Mangiferin for the Management of Liver Diseases: A Review
Abstract
:1. Introduction
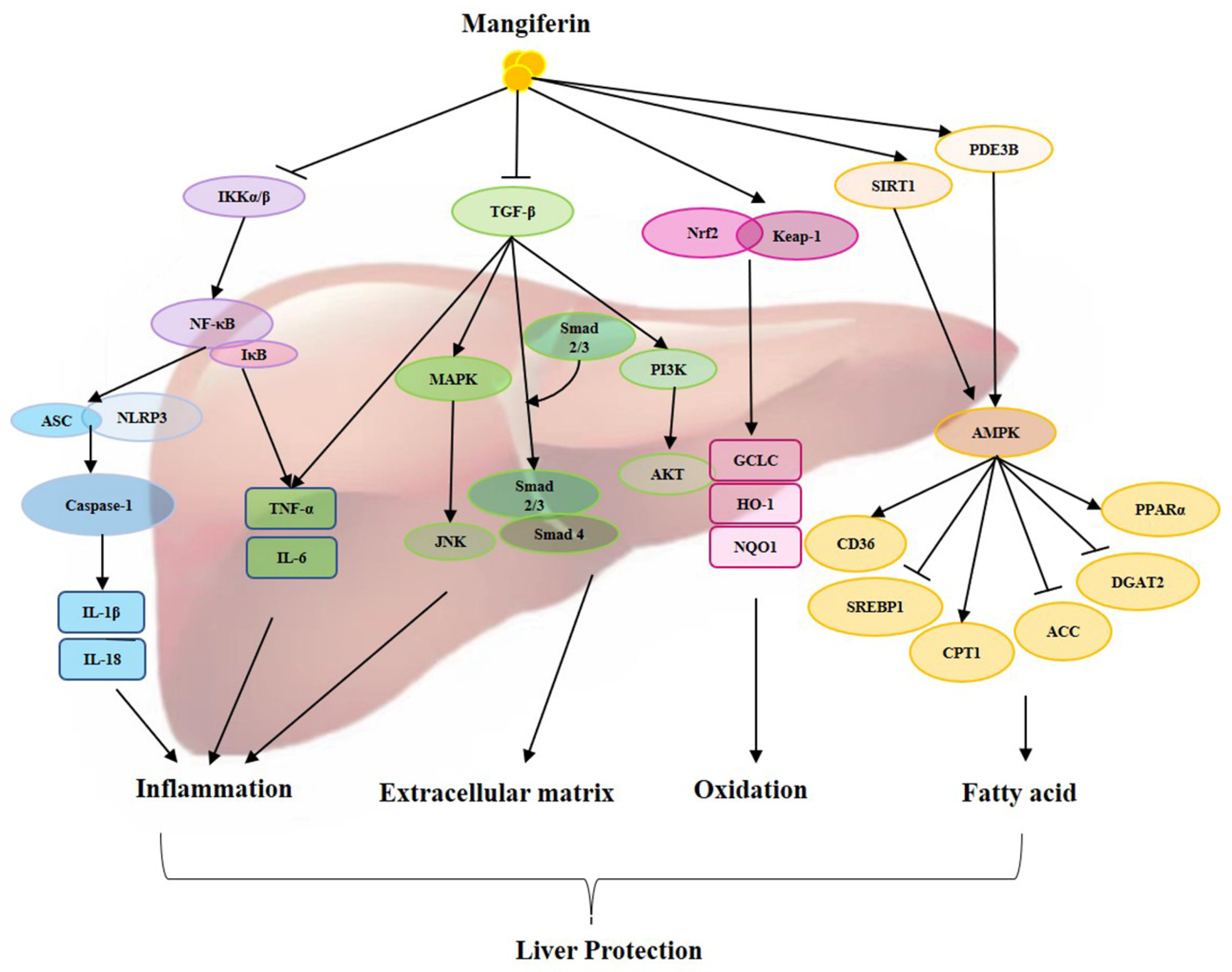
2. Effect Targets of Mangiferin in Liver
2.1. NF-κB
2.2. TGF-β/Smad
2.3. Nrf-2/Keap-1
2.4. NLRP3
2.5. AMPK
2.6. Other Targets
3. Mangiferin Protects against Liver Disease
3.1. Liver Injury
3.2. Nonalcoholic Fatty Liver Disease
3.3. Alcoholic Liver Disease
3.4. Liver Fibrosis
3.5. Hepatocellular Carcinoma
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Trefts, E.; Gannon, M.; Wasserman, D.H. The liver. Curr. Biol. 2017, 27, R1147–R1151. [Google Scholar] [CrossRef]
- Ivana, M.; Ankica, V.; Aleksandra, B.; Marina, D.; Milos, K.; Aleksandra, R.S.; Ivana, G.; Olja, S.; Vladimir, D.; Nebojsa, L.; et al. Gut-liver axis, gut microbiota, and its modulation in the management of liver diseases: A review of the literature. Int. J. Mol. Sci. 2019, 20, 395. [Google Scholar]
- Bajaj, J.S. Alcohol, liver disease and the gut microbiota. Nat. Rev. Gastroenterol. Hepatol. 2019, 16, 235–246. [Google Scholar] [CrossRef] [PubMed]
- Devarbhavi, H.; Asrani, S.K.; Arab, J.P.; Nartey, Y.A.; Pose, E.; Kamath, P.S. Global burden of liver disease: 2023 update. J. Hepatol. 2023, S0168-8278, 00194. [Google Scholar] [CrossRef]
- Roehlen, N.; Crouchet, E.; Baumert, T.F. Liver fibrosis: Mechanistic concepts and therapeutic perspectives. Cells 2020, 9, 875. [Google Scholar] [CrossRef] [Green Version]
- Anwanwan, D.; Singh, S.K.; Singh, S.; Saikam, V.; Singh, R. Challenges in liver cancer and possible treatment approaches. Biochim. Biophys. Acta Rev. Cancer 2020, 1873, 188314. [Google Scholar] [CrossRef]
- Imran, M.; Arshad, M.S.; Butt, M.S.; Kwon, J.H.; Arshad, M.U.; Sultan, M.T. Mangiferin: A natural miracle bioactive compound against lifestyle related disorders. Lipids. Health Dis. 2017, 16, 84. [Google Scholar] [CrossRef]
- Dong, M.; Li, L.; Li, G.; Song, J.; Liu, B.; Liu, X.; Wang, M. Mangiferin protects against alcoholic liver injury via suppression of inflammation-induced adipose hyperlipolysis. Food Funct. 2020, 11, 8837–8851. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Liu, C.; Yin, L.; Huang, C.; Fan, S. Mangiferin relieves CCl4-induced liver fibrosis in mice. Sci. Rep. 2023, 13, 4172. [Google Scholar] [CrossRef]
- Tan, H.Y.; Wang, N.; Li, S.; Hong, M.; Guo, W.; Man, K.; Cheng, C.S.; Chen, Z.; Feng, Y. Repression of WT1-Mediated LEF1 transcription by mangiferin governs β-catenin-independent wnt signalling inactivation in hepatocellular carcinoma. Cell. Physiol. Biochem. 2018, 47, 1819–1834. [Google Scholar] [CrossRef]
- Vidhushini, S.; Sugumar, M.; Malarvizhi, R.; Nithya, P.; Hannah, R.V. Antidiabetic effect of mangiferin in combination with oral hypoglycemic agents metformin and gliclazide. Phytomedicine 2019, 59, 152901. [Google Scholar]
- Sauvik, B.; Ahmmed, S.M.; Bishnu, P.S.; Pulok, K.M. Soya phospholipid complex of mangiferin enhances its hepatoprotectivity by improving its bioavailability and pharmacokinetics. Sci. Food Agric. 2014, 94, 1380–1388. [Google Scholar]
- De Souza Basso, B.; Haute, G.V.; Ortega-Ribera, M.; Luft, C.; Antunes, G.L.; Bastos, M.S.; Carlessi, L.P.; Levorse, V.G.; Cassel, E.; Donadio, M.V.F.; et al. Methoxyeugenol deactivates hepatic stellate cells and attenuates liver fibrosis and inflammation through a PPAR-ɣ and NF-kB mechanism. J. Ethnopharmacol. 2021, 280, 114433. [Google Scholar] [CrossRef]
- Xiao, C.; Ghosh, S. NF-kappaB, an evolutionarily conserved mediator of immune and inflammatory responses. Adv. Exp. Med. Biol. 2005, 560, 41–45. [Google Scholar] [PubMed]
- Luedde, T.; Schwabe, R.F. NF-κB in the liver--linking injury, fibrosis and hepatocellular carcinoma. Nat. Rev. Gastroenterol. Hepatol. 2011, 8, 108–118. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Broitman, S.A.; Gottlieb, L.S.; Zamcheck, N. Influence of neomycin and ingested endotoxin in the pathogenesis of choline deficiency cirhosis in the adult rat. J. Exp. Med. 1964, 119, 633–642. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rajagopal, C.; Lankadasari, M.B.; Aranjani, J.M.; Harikumar, K.B. Targeting oncogenic transcription factors by polyphenols: A novel approach for cancer therapy. Pharmacol. Res. 2018, 130, 273–291. [Google Scholar] [CrossRef]
- Abdalla, Y.; Abdalla, A.; Hamza, A.A.; Amin, A. Safranal prevents liver cancer through inhibiting oxidative stress and alleviating inflammation. Front. Pharmacol. 2021, 12, 777500. [Google Scholar] [CrossRef]
- Núñez Selles, A.J.; Daglia, M.; Rastrelli, L. The potential role of mangiferin in cancer treatment through its immunomodulatory, anti-angiogenic, apoptopic, and gene regulatory effects. BioFactors 2016, 42, 475–491. [Google Scholar] [CrossRef]
- Pal, P.B.; Sinha, K.; Sil, P.C. Mangiferin, a natural xanthone, protects murine liver in Pb(II) induced hepatic damage and cell death via MAP kinase, NF-κB and mitochondria dependent pathways. PLoS ONE 2013, 8, e56894. [Google Scholar] [CrossRef] [Green Version]
- Chowdhury, A.; Lu, J.; Zhang, R.; Nabila, J.; Gao, H.; Wan, Z.; Adelusi Temitope, I.; Yin, X.; Sun, Y. Mangiferin ameliorates acetaminophen-induced hepatotoxicity through APAP-Cys and JNK modulation. Biomed. Pharmacother. 2019, 117, 109097. [Google Scholar] [CrossRef]
- Xu, L.; Tian, D.; Zheng, Y. Pleiotropic roles of TGF-β/Smad signaling in the progression of chronic liver disease. Crit. Rev. Eukaryot. Gene Exp. 2013, 23, 237–255. [Google Scholar] [CrossRef]
- Pouponnot, C.; Jayaraman, L.; Massagué, J. Physical and functional interaction of SMADs and p300/CBP. J. Biol. Chem. 1998, 273, 22865–22868. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Graff, J.M.; Bansal, A.; Melton, D.A. Xenopus mad proteins transduce distinct subsets of signals for the TGF beta superfamily. Cell 1996, 85, 479–487. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Massagué, J.; Seoane, J.; Wotton, D. Smad transcription factors. Genes Dev. 2005, 19, 2783–2810. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Peng, D.; Fu, M.; Wang, M.; Wei, Y.; Wei, X. Targeting TGF-β signal transduction for fibrosis and cancer therapy. Mol. Cancer 2022, 21, 104. [Google Scholar] [CrossRef]
- Krasagakis, K.; Garbe, C.; Zouboulis, C.C.; Orfanos, C.E. Growth control of melanoma cells and melanocytes by cytokines. Recent. Results Cancer Res. 1995, 139, 169–182. [Google Scholar] [PubMed]
- Wang, M.L.; Wu, D.B.; Tao, Y.C.; Chen, L.L.; Liu, C.P.; Chen, E.Q.; Tang, H. The truncated mutant HBsAg expression increases the tumorigenesis of hepatitis B virus by regulating TGF-β/Smad signaling pathway. Virol. J. 2018, 15, 61. [Google Scholar] [CrossRef] [Green Version]
- Yoshida, K.; Matsuzaki, K.; Murata, M.; Yamaguchi, T.; Suwa, K.; Okazaki, K. Clinico-pathological importance of TGF-β/Phospho-Smad signaling during human hepatic fibrocarcinogenesis. Cancers 2018, 10, 183. [Google Scholar] [CrossRef] [Green Version]
- Andreotti, M.; Pirillo, M.F.; Liotta, G.; Jere, H.; Maulidi, M.; Sagno, J.B.; Luhanga, R.; Amici, R.; Mancini, M.G.; Gennaro, E.; et al. The impact of HBV or HCV infection in a cohort of HIV-infected pregnant women receiving a nevirapine-based antiretroviral regimen in Malawi. BMC Infect. Dis. 2014, 14, 180. [Google Scholar] [CrossRef] [Green Version]
- Mu, M.; Zuo, S.; Wu, R.M.; Deng, K.S.; Lu, S.; Zhu, J.J.; Zou, G.L.; Yang, J.; Cheng, M.L.; Zhao, X.K. Ferulic acid attenuates liver fibrosis and hepatic stellate cell activation via inhibition of TGF-β/Smad signaling pathway. Drug. Des. Devel. Ther. 2018, 12, 4107–4115. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jiang, Y.; Wang, C.; Li, Y.Y.; Wang, X.C.; An, J.D.; Wang, Y.J.; Wang, X.J. Mistletoe alkaloid fractions alleviates carbon tetrachloride-induced liver fibrosis through inhibition of hepatic stellate cell activation via TGF-β/Smad interference. J. Ethnopharmacol. 2014, 158, 230–238. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.L.; Zhang, X.Y.; Ge, X.Q.; Liu, M.X. Mangiferin prevents hepatocyte epithelial-mesenchymal transition in liver fibrosis via targeting HSP27-mediated JAK2/STAT3 and TGF-β1/Smad pathway. Phytother. Res. 2022, 36, 4167–4182. [Google Scholar] [CrossRef] [PubMed]
- Tao, Y.; Tian, X.; Luo, J.; Zhu, H.; Chu, Y.; Pei, L. Mangiferin inhibits chronic stress-induced tumor growth in colorectal liver metastases via WAVE2 signaling pathway. Heliyon 2023, 9, e13753. [Google Scholar] [CrossRef] [PubMed]
- Sies, H.; Berndt, C.; Jones, D.P. Oxidative Stress. Annu. Rev. Biochem. 2017, 86, 715–748. [Google Scholar] [CrossRef]
- Bellezza, I.; Giambanco, I.; Minelli, A.; Donato, R. Nrf2-Keap1 signaling in oxidative and reductive stress. Biochim. Biophys. Acta Mol. Cell. Res. 2018, 1865, 721–733. [Google Scholar] [CrossRef]
- Martin, F.; van Deursen, J.M.; Shivdasani, R.A.; Jackson, C.W.; Troutman, A.G.; Ney, P.A. Erythroid maturation and globin gene expression in mice with combined deficiency of NF-E2 and nrf-2. Blood 1998, 91, 3459–3466. [Google Scholar] [CrossRef] [Green Version]
- Itoh, K.; Chiba, T.; Takahashi, S.; Ishii, T.; Igarashi, K.; Katoh, Y.; Oyake, T.; Hayashi, N.; Satoh, K.; Hatayama, I.; et al. An Nrf2/small Maf heterodimer mediates the induction of phase II detoxifying enzyme genes through antioxidant response elements. Biochem. Biophys. Res. Commun. 1997, 236, 313–322. [Google Scholar] [CrossRef]
- Itoh, K.; Wakabayashi, N.; Katoh, Y.; Ishii, T.; Igarashi, K.; Engel, J.D.; Yamamoto, M. Keap1 represses nuclear activation of antioxidant responsive elements by Nrf2 through binding to the amino-terminal Neh2 domain. Genes Dev. 1999, 13, 76–86. [Google Scholar] [CrossRef] [Green Version]
- Wakabayashi, N.; Itoh, K.; Wakabayashi, J.; Motohashi, H.; Noda, S.; Takahashi, S.; Imakado, S.; Kotsuji, T.; Otsuka, F.; Roop, D.R.; et al. Keap1-null mutation leads to postnatal lethality due to constitutive Nrf2 activation. Nat. Genet. 2003, 35, 238–245. [Google Scholar] [CrossRef]
- Sadhukhan, P.; Saha, S.; Dutta, S.; Sil, P.C. Mangiferin ameliorates cisplatin induced acute kidney injury by upregulating Nrf-2 via the activation of PI3K and exhibits synergistic anticancer activity with cisplatin. Front. Pharmacol. 2018, 9, 638. [Google Scholar] [CrossRef] [Green Version]
- Waris, G.; Huh, K.W.; Siddiqui, A. Mitochondrially associated hepatitis B virus X protein constitutively activates transcription factors STAT-3 and NF-kappa B via oxidative stress. Mol. Cell. Biol. 2001, 21, 7721–7730. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, L.; Li, K.; Liu, Q.; Quiles, J.L.; Filosa, R.; Kamal, M.A.; Wang, F.; Kai, G.; Zou, X.; Teng, H.; et al. Protective effects of raspberry on the oxidative damage in HepG2 cells through Keap1/Nrf2-dependent signaling pathway. Food Chem. Toxicol. 2019, 133, 110781. [Google Scholar] [CrossRef] [PubMed]
- Das, J.; Ghosh, J.; Roy, A.; Sil, P.C. Mangiferin exerts hepatoprotective activity against D-galactosamine induced acute toxicity and oxidative/nitrosative stress via Nrf2-NFκB pathways. Toxicol. Appl. Pharmacol. 2012, 260, 35–47. [Google Scholar] [CrossRef] [PubMed]
- Pan, C.W.; Pan, Z.Z.; Hu, J.J.; Chen, W.L.; Zhou, G.Y.; Lin, W.; Jin, L.X.; Xu, C.L. Mangiferin alleviates lipopolysaccharide and D-galactosamine-induced acute liver injury by activating the Nrf2 pathway and inhibiting NLRP3 inflammasome activation. Eur. J. Pharmacol. 2016, 770, 85–91. [Google Scholar] [CrossRef]
- Szabo, G.; Petrasek, J. Inflammasome activation and function in liver disease. Nat. Rev. Gastroenterol. Hepatol. 2015, 12, 387–400. [Google Scholar] [CrossRef]
- Yu, L.; Li, Y.; Du, C.; Zhao, W.; Zhang, H.; Yang, Y.; Sun, A.; Song, X.; Feng, Z. Pattern recognition receptor-mediated chronic inflammation in the development and progression of obesity-related metabolic diseases. Mediat. Inflamm. 2019, 2019, 5271295. [Google Scholar] [CrossRef]
- Mohamadi, Y.; Mousavi, M.; Khanbabaei, H.; Salarinia, R.; Javankiani, S.; Hassanzadeh, G.; Momeni, F. The role of inflammasome complex in ischemia-reperfusion injury. J. Cell. Biochem. 2018, 124, 755–764. [Google Scholar] [CrossRef]
- Guan, Y.; Gu, Y.; Li, H.; Liang, B.; Han, C.; Zhang, Y.; Liu, Q.; Wei, W.; Ma, Y. NLRP3 inflammasome activation mechanism and its role in autoimmune liver disease. Acta Biochim. Biophys. Sin. 2022, 54, 1577–1586. [Google Scholar] [CrossRef]
- He, Y.; Hara, H.; Núñez, G. Mechanism and regulation of NLRP3 inflammasome activation. Trends Biochem. Sci. 2016, 41, 1012–1021. [Google Scholar] [CrossRef] [Green Version]
- Swanson, K.V.; Deng, M.; Ting, J.P. The NLRP3 inflammasome: Molecular activation and regulation to therapeutics. Nat. Rev. Immunol. 2019, 19, 477–489. [Google Scholar] [CrossRef]
- Kubes, P.; Mehal, W.Z. Sterile inflammation in the liver. Gastroenterology 2012, 143, 1158–1172. [Google Scholar] [CrossRef]
- Wree, A.; McGeough, M.D.; Peña, C.A.; Schlattjan, M.; Li, H.; Inzaugarat, M.E.; Messer, K.; Canbay, A.; Hoffman, H.M.; Feldstein, A.E. NLRP3 inflammasome activation is required for fibrosis development in NAFLD. J. Mol. Med. 2014, 92, 1069–1082. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yong, Z.; Ruiqi, W.; Hongji, Y.; Ning, M.; Chenzuo, J.; Yu, Z.; Zhixuan, X.; Qiang, L.; Qibing, L.; Weiying, L.; et al. Mangiferin ameliorates HFD-induced NAFLD through regulation of the AMPK and NLRP3 inflammasome signal pathways. J. Immunol. Res. 2021, 2021, 4084566. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Wu, C.; Guo, H.; Chu, C.; Hu, M.; Zhou, C. Mangiferin improves hepatic damage-associated molecular patterns, lipid metabolic disorder and mitochondrial dysfunction in alcohol hepatitis rats. Food Funct. 2019, 10, 3514–3534. [Google Scholar] [CrossRef] [PubMed]
- Carling, D. AMPK signalling in health and disease. Curr. Opin. Cell. Biol. 2017, 45, 31–37. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.W.; Choe, S.S.; Jang, H.; Kim, J.; Jeong, H.W.; Jo, H.; Jeong, K.H.; Tadi, S.; Park, M.G.; Kwak, T.H.; et al. AMPK activation with glabridin ameliorates adiposity and lipid dysregulation in obesity. J. Lipid Res. 2012, 53, 1277–1286. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Iseli, T.J.; Walter, M.; van Denderen, B.J.; Katsis, F.; Witters, L.A.; Kemp, B.E.; Michell, B.J.; Stapleton, D. AMP-activated protein kinase beta subunit tethers alpha and gamma subunits via its C-terminal sequence (186–270). J. Biol. Chem. 2005, 280, 13395–13400. [Google Scholar] [CrossRef] [Green Version]
- Smith, B.K.; Marcinko, K.; Desjardins, E.M.; Lally, J.S.; Ford, R.J.; Steinberg, G.R. Treatment of nonalcoholic fatty liver disease: Role of AMPK. Am. J. Physiol. Endocrinol. Metab. 2016, 311, E730–E740. [Google Scholar] [CrossRef] [Green Version]
- Abdul-Ghani, M.A.; DeFronzo, R.A. Pathogenesis of insulin resistance in skeletal muscle. J. Biomed. Biotechnol. 2010, 2010, 476279. [Google Scholar] [CrossRef] [Green Version]
- Lu, X.; Xuan, W.; Li, J.; Yao, H.; Huang, C.; Li, J. AMPK protects against alcohol-induced liver injury through UQCRC2 to up-regulate mitophagy. Autophagy 2021, 17, 3622–3643. [Google Scholar] [CrossRef]
- Garcia, D.; Hellberg, K.; Chaix, A.; Wallace, M.; Herzig, S.; Badur, M.G.; Lin, T.; Shokhirev, M.N.; Pinto, A.F.M.; Ross, D.S.; et al. Genetic liver-specific AMPK activation protects against diet-induced obesity and NAFLD. Cell Rep. 2019, 26, 192–208.e6. [Google Scholar] [CrossRef] [Green Version]
- Shackelford, D.B.; Shaw, R.J. The LKB1-AMPK pathway: Metabolism and growth control in tumour suppression. Nat. Rev. Cancer 2009, 9, 563–575. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Xu, S.; Mihaylova, M.M.; Zheng, B.; Hou, X.; Jiang, B.; Park, O.; Luo, Z.; Lefai, E.; Shyy, J.Y.; et al. AMPK phosphorylates and inhibits SREBP activity to attenuate hepatic steatosis and atherosclerosis in diet-induced insulin-resistant mice. Cell Metab. 2011, 13, 376–388. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, Y.; Liu, X.; Han, L.; Gao, X.; Liu, E.; Wang, T. Regulation of lipid and glucose homeostasis by mango tree leaf extract is mediated by AMPK and PI3K/AKT signaling pathways. Food Chem. 2013, 141, 2896–2905. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Chen, Q.; Liu, M.Y.; Ruan, J.Y.; Yu, H.Y.; Li, J.; Wang, T. Effects of benzophenones from mango leaves on lipid metabolism. Chem. Pharm. Bull. 2019, 67, 634–639. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lim, J.; Liu, Z.; Apontes, P.; Feng, D.; Pessin, J.E.; Sauve, A.A.; Angeletti, R.H.; Chi, Y. Dual mode action of mangiferin in mouse liver under high fat diet. PLoS ONE 2014, 9, e90137. [Google Scholar] [CrossRef] [Green Version]
- Saha, S.; Rashid, K.; Sadhukhan, P.; Agarwal, N.; Sil, P.C. Attenuative role of mangiferin in oxidative stress-mediated liver dysfunction in arsenic-intoxicated murines. BioFactors 2016, 42, 515–532. [Google Scholar] [CrossRef]
- Wang, C.; Ma, C.; Gong, L.; Guo, Y.; Fu, K.; Zhang, Y.; Zhou, H.; Li, Y. Macrophage polarization and its role in liver disease. Front. Immunol. 2021, 12, 803037. [Google Scholar] [CrossRef] [PubMed]
- El-Sayyad, S.M.; Soubh, A.A.; Awad, A.S.; El-Abhar, H.S. Mangiferin protects against intestinal ischemia/reperfusion-induced liver injury: Involvement of PPAR-γ, GSK-3β and Wnt/β-catenin pathway. Eur. J. Pharmacol. 2017, 809, 80–86. [Google Scholar] [CrossRef]
- Li, S.; Tan, H.Y.; Wang, N.; Zhang, Z.J.; Lao, L.; Wong, C.W.; Feng, Y. The role of oxidative stress and antioxidants in liver diseases. Int. J. Mol. Sci. 2015, 16, 26087–26124. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, J.W.; Yang, D.; Jeong, H.; Park, I.S.; Lee, M.H.; Lim, C.W.; Kim, B. Dietary zerumbone, a sesquiterpene, ameliorates hepatotoxin-mediated acute and chronic liver injury in mice. Phytother. Res. 2019, 33, 1538–1550. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Luo, Q.; Tao, Y.; Sun, X.; Liu, C. Pharmacotherapies for drug-induced liver injury: A current literature review. Front. Pharmacol. 2021, 12, 806249. [Google Scholar] [CrossRef] [PubMed]
- Videla, L.A. Oxidative stress signaling underlying liver disease and hepatoprotective mechanisms. World J. Hepatol. 2009, 1, 72–78. [Google Scholar] [CrossRef] [PubMed]
- Seen, S. Chronic liver disease and oxidative stress—A narrative review. Expert. Rev. Gastroenterol. Hepatol. 2021, 15, 1021–1035. [Google Scholar] [CrossRef]
- Luster, M.I.; Simeonova, P.P.; Gallucci, R.; Matheson, J. Tumor necrosis factor alpha and toxicology. Crit. Rev. Toxicol. 1999, 29, 491–511. [Google Scholar] [CrossRef]
- Li, S.; Tan, H.Y.; Wang, N.; Cheung, F.; Hong, M.; Feng, Y. The potential and action mechanism of polyphenols in the treatment of liver diseases. Oxid. Med. Cell Longev. 2018, 2018, 8394818. [Google Scholar] [CrossRef] [Green Version]
- Rice-Evans, C.A.; Miller, N.J.; Paganga, G. Structure-antioxidant activity relationships of flavonoids and phenolic acids. Free. Radic. Biol. Med. 1996, 20, 933–956. [Google Scholar] [CrossRef]
- Ullah, H.; Khan, A.; Baig, M.W.; Ullah, N.; Ahmed, N.; Tipu, M.K.; Ali, H.; Khan, S. Poncirin attenuates CCL4-induced liver injury through inhibition of oxidative stress and inflammatory cytokines in mice. BMC Complement. Med. Ther. 2020, 20, 115. [Google Scholar] [CrossRef] [Green Version]
- Li, S.; Jin, S.; Chen, W.; Yu, J.; Fang, P.; Zhou, G.; Li, J.; Jin, L.; Chen, Y.; Chen, P.; et al. Mangiferin alleviates endoplasmic reticulum stress in acute liver injury by regulating the miR-20a/miR-101a-Nrf2 axis. J. Biochem. 2020, 168, 365–374. [Google Scholar] [CrossRef]
- Yang, S.; Kuang, G.; Zhang, L.; Wu, S.; Zhao, Z.; Wang, B.; Yin, X.; Gong, X.; Wan, J. Mangiferin attenuates LPS/D-GalN-induced acute liver injury by promoting HO-1 in kupffer cells. Front. Immunol. 2020, 11, 285. [Google Scholar] [CrossRef]
- Li, L.; Zhen, Y.; Chen, X.; Cao, L.; Song, J.; Liu, X.; Wang, M. Mangiferin protects DNase 2 abundance via Nrf2 activation to prevent cytosolic mtDNA accumulation during liver injury. Mol. Nutr. Food Res. 2023, e2200885. [Google Scholar] [CrossRef]
- Cotter, T.G.; Rinella, M. Nonalcoholic fatty liver disease 2020: The state of the disease. Gastroenterology 2020, 158, 1851–1864. [Google Scholar] [CrossRef] [PubMed]
- Calzadilla Bertot, L.; Adams, L.A. The natural course of non-alcoholic fatty liver disease. Int. J. Mol. Sci. 2016, 17, 774. [Google Scholar] [CrossRef] [Green Version]
- Videla, L.A.; Tapia, G.; Rodrigo, R.; Pettinelli, P.; Haim, D.; Santibañez, C.; Araya, A.V.; Smok, G.; Csendes, A.; Gutierrez, L.; et al. Liver NF-kappaB and AP-1 DNA binding in obese patients. Obesity 2009, 17, 973–979. [Google Scholar] [CrossRef] [PubMed]
- Romero-Gómez, M. Non-alcoholic steatohepatitis. Med. Clin. 2022, 159, 388–395. [Google Scholar] [CrossRef] [PubMed]
- Byrne, C.D.; Targher, G. NAFLD: A multisystem disease. J. Hepatol. 2015, 62 (Suppl. S1), S47–S64. [Google Scholar] [CrossRef] [Green Version]
- Sarkar, P.; Bhowmick, A.; Kalita, M.C.; Banu, S. Effects of resveratrol and mangiferin on PPARγ and FALDH gene expressions in adipose tissue of streptozotocin-nicotinamide-induced diabetes in rats. J. Diet. Suppl. 2019, 16, 659–675. [Google Scholar] [CrossRef]
- Singh, A.K.; Raj, V.; Keshari, A.K.; Rai, A.; Kumar, P.; Rawat, A.; Maity, B.; Kumar, D.; Prakash, A.; De, A.; et al. Isolated mangiferin and naringenin exert antidiabetic effect via PPAR(γ)/GLUT4 dual agonistic action with strong metabolic regulation. Chem. Biol. Interact. 2018, 280, 33–44. [Google Scholar] [CrossRef]
- Saleh, S.; El-Maraghy, N.; Reda, E.; Barakat, W. Modulation of diabetes and dyslipidemia in diabetic insulin-resistant rats by mangiferin: Role of adiponectin and TNF-α. An. Acad. Bras. Cienc. 2014, 86, 1935–1948. [Google Scholar] [CrossRef] [Green Version]
- Zhong, Y.; Xu, Y.; Tan, Y.; Zhang, X.; Wang, R.; Chen, D.; Wang, Z.; Zhong, X. Lipidomics of the erythrocyte membrane and network pharmacology to explore the mechanism of mangiferin from Anemarrhenae rhizoma in treating type 2 diabetes mellitus rats. J. Pharm. Biomed. Anal. 2023, 230, 115386. [Google Scholar] [CrossRef]
- Niu, Y.; Li, S.; Na, L.; Feng, R.; Liu, L.; Li, Y.; Sun, C. Mangiferin decreases plasma free fatty acids through promoting its catabolism in liver by activation of AMPK. PLoS ONE 2012, 7, e30782. [Google Scholar] [CrossRef] [Green Version]
- Noh, J.W.; Lee, H.Y.; Lee, B.C. Mangiferin ameliorates obesity-associated inflammation and autophagy in high-fat-diet-fed mice: In silico and in vivo approaches. Int. J. Mol. Sci. 2022, 23, 15329. [Google Scholar] [CrossRef]
- Guo, F.; Huang, C.; Liao, X.; Wang, Y.; He, Y.; Feng, R.; Li, Y.; Sun, C. Beneficial effects of mangiferin on hyperlipidemia in high-fat-fed hamsters. Mol. Nutr. Food Res. 2011, 55, 1809–1818. [Google Scholar] [CrossRef]
- Wang, C.; Jiang, J.D.; Wu, W.; Kong, W.J. The compound of mangiferin-berberine salt has potent activities in modulating lipid and glucose metabolisms in hepG2 cells. Biomed. Res. Int. 2016, 2016, 8753436. [Google Scholar] [CrossRef] [Green Version]
- Pimpin, L.; Cortez-Pinto, H.; Negro, F.; Corbould, E.; Lazarus, J.V.; Webber, L.; Sheron, N. Burden of liver disease in Europe: Epidemiology and analysis of risk factors to identify prevention policies. J. Hepatol. 2018, 69, 718–735. [Google Scholar] [CrossRef] [PubMed]
- Han, J.; Yi, J.; Liang, F.; Jiang, B.; Xiao, Y.; Gao, S.; Yang, N.; Hu, H.; Xie, W.F.; Chen, W. X-3, a mangiferin derivative, stimulates AMP-activated protein kinase and reduces hyperglycemia and obesity in db/db mice. Mol. Cell Endocrinol. 2015, 405, 63–73. [Google Scholar] [CrossRef] [PubMed]
- Toledo, R.C.L.; Brito, L.F.; Caetano, M.M.M.; Nakajima, V.M.; da Silva, B.P.; Soares, F.E.F.; Martino, H.S.D.; de Queiroz, J.H. Acute treatment with mangifera indica L. leaf extract attenuates liver inflammation in rats fed a cafeteria diet. Food Funct. 2019, 10, 4861–4867. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Zhu, Y.Y.; Wang, L.; Teng, T.; Zhou, M.; Wang, S.G.; Tian, Y.Z.; Du, L.; Yin, X.X.; Sun, Y. Mangiferin ameliorates fatty liver via modulation of autophagy and inflammation in high-fat-diet induced mice. Biomed. Pharmacother. 2017, 96, 328–335. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Liu, M.; Yu, H.; Wang, W.; Han, L.; Chen, Q.; Ruan, J.; Wen, S.; Zhang, Y.; Wang, T. Mangiferin improves hepatic lipid metabolism mainly through its metabolite-norathyriol by modulating SIRT-1/AMPK/SREBP-1c signaling. Front. Pharmacol. 2018, 9, 201. [Google Scholar] [CrossRef]
- Singal, A.K.; Bataller, R.; Ahn, J.; Kamath, P.S.; Shah, V.H. ACG clinical guideline: Alcoholic liver disease. Am. J. Gastroenterol. 2018, 113, 175–194. [Google Scholar] [CrossRef] [PubMed]
- Osna, N.A.; Rasineni, K.; Ganesan, M.; Donohue, T.M., Jr.; Kharbanda, K.K. Pathogenesis of alcohol-associated liver disease. J. Clin. Exp. Hepatol. 2022, 12, 1492–1513. [Google Scholar] [CrossRef]
- Sun, M.; Kisseleva, T. Reversibility of liver fibrosis. Clin. Res. Hepatol. Gastroenterol. 2015, 39 (Suppl. S1), S60–S63. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bataller, R.; Brenner, D.A. Liver fibrosis. J. Clin. Investig. 2005, 115, 209–218. [Google Scholar] [CrossRef] [PubMed]
- Ge, X.; Antoine, D.J.; Lu, Y.; Arriazu, E.; Leung, T.M.; Klepper, A.L.; Branch, A.D.; Fiel, M.I.; Nieto, N. High mobility group box-1 (HMGB1) participates in the pathogenesis of alcoholic liver disease (ALD). J. Biol. Chem. 2014, 289, 22672–22691. [Google Scholar] [CrossRef] [Green Version]
- Kwon, H.J.; Won, Y.S.; Park, O.; Chang, B.; Duryee, M.J.; Thiele, G.E.; Matsumoto, A.; Singh, S.; Abdelmegeed, M.A.; Song, B.J.; et al. Aldehyde dehydrogenase 2 deficiency ameliorates alcoholic fatty liver but worsens liver inflammation and fibrosis in mice. Hepatology 2014, 60, 146–157. [Google Scholar] [CrossRef] [Green Version]
- Enomoto, N.; Ikejima, K.; Yamashina, S.; Hirose, M.; Shimizu, H.; Kitamura, T.; Takei, Y.; Sato, N.; Thurman, R.G. Kupffer cell sensitization by alcohol involves increased permeability to gut-derived endotoxin. Alcohol. Clin. Exp. Res. 2001, 25, 51S–54S. [Google Scholar] [CrossRef]
- Méndez-Sánchez, N.; Valencia-Rodríguez, A.; Coronel-Castillo, C.; Vera-Barajas, A.; Contreras-Carmona, J.; Ponciano-Rodríguez, G.; Zamora-Valdés, D. The cellular pathways of liver fibrosis in non-alcoholic steatohepatitis. Ann. Transl. Med. 2020, 8, 400. [Google Scholar] [CrossRef]
- Chidambaranathan-Reghupaty, S.; Fisher, P.B.; Sarkar, D. Hepatocellular carcinoma (HCC): Epidemiology, etiology and molecular classification. Adv. Cancer Res. 2021, 149, 1–61. [Google Scholar]
- El-Serag, H.B. Epidemiology of viral hepatitis and hepatocellular carcinoma. Gastroenterology 2012, 142, 1264–1273.e1. [Google Scholar] [CrossRef] [Green Version]
- Yang, J.D.; Hainaut, P.; Gores, G.J.; Amadou, A.; Plymoth, A.; Roberts, L.R. A global view of hepatocellular carcinoma: Trends, risk, prevention and management. Nat. Rev. Gastroenterol. Hepatol. 2019, 16, 589–604. [Google Scholar] [CrossRef] [PubMed]
- Younossi, Z.M.; Koenig, A.B.; Abdelatif, D.; Fazel, Y.; Henry, L.; Wymer, M. Global epidemiology of nonalcoholic fatty liver disease-Meta-analytic assessment of prevalence, incidence, and outcomes. Hepatology 2016, 64, 73–84. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Park, J.W.; Chen, M.; Colombo, M.; Roberts, L.R.; Schwartz, M.; Chen, P.J.; Kudo, M.; Johnson, P.; Wagner, S.; Orsini, L.S.; et al. Global patterns of hepatocellular carcinoma management from diagnosis to death: The BRIDGE Study. Liver Int. 2015, 35, 2155–2166. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arlt, V.M.; Stiborova, M.; Schmeiser, H.H. Aristolochic acid as a probable human cancer hazard in herbal remedies: A review. Mutagenesis 2002, 17, 265–277. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, G.; Shang, X.; Cui, G.; Zhao, L.; Zhao, H.; Wang, N. Mangiferin attenuated diethynitrosamine-induced hepatocellular carcinoma in sprague-dawley rats via alteration of oxidative stress and apoptotic pathway. J. Environ. Pathol. Toxicol. Oncol. 2019, 38, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Xing, X.; Li, D.; Chen, D.; Zhou, L.; Chonan, R.; Yamahara, J.; Wang, J.; Li, Y. Mangiferin treatment inhibits hepatic expression of acyl-coenzyme A: Diacylglycerol acyltransferase-2 in fructose-fed spontaneously hypertensive rats: A link to amelioration of fatty liver. Toxicol. Appl. Pharmacol. 2014, 280, 207–215. [Google Scholar] [CrossRef]
| Compounds | Model | Cell Lines/Animals | Mechanism | Reference |
|---|---|---|---|---|
| Mangiferin | LPS and D-GalN-induced acute liver injury | BALB/c, Primary hepatocytes | Nrf2↑ NLRP3↓ ASC↓ Caspase-1↓ IL-1β↓ HO-1↑ TNF-α↓ MCP-1↓ ROS↓ | [45] |
| Mangiferin | Against arsenic (As)-induced oxidative damages in the murine liver | Wistar rats, HepG2 cells | ROS↓ Caspase↓ AKT, Nrf2↑ PI3K↓ ERK1/2↓ | [68] |
| Mangiferin | Mesenteric ischemia/reperfusion (I/R)-induced liver injury | Wistar rats | β-catenin↑ PPAR-γ↑ GSK-3β↓ NF-κB-p65↓ IL-6↓ Bcl-2↑ IL-1β↓ Caspase-3↓ | [70] |
| Mangiferin | LPS/D-GalN-induced acute liver injury mouse model | BALB/c | HO-1↑ TNF-α↓ TLR4/NF-κB↓ | [78] |
| Mangiferin | CCl4-injured liver model, BDL | C57BL/6J, knockdown of Nrf2 C57BL/6J, Primary hepatocytes | Nrf2↑ HO-1↑ NQO1↑ TNF-α↓ IL-1β↓ IL-6↓ ROS↓ mtDNA↓ NF-κB↓ TLR9/MyD88↓ | [82] |
| Mangiferin | LPS/D-GalN-induced acute liver injury mouse model | BALB/c, Primary hepatocytes | miR-20a↓ miR-101a↓ Nrf2↑ | [80] |
| Mangiferin | High-fat diet (HFD) (NAFLD) | C57BL/6J, HepG2 cells | AMPK↑ NLRP3↓ Caspase-1↓ IL-1β↓ | [54] |
| Mangiferin | HFD (NAFLD) | Kunming mice | NF-κB↓ p-JNK↓ AMPK/mTOR↑ IRS/PI3K/AKT↑ | [99] |
| Mangiferin | HFD (hyperlipidemia) | C57BL6/J | Dhtkd1↑ Cox6b1↑ Scd1↓ Acac1↓ | [67] |
| Mangiferin | STZ-nicotinamide-NA-induced diabetic rats (Type 2 diabetes) | Albino Wistar rats | HbA1c↓ PPAR-γ↑ FALDH↑ | [88] |
| Mangiferin-berberine (MB) salt | OA induced HepG2 cells | HepG2 cells | p-AMPK𝛼 (Thr172)/p-ACC (Ser79) ↑ CPT1↑ | [95] |
| Isolated from the leaves of Salacia oblonga | STZ-induced diabetes rat | Albino rats | PPAR-γ/GLUT4↑ | [89] |
| Mangiferin | HFD-Fr-STZ (type 2 diabetes) | Wistar rats | TNF-α↓ PPAR-α↑ SREBP-1↓ AMPK↑ MTP↓ GSK3β↑ | [90] |
| Mangiferin | T2DM model rats, IR model of HepG2 cells | SD rats, HepG2 cells | PPAR-γ↑ GLUT4↑ AMPK↑ TNF-α↓ NF-κB↓ NLRP3↓ COX2↓ MAPK IL-6↓ IL-1β↓ NO↓ PI3K/AKT↑ | [91] |
| Mangiferin | HFD, oleic acid (lipid metabolism) | Wistar rats, HepG2 cells | DGAT↓ CPT1↑ CD36↑ ACC↓ AMPK↑ | [92] |
| Mangiferin | HFD (obesity and glucose metabolism) | C57BL/6 | F4/80↓ TNF-α↓ FGF21↑ ATG7↑ JNK↑ IL-6 | [93] |
| Mangiferin | HFD (hyperlipidemia) | hamsters | CPT1↑ PPAR-α↑ CD36↑ ACC↓ SREBP-1↓ MTP↓ DGAT2↓ | [94] |
| X-3, a derivative of mangiferin, | db/db (C57BL/KsJ), db/ (+) (C57BL/KsJ) mice, | C57BL/6J, Murine 3T3-L1 preadipocytes, LKB1-deficient Hela cells, and HEK293 cells | AMPK↑ AKT↑ ACC↑ | [97] |
| Mangiferin | HFD (liver inflammation caused by fat-rich diets) | Wistar rats | PPAR-α↑ HSP27↑ p65↓ IκB↓ TNF-α↓ IL-1β↓ iNOS↓ IL-10↓ | [98] |
| Mangiferin | Gavaged with a single dose of ethanol | C57BL/6 | PDE3B↑ NF-κB↓ AMPK/TBK1↑ CD36↓ FATP2↓ FATP5↓ ATGL↓ SREBP-1↓ p65↓ p100↓ ULK1↑ | [8] |
| Mangiferin | Alcohol hepatitis (AH) rats | Sprague–Dawley rats | PPAR-γ↑ OPA-1↑ Cav-1↑ EB1↓ NF-κB↓ p65↓ NLRP3↓ Cas-1↓ IL-1β↓ HSP90↓ HMGB1↓ SYK↓ CCL20↓ C-CAS-3↓ C-PARP↓ STARD1↓ | [55] |
| Mangiferin | CCl4-injured liver model | C57BL/6J | IL-6 IL-1β↓ α-SMA↓ TGF-β↓ MMP-2↓ ABCB4↓ ABCB11↓ SULT2A1↓ p-IκB↓ p-p65↓ NF-κB↓ | [9] |
| Mangiferin | CCl4-injured liver model | rats | HSP27↓ JAK/STAT↓ Smad/TGF-β↓ | [33] |
| Mangiferin | D-GAL induced hepatic pathophysiology | Swiss albino male rats | TNF-α↓ IFN-γ↓ IL-1β↓ IL-6↓ IL-12↓ IL-18↓ IL-10↓ iNOS↓ NF-κB↓ | [44] |
| Mangiferin | Orthotopic HCC implantation murine model, | MHCC97L Cells, BALB/c-nu/nu, | WTI↓ LEF1↓ MYC↓ Axcin2↓ MMP2↓ CCND1↓ | [10] |
| Mangiferin | DEN-induced hepatocellular carcinoma | Sprague Dawley rats | ROS↓ Bcl-2↓ Bax↑ Caspase-3↑ 8-OHdG↓ NF-κB | [115] |
| Mangiferin | APAP-induced hepatotoxicity | C57BL/6 | TNF-α↓ IL-6↓ MCP-1↓ CXCL-1↓ CXCL-2↓ IL-1β↓ NF-κB↓ AMPK↑ | [21] |
| Mangiferin | Galactosamine-induced hepatic toxicity | SHRs/NCrlVr | DGAT↓ CPT1↑ PPAR-α↑ CD36↑ MTTP↓ | [116] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, L.; Dong, Y.; Liu, X.; Wang, M. Mangiferin for the Management of Liver Diseases: A Review. Foods 2023, 12, 2469. https://doi.org/10.3390/foods12132469
Li L, Dong Y, Liu X, Wang M. Mangiferin for the Management of Liver Diseases: A Review. Foods. 2023; 12(13):2469. https://doi.org/10.3390/foods12132469
Chicago/Turabian StyleLi, Lisi, Yujia Dong, Xifu Liu, and Meng Wang. 2023. "Mangiferin for the Management of Liver Diseases: A Review" Foods 12, no. 13: 2469. https://doi.org/10.3390/foods12132469





