.; Bernardi, L.; Frontoni, S.; Pop-Busui, R.; Stevens, M.; Kempler, P.; Hilsted, J.; Tesfaye, S.; et al. Cardiovascular autonomic neuropathy in diabetes: Clinical impact, assessment, diagnosis, and management. Diabetes Metab. Res. Rev. 2011, 27, 639–653. Available online: https://onlinelibrary.wiley.com/doi/abs/10.1002/dmrr.1239 (accessed on 23 February 2022). [CrossRef] [PubMed]
- Pop-Busui, R.; Boulton, A.J.; Feldman, E.L.; Bril, V.; Freeman, R.; Malik, R.A.; Sosenko, J.M.; Ziegler, D. Diabetic Neuropathy: A Position Statement by the American Diabetes Association. Diabetes Care 2017, 40, 136–154. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6977405/ (accessed on 23 February 2022). [CrossRef]
- Ziegler, D.; Keller, J.; Maier, C.; Pannek, J. Diabetic Neuropathy. Exp. Clin. Endocrinol. Diabetes 2014, 122, 406–415. Available online: http://www.thieme-connect.de/DOI/DOI?10.1055/s-0034-1366435 (accessed on 23 February 2022). [CrossRef] [PubMed]
- Low, P.A.; Denq, J.-C.; Opfer-Gehrking, T.L.; Dyck, P.J.; O’Brien, P.C.; Slezak, J.M. Effect of age and gender on sudomotor and cardiovagal function and blood pressure response to tilt in normal subjects. Muscle Nerve 1997, 20, 1561–1568. Available online: https://onlinelibrary.wiley.com/doi/abs/10.1002/%28SICI%291097-4598%28199712%2920%3A12%3C1561%3A%3AAID-MUS11%3E3.0.CO%3B2-3 (accessed on 23 February 2022). [CrossRef]
- Pop-Busui, R. What Do We Know and We Do Not Know About Cardiovascular Autonomic Neuropathy in Diabetes. J. Cardiovasc. Transl. Res. 2012, 5, 463–478. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3634565/ (accessed on 23 February 2022). [CrossRef]
- Zhang, Z.; Ma, Y.; Fu, L.; Li, L.; Liu, J.; Peng, H.; Jiang, H. Combination of Composite Autonomic Symptom Score 31 and Heart Rate Variability for Diagnosis of Cardiovascular Autonomic Neuropathy in People with Type 2 Diabetes. J. Diabetes Res. 2020, 2020, 5316769. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7648703/ (accessed on 23 February 2022). [CrossRef]
- dos Santos Corrêa, A.P.; AntunesFetter, C.; Ramos Figueira, F.; Axmann de Castro, M.; Pinto Ribeiro, J.; D’AgordSchaan, B. Effect of Acute Inspiratory Muscle Exercise on Blood Flow of Resting and Exercising Limbs and Glucose Levels in Type 2 Diabetes. PLoS ONE 2015, 10, e0121384. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4372446/ (accessed on 31 March 2021).
- Gandhi, P.G.; Gundu, H.R. Detectionofneuropathyusing a sudomotor test in type 2 diabetes. Degener. Neurol. Neuromuscul. Dis. 2015, 5, 1–7. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7337199/ (accessed on 23 February 2022).
- Robles Cabrera, A. Regulación Autonómica de la Frecuencia Cardiaca y la Presión Arterial Sistólica Ante el Ortostatismo y la Respiración Rítmica Controlada en Sujetos Diabéticos de Corta (<2 años de diagnóstico) y Larga Evolución (>2 Años de Diagnóstico) del Instituto Nacional de Ciencias Médicas y Nutrición Salvador Zubirán Durante el Período Comprendido Entre Agosto de 2008 a Agosto de 2014. 2015. Available online: https://repositorioinstitucional.buap.mx/handle/20.500.12371/8878 (accessed on 23 February 2022).
- Wu, H.-T.; Hsu, P.-C.; Sun, C.-K.; Wang, H.-J.; Liu, C.-C.; Chen, H.-R.; Liu, A.-B.; Tang, C.-J.; Lo, M.-T. Assessment of autonomic dysfunction in patients with type 2 diabetes using reactive hyperemia. J. Theor. Biol. 2013, 330, 9–17. Available online: https://www.sciencedirect.com/science/article/pii/S0022519313001483 (accessed on 23 February 2022). [CrossRef]
- Palazzo, P.; Maggio, P.; Altavilla, R.; Di Flaviani, A.; Giordani, I.; Malandrucco, I.; Picconi, F.; Passarelli, F.; Pasqualetti, P.; Ercolani, M.; et al. Cerebral Hemodynamics and Systemic Endothelial Function Are Already Impaired in Well-Controlled Type 2 Diabetic Patients, with Short-Term Disease. PLoS ONE 2013, 8, e83287. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3877017/ (accessed on 23 February 2022). [CrossRef]
- Sardu, C.; Paolisso, P.; Santamaria, M.; Sacra, C.; Pieretti, G.; Rizzo, M.R.; Barbieri, M.; Scisciola, L.; Nicoletti, G.; Paolisso, G.; et al. Cardiac syncope recurrence in type 2 diabetes mellitus patients vs. normoglycemics patients: The CARVAS study. Diabetes Res. Clin. Pract. 2019, 151, 152–162. Available online: https://www.sciencedirect.com/science/article/pii/S0168822719302578 (accessed on 23 February 2022). [CrossRef] [PubMed]
- Kandhai-Ragunath, J.J.; Jørstad, H.T.; de Man, F.H.; Peters, R.J.; von Birgelen, C. Approaches for non-invasive assessment of endothelial function: Focus on peripheral arterial tonometry. Neth. Heart J. 2013, 21, 214–218. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3636344/ (accessed on 23 February 2022). [CrossRef] [PubMed]
- Wilk, G.; Osmenda, G.; Matusik, P.; Nowakowski, D.; Jasiewicz-Honkisz, B.; Ignacak, A.; Czesnikiewicz-Guzik, M.; Guzik, T.J. Endothelial function assessment in atherosclerosis. Comparison of brachial artery flow-mediated vasodilation and peripheral arterial tonometry. Pol. Arch. Med. Wewn. 2013, 123, 443–452. Available online: http://scielo.sld.cu/pdf/ibi/v20n4/ibi08401.pdf (accessed on 23 February 2022). [CrossRef]
- Olamaei, N. Reactive Hyperemia as Endothelial Function Determinant Using Plethysmography Methods; Biomedical Engineering Institute Faculty of Medicine, Montreal University: Montreal, QC, Canada, 2009; Available online: https://papyrus.bib.umontreal.ca/xmlui/bitstream/handle/1866/2854/OlamaeiNina_2009_memoire.pdf (accessed on 24 February 2022).
- Figueroa, A.; González, J.; Rodríguez, R.; Jiménez Rivero, V.; Rodríguez Álvarez, J. Evaluación clínica del pletismógrafo digital Angiodin PD 3000: Estudio fase II. Rev. Cuba. Investig. Biomédicas 2001, 20, 279–286. Available online: http://scielo.sld.cu/pdf/ibi/v20n4/ibi08401.pdf (accessed on 23 February 2022).
- Jasperse, J.; Shoemaker, K.; Gray, E.; Clifford, P.h. Positional differences in reactive hyperemia provide insight into initial phase of exercise hyperemia. J. Appl. Physiol. 2015, 119, 569–575. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4556838/ (accessed on 23 February 2022). [CrossRef]
- Shibata, N.; Matsunaga, T.; Kudo, D.; Sasaki, K.; Mizutani, T.; Sato, M.; Chida, S.; Hatakeyama, K.; Watanabe, M.; Shimada, Y. Postcontraction hyperemia after electrical stimulation: Potential utility in rehabilitation of patients with upper extremity paralysis. Biomed. Res. 2016, 37, 73–76. Available online: https://www.ncbi.nlm.nih.gov/pubmed/26912142 (accessed on 23 February 2022). [CrossRef]
- Pettit, A.P.; Kipen, H.; Laumbach, R.; Ohman-Strickland, P.; Kelly-McNeill, K.; Cepeda, C.; Fan, Z.-H.; Amorosa, L.; Lubitz, S.; Schneider, S.; et al. Disrupted Nitric Oxide Metabolism from Type II Diabetes and Acute Exposure to Particulate Air Pollution. PLoS ONE 2015, 10, e0144250. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4682772/ (accessed on 23 February 2022). [CrossRef]
- Low, B.H.; Lin, Y.D.; Huang, B.W.; Chia, T.; Bau, J.G.; Huang, H.Y. Impaired Microvascular Response to Muscle Stretching in Chronic Smokers With Type 2 Diabetes. Front. Bioeng. Biotechnol. 2020, 8, 602. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7300253/ (accessed on 23 February 2022). [CrossRef] [PubMed]
- Shimada, S.; Todoki, K.; Omori, Y.; Toyama, T.; Matsuo, M.; Wada-Takahashi, S.; Takahashi, S.-S.; Lee, M.-C. Contribution of nitrergic nerve in canine gingival reactive hyperemia. J. Clin. Biochem. Nutr. 2015, 56, 98–104. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4345180/ (accessed on 22 February 2022). [CrossRef]
- Krishnan, A.; Lucassen, E.B.; Hogeman, C.; Blaha, C.; Leuenberger, U.A. Effects of Limb Posture on Reactive Hyperemia. Eur. J. Appl. Physiol. 2011, 111, 1415–1420. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3285391/ (accessed on 22 February 2022). [CrossRef]
- Schreuder, T.H.A.; Green, D.J.; Hopman, M.T.E.; Thijssen, D.H.J. Acute impact of retrograde shear rate on brachial and superficial femoral artery flow-mediated dilation in humans. Physiol. Rep. 2014, 2, e00193. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3967676/ (accessed on 22 February 2022). [CrossRef] [PubMed]
- Thijssen, D.H.J.; Atkinson, C.L.; Ono, K.; Sprung, V.S.; Spence, A.; Pugh, C.J.A.; Green, D.J. Sympathetic nervous system activation, arterial shear rate, and flow-mediated dilation. J. Appl. Physiol. 2014, 116, 1300–1307. Available online: https://journals.physiology.org/doi/full/10.1152/japplphysiol.00110.2014 (accessed on 1 March 2022). [CrossRef] [PubMed]
- Yoshida, M.; Tomiyama, H.; Shiina, K.; Odaira, M.; Yamashina, A. The difference of the influence of autonomic nervous activation caused by reactive hyperemia on two different endothelial function tests. Eur. Heart J. 2013, 34, 1436. Available online: https://academic.oup.com/eurheartj/article/34/suppl_1/P1436/2860620 (accessed on 1 March 2022). [CrossRef]
- Lambert, E.A.; Teede, H.; Sari, C.I.; Jona, E.; Shorakae, S.; Woodington, K.; Hemmes, R.; Eikelis, N.; Straznicky, N.E.; De Courten, B.; et al. Sympathetic activation and endothelial dysfunction in polycystic ovary syndrome are not explained by either obesity or insulin resistance. Clin. Endocrinol. 2015, 83, 812–819. Available online: https://onlinelibrary.wiley.com/doi/abs/10.1111/cen.12803 (accessed on 22 February 2022). [CrossRef]
- Short, K.R.; Blackett, P.R.; Gardner, A.W.; Copeland, K.C. Vascular health in children and adolescents: Effects of obesity and diabetes. Vasc. Health Risk Manag. 2009, 5, 973–990. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2788602/ (accessed on 22 February 2022).
- Petrofsky, J.; Lee, S.; Cuneo, M. Effects of aging and type 2 diabetes on resting and post occlusive hyperemia of the forearm; the impact of rosiglitazone. BMC Endocr. Disord. 2005, 5, 4. Available online: http://www.biomedcentral.com/1472-6823/5/4 (accessed on 22 February 2022). [CrossRef]
- Petrofsky, J.; Berk, L.; Al-Nakhli, H. The Influence of Autonomic Dysfunction Associated with Aging and Type 2 Diabetes on Daily Life Activities. Exp. Diabetes Res. 2012, 2012, 1–12. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3332074/ (accessed on 31 March 2021). [CrossRef]
- Mori, H.; Okada, Y.; Kawaguchi, M.; Iwata, S.; Yoshikawa, M.; Sonoda, S.; Sugai, K.; Tanaka, K.; Hajime, M.; Narisawa, M.; et al. A Study of the Vascular Endothelial Function in Patients with Type 2 Diabetes Mellitus and Rheumatoid Arthritis. Intern. Med. 2019, 58, 1383–1390. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6548912/ (accessed on 22 February 2022). [CrossRef]
- Lorenzo, S.; Minson, C.T. Human cutaneous reactive hyperaemia: Role of BKCa channels and sensory nerves. J. Physiol. 2007, 585 Pt 1, 295. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2375471/ (accessed on 1 March 2023). [CrossRef]
- Nouri, M.N.; Ahmed, A.; Bril, V.; Orszag, A.; Ng, E.; Nwe, P.; Perkins, B.A. Diabetic neuropathy and axon reflex-mediated neurogenic vasodilatation in type 1 diabetes. PLoS ONE 2012, 7, e34807. Available online: https://pubmed.ncbi.nlm.nih.gov/22529938/ (accessed on 4 March 2023).
- Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology. Heart Rate Variability: Standards of Measurement, Physiological Interpretation and Clinical Use. Circulation 1996, 93, 1043–1065. Available online: https://www.ahajournals.org/doi/full/10.1161/01.CIR.93.5.1043 (accessed on 9 January 2022). [CrossRef]
- Seyhood, G.; Talib, B.; Ghanim, K. Detection of diabetic autonomic neuropathy in diabetes mellitus type 2. Med. J. Babylon 2008, 5, 71–86. Available online: https://www.iasj.net/iasj/download/8cee54e23bb75090 (accessed on 23 February 2022).
- Di Carli, M.F.; Bianco-Batlles, D.; Landa, M.E.; Kazmers, A.; Groehn, H.; Muzik, O.; Grunberger, G. Effects of Autonomic Neuropathy on Coronary Blood Flow in Patients with Diabetes Mellitus. Circulation 1999, 100, 813–819. Available online: https://www.ahajournals.org/doi/10.1161/01.cir.100.8.813?url_ver=Z39.88-2003&rfr_id=ori:rid:crossref.org&rfr_dat=cr_pub%20%200pubmed (accessed on 22 February 2022). [CrossRef]
- Tuttolomondo, A.; Del Cuore, A.; La Malfa, A.; Casuccio, A.; Daidone, M.; Maida, C.D.; Di Raimondo, D.; Di Chiara, T.; Puleo, M.G.; Norrito, R.; et al. Assessment of heart rate variability (HRV) in subjects with type 2 diabetes mellitus with and without diabetic foot: Correlations with endothelial dysfunction indices and markers of adipo-inflammatory dysfunction. Cardiovasc. Diabetol. 2021, 20, 142. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8281716/ (accessed on 21 December 2021). [CrossRef]
- Murata, M.; Tamemoto, H.; Otani, T.; Jinbo, S.; Ikeda, N.; Kawakami, M.; Ishikawa, S. Endothelial impairment and bone marrow-derived CD34+/133+ cells in diabetic patients with erectile dysfunction. J. Diabetes Investig. 2012, 3, 526–533. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4015432/ (accessed on 22 February 2022). [CrossRef]
- Hartmann, A.; Schlottog, B.; Jungmann, E.; Böhm, B.O.; Usadel, K.H.; Kaltenbach, M. Somatic pain threshold and reactive hyperemia in autonomic diabetic neuropathy and silent myocardial ischemia. Int. J. Cardiol. 1993, 42, 121–127. Available online: https://www.sciencedirect.com/science/article/pii/016752739390081Q (accessed on 22 February 2022). [CrossRef]
- Ando, A.; Miyamoto, M.; Saito, N.; Kotani, K.; Kamiya, H.; Ishibashi, S.; Tavakoli, M. Small fibre neuropathy is associated with impaired vascular endothelial function in patients with type 2 diabetes. Front. Endocrinol. 2021, 12, 653277. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8079951/ (accessed on 4 March 2023). [CrossRef]
- Sheila, R.P.; Sheelajoice, P.; Subbalakshmi, N.K.; Hemalatha, H.; Kishan, K. Validity of standard deviation and maximum: Minimum ratio in quantifying cardiac autonomic function following acute unilateral brachial artery occlusion. J. Chin. Clin. Med. 2011, 6, 28–32. Available online: https://www.cabdirect.org/cabdirect/abstract/20113239485 (accessed on 21 February 2022).
- Petrofsky, J.S. The Effect of Type-2-Diabetes-Related Vascular Endothelial Dysfunction on Skin Physiology and Activities of Daily Living. J. Diabetes Sci. Technol. 2011, 5, 657–667. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3192631/ (accessed on 21 February 2022). [CrossRef]
- Akasaka, T.; Sueta, D.; Tabata, N.; Takashio, S.; Yamamoto, E.; Izumiya, Y.; Tsujita, K.; Kojima, S.; Kaikita, K.; Matsui, K.; et al. Effects of the Mean Amplitude of Glycemic Excursions and Vascular Endothelial Dysfunction on Cardiovascular Events in Nondiabetic Patients with Coronary Artery Disease. J. Am. Heart Assoc. 2017, 6, e004841. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5524064/ (accessed on 21 February 2022). [CrossRef]
- Marcinkevics, Z.; Aglinska, A.; Rubins, U.; Grabovskis, A. Remote Photoplethysmography for Evaluation of Cutaneous Sensory Nerve Fiber Function. Sensors 2021, 21, 1272. Available online: https://www.mdpi.com/1424-8220/21/4/1272/htm (accessed on 21 February 2022). [CrossRef] [PubMed]
- Wei, H.-C.; Hu, W.-R.; Ta, N.; Xiao, M.-X.; Tang, X.-J.; Wu, H.-T. Prognosis of Diabetic Peripheral Neuropathy via Decomposed Digital Volume Pulse from the Fingertip. Entropy 2020, 22, 754. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7517300/ (accessed on 31 March 2021). [CrossRef] [PubMed]




| Study | Aim | RH Method | ANS Method | Results |
|---|---|---|---|---|
| Studies That Made Comparisons between Healthy Subjects and Diabetic Patients | ||||
| [33] Wu HT et al. (2013). | To assess the impacts of age and diabetes on autonomic function at baseline and during reactive hyperemia. | Piezoresistive sensor using air pressure sensation system (APSS): occlusion pressure at 200 mmHg in the upper limbs for 4 min. | Frequency analysis and non-linear Poincaré method of HRV. | Dilatation index (DI): 1.88 ± 0.54 (healthy young subjects), 1.67 ± 0.56 (healthy upper-middle-aged subjects), and 1.38 ± 0.51 (type 2 diabetic patients). There was no significant difference. Changes in SSR(1–10) before and after the induction of reactive hyperemia, showing a significant difference in type 2 diabetic patients. A direct correlation between autonomic nervous system and endothelium function after RH (r = 0.29; p = 0.033) in all subjects of the study. |
| [57] Seyhood G et al. (2008). | To detect autonomic neuropathy in type 2 diabetic patients. | FMD: occlusion pressure at 250 mmHg in the upper limbs for 5 min. | Cardiovascular autonomic reflex test. | FMD% was significantly (p < 0.001) lower (64%) in diabetic patients in comparison to control subjects. The FMD% in diabetic patients was significantly correlated with the heart rate response to the Valsalva maneuver (Ω = 0.48; p < 0.01), HR variation during deep breathing (Ω = 0.59; p < 0.001), the immediate HR response to standing (Ω = 0.37; p < 0.02), the blood pressure response to standing (Ω = 0.64; p < 0.001), and the blood pressure response to hand gripping (Ω = 0.41; p < 0.01). FMD% was significantly (p < 0.001) and negatively (r = −0.67) correlated with the plasma nitrite concentration. |
| [15] Baqar S et al. (2019). | To compare sympathetic nervous system activity and endothelial function based on low sodium intake. | Peripheral arterial tonometry: suprasystolic occlusion pressure at 60 mmHg or 200 mmHg in the upper limbs for 5 min and 5–10 min post-occlusion. | Muscle sympathetic nerve activity (MSNA) | The diabetic patients with cardiometabolic risk factors (T2DM+) had higher systolic blood pressures (p = 0.04) and heart rates (p = 0.002) than normal glucose tolerance (NGT), impaired glucose tolerance (IGT) and treatment-naive diabetic patients (T2DM−). The endothelial-function-derived measure of the PAT ratio (p = 0.04) was lower in the T2DM+ group, and the reactive hyperemic index trended towards being lower in the T2DM+ group (p = 0.08). Despite the lower burst incidence, the baroreflex effectiveness index was shown to be lower in the T2DM+ group compared to all other groups (p = 0.0002). |
| [30] Dos Santos AP et al. (2015). | To evaluate the effects of inspiratory loading on blood flow in diabetic patients. | FMD: occlusion pressure at 250 mmHg in the upper limbs for 5 min. | Cardiovascular autonomic reflex test | FMD% was decreased in both T2DM (6 ± 2) and T2DM with CAN (4 ± 3) patients compared to healthy subjects (12 ± 2). Resting HR was higher (p < 0.05) in T2DM with CAN (80 ± 7) compared to healthy subjects (68 ± 6), and it had a trend to be higher than in T2DM. |
| [58] Di Carli MF et al. (1999). | To evaluate the sympathetic regulation of myocardial blood flow in diabetic patients with autonomic neuropathy. | FMD: occlusion pressure at 300 mmHg in the upper limbs for 5 min. | Cardiovascular autonomic reflex test and radioimaging of the sympathetic innervation of the heart. | FMD% was lower in diabetic patients with (7.0 ± 3.4; p = 0.018) and without (7.8 ± 8.1; p = 0.045) sympathetic nerve dysfunction than in the healthy volunteers (13.7 ± 4.1). FMD% was similar in both groups of diabetic patients. |
| [43] Low BH et al. (2020). | To investigate the microvascular response of the skin to muscle stretching stimuli in diabetic smokers. | Laser Doppler: reactive hyperemia in the cutaneous microcirculation after a 10 s stretching stimulus. | Spectral analysis of laser Doppler. | After a 10 s stretching stimulus, blood flow was significantly elevated within the first 10 s, followed by a gradual decrease. The blood flow of the calf skin after the stretch was not significantly different between normal subjects and diabetic non-smokers; however, there were significant differences between diabetic smokers and diabetic non-smokers (p < 0.01) and between diabetic smokers and normal subjects (p < 0.01). The intensity of the spectrum of the neurogenic activity reached its maximum value immediately after the stretching stimulus (p < 0.05) and then decreased gradually in the next periods for diabetic subjects, while there was almost no neurogenic response for the normal subjects. Compared to the normal subjects, the intensity of the neurogenic activity at baseline was significantly lower for smokers (p < 0.05). |
| [59] Tuttolomondo A et al. (2021). | To evaluate the alteration of the sympatho-vagal balance and correlation with endothelial dysfunction in patients with diabetes mellitus with and without diabetic foot syndrome and in healthy subjects. | Peripheral arterial tonometry: suprasystolic occlusion pressure at 60 mmHg or 200 mmHg in the upper limbs for 5 min. | HRV (SDNN, RMSSD, and spectral analysis). | Diabetic patients with diabetes foot syndrome (DFS) showed a higher HF (21.23 ± 14.68 vs. 11.10 ± 11.58; p = 0.002), a lower LF/HF ratio (1.63 ± 1.66 vs. 3.18 ± 2.82; p = 0.001), and a lower RH (1.60 ± 0.33 vs. 2.01 ± 0.69; p < 0.0005) than the diabetic controls and a lower RH (1.60 ± 0.33 vs. 2.20 ± 0.38; p = 0.00) and a similar HF% (21.23 ± 14.68 vs. 14.70 ± 11.15; p = 0.82) and LF/HF ratio (1.63 ± 1.66 vs. 1.07 ± 0.44; p = 0.59) compared to the healthy controls. Diabetic control patients showed a higher LF/HF ratio (3.18 ± 2.82 vs. 1.07 ± 0.44; p < 0.0005) and a comparable RH (2.01 ± 0.69 vs. 2.20 ± 0.38; p = 0.315) compared to the healthy controls. In DFS, there was a statistically significant negative correlation between the RH and RMSDD (Pearson index: −0.47; p = 0.0001), the standard deviation (Pearson index: −0.374; p = 0.002), and HF% (Pearson index: −0.395; p = 0.001) and a positive correlation with the LF/HF ratio (Pearson index: 0.280; p = 0.026). |
| Studies That did not compare healthy subjects and diabetic patients | ||||
| [60] Murata M et al. (2012). | To determine the relationship between endothelial dysfunction and erectile dysfunction (ED) in type 2 diabetic patients. | FMD: suprasystolic occlusion pressure at 50 mmHg in the upper limbs for 5 min. | HRV (CV of RR intervals) | FMD was significantly lower (p = 0.038) in the diabetic patients with ED (2.84 ± 0.34%) than in those without ED (3.82 ± 0.39%). In the diabetic patients without autonomic neuropathy, FMD was significantly reduced in the diabetic patients with ED compared to those without ED (2.43 ± 0.38% vs. 3.92 ± 0.41%, p = 0.015). In contrast, in the diabetic patients with autonomic neuropathy, there was no difference in FMD between the diabetic patients with and without ED. |
| [61] Hartmann A et al. (1993). | To investigate the pain threshold and reactive hyperemia in diabetic patients with and without autonomic neuropathy and in patients with silent myocardial ischemia. | Peripheral arterial tonometry: suprasystolic occlusion pressure in the upper limbs for 5 min. | HRV at rest and during deep breaths (CV of RR and RMSSD intervals) | The maximum hyperemia was obtained 15 s after the deflation of the cuff, and hyperemia returned to baseline 300 s after the termination of forearm ischemia. Total post-ischemic (with exercise) reactive hyperemia was significantly higher (p < 0.01) in patients with silent myocardial ischemia (219.56 ± 90.5 mL/l00 mL of tissue) compared to diabetic patients with neuropathy (179.6 ± 79 mL/100 mL of tissue) and without neuropathy (180.8 ± 58 mL/100 mL of tissue). There was no significant difference in total reactive hyperemia in diabetic patients with and without neuropathy. |
| [20] Barwick AL et al. (2016). | To examine the relationships between clinically detectable peripheral sensory neuropathy, cardiac autonomic dysfunction, and RH. | Laser Doppler: occlusion pressure at 220 mmHg in the upper limbs for 3 min and 4 min of post-occlusion recording. | HRV (SDNN, RMSSD, and spectral analysis). | The presence of neuropathy was associated with a longer latency to peak after occlusion but not with changes in the peak magnitude. HRV parameters did not predict responses. |
| [62] Ando A et al. (2021). | To investigate the association of RH and other physiological vascular markers with neuropathy markers. | Peripheral arterial tonometry and FMD: occlusion of the non-dominant arm maintained for 5 min above 200 mmHg or at least 50 mmHg above the systolic blood pressure. | CV of RR intervals at rest and during deep breathing. | RH had no significant statistical different between diabetic patients with (FMD: 1.6 ± 1.2; PAT: 1.7 ± 0.5) and without neuropathy (FMD: 1.8 ± 1.3; p = 0.39; PAT: 1.7 ± 0.4; p = 0.78). RH-PAT had significant statistical relations with electrophysiological grading (Spearman’s coefficients: 0.22; p < 0.05). |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
López-Galán, E.; Montoya-Pedrón, A.; Barrio-Deler, R.; Sánchez-Hechavarría, M.E.; Muñoz-Bustos, M.E.; Muñoz-Bustos, G.A. Reactive Hyperemia and Cardiovascular Autonomic Neuropathy in Type 2 Diabetic Patients: A Systematic Review of Randomized and Nonrandomized Clinical Trials. Medicina 2023, 59, 770. https://doi.org/10.3390/medicina59040770
López-Galán E, Montoya-Pedrón A, Barrio-Deler R, Sánchez-Hechavarría ME, Muñoz-Bustos ME, Muñoz-Bustos GA. Reactive Hyperemia and Cardiovascular Autonomic Neuropathy in Type 2 Diabetic Patients: A Systematic Review of Randomized and Nonrandomized Clinical Trials. Medicina. 2023; 59(4):770. https://doi.org/10.3390/medicina59040770
Chicago/Turabian StyleLópez-Galán, Erislandis, Arquímedes Montoya-Pedrón, Rafael Barrio-Deler, Miguel Enrique Sánchez-Hechavarría, Mario Eugenio Muñoz-Bustos, and Gustavo Alejandro Muñoz-Bustos. 2023. "Reactive Hyperemia and Cardiovascular Autonomic Neuropathy in Type 2 Diabetic Patients: A Systematic Review of Randomized and Nonrandomized Clinical Trials" Medicina 59, no. 4: 770. https://doi.org/10.3390/medicina59040770






