1. Introduction
Of the thousands of edible plant species known on Earth, only 150 to 200 species are utilized by humans, both for direct consumption and to produce food ingredients such as oils, proteins, spices, etc. [
1]. Many plants with food potential have yet to be developed and are known as “non-conventional edible plants” (NCEPs). Recently, these NCEPs are becoming increasingly important, especially considering that the demand for food supplies will increase dramatically due to population growth.
Cinnamomum camphora (L.) Presl, the plant of
Lauraceae, commonly known as camphor tree, is widely distributed in East Asia [
2]. In particular, as a massive broad-leaved evergreen tree with a broad sweeping crown,
C. camphora is widely cultivated as a garden tree and street tree in the southern region of the Yangtze River in China [
3].
C. camphora has long been known for its great potential applications in the food industry for the essential oils extracted from its roots, barks, branches, leaves and fruits [
4,
5,
6,
7]. Additionally, it was reported that the annual yield of
C. camphora seeds in China was over one million tons [
3]. Specifically, the hot-pressed oil from
C. camphora seeds has been used as a cooking oil in China’s Anfu County since the 1960s [
8]. Therefore, it is of great significance to explore
C. camphora seeds as sustainable and efficient alternative food sources.
C. camphora seeds comprise an outer shell and a kernel. In recent years, there has been a growing industry interest in
C. camphora seed kernels (CCSKs) due to their excellent pharmacological and nutritional properties. The high nutritional value of CCSKs is attributed to its high content of oil (59.34%), proteins (19.34%), dietary fibers (10.91%) and phenolics (0.97%) [
3]. In particular, CCSK oil is rich in fatty acids with 8–12 carbon chains (more than 95%), significantly higher than those of coconut oil (62%) and palm kernel oil (55%). Also, CCSK oil has been found to be safe and regulate glucose and lipid metabolism [
8,
9,
10]. In this respect, the oil has the potential for use in structured lipids and pharmaceuticals. Moreover, the protein isolated from CCSK has been reported to have comparable nutritional value to soybean protein as it contains sufficient essential amino acids (EAA) [
3]. The EAA in CCSK protein isolate meet the recommendations of the FAO and WHO for adults. Thus, CCSKs may be an excellent alternative source of plant protein. CCSKs are also a valuable source of phytochemical constituents, especially phenolics and flavonoids. An 80% ethanol extract from CCSKs exhibited various potential bioactive properties, including antioxidant, anti-diabetic, anti-obesity, anti-cholinergic and anti-inflammatory activities [
2,
11].
It should be mentioned that the chemical profiles of plants depend on many factors, including degree of maturity, growing location and weather conditions. For example, Liu et al. [
12] reported that the yield of peony seeds and the content of the main fatty acids of peony seed oil significantly depended on the cultivation areas. Similarly, Chinese black truffle collected from different geographical regions showed varying degrees of nutritional value and biological activity [
13].
C. camphora is mainly cultivated in China, including fourteen provinces (Sichuan, Yunnan, Guizhou, Chongqing, Guangxi, Hunan, Guangdong, Hubei, Anhui, Jiangxi, Fujian, Jiangsu, Zhejiang, Shanghai). Although there are previous studies describing the chemical compositions and bioactivities of CCSK, there is limited information on the samples collected from different geographical regions.
The study aimed to systematically investigate the physical characteristic (weights), chemical composition (oil, proteins, dietary fibers and ash) and nutritional value (minerals, phenolics, flavonoids, fatty acids and amino acids) of CCSK samples collected from different geographical regions in China. As a growing trend in the recycling of nutritional food ingredients from plants, this study will improve the chemical profiles and nutritional value of CCSKs and promote their effective utilization.
2. Materials and Methods
2.1. Collection of Plant Material
In China, the maturity of
C. camphora fruits is the highest in December, and the amount of fallen fruits is also the largest. Hence, fourteen samples of
C. camphora fruit were collected from the Yangtze River basin and its southern region in China from 1–31 December 2021. The geographical locations of the studied accessions are shown in
Table 1. After being taken to the laboratory, the
C. camphora fruits were manually cleaned, then all flesh was removed and dried at 35 °C for 48 h. Then, the seed shell was removed to obtain the CCSK and stored at 4 °C.
2.2. Chemical Reagents
Folin–Ciocalteu was purchased from Sigma-Aldrich Trading Co., Ltd. (Shanghai, China). Thirty-seven fatty acid methyl ester mixed standards were purchased from Shanghai Anpel Experimental Technology Co., Ltd. (Shanghai, China). Isopropyl alcohol, acetonitrile and methanol (HPLC-grade) were purchased from Thermo Fisher Technology Co. (Shanghai, China). All other solvents and reagents used were of analytical grade.
2.3. Measurement of Fruit, Seed and Kernel Weights
The weight of 1000 fruits (wet, W1) of the sample was immediately measured after being taken to the laboratory. The weight of 1000 seeds (wet, W2) was measured after removal of flesh and then the weight of 1000 seeds (dry, W3) was measured after dried. After the dry seed shell was removed, the weight of 1000 kernels (dry, W4) was obtained. Based on the above, the seed rate of the fruit (wet, R1) and kernel rate of the seed (dry, R2) were calculated [
14].
2.4. Determination of Proximate Composition of CCSK
The proximate composition of CCSK, including oil, protein, dietary fiber, moisture and ash, was determined by using a series of Chinese National Standards. Lipid content was measured by using Soxhlet extraction (GB 5009.6—2016). Protein content was determined using the Kjeldahl method (N × 6.25) (GB 5009.5—2016). Dietary fiber content was determined using enzymatic hydrolysis, followed by the weighting method (GB 5009.88—2016). Moisture content was determined by using an HX204 Moisture Analyzer (METTLER TOLEDO, Zurich, Switzerland) according to GB 5009.3—2016. Ash content was evaluated via incineration of the sample at 550 °C for 4 h (GB 5009.4—2016).
2.5. Determination of Minerals and Heavy Metals of CCSK
The minerals and heavy metals in CCSKs were determined according to Chinese National Standard GB 5009.268—2016 by using an inductively coupled plasma optical emission spectrometer (ICP-OES 725ES, Agilent, Santa Clara, CA, USA). Prior to analysis, the samples were decomposed with 8 mL of nitric acid in a Microwave Digestion System (MARS6, CEM, Charlotte, NC, USA) for 1 h. Then, the digestion solution was transferred into a 100 mL volumetric flask, diluted with ddH2O to the volume. The main parameters of ICP-OES were set as follows: radio-frequency power, 1.20 KW; plasma gas flow, 15.0 L/min; auxiliary gas flow, 1.5 L/min; observed height, 10 mm; atomizer pressure, 240 KPa.
2.6. Determination of Phytochemical Constituents of CCSKs
2.6.1. Preparation of Phytochemical Extracts
The extracts were prepared by mixing the defatted CCSKs with 80% aqueous–ethanol solution at a ratio of 1:20 (
w/
v) [
3]. The mixture was stirred at room temperature for 2 h with the speed of 300 rpm and then filtered with an aspirator filter pump. The residue was re-extracted 5 times by using the above procedure. Both extracts were combined and stored at 4 °C for further analysis.
2.6.2. Total Phenolic Content (TPC)
Folin–Ciocalteu method was used to determine the TPC [
2]. Briefly, a 40 μL aliquot of the extract was mixed with 20 μL of Folin–Ciocalteu reagent and reacted at room temperature for 6 min. Then, 200 μL of 7% Na
2CO
3 was added to neutralize the reaction. After incubation at room temperature in the dark for 1 h, the absorbance value was measured at 760 nm using a microplate reader (ReadMax 1200, Flicker Biotechnology Co., Ltd., Shanghai, China). Results were expressed as gram of gallic acid equivalent (GAE) per 100 g of CCSK (g GAE/100 g).
2.6.3. Total Flavonoid Content (TFC)
TFC was determined using the method of Tian et al. [
15]. Briefly, a 2.5 mL aliquot of the extract was mixed with 300 μL of NaNO
2 (5%) and incubated at room temperature for 6 min. Then, 300 μL of AlCl
3 (5%) was added and incubated at room temperature for 6 min, followed by mixing with 4.4 mL of NaOH (4%), and incubation for 10 min. The absorbance value was measured at 510 nm. Results were expressed as gram of rutin equivalent (RE) per 100 g of CCSK (g RE/100 g).
2.6.4. HPLC Profile
The Agilent 1260 HPLC system combined with a UV detector was used to analyze 80% ethanol extracts of CCSKs [
16]. Prior to analysis, the extract solutions were filtered through 0.45 mm membrane filters. Samples (10 μL) were injected onto an amethyst (Sepax Technology Co., Ltd., Newark, DE, USA) C18-H reverse-phase column (250 mm × 4.6 mm × 5 μm) at 30 °C. The mobile phases consisted of 0.1% (
v/
v) formic acid in water (A) and acetonitrile (B). The gradient conditions were as follows: 15–40% B at 0–20 min; 40–50% B at 20–25 min; 50–95% B at 25–28 min; 95–15% B at 28–30 min. The flow rate was 1.0 mL/min and the absorbance was recorded at 280 nm.
2.7. Determination of In Vitro Antioxidant Activity
The DPPH free-radical-scavenging method was used to determine antioxidant activity [
17]. The extract solution (150 μL, 1 mg/mL) was mixed with DPPH solution (150 μL, 0.1 mmol/L in 95% ethanol) and incubated in dark at room temperature for 30 min. The absorbance was measured at 517 nm by using a microplate reader (ReadMax 1200, Flicker Biotechnology Co., Ltd., Shanghai, China). The results were expressed as μmol of Trolox equivalent (TE) per gram of extract (μmol TE/g extract).
2.8. Determination of Fatty Acid Composition in Kernel Oil of CCSKs
Methyl esterification of the kernel oil was analyzed using an Agilent 7890B gas chromatograph with a DB-23 column (60 m × 250 μm × 0.25 μm, Agilent, Santa Clara, CA, USA) [
18]. The major parameters were set as follows: carrier gas, nitrogen; flow rate, 2 mL/min; injection temperature, 250 °C; detection temperature, 250 °C. The column temperature was first kept at 50 °C for 1 min, increased to 175 °C at 20 °C/min and kept at 175 °C for 5 min, increased from to 230 °C at 4 °C/min and then kept at 230 °C for 10 min. The fatty acid profiles of the kernel oil were obtained by comparing the gas chromatograms of samples with thirty-seven fatty acid methyl ester mixed standards (Anpel, Shanghai, China) using normalization method.
2.9. Determination of Amino Acid Composition in Kernel Protein of CCSKs
The amino acid composition of CCSKs was determined using an amino acid auto-analyzer (S-433D, Sykam GmbH, Eresing, Germany) [
19]. Prior to analysis, 0.1 g aliquot of each sample was mixed with 10 mL of HCl (6.0 M) and 1.0 g phenol in sealed glass tubes at 110 °C for 24 h. The hydrolyzed samples were filtered, placed in 50 mL volumetric flasks and diluted with ddH
2O to the volume. The hydrolyzed sample (1 mL) was transferred to a glass dish and evaporated at 60 °C. Then, 4 mL of sample dilution buffer (0.12 N, pH 2.2) was added and filtered through a 0.22 μm PTFE filter.
2.10. Protein Profiles
The kernel protein extraction was carried out based on a previous study [
3]. Sodium dodecyl sulphate–polyacrylamide gel electrophoresis (SDS-PAGE) under reducing condition was performed.
2.11. Statistical Analysis
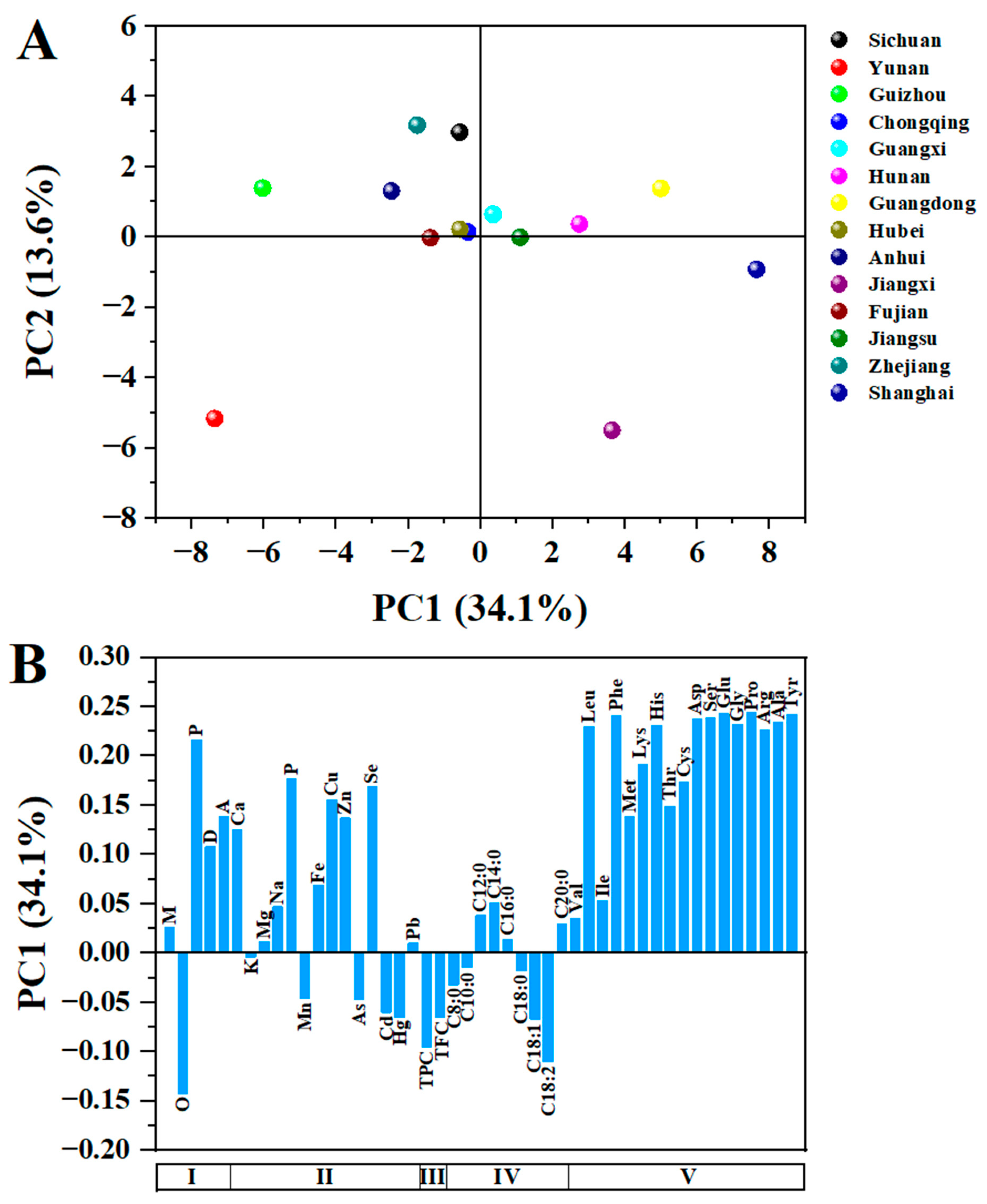
All experiments were conducted in triplicate. Data were expressed as means ± standard deviation (SD) and subjected to one-way analysis of variance (ANOVA), followed by Tukey’s test for comparison of means (p < 0.05) using SPSS software. Principal component analysis (PCA) was performed in Origin 2023 software.
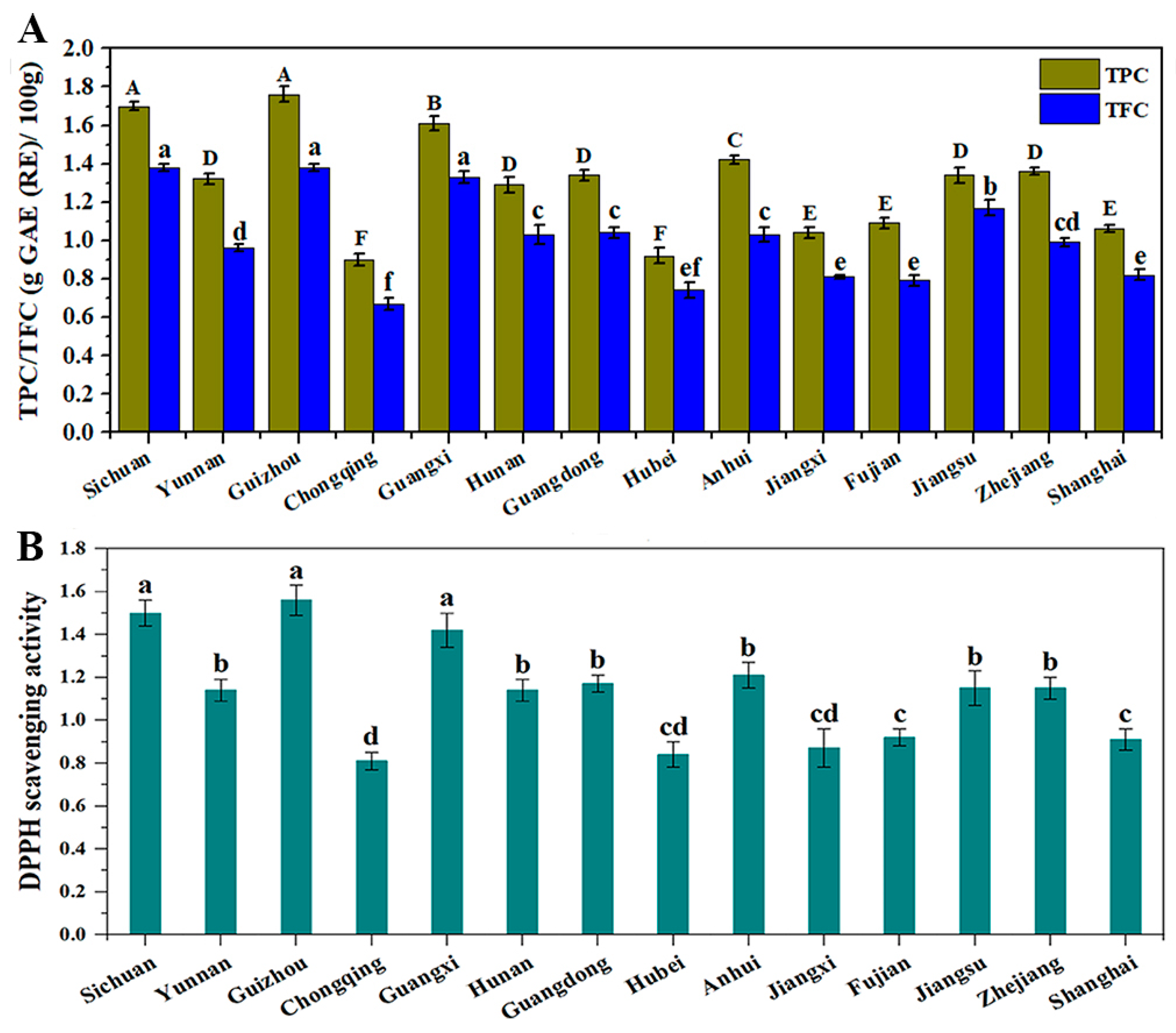
4. Conclusions
In this study, the physical characteristics, chemical compositions and nutritional values of fourteen CCSK samples collected from different geographical regions in China were investigated. The results showed that the weights (1000 fruits, 1000 seeds and 1000 kernels), proximate compositions, minerals, phenolics, flavonoids and amino acid contents were significantly affected by geographical factors, such as average altitude and weather conditions. Proximate composition analysis found that CCSKs had the high contents of oil (46.29–62.08%), protein (12.33–22.17%) and dietary fiber (5.66–8.03%), among which Chongqing, Shanghai and Fujian had the highest contents of oil, protein and dietary fiber, respectively. The main minerals in the CCSK samples were K (4345.05–7186.89 mg/kg), P (2735.86–5385.36 mg/kg), Ca (1412.27–3327.37 mg/kg) and Mg (2028.65–3147.32 mg/kg). The highest TPC and TFC values were found in Guizhou (1.76 g GAE/100 g and 1.38 g RE/100 g, respectively). The ratio of essential amino acids to total amino acids in the CCSK sample from Guizhou was the highest (36.20%). The antioxidant activity varied greatly with the sample location, and was highly correlated with TPC and TFC. The results suggested that CCSKs were rich in phytochemical components and had high nutritional value, and may be suitable to be used as ingredients for the food industry. However, given the heavy metal content in CCSKs, great caution should be exercised.