Chemical Profile and Biological Activities of Brassica rapa and Brassica napus Ex Situ Collection from Portugal
Abstract
:1. Introduction
2. Material and Methods
2.1. Plant Material
2.2. Methanol Extracts
2.3. Liquid Chromatography and Tandem Mass Spectrometry
2.4. Evaluation of Biological Activities
2.4.1. Antioxidant Activity
- Radical-scavenging capacity assay (DPPH•)
- Ferric-ion-reducing antioxidant power assay
- Total phenolic content by Folin–Ciocalteu reagent
2.4.2. Antibacterial Activity
2.5. Statistical Analysis
3. Results and Discussion
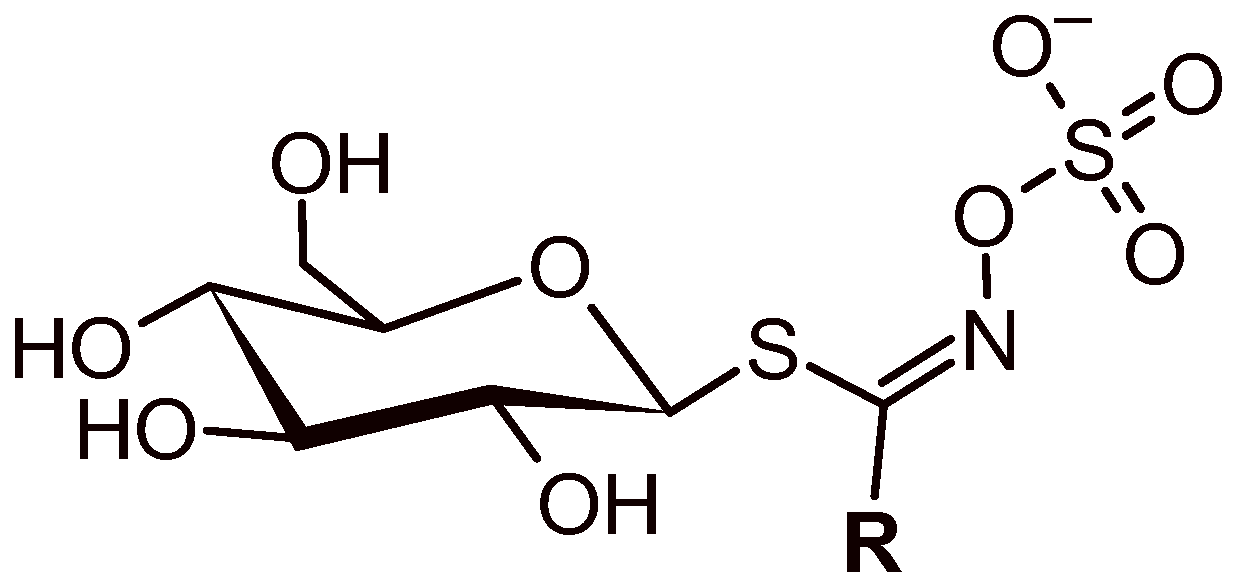
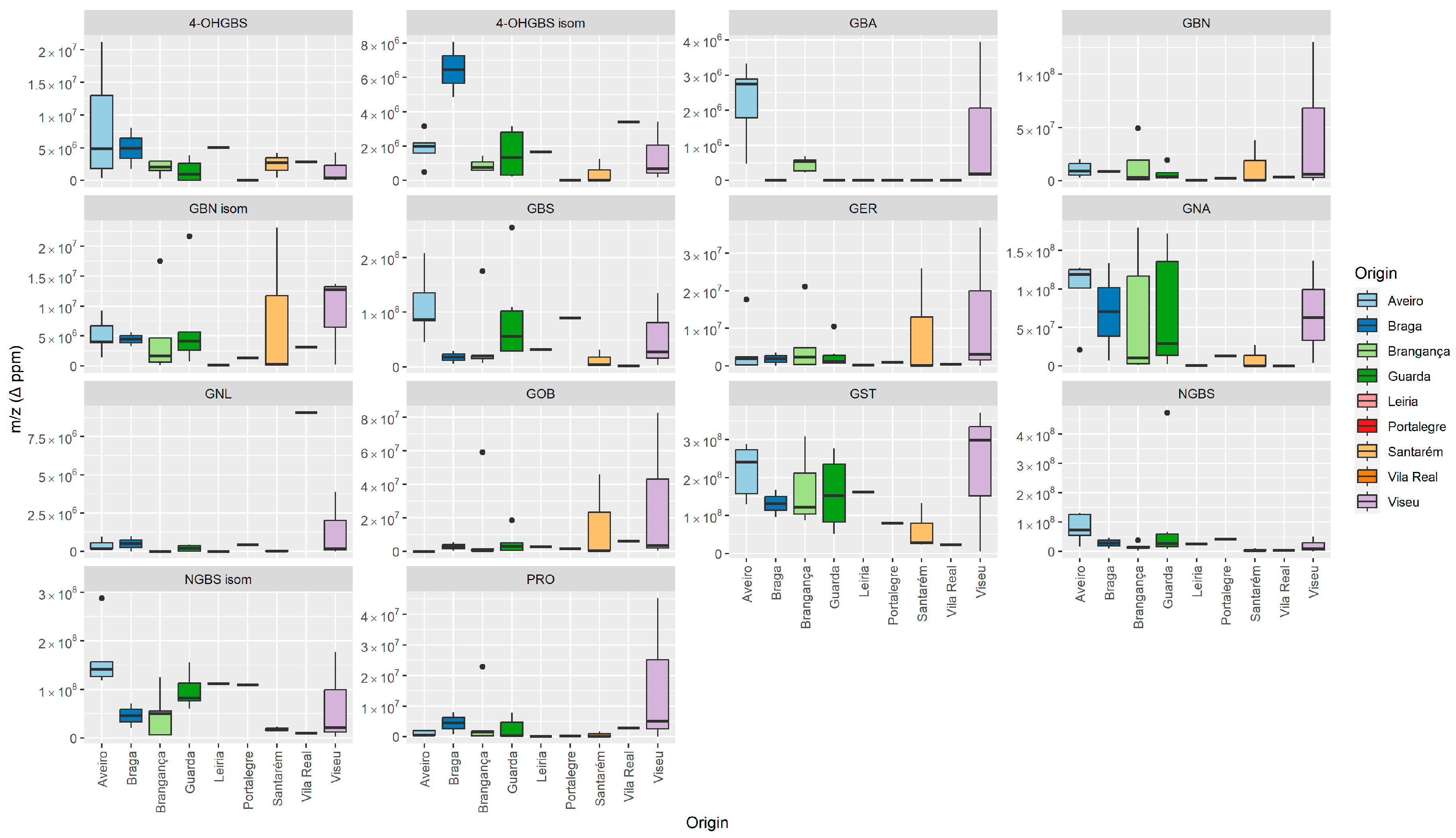
3.1. GLS’ Profile by Liquid Chromatography–Tandem Mass Spectrometry
3.2. In Vitro Antioxidant Activity and Total Phenolic Content
3.3. Antibacterial Activity
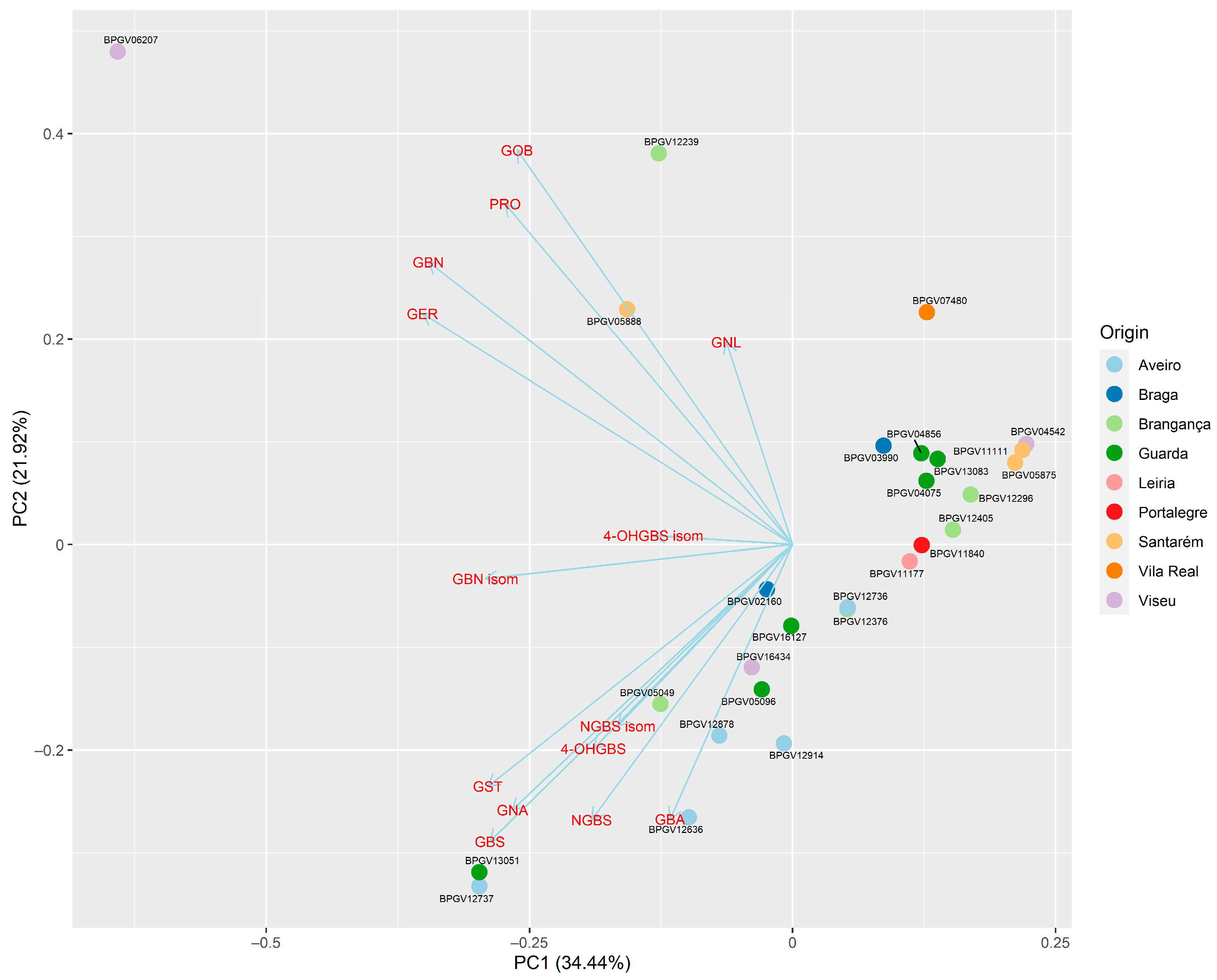
3.4. Principal Component Analysis (PCA) to Assess Overall Variation
3.5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- FAOSTAT. Food and Agriculture Organization of the United Nations. FAOSTAT Statistical Database. FAO DATABASE. 2014. Available online: http://www.fao.org/faostat/en/ (accessed on 5 May 2021).
- Mordor Intelligence Research & Advisory. Análise de Tamanho e Participação do Mercado de Couves e Outras Brássicas—Tendências e Previsões de Crescimento (2023–2028). Mordor Intelligence. 2023. Available online: https://www.mordorintelligence.com/pt/industry-reports/cabbages-and-other-brassicas-market (accessed on 17 January 2024).
- FAO. Fruit and vegetables—Your dietary essentials. In The International Year of Fruits and Vegetables; Background Paper; FAO: Rome, Italy, 2021. [Google Scholar] [CrossRef]
- FAOSTAT. Food and Agriculture Organization of the United Nations. FAOSTAT Statistical Database. FAO DATABASE. 2018. Available online: http://www.fao.org/faostat/en/#data (accessed on 30 March 2022).
- INE. Base de Dados—Produção das Principais Culturas Agrícolas. 2022. Available online: https://www.ine.pt/xportal/xmain?xpid=INE&xpgid=ine_indicadores&indOcorrCod=0000020&xlang=pt&contexto=bd&selTab=tab2 (accessed on 12 May 2022).
- Prakash, S.; Wu, X.-M.; Bhat, S.R. History, Evolution, and Domestication of Brassica Crops. In Plant Breeding Reviews; Janick, J., Ed.; Wiley Online Books: Hoboken, NJ, USA, 2011; Volume 35, pp. 19–84. [Google Scholar] [CrossRef]
- Guo, Y.; Chen, S.; Li, Z.; Wallace, C.A. Center of Origin and Centers of Diversity in an Ancient Crop, Brassica rapa (Turnip Rape). J. Hered. 2014, 105, 555–565. [Google Scholar] [CrossRef]
- Prakash, S.; Bhat, S.R.; Quiros, C.F.; Kirti, P.B.; Chopra, V.L. Brassica and Its Close Allies: Cytogenetics and Evolution. In Plant Breeding Reviews; Janick, J., Ed.; John Wiley & Sons: Hoboken, NY, USA, 2009; pp. 21–187. [Google Scholar] [CrossRef]
- Ayadi, J.; Debouba, M.; Rahmani, R.; Bouajila, J. Brassica Genus Seeds: A Review on Phytochemical Screening and Pharmacological Properties. Molecules 2022, 27, 6008. [Google Scholar] [CrossRef] [PubMed]
- Das, G.; Tantengco, O.A.G.; Tundis, R.; Robles, J.A.H.; Loizzo, M.R.; Shin, H.S.; Patra, J.K. Glucosinolates and Omega-3 Fatty Acids from Mustard Seeds: Phytochemistry and Pharmacology. Plants 2022, 11, 2290. [Google Scholar] [CrossRef]
- Bird, K.A.; An, H.; Gazave, E.; Gore, M.A.; Pires, J.C.; Robertson, L.D.; Labate, J.A. Population Structure and Phylogenetic Relationships in a Diverse Panel of Brassica rapa L. Front. Plant Sci. 2017, 13, 321. [Google Scholar] [CrossRef]
- McAlvay, A.C.; Ragsdale, A.P.; Mabry, M.E.; Qi, X.; Bird, K.A.; Velasco, P.; An, H.; Pires, J.C.; Emshwiller, E. Brassica rapa Domestication: Untangling Wild and Feral Forms and Convergence of Crop Morphotypes. Mol. Biol. Evol. 2021, 38, 3358–3372. [Google Scholar] [CrossRef]
- Hayward, A. Introduction-oilseed Brassicas. In Genetics, Genomics and Breeding of Oilseed Brassicas; Science Publishers, Inc.: Enfield, NH, USA, 2011; pp. 1–13. [Google Scholar]
- Halkier, B.A.; Gershenzon, J. Biology and biochemistry of glucosinolates. Annu. Rev. Plant Biol. 2006, 57, 303–333. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, E.; Alegria, C.; Abreu, M. Benefits of Brassica nutraceutical compounds in human health. In Brassicaceae: Characterization, Functional Genomics and Health Benefits; Lang, M., Ed.; Nova Publishers, Inc.: New York, NY, USA, 2013; Chapter 2; pp. 19–65. [Google Scholar]
- Gonçalves, E.M.; Alegria, C.; Ramos, A.C.; Abreu, M. Advances in understanding and improving the nutraceutical properties of broccoli and other Brassicas. In Understanding and Optimising the Nutraceutical Properties of Fruit and Vegetables; Preedy, V.R., Patel, V.B., Eds.; Burleigh Dodds Science Publishing: Cambridge, UK, 2022; pp. 1–31. ISBN 978-1-78676-850-6. Available online: www.bdspublishing.com (accessed on 28 February 2023). [CrossRef]
- Fahey, J.W.; Zalcmann, A.T.; Talalay, P. The chemical diversity and distribution of glucosinolates and isothiocyanates among plants. Phytochemistry 2001, 56, 5–51. [Google Scholar] [CrossRef] [PubMed]
- Holst, B.; Williamson, G. A critical review of the bioavailability of glucosinolates and related compounds. Nat. Prod. Rep. 2004, 21, 425–447. [Google Scholar] [CrossRef]
- Padilla, G.; Cartea, M.E.; Velasco, P.; de Haro, A.; Ordás, A. Variation of glucosinolates in vegetable crops of Brassica rapa. Phytochemistry 2007, 68, 536–545. [Google Scholar] [CrossRef]
- Wagner, A.E.; Terschluessen, A.M.; Rimbach, G. Health promoting effects of brassica-derived phytochemicals: From chemo-preventive and anti-inflammatory activities to epigenetic regulation. Oxid. Med. Cell. Longev. 2013, 2013, 964539. [Google Scholar] [CrossRef]
- Burrows, G.E.; Tyrl, R.J. Toxic Plants of North America, 2nd ed.; Wiley-Blackwell: Ames, IA, USA, 2013; ISBN 978-0-813-82034-7. [Google Scholar]
- Shapiro, T.A.; Fahey, J.W.; Dinkova-Kostova, A.T.; Holtzclaw, W.D.; Stephenson, K.K.; Wade, K.L.; Ye, L.; Talalay, P. Safety, tolerance, and metabolism of broccoli sprout glucosinolates and isothiocyanates: A clinical phase I study. Nutr. Cancer 2006, 55, 53–62. [Google Scholar] [CrossRef] [PubMed]
- Burčul, F.; Generalić Mekinić, I.; Radan, M.; Rollin, P.; Blažević, I. Isothiocyanates: Cholinesterase inhibiting, antioxidant, and anti-inflammatory activity. J. Enzym. Inhib. Med. Chem. 2018, 33, 577–582. [Google Scholar] [CrossRef] [PubMed]
- Esteve, M. Mechanisms Underlying Biological Effects of Cruciferous Glucosinolate-Derived Isothiocyanates/Indoles: A Focus on Metabolic Syndrome. Front. Nutr. 2020, 7, 111. [Google Scholar] [CrossRef] [PubMed]
- Romani, A.; Vignolini, P.; Isolani, L.; Ieri, F.; Heimler, D. HPLC-DAD/MS characterization of flavonoids and hydroxycinnamic derivatives in turnip tops (Brassica rapa L. Subsp. sylvestris L.). J. Agric. Food Chem. 2006, 54, 1342–1346. [Google Scholar] [CrossRef] [PubMed]
- Dias, J.S. Taxonomia das Couve Galaico-Portuguesas Utilizando Caracteres Morfológicos, Isoenzimas e RFLPs. Ph.D. Thesis, Universidade Técnica de Lisboa, Lisboa, Portugal, 1992. [Google Scholar]
- Dias, J.S. Portuguese perennial kale: A relic leafy vegetable crop. Genet. Resour. Crop Evol. 2012, 59, 1201–1206. [Google Scholar] [CrossRef]
- Negri, V.; Maxted, N.; Vetelainen, M. European landrace conservation: An introduction. In European Landraces: On-Farm Conservation, Management and Use; Bioversity Technical, Bulletin; Vetelainen, M., Negri, V., Maxted, N., Eds.; Bioversity International: Rome, Italy, 2009; Volume 15, pp. 1–22. [Google Scholar]
- Cartea, E.; Cámara-Martos, F.; Obregón, S.; Rubén Badenes-Pérez, F.; De Haro, A. Advances in Breeding in Vegetable Brassica rapa Crops; IntechOpen: London, UK, 2021. [Google Scholar] [CrossRef]
- Lopes, V.R.; Barata, A.M.; Nunes, E.; Cartea, M.H.; Soengas, P.; Allender, C.; Bas, N. Assessment of Genetic Diversity in Iberian Landraces of Brassica oleracea by Molecular Markers. In Proceedings of the 6th International Symposium on Brassica and 18th Crucifer Genetic Workshop, Catania, Italy, 12–16 November 2012; p. 98. [Google Scholar]
- Branca, F.; Coelho, P.S.; De Haro, A.; Rosa, E.; Lopes, V.R.; Scalzo, R. Collection, Characterization and Evaluation of Wild and Cultivated Brassicas. 2016. Available online: https://www.researchgate.net/profile/Ferdinando_Branca/ (accessed on 30 March 2020). [CrossRef]
- Coelho, P.S.; Monteiro, A.A.; Lopes, V.R.; Branca, F. New sources of resistance to downy mildew in a collection of wild and cultivated brassicas. Acta Hortic. 2018, 1202, 93–100. [Google Scholar] [CrossRef]
- Tribulato, A.; Donzella, E.; Sdouga, D.; Lopes, V.R.; Branca, F. Bio-morphological characterization of Mediterranean wild and cultivated Brassica species. Acta Hortic. 2018, 1202, 9–16. [Google Scholar] [CrossRef]
- Coelho, P.S.; Carlier, J.D.; Monteiro, A.A.; Leitão, J.M. A major QTL conferring downy mildew resistance in ‘Couve Algarvia’ (Brassica oleracea var. tronchuda) is located on chromosome 8. Acta Hortic. 2023, 1362, 289–296. [Google Scholar] [CrossRef]
- Subramanian, P.; Kim, S.H.; Hahn, B.S. Brassica biodiversity conservation: Prevailing constraints and future avenues for sustainable distribution of plant genetic resources. Front. Plant Sci. 2023, 14, 1220134. [Google Scholar] [CrossRef]
- Conversa, G.; Lazzizera, C.; Bonasia, A.; La Rotonda, P.; Elia, A. Nutritional Characterization of Two Rare Landraces of Tur-nip (Brassica rapa. var. rapa) Tops and Their On-Farm Conservation in Foggia Province. Sustainability 2020, 12, 3842. [Google Scholar] [CrossRef]
- Aires, A.; Fernandes, C.; Carvalho, R.; Bennett, R.N.; Saavedra, M.J.; Rosa, E.A.S. Seasonal Effects on Bioactive Compounds and Antioxidant Capacity of Six Economically Important Brassica Vegetables. Molecules 2011, 16, 6816–6832. [Google Scholar] [CrossRef] [PubMed]
- OECD. Brassica crops (Brassica species). In Safety Assessment of Transgenic Organisms in the Environment; OECD Consensus Documents; OECD Publishing: Paris, France, 2016; Volume 5. [Google Scholar] [CrossRef]
- Kondo, S.; Yoshikawa, H.; Katayama, R. Antioxidant activity in astringent and non-astringent persimmons. J. Hortic. Sci. Biotechnol. 2004, 79, 390–394. [Google Scholar] [CrossRef]
- Deighton, N.; Brennan, R.; Finn, C.; Davies, H.V. Antioxidant properties of domesticated and wild Rubus species. J. Sci. Food Agric. 2000, 80, 1307–1313. [Google Scholar] [CrossRef]
- Slinkard, K.; Singleton, V.L. Total Phenol Analysis: Automation and Comparison with Manual Methods. Am. J. Enol. Vitic. 1977, 28, 49–55. [Google Scholar] [CrossRef]
- Asami, D.K.; Hong, Y.J.; Barrett, D.M.; Mitchell, A.E. Processing-induced changes in total phenolics and procyanidins in clingstone peaches. J. Sci. Food Agric. 2003, 83, 56–63. [Google Scholar] [CrossRef]
- Pires, T.C.P.; Dias, M.I.; Barros, L.; Alves, M.J.; Oliveira, M.B.P.; Santos-Buelga, C.; Ferreira, I.C.R. Antioxidant and antimicrobial properties of dried Portuguese apple variety (Malus domestica Borkh. cv Bravo de Esmolfe). Food Chem. 2018, 240, 701–706. [Google Scholar] [CrossRef] [PubMed]
- STATISTICA (Data Analysis Software System), Version 12; StatSoft. Inc. Available online: https://statistica.software.informer.com/12.0/ (accessed on 1 November 2021).
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2021; Available online: https://www.R-project.org/ (accessed on 1 November 2021).
- Wickham, H.; Averick, M.; Bryan, J.; Chang, W.; McGowan, L.D.; François, R.; Grolemund, G.; Hayes, A.; Henry, L.; Hester, J.; et al. Welcome to the Tidyverse. J. Open Source Softw. 2019, 4, 1686. [Google Scholar] [CrossRef]
- Wickham, H. ggplot2: Elegant Graphics for Data Analysis; Springer: New York, NY, USA, 2016. [Google Scholar]
- Olivoto, T.; Lúcio, A.D. metan: An R package for multi-environment trial analysis. Methods Ecol. Evol. 2020, 11, 783–789. [Google Scholar] [CrossRef]
- Mocniak, L.E.; Elkin, K.R.; Dillard, S.L.; Bryant, R.B.; Soder, E.J. Building comprehensive glucosinolate profiles for brassica varieties. Talanta 2023, 251, 123814. [Google Scholar] [CrossRef]
- Stefanucci, A.; Zengin, G.; Llorent-Martinez, E.J.; Dimmito, M.P.; Della Valle, A.; Pieretti, S.; Ak, G.; Sinan, K.I.; Mollica, A. Chemical characterization, antioxidant properties and enzyme inhibition of Rutabaga root’s pulp and peel (Brassica napus L.). Arab. J. Chem. 2020, 13, 7078–7086. [Google Scholar] [CrossRef]
- Nawaz, H.; Shad, M.A.; Muzaffar, S. Phytochemical Composition and Antioxidant Potential of Brassica. In Brassica Germplasm—Characterization, Breeding and Utilization; InTech: Houston, TX, USA, 2018. [Google Scholar] [CrossRef]
- Serrano, C.; Matos, O.; Teixeira, B.; Ramos, C.; Neng, N.; Nogueira, J.; Nunes, M.L.; Marques, A. Antioxidant and antimicrobial activity of Satureja montana L. extracts. J. Sci. Food Agric. 2011, 91, 1554–1560. [Google Scholar] [CrossRef] [PubMed]
- Arnao, M.B. Some Methodological Problems in the Determination of Antioxidant Activity Using Chromogen Radicals: A Practical Case. Trends Food Sci. Technol. 2000, 11, 419–421. [Google Scholar] [CrossRef]
- Prior, R.L.; Wu, X.; Schaich, K. Standardized Methods for the Determination of Antioxidant Capacity and Phenolics in Foods and Dietary Supplements. J. Agric. Food Chem. 2005, 53, 4290–4302. [Google Scholar] [CrossRef] [PubMed]
- Beltagy, A.M. Investigation of new antimicrobial and antioxidant activities of Brassica rapa L. Int. J. Pharm. Pharm. Sci. 2014, 6, 19–25. [Google Scholar]
- Paul, S.; Geng, C.-A.; Yang, T.-H.; Yang, Y.-P.; Chen, J.-J. Phytochemical and health-beneficial progress of turnip (Brassica rapa). J. Food Sci. 2019, 84, 19–30. [Google Scholar] [CrossRef]
- Barbieri, G.; Pernice, R.; Maggio, A.; De Pascale, S.; Fogliano, V. Glucosinolates profile of Brassica rapa L. subsp. Sylvestris L. Janch. var. esculenta Hort. Food Chem. 2008, 107, 1687–1691. [Google Scholar] [CrossRef]

| Accession | County Origin | Portugal Region | Species | Sample Analized | Harvest Year |
|---|---|---|---|---|---|
| BPGV03990 | Braga | North-west | Brassica rapa | turnip | 2019 |
| BPGV12376 | Bragança | Far northeast | Brassica rapa | turnip | 2019 |
| BPGV12405 | Bragança | Far northeast | Brassica rapa | turnip | 2019 |
| BPGV12239 | Bragança | Far northeast | Brassica rapa | turnip | 2019 |
| BPGV12296 | Bragança | Far northeast | Brassica rapa | turnip | 2019 |
| BPGV16127 | Guarda | Central-north inland | Brassica rapa | turnip | 2019 |
| BPGV11177 | Leiria | Littoral western central | Brassica rapa | turnip | 2019 |
| BPGV05875 | Santarém | Centre | Brassica rapa | turnip | 2019 |
| BPGV11111 | Santarém | Centre | Brassica rapa | turnip | 2019 |
| BPGV04542 | Viseu | Centre-north | Brassica rapa | turnip | 2019 |
| BPGV07301 | Vila Real | Northern inland | Brassica rapa | turnip | 2019 |
| BPGV07480 | Vila real | Northern inland | Brassica rapa | turnip | 2019 |
| BPGV06987A | Bragança | Far northeast | Brassica napus var. napobrassica (L.) Rchb. | turnip | 2019 |
| BPGV06987B | Bragança | Far northeast | Brassica napus var. napobrassica (L.) Rchb. | turnip | 2019 |
| BPGV02906 | Vila real | Northern inland | Brassica napus var. napobrassica (L.) Rchb. | turnip | 2019 |
| BPGV05888 | Santarém | Centre | Brassica rapa sylvestris | turnip | 2019 |
| BPGV12636 | Aveiro | Littoral Centre | Brassica rapa | turnip | 2020 |
| BPGV12736 | Aveiro | Littoral Centre | Brassica rapa | turnip | 2020 |
| BPGV12737 | Aveiro | Littoral Centre | Brassica rapa | turnip | 2020 |
| BPGV12878 | Aveiro | Littoral Centre | Brassica rapa | turnip | 2020 |
| BPGV12914 | Aveiro | Littoral Centre | Brassica rapa | turnip | 2020 |
| BPGV05049 | Bragança | Far northeast | Brassica rapa | turnip | 2020 |
| BPGV13051 | Guarda | Central-north inland | Brassica rapa | turnip | 2020 |
| BPGV13083 | Guarda | Central-north inland | Brassica rapa | turnip | 2020 |
| BPGV05096 | Guarda | Central-north inland | Brassica rapa | turnip | 2020 |
| BPGV11840 | Portalegre | Centre-south inland | Brassica rapa | turnip | 2020 |
| BPGV06207 | Viseu | Centre-north | Brassica rapa | turnip | 2020 |
| BPGV16434 | Viseu | Centre-north | Brassica rapa | turnip | 2020 |
| BPGV04856 | Guarda | Central-north inland | Brassica rapa subsp. rapa | turnip | 2020 |
| BPGV04075 | Guarda | Central-north inland | Brassica rapa subsp. rapa | turnip | 2020 |
| BPGV02160 | Braga | North-west | Brassica rapa subsp. rapa | turnip | 2020 |
| BPGV02578 | Braga | North-west | Brassica napus | seeds | 2021 |
| BPGV04238 | Braganca | Far northeast | Brassica napus | seeds | 2021 |
| BPGV02636 | Viana Castelo | Littoral North-west | Brassica napus | seeds | 2021 |
| BPGV02740 | Viana Castelo | Littoral North-west | Brassica napus | seeds | 2021 |
| BPGV02745 | Viana Castelo | Littoral North-west | Brassica napus | seeds | 2021 |
| BPGV02707 | Vila Real | Northern inland | Brassica napus | seeds | 2021 |
| Semisystematic Name (Glucosinolate Group) | Trivial Name (Abbreviation) | TR (min) | Ionic Formula | [M − H]− (m/z (Δ ppm) | MS/MS [m/z (Δ ppm) (Attribution)] |
|---|---|---|---|---|---|
| 2(R)-2-Hydroxyl but- -3-enyl (hydroxyalkenyl) | Progoitrin (PRO) | 1.38 | [C11H19N1O10S2]− | 388.0380; −0.7 | 308.0805; −2.4; (C11H18N1O7S1)− 301.0124; −0.5; (C8H13O8S2)− 274.9904; −1.0; (C6H11O8S2)− 259.0131; −0.6; (C6H11O9S1)− 241.0029; −2.3; (C6H9O8S1)− 195.0334; −0.6; (C6H11O5S1)− 194.9794; −1.3; (C6H7O4S2)−*F2b 179.0560; −0.7; (C5H11O6)− 146.0278; +1.9; (C5H8N1O2S1)−*F1 135.9706; +3.1; (C2H2N1O4S1)− |
| 2-Hydroxypent- -4-enyl (hydroxyalkenyl) | Gluco- napoleiferin (GNL) | 1.45 | [C12H20N1O10S2]− | 402.0543; −2.3 | 322.0950; −2.4; (C12H20N1O7S1)− 274.9902; −1.9; (C6H11O8S2)− 259.0131; −2.1; (C6H11O9S1)− 208.9952; −2.2; (C6H9O4S2)−*F2b 195.0333; −0.9; (C6H11O5S1)− 160.0437; +0.2; (C6H10N1O2S)−*F1 135.9713; −2.0; (C2H2N1O4S)− |
| But-3-enyl (alkenyl) | Gluconapin (GNA) | 1.47 | [C11H18N1O9S2]− | 372.0432; −1.0 | 292.0861; −0.4; (C11H18N1O6S)− 274.9905; −1.5; (C6H11O8S2)− 259.0133; −1.3; (C6H11O9S1)− 241.0025; −0.5; (C6H9O8S1)− 195.0333; −0.9; (C6H11O5S1)− 178.9834; −4.4; (C5H7O3S2)−*F2b 130.0334; −1.2; (C5H8NOS)−*F1 |
| 4-Hydroxyindol- -3-ylmethyl (indole) | 4-Hydroxy- glucobrassicin (4-OHGBS) | 1.55 | [C16H19N2O10S2]− | 463.0491; −0.9 | 383.0921; −0.6; (C16H19N2O7S)− 274.9903; −0.4; (C6H11O8S2)− 267.0081; −0.2; (C10H7N2O5S)− *F2a 259.0128; −0.4; (C6H11O9S1)− 221.0390; −0.3; (C10H9N2O2S1)−*F1 |
| Pent-4-enyl (alkenyl) | Gluco- brassicanapin (GBN) | 2.20 | [C12H20N1O9S2]− | 386.0583; −0.6 | 306.1011; −1.1; (C12H20N1O6S)− 274.9900; −0.3; (C6H11O8S2)− 259.0131; −0.6; (C6H11O9S1)− 241.0021; −1.1; (C6H9O8S1)− 195.0332; −0.4; (C6H11O5S)− 190.0187; −3.8; (C6H8NO4S1)−*F2a 144.0484; −2.9; (C6H10NOS1)−*F1 |
| Hydroxyindol- -3-ylmethyl (indole) | Hydroxy- glucobrassicin isomer (OHGBS isomer) | 3.02 | [C16H19N2O10S2]− | 463.0493; −1.1 | 383.0917; −0.4; (C16H19N2O7S)− 274.9902; −0.3; (C6H11O8S2)− 267.0092; −4.2; (C10H7N2O5S)−*F2a 259.0131; −0.6; (C6H11O9S1)− 221.0391; −0.4; (C10H9N2O4S1)−*F1 195.0339; −4.4; (C6H11O5S)− |
| 4(Methylsulphanyl)- -butyl (sulphur containing) | Glucoerucin (GER) | 3.21 | [C12H22N1O9S3]− | 420.0467; −1.2 | 340.0913; −5.4; (C12H22N1O6S2)− 331.0716; −1.4; (C11H22N1O6S2)− 274.9900; −1.5; (C6H11O8S2)− 259.0140; −4.3; (C6H11O9S1)− 226.9884; −3.5; (C6H11O3S3)−*F2b 195.0340; −4.9; (C6H11O5S)− 178.0366; −0.1; (C6H12N1OS2)−*F1 130.0334; −1.2; (C5H8NOS)− |
| Pentenyl (alkenyl) | Gluco- brassicanapin isomer (GBN isomer) | 3.38 | [C12H20N1O9S2]− | 386.0595; −2.6 | 306.1024; −2.5; (C12H20N1O6S)− 274.9905; −2.6; (C6H11O8S2)− 259.0133; −1.3; (C6H11O9S1)− 241.0035; −2.0; (C6H9O8S1)− 195.0339; −2.5; (C6H11O5S)− 190.0187; −3.8; (C6H8NO4S)−*F2a 144.0487; −0.9; (C6H10NOS)−*F1 |
| Indol-3-ylmethyl (indole) | Glucobrassicin (GBS) | 3.40 | [C16H19N2O9S2]− | 447.0547; −2.0 | 367.0962; −2.0; (C16H19N2O6S1)− 274.9909; −3.1; (C6H11O8S2)− 259.0138; −3.4; (C6H11O9S1)− 253.9961; −3.9; (C10H8N1O3S2)−*F2b 205.0440; −0.5; (C10H9N2OS)−*F1 172.0223; −2.1; (C10H6N1S1)− |
| 2(R)-Hydroxy- -2-phenylethyl (aromatic) | Glucobarbarin (GBA) | 3.70 | [C15H20N1O10S2]− | 438.0534; −3.7 | 358.0996; −7.4; (C15H20N1O7S1)− 274.9909; −3.1; (C6H11O8S2)− 259.0140; −4.1; (C6H11O9S1)− 244.9941; −2.8; (C9H9O4S2)−*F2b 196.0442; −2.0; (C9H10NO2S1)−*F1 172.0223; −2.1; (C10H6N1S1)− |
| 5-(Methylsulfanyl)- -pentyl (sulfur containing) | Glucoberteroin (GOB) | 4.46 | [C13H24N1O9S3]− | 434.0626; −1.7 | 354.1060; −2.8; (C13H24N1O6S2)− 274.9903; −0.7; (C6H11O8S2)− 259.0140; −1.8; (C6H11O9S1)− 241.0035; −1.1; (C7H13O3S3)−*F2b 195.0335; −1.3; (C6H11O5S1)− 192.0524; −1.3; (C7H14N1O1S2)−*F1 |
| 2-Phenylethyl (aromatic) | Gluconasturtiin (GST) | 4.47 | [C15H20N1O9S2]− | 422.0593; −2.0 | 342.1026; −2.8; (C15H20N1O6S1)− 274.9912; −3.9; (C6H11O8S2)− 259.0295; −3.6; (C6H11O9S1)− 241.0032; −3.5; (C6H9O8S1)− 229.0006; −3.1; (C9H9O3S2)−*F2b 226.0184; −1.8; (C9H8N1O4S1)−*F2a 180.0485; −1.8; (C9H10N1O1S1)−*F1 |
| 4-Methoxyindol- -3-ylmethyl (indole) | 4-methoxy- glucobrassicin (NGBS) | 4.78 | [C17H21N2O10S2]− | 477.0655; −2.5 | 284.0062; −2.0; (C11H10N1O4S2)−*F2b 274.9911; −3.9; (C6H11O8S2)− 259.0138; −0.9; (C6H11O9S1)− 235.0560; −5.8; (C11H11N2O2S1)−*F1 205.0441; −0.2; (C10H9N2O1S1)− 203.0286; −0.5; (C10H7N2O1S1)− |
| 1-Methoxyindol- -3-ylmethyl (indole) | 1-methoxy- -glucobrassicin (NGBS isomer) | 5.62 | [C17H21N2O10S2]− | 477.0664; −3.7 | 446.0480; −4.6; (C16H18N2O9S2)−• 274.9900; −0.9; (C6H11O8S2)− 259.0136; −2.6; (C6H11O9S1)− 235.0559; −5.2; (C11H11N2O2S1)−*F1 205.0437; −1.9; (C10H9N2O1S1)− 195.0344; −6.0; (C6H11O5S)− |
| Hydroxyciannamic acids | |||||
| Sucrose | 1.13 | [C12H21O11]− | 341.1091; −0.5 | 179.0562; −0.7; (C8H11O6)− | |
| Gentisoyl glucoside | 1.93 | [C13H15O9]− | 315.0726; −1.5 | 153.02187; +4,7; (C7H5O4)− | |
| 1-O-Sinapolyl- glucose | 4.80 | [C17H21O10]− | 385.1139; −0.2 | 205.0510; −1.8; (C11H9O4)− 179.0723; −5.3; (C10H11O3)− 164.0480; −0.4; (C9H8O3)−• | |
| Sinapic acid | 6.17 | [C11H11O5]− | 223.0612; −1.9 | 208.0375; −0.7; (C10H8O5)−• 179.0708; −7.7; (C10H11O3)− 164.0469; −5.7; (C9H8O3)−• | |
| 1,2-Disinapoyl-gentiobiose | 6.73 | [C34H41O19]− | 753.2252; −0.5 | 529.1571; −0.8; (C23H29O14)− 223.0615; −1.3; (C11H11O5)− 205.0510; −1.4; (C11H9O4)− | |
| 1-Feruloyl- -1-sinapoyl- gentiobiose | 6.88 | [C33H39O18]− | 723.2145; −0.4 | 449.1471; −2.7; (C22H27O13)− 223.0615; −3.6; (C11H11O5)− 205.0517; −5.0; (C11H9O4)− 175.0463; −1.6; (C10H7O3)− | |
| 1,2,2′-Trisinapoyl- Gentiobiose | 7.46 | [C45H51O23]− | 959.2855; −2.5 | 735.2155; −1.0; (C34H39O18)− 529.1578; −2.8; (C23H29O14)− 223.0622; −4.7; (C11H11O5)− 205.0505; −0.5; (C11H9O4)− | |
| 1,2′-Disinapoyl-2- -feruloyl- Gentiobiose | 8.15 | [C44H49O22]− | 929.2721; −3.8 | 705.2057; −2.9; (C33H37O17)− 529.1575; −2.3; (C23H29O14)− 205.0518; −5.5; (C11H9O4)− |
| Accession | TPC (mg GAE·g−1) DW | FRAP (µmolFe2+·g−1) DW | DPPH (mmolTE·g−1) DW |
|---|---|---|---|
| BPGV03990 | 7.85 ± 0.39 a,b,c,d,e,f | 79.4 ± 2.99 a,b | 157.59 ± 0.16 b,c |
| BPGV12376 | 9.66 ± 0.11 c,d,e,f,g,h,i | 73.50 ± 4.65 a,b,c,d | 208.28 ± 8.77 b,c,d,e |
| BPGV12405 | 11.91 ± 1.19 h,i,j,l | 102.96 ± 3.77 a,b,c | 177.96 ± 1.61 b,c,d,e |
| BPGV12239 | 8.66 ± 2.60 a,b,c,d,e,f,g,h | 51.08 ± 11.93 a,b,c,d | 187.11 ± 2.01 b,c,d,e |
| BPGV12296 | 10.47 ± 0.77 f,g,h,i,j | 88.83 ± 12.23 a,b,c | 212.60 ± 0.08 b,c,d,e |
| BPGV16127 | 10.05 ± 0.54 d,e,f,g,h,i | 97.64 ± 3.32 a | 202.36 ± 1.69 b,c,d,e |
| BPGV11177 | 8.17 ± 0.06 a,b,c,d,e,f,g | 63.61 ± 6.85 a | 186.72 ± 4.02 b,c,d,e |
| BPGV05875 | 12.10 ± 0.18 h,i,j,l | 106.41 ± 0.29 a,b,c,d | 188.48 ± 0.08 b,c,d,e |
| BPGV11111 | 11.76 ± 2.52 h,i,j,l | 74.80 ± 21.11 a | 179.15± 9.41 b,c,d,e |
| BPGV04542 | 9.13 ± 0.45 c,d,e,f,g,h,i | 68.48 ± 2.79 a | 184.10 ± 6.44 b,c,d,e |
| BPGV07301 | 5.38 ± 0.97 a | 59.70 ± 16.08 a,b,c,d | 230.40 ± 7.40 e |
| BPGV07480 | 5.61 ± 0.88 a,b | 51.03 ± 1.50 a,b,c | 194.28 ± 30.97 b,c,d,e |
| BPGV06987A | 6.89 ± 0.22 a,b,c,d | 60.68 ± 4.57 a,b,c,d | 202.36 ± 1.69 b,c,d,e |
| BPGV06987B | 10.52 ± 0.49 i,j | 72.00 ± 5.70 a,b,c | 195.65 ± 5.07 b,c,d,e |
| BPGV02906 | 6.72 ± 0.67 a,b,c,d | 55.17 ± 4.94 a,b,c | 213.91 ± 3.38 b,c,d,e |
| BPGV05888 | 7.01 ± 0.55 a,b,c,d,e | 59.74 ± 8.60 a,b,c | 196.59 ± 6.80 b,c,d,e |
| BPGV12636 | 7.91 ± 0.51 a,b,c,d,e,f | 64.43 ± 7.80 a,b,c,d,e | 210.08 ± 7.56 b,c,d,e |
| BPGV12736 | 6.41 ± 0.09 a,b,c | 44.01 ± 10.69 a,b,c | 156.91 ± 2.95 b |
| BPGV12737 | 13.72 ± 0.71 j,l,m | 123.92 ± 14.21 e | 228.3 ± 0.16 e |
| BPGV12878 | 11.73 ± 0.07 h,i,j,l | 85.04 ± 2.73 c,d,e | 225.44 ± 1.50 e |
| BPGV12914 | 18.00 ± 0.64 n | 127.97 ± 0.15 e | 218.72 ± 2.52 d,e |
| BPGV05049 | 12.58 ± 0.00 i,j,l | 69.31 ± 7.01 a,b,c,d | 94.00 ± 17.70 a |
| BPGV13051 | 8.75 ± 0.42 a,b,c,d,e,f,g,h | 65.94 ± 22.57 a,b,c,d | 193.16 ± 32.34 b,c,d,e |
| BPGV13083 | 15.12 ± 0.03 l,m,n | 124.06 ± 38.57 e | 184.67 ± 51.43 b,c,d,e |
| BPGV05096 | 9.79 ± 0.29 c,d,e,f,g,h,i | 83.75 ± 5.55 b,c,d,e | 216.71 ± 6.22 d,e |
| BPGV11840 | 16.90 ± 0.83 m,n | 104.37 ± 0.42 d,e | 210.23 ± 3.16 b,c,d,e |
| BPGV16434 | 7.09 ± 0.46 a,b,c,d,e,f | 56.92 ± 20.44 a,b,c,d | 168.89 ± 1.45 b,c,d |
| BPGV06207 | 16.92 ± 0.66 m,n | 130.83 ± 24.98 e | 232.53 ± 1.56 e |
| BPGV04856 | 8.99 ± 0,45 h,i,j,l | 63.61 ± 5.34 a,b,c,d,e | 190.51 ± 11.10 b,c,d,e |
| BPGV04075 | 12.06 ± 0.58 b,c,d,e,f,g,h | 70.51 ± 1.21 a,b,c,d | 211.63 ± 9.01 b,c,d,e |
| BPGV02160 | 11.43 ± 0.44 g,h,i,j | 81.92 ± 6.55 b,c,d,e | 221.07 ± 1.13 d,e |
| Accession | TPC (mg GAE·g−1) DW | FRAP (mmolFe2+·g−1) DW | DPPH (mmol·TE g−1) DW |
|---|---|---|---|
| BPGV02578 | 27.09 ± 2.69 a | 3.18 ± 0.15 a | 330.88 ± 40.58 a |
| BPGV04238 | 24.90 ± 5.16 a | 3.01 ± 0.35 a | 280.38 ± 65.11 a |
| BPGV02636 | 23.04 ± 0.94 a | 2.74 ± 0.16 a | 246.44 ± 2.59 a |
| BPGV02740 | 27.39 ± 0.20 a | 3.12± 0.50 a | 314.81 ± 12.71 a |
| BPGV02745 | 25.12 ± 0.37 a | 2.89 ± 0.01 a | 296.73 ± 8.55 a |
| BPGV02707 | 24.40 ± 0.72 a | 2.99 ± 0.66 a | 286.37 ± 8.36 a |
| Gram-Negative Bacteria | Gram-Positive Bacteria | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Sample | Enterobacter Cloacae | Escherichia coli | Pseudomonas aeruginosa | Salmonella enterica | Yersinia enterocolitica | Bacillus cereus | Listeria monocytogenes | Staphylococcus aureus | |
| BPGV03990 | MIC | >10 | 10 | >10 | 2.5 | >10 | >10 | >10 | 2.5 |
| MBC | >10 | >10 | >10 | >10 | >10 | >10 | >10 | >10 | |
| BPGV12239 | MIC | >10 | 5 | >10 | 2.5 | >10 | >10 | >10 | 5 |
| MBC | >10 | >10 | >10 | >10 | >10 | >10 | >10 | >10 | |
| BPGV12296 | MIC | >10 | 10 | >10 | 5 | 10 | >10 | >10 | 5 |
| MBC | >10 | >10 | >10 | >10 | >10 | >10 | >10 | >10 | |
| BPGV12376 | MIC | >10 | 10 | >10 | 5 | 10 | >10 | >10 | 5 |
| MBC | >10 | >10 | >10 | >10 | >10 | >10 | >10 | >10 | |
| BPGV12405 | MIC | >10 | 10 | >10 | 5 | >10 | >10 | >10 | 2.5 |
| MBC | >10 | >10 | >10 | >10 | >10 | >10 | >10 | >10 | |
| BPGV16127 | MIC | >10 | 10 | >10 | 5 | 10 | >10 | >10 | 2.5 |
| MBC | >10 | >10 | >10 | >10 | >10 | >10 | >10 | >10 | |
| BPGV11177 | MIC | >10 | 10 | >10 | 5 | 10 | >10 | >10 | 2.5 |
| MBC | >10 | >10 | >10 | >10 | >10 | >10 | >10 | >10 | |
| BPGV05875 | MIC | >10 | 10 | >10 | 5 | 5 | >10 | >10 | 2.5 |
| MBC | >10 | >10 | >10 | >10 | >10 | >10 | >10 | >10 | |
| BPGV11111 | MIC | >10 | 10 | >10 | 5 | >10 | >10 | >10 | 2.5 |
| MBC | >10 | >10 | >10 | >10 | >10 | >10 | >10 | >10 | |
| BPGV7301 | MIC | >10 | 10 | >10 | 2.5 | 10 | >10 | >10 | 2.5 |
| MBC | >10 | >10 | >10 | >10 | >10 | >10 | >10 | >10 | |
| BPGV07480 | MIC | >10 | 10 | >10 | 2.5 | 10 | >10 | >10 | 2.5 |
| MBC | >10 | >10 | >10 | >10 | >10 | >10 | >10 | >10 | |
| BPGV04542 | MIC | >10 | 10 | >10 | 10 | 10 | >10 | >10 | 2.5 |
| MBC | >10 | >10 | >10 | >10 | >10 | >10 | >10 | >10 | |
| BPGV05888 | MIC | >10 | >10 | >10 | 5 | >10 | >10 | >10 | 2.5 |
| MBC | >10 | >10 | >10 | >10 | >10 | >10 | >10 | >10 | |
| BPGV06987 A | MIC | >10 | 10 | >10 | 5 | >10 | >10 | >10 | 5 |
| MBC | >10 | >10 | >10 | >10 | >10 | >10 | >10 | >10 | |
| BPGV06987 B | MIC | >10 | 10 | >10 | 5 | >10 | >10 | >10 | 10 |
| MBC | >10 | >10 | >10 | >10 | >10 | >10 | >10 | >10 | |
| BPGV02906 | MIC | >10 | 5 | >10 | 5 | >10 | >10 | >10 | 2.5 |
| MBC | >10 | >10 | >10 | >10 | >10 | >10 | >10 | >10 | |
| Streptomicin 1 mg/mL | MIC | 0.007 | 0.01 | 0.06 | 0.007 | 0.007 | 0.007 | 0.007 | 0.007 |
| MBC | 0.007 | 0.01 | 0.06 | 0.007 | 0.007 | 0.007 | 0.007 | 0.007 | |
| Methicilin 1 mg/mL | MIC | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | 0.007 |
| MBC | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | 0.007 | |
| Ampicillin 10 mg/mL | MIC | 0.15 | 0.15 | 0.63 | 0.15 | 0.15 | n.d. | 0.15 | 0.15 |
| MBC | 0.15 | 0.15 | 0.63 | 0.15 | 0.15 | n.d. | 0.15 | 0.15 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Serrano, C.; Oliveira, M.C.; Lopes, V.R.; Soares, A.; Molina, A.K.; Paschoalinotto, B.H.; Pires, T.C.S.P.; Serra, O.; Barata, A.M. Chemical Profile and Biological Activities of Brassica rapa and Brassica napus Ex Situ Collection from Portugal. Foods 2024, 13, 1164. https://doi.org/10.3390/foods13081164
Serrano C, Oliveira MC, Lopes VR, Soares A, Molina AK, Paschoalinotto BH, Pires TCSP, Serra O, Barata AM. Chemical Profile and Biological Activities of Brassica rapa and Brassica napus Ex Situ Collection from Portugal. Foods. 2024; 13(8):1164. https://doi.org/10.3390/foods13081164
Chicago/Turabian StyleSerrano, Carmo, M. Conceição Oliveira, V. R. Lopes, Andreia Soares, Adriana K. Molina, Beatriz H. Paschoalinotto, Tânia C. S. P. Pires, Octávio Serra, and Ana M. Barata. 2024. "Chemical Profile and Biological Activities of Brassica rapa and Brassica napus Ex Situ Collection from Portugal" Foods 13, no. 8: 1164. https://doi.org/10.3390/foods13081164
APA StyleSerrano, C., Oliveira, M. C., Lopes, V. R., Soares, A., Molina, A. K., Paschoalinotto, B. H., Pires, T. C. S. P., Serra, O., & Barata, A. M. (2024). Chemical Profile and Biological Activities of Brassica rapa and Brassica napus Ex Situ Collection from Portugal. Foods, 13(8), 1164. https://doi.org/10.3390/foods13081164









