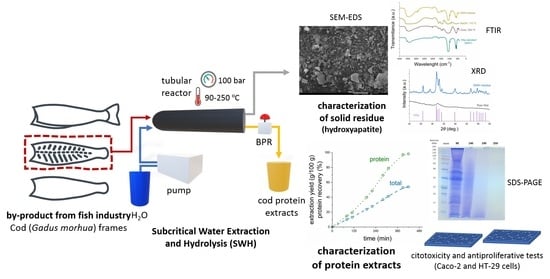
1. Introduction
Atlantic codfish (
Gadus morhua) is among the most important commercial fishes worldwide and the most consumed fish in Portugal. According to recent statistics, Portugal consumes around 70,000 tons per year [
1]. This produces large quantities of by-products, mostly skin and bones, which might cause environmental problems when disposed of and should be adequately valorized.
Codfish frames (backbone with residual meat) are regarded as rich in minerals, such as calcium and phosphorus, and collagen proteins, although some carbohydrate and lipids are also found [
2]. The bone tissue is mainly built up of an organic extra cellular matrix covered with hydroxyapatite (HAp), a calcium phosphate with chemical formula Ca
10(PO
4)
6(OH)
2. Both collagen and HAp can be used in a broad range of economically interesting applications as food ingredients, nutraceutics, or cosmetics [
3]. Hence, there is a great potential for the conversion of cod frames into different bioproducts with high market value.
Collagen has a wide range of applications as emulsifier, foaming and gelling agent in the food, pharmaceutical, cosmetic, and photographic industries, among others. The most common sources are mainly from porcine or bovine origin. However, fish collagen is less restricted due to religious concerns and transmissible diseases, such as bovine spongiform encephalopathy (BSE) and food and mouth disease (FMD) [
4]; thus, it has gained interest in the last years. Fish collagen can be also hydrolyzed to increase its digestibility in nutraceutical preparations and obtain active peptides with biologically active properties (antioxidative, antihypertensive and antimicrobial properties) [
5]. Conventional methods for collagen extraction and hydrolysis use thermal treatments and strong acids or alkalis to increase collagen solubility, followed by salt precipitation to allow isolation. Loss of functional and nutritional properties due to the degradation of active molecules with the extreme temperatures and pHs involved, restrains the field of application of these hydrolysates to animal feed and fertilizers, where quality standards are lower [
6]. Diluted organic acids such as acetic or citric acid have emerged as alternative extraction solvents, at the cost of increased processing times. Different authors report complex extraction methods consisting of pretreatment, extraction, and purification steps during several days to obtain relatively low yields [
7], which is not affordable at industrial scale. Simultaneous or subsequent hydrolysis with proteases has been also introduced to obtain collagen fragments with increased solubility and potential biological properties. Jafarpour et al. [
8] have recently investigated the valorization of cod frames by means of enzymatic extraction and hydrolysis. Other authors have proposed the intensification of the process through extrusion methods [
7], ultrasound assisted extraction (UAE) [
9], or the application of pulsed electric fields [
10].
Calcium phosphates such as hydroxyapatite (HAp) present very high value due to their properties and technological applications, being well known for their use as bioceramics [
11]. HAp is probably the most important calcium phosphate due to its high biocompatibility, and for this reason it is widely used in the medical field for hard tissue engineering [
12]. Other applications of HAp include soil remediation [
13], support for nanometallic catalysts [
14], and development of fire-proof materials [
15]. Most of the HAp and other calcium phosphates are currently produced by chemical synthesis, using different methods [
11]. However, natural by-products and biowastes have been also investigated as a source for these materials, with economic and environmental advantages [
11,
16]. Bovine bones are extensively used to obtain HAp bioceramics [
16], although other biowastes such as pig teeth and bones [
17,
18], chicken bones [
19], eggshells [
20], fish bones [
21,
22], and seafood shells [
23] have been investigated as potential HAp sources. Different methods for HAp production from biowastes have been explored, such as hydrothermal conversion of calcified materials with phosphorus precursors [
16], enzymatic hydrolysis [
24], extraction with ionic liquids [
25], and the most common thermal treatment at temperatures of 600–1200 °C. The thermal decomposition method is simpler and cost-effective and has reached commercial status [
11]. However, collagen proteins and other valuable compounds that might be present in the raw material are destroyed. Hence, previous extraction steps are necessary for an integral valorization of bone residues.
Subcritical water hydrolysis (SBW) represents a green alternative to both protein extraction and hydrolysis processes and HAp production through conventional chemical synthesis or thermal decomposition. SBW uses water at 100–274 °C and pressure above saturation value but less than critical value. At these conditions, the dielectric constant of water decreases with increasing temperature due to hydrogen bond dissociation, allowing water to act as an effective solvent for moderately polar to non-polar substances. Additionally, the ionic product of water (K
W) increases with temperature, being three orders of magnitude higher than that at ambient conditions. This leads to the increase of hydronium and hydroxide ions concentrations and allows water to act as an acid or base catalyst, facilitating the hydrolysis of lignocellulosic polymers and proteins to smaller oligomers and peptides, without the use of additional catalysts [
26,
27].
SBW has been widely investigated as a green method for biomass conversion, hemicellulose fractionation and extraction of phenolics [
28]. However, SBW can be also applied to proteinaceous materials such as fish and fish wastes, obtaining amino acids and small peptides with interesting physicochemical and bioactive properties. Yoshida et al. [
29] reported the SBW of fish meat from horse mackerel in a batch reactor, sequentially producing amino acids, organic acids, and fish oil when increasing temperature from 200 to 400 °C. Other authors have also investigated the SBW of fish waste from different origins, such as white croaker entrails [
30,
31], bonito [
31], squid viscera [
32,
33], or mackerel meat [
34], bones and skin [
35,
36], among others. Ueno et al. [
37] investigated the production of antihypertensive peptides through continuous SBW at 160–200 °C and 20 bar, using cold-water fish gelatin as a fish waste model. Cho et al. [
38] studied the production of shrimp (
Penaeus japonicus) hydrolysates through a batch SBW process at 100–200 °C and evaluated their physicochemical and biofunctional properties, finding strong free-radical scavenging and antioxidant activities that increased with temperature up to 200 °C. Ahmed and Chun [
39] compared the hydrolysates obtained from tuna skin and from previously isolated tuna skin collagen through SBW process at 150–300 °C and 50–100 bar. Maximum antioxidant and antimicrobial activities were found at 280 °C and 80 bar for both materials, although hydrolysates from previously isolated collagen showed better results. Asaduzzaman et al. [
35] reported the production of bioactive low-molecular-weight peptides from mackerel (
Scomber japonicus) bone and skin, through a combination of pepsin assisted extraction of collagen and subsequent SBW of the extracts. SBW process was able to increase the antioxidant activity of the extracts, compared to the initially isolated collagen. In a previous work [
40], we have recently applied a sequential supercritical fluid extraction and SBW process to the fractionation of sardine (
Sardina pilchardus) waste into a fish oil rich in omega-3 polyunsaturated fatty acids, and a fish protein hydrolysate fraction rich in peptides and amino acids. Bioactive properties of the hydrolysates were affected by the temperature. The highest antioxidant activity and in vitro antiproliferative effect were found in the extracts obtained at the highest temperature studied, of 250 °C. In all these works, the molecular profile of the hydrolysates showed that low-molecular-weight peptides are associated with the biofunctional activity of the extracts.
Other authors have applied the SBW process to completely remove protein and organic compounds from bone materials, focusing on the production of HAp from the deproteinized residue. Barakat et al. [
41] obtained pure HAp nanoflakes from bovine bones by SBW treatment at 275 °C and holding time 1 h, although less crystallinity was obtained with SBW method compared to conventional direct heating treatment at 750–1200 °C. SBW was also used for the deproteinization of shrimp cephalotorax wastes, obtaining highly pure calcareous chitin and promoting the formation of HAp nanocrystals with needle and flake shapes [
42].
In this work, the extraction and hydrolysis of protein from cod frames and the simultaneous isolation of HAp using subcritical water have been investigated. To date, SBW process has been applied to the extraction and hydrolysis of collagen from different fish and fish wastes [
29,
30,
31,
32,
33,
34,
35,
36,
37,
38,
39,
40]. However, the production of HAp from these sources is scarcely reported and no mention has been found in the literature about the simultaneous collagen and HAp isolation from bone materials. Thus, the aim of this study was to isolate the main components of the cod frames and characterize their potential application as functional ingredients, to promote a more effective use of the waste generated during codfish processing.
2. Materials and Methods
2.1. Materials
Atlantic codfish (Gadus morhua) frames resulting from the industrial fish filleting process and consisting of the fish backbone and adhered muscle were provided by Pascoal and Filhos S.A. (Gafanha da Nazaré, Portugal). Each batch of fish waste was received fresh, immediately lyophilized, and kept at −20 °C. Prior to experiments, samples were grinded (IKA Tube-Mill Control, IKA®-Werke GmbH & Co. KG, Staufen, Germany) and manually crushed to a fine powder (ca. 5 mm). During this process, bones from different fish and from different parts of the same fish were mixed, to ensure that the samples were as homogenous as possible.
Lowry reagent, 2N Folin-Ciocalteu’s reagent, Bovine Serum Albumin (BSA) standard, phenol (99%), d(+)-glucose monohydrate, Bradford reagent and Lipopolysacccharides from Escherichia coli O55:B5 (LPS) were obtained from Sigma-Aldrich (Saint Louis, MO, USA). Human colorectal adenocarcinoma cell lines, Caco-2 and HT-29 were obtained from Deutsche Sammlung von Mikroorganismen und Zellkulturen (Braunschweig, Germany) and American Type Culture Collection (Manassas, VA, USA), respectively. Cell culture media (Dulbecco’s Modified Eagle Medium, DMEM, and Roswell Park Memorial Institute, RPMI) and supplements (heat inactivated Fetal Bovine Serum, FBS, and non-essential amino acids, NEAA) were purchased from Invitrogen (Gibco, Invitrogen Corporation, Paisley, UK). CellTiter 96 Aqueous One Solution Cell Proliferation Assay (MTS) was obtained from Promega (Madison, WI, USA). Human IL-8 (CXCL8) Mini TMB ELISA Development Kit and TNF-α human recombinant were obtained from Peprotech (London, UK). Human IL-1β was obtained from Sino Biological (Düsseldorf, Germany). All other reagents and solvents used in the present study were of analytical grade and purchased from available suppliers.
2.2. Subcritical Water Extraction and Hydrolysis (SBW)
The subcritical water hydrolysis (SBW) of codfish frames was performed in a lab-scale apparatus described in previous publications [
27]. Briefly, it consists of a distilled water reservoir, a Knauer 1800 high-pressure pump (Knauer Wissenschaftliche Geräte GmbH, Berlin, Germany), and a high-pressure reactor (High Pressure Equipment Co., Erie, PA, USA) with 510 mm length, 50 mm O.D., and 26 mm I.D., placed in a Nabertherm TR 240 electrical oven (Nabertherm GmbH, Lilienthal, Germany). Pressure of the system was controlled by a Tescom back-pressure regulator (BPR). Water mass flow and total mass of water fed to the system was recorded by means of a Rheonik RHM 007 mass flow meter (Rheonik Messtechnik GmbH, Odelzhausen, Germany).
In a typical experiment, 60 g of grinded codfish frames were loaded into the reactor, which was subsequently placed in the electrical oven and connected to the high-pressure circuit. Then, the pump was turned on at a flow rate of 10 mL/min and the BPR was set at 100 bar to pressurize the system. When pressure reached 100 bar, the BPR opened and extract collection started, initiating the experiment. The preheating wires and the electrical oven were turned on at that point, and outlet temperature was increased from ambient conditions to 250 °C. Samples were continuously collected in different flasks at specific target temperatures (90, 140, 190 and 250 °C), which were maintained constant for 30 min. For each sample, 25 mL were taken in triplicate, lyophilized, and weighed to calculate the extraction yield. The rest were frozen and kept at −18 °C for further analysis. The residue that remained in the reactor after SBW experiments was washed with water, dried at 105 °C overnight, weighed and stored at 4 °C for further analysis.
2.3. Proximate Composition
Moisture was determined in a Kern DAB 100-3 thermogravimetric balance (Kern & Sohn GmbH, Balingen, Germany) at 105 °C. Ash content was gravimetrically analyzed through calcination in a muffle at 550 °C during 4 h.
The protein content was determined by elemental analysis of nitrogen in a Flash EA 1112 CHNS analyzer (Thermo Fisher Scientific, Marietta, OH, USA), applying an appropriated nitrogen-to-protein conversion factor (NPCF).
A modified Lowry method [
40] was also used to provide a second measurement of the protein content of codfish frames and protein extracts. Protein extraction from codfish frames was performed by alkaline hydrolysis with 4.2 M NaOH at 95 °C during 1 h (S:L = 1:20). Lyophilized protein extracts were redissolved in distilled water at a known concentration. Absorbance at 750 nm was read in a Genesys 50 spectrophotometer (Thermo Fisher Scientific, Marietta, OH, USA) and protein concentration in the samples was calculated based on a calibration curve made with BSA standard at concentrations ranging from 40 to 400 μg/mL. Blank runs were also performed with distilled water instead of the protein sample.
Fat content was measured through the Bligh and Dyer [
43] method. Soxhlet extraction with
n-hexane was also performed in order to estimate the amount of non-polar lipids, following the method described by Pedras et al. [
27]. Total sugars were determined by phenol-sulphuric acid colorimetric method, which is described elsewhere [
27].
2.4. Amino Acid Profile
The amino acid profile of codfish frames and protein extracts obtained after SBW process was analyzed using a Dionex ICS-3000 high-performance liquid chromatography–ion chromatography (HPLC-IC) system (Thermo Fisher Scientific, Marietta, OH, USA). The chromatographic separation was carried out at 30 °C in a Dionex AminoPac PA10 IC column (250 × 4 mm), with a 50 × 4 mm pre-column. Detection was performed in an electrochemical (ED) detector. Solvent flow was 0.8 mL/min and gradient elution was achieved with solvents (S) S1: water, S2: 250 mM NaOH, S3: 1 M sodium acetate, S4: 0.1 M acetic acid. The course of the gradient was as follows: elution time (t) = 0: 76% S1, 24% S2; t = 8 min: 64% S1, 36% S2; t = 18 min: 40% S1, 20% S2, 40% S3; t = 23 min: 14% S1, 16% S2, 70% S3; t = 45 min: 100% S4; t = 47.1 min: 20% S1, 80% S2; t = 49.3 min: 76% S1, 24% S2 (maintained for 15 min). Sample preparation consisted of an alkaline digestion with 4.2 M NaOH for 24 h at 110 °C (S:L = 1:20). After proper neutralization and dilution, 10 µL were injected and HPLC-IC analysis was performed in triplicate. Individual amino acids were identified and quantified using calibration curves constructed with the corresponding standard compounds and using norleucine as internal standard. All compounds were obtained from Sigma-Aldrich (Saint Louis, MO, USA).
Free amino acids present in the SBW extracts were determined following the same analytical procedure but omitting the alkaline digestion step. Amino acid profile of codfish frames was used to calculate a corrected nitrogen-to-protein factor, following the NREL protocols [
44]. Additionally, the weight percentage of collagen in the codfish frames and the SBW extracts was estimated from the hydroxyproline content as in Equation (1) [
45]:
where % Hyp is the weight percentage of hydroxyproline in each sample. This value was used to monitor the collagen purity of the extracts.
2.5. Mineral Profile
To evaluate the mineral composition and the potential presence of heavy metals in the codfish frames, the obtained protein extracts, and the isolated HAp, inductively coupled plasma–atomic emission spectrometry (ICP-AES) analysis was performed using a Horiba Jobin-Yvon Ultima ICP (Horiba Ltd., Kyoto, Japan), equipped with a 40.68 MHz RF generator and a Czerny-Turner monochromator. Argon was used to generate the plasma. To prepare the samples, the mineralized residue from ash determination was dissolved in 6 M nitric acid at a known concentration and subsequently diluted with distilled water.
2.6. SDS-PAGE
Sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) of the protein extracts obtained after SBW process of codfish frames was conducted by the method of Laemmli [
46], with some modifications, to separate the proteins by their molecular weight. The hydrolysates, 100 μg protein of each sample, were mixed with the loading buffer (50 mM Tris-HCl, pH 6.8, 2% (
w/
v) SDS (
w/
v), 10% glycerol (
v/
v), and 0.01% (
w/
v) bromophenol blue), and then loaded onto two different polyacrylamide gels: (i) a 7.5% precast gel, and (ii) a 15% resolving gel and 4.5% stacking gel. The electrophoresis was conducted at a constant voltage of 120 V. After electrophoresis, the gel was stained with 0.25% (
w/
v) Coomassie Blue R-250 in 8% (
v/
v) acetic acid and 46% (
v/
v) ethanol for 30 min and subsequently destained with a solution containing 10% (
v/
v) acetic acid and 30% (
v/
v) ethanol. A molecular-weight protein marker (NZYColour Protein Marker II) (NZYTech, Lda.—Genes and Enzymes, Lisbon, Portugal) was used to estimate the mass of the protein bands.
2.7. Size Exclusion–Gel Permeation Chromatography (SEC-GPC)
The extracts obtained after SBW treatment of codfish frames were filtrated through nylon syringe filters (0.22 µm pore diameter, ALWSCI Corporation, Zhejiang, China) and submitted to size exclusion–gel permeation chromatography (SEC-GPC) to estimate their molecular mass distribution. The equipment used was a Knauer Smartline HPLC system. For each sample, 20 µL were injected into a Phenomenex Yarra SEC-2000 column (300 × 7.8 mm i.d.) maintained at a constant temperature of 25 °C, and eluted with 0.1 M phosphate buffer (pH 6.8) including 0.025% sodium azide at a flow rate of 1.0 mL/min. The relative intensity of the eluate was monitored with a UV/Vis detector at 280 nm. The retention times of molecular mass standards (Protein Standard Mix 69385 from Sigma-Aldrich (Saint Louis, MO, USA)) were used to calculate the molecular mass distribution of the SBW extracts according to the retention time of the different peaks present in the chromatogram.
2.8. Cell Viability
To evaluate the potential of codfish SBW extracts for nutraceutical applications, cell viability assays were performed with the SBW extracts obtained at different temperatures. Human colorectal adenocarcinoma cell lines, Caco-2, were cultured following previously described methods [
47]. Firstly, cytotoxicity effects were assessed through MTS assay using confluent Caco-2 cells as a model of the human intestinal epithelium. Briefly, cells were seeded in 96-well plates at a density of 2 × 10
4 cells/well and allowed to reach confluence for 7 days. Subsequently, the cells were incubated for 24 h with the extracts diluted in culture medium (DMEM + 0.5% FBS + 1% NEAA). The range of concentrations tested was 0.39–50 mg/mL. Cells incubated with only culture medium were considered as control. After 24 h of treatment, the medium was removed and cell viability was measured using MTS reagent, according to a previously described method [
47]. Three independent experiments were performed in a microplate spectrophotometer at 490 nm (EPOCH 2, BioTek Instruments, Winooski, VT, USA). Cell viability was expressed in terms of percentage of viable cells relative to the control and half maximal effective concentration (EC
50) values were calculated from dose–response curves, as the concentration of sample necessary to decrease cell proliferation by 50%.
2.9. Anti-Inflammatory Potential of Hydrolysates
Anti-inflammatory properties of codfish SBW extracts were tested in Caco-2 cell line. Cells were plated at a density of 100,000 cells/cm2 and allowed to grow for 21 days, with medium (DMEM supplemented with 1% NEAA, 10% FBS and 1% PS) renewed 3 times a week. On the 21st day cells were ready to be used as a model of inflammation of the intestinal mucosa. To mimic the inflammation, cells were stimulated for 48 h with a cocktail of cytokines (25 ng/mL IL-1β, 50 ng/mL TNF-α and 10 ng/mL LPS) and extracts diluted in medium (DMEM supplemented with 1% NEAA and 0.5% FBS). The extract concentration tested was 25 mg/mL since it did not show any cytotoxic effect. Cells incubated only with medium were considered as negative control and cells incubated with medium and the cocktail of cytokines were considered positive control. After 48 h of treatment the medium was collected and centrifuged at 2000× g for 10 min. The supernatant was recovered and stored at −80 °C until the ELISA.
2.10. Quantification of IL-8
According to the cocktail of cytokines used, ELISA was performed to detect the presence of IL-8. The assessment of the IL-8 levels in the supernatant were performed using the human IL-8 (CXCL8) Mini TMB ELISA Development Kit (Peprotech, London, UK), according to the protocol provided by the manufacturer. Briefly, one day prior to running the assay, 96-well plates were coated with the capture antibody. Following overnight incubation at room temperature, the plates were washed with wash buffer (PBS (Thermo Fisher Scientific, Marietta, OH, USA) containing 0.05% Tween-20 (Sigma-Aldrich, St. Louis, MO, USA) and then incubated for 1 h at room temperature (RT) with block buffet (1% BSA in PBS) to inhibit nonspecific binding. After washing, 100 mL of standard dilution or extracts was added to each well in triplicate and incubated for 2 h at RT. After washing the plates, 100 mL of detection antibody was added to each well. The plates were then incubated for 2 h at RT, and then washed again, followed by the incubation with 100 mL of streptavidin-horseradish peroxidase (HRP) conjugated for 30 min at RT. Next, after additional washing, 3,3’,5,5’-tetramethylbenzidine (TMB) substrate solution was added and the plates were incubated in the dark at RT for the color development (approximately 20 min). The reaction was stopped by the addition of 100 mL of HCL 1M, and the absorbance was measured at 450 nm with wavelength correction set at 620 nm in a microplate spectrophotometer (EPOCH 2, BioTek Instruments).
The results (mean ± SD of at least three experiments) were expressed in percentage normalized to the positive control.
2.11. Hydroxyapatite Isolation and Characterization
The deproteinized residue remaining in the SBW reactor was dried and submitted to different instrumental techniques to determine the presence and purity of HAp. For comparison, the product obtained after direct heating of codfish frames at 550 °C during 5 h, and the residue obtained after alkaline digestion (4.2 M NaOH, 110 °C, 24 h, S:L = 1:20) were analyzed by the same instrumental techniques, Fourier Transform-Infrared (FT-IR) spectroscopy, X-ray diffraction (XRD), and Field Emission Gun Scanning Electron Microscopy (FEG-SEM)as follows.
Spectroscopic characterization of codfish frames, lyophilized protein extracts, and the remaining residue after SBW process was performed by FT-IR spectroscopy. The spectra between 4000 and 400 cm
−1 of all the samples were recorded as KBr discs, using a Perkin-Elmer Spectrum Two FT-IR spectrometer (Waltham, MA, USA). KBr discs were pressed using 5 mg of the sample mixed with 200 mg of desiccated KBr. The FT-IR spectra of Calcium phosphate hydroxide (HAp) from the National Institute of Standards and Technology (NIST) was used as a reference standard [
48]. Data about the phase and crystallinity of the samples from the remaining residue were determined by XRD using a benchtop X-ray Diffractometer Rigaku, model MiniFlex II, with Cu K-α radiation (30 KV/15 mA) (Tokyo, Japan). Measurements were taken at a scan speed of 1°/min and 0.02° steps, between 20 and 55°. The NIST standard for HAp (SRM 2910b [
49]) was used to identify the phases and compare the samples. FEG-SEM was performed in a JEOL 7001F equipment (Jeol Ltd., Tokyo, Japan) with Oxford light elements and Energy-Dispersive Detector (EDS). Samples were gold-sputtered in Ar plasma prior to analysis.
2.12. Statistical Analysis
SBW experiments were duplicated. Analytical data are expressed as mean ± standard deviation (SD) of triplicates. Significant differences were detected through one-way analysis of variance and Fisher’s LSD test, using Statgraphics software (version 18 for Windows).
GraphPad Prism 6 software was used to calculate the EC50 values of cytotoxicity. Results of potential anti-inflammatory analyses are the averages of at least three independent experiments and are reported as mean % positive control ± SD. Differences between the controls and the experimental concentrations were assessed using t-test (p < 0.05) using the GraphPad Prims 6 software.