Nutritional Profiling and Preliminary Bioactivity Screening of Five Micro-Algae Strains Cultivated in Northwest Europe
Abstract
:1. Introduction
2. Materials and Methods
2.1. Algae Biomass
2.2. Biochemical Analysis
2.3. In Vitro Digestibility
2.4. Angiotensin-1-Converting Enzyme (ACE-1; EC 3.4.15.1) Inhibition Assay
2.5. Statistics
3. Results and Discussion
3.1. Sample Pretreatment and Biochemical Composition
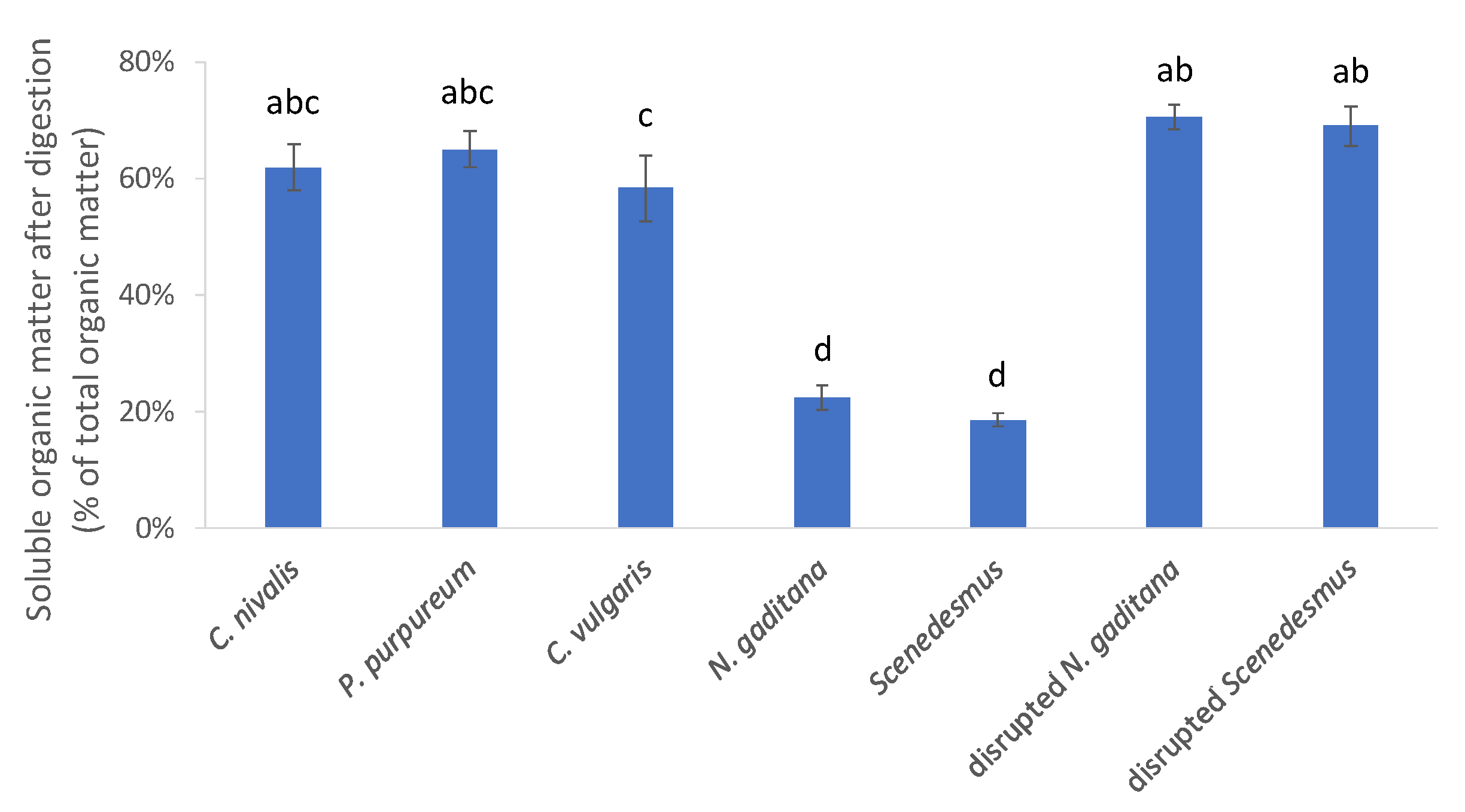
3.2. Carbohydrate Composition and Digestibility
3.3. Lipid Composition and Digestibility
3.4. Protein Composition and Digestibility
3.5. Mineral Content
3.6. Bioactivity Screening-ACE-1 Inhibition
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Allnutt, F.C.T. Promising future products from microalgae for commercial applications. In Sustainable Downstream Processing of Microalgae for Industrial Application; Gayen, K., Bhowmick, T.K., Maity, S.K., Eds.; CRC Press: Boca Raton, FL, USA, 2019; p. 30. ISBN 9780429027970. [Google Scholar]
- Paliwal, C.; Nesamma, A.A.; Jutur, P.P. Industrial scope with high-value biomolecules from microalgae. In Sustainable Downstream Processing of Microalgae for Industrial Application; CRC Press: Boca Raton, FL, USA, 2019; pp. 83–98. [Google Scholar]
- Madeira, M.S.; Cardoso, C.; Lopes, P.A.; Coelho, D.; Afonso, C.; Bandarra, N.M.; Prates, J.A.M. Microalgae as feed ingredients for livestock production and meat quality: A review. Livest. Sci. 2017, 205, 111–121. [Google Scholar] [CrossRef]
- European Food Safety Authority. Scientific opinion on dietary reference values for fats, including saturated fatty acids, polyunsaturated fatty acids, monounsaturated fatty acids, trans fatty acids, and cholesterol. EFSA J. 2010, 8, 1461. [Google Scholar] [CrossRef] [Green Version]
- Tibbetts, S.M.; Yasumaru, F.; Lemos, D. In vitro prediction of digestible protein content of marine microalgae (Nannochloropsis granulata) meals for Pacific white shrimp (Litopenaeus vannamei) and rainbow trout (Oncorhynchus mykiss). Algal Res. 2017, 21, 76–80. [Google Scholar] [CrossRef] [Green Version]
- Wild, K.J.; Steingaß, H.; Rodehutscord, M. Variability in nutrient composition and in vitro crude protein digestibility of 16 microalgae products. J. Anim. Physiol. Anim. Nutr. 2018, 102, 1306–1319. [Google Scholar] [CrossRef] [PubMed]
- Massa, M.; Buono, S.; Langellotti, A.L.; Martello, A.; Russo, G.L.; Troise, D.A.; Sacchi, R.; Vitaglione, P.; Fogliano, V. Biochemical composition and in vitro digestibility of Galdieria sulphuraria grown on spent cherry-brine liquid. New Biotechnol. 2019, 53, 9–15. [Google Scholar] [CrossRef]
- Batista, A.P.; Niccolai, A.; Bursic, I.; Sousa, I.; Raymundo, A.; Rodolfi, L.; Biondi, N.; Tredici, M.R. Microalgae as functional ingredients in savory food products: Application to wheat crackers. Foods 2019, 8, 611. [Google Scholar] [CrossRef] [Green Version]
- Bernaerts, T.M.M.; Verstreken, H.; Dejonghe, C.; Gheysen, L.; Foubert, I.; Grauwet, T.; Van Loey, A.M. Cell disruption of Nannochloropsis sp. improves in vitro bioaccessibility of carotenoids and ω3-LC-PUFA. J. Funct. Foods 2020, 65, 103770. [Google Scholar] [CrossRef]
- Cavonius, L.R.; Albers, E.; Undeland, I. In vitro bioaccessibility of proteins and lipids of pH-shift processed Nannochloropsis oculata microalga. Food Funct. 2016, 7, 2016–2024. [Google Scholar] [CrossRef] [Green Version]
- Bernaerts, T.M.M.; Gheysen, L.; Foubert, I.; Hendrickx, M.E.; Van Loey, A.M. The potential of microalgae and their biopolymers as structuring ingredients in food: A review. Biotechnol. Adv. 2019, 37. [Google Scholar] [CrossRef]
- Niccolai, A.; Chini Zittelli, G.; Rodolfi, L.; Biondi, N.; Tredici, M.R. Microalgae of interest as food source: Biochemical composition and digestibility. Algal Res. 2019, 42, 101617. [Google Scholar] [CrossRef]
- Bleakley, S.; Hayes, M. Functional and Bioactive Properties of Protein Extracts Generated from Spirulina platensis and Isochrysis galbana T-Iso. Appl. Sci. 2021, 11, 3964. [Google Scholar] [CrossRef]
- Ryckebosch, E.; Muylaert, K.; Foubert, I. Optimization of an analytical procedure for extraction of lipids from microalgae. J. Am. Oil Chem. Soc. 2012, 89, 189–198. [Google Scholar] [CrossRef]
- ISO. ISO 12966-2:2017(en) Animal and Vegetable Fats And Oils—Gas Chromatography of Fatty Acid Methyl Esters—Part 2: Preparation of Methyl Esters of Fatty Acids. Available online: https://www.iso.org/obp/ui/#iso:std:iso:12966:-2:ed-2:v1:en (accessed on 1 August 2019).
- Soetemans, L.; Gianotten, N.; Bastiaens, L. Agri-food side-stream inclusion in the diet of alphitobius diaperinus. Part 2: Impact on larvae composition. Insects 2020, 11, 190. [Google Scholar] [CrossRef] [Green Version]
- Verspreet, J.; Soetemans, L.; Bastiaens, L. Searching for appropriate storage conditions for short-term wet preservation of porphyridium purpureum. Appl. Sci. 2020, 10, 8315. [Google Scholar] [CrossRef]
- Naozuka, J.; Mesquita Silva da Veiga, M.A.; Vitoriano Oliveira, P.; de Oliveira, E. Determination of chlorine, bromine and iodine in milk samples by ICP-OES. J. Anal. At. Spectrom. 2003, 18, 917. [Google Scholar] [CrossRef]
- Minekus, M.; Alminger, M.; Alvito, P.; Ballance, S.; Bohn, T.; Bourlieu, C.; Carrière, F.; Boutrou, R.; Corredig, M.; Dupont, D.; et al. A standardised static in vitro digestion method suitable for food-an international consensus. Food Funct. 2014, 5, 1113–1124. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brodkorb, A.; Egger, L.; Alminger, M.; Alvito, P.; Assunção, R.; Ballance, S.; Bohn, T.; Bourlieu-Lacanal, C.; Boutrou, R.; Carrière, F.; et al. INFOGEST static in vitro simulation of gastrointestinal food digestion. Nat. Protoc. 2019, 14, 991–1014. [Google Scholar] [CrossRef]
- Spellman, D.; McEvoy, E.; O’Cuinn, G.; FitzGerald, R.J. Proteinase and exopeptidase hydrolysis of whey protein: Comparison of the TNBS, OPA and pH stat methods for quantification of degree of hydrolysis. Int. Dairy J. 2003, 13, 447–453. [Google Scholar] [CrossRef]
- Leni, G.; Soetemans, L.; Caligiani, A.; Sforza, S.; Bastiaens, L. Degree of hydrolysis affects the techno-functional properties of lesser mealworm protein hydrolysates. Foods 2020, 9, 381. [Google Scholar] [CrossRef] [Green Version]
- Kangani, C.O.; Kelley, D.E.; DeLany, J.P. New method for GC/FID and GC–C-IRMS analysis of plasma free fatty acid concentration and isotopic enrichment. J. Chromatogr. B 2008, 873, 95–101. [Google Scholar] [CrossRef] [Green Version]
- Dave, L.; Hayes, M.; Mora, L.; Montoya, C.; Moughan, P.; Rutherfurd, S. Gastrointestinal endogenous protein-derived bioactive peptides: An in vitro study of their gut modulatory potential. Int. J. Mol. Sci. 2016, 17, 482. [Google Scholar] [CrossRef] [Green Version]
- González-Fernández, C.; Sialve, B.; Bernet, N.; Steyer, J.P. Thermal pretreatment to improve methane production of Scenedesmus biomass. Biomass Bioenergy 2012, 40, 105–111. [Google Scholar] [CrossRef]
- González-Fernández, C.; Sialve, B.; Bernet, N.; Steyer, J.P. Comparison of ultrasound and thermal pretreatment of Scenedesmus biomass on methane production. Bioresour. Technol. 2012, 110, 610–616. [Google Scholar] [CrossRef]
- Reshma, R.; Arumugam, M. Selective degradation of the recalcitrant cell wall of Scenedesmus quadricauda CASA CC202. Planta 2017, 246, 779–790. [Google Scholar] [CrossRef] [PubMed]
- Geresh, S.; Arad, S.M.; Levy-Ontman, O.; Zhang, W.; Tekoah, Y.; Glaser, R. Isolation and characterization of poly- and oligosaccharides from the red microalga Porphyridium sp. Carbohydr. Res. 2009, 344, 343–349. [Google Scholar] [CrossRef] [PubMed]
- Bernaerts, T.M.M.; Gheysen, L.; Kyomugasho, C.; Jamsazzadeh Kermani, Z.; Vandionant, S.; Foubert, I.; Hendrickx, M.E.; Van Loey, A.M. Comparison of microalgal biomasses as functional food ingredients: Focus on the composition of cell wall related polysaccharides. Algal Res. 2018, 32, 150–161. [Google Scholar] [CrossRef]
- Scholz, M.J.; Weiss, T.L.; Jinkerson, R.E.; Jing, J.; Roth, R.; Goodenough, U.; Posewitz, M.C.; Gerken, H.G. Ultrastructure and composition of the Nannochloropsis gaditana cell wall. Eukaryot. Cell 2014, 13, 1450–1464. [Google Scholar] [CrossRef] [Green Version]
- Müller, T.; Bleiß, W.; Martin, C.-D.; Rogaschewski, S.; Fuhr, G. Snow algae from northwest Svalbard: Their identification, distribution, pigment and nutrient content. Polar Biol. 1998, 20, 14–32. [Google Scholar] [CrossRef]
- Hoham, R.W.; Blinn, D.W. Distribution of cryophilic algae in an arid region, the American Southwest. Phycologia 1979, 18, 133–145. [Google Scholar] [CrossRef]
- Koo, K.M.; Jung, S.; Lee, B.S.; Kim, J.B.; Jo, Y.D.; Choi, H.-I.; Kang, S.Y.; Chung, G.H.; Jeong, W.J.; Ahn, J.W. The mechanism of starch over-accumulation in Chlamydomonas reinhardtii high-starch mutants identified by comparative transcriptome analysis. Front. Microbiol. 2017, 8, 858. [Google Scholar] [CrossRef] [PubMed]
- Rengel, R.; Smith, R.T.; Haslam, R.P.; Sayanova, O.; Vila, M.; León, R. Overexpression of acetyl-CoA synthetase (ACS) enhances the biosynthesis of neutral lipids and starch in the green microalga Chlamydomonas reinhardtii. Algal Res. 2018, 31, 183–193. [Google Scholar] [CrossRef] [Green Version]
- Pandey, A.; Gupta, A.; Sunny, A.; Kumar, S.; Srivastava, S. Multi-objective optimization of media components for improved algae biomass, fatty acid and starch biosynthesis from Scenedesmus sp. ASK22 using desirability function approach. Renew. Energy 2020, 150, 476–486. [Google Scholar] [CrossRef]
- Kessler, E. Physiological and biochemical contributions to the taxonomy of the genera Ankistrodesmus and Scenedesmus-V. Starch hydrolysis and new assignment of strains. Arch. Microbiol. 1980, 126, 11–14. [Google Scholar] [CrossRef]
- Kaplan, E.; Sayar, N.A.; Kazan, D.; Sayar, A.A. Assessment of different carbon and salinity level on growth kinetics, lipid, and starch composition of Chlorella vulgaris SAG 211-12. Int. J. Green Energy 2020, 17, 290–300. [Google Scholar] [CrossRef]
- Nordin, N.; Yusof, N.; Maeda, T.; Mustapha, N.A.; Yusoff, M.Z.M.; Khairuddin, R.F.R. Mechanism of carbon partitioning towards starch and triacylglycerol in Chlorella vulgaris under nitrogen stress through whole-transcriptome analysis. Biomass Bioenergy 2020, 138, 105600. [Google Scholar] [CrossRef]
- Fernandes, B.; Dragone, G.; Abreu, A.P.; Geada, P.; Teixeira, J.; Vicente, A. Starch determination in Chlorella vulgaris-a comparison between acid and enzymatic methods. J. Appl. Phycol. 2012, 24, 1203–1208. [Google Scholar] [CrossRef] [Green Version]
- Brányiková, I.; Maršálková, B.; Doucha, J.; Brányik, T.; Bišová, K.; Zachleder, V.; Vítová, M. Microalgae-novel highly efficient starch producers. Biotechnol. Bioeng. 2011, 108, 766–776. [Google Scholar] [CrossRef]
- Teuling, E.; Wierenga, P.A.; Agboola, J.O.; Gruppen, H.; Schrama, J.W. Cell wall disruption increases bioavailability of Nannochloropsis gaditana nutrients for juvenile Nile tilapia (Oreochromis niloticus). Aquaculture 2019, 499, 269–282. [Google Scholar] [CrossRef]
- Vogler, B.W.; Brannum, J.; Chung, J.W.; Seger, M.; Posewitz, M.C. Characterization of the Nannochloropsis gaditana storage carbohydrate: A 1,3-beta glucan with limited 1,6-branching. Algal Res. 2018, 36, 152–158. [Google Scholar] [CrossRef]
- Blazek, J.; Gilbert, E.P. Effect of enzymatic hydrolysis on native starch granule structure. Biomacromolecules 2010, 11, 3275–3289. [Google Scholar] [CrossRef]
- Waterschoot, J.; Gomand, S.V.; Fierens, E.; Delcour, J.A. Starch blends and their physicochemical properties. Starch Staerke 2015, 67, 1–13. [Google Scholar] [CrossRef]
- Yu, S.; Blennow, A.; Bojko, M.; Madsen, F.; Olsen, C.E.; Engelsen, S.B. Physico-chemical characterization of floridean starch of red algae. Starch Staerke 2002, 54, 66–74. [Google Scholar] [CrossRef]
- Gebreyohannes, A.Y.; Dharmjeet, M.; Swusten, T.; Mertens, M.; Verspreet, J.; Verbiest, T.; Courtin, C.M.; Vankelecom, I.F.J. Simultaneous glucose production from cellulose and fouling reduction using a magnetic responsive membrane reactor with superparamagnetic nanoparticles carrying cellulolytic enzymes. Bioresour. Technol. 2018, 263, 532–540. [Google Scholar] [CrossRef] [PubMed]
- Grundy, M.M.L.; Edwards, C.H.; Mackie, A.R.; Gidley, M.J.; Butterworth, P.J.; Ellis, P.R. Re-evaluation of the mechanisms of dietary fibre and implications for macronutrient bioaccessibility, digestion and postprandial metabolism. Br. J. Nutr. 2016, 116, 816–833. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Staudacher, H.M.; Whelan, K. The low FODMAP diet: Recent advances in understanding its mechanisms and efficacy in IBS. Gut 2017, 66, 1517–1527. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Staudacher, H.M.; Irving, P.M.; Lomer, M.C.E.; Whelan, K. Mechanisms and efficacy of dietary FODMAP restriction in IBS. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 256–266. [Google Scholar] [CrossRef]
- Camacho-Rodríguez, J.; Cerón-García, M.C.; Macías-Sánchez, M.D.; Fernández-Sevilla, J.M.; López-Rosales, L.; Molina-Grima, E. Long-term preservation of concentrated Nannochloropsis gaditana cultures for use in aquaculture. J. Appl. Phycol. 2016, 28, 299–312. [Google Scholar] [CrossRef]
- Camacho-Rodríguez, J.; González-Céspedes, A.M.; Cerón-García, M.C.; Fernández-Sevilla, J.M.; Acién-Fernández, F.G.; Molina-Grima, E. A quantitative study of eicosapentaenoic acid (EPA) production by Nannochloropsis gaditana for aquaculture as a function of dilution rate, temperature and average irradiance. Appl. Microbiol. Biotechnol. 2014, 98, 2429–2440. [Google Scholar] [CrossRef]
- Ma, X.N.; Chen, T.P.; Yang, B.; Liu, J.; Chen, F. Lipid production from Nannochloropsis. Mar. Drugs 2016, 14, 61. [Google Scholar] [CrossRef] [Green Version]
- Rebolloso Fuentes, M.M.; Acién Fernández, G.G.; Sánchez Pérez, J.A.; Guil Guerrero, J.L. Biomass nutrient profiles of the microalga Porphyridium cruentum. Food Chem. 2000, 70, 345–353. [Google Scholar] [CrossRef]
- Guihéneuf, F.; Stengel, D.B. Towards the biorefinery concept: Interaction of light, temperature and nitrogen for optimizing the co-production of high-value compounds in Porphyridium purpureum. Algal Res. 2015, 10, 152–163. [Google Scholar] [CrossRef]
- Semih, O.; Ruhsen, P. Fatty acid Composition of Chlorella and Spirulina Microalgae Species. J. AOAC Int. 2001, 84, 1708–1714. [Google Scholar]
- Canelli, G.; Tarnutzer, C.; Carpine, R.; Neutsch, L.; Bolten, C.J.; Dionisi, F.; Mathys, A. Biochemical and nutritional evaluation of Chlorella and Auxenochlorella biomasses relevant for food application. Front. Nutr. 2020, 7, 1–9. [Google Scholar] [CrossRef]
- Lu, N.; Wei, D.; Jiang, X.L.; Chen, F.; Yang, S.T. Fatty acids profiling and biomarker identification in Snow Alga Chlamydomonas Nivalis by NaCl stress using GC/MS and multivariate statistical analysis. Anal. Lett. 2012, 45, 1172–1183. [Google Scholar] [CrossRef]
- Kudahettige, N.P.; Pickova, J.; Gentili, F.G. Stressing Algae for Biofuel Production: Biomass and Biochemical Composition of Scenedesmus dimorphus and Selenastrum minutum Grown in Municipal Untreated Wastewater. Front. Energy Res. 2018, 6, 132. [Google Scholar] [CrossRef]
- Zhang, Y.; Wu, H.; Sun, M.; Peng, Q.; Li, A. Photosynthetic physiological performance and proteomic profiling of the oleaginous algae Scenedesmus acuminatus reveal the mechanism of lipid accumulation under low and high nitrogen supplies. Photosynth. Res. 2018, 138, 73–102. [Google Scholar] [CrossRef] [PubMed]
- Gille, A.; Trautmann, A.; Posten, C.; Briviba, K. Bioaccessibility of carotenoids from Chlorella vulgaris and Chlamydomonas reinhardtii. Int. J. Food Sci. Nutr. 2016, 67, 507–513. [Google Scholar] [CrossRef]
- Coelho, D.; Lopes, P.A.; Cardoso, V.; Ponte, P.; Brás, J.; Madeira, M.S.; Alfaia, C.M.; Bandarra, N.M.; Gerken, H.G.; Fontes, C.M.G.A.; et al. Novel combination of feed enzymes to improve the degradation of Chlorella vulgaris recalcitrant cell wall. Sci. Rep. 2019, 9, 5382. [Google Scholar] [CrossRef] [Green Version]
- Rego, D.; Costa, L.; Pereira, M.T.; Redondo, L.M. Cell Membrane Permeabilization Studies of Chlorella sp. by Pulsed Electric Fields. IEEE Trans. Plasma Sci. 2015, 43, 3483–3488. [Google Scholar] [CrossRef]
- Balduyck, L.; Stock, T.; Bijttebier, S.; Bruneel, C.; Jacobs, G.; Voorspoels, S.; Muylaert, K.; Foubert, I. Integrity of the microalgal cell plays a major role in the lipolytic stability during wet storage. Algal Res. 2017, 25, 516–524. [Google Scholar] [CrossRef]
- Bleakley, S.; Hayes, M. Algal proteins: Extraction, application, and challenges concerning production. Foods 2017, 6, 33. [Google Scholar] [CrossRef] [Green Version]
- Bernaerts, T.M.M.; Kyomugasho, C.; Van Looveren, N.; Gheysen, L.; Foubert, I.; Hendrickx, M.E.; Van Loey, A.M. Molecular and rheological characterization of different cell wall fractions of Porphyridium cruentum. Carbohydr. Polym. 2018, 195, 542–550. [Google Scholar] [CrossRef] [PubMed]
- Canelli, G.; Murciano Martínez, P.; Maude Hauser, B.; Kuster, I.; Rohfritsch, Z.; Dionisi, F.; Bolten, C.J.; Neutsch, L.; Mathys, A. Tailored enzymatic treatment of Chlorella vulgaris cell wall leads to effective disruption while preserving oxidative stability. LWT 2021, 143, 111157. [Google Scholar] [CrossRef]
- Muys, M.; Sui, Y.; Schwaiger, B.; Lesueur, C.; Vandenheuvel, D.; Vermeir, P.; Vlaeminck, S.E. High variability in nutritional value and safety of commercially available Chlorella and Spirulina biomass indicates the need for smart production strategies. Bioresour. Technol. 2019, 275, 247–257. [Google Scholar] [CrossRef]
- Lourenço, S.O.; Barbarino, E.; Lavín, P.L.; Lanfer Marquez, U.M.; Aidar, E. Distribution of intracellular nitrogen in marine microalgae: Calculation of new nitrogen-to-protein conversion factors. Eur. J. Phycol. 2004, 39, 17–32. [Google Scholar] [CrossRef]
- Fowden, L. The composition of the bulk proteins of Chlorella. Biochem. J. 1952, 50, 355–358. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Manor, M.L.; Kim, J.; Derksen, T.J.; Schwartz, R.L.; Roneker, C.A.; Bhatnagar, R.S.; Lei, X.G. Defatted microalgae serve as a dual dietary source of highly bioavailable iron and protein in an anemic pig model. Algal Res. 2017, 26, 409–414. [Google Scholar] [CrossRef]
- Bohn, L.; Meyer, A.S.; Rasmussen, S.K. Phytate: Impact on environment and human nutrition. A challenge for molecular breeding. J. Zhejiang Univ. Sci. B 2008, 9, 165–191. [Google Scholar] [CrossRef] [Green Version]
- Barbera, E.; Teymouri, A.; Bertucco, A.; Stuart, B.J.; Kumar, S. Recycling minerals in microalgae cultivation through a combined flash hydrolysis–precipitation process. ACS Sustain. Chem. Eng. 2017, 5, 929–935. [Google Scholar] [CrossRef]
- Fitzgerald, C.; Mora-Soler, L.; Gallagher, E.; O’Connor, P.; Prieto, J.; Soler-Vila, A.; Hayes, M. Isolation and characterization of bioactive pro-peptides with in vitro renin inhibitory activities from the Macroalga Palmaria palmata. J. Agric. Food Chem. 2012, 60, 7421–7427. [Google Scholar] [CrossRef]
- Fitzgerald, C.; Aluko, R.E.; Hossain, M.; Rai, D.K.; Hayes, M. Potential of a renin inhibitory peptide from the red seaweed palmaria palmata as a functional food ingredient following confirmation and characterization of a hypotensive effect in spontaneously hypertensive rats. J. Agric. Food Chem. 2014, 62, 8352–8356. [Google Scholar] [CrossRef] [PubMed]




| C. nivalis | P. purpureum | C. vulgaris | Disrupted N. gaditana | Disrupted Scenedesmus | |
|---|---|---|---|---|---|
| Carbohydrates (% dm) 1 | 38.4 ± 0.5 | 51.2 ± 1.9 | 16.0 ± 3.2 | 15.8 ± 0.4 | 45.4 ± 6.9 |
| Proteins (% dm) 1 | 33.4 ± 0.9 | 22.2 ± 0.1 | 45.5 ± 1.3 | 46.2 ± 0.3 | 25.7 ± 4.4 |
| Lipids (% dm) | 22.2 ± 0.4 | 13.2 ± 0.9 | 21.8 ± 0.3 | 31.9 ± 0.6 | 17.5 ± 0.5 |
| Ash (% dm) | 3.6 ± 0.1 | 8.4 ± 0.1 | 8.2 ± 0.3 | 6.4 ± 0.0 | 3.0 ± 0.1 |
| Fatty Acid Levels (mg/g Dry Algae) | C. nivalis | P. purpureum | C. vulgaris | Disrupted N. gaditana | Disrupted Scenedesmus |
|---|---|---|---|---|---|
| C12:0 | 0.15 ± 0.01 | 0 | 0.25 ± 0.03 | 0.44 ± 0.04 | 0.20 ± 0.01 |
| C14:0 | 0.27 ± 0.02 | 0.04 ± 0.04 | 0.36 ± 0.01 | 4.05 ± 0.08 | 0.20 ± 0.01 |
| C16:0 | 17.03 ± 0.74 | 14.83 ± 2.18 | 23.89 ± 0.95 | 17.87 ± 0.73 | 14.06 ± 0.41 |
| C16:1 (cis 9) | 0.41 ± 0.35 | 0.04 ± 0.04 | 0 | 31.49 ± 0.97 | 0.24 ± 0.01 |
| C17:0 | 4.75 ± 0.28 | 0 | 2.21 ± 0.11 | 0.23 ± 0.01 | 1.24 ± 0.04 |
| C18:0 | 0.96 ± 0.07 | 0.70 ± 0.12 | 1.70 ± 0.07 | 0.34 ± 0.02 | 0.94 ± 0.03 |
| C18:1 (cis-9) | 20.88 ± 1.08 | 1.82 ± 0.50 | 28.26 ± 1.08 | 1.94 ± 0.12 | 16.68 ± 0.77 |
| C18:1 (cis-11) | 11.15 ± 0.59 | 0.16 ± 0.03 | 1.84 ± 0.06 | 0.27 ± 0.01 | 1.11 ± 0.03 |
| C18:2 (cis,cis-9,12) or LA | 12.57 ± 0.83 | 4.62 ± 0.18 | 15.21 ± 0.78 | 1.37 ± 0.08 | 8.54 ± 0.49 |
| C18:3 (cis 6,9,12) | 0 | 0.14 ± 0.08 | 0.70 ± 0.06 | 0.82 ± 0.07 | 0.31 ± 0.07 |
| C18:3 (cis 9,12,15) or ALA | 26.98 ± 2.07 | 0.57 ± 0.09 | 22.39 ± 1.42 | 0.08 ± 0.03 | 12.06 ± 0.61 |
| C20:0 | 0 | 0 | 0 | 0.11 ± 0.01 | 0.12 ± 0.03 |
| C20:1 (cis11) | 0 | 0 | 0.50 ± 0.15 | 0 | 0.33 ± 0.03 |
| C20:2 (cis 11,14) | 0 | 0.29 ± 0.04 | 0 | 0 | 0 |
| C20:3 (cis8,11,14) | 0 | 0.55 ± 0.08 | 0 | 0.90 ± 0.09 | 0 |
| C20:4 (cis 5,8,11,14) | 0 | 8.24 ± 1.52 | 0 | 3.16 ± 0.27 | 0 |
| C22:1 (cis 13) | 0 | 0 | 0.15 ± 0.04 | 0.44 ± 0.02 | 0.09 ± 0.01 |
| C20:5 (cis 5,8,11,14,17) or EPA | 0 | 3.61 ± 0.76 | 0 | 54.87 ± 4.67 | 0 |
| C22:6 (cis 4,7,10,13,16,19) or DHA | 0 | 0 | 0 | 0.20 ± 0.03 | 0 |
| Sum unidentified peaks | 23.03 ± 1.69 | 1.40 ± 0.54 | 25.50 ± 1.93 | 20.79 ± 1.41 | 16.41 ± 0.94 |
| omega-3 fatty acids | 27.0 ± 2.1 | 4.2 ± 0.8 | 22.4 ± 1.4 | 55.1 ± 4.7 | 12.1 ± 0.6 |
| omega-6 fatty acids | 12.6 ± 0.8 | 13.8 ± 1.5 | 15.9 ± 0.8 | 6.2 ± 0.3 | 8.9 ± 0.5 |
| sum SFA | 23.2 ± 0.8 | 15.6 ± 2.2 | 28.4 ± 1.0 | 23.0 ± 0.7 | 16.8 ± 0.4 |
| sum MUFA | 32.4 ± 1.3 | 2.0 ± 0.5 | 30.8 ± 1.1 | 34.1 ± 1.0 | 18.5 ± 0.8 |
| sum PUFA | 39.6 ± 2.2 | 18.0 ± 1.7 | 38.3 ± 1.6 | 61.4 ± 4.7 | 20.9 ± 0.8 |
| C. nivalis | P. purpureum | C. vulgaris | Disrupted N. gaditana | Disrupted Scenedesmus | |
|---|---|---|---|---|---|
| Sum amino acids (mg/g dm) 1 | 334 ± 9 | 222 ± 1 | 455 ± 13 | 462 ± 3 | 257 ± 44 |
| Sum essential amino acids (mg/g dm) | 100 ± 1 | 71 ± 4 | 146 ± 2 | 150 ± 2 | 77 ± 14 |
| Arginine (mg/g dm) | 94 ± 1 | 61 ± 3 | 128 ± 11 | 138 ± 8 | 69 ± 4 |
| Lysine (mg/g dm) 2 | 17 ± 0 | 12 ± 1 | 23 ± 1 | 27 ± 1 | 14 ± 5 |
| Alanine (mg/g dm) | 22 ± 1 | 17 ± 2 | 38 ± 1 | 34 ± 1 | 21 ± 5 |
| Threonine (mg/g dm) 2 | 17 ± 0 | 10 ± 0 | 22 ± 1 | 22 ± 0 | 15 ± 2 |
| Glycine (mg/g dm) | 17 ± 0 | 12 ± 2 | 27 ± 1 | 24 ± 1 | 15 ± 4 |
| Valine (mg/g dm) 2 | 25 ± 1 | 15 ± 3 | 27 ± 0 | 28 ± 1 | 14 ± 5 |
| Serine (mg/g dm) | 16 ± 0 | 12 ± 0 | 18 ± 1 | 19 ± 0 | 14 ± 1 |
| Proline (mg/g dm) | 16 ± 0 | 9 ± 0 | 33 ± 0 | 34 ± 0 | 14 ± 3 |
| Isoleucine (mg/g dm) 2 | 13 ± 1 | 11 ± 0 | 19 ± 0 | 22 ± 1 | 9 ± 4 |
| Leucine (mg/g dm) 2 | 28 ± 0 | 19 ± 0 | 41 ± 0 | 41 ± 1 | 19 ± 11 |
| Methionine (mg/g dm) 2 | 2 ± 0 | 5 ± 1 | 8 ± 0 | 7 ± 1 | 3 ± 1 |
| Histidine (mg/g dm) 2 | 6 ± 0 | 3 ± 0 | 11 ± 0 | 10 ± 0 | 4 ± 1 |
| Phenylalanine (mg/g dm) 2 | 17 ± 0 | 10 ± 0 | 24 ± 0 | 23 ± 0 | 14 ± 4 |
| Glutamate and glutamine (mg/g dm) | 45 ± 6 | 24 ± 2 | 52 ± 1 | 50 ± 0 | 37 ± 4 |
| Aspartate and asparagine (mg/g dm) | 37 ± 2 | 23 ± 1 | 40 ± 1 | 40 ± 1 | 27 ± 3 |
| Cystine (mg/g dm) 3 | 2 ± 0 | 0 ± 0 | 3 ± 0 | 2 ± 0 | 1 ± 1 |
| Tyrosine (mg/g dm) | 10 ± 0 | 9 ± 0 | 16 ± 0 | 16 ± 0 | 8 ± 1 |
| C. nivalis | P. purpureum | C. vulgaris | Disrupted N. gaditana | Disrupted Scenedesmus | |
|---|---|---|---|---|---|
| P (mg/g dm) | 7.20 ± 0.11 | 7.73 ± 0.08 | 10.44 ± 0.16 | 13.35 ± 0.15 | 5.19 ± 0.04 |
| K (mg/g dm) | 5.27 ± 0.10 | 9.84 ± 0.26 | 7.17 ± 0.03 | 13.35 ± 0.30 | 7.59 ± 0.20 |
| Ca (mg/g dm) | 3.99 ± 0.04 | 8.72 ± 0.17 | 1.04 ± 0.01 | 3.05 ± 0.10 | 1.81 ± 0.05 |
| Na (mg/g dm) | 1.07 ± 0.03 | 2.91 ± 0.09 | 17.11 ± 0.21 | 3.39 ± 0.10 | 0.13 ± 0.00 |
| Cl (mg/g dm) | 0.94 ± 0.03 | 1.75 ± 0.10 | 18.57 ± 0.35 | 3.03 ± 0.01 | 0.47 ± 0.02 |
| Fe (mg/g dm) | 0.10 ± 0.00 | 0.11 ± 0.01 | 1.12 ± 0.00 | 0.29 ± 0.00 | 0.37 ± 0.00 |
| After digestion | |||||
| Soluble orthophosphate (mg P/g dm) | 4.83 ± 0.28 | - 1 | 6.03 ± 1.94 | 7.25 ± 0.92 | 2.35 ± 0.08 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Verspreet, J.; Soetemans, L.; Gargan, C.; Hayes, M.; Bastiaens, L. Nutritional Profiling and Preliminary Bioactivity Screening of Five Micro-Algae Strains Cultivated in Northwest Europe. Foods 2021, 10, 1516. https://doi.org/10.3390/foods10071516
Verspreet J, Soetemans L, Gargan C, Hayes M, Bastiaens L. Nutritional Profiling and Preliminary Bioactivity Screening of Five Micro-Algae Strains Cultivated in Northwest Europe. Foods. 2021; 10(7):1516. https://doi.org/10.3390/foods10071516
Chicago/Turabian StyleVerspreet, Joran, Lise Soetemans, Caoimhe Gargan, Maria Hayes, and Leen Bastiaens. 2021. "Nutritional Profiling and Preliminary Bioactivity Screening of Five Micro-Algae Strains Cultivated in Northwest Europe" Foods 10, no. 7: 1516. https://doi.org/10.3390/foods10071516






