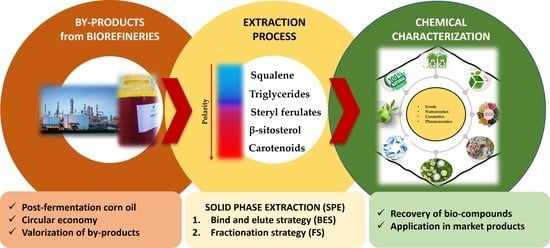
Valorization of By-Products from Biofuel Biorefineries: Extraction and Purification of Bioactive Molecules from Post-Fermentation Corn Oil
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Sample Treatment
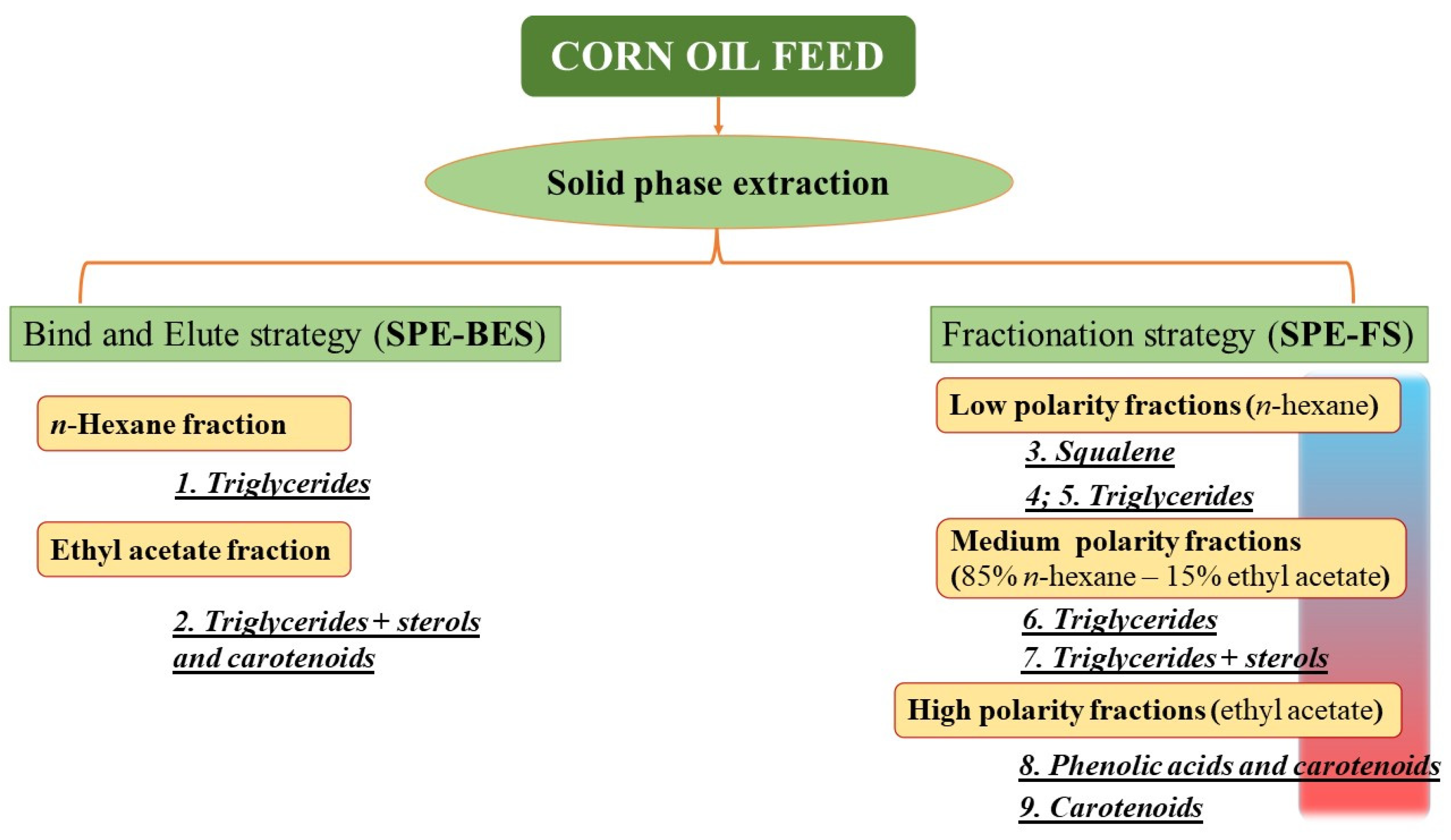
2.3. Solid Phase Extraction (SPE)
2.4. Purification Step
2.4.1. Phytosterols Purification
2.4.2. Selective Extraction of Carotenoids on RP18 Silica Gel
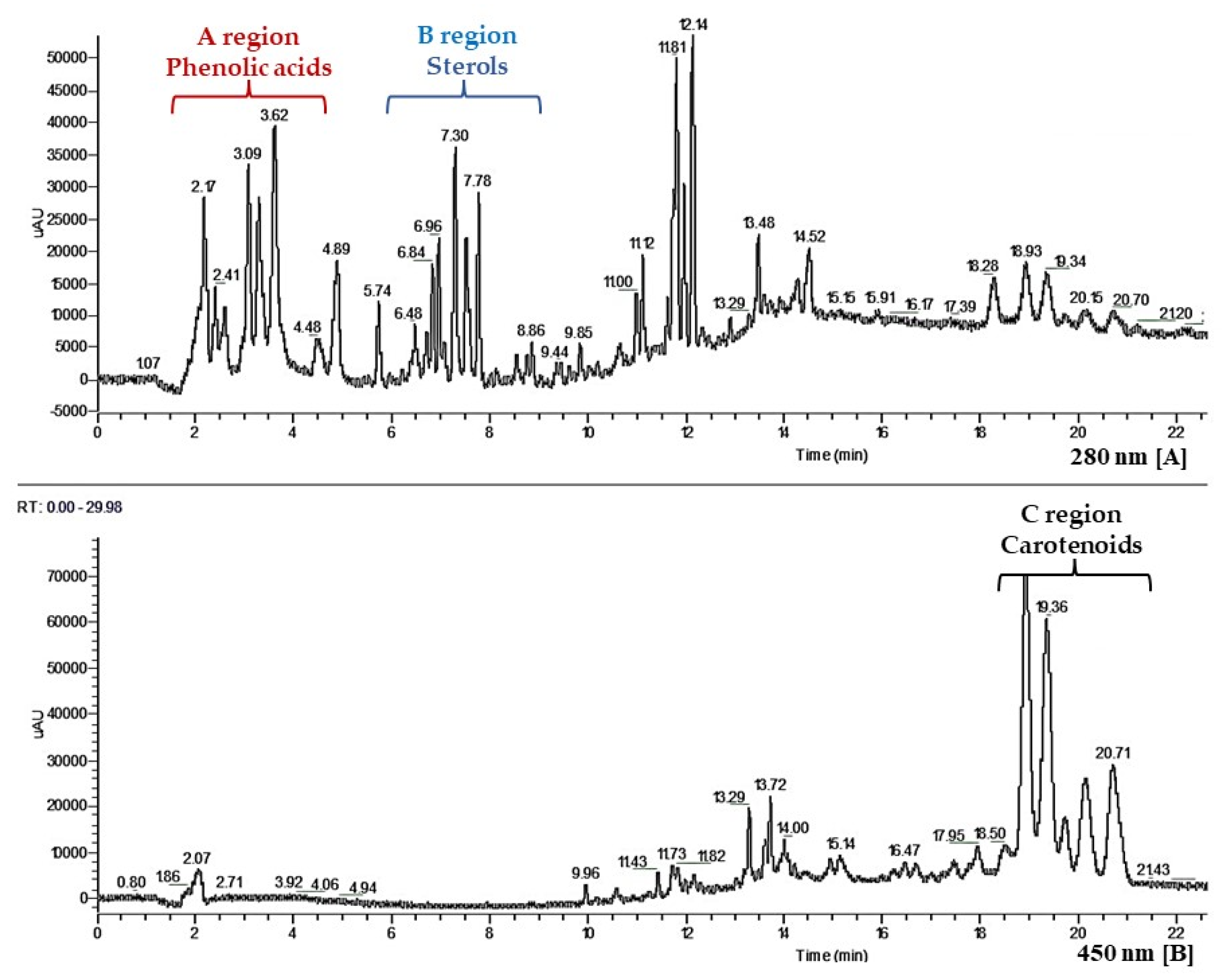
2.5. High-Performance Liquid Chromatography (HPLC) Analysis
2.6. High-Performance Liquid Chromatography Coupled to Mass Spectrometer (LC–MS/MS)
2.7. Semipreparative HPLC-Refractive Index Detector
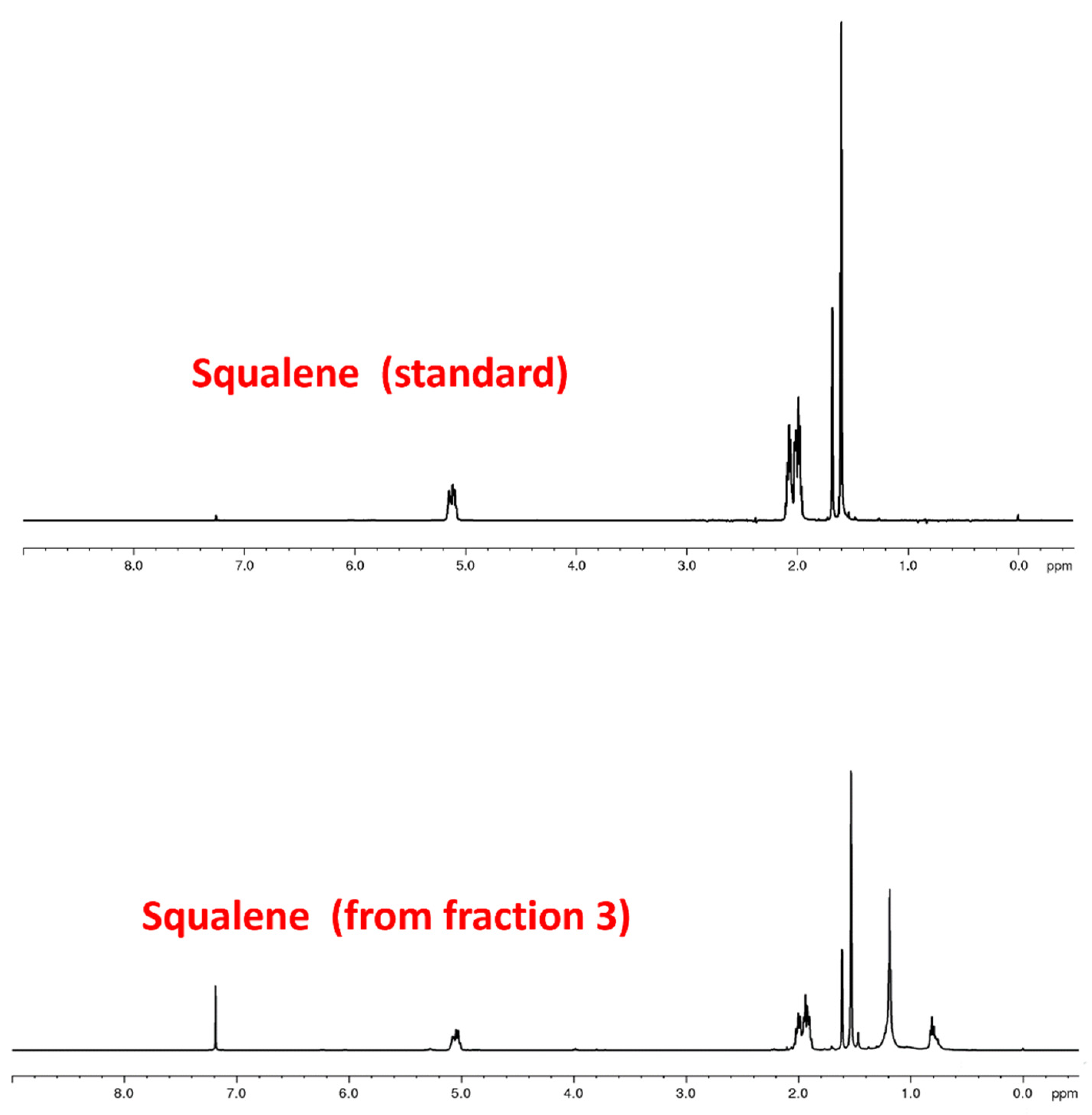
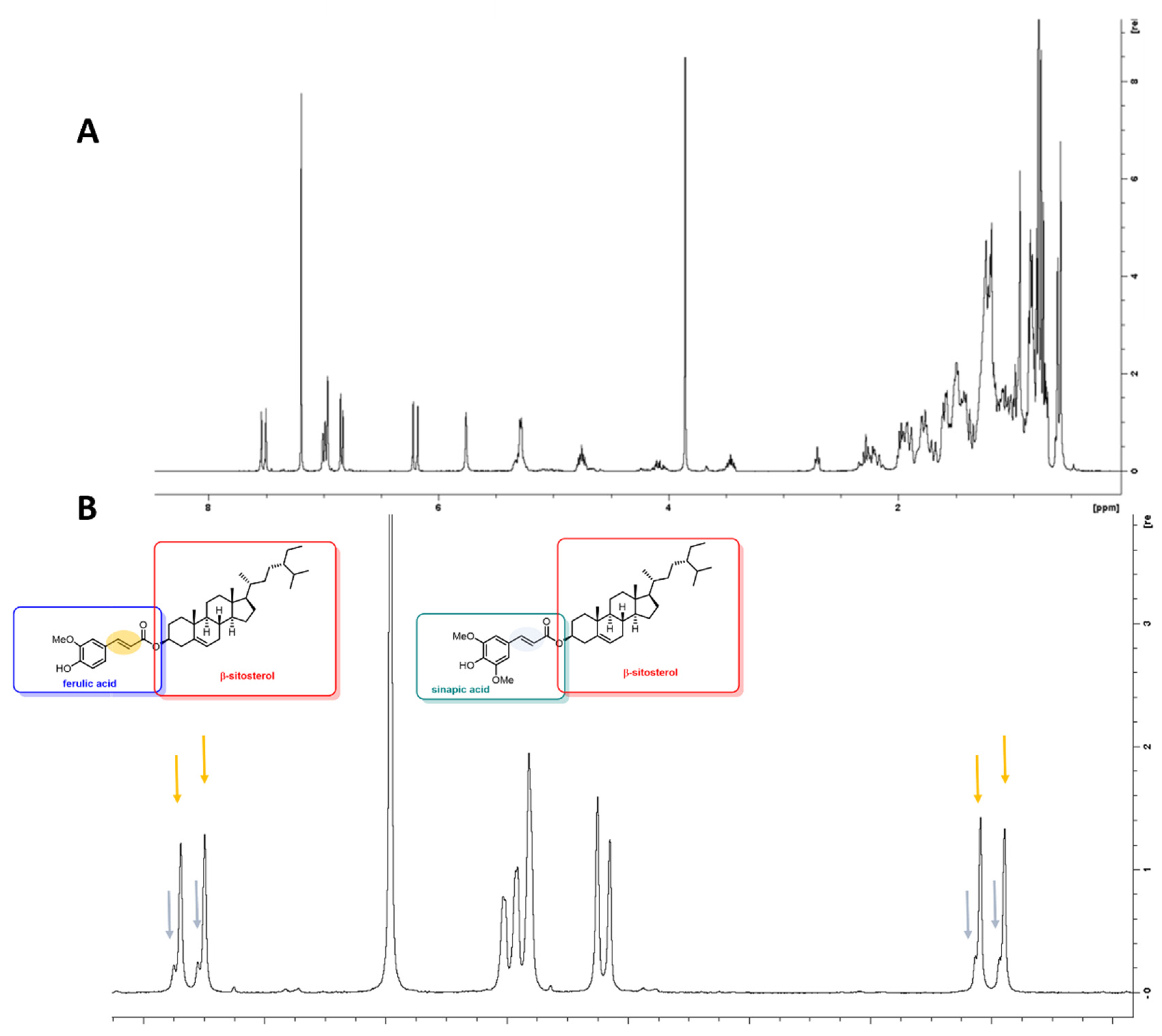
2.8. 1H- and 13C-NMR Analysis
2.9. Total Phenolic Content (TPC) Evaluation
2.10. Total Flavonoid Content (TFC) Evaluation
2.11. DPPH Radical Scavenging Assay
2.12. Trolox Equivalent Antioxidant Capacity (TEAC) Assay
2.13. α-Amylase Inhibitory Activity
2.14. Tyrosinase Inhibitory Activity
2.15. Cholinesterase Inhibitory Activity
2.16. α-Glucosidase Inhibitory Activity
2.17. Statistical Analysis
3. Results and Discussion
3.1. Solid Phase Extraction (SPE) and HPLC Analysis
3.2. Antioxidant and Enzymatic Activity
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- European Commission. Communication from the Commission to the European Parliament, the Council, the European Economic and Social Committee and the Committee of the Regions. In A New Circular Economy Action Plan for a Cleaner and More Competitive Europe; European Commission: Brussels, Belgium, 2020. [Google Scholar]
- Oreopoulou, V.; Tzia, C. Utilization of plant by-products for the recovery of proteins, dietary fibers, antioxidants, and colorants. In Utilization of By-Products and Treatment of Waste in the Food Industry; Oreopoulou, V., Russ, W., Eds.; Springer: New York, NY, USA, 2007; Volume 3, pp. 209–232. [Google Scholar]
- Mirabella, N.; Castellani, V.; Sala, S. Current options for the valorization of food manufacturing waste: A review. J. Clean. Prod. 2014, 65, 28–41. [Google Scholar] [CrossRef] [Green Version]
- Ben-Othman, S.; Jõudu, I.; Bhat, R. Bioactives from agri-food wastes: Present insights and future challenges. Molecules 2020, 25, 510. [Google Scholar] [CrossRef] [Green Version]
- Marchetti, J.M. A summary of the available technologies for biodiesel production based on a comparison of different feedstock’s properties. Process Saf. Environ. Prot. 2012, 90, 157–163. [Google Scholar] [CrossRef]
- ePURE. European Renewable Ethanol—Key Figures. 2020. Available online: https://www.google.com.hk/url?sa=t&rct=j&q=&esrc=s&source=web&cd=&ved=2ahUKEwj77ou2g5_1AhUUT2wGHViIAmYQFnoECAoQAQ&url=https%3A%2F%2Fwww.epure.org%2Fwp-content%2Fuploads%2F2021%2F09%2F210823-DEF-PR-European-renewable-ethanol-Key-figures-2020-web.pdf&usg=AOvVaw1upJjrrr-43EX96SRZinr1 (accessed on 22 November 2021).
- Di Lena, G.; Ondrejíčková, P.; del Pulgar, J.S.; Cyprichová, V.; Ježovič, T.; Lucarini, M.; Lombardi Boccia, G.; Ferrari Nicoli, S.; Gabrielli, P.; Aguzzi, A.; et al. Towards a Valorization of Corn Bioethanol Side Streams: Chemical Characterization of Post Fermentation Corn Oil and Thin Stillage. Molecules 2020, 25, 3549. [Google Scholar] [CrossRef]
- Di Lena, G.; Sanchez del Pulgar, J.; Lombardi Boccia, G.; Casini, I.; Ferrari Nicoli, S. Corn Bioethanol Side Streams: A Potential Sustainable Source of Fat-Soluble Bioactive Molecules for High-Value Applications. Foods 2020, 9, 1788. [Google Scholar] [CrossRef] [PubMed]
- Martins, N.; Barros, L.; Dueñas, M.; Santos-Buelga, C.; Ferreira, I.C.F.R. Characterization of phenolic compounds and antioxidant properties of Glycyrrhiza glabra L. rhizomes and roots. RSC Adv. 2015, 5, 26991–26997. [Google Scholar] [CrossRef] [Green Version]
- Mocan, A.; Schafberg, M.; Crișan, G.; Rohn, S. Determination of lignans and phenolic components of Schisandra chinensis (Turcz.) Baill. using HPLC-ESI-ToF-MS and HPLC-online TEAC: Contribution of individual components to overall antioxidant activity and comparison with traditional antioxidant assays. J. Funct. Foods 2016, 24, 579–594. [Google Scholar] [CrossRef]
- Zengin, G. A study on in vitro enzyme inhibitory properties of Asphodeline anatolica: New sources of natural inhibitors for public health problems. Ind. Crop. Prod. 2016, 83, 39–43. [Google Scholar] [CrossRef]
- Carradori, S.; Cairone, F.; Garzoli, S.; Fabrizi, G.; Iazzetti, A.; Giusti, A.M.; Menghini, L.; Uysal, S.; Ak, G.; Zengin, G.; et al. Phytocomplex Characterization and Biological Evaluation of Powdered Fruits and Leaves from Elaeagnus angustifolia. Molecules 2020, 25, 2021. [Google Scholar] [CrossRef] [PubMed]
- Chiavaroli, A.; Recinella, L.; Ferrante, C.; Locatelli, M.; Carradori, S.; Macchione, N.; Zengin, G.; Leporini, L.; Leone, S.; Martinotti, S.; et al. Crocus sativus, Serenoa repens and Pinus massoniana extracts modulate inflammatory response in isolated rat prostate challenged with LPS. J. Biol. Regul. Homeost. Agents 2017, 31, 531–541. [Google Scholar]
- Azzouz, A.; Kailasa, S.K.; Lee, S.S.; Rascón, A.J.; Ballesteros, E.; Zhang, M.; Kim, K.-H. Review of nanomaterials as sorbents in solid-phase extraction for environmental samples. TrAC Trends Anal. Chem. 2018, 108, 347–369. [Google Scholar] [CrossRef]
- Nergiz, C.; Çelikkale, D. The effect of consecutive steps of refining on squalene content of vegetable oils. J. Food Sci. Technol. 2011, 48, 382–385. [Google Scholar] [CrossRef] [Green Version]
- Huang, Z.-R.; Lin, Y.-K.; Fang, J.-Y. Biological and Pharmacological Activities of Squalene and Related Compounds: Potential Uses in Cosmetic Dermatology. Molecules 2009, 14, 540–554. [Google Scholar] [CrossRef]
- Espinosa, R.R.; Inchingolo, R.; Alencar, S.M.; Rodriguez-Estrada, M.T.; Castro, I.A. Antioxidant activity of phenolic compounds added to a functional emulsion containing omega-3 fatty acids and plant sterol esters. Food Chem. 2015, 182, 95–104. [Google Scholar] [CrossRef]
- García-Llatas, G.; Rodríguez-Estrada, M.T. Current and new insights on phytosterol oxides in plant sterol-enriched food. Chem. Phys. Lipids 2011, 164, 607–624. [Google Scholar] [CrossRef] [PubMed]
- Yuan, C.; Xie, Y.; Jin, R.; Ren, L.; Zhou, L.; Zhu, M.; Ju, Y.J. Simultaneous analysis of tocopherols, phytosterols, and squalene in vegetable oils by high-performance liquid chromatography. Food Anal. Methods 2017, 10, 3716–3722. [Google Scholar] [CrossRef]
- Wester, I. Cholesterol-lowering effect of plant sterols. Eur. J. Lipid Sci. Technol. 2000, 102, 37–44. [Google Scholar] [CrossRef]
- Kurilich, A.C.; Juvik, J.A. Quantification of Carotenoid and Tocopherol Antioxidants in Zea mays. J. Agric. Food Chem. 1999, 47, 1948–1955. [Google Scholar] [CrossRef] [PubMed]
- Moreau, R.A.; Johnston, D.B.; Hicks, K.B. A comparison of the levels of lutein and zeaxanthin in corn germ oil, corn fiber oil and corn kernel oil. J. Am. Oil Chem. Soc. 2007, 84, 1039–1044. [Google Scholar] [CrossRef]
- Mocan, A.; Moldovan, C.; Zengin, G.; Bender, O.; Locatelli, M.; Simirgiotis, M.; Atalay, A.; Vodnar, D.C.; Rohn, S.; Crișan, G. UHPLC-QTOF-MS analysis of bioactive constituents from two Romanian Goji (Lycium barbarum L.) berries cultivars and their antioxidant, enzyme inhibitory, and real-time cytotoxicological evaluation. Food Chem. Toxicol. 2018, 115, 414–424. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.; Lu, M.; Zhang, T.; Zhang, Z.; Jin, Q.; Chang, M.; Wang, X. Evaluation of the antioxidant properties of micronutrients in different vegetable oils. Eur. J. Lipid Sci. Technol 2020, 122, 1900079–1900087. [Google Scholar] [CrossRef]
- De Bruno, A.; Romeo, R.; Piscopo, A.; Poiana, M. Antioxidant quantification in different portions obtained during olive oil extraction process in an olive oil press mill. J. Sci. Food Agric. 2021, 101, 1119–1126. [Google Scholar] [CrossRef] [PubMed]
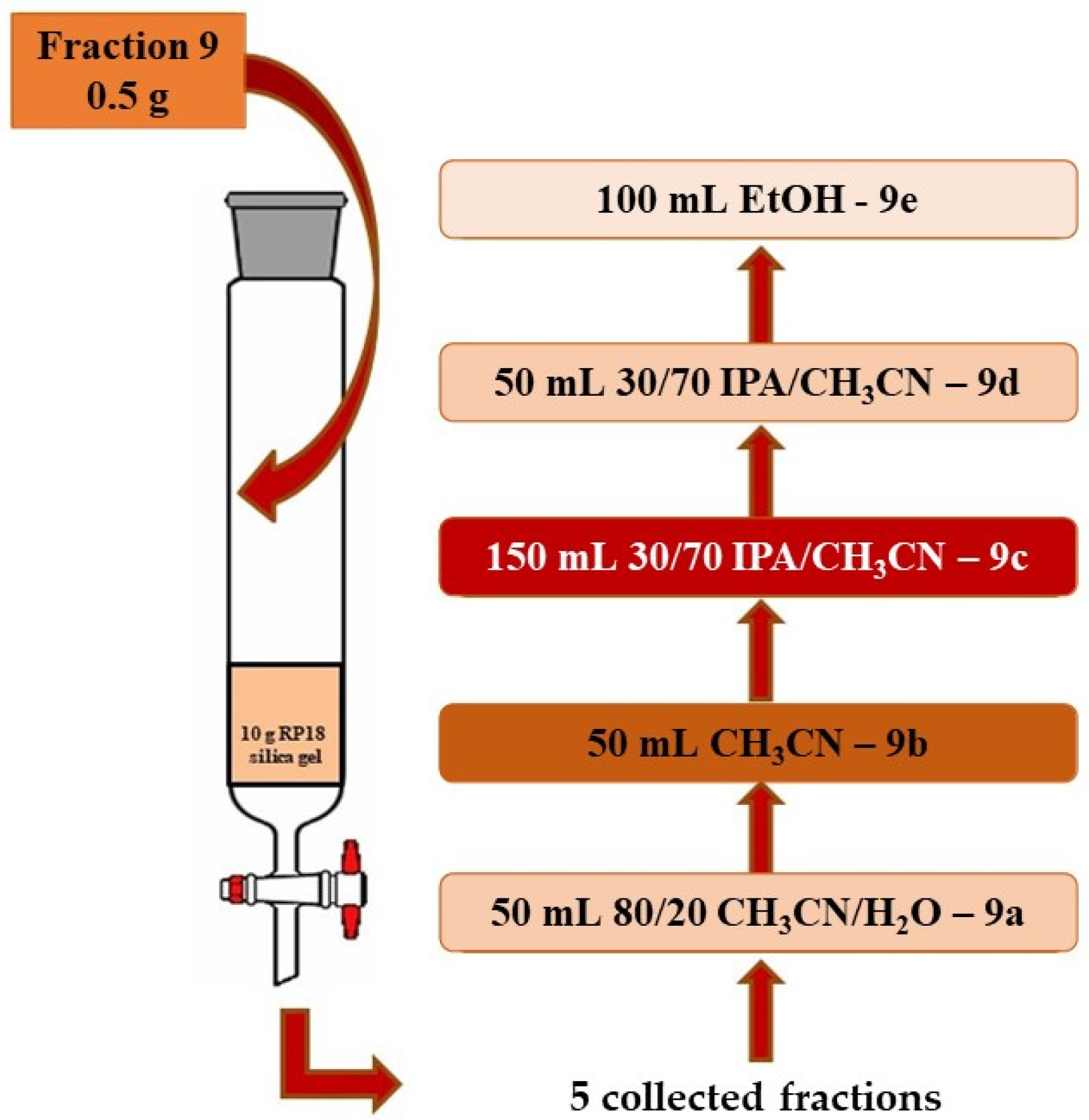
| Fraction Polarity | Vol (L) | Mobile Phase | Yield % |
|---|---|---|---|
| 3 | 0–0.25 | n-hexane | 0.01 |
| 4 | 0.25–0.50 | n-hexane | 5.90 |
| 5 | 0.50–1.50 | n-hexane | 44.50 |
| 6 | 0–1.00 | n-hex.:Et. Acet. 85:15 | 41.70 |
| 7 | 1.00–1.50 | n-hex.:Et. Acet. 85:15 | 3.10 |
| 8 | 0–1.00 | Ethyl acetate | 2.80 |
| 9 | 1.00–1.50 | Ethyl acetate | 0.60 |
| Fractions | γ-T3 a | α-T3 | δ-T | γ-T a | α-T | ERG | AVN * | STG + CAMP | β-SITO | SQUA |
|---|---|---|---|---|---|---|---|---|---|---|
| 2 | - | - | - | - | - | 1.54 | 8.79 | 4.71 | 31.39 | |
| 3 | - | - | - | - | - | - | - | - | - | 44.70 |
| 7 | - | - | - | - | - | 3.74 | 8.38 | 13.42 | 37.50 | 0.03 |
| 8 | - | - | - | - | - | 2.77 | 14.78 | 10.86 | 59.94 | - |
| 9 | - | - | - | - | - | 4.88 | 9.27 | 9.57 | 43.50 | - |
| 9a | - | - | - | - | - | - | - | - | - | - |
| 9b | - | - | - | - | - | - | - | - | - | - |
| 9c | - | - | - | - | - | 11.28 | 19.24 | 15.70 | 99.37 | - |
| 9d | 0.15 | - | - | - | - | 16.88 | 41.63 | 101.09 | 271.07 | - |
| 9e | 0.20 | 0.04 | - | - | - | 4.17 | 9.95 | 18.79 | 73.56 | - |
| Crude corn oil | 0.20 | 0.21 | 0.02 | 0.72 | 0.22 | 0.38 | 1.43 | 1.03 | 4.18 | 0.74 |
| Fractions | Lutein | Zeaxanthin | N.I.C. 1 | N.I.C. 2 | N.I.C. 3 | N.I.C. 4 | N.I.C. 5 | N.I.C. 6 | N.I.C. 7 | N.I.C. 8 | N.I.C. 9 | Total |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 2 | 70.1 | 63.6 | 3.9 | 4.4 | 10.9 | 9.7 | 2.7 | 6.5 | 20.5 | 8.6 | 23.5 | 224.4 |
| 8 | - | - | - | - | - | - | - | - | - | - | - | - |
| 9 | 822.0 | 654.0 | 22.8 | 29.3 | 50.5 | 84.2 | 25.9 | 47.1 | 226.2 | 120 | 304.2 | 2386 |
| 9a | - | - | - | - | - | - | - | - | - | - | - | - |
| 9b | - | - | - | - | - | - | - | - | - | - | - | - |
| 9c | 2573.0 | 2151.0 | 69.1 | 97.5 | 196.8 | 332.5 | 85.1 | 182.3 | 230 | 1548 | 437.9 | 7903 |
| 9d | - | - | - | - | - | - | - | - | - | - | - | - |
| 9e | - | - | - | - | - | - | - | - | - | - | - | - |
| Crude corn oil | 133.7 | 134.1 | 3.3 | 7.5 | 10.4 | 26.1 | 13.4 | - | 53.6 | 12.4 | 45.4 | 440 |
| Fractions | Ferulic Acid | p-Coumaric Acid | Vanillic Acid | Syringic Acid | Sinapic Acid | Sum |
|---|---|---|---|---|---|---|
| 2 | 56 | 51 | 4 | - | - | 111 |
| 8 | - | - | - | - | - | - |
| 9 | 728 | 554 | 189 | 27 | 10 | 1508 |
| Crude corn oil | 7.83 | 3.89 | 4.54 | 0.91 | 0.89 | 18.06 |
| Fractions 1 | TPC (mgGAE/g) 2 | TFC (mgRE/g) 3 | DPPH (mgTE/g) 4 | ABTS (mgTE/g) 4 |
|---|---|---|---|---|
| 9 | 14.04 ± 0.03 b | 3.50 ± 0.19 a | 5.69 ± 0.38 b | 14.94 ± 0.48 a |
| 9c | 15.11 ± 0.10 a | 0.46 ± 0.07 b | 10.33 ± 0.27 a | 14.00 ± 0.05 b |
| Fractions 1 | AChE Inhibition (mgGALAE/g) 2 | α-Amylase Inhibition (mmolACAE/g) 3 | α-Glucosidase Inhibition (mmolACAE/g) 3 | Tyrosinase Inhibition (mgKAE/g) 4 |
|---|---|---|---|---|
| 9 | 0.76 ± 0.05 | 0.32 ± 0.02 b | 0.29 ± 0.02 a | 19.42 ± 0.96 a |
| 9c | - | 0.38 ± 0.01 a | 0.48 ± 0.21 a | 16.72 ± 0.23 b |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cairone, F.; Cesa, S.; Ciogli, A.; Fabrizi, G.; Goggiamani, A.; Iazzetti, A.; Di Lena, G.; Sanchez del Pulgar, J.; Lucarini, M.; Cantò, L.; et al. Valorization of By-Products from Biofuel Biorefineries: Extraction and Purification of Bioactive Molecules from Post-Fermentation Corn Oil. Foods 2022, 11, 153. https://doi.org/10.3390/foods11020153
Cairone F, Cesa S, Ciogli A, Fabrizi G, Goggiamani A, Iazzetti A, Di Lena G, Sanchez del Pulgar J, Lucarini M, Cantò L, et al. Valorization of By-Products from Biofuel Biorefineries: Extraction and Purification of Bioactive Molecules from Post-Fermentation Corn Oil. Foods. 2022; 11(2):153. https://doi.org/10.3390/foods11020153
Chicago/Turabian StyleCairone, Francesco, Stefania Cesa, Alessia Ciogli, Giancarlo Fabrizi, Antonella Goggiamani, Antonia Iazzetti, Gabriella Di Lena, Jose Sanchez del Pulgar, Massimo Lucarini, Luca Cantò, and et al. 2022. "Valorization of By-Products from Biofuel Biorefineries: Extraction and Purification of Bioactive Molecules from Post-Fermentation Corn Oil" Foods 11, no. 2: 153. https://doi.org/10.3390/foods11020153
APA StyleCairone, F., Cesa, S., Ciogli, A., Fabrizi, G., Goggiamani, A., Iazzetti, A., Di Lena, G., Sanchez del Pulgar, J., Lucarini, M., Cantò, L., Zengin, G., & Ondrejíčková, P. (2022). Valorization of By-Products from Biofuel Biorefineries: Extraction and Purification of Bioactive Molecules from Post-Fermentation Corn Oil. Foods, 11(2), 153. https://doi.org/10.3390/foods11020153