Improving the Quality and Safety of Fish Products with Edible Coatings Incorporating Piscicolin CM22 from a Psychrotolerant Carnobacterium maltaromaticum Strain
Abstract
1. Introduction
2. Materials and Methods
2.1. Bacterial Strains and Culture Conditions
2.2. Preparation of Piscicolin CM22
2.3. Enzymatic and Physicochemical Stability of Piscicolin CM22
2.4. Edible Coatings (ECs)
2.5. Physicochemical Characterization of Coatings
2.6. In Vitro Antimicrobial Activity of Edible Coatings (ECs)
2.7. Food Effectiveness Trials
2.7.1. Influence on the Shelf Life of Raw Tuna and Salmon
2.7.2. Challenge Test in Food
2.8. Statistical Analyses
3. Results
3.1. In Vitro Trials
3.2. Shelf-Life Trials
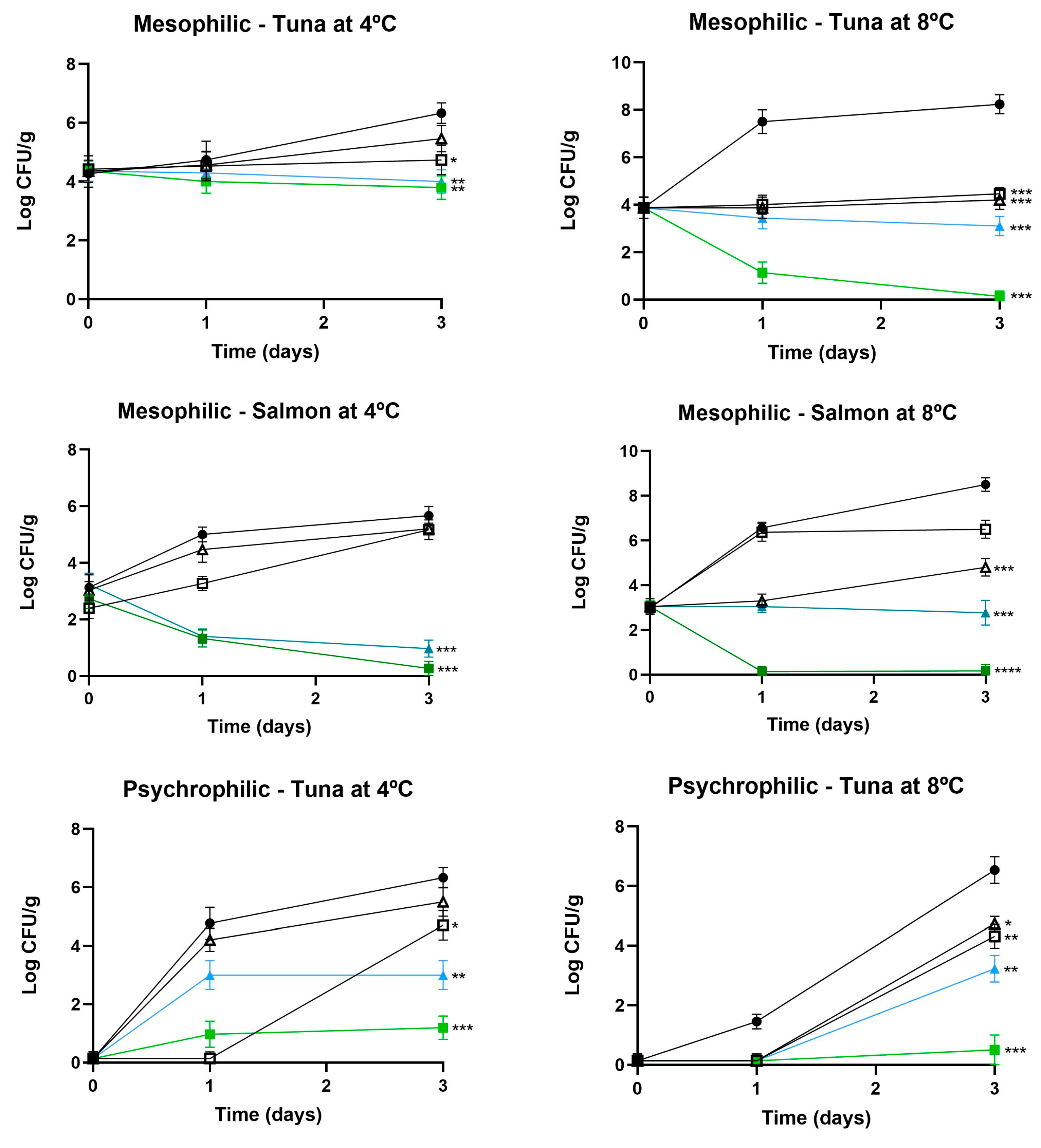
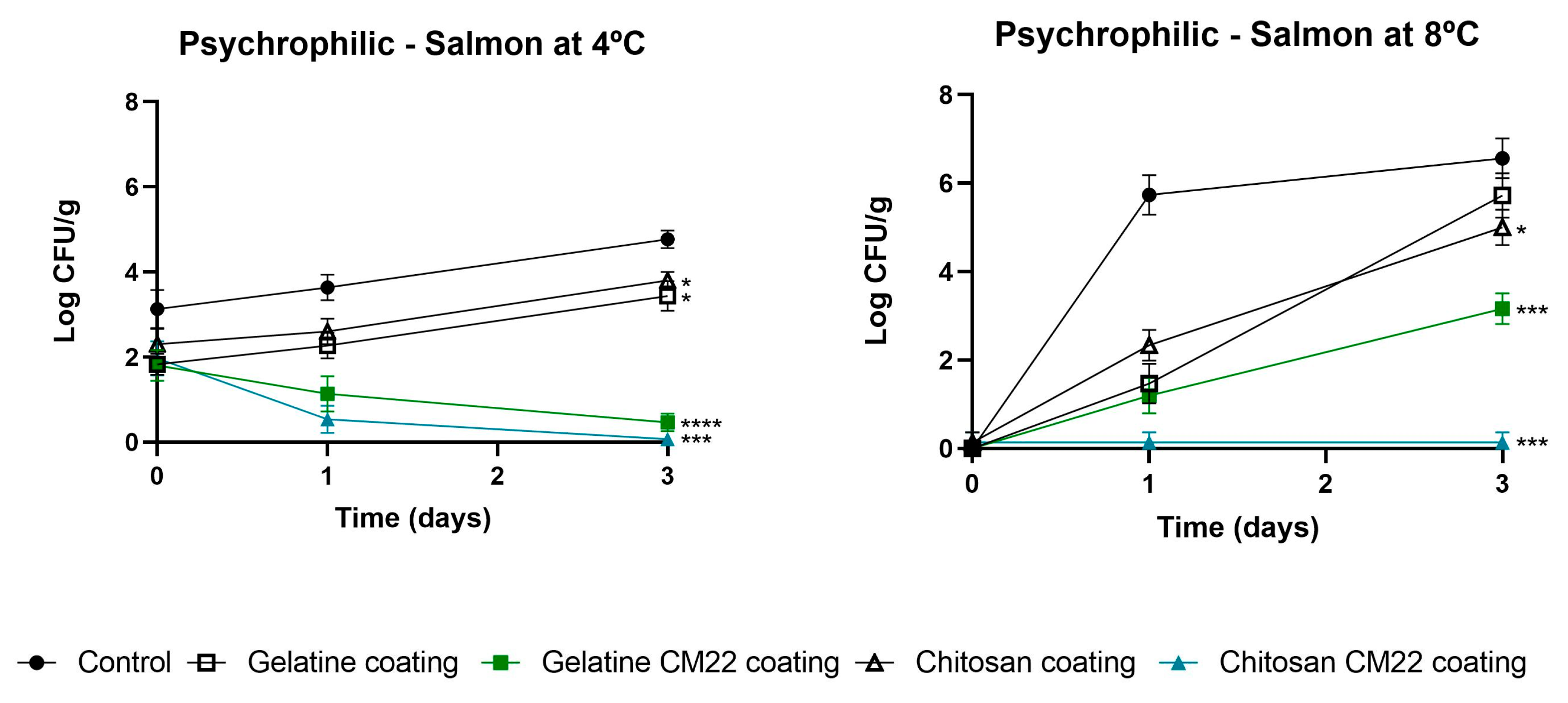
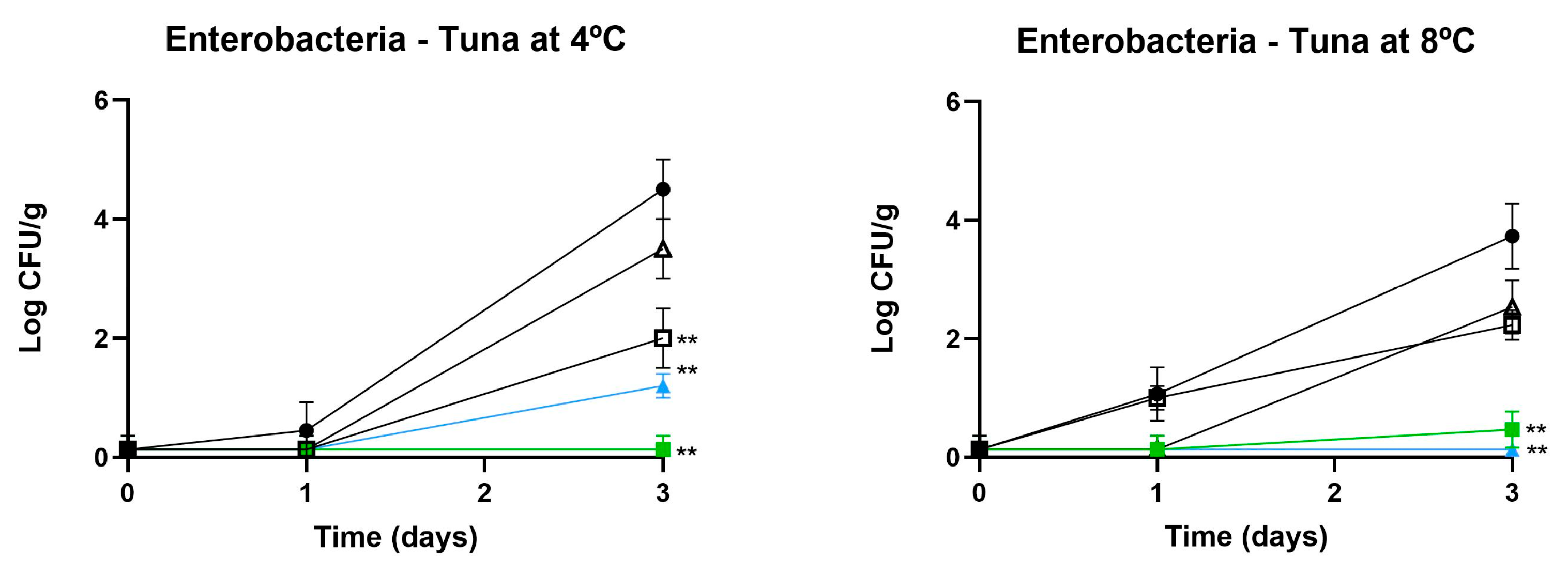
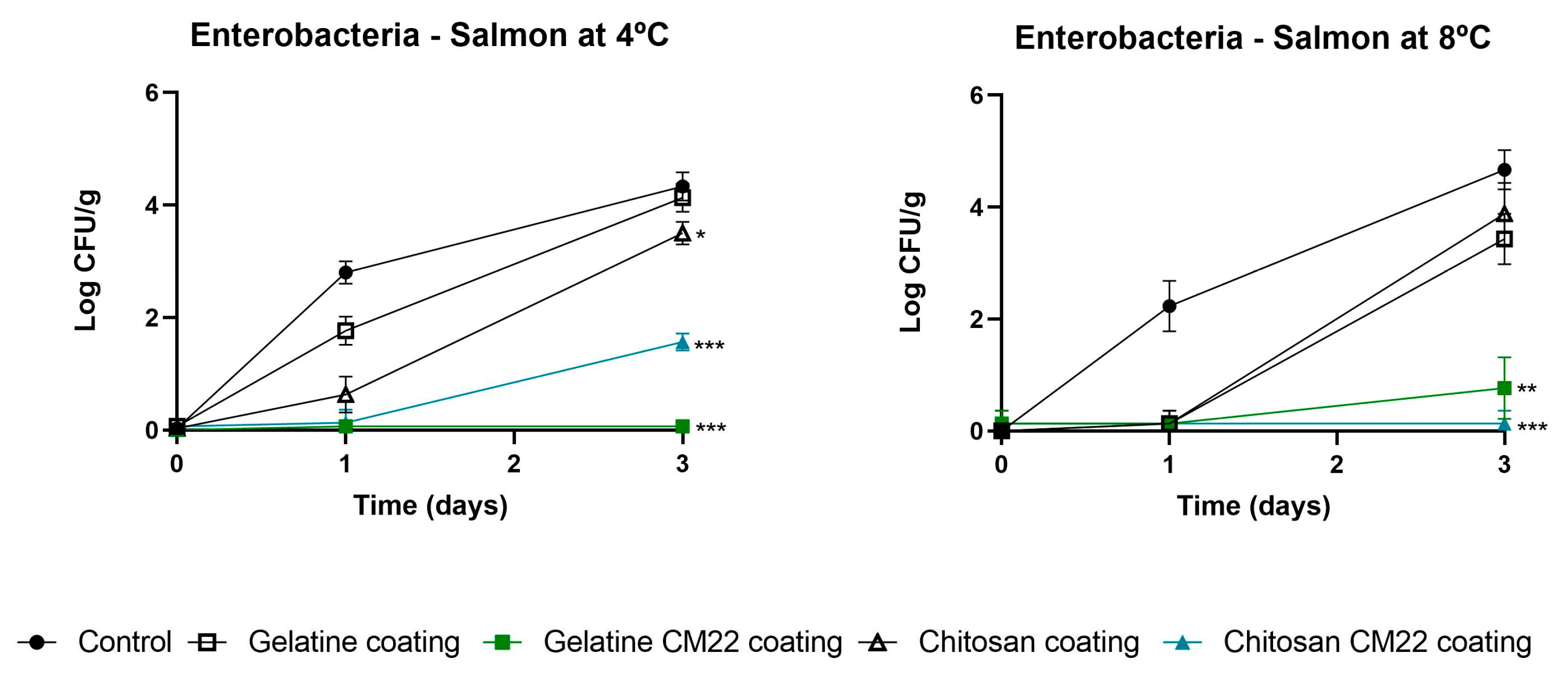
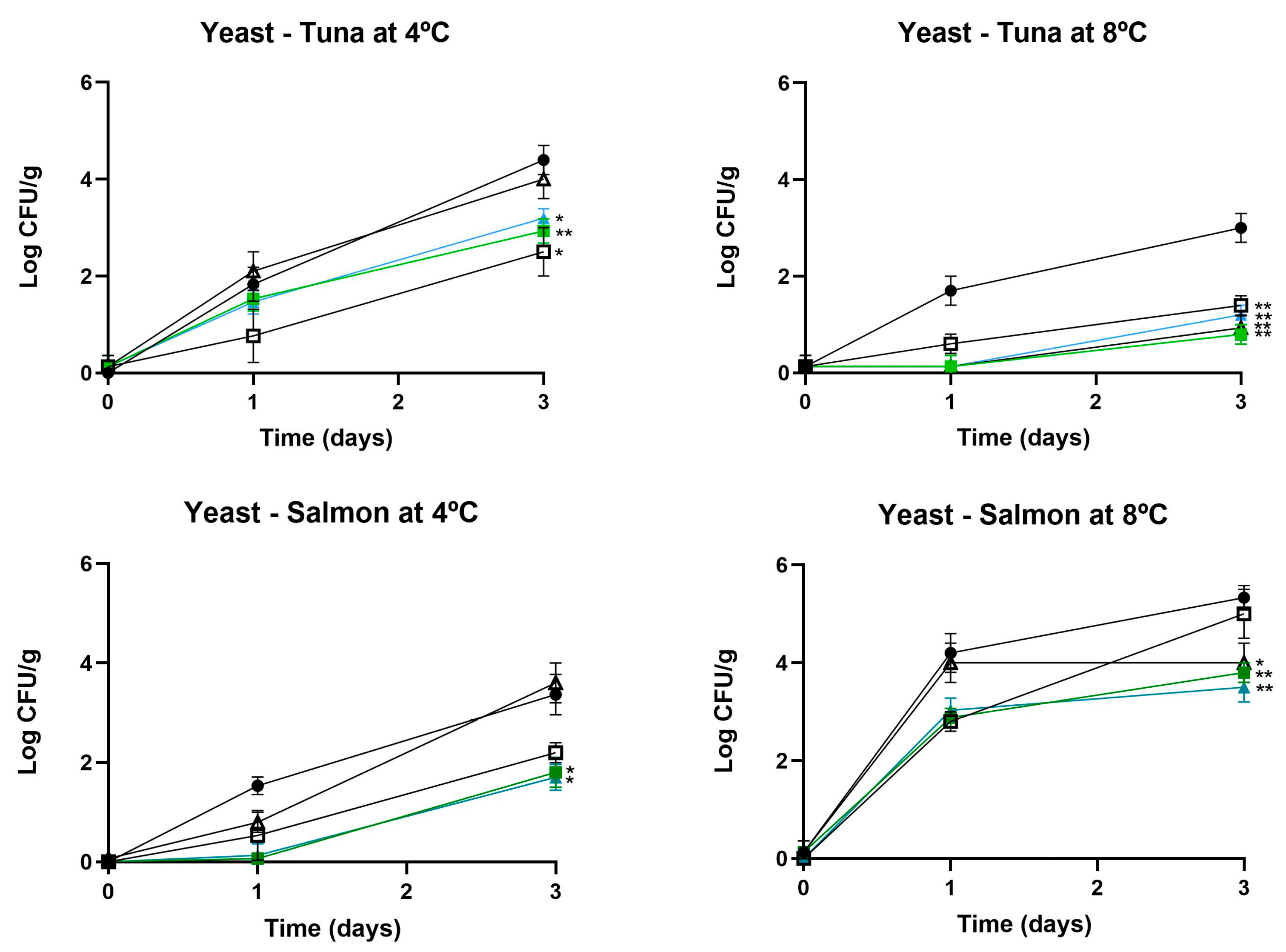
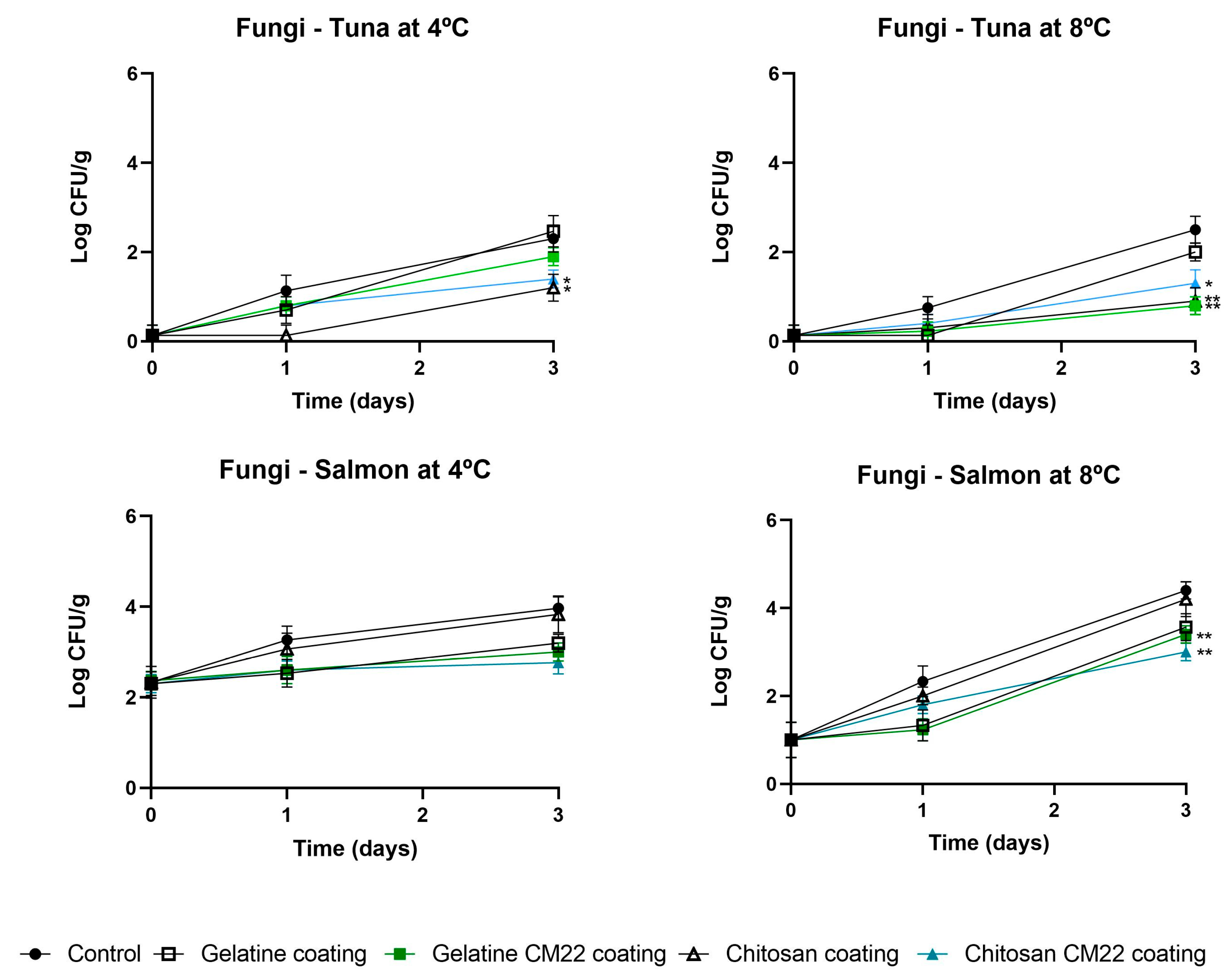
3.2.1. Influence on Microbial Counts
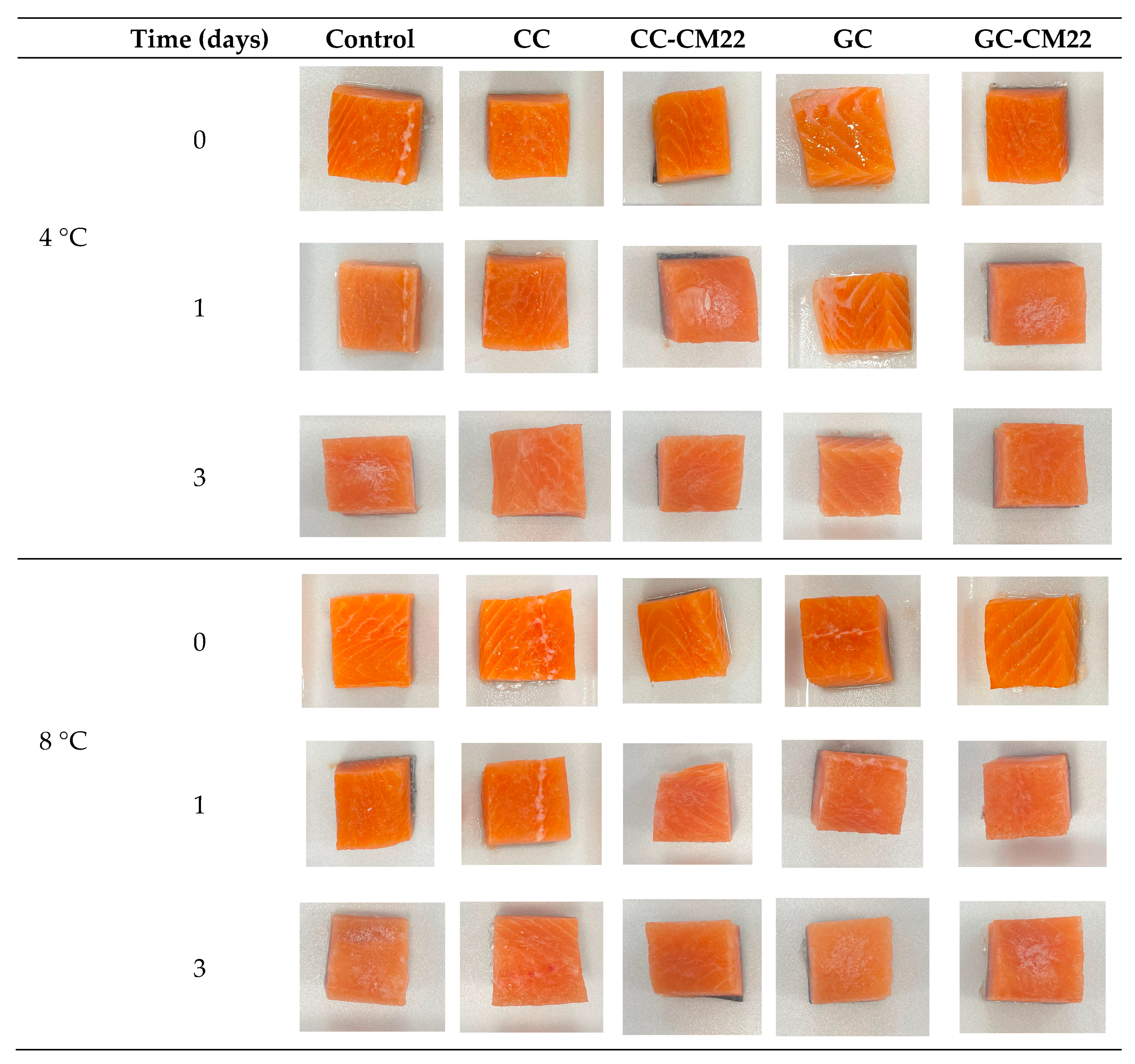
3.2.2. Influence in Technological Properties
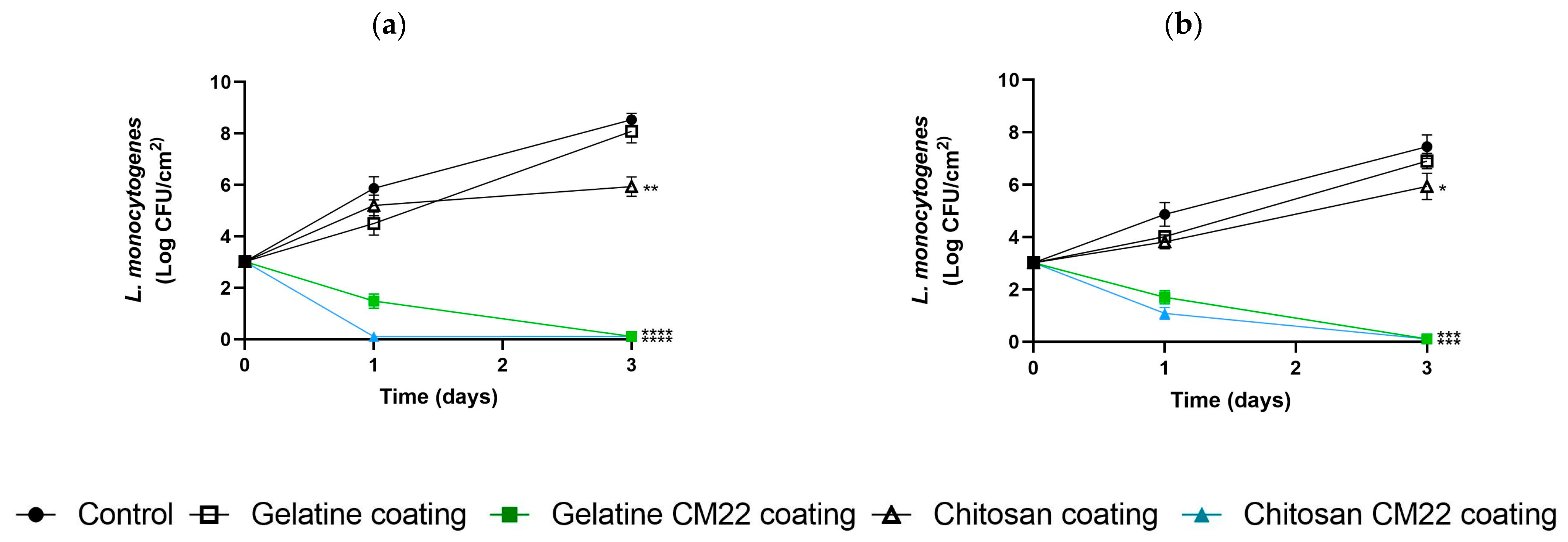
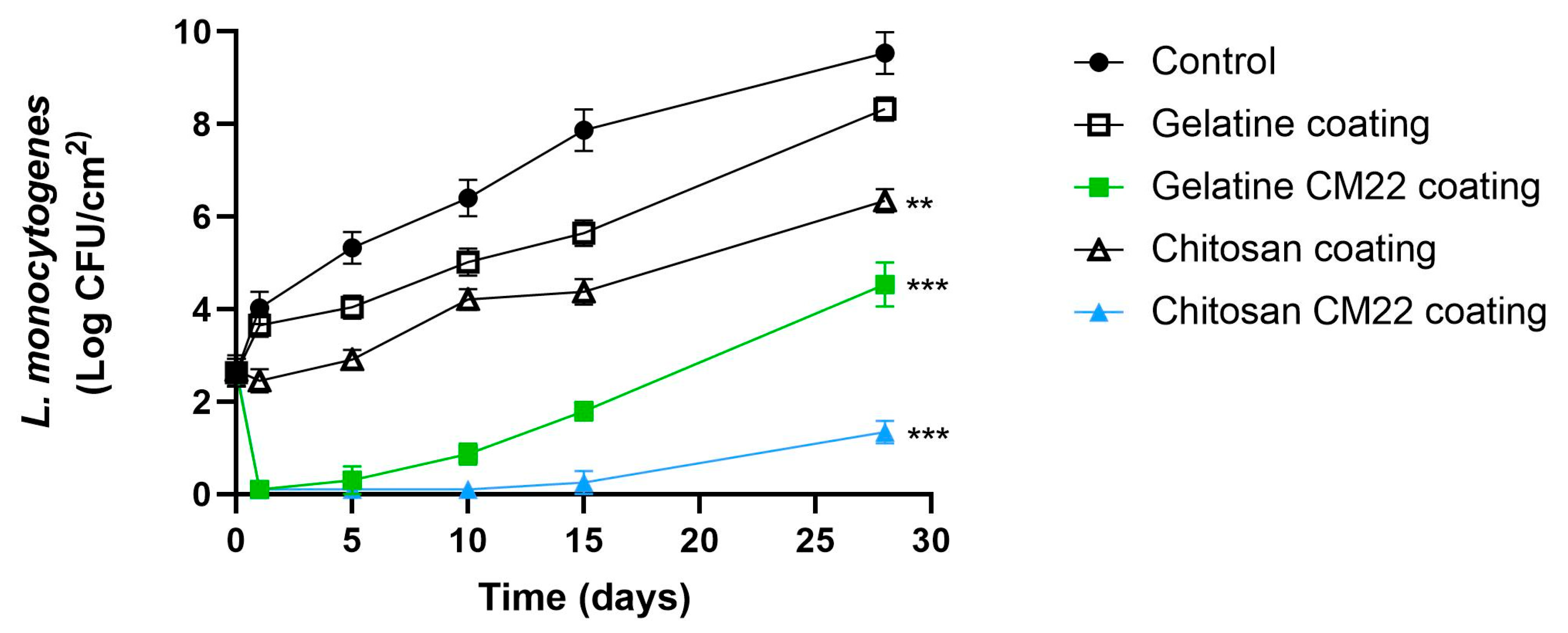
3.3. Challenge Test
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Jami, E.; White, B.A.; Mizrahi, I. Potential Role of the Bovine Rumen Microbiome in Modulating Milk Composition and Feed Efficiency. PLoS ONE 2014, 9, e85423. [Google Scholar] [CrossRef] [PubMed]
- Gudmundsdóttir, S.; Gudbjörnsdóttir, B.; Einarsson, H.; Kristinsson, K.G.; Kristjánsson, M. Contamination of Cooked Peeled Shrimp (Pandalus borealis) by Listeria monocytogenes during Processing at Two Processing Plants. J. Food Prot. 2006, 69, 1304–1311. [Google Scholar] [CrossRef] [PubMed]
- Poole, S.; Norhana, M.N.W.; Poole, S.E.; Deeth, H.C.; Dykes, G.A. Prevalence, Persistence and Control of Salmonella and Listeria in Shrimp and Shrimp Products: A Review Prevalence, Persistence and Control of Salmonella and Listeria in Shrimp and Shrimp Products: A Review. Food Control 2010, 21, 343–361. [Google Scholar] [CrossRef]
- Jiang, M.; Liu, S.; Wang, Y. Effects of Antimicrobial Coating from Catfish Skin Gelatin on Quality and Shelf Life of Fresh White Shrimp (Penaeus vannamei). J. Food Sci. 2011, 76, 204–209. [Google Scholar] [CrossRef] [PubMed]
- Bremer, P.J.; Monk, I.; Butler, R. Inactivation of Listeria monocytogenes/Flavobacterium spp. Biofilms Using Chlorine: Impact of Substrate, PH, Time and Concentration. Lett. Appl. Microbiol. 2002, 35, 321–325. [Google Scholar] [CrossRef]
- Hierro, E.; Ganan, M.; Barroso, E.; Fernández, M. Pulsed Light Treatment for the Inactivation of Selected Pathogens and the Shelf-Life Extension of Beef and Tuna Carpaccio. Int. J. Food Microbiol. 2012, 158, 42–48. [Google Scholar] [CrossRef]
- Danielski, G.M.; Henrique, P.; Maria, C.; Cavalari, D.A.; Daube, G.; Clinquart, A.; Ernlund, R.; Macedo, F. De Carnobacterium maltaromaticum as Bioprotective Culture in Vitro and in Cooked Ham. Meat Sci. 2020, 162, 108035. [Google Scholar] [CrossRef]
- Reis, F.B.; De Souza, V.M.; Thomaz, M.R.S.; Fernandes, L.P.; De Oliveira, W.P.; Martinis, E.C.P. De International Journal of Food Microbiology Use of Carnobacterium maltaromaticum Cultures and Hydroalcoholic Extract of Lippia sidoides Cham. against Listeria monocytogenes in Fi Sh Model Systems. Int. J. Food Microbiol. 2011, 146, 228–234. [Google Scholar] [CrossRef]
- Baños, A.; Ariza, J.J.; Nuñez, C.; Gil-Martínez, L.; García-López, J.D.; Martínez-Bueno, M.; Valdivia, E. Effects of Enterococcus faecalis UGRA10 and the Enterocin AS-48 against the Fish Pathogen Lactococcus garvieae. Studies in Vitro and in Vivo. Food Microbiol. 2019, 77, 69–77. [Google Scholar] [CrossRef]
- Mills, S.; Stanton, C.; Hill, C.; Ross, R.P. New Developments and Applications of Bacteriocins and Peptides in Foods. Annu. Rev. Food Sci. Technol. 2011, 2, 299–329. [Google Scholar] [CrossRef]
- Ingham, A.; Ford, M.; Moore, R.J.; Tizard, M. The Bacteriocin Piscicolin 126 Retains Antilisterial Activity in Vivo. J. Antimicrob. Chemother. 2003, 51, 1365–1371. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Zhang, J.; Zhao, L.; Tang, W.; Li, J.; Tang, T.; Sun, X. Heliyon Characterization of a Novel Circular Bacteriocin from Bacillus Velezensis 1-3, and Its Mode of Action against Listeria monocytogenes. Heliyon 2024, 10, e29701. [Google Scholar] [CrossRef] [PubMed]
- González, E.; David, G.J.; López, G.; Teso, C.; Irene, P.; Hernández, J.; Manuel, J.; Sánchez, P.; Valdivia, E.; Lopez, M.M.; et al. Genomic Characterization of Piscicolin CM22 Produced by Carnobacterium maltaromaticum CM22 Strain Isolated from Salmon (Salmo Salar). Probiotics Antimicrob. Proteins 2024. [Google Scholar] [CrossRef] [PubMed]
- Gursky, L.J.; Martin, N.I.; Derksen, D.J.; Van Belkum, M.J.; Kaur, K.; Vederas, J.C.; Stiles, M.E.; Mcmullen, L.M. Production of Piscicolin 126 by Carnobacterium maltaromaticum UAL26 Is Controlled by Temperature and Induction Peptide Concentration. Arch. Microbiol. 2006, 186, 317–325. [Google Scholar] [CrossRef]
- Gibbs, G.M.; Davidson, B.E.; Hillier, A.J. Novel Expression System for Large-Scale Production and Purification of Recombinant Class IIa Bacteriocins and Its Application to Piscicolin 126. Appl. Environ. Microbiol. 2004, 70, 3292–3297. [Google Scholar] [CrossRef]
- Surya, T.; Jeyasekaran, G.; Jeya, R.; Alsalhi, M.S.; Devanesan, S.; Sivaraman, B.; Arisekar, U.; Huong, T. Effect of Antibiotics and Sanitizers on Salmonella Biofilms Associated with Seafood Contact Surfaces. Microbiol. Res. 2023, 266, 127213. [Google Scholar] [CrossRef]
- Dhowlaghar, N.; Abeysundara, P.D.E.A.; Nannapaneni, R. Growth and Biofilm Formation by Listeria monocytogenes in Catfish Mucus Extract on Four Food Contact Surfaces at 22 and 10 °C and Their Reduction by Commercial Disinfectants. J. Food Prot. 2018, 81, 59–67. [Google Scholar] [CrossRef]
- Wu, S. International Journal of Biological Macromolecules Effect of Chitosan-Based Edible Coating on Preservation of White Shrimp during Partially Frozen Storage. Int. J. Biol. Macromol. 2014, 65, 325–328. [Google Scholar] [CrossRef]
- Falguera, V.; Quintero, J.P.; Jiménez, A.; Aldemar-Muñoz, J.; Ibarz, A. Edible Films and Coatings: Structures, Active Functions and Trends in Their Use. Trends Food Sci. Technol. 2011, 22, 292–303. [Google Scholar] [CrossRef]
- Amankwaah, C.; Li, J.; Lee, J.; Pascall, M.A. Development of Antiviral and Bacteriostatic Chitosan-Based Food Packaging Material with Grape Seed Extract for Murine Norovirus, Escherichia coli and Listeria innocua Control. Food Sci. Nutr. 2020, 8, 6174–6181. [Google Scholar] [CrossRef]
- Jasour, M.S.; Ehsani, A.; Samaneh, S. Chitosan Coating Incorporated with the Lactoperoxidase System: An Active Edible Coating for Fish Preservation. J. Sci. Food Agric. 2014, 95, 1373–1378. [Google Scholar] [CrossRef] [PubMed]
- Olaimat, A.N.; Fang, Y.; Holley, R.A. International Journal of Food Microbiology Inhibition of Campylobacter Jejuni on Fresh Chicken Breasts by κ-Carrageenan/Chitosan-Based Coatings Containing Allyl Isothiocyanate or Deodorized Oriental Mustard Extract. Int. J. Food Microbiol. 2014, 187, 77–82. [Google Scholar] [CrossRef] [PubMed]
- Gniewosz, M.; Kra, K.; Woreta, M.; Kosakowska, O. Antimicrobial Activity of a Pullulan—Caraway Essential Oil Coating on Reduction of Food Microorganisms and Quality in Fresh Baby Carrot. J. Food Sci. 2013, 78, M1242–M1248. [Google Scholar] [CrossRef] [PubMed]
- Murillo-Martínez, M.M.; Tello-Solís, S.R.; García-Sánchez, M.A.; Ponce-Alquicira, E. Antimicrobial Activity and Hydrophobicity of Edible Whey Protein Isolate Films Formulated with Nisin and/or Glucose Oxidase. J. Food Sci. 2013, 78, M560–M566. [Google Scholar] [CrossRef]
- Galvez, A.; Maqueda, M.; Valdivia, E.; Quesada, A.; Montoya, E. Characterization and Partial Purification of a Broad Spectrum Antibiotic AS-48 Produced by Streptococcus faecalis. Can. J. Microbiol. 1986, 32, 765–771. [Google Scholar] [CrossRef]
- Zhao, R.; Lu, Y.; Ran, J.; Li, G.; Lei, S.; Zhu, Y.; Xu, B. Purification and Characterization of Bacteriocin Produced by Lactobacillus Rhamnosus Zrx01. Food Biosci. 2020, 38, 100754. [Google Scholar] [CrossRef]
- Ahn, Y.N. Effect of Alkyl Branches on the Thermal Stability of Quaternary Ammonium Cations in Organic Electrolytes for Electrochemical Double. Phys. Chem. Chem. Phys. 2017, 19, 19959–19966. [Google Scholar] [CrossRef]
- Tan, C.; Faqir, Y.; Zeng, Y.; Huang, Y.; Ocloo, E.A.; Kaleri, A.R.; Kalhoro, M.T.; Ma, J.; Aslam, M. Investigation of Biomechanical Characteristics of Novel Chitosan from Dung Beetle and Its Application Potential on Stored Tomato Fruit. J. Food Meas. Charact. 2022, 16, 4551–4563. [Google Scholar] [CrossRef]
- Ma, J.; Xin, C.; Tan, C. Preparation, Physicochemical and Pharmaceutical Characterization of Chitosan from Catharsius Molossus Residue. Int. J. Biol. Macromol. 2015, 80, 547–556. [Google Scholar] [CrossRef]
- Tagg, J.R.; Mcgiven, A.R. Assay System for Bacteriocins. Appl. Microbiol. 1971, 21, 943. [Google Scholar] [CrossRef]
- Baños-Arjona, A.; García-López, J.D.; Núñez, C.; Martínez-Bueno, M.; Maqueda, M.; Valdivia, E. Biocontrol of Listeria monocytogenes in Fish by Enterocin AS-48 and Listeria Lytic Bacteriophage P100. LWT - Food Science and Technology 2016, 66, 672–677. [Google Scholar] [CrossRef]
- Thambiliyagodage, C.; Jayanetti, M.; Mendis, A.; Ekanayake, G. Recent Advances in Chitosan-Based Applications—A Review. Materials 2023, 16, 2073. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, A.; El Galiou, O.; Zantar, S.; Arakrak, A.; Laglaoui, A.; Zerrouk, M.H. Edible Films and Coatings: Major Challenges and Potential Applications in Food Packaging. A Review. In Food Packaging: The Smarter Way; Springer: Singapore, 2022; pp. 187–224. [Google Scholar] [CrossRef]
- Nitikornwarakul, C.; Wangpradid, R.; Rakkapao, N. Impact of Molar Composition on the Functional Properties of Glutinous Rice Starch–Chitosan Blend: Natural-Based Active Coating for Extending Mango Shelf Life. Polymers 2024, 16, 1375. [Google Scholar] [CrossRef] [PubMed]
- Qin, Y.; Li, P.; Guo, Z. Cationic Chitosan Derivatives as Potential Antifungals: A Review of Structural Optimization and Applications. Carbohydr. Polym. 2020, 236, 116002. [Google Scholar] [CrossRef]
- Palou, L.; Valencia-Chamorro, S.A.; Pérez-Gago, M.B. Antifungal Edible Coatings for Fresh Citrus Fruit: A Review. Coatings 2015, 5, 962–986. [Google Scholar] [CrossRef]
- Anderson, S. Salmon Color and the Consumer. In Proceedings of the IIFET 2000 Proceedings, Corvallis, OR, USA, 10–14 July 2000; p. 3. [Google Scholar]
- Fang, T.; Wu, Y.; Xie, Y.; Sun, L.; Qin, X.; Liu, Y.; Li, H.; Dong, Q.; Wang, X. Inactivation and Subsequent Growth Kinetics of Listeria monocytogenes after Various Mild Bactericidal Treatments. Front. Microbiol. 2021, 12, 646735. [Google Scholar] [CrossRef]
- Russo, G.L.; Langellotti, A.L.; Torrieri, E.; Masi, P. Emerging Technologies in Seafood Processing: An Overview of Innovations Reshaping the Aquatic Food Industry. Compr. Rev. Food Sci. Food Saf. 2024, 23, e13281. [Google Scholar] [CrossRef]
- Moreira, R.; Pereda, M.; Marcovich, N.E.; Roura, S.I. Antimicrobial Effectiveness of Bioactive Packaging Materials from Edible Chitosan and Casein Polymers: Assessment on Carrot, Cheese, and Salami. J. Food Sci. 2011, 76, 54–63. [Google Scholar] [CrossRef]
- Petrou, S.; Tsiraki, M.; Giatrakou, V.; Savvaidis, I.N. International Journal of Food Microbiology Chitosan Dipping or Oregano Oil Treatments, Singly or Combined on Modi Fi Ed Atmosphere Packaged Chicken Breast Meat. Int. J. Food Microbiol. 2012, 156, 264–271. [Google Scholar] [CrossRef]
- Günlü, A.; Koyun, E. Effects of Vacuum Packaging and Wrapping with Chitosan-Based Edible Film on the Extension of the Shelf Life of Sea Bass (Dicentrarchus labrax) Fillets in Cold Storage (4 °C). Food Bioprocess Technol. 2013, 6, 1713–1719. [Google Scholar] [CrossRef]
- Lungu, B.; Johnson, M.G. Potassium Sorbate Does Not Increase Control of Listeria monocytogenes When Added to Zein Coatings with Nisin on the Surface of Full Fat Turkey Frankfurter Pieces in a Model System at 4 °C. J. Food Sci. 2002, 70, M95–M99. [Google Scholar] [CrossRef]
- Gill, A.O.; Holley, R.A. Surface Application of Lysozyme, Nisin, and EDTA to Inhibit Spoilage and Pathogenic Bacteria on Ham and Bologna. J. Food Prot. 2000, 63, 1338–1346. [Google Scholar] [CrossRef] [PubMed]
- Min, B.J.; Han, I.Y.; Dawson, P.L. Antimicrobial Gelatin Films Reduce Listeria monocytogenes on Turkey Bologna 1. Poult. Sci. 2010, 89, 1307–1314. [Google Scholar] [CrossRef] [PubMed]
- Beverlya, R.L.; Janes, M.E.; Prinyawiwatkula, W.; No, H.K. Edible Chitosan Films on Ready-to-Eat Roast Beef for the Control of Listeria monocytogenes. Food Microbiol. 2008, 25, 534–537. [Google Scholar] [CrossRef]
- Ojagh, S.M.; Rezaei, M.; Hadi Razavi, S.; Hashem Hosseini, S.M. Effect of Chitosan Coatings Enriched with Cinnamon Oil on the Quality of Refrigerated Rainbow Trout. Food Chem. 2010, 120, 193–198. [Google Scholar] [CrossRef]
- Meade, E.; Slattery, M.A.; Garvey, M. Bacteriocins, Potent Antimicrobial Peptides and the Fight against Multi Drug Resistant Species: Resistance Is Futile? Antibiotics 2020, 9, 32. [Google Scholar] [CrossRef]
- Jack, R.W.; Wan, J.; Gordon, J.; Harmark, K.; Davidson, B.E.; Hillier, A.J.; Wettenhall, R.E.H.; Hickey, M.W.; Coventry, M.J. Characterization of the Chemical and Antimicrobial Properties of Piscicolin 126, a Bacteriocin Produced by Carnobacterium Piscicola JG126. Appl. Environ. Microbiol. 1996, 62, 2897–2903. [Google Scholar] [CrossRef]
- Karamah, E.F.; Ismaningtyas, N.; Putri Ilmiyah, A. The Effect of Exposure Time and Water Replacement in the Application of Ozonated Water to Maintain the Quality of Tuna. IOP Conf. Ser. Mater. Sci. Eng. 2019, 509, 012092. [Google Scholar] [CrossRef]
- Chan, S.S.; Jakobsen, A.N.; Roth, B.; Jessen, F. Water Holding Properties of Atlantic Salmon. Compr. Rev. Food Sci. Food Saf. 2021, 21, 477–498. [Google Scholar] [CrossRef]
- Kong, M.; Chen, X.G.; Xing, K.; Park, H.J. Antimicrobial Properties of Chitosan and Mode of Action: A State of the Art Review. Int. J. Food Microbiol. 2010, 144, 51–63. [Google Scholar] [CrossRef]
- Duan, J.; Park, S.I.; Daeschel, M.A.; Zhao, Y. Antimicrobial Chitosan-Lysozyme (CL) Films and Coatings for Enhancing Microbial Safety of Mozzarella Cheese. J. Food Sci. 2007, 72, M355–M362. [Google Scholar] [CrossRef] [PubMed]
- Masterson, K.; Major, I.; Lynch, M.; Rowan, N. Synergy Assessment of Four Antimicrobial Bioactive Compounds for the Combinational Treatment of Bacterial Pathogens. Biomedicines 2023, 11, 2216. [Google Scholar] [CrossRef] [PubMed]
- Wan, J.; Harmark, K.; Davidson, B.E.; Hillier, A.J.; Gordon, J.B.; Wilcock, A.; Hickey, M.W.; Coventry, M.J. Inhibition of Listeria monocytogenes by Piscicolin 126 in Milk and Camembert Cheese Manufactured with a Thermophilic Starter. J. Appl. Microbiol. 1997, 82, 273–280. [Google Scholar] [CrossRef] [PubMed]
- Acedo, J.Z.; Towle, K.M.; Lohans, C.T.; Miskolzie, M.; McKay, R.T.; Doerksen, T.A.; Vederas, J.C.; Martin-Visscher, L.A. Identification and Three-Dimensional Structure of Carnobacteriocin XY, a Class IIb Bacteriocin Produced by Carnobacteria. FEBS Lett. 2017, 591, 1349–1359. [Google Scholar] [CrossRef]
- De Niederhäusern, S.; Camellini, S.; Sabia, C.; Iseppi, R. Antilisterial Activity of Bacteriocins Produced by Lactic Bacteria Isolated from Dairy Products. Foods 2000, 9, 1757. [Google Scholar] [CrossRef]
- Regiane, L.F.; Ka, R.; Furlaneto, C. Antimicrobial Activity of Enterocins against Listeria sp. and Other Food Spoilage Bacteria. Biotechnol. Lett. 2020, 7, 797–806. [Google Scholar] [CrossRef]
- Martin-visscher, L.A.; Yoganathan, S.; Sit, C.S.; Lohans, C.T.; Vederas, J.C. The Activity of Bacteriocins from Carnobacterium maltaromaticum UAL307 against Gram-Negative Bacteria in Combination with EDTA treatment. FEMS Microbiol. Lett. 2011, 317, 152–159. [Google Scholar] [CrossRef]
- Gonzales-Barron, U.; Cadavez, V.; De Oliveira Mota, J.; Guillier, L.; Sanaa, M. A Critical Review of Risk Assessment Models for Listeria monocytogenes in Produce. Foods 2024, 13, 1111. [Google Scholar] [CrossRef]
- Heir, E.; Jensen, M.R.; Aasli, A.W.; Berget, I.; Holck, A.L. Reduction and Growth Inhibition of Listeria monocytogenes by Use of Anti-Listerial Nisin, P100 Phages and Buffered Dry Vinegar Fermentates in Standard and Sodium-Reduced Cold-Smoked Salmon. Foods 2023, 12, 4391. [Google Scholar] [CrossRef]
- Chen, R.; Skeens, J.W.; Wiedmann, M.; Guariglia-Oropeza, V. The Efficacy of Nisin against Listeria monocytogenes on Cold-Smoked Salmon at Natural Contamination Levels Is Concentration-Dependent and Varies by Serotype. Front. Microbiol. 2022, 13, 930400. [Google Scholar] [CrossRef]
- Leistner, L. Basic Aspects of Food Preservation by Hurdle Technology. Int. J. Food Microbiol. 2000, 55, 181–186. [Google Scholar] [CrossRef]


| Treatments | Bacteriocin Activity a |
|---|---|
| Control (only bacteriocin) | 26 ± 0.5 |
| Heat | |
| 60 °C for 30 min | 26 ± 1 |
| 80 °C for 30 min | 25 ± 0.5 |
| 100 °C for 30 min | 20 ± 1 |
| 121 °C for 15 min | - |
| pH | |
| 2 | 25 ± 0.5 |
| 3 | 26 ± 1 |
| 5 | 26 ± 1 |
| 7 | 26 ± 0.5 |
| 9 | 35 ± 0.2 |
| 10 | 20 ± 0.5 |
| 11 | - |
| Enzymes | |
| Trypsin, papain, and proteinase K | - |
| Edible Coatings (ECs) | TF μm | FS, % | FWS, % | WVP, mg cm−2 h−1 |
|---|---|---|---|---|
| Gelatin coating (GC) | 32.5 ± 0.6 | 8.7 ± 1.2 | 63.4 ± 2.6 | 8.7 ± 1.3 |
| Gelatin-CM22 coating (GC-CM22) | 31.9 ± 1.3 | 7.8 ± 0.9 | 60.9 ± 4.1 | 8.8 ± 1.7 |
| Chitosan coating (CC) | 21.7 ± 0.9 | 5.3 ± 0.7 | 39.7 ± 1.4 | 4.2 ± 1.1 |
| Chitosan-CM22 coating (CC-CM22) | 22.3 ± 1.8 | 4.8 ± 0.6 | 41.2 ± 2.3 | 4.3 ± 1.5 |
| Strain | Inhibition of Halo (mm) | ||||
|---|---|---|---|---|---|
| GC | GC-CM22 | CC | CC-CM22 | CM22 | |
| L. monocytogenes DZSM 112143 | 0 | 19 ± 1 | 10 ± 1 | 28 ± 1 | 24 ± 1 |
| L. monocytogenes DMC 04 | 0 | 19 ± 1.5 | 11 ± 1 | 29 ± 1.4 | 25 ± 1.4 |
| L. monocytogenes DMC 05 | 0 | 19 ± 2 | 11 ± 1.2 | 29 ± 2 | 22 ± 2 |
| L. monocytogenes CECT 4032 | 0 | 22 ± 1 | 11 ± 1.2 | 27 ± 1 | 25 ± 1 |
| L. monocytogenes CECT 5342 | 0 | 23 ± 1.3 | 12 ± 1 | 26 ± 1.5 | 26 ± 1.5 |
| Time (Days) | ||||
|---|---|---|---|---|
| Salmon | 0 | 1 | 3 | |
| Control | 6.72 | 6.72 | 6.85 | |
| Gelatin coating (GC) | 6.72 | 6.72 | 6.83 | |
| Gelatin coating with CM22 (GC-CM22) | 4 °C | 6.94 | 6.94 | 6.75 |
| Chitosan coating (CC) | 6.79 | 6.79 | 6.77 | |
| Chitosan coating with CM22 (CC-CM22) | 6.87 | 6.87 | 6.86 | |
| Control | 6.85 | 6.85 | 7.15 | |
| Gelatin coating (GC) | 6.88 | 6.88 | 6.81 | |
| Gelatin coating with CM22 (GC-CM22) | 8 °C | 6.87 | 6.87 | 6.81 |
| Chitosan coating (CC) | 6.75 | 6.75 | 6.78 | |
| Chitosan coating CM22 (CC-CM22) | 6.73 | 6.73 | 6.79 | |
| Tuna | ||||
| Control | 6.54 | 6.54 | 6.59 | |
| Gelatin coating (GC) | 6.38 | 6.38 | 6.67 | |
| Gelatin coating with CM22 (GC-CM22) | 4 °C | 6.71 | 6.71 | 6.72 |
| Chitosan coating (CC) | 6.38 | 6.38 | 6.35 | |
| Chitosan coating with CM22 (CC-CM22) | 6.42 | 6.42 | 6.64 | |
| Control | 6.57 | 6.57 | 6.56 | |
| Gelatin coating (GC) | 6.53 | 6.53 | 6.77 | |
| Gelatin coating CM22 (GC-CM22) | 8 °C | 6.68 | 6.68 | 6.76 |
| Chitosan coating (CC) | 6.74 | 6.74 | 6.79 | |
| Chitosan coating with CM22 (CC-CM22) | 6.49 | 6.49 | 6.77 | |
| Parameter | Time (Days) | Control | CC | CC-CM22 | GC | GC-CM22 | |
|---|---|---|---|---|---|---|---|
| L* | 0 | 50.77 ± 0.87 | 52.94 ± 0.87 | 52.93 ± 0.87 | 51.29 ± 0.87 | 51.31 ± 0.87 | |
| 4 °C | 1 | 53.68 ± 1.92 | 51.68 ± 2.34 | 52.57 ± 0.98 | 51.15 ± 3.01 | 51.4 ± 3.01 | |
| 3 | 42.41 a ± 2.03 | 52.20 b,c ± 3.73 | 55.66 ± 4.03 | 50.35 c ± 2.27 | 50.38 b ± 2.27 | ||
| 0 | 52.63 ± 1.08 | 52.94 ± 1.37 | 52.94 ± 0.13 | 51.25 ± 0.88 | 51.11 ± 0.88 | ||
| 8 °C | 1 | 42.49 a ± 0.97 | 43.83 a ± 1.02 | 49.33 b ± 0.90 | 50.35 b ± 0.53 | 50.96 b ± 0.53 | |
| 3 | 38.66 a ± 0.85 | 48.65 b ± 0.48 | 48.55 b ± 1.47 | 50.55 c ± 0.48 | 49.81 b,c ± 0.48 | ||
| a* | 0 | 24.35 b ± 0.98 | 21.81 a ± 0.98 | 21.81 a ± 0.98 | 24.76 b ± 0.98 | 24.93 b ± 0.98 | |
| 4 °C | 1 | 11.15 a ± 0.75 | 15.65 b ± 0.10 | 15.85 b ± 1.31 | 19.85 c ± 0.7 | 19.82 c ± 0.7 | |
| 3 | 12.71 a ± 0.65 | 14.11 a,b ± 0.85 | 13.50 a ± 1.44 | 17.69 b ± 1.13 | 17.57 b ± 1.13 | ||
| 0 | 23.44 ± 1.75 | 21.81 ± 1.01 | 21.81 ± 0.66 | 24.27 ± 0.43 | 24.96 ± 0.43 | ||
| 8 °C | 1 | 13.42 a ± 0.41 | 18.55 b ± 0.74 | 17.58 b ± 0.34 | 18.21 b ± 0.65 | 13.20 a ± 0.65 | |
| 3 | 11.49 a ± 0.13 | 15 b ± 0.34 | 15.52 b ± 0.42 | 11.51 a ± 0.70 | 11.52 a ± 0.70 | ||
| b* | 0 | 13.35 ± 0.40 | 14.46 ± 0.40 | 14.46 ± 0.40 | 14.9 ± 0.40 | 14.58 ± 0.40 | |
| 4 °C | 1 | 12.58 ± 0.68 | 14.71 ± 0.67 | 13.75 ± 1.34 | 14.88 ± 1.75 | 13.71 ± 1.75 | |
| 3 | 6.31 a ± 0.88 | 15.27 b ± 1.88 | 13.3 b ± 1.11 | 13.63 b ± 0.58 | 13.64 b ± 0.58 | ||
| 0 | 13.52 ± 0.33 | 13.46 ± 0.69 | 13.46 ± 0.47 | 14.81 ± 0.23 | 13.57 ± 0.23 | ||
| 8 °C | 1 | 8.79 a ± 0.24 | 11.97 b ± 0.27 | 13.28 b ± 0.17 | 12.68 b ± 0.47 | 13.68 b ± 0.47 | |
| 3 | 1.49 a ± 0.1 | 9.51 b ± 0.3 | 9.48 b ± 0.26 | 10.48 b ± 0.46 | 10.48 b ± 0.46 |
| Parameter | Time (Days) | Control | CC | CC-CM22 | GC | GC-CM22 | |
|---|---|---|---|---|---|---|---|
| L* | 0 | 27.05 a ± 0.87 | 40.55 b ± 0.87 | 40.55 b ± 0.87 | 27.76 a ± 0.87 | 27.45 a ± 0.87 | |
| 4 °C | 1 | 26.62 a ± 1.92 | 38.16 b ± 2.34 | 38.57 b ± 0.98 | 28.79 a ± 3.01 | 28.44 a ± 3.01 | |
| 3 | 25.17 a ± 2.03 | 38.60 b ± 3.73 | 40.04 b ± 4.03 | 28.6 a ± 2.27 | 28.31 a ± 2.27 | ||
| 0 | 27.5 a ± 1.08 | 42.55 b ± 1.37 | 42.55 b ± 0.13 | 27.5 b ± 0.88 | 27.48 a ± 0.88 | ||
| 8 °C | 1 | 11.50 a ± 0.97 | 37.90 c ± 1.02 | 33.66 c ± 0.90 | 22.2 b ± 0.53 | 23.66 b ± 0.53 | |
| 3 | 18.48 a ± 0.85 | 38.45 b ± 0.48 | 38.47 b ± 1.47 | 18.46 a ± 0.48 | 18.49 a ± 0.48 | ||
| a* | 0 | 16.24 b ± 0.98 | 15.47 b ± 0.98 | 15.47 b ± 0.98 | 5.71 a ± 0.98 | 5.13 a ± 0.98 | |
| 4 °C | 1 | 16.89 b ± 0.75 | 16.07 b ± 0.10 | 16.35 b ± 1.31 | 9.61 a ± 0.7 | 9.97 a ± 0.7 | |
| 3 | 15.14 b ± 0.65 | 16.51 b ± 0.85 | 16.24 b ± 1.44 | 11.52 a ± 1.13 | 12.4 a ± 1.13 | ||
| 0 | 16.64 b ± 1.75 | 16.79 b ± 1.01 | 16.79 b ± 0.66 | 5.99 a ± 0.43 | 5.08 a ± 0.43 | ||
| 8 °C | 1 | 9.24 a ± 0.41 | 14.49 b ± 0.74 | 14.63 b ± 0.34 | 8.11 a ± 0.65 | 8.49 a ± 0.65 | |
| 3 | 0.52 a ± 0.13 | 12.52 b ± 0.34 | 12.52 b ± 0.42 | 0.53 a ± 0.70 | 0.53 a ± 0.70 | ||
| b* | 0 | 5.29 a ±0.40 | 5.47 b ± 0.40 | 5.55 b ± 0.40 | 2.16 a ± 0.40 | 2.81 a ± 0.40 | |
| 4 °C | 1 | 6.66 a ±0.68 | 5.06 a ± 0.67 | 6.04 a ± 1.34 | 6.60 a ± 1.75 | 10.27 b ± 1.75 | |
| 3 | 2.07 a ±0.88 | 4.51 b ± 1.88 | 4.44 b ± 1.11 | 1.51 a ± 0.58 | 15.68 c ± 0.58 | ||
| 0 | 5.78 a ±0.33 | 5.14 b ± 0.69 | 5.14 b ± 0.47 | 2.05 a ± 0.23 | 2.77 a ± 0.23 | ||
| 8 °C | 1 | 6.94 a ±0.24 | 5.14 b ± 0.27 | 5.11 b ± 0.17 | 1.17 a ± 0.47 | 1.43 a ± 0.47 | |
| 3 | 1.48 a ±0.1 | 4.51 b ± 0.3 | 4.48 b ± 0.26 | 1.52 a ± 0.46 | 1.49 a ± 0.46 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
González-Gragera, E.; García-López, J.D.; Boutine, A.; García-Marín, M.L.; Fonollá, J.; Gil-Martínez, L.; Fernández, I.; Martínez-Bueno, M.; Baños, A. Improving the Quality and Safety of Fish Products with Edible Coatings Incorporating Piscicolin CM22 from a Psychrotolerant Carnobacterium maltaromaticum Strain. Foods 2024, 13, 3165. https://doi.org/10.3390/foods13193165
González-Gragera E, García-López JD, Boutine A, García-Marín ML, Fonollá J, Gil-Martínez L, Fernández I, Martínez-Bueno M, Baños A. Improving the Quality and Safety of Fish Products with Edible Coatings Incorporating Piscicolin CM22 from a Psychrotolerant Carnobacterium maltaromaticum Strain. Foods. 2024; 13(19):3165. https://doi.org/10.3390/foods13193165
Chicago/Turabian StyleGonzález-Gragera, Elías, José David García-López, Abdelkader Boutine, María Luisa García-Marín, Juristo Fonollá, Lidia Gil-Martínez, Inmaculada Fernández, Manuel Martínez-Bueno, and Alberto Baños. 2024. "Improving the Quality and Safety of Fish Products with Edible Coatings Incorporating Piscicolin CM22 from a Psychrotolerant Carnobacterium maltaromaticum Strain" Foods 13, no. 19: 3165. https://doi.org/10.3390/foods13193165
APA StyleGonzález-Gragera, E., García-López, J. D., Boutine, A., García-Marín, M. L., Fonollá, J., Gil-Martínez, L., Fernández, I., Martínez-Bueno, M., & Baños, A. (2024). Improving the Quality and Safety of Fish Products with Edible Coatings Incorporating Piscicolin CM22 from a Psychrotolerant Carnobacterium maltaromaticum Strain. Foods, 13(19), 3165. https://doi.org/10.3390/foods13193165






