Detection of Orange Essential Oil, Isopropyl Myristate, and Benzyl Alcohol in Lemon Essential Oil by FTIR Spectroscopy Combined with Chemometrics
Abstract
:1. Introduction
2. Materials and Methods
2.1. Equipment
2.2. Essential Oils and Chemicals
2.3. Sample Preparation
2.4. FTIR Measurements
2.5. Chemometrics
2.5.1. Hierarchical Cluster Analyses
2.5.2. Quantification of Adulterants in the Lemon Essential Oils
3. Results
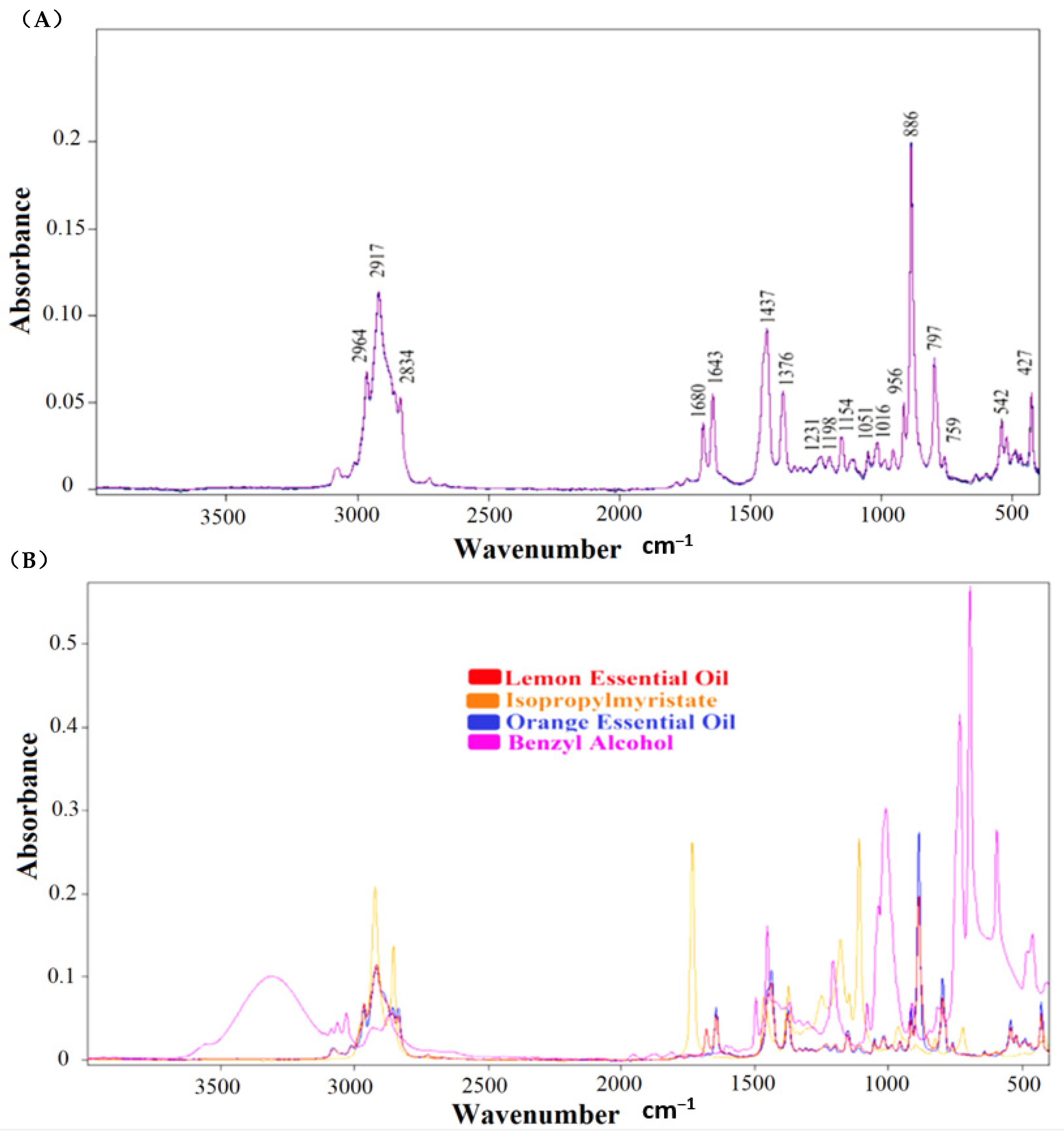
3.1. Characterization of Lemon Essential Oils by FTIR Spectroscopy
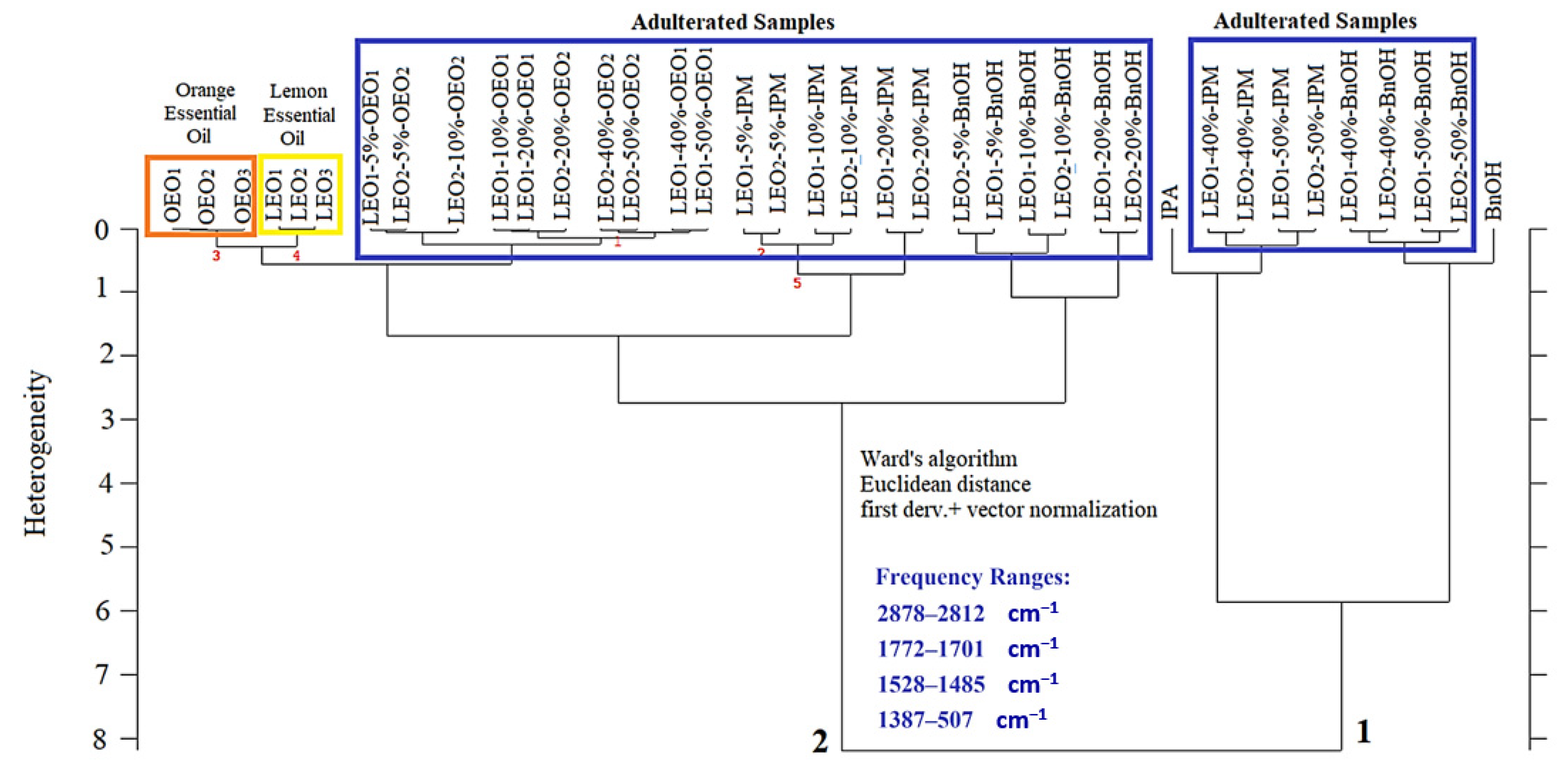
3.2. Determination of Authentic Lemon Essential Oil by Hierarchical Cluster Analysis
3.3. Quantification of OEO, BnOH, and IPM in LEO by Using PLSR and PCR Models
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Perdones, A.; Sánchez-González, L.; Chiralt, A.; Vargas, M. Effect of chitosan-lemon essential oil coatings on storage-keeping quality of strawberry. Postharvest Biol. Technol. 2012, 70, 32–41. [Google Scholar] [CrossRef]
- Boren, K.E.; Young, D.G.; Wooley, C.L.; Smith, B.L.; Carlson, R.E. Detecting essential oil adulteration. J. Environ. Anal. Chem. 2015, 2, 1–4. [Google Scholar]
- Mustafa, N. Citrus Essential Oils: Current and prospective uses in the food industry. Recent Pat. Food. Nutr. Agric. 2015, 7, 115–127. [Google Scholar] [CrossRef] [PubMed]
- Hao, C.W.; Lai, W.S.; Ho, C.T.; Sheen, L.Y. Antidepressant-like effect of lemon essential oil is through a modulation in the levels of norepinephrine, dopamine, and serotonin in mice: Use of the tail suspension test. J. Funct. Foods 2013, 5, 370–379. [Google Scholar] [CrossRef]
- Huynh, T.V.; Caffin, N.; Dykes, G.; Bhandari, B. Optimization of the microencapsulation of lemon myrtle oil using response surface methodology. Dry. Technol. 2008, 26, 357–368. [Google Scholar] [CrossRef]
- Do, T.K.T.; Hadji-Minaglou, F.; Antoniotti, S.; Fernandez, X. Authenticity of essential oils. TrAC Trends Anal. Chem. 2015, 66, 146–157. [Google Scholar] [CrossRef]
- Tony, C.; Oct, B.; Adulteration, O.; Organisation, I.S.; Which, H.; Trade, T.P.; Pharmacopoeia, B.; Uk, M.C.; Pharmacopoeia, E.; Pharmacopoeia, U.S.; et al. The adulteration of essential oils-and the consequences to aromatherapy & natural perfumery practice. By Tony Burfield October 2003. In Proceedings of the the International Federation of Aromatherapists Annual AGM, London, UK, 11 October 2003. [Google Scholar]
- Schipilliti, L.; Dugo, P.; Bonaccorsi, I.; Mondello, L. Authenticity control on lemon essential oils employing Gas Chromatography-Combustion-Isotope Ratio Mass Spectrometry (GC-C-IRMS). Food Chem. 2012, 131, 1523–1530. [Google Scholar] [CrossRef]
- International Standard ISO 855:2003 Oil of Lemon [Citrus limon (L.) Burm. f.], Obtained by Expression; ISO: Geneva, Switzerland, 2003; Available online: https://www.iso.org/standard/32042.html (accessed on 10 December 2020).
- Mehl, F.; Marti, G.; Boccard, J.; Debrus, B.; Merle, P.; Delort, E.; Baroux, L.; Raymo, V.; Velazco, M.I.; Sommer, H.; et al. Differentiation of lemon essential oil based on volatile and non-volatile fractions with various analytical techniques: A metabolomic approach. Food Chem. 2014, 143, 325–335. [Google Scholar] [CrossRef]
- Lifshitz, A.; Stepak, Y.; Basker, H.B. Characterization of Israel Lemon Oil and Detection of its Adulteration. J. Food Sci. 1969, 34, 254–257. [Google Scholar] [CrossRef]
- Alvarez-Ordonez, A.; Prieto, M. Fourier Transform Infrared Spectroscopy in Food Microbiology; Hartel, R.W., Clark, J.P., Rodriguez-Lazaro, D., Topping, D., Eds.; Springer: New York, NY, USA, 2012; ISBN 9781461438120. [Google Scholar]
- Rodriguez-Saona, L.E.; Allendorf, M.E. Use of FTIR for rapid authentication and detection of adulteration of food. Annu. Rev. Food Sci. Technol. 2011, 2, 467–483. [Google Scholar] [CrossRef]
- Berechet, M.D.; Calinescu, I.; Stelescu, M.D.; Manaila, E.; Craciun, G.; Purcareanu, B.; Mihaiescu, D.E.; Rosca, S.; Fudulu, A.; Niculescu-Aron, I.G.; et al. Composition of the essential oil of Rosa damascena Mill. cultivated in Romania. Rev. Chim. 2015, 66, 1986–1991. [Google Scholar]
- Li, S.; Zhu, X.; Zhang, J.; Li, G.; Su, D.; Shan, Y. Authentication of Pure Camellia Oil by Using Near Infrared Spectroscopy and Pattern Recognition Techniques. J. Food Sci. 2012, 77, 374–380. [Google Scholar] [CrossRef] [PubMed]
- Minteguiaga, M.; Dellacassa, E.; Iramain, M.A.; Catalán, C.A.N.; Brandán, S.A. FT-IR, FT-Raman, UV–Vis, NMR and structural studies of carquejyl acetate, a distinctive component of the essential oil from Baccharis trimera (less.) DC. (Asteraceae). J. Mol. Struct. 2019, 1177, 499–510. [Google Scholar] [CrossRef]
- Bombarda, I.; Dupuy NJ, P.D.; Le Van Da, J.P.; Gaydou, E.M. Comparative chemometric analyses of geographic origins and compositions of lavandin var. Grosso essential oils by mid infrared spectroscopy and gas chromatography. Anal. Chim. Acta 2008, 613, 31–39. [Google Scholar] [CrossRef] [PubMed]
- Andersen, F.A. Final report on the safety assessment of Benzyl Alcohol, Benzoic Acid, and Sodium Benzoate. Int. J. Toxicol. 2001, 20, 23–50. [Google Scholar]
- Myristate, M.; Myristate, I. Final report on the safety assessment of myristyl myristate and isopropyl myristate. Int. J. Toxicol. 1982, 1, 55–80. [Google Scholar]
- Sivakesava, S.; Irudayaraj, J. Detection of inverted beet sugar adulteration of honey by FTIR spectroscopy. J. Sci. Food Agric. 2001, 81, 683–690. [Google Scholar] [CrossRef]
- Rohman, A.; Man, Y.B.C. Application of Fourier transform infrared spectroscopy for authentication of functional food oils. Appl. Spectrosc. Rev. 2012, 47, 1–13. [Google Scholar] [CrossRef]
- Boughendjioua, H. Fourier transformed infrared spectroscopy analysis of constituents of lemon essential oils from Algeria. Am. J. Opt. Photonics 2017, 5, 30. [Google Scholar] [CrossRef] [Green Version]
- Hasani, S.; Ojagh, S.M.; Ghorbani, M. Nanoencapsulation of lemon essential oil in Chitosan-Hicap system. Part 1: Study on its physical and structural characteristics. Int. J. Biol. Macromol. 2018, 115, 143–151. [Google Scholar] [CrossRef]
- Benoudjit, F.; Maameri, L.; Ouared, K.; History, A. Evaluation of the quality and composition of lemon (Citrus limon) peel essential oil from an Algerian fruit juice industry ARTICLE INFO ABSTRACT/RESUME. Alger. J. Environ. Sci. Technol. Dec. Ed. 2020, 6, 1575–1581. [Google Scholar]
- Taylan, O.; Cebi, N.; Tahsin Yilmaz, M.; Sagdic, O.; Bakhsh, A.A. Detection of lard in butter using Raman spectroscopy combined with chemometrics. Food Chem. 2020, 332, 127344. [Google Scholar] [CrossRef] [PubMed]
- Cebi, N.; Yilmaz, M.T.; Sagdic, O. A rapid ATR-FTIR spectroscopic method for detection of sibutramine adulteration in tea and coffee based on hierarchical cluster and principal component analyses. Food Chem. 2017, 229, 517–526. [Google Scholar] [CrossRef] [PubMed]
- Gok, S.; Severcan, M.; Goormaghtigh, E.; Kandemir, I.; Severcan, F. Differentiation of Anatolian honey samples from different botanical origins by ATR-FTIR spectroscopy using multivariate analysis. Food Chem. 2015, 170, 234–240. [Google Scholar] [CrossRef] [PubMed]
- Cebi, N.; Durak, M.Z.; Toker, O.S.; Sagdic, O.; Arici, M. An evaluation of Fourier transforms infrared spectroscopy method for the classification and discrimination of bovine, porcine and fish gelatins. Food Chem. 2016, 190, 1109–1115. [Google Scholar] [CrossRef] [PubMed]
- Ropodi, A.I.; Pavlidis, D.E.; Mohareb, F.; Panagou, E.Z.; Nychas, G.J.E. Multispectral image analysis approach to detect adulteration of beef and pork in raw meats. Food Res. Int. 2015, 67, 12–18. [Google Scholar] [CrossRef]
- Schulz, H.; Özkan, G.; Baranska, M.; Krüger, H.; Özcan, M. Characterisation of essential oil plants from Turkey by IR and Raman spectroscopy. Vib. Spectrosc. 2005, 39, 249–256. [Google Scholar] [CrossRef]
- Liu, W.; Zhang, B.; Xin, Z.; Ren, D.; Yi, L. GC-MS fingerprinting combined with chemometric methods reveals key bioactive components in Acori tatarinowii rhizoma. Int. J. Mol. Sci. 2017, 18. [Google Scholar] [CrossRef]
- Wu, D.; Nie, P.; Cuello, J.; He, Y.; Wang, Z.; Wu, H. Application of visible and near infrared spectroscopy for rapid and non-invasive quantification of common adulterants in Spirulina powder. J. Food Eng. 2011, 102, 278–286. [Google Scholar] [CrossRef]
- Das, R.S.; Agrawal, Y.K. Raman spectroscopy: Recent advancements, techniques and applications. Vib. Spectrosc. 2011, 57, 163–176. [Google Scholar] [CrossRef]
- Rodriguez-Saona, L.E.; Pujolras, M.P.; Giusti, M.M. Targeted and Non-Targeted Analysis. In Analytical Separation Science; Wiley-VCH: Weinheim, Germany, 2015; pp. 1401–1436. [Google Scholar]
- Agarwal, A.; Shah, D.; Shen, D.; Song, D. On robustness of principal component regression. arXiv 2019, arXiv:1902.10920. [Google Scholar]
- Benson, I.B. Food Authenticity and Traceability; Woodhead Publishing Limited: Cambridge, UK, 2003; ISBN 9781855735262. [Google Scholar]
- Chen, G.; Kocaoglu-Vurma, N.A.; Harper, W.J.; Rodriguez-Saona, L.E. Application of infrared microspectroscopy and multivariate analysis for monitoring the effect of adjunct cultures during Swiss cheese ripening. J. Dairy Sci. 2009, 92, 3575–3584. [Google Scholar] [CrossRef] [PubMed]
- Hoon, A. The Evaluation of Fourier Transform Infrared (FT-IR) Spectroscopy and Multivariate Data Analysis Techniques for Quality Control at an Industrial Cellar; Stellenbosch University: Stellenbosch, South Africa, 2015; pp. 25–26. [Google Scholar]
- Schulz, H.; Schrader, B.; Quilitzsch, R.; Steuer, B. Quantitative analysis of various citrus oils by ATR/FT-IR and NIR-FT Raman spectroscopy. Appl. Spectrosc. 2002, 56, 117–124. [Google Scholar] [CrossRef]
- McHale, D.; Sheridan, J.B. Detection of adulteration of cold-pressed lemon oil. Flavour Fragr. J. 1988, 3, 127–133. [Google Scholar] [CrossRef]
- Sivakesava, S.; Irudayaraj, J. Prediction of inverted cane sugar adulteration of honey by Fourier transform infrared spectroscopy. J. Food Sci. 2001, 66, 972–978. [Google Scholar] [CrossRef]
- Jentzsch, P.; Ramos, L.; Ciobotă, V. Handheld Raman Spectroscopy for the Distinction of Essential Oils Used in the Cosmetics Industry. Cosmetics 2015, 2, 162–176. [Google Scholar] [CrossRef] [Green Version]
- Sandasi, M.; Kamatou, G.P.P.; Gavaghan, C.; Baranska, M.; Viljoen, A.M. A quality control method for geranium oil based on vibrational spectroscopy and chemometric data analysis. Vib. Spectrosc. 2011, 57, 242–247. [Google Scholar] [CrossRef]
- Schulz, H.; Quilitzsch, R.; Krüger, H. Rapid evaluation and quantitative analysis of thyme, origano and chamomile essential oils by ATR-IR and NIR spectroscopy. J. Mol. Struct. 2003, 661–662, 299–306. [Google Scholar] [CrossRef]
- Agatonovic-Kustrin, S.; Ristivojevic, P.; Gegechkori, V.; Litvinova, T.M.; Morton, D.W. Essential Oil Quality and Purity Evaluation via FT-IR Spectroscopy and Pattern Recognition Techniques. Appl. Sci. 2020, 10, 7294. [Google Scholar] [CrossRef]

| Samples | Calibration Technique | Spectra | Equation | R2 | LOGPress | SECV | Bias | ||
|---|---|---|---|---|---|---|---|---|---|
| Calibration | Validation | Calibration | Validation | ||||||
| OE1LEO1 (OEO1 adulterated LEO1) | PLSR | Raw | y = 0.9996x + 0.0123 | y = 1.0318x − 0.4627 | R² = 0.9996 | R² = 0.9984 | 2.14 | 4.14 | 1.37 |
| First derivative. | y = 0.9995x + 0.0139 | y = 0.9676x + 0.4399 | R² = 0.9995 | R² = 0.9950 | 2.20 | 4.44 | 1.50 | ||
| Second derivative. | y = 0.9992x + 0.0222 | y = 0.9659x + 0.4985 | R² = 0.9992 | R² = 0.9970 | 2.40 | 4.90 | 1.62 | ||
| PCR | Raw | y = 0.9995x + 0.0129 | y = 1.0161x − 0.2343 | R² = 0.9995 | R² = 0.9988 | 2.13 | 4.09 | 0.75 | |
| First derivative. | y = 0.9988x + 0.0337 | y = 0.9672x + 0.445 | R² = 0.9988 | R² = 0.9950 | 2.19 | 4.42 | 1.52 | ||
| Second derivative. | y = 0.9995x + 0.0143 | y = 0.9761x + 0.1554 | R² = 0.9995 | R² = 0.9985 | 0.62 | 0.72 | 0.38 | ||
| OE2LEO2 (OEO2 adulterated LEO2) | PLSR | Raw | y = 0.9995x + 0.0152 | y = 1.0049x + 0.0012 | R² = 0.9995 | R² = 0.9938 | 1.58 | 2.18 | 1.80 |
| First derivative. | y = 0.9995x + 0.0149 | y = 0.9968x + 0.0991 | R² = 0.9995 | R² = 0.9977 | 1.60 | 2.22 | 1.17 | ||
| Second derivative. | y = 0.9985x + 0.0434 | y = 0.9953x + 0.13 | R² = 0.9985 | R² = 0.9971 | 1.83 | 2.92 | 1.32 | ||
| PCR | Raw | y = 0.9993x + 0.0194 | y = 1.0049x + 0.0005 | R² = 0.9993 | R² = 0.9938 | 1.47 | 1.92 | 1.80 | |
| First derivative. | y = 0.9986x + 0.0399 | y = 0.9968x + 0.0972 | R² = 0.9986 | R² = 0.9977 | 1.54 | 2.09 | 1.17 | ||
| Second derivative. | y = 0.9983x + 0.0492 | y = 0.9953x + 0.1275 | R² = 0.9983 | R² = 0.9971 | 1.61 | 2.26 | 1.32 | ||
| OEO3LEO3 (OEO3 adulterated LEO3) | PLSR | Raw | y = 0.9995x + 0.013 | y = 0.9942x + 0.1032 | R² = 0.9995 | R² = 0.9969 | 1.92 | 3.21 | 0.81 |
| First derivative. | y = 0.9996x + 0.0123 | y = 0.9758x + 0.3435 | R² = 0.9996 | R² = 0.9964 | 1.82 | 2.87 | 1.13 | ||
| Second derivative. | y = 0.9993x + 0.0193 | y = 0.9869x + 0.1531 | R² = 0.9993 | R² = 0.998 | 1.37 | 1.70 | 0.74 | ||
| PCR | Raw | y = 0.9979x + 0.0601 | y = 0.9942x + 0.1031 | R² = 0.9979 | R² = 0.9969 | 1.97 | 3.40 | 0.81 | |
| First derivative. | y = 0.998x + 0.0564 | y = 0.9757x + 0.344 | R² = 0.998 | R² = 0.9964 | 1.54 | 2.08 | 1.14 | ||
| Second derivative. | y = 0.9979x + 0.0591 | y = 0.9669x + 0.4802 | R² = 0.9979 | R² = 0.9949 | 1.67 | 2.43 | 1.53 | ||
| BA1LEO1 (Benzylalcohol adulterated LEO1) | PLSR | Raw | y = 0.9999x + 0.0024 | y = 0.9981x + 0.0139 | R² = 0.9999 | R² = 0.9999 | 1.24 | 0.92 | 0.27 |
| First derivative. | y = 0.9999x + 0.0035 | y = 0.9464x + 0.9899 | R² = 0.9999 | R² = 0.9985 | 1.56 | 2.14 | 1.90 | ||
| Second derivative. | y = 0.9998x + 0.0044 | y = 0.9192x + 1.1921 | R² = 0.9998 | R² = 0.9953 | 1.84 | 2.94 | 3.15 | ||
| PCR | Raw | y = 0.9999x + 0.0024 | y = 0.9965x + 0.0367 | R² = 0.9999 | R² = 0.9999 | 0.43 | 0.58 | 0.27 | |
| First derivative. | y = 0.9998x + 0.0043 | y = 0.9396x + 1.0674 | R² = 0.9998 | R² = 0.9981 | 1.67 | 2.42 | 2.17 | ||
| Second derivative. | y = 0.9998x + 0.0056 | y = 0.9228x + 1.2914 | R² = 0.9998 | R² = 0.9968 | 1.89 | 3.12 | 2.84 | ||
| BA1LEO2 (Benzylalcohol adulterated LEO) | PLSR | Raw | y = 0.9999x + 0.0028 | y = 1.0079x − 0.1288 | R² = 0.9999 | R² = 0.9998 | 1.12 | 1.28 | 0.33 |
| First derivative. | y = 0.9999x + 0.0032 | y = 0.9646x + 0.6987 | R² = 0.9999 | R² = 0.9993 | 1.22 | 1.44 | 1.52 | ||
| Second derivative. | y = 0.9999x + 0.0036 | y = 0.9486x + 0.9728 | R² = 0.9999 | R² = 0.9986 | 1.53 | 2.05 | 1.80 | ||
| PCR | Raw | y = 0.9999x + 0.0028 | y = 1.0058x − 0.0992 | R² = 0.9999 | R² = 0.9998 | 0.95 | 1.06 | 0.31 | |
| First derivative. | y = 0.9999x + 0.0037 | y = 0.9578x + 0.806 | R² = 0.9999 | R² = 0.9990 | 1.36 | 1.70 | 1.47 | ||
| Second derivative. | y = 0.9998x + 0.0043 | y = 0.9442x + 0.9984 | R² = 0.9998 | R² = 0.9984 | 1.60 | 2.24 | 2.00 | ||
| BA1LEO3 (Benzylalcohol adulterated LEO3) | PLSR | Raw | y = 0.9999x + 0.0030 | y = 1.0054x − 0.085 | R² = 0.9999 | R² = 0.9998 | 1.13 | 1.30 | 0.29 |
| First derivative. | y = 0.9999x + 0.0028 | y = 0.9671x + 0.6537 | R² = 0.9999 | R² = 0.9994 | 1.15 | 1.33 | 1.12 | ||
| Second derivative. | y = 0.9999x + 0.0025 | y = 0.9394x + 0.8944 | R² = 0.9999 | R² = 0.9974 | 1.53 | 2.05 | 2.35 | ||
| PCR | Raw | y = 0.9999x + 0.0023 | y = 1.0027x − 0.0473 | R² = 0.9999 | R² = 0.9998 | 1.01 | 1.13 | 0.29 | |
| First derivative. | y = 0.9999x + 0.0031 | y = 0.9621x + 0.7607 | R² = 0.9999 | R² = 0.9992 | 1.27 | 1.52 | 1.29 | ||
| Second derivative. | y = 0.9999x + 0.0028 | y = 0.9467x + 0.963 | R² = 0.9999 | R² = 0.9987 | 1.55 | 2.11 | 1.89 | ||
| IPM1LEO1 (isopropylmyristate adulterated LEO1) | PLSR | Raw | y = 0.9997x + 0.009 | y = 0.9292x + 0.9864 | R² = 0.9997 | R² = 0.9957 | 1.98 | 3.47 | 2.87 |
| First derivative. | y = 0.9999x + 0.0034 | y = 0.9055x + 1.4095 | R² = 0.9999 | R² = 0.9923 | 2.08 | 3.90 | 3.78 | ||
| Second derivative. | y = 0.9996x + 0.0108 | y = 0.9524x + 0.8724 | R² = 0.9996 | R² = 0.9994 | 2.00 | 3.54 | 2.35 | ||
| PCR | Raw | y = 0.9997x + 0.009 | y = 0.9307x + 0.9623 | R² = 0.9997 | R² = 0.9959 | 1.99 | 3.49 | 2.82 | |
| First derivative. | y = 0.9994x + 0.0162 | y = 0.9052x + 1.4117 | R² = 0.9994 | R² = 0.9923 | 2.11 | 4.01 | 3.79 | ||
| Second derivative. | y = 0.999x + 0.0281 | y = 0.8821x + 1.7718 | R² = 0.999 | R² = 0.9971 | 2.33 | 5.21 | 4.72 | ||
| IPM1LEO2 (isopropylmyristate adulterated LEO2) | PLSR | Raw | y = 0.9997x + 0.0086 | y = 0.9328x + 0.9419 | R² = 0.9997 | R² = 0.9961 | 1.92 | 3.21 | 2.73 |
| First derivative. | y = 0.9999x + 0.0031 | y = 0.9132x + 1.3 | R² = 0.9999 | R² = 0.9936 | 2.02 | 3.62 | 3.47 | ||
| Second derivative. | y = 1x + 0.0008 | y = 0.8924x + 1.6224 | R² = 1 | R² = 0.9994 | 2.23 | 4.63 | 4.31 | ||
| PCR | Raw | y = 0.9997x + 0.0086 | y = 0.9339x + 0.9249 | R² = 0.9997 | R² = 0.9962 | 1.92 | 3.23 | 2.70 | |
| First derivative. | y = 0.9995x + 0.0142 | y = 0.913x + 1.3013 | R² = 0.9995 | R² = 0.9935 | 2.04 | 3.73 | 3.48 | ||
| Second derivative. | y = 0.9992x + 0.0238 | y = 0.8923x + 1.6221 | R² = 0.9992 | R² = 0.9994 | 2.27 | 4.80 | 4.31 | ||
| IPM1LEO3 (isopropylmyristate adulterated LEO3) | PLSR | Raw | y = 0.9995x + 0.0148 | y = 0.9171x + 1.1763 | R² = 0.9995 | R² = 0.9937 | 2.15 | 4.20 | 3.73 |
| First derivative. | y = 0.9997x + 0.0075 | y = 0.8987x + 1.5123 | R² = 0.9906 | R² = 0.9997 | 2.15 | 4.21 | 4.06 | ||
| Second derivative. | y = 0.9998x + 0.0049 | y = 0.8795x + 1.8146 | R² = 0.9998 | R² = 0.9960 | 2.33 | 5.15 | 4.84 | ||
| PCR | Raw | y = 0.9995x + 0.0149 | y = 0.918x + 1.1602 | R² = 0.9938 | R² = 0.9995 | 2.09 | 3.92 | 3.34 | |
| First derivative. | y = 0.9992x + 0.0215 | y = 0.8984x + 1.5149 | R² = 0.9992 | R² = 0.9906 | 2.16 | 4.28 | 4.08 | ||
| Second derivative. | y = 0.9993x + 0.0194 | y = 0.8794x + 1.8155 | R² = 0.9993 | R² = 0.9959 | 2.33 | 5.21 | 4.85 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cebi, N.; Taylan, O.; Abusurrah, M.; Sagdic, O. Detection of Orange Essential Oil, Isopropyl Myristate, and Benzyl Alcohol in Lemon Essential Oil by FTIR Spectroscopy Combined with Chemometrics. Foods 2021, 10, 27. https://doi.org/10.3390/foods10010027
Cebi N, Taylan O, Abusurrah M, Sagdic O. Detection of Orange Essential Oil, Isopropyl Myristate, and Benzyl Alcohol in Lemon Essential Oil by FTIR Spectroscopy Combined with Chemometrics. Foods. 2021; 10(1):27. https://doi.org/10.3390/foods10010027
Chicago/Turabian StyleCebi, Nur, Osman Taylan, Mona Abusurrah, and Osman Sagdic. 2021. "Detection of Orange Essential Oil, Isopropyl Myristate, and Benzyl Alcohol in Lemon Essential Oil by FTIR Spectroscopy Combined with Chemometrics" Foods 10, no. 1: 27. https://doi.org/10.3390/foods10010027