3.1. Protein Content
Cricket powder as a raw material was analyzed for its protein and lipid content, which were found to be 71.2% and 10.9%, respectively. The protein content in this study was slightly higher than that in studies by Udomsil et al. [
31] and Stone et al. [
33], who reported a protein content of crickets of around 60–70%. This may be because the samples in the present study were from cricket powder, the production of which involved removing small parts containing less protein, such as legs and wings, before grinding and sieving. However, Stone et al. [
33] reported a higher fat content, 16.1%, compared with the 10.91% found in this study. Crickets are known to be an alternative protein source with high fat content [
31]. The high fat content could possibly inhibit the extraction of protein and generate an off-flavor [
6].
In the present study, the defatting process was carried out before extraction using the AEAP method and isolation with ammonium sulfate added to optimize the protein content. The protein content of cricket powder, defatted cricket powder, cricket protein extracted using AEAP, and cricket protein extracted with various concentrations of ammonium sulfate (0%, 20%, 40%, and 60%,
w/
v) is shown in
Table 1. The protein content in this study was calculated using 6.25 as Kp and reported as crude protein content. In fact, previous reports indicated that overestimation of cricket protein content could occur if a Kp value of 6.25 is used, due to the presence of nonprotein nitrogens like phospholipids, chitin, nucleic acids, and ammonia [
17,
34,
35]. However, the recommended values (5.09 to 5.60) were not in agreement based on the total true protein from amino acid content and total nitrogen in each sample [
20,
35,
36]. Therefore, the Kp value of 6.25 was used in the present study for comparison with other studies that used this value to calculate cricket protein content [
3,
19,
31,
33,
37,
38,
39,
40].
It was found that the protein content increased after the defatting, extraction, and isolation steps. In general, some lipids that bind to proteins may inhibit protein extraction [
41]. Therefore, the defatting process would help to remove lipids and enhance the protein content after extraction [
6]. The alkaline extraction–acid precipitation (AEAP) process increased the protein content from approximately 78% to 82%. This occurred due to the utilization of sodium hydroxide, which can increase protein solubility and separate protein from other components, such as fat [
42]. In addition, hydrochloric acid was used to bring the protein back to its isoelectric point, where it has a net charge of 0, and thus it was precipitated, thereby increasing the protein extraction efficiency [
42]. The protein content of the AEAP sample was higher compared with the cricket protein concentration reported by Ndiritu et al. [
19] and the cricket protein hydrolysate by Hall et al. [
3].
The combination of AEAP with the inclusion of various concentrations of ammonium sulfate showed a positive trend by enhancing the content of cricket protein (
Table 1). In addition, there was no significant difference (
p > 0.05) between AEAP cricket protein powder and that with 20% (
w/
v) ammonium sulfate (20 AS). However, there was a significant difference between the samples extracted with 40% and 60% ammonium sulfate (40 and 60 AS). The highest protein content of 94.0% was found in the 60 AS sample, which could be classified as cricket protein isolate (CPI) since it exceeded 90% as the minimum requirement for protein isolate classification [
43]. The enhanced protein content with the inclusion of ammonium sulfate could be explained by the salting-in and salting-out mechanisms [
44]. Briefly, salting-in occurs when the presence of the salt ion of ammonium sulfate (NH
4+) causes an increase in protein solubility. However, a gradual addition of ammonium sulfate would cause competition between salt ions (SO
42−) and protein molecules in binding with water molecules. Ultimately, this competition could force the protein molecules to expose their hydrophobic groups, leading to aggregation and precipitation (salting-out). The increase in protein content via the inclusion of 60% ammonium sulfate in this study is in agreement with a study by Waglay et al. [
14], who reported that the inclusion of 40% to 60% ammonium sulfate successfully enhanced potato protein content from 70.1% to 98.6%, as well as a study by Zhang et al. [
45], who used 60% ammonium sulfate to extract potato protein and found up to 83.2% protein content. Moreover, another study by Ersus et al. [
46] also mentioned the effectiveness of 85% ammonium sulfate in increasing the content of protein extracted from mallow leaf from 43.5% to 61.7%.
Based on the findings of the present study, the extraction yields of the protein samples from all treatments after freeze-drying were roughly calculated at around 16% to 24% (w/w), which could be applied to produce CPI and for use in the food industry. In addition, the high CPI value obtained in this study has not been reported elsewhere. Therefore, the physicochemical and functional properties of cricket powder, defatted cricket powder, AEAP samples, and CPI samples (inclusion of 60% ammonium sulfate) were further analyzed to determine the potential applications of CPI in the food industry.
3.2. Surface Hydrophobicity and Zeta Potential
Surface hydrophobicity (H
0), an important physicochemical parameter used to determine protein characteristics, is strongly related to the functional properties of proteins, such as foaming. The H
0 and zeta potential of cricket proteins are given in
Table 2. It was found that after defatting and extracting via AEAP and AS, the H
0 of the samples increased. According to Santiago et al. [
4] and Jiang et al. [
10], heat treatment and alkaline extraction can unfold proteins and thus expose their hydrophobic regions. On the other hand, including a high concentration of salt can enhance H
0 due to the salting-out mechanism, which breaks the hydration layer on the surface and disrupts the charge balance of protein molecules. Moreover, the presence of salt ions from ammonium sulfate may promote competition with protein molecules in binding with water molecules, thus leading to aggregation of the protein and ultimately increased hydrophobicity [
47].
The zeta potential reflects the compositional and structural variations in proteins, which are related to their functional properties, such as solubility [
28]. External factors such as pH and ionic strength affect the zeta potential [
10]. As shown in
Table 2, the zeta potential of cricket powder decreased to a negative value after defatting and protein extraction. However, the zeta potential of the CPI sample (−15.30) was higher than that of the defatted (−19.00) and AEAP (−23.12) samples. Negative zeta potential values reflect the greater electrostatic repulsion and separation distance between protein molecules in a suspension [
10]. According to Zhu et al. [
26], the greater electrostatic repulsion of the AEAP sample was probably due to the alkaline extraction–acid precipitation method, which increases the availability of hydrophobic zones. The use of extreme acid and alkaline pH during AEAP causes unfolding of the protein structure due to the change in electrostatic repulsion accompanied by the breaking of disulfide bonds, the dissociation of subunits, and the exposure of more hydrophobic groups of protein compounds, ultimately leading to increased hydrophobicity and flexibility of protein molecules [
48]. The result in this study shows that the most negative zeta potential values were observed with the AEAP technique compared with other samples. In addition, it was found that the samples with ammonium sulfate added showed fewer negative zeta potential values. This may be explained by the fact that the presence of salt ions may reduce the electrostatic repulsion between protein molecules (salting-out), as mentioned above [
4].
3.3. Protein Solubility
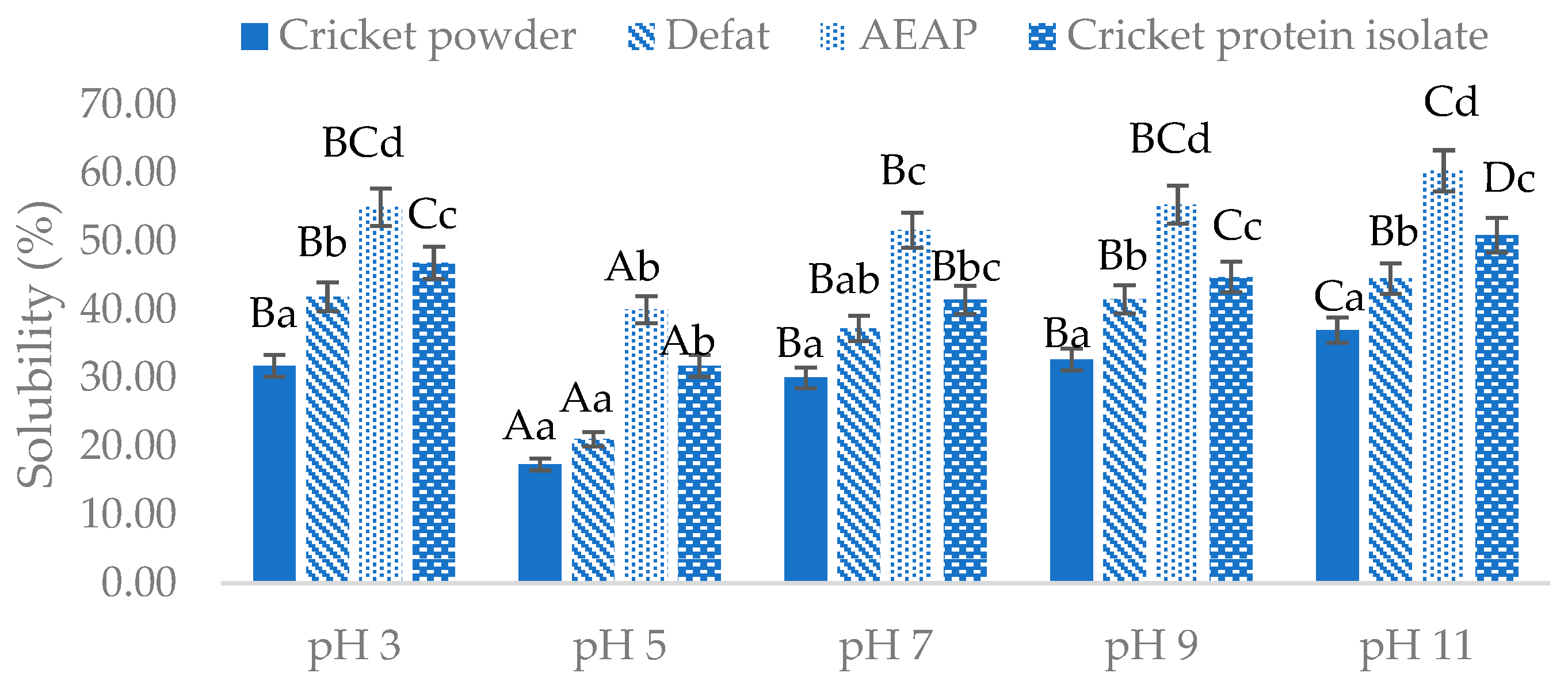
The protein solubility of cricket powder and extracted cricket protein at pH 3, 5, 7, 9, and 11 is presented in
Figure 1. The lowest solubility of all samples was found at pH 5. This result is in agreement with a study by Zielińska et al. [
27], who reported the lowest solubility was found at pH 5, while Stone et al. [
33] and Ruggeri et al. [
49] reported that the isoelectric point (pI) of cricket protein was around pH 4. Shifting the pH of the protein toward its isoelectric point would reduce the electrostatic repulsion between protein molecules and force the protein to aggregate and precipitate, resulting in lower solubility [
50]. As shown in
Figure 1, cricket protein was found to be more soluble in an alkaline than an acidic environment, as evidenced by the highest solubility for all samples being obtained at pH 11: 37.03%, 44.60%, 60.31%, and 50.94% for the cricket powder, defatted cricket powder, AEAP, and CPI samples, respectively.
Furthermore, the protein solubility results in
Figure 1 are strongly related to the zeta potential results shown in
Table 2. The change in the zeta potential of the cricket powder to a greater negative value occurred after the defatting, AEAP extraction, and isolating processes. This indicates greater electrostatic repulsion between protein molecules, resulting in better protein solubility [
51]. Another important factor that strongly impacts solubility is ionic strength. The presence of salt ions affects the solubility of proteins, as evidenced by the high salt concentration in the CPI sample decreasing the solubility of protein [
52]. This also correlates with the salting-out mechanism mentioned earlier [
44].
3.4. Secondary Structure
Circular dichroism (CD) spectroscopy is a widely used technique for determining the secondary structures of protein samples.
Table 3 displays the secondary structures of proteins in cricket powder and extracted cricket protein powder. It was found that the samples contained different α-helix, β-sheet, β-turn, and random coil structures. As shown in
Table 3, random coil was the most dominant secondary structure in all samples. The reduction in α-helix occurred after the defatting, AEAP, and isolation (addition of ammonium sulfate) steps. The reduced α-helix content reflects the breaking of intermolecular hydrogen bonds, resulting in a less ordered protein structure and elevating its flexibility [
53]. This finding was similar to that of a study by Mao et al. [
54], who reported that alkaline extraction and acid precipitation changed the secondary structure of protein by breaking its hydrogen bonds.
As shown in
Table 3, the lowest α-helix content was found in the CPI sample. This is because the addition of salt ions (ammonium sulfate) caused the rearrangement of protein molecules by exposing the buried hydrophobic region, turning them into other forms, such as β-turn and random coil [
55]. According to Zhao et al. [
56], α-helix and β-sheet are stabilized by intra- and intermolecular hydrogen bonds. Including a low concentration of ammonium sulfate in the form of cations (NH
4 +) increases the solubility and the α-helix content.
However, in the presence of a high concentration of anions (SO
42−), the hydrogen bonding between proteins and water molecules is weakened, leading to unfolding and stretching of the protein structure, thereby decreasing the α-helix content [
56]. The exposure of the hydrophobic region of the cricket protein in this study was confirmed via measuring the surface hydrophobicity, with the CPI sample showing the highest surface hydrophobicity (
Table 2). The reduction in α-helix structure due to salt ion inclusion in this study was in agreement with Sun et al. [
57], who found that adding NaCl could reduce the α-helix structure of oleosin. Furthermore, Zhang et al. [
47] reported that including 0.005 g/mL sodium chloride reduced the α-helix content by loosening the protein structure and destroying the secondary structures of mixed gels of wheat gluten protein and potato protein isolate.
3.5. Water-Holding Capacity (WHC) and Oil-Holding Capacity (OHC)
WHC is a functional property that is defined as the capability of a protein to retain water molecules [
58]. Similarly, OHC is defined as the ability of a protein to retain absorbed oil [
27]. Both of these functional properties are strongly determined by the composition, pH condition, and ionic strength of protein [
59]. The WHC and OHC of the cricket powder and extracted cricket protein powder are shown in
Table 4. A negative correlation between WHC and OHC was observed. The extraction and isolation processes reduced the WHC of the protein samples (AEAP and CPI) and positively affected the OHC, as evidenced by the elevated OHC values for the AEAP and CPI samples.
The increased WHC after defatting could be related to the increase in protein content from 71.2% to 77.6% (
Table 1) and is correlated with the removal of fat content. This results in the exposure of the hydrophilic regions of protein, which can bind with water molecules [
60]. On the other hand, the decreased WHC of the AEAP and CPI samples might be due to the change in the secondary structure of cricket protein after alkaline extraction–acid precipitation and the inclusion of ammonium sulfate. It can be explained that α-helix structure is mainly stabilized by hydrogen bonds between the carbonyl oxygen (-CO) and amino hydrogen (NH-) of the polypeptide chains, which can easily be destroyed under extreme acidic or alkaline conditions [
61]. The results of this study agree with this theory, with a decrease in α-helix content from 39.6 to 23.4 for AEAP samples and 1.0 for CPI samples. The loss of α-helix structure leads to the weakening of hydrogen bonds, thereby reducing protein–water interactions, which was reflected in the reduced water-holding capacity of the AEAP and CPI samples [
62]. Ogunwolu et al. [
63] also noted that the presence of salt ions reduces WHC, since the inclusion of a large number of salt ions promotes salting-out, ultimately resulting in dehydration and reduced WHC. The WHC of cricket powder in this study was quite similar to the WHC of freeze-dried edible cricket powder (2.03 g/g) reported by Jeong et al. [
64], while it was higher than the WHC of cashew nut protein isolate (1.75 g/g) reported by Liu et al. [
65] and lower than that of walnut protein (5 g/g) reported by Hu et al. [
66].
On the other hand, the OHC values in this study increased after defatting and extraction (
Table 4). The enhanced OHC could be explained by the increased amount of protein and its exposed hydrophobic amino acids interacting and binding with the oil. Other important factors affecting OHC were the heat treatment and the extreme pH due to the use of sodium hydroxide (alkaline) and hydrochloric acid (acidic) during extraction. These can promote partial denaturation of protein, causing the exposure of more hydrophobic amino acids to interact with the oil. The OHC of CPI (3.78 g oil/g sample) was higher compared with those in previous studies on cricket protein samples by Zielińska et al. [
8] and Damasceno et al. [
67], in which the values were 3.10 and 2.20 g oil/g, respectively. Meanwhile, the OHC in this study was lower compared with that of the 6.7 g oil/g sample of mealworm protein hydrolysates reported by Leni et al. [
50]. Furthermore, a study by Sun et al. [
57] noted that including salt ions would decrease the zeta potential of protein solution and cause the exposure of more hydrophobic groups of the protein, which would enhance its ability to entrap oil.
3.6. Emulsifying Properties
Emulsification is defined as the ability of the protein to absorb and stabilize the oil–water interface. There are many aspects involved in the formation of a protein emulsion system, such as the movement of protein from the aqueous phase to the newly formed interface and conformational rearrangement. In general, proteins that are soluble, amphiphilic, and flexible are considered to be effective emulsifiers [
68].
Table 5 gives the emulsion activity index (EAI) and emulsion stability index (ESI) of cricket powder and extracted cricket protein powder. It was found that the EAI and ESI of cricket powder increased after the defatting process. These values increased because the fat was removed, which increased the protein content. Kim et al. [
69] explained that removing the fat from the protein would increase the emulsifying properties of cricket protein. Siddiq et al. [
70] explained that the enhanced EAI and ESI after defatting was strongly correlated with the increased solubility of protein. The increased protein solubility was observed after defatting (
Figure 1), which allowed the protein molecules to prevent the coalescence of fat by constructing a protective barrier around fat droplets, thus increasing the emulsifying properties. However, it was found that the EAI and ESI of extracted protein samples (AEAP and CPI) were decreased. Ogunwolu et al. [
63] noted that increasing the protein concentration would decrease the EAI of protein, since at high concentrations, the activation energy barrier does not allow protein to migrate and it takes place in a diffusion-dependent manner.
The reduced EAI and ESI of the AEAP and CPI samples may be related to the loss of the α-helix structure of the samples. Xue et al. [
71] stated that protein denaturation can occur under extreme pH during alkaline extraction–acid precipitation, which changes the secondary structures of the protein by reducing its α-helix content. Studies conducted by Guo et al. [
72] and Yan et al. [
73] showed that the loss of α-helix structure increased surface hydrophobicity. Excessive hydrophobicity can influence the stability of particles on the surface of oil droplets, which can affect the balance of hydrophobic and hydrophilic regions in the emulsion system. This excessive hydrophobicity might be correlated with the H
0 of the AEAP (746.11) and CPI (956.22) samples, which were higher compared with that of the cricket powder (71.42). The EAI values in this study were higher than those in a study by Dion-Poulin et al. [
7], who reported values of around 11.86–13.32 m
2/g. Furthermore, the excessive salt ions being another cause of the exposure of hydrophobic regions of protein is in line with studies by Hu et al. [
66] and Lawal et al. [
74].
3.7. Foaming Properties
Foaming capacity (FC) is defined as the capability of protein molecules to be dissolved and unfolded rapidly in order to form a cohesive layer around a gas bubble, while foaming stability (FS) is the ability of protein molecules to form a stable foam by forming a continuous intermolecular polymer that traps air [
10].
Table 6 shows the FC and FS of cricket powder and extracted cricket protein powder. Interestingly, it was found that cricket powder was unable to form a foaming system. This may be explained by the fact that the fat content can interrupt protein interactions at the air–water interface and inhibit the generation of a foaming system [
58]. Thus, the defatting step is necessary to optimize the construction of a foaming system.
The results in
Table 6 show that the defatted sample could form foam, but the stability of the foam was not very good. Moreover, samples from AEAP and ammonium sulfate inclusion showed higher FC and FS compared with the defatted sample. The CPI sample showed the highest FC and had the most stable foaming system. The results for foaming properties in this study were correlated with the results for protein solubility (
Figure 1) and surface hydrophobicity (
Table 2). This can be explained by the alkaline extraction method causing partial protein unfolding, which increases the ability of protein to undergo rapid conformational changes at the interface and rapid adsorption at the air–water interface [
59]. In addition, including ammonium sulfate as salt ions may increase both FC and FS due to the exposure of hydrophilic residue and higher protein solubility during the isolation step [
10]. These findings are positively related to those of a study by Yuliana et al. [
75], who reported increasing FS of cashew nut shell protein from 56.03% to 76.91% at 60 min after the addition of salt, which increased the surface activity and solubility of the protein.
The FC and FS of CPI in this study were quite similar to those in a study by Jeong et al. [
64], who reported values of 61.1% and 73.9% (30 min), respectively, for cricket protein concentrate. However, the CPI in this study showed remarkable foaming stability. This may be correlated with the high protein content in CPI, which was more than 90%. This may have increased the stability of the foaming system, since it exhibited faster adsorption and a smaller and more homogeneously distributed bubble size, which eventually led to a more stable foaming system [
76]. Moreover, the high protein content of CPI would provide an advantage in protecting against the coalescence of bubbles due to a more stretchable interface in the foaming process [
76].