Application of Strain Selection Technology in Alcoholic Beverages: A Review
Abstract
:1. Introduction
2. Type and Diversity Characteristics of Microorganisms in Alcoholic Beverages
2.1. Types and Diversity Characteristics of Microorganisms in Yellow Wine
2.2. Types and Diversity Characteristics of Microorganisms in Wine
2.3. Types and Diversity Characteristics of Microorganisms in Beer
3. Strain Selection Techniques
3.1. Natural Selection
3.2. Induced Mutation
3.3. Crossing Techniques
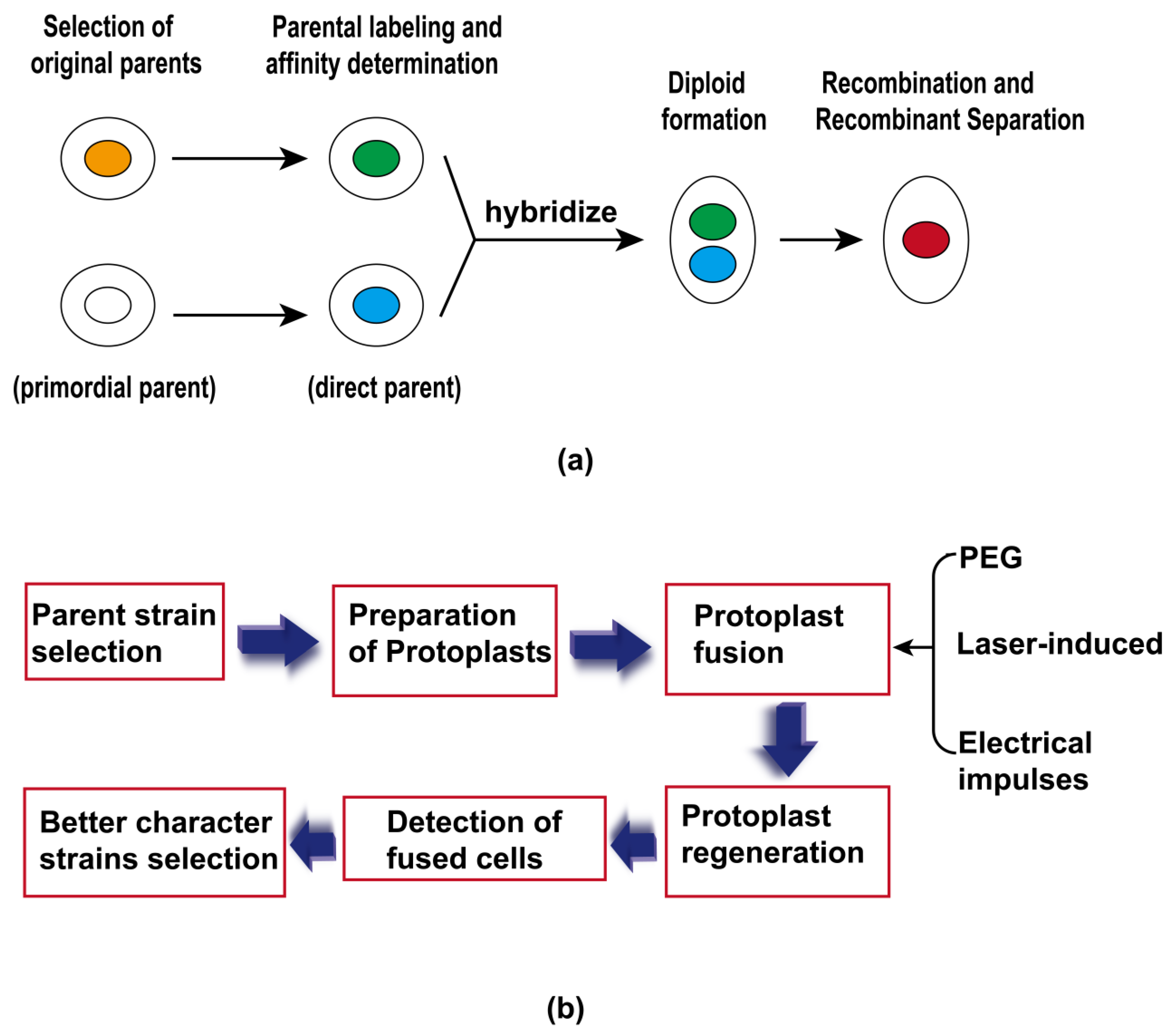
3.4. Protoplast Fusion Technique
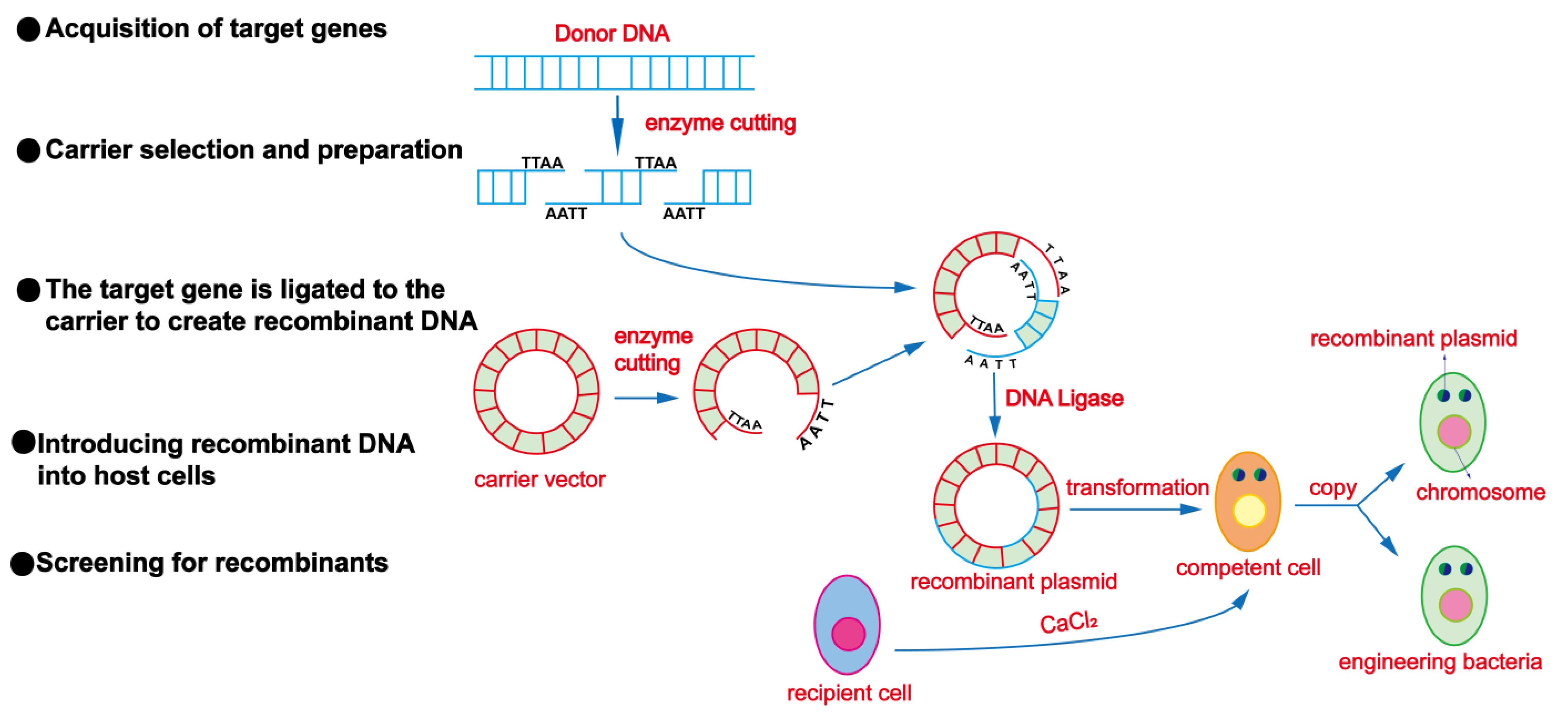
3.5. Genetic Engineering Techniques
3.6. Genome Shuffling Technology
4. Application of Strain Selection Technology in Improving the Quality of Alcoholic Beverages
5. Conclusions and Outlook
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Patel, P.; Butani, K.; Kumar, A.; Singh, S.; Prajapati, B.G. Effects of Fermented Food Consumption on Non-Communicable Diseases. Foods 2023, 12, 687. [Google Scholar] [CrossRef] [PubMed]
- Marco, M.L.; Heeney, D.; Binda, S.; Cifelli, C.J.; Cotter, P.D.; Foligné, B.; Gänzle, M.; Kort, R.; Pasin, G.; Pihlanto, A.; et al. Health benefits of fermented foods: Microbiota and beyond. Curr. Opin. Biotechnol. 2017, 44, 94–102. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.; Qi, X.; Chen, M.; Lu, D.; Chen, B. Discrimination of Chinese yellow wine from different origins based on flavor fingerprint. Acta Chromatogr. 2019, 32, 1–6. [Google Scholar] [CrossRef]
- Zhou, M.J.; Bu, T.T.; Zheng, J.X.; Liu, L.; Yu, S.F.; Li, S.S.; Wu, J.P. Peptides in Brewed Wines: Formation, Structure, and Function. J. Agric. Food Chem. 2021, 69, 2647–2657. [Google Scholar] [CrossRef] [PubMed]
- Caspermeyer, J. The Evolution of Beer. Mol. Biol. Evol. 2016, 33, 295. [Google Scholar] [CrossRef] [PubMed]
- Hinojosa-Avila, C.R.; Garcia-Gamboa, R.; Chedraui-Urrea, J.J.T.; Garcia-Cayuela, T. Exploring the potential of probiotic-enriched beer: Microorganisms, fermentation strategies, sensory attributes, and health implications. Food Res. Int. 2024, 175, 113717. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.; Li, L.M.; Xiang, P.; Zhang, T.; Peng, L.X.; Zou, L.; Li, Q. Phages in Fermented Foods: Interactions and Applications. Fermentation 2023, 9, 201. [Google Scholar] [CrossRef]
- van Wyk, N.; Grossmann, M.; Wendland, J.; von Wallbrunn, C.; Pretorius, I.S. The Whiff of Wine Yeast Innovation: Strategies for Enhancing Aroma Production by Yeast during Wine Fermentation. J. Agric. Food Chem. 2019, 67, 13496–13505. [Google Scholar] [CrossRef]
- Mendes Ferreira, A.; Mendes-Faia, A. The Role of Yeasts and Lactic Acid Bacteria on the Metabolism of Organic Acids during Winemaking. Foods 2020, 9, 1231. [Google Scholar] [CrossRef]
- Munford, A.R.G.; Chaves, R.D.; Sant’Ana, A.S. Inactivation kinetics of beer spoilage bacteria (Lactobacillus brevis, Lactobacillus casei, and Pediococcus damnosus) during acid washing of brewing yeast. Food Microbiol. 2020, 91, 103513. [Google Scholar] [CrossRef]
- Steensels, J.; Meersman, E.; Snoek, T.; Saels, V.; Verstrepen, K.J. Large-scale selection and breeding to generate industrial yeasts with superior aroma production. Appl. Environ. Microbiol. 2014, 80, 6965–6975. [Google Scholar] [CrossRef]
- Su, J.; Wang, T.; Wang, Y.; Li, Y.Y.; Li, H. The use of lactic acid-producing, malic acid-producing, or malic acid-degrading yeast strains for acidity adjustment in the wine industry. Appl. Microbiol. Biotechnol. 2014, 98, 2395–2413. [Google Scholar] [CrossRef] [PubMed]
- Tilloy, V.; Ortiz-Julien, A.; Dequin, S. Reduction of ethanol yield and improvement of glycerol formation by adaptive evolution of the wine yeast Saccharomyces cerevisiae under hyperosmotic conditions. Appl. Environ. Microbiol. 2014, 80, 2623–2632. [Google Scholar] [CrossRef]
- Su, Y.; Gamero, A.; Rodríguez, M.E.; Lopes, C.A.; Querol, A.; Guillamón, J.M. Interspecific hybridisation among diverse Saccharomyces species: A combined biotechnological solution for low-temperature and nitrogen-limited wine fermentations. Int. J. Food Microbiol. 2019, 310, 108331. [Google Scholar] [CrossRef]
- Yang, Y.; Hu, W.; Xia, Y.; Mu, Z.; Tao, L.; Song, X.; Zhang, H.; Ni, B.; Ai, L. Flavor Formation in Chinese Rice Wine (Huangjiu): Impacts of the Flavor-Active Microorganisms, Raw Materials, and Fermentation Technology. Front. Microbiol. 2020, 11, 580247. [Google Scholar] [CrossRef] [PubMed]
- Shuai, H.; Xiangzhao, M.; Pei, L.; Hong, L.; Zuyuan, D.; Ning, L.; Jichen, H.; Cuifang, Q. Research into the functional components and antioxidant activities of North China rice wine (Ji Mo Lao Jiu). Food Sci. Nutr. 2013, 1, 307–314. [Google Scholar]
- Lv, X.-C.; Chen, Z.-C.; Jia, R.-B.; Liu, Z.-B.; Zhang, W.; Chen, S.-J.; Rao, P.-F.; Ni, L. Microbial community structure and dynamics during the traditional brewing of Fuzhou Hong Qu glutinous rice wine as determined by culture-dependent and culture-independent techniques. Food Control 2015, 57, 216–224. [Google Scholar] [CrossRef]
- Wu, Z.; Xu, E.; Long, J.; Pan, X.; Xu, X.; Jin, Z.; Jiao, A. Comparison between ATR-IR, Raman, concatenated ATR-IR and Raman spectroscopy for the determination of total antioxidant capacity and total phenolic content of Chinese rice wine. Food Chem. 2016, 194, 671–679. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.; Meng, L.; Sun, Z.; Sun, S.; Huang, X.; Lin, N.; Zhang, J.; Lu, W.; Yang, Q.; Chi, J.; et al. Yellow Wine Polyphenolic Compound Protects Against Doxorubicin-Induced Cardiotoxicity by Modulating the Composition and Metabolic Function of the Gut Microbiota. Circ. Heart Fail. 2021, 14, e008220. [Google Scholar] [CrossRef]
- Yang, J.; Song, J.; Zhou, J.; Lin, H.; Wu, Z.; Liu, N.; Xie, W.; Guo, H.; Chi, J. Functional components of Chinese rice wine can ameliorate diabetic cardiomyopathy through the modulation of autophagy, apoptosis, gut microbiota, and metabolites. Front. Cardiovasc. Med. 2022, 9, 940663. [Google Scholar] [CrossRef]
- Zhao, C.; Su, W.; Mu, Y.; Luo, L.; Zhao, M.; Qiu, S.; Su, G.; Jiang, L. Effects of Jiuqu inoculating Rhizopus oryzae Q303 and Saccharomyces cerevisiae on chemical components and microbiota during black glutinous rice wine fermentation. Int. J. Food Microbiol. 2023, 385, 110012. [Google Scholar] [CrossRef] [PubMed]
- Tian, S.; Zeng, W.; Fang, F.; Zhou, J.; Du, G. The microbiome of Chinese rice wine (Huangjiu). Curr. Res. Food Sci. 2022, 5, 325–335. [Google Scholar] [CrossRef] [PubMed]
- Londoño-Hernández, L.; Ramírez-Toro, C.; Ruiz, H.A.; Ascacio-Valdés, J.A.; Aguilar-Gonzalez, M.A.; Rodríguez-Herrera, R.; Aguilar, C.N. Rhizopus oryzae—Ancient microbial resource with importance in modern food industry. Int. J. Food Microbiol. 2017, 257, 110–127. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.; Wu, F.; Guo, J.; Ye, M.; Hu, H.; Guo, J.; Liu, X. Effects of glutinous rice protein components on the volatile substances and sensory properties of Chinese rice wine. J. Sci. Food Agric. 2020, 100, 3297–3307. [Google Scholar] [CrossRef] [PubMed]
- Chang, P.K.; Scharfenstein, L.L.; Solorzano, C.D.; Abbas, H.K.; Hua, S.S.; Jones, W.A.; Zablotowicz, R.M. High sequence variations in the region containing genes encoding a cellular morphogenesis protein and the repressor of sexual development help to reveal origins of Aspergillus oryzae. Int. J. Food Microbiol. 2015, 200, 66–71. [Google Scholar] [CrossRef] [PubMed]
- Rui, Y.; Wan, P.; Chen, G.; Xie, M.; Sun, Y.; Zeng, X.; Liu, Z. Analysis of bacterial and fungal communities by Illumina MiSeq platforms and characterization of Aspergillus cristatus in Fuzhuan brick tea. LWT 2019, 110, 168–174. [Google Scholar] [CrossRef]
- Liu, S.; Yang, L.; Zhou, Y.; He, S.; Li, J.; Sun, H.; Yao, S.; Xu, S. Effect of mixed moulds starters on volatile flavor compounds in rice wine. LWT 2019, 112, 108215. [Google Scholar] [CrossRef]
- Liu, Y.; Yue, Z.; Jinglei, L.; Shan, L.; Shudong, H.; Hanju, S.; Shengfei, Y.; Shangying, X. Effect of enzymes addition on the fermentation of Chinese rice wine using defined fungal starter. LWT 2021, 143, 111101. [Google Scholar]
- Yang, Y.; Zhong, H.; Yang, T.; Lan, C.; Zhu, H. Characterization of the key aroma compounds of a sweet rice alcoholic beverage fermented with Saccharomycopsis fibuligera. J. Food Sci. Technol. 2021, 58, 3752–3764. [Google Scholar] [CrossRef] [PubMed]
- Jolly, N.P.; Varela, C.; Pretorius, I.S. Not your ordinary yeast: Non-Saccharomyces yeasts in wine production uncovered. FEMS Yeast Res. 2014, 14, 215–237. [Google Scholar] [CrossRef]
- Pretorius, I.S. Tailoring wine yeast for the new millennium: Novel approaches to the ancient art of winemaking. Yeast 2000, 16, 675–729. [Google Scholar] [CrossRef] [PubMed]
- Pretorius, I.S.; Bauer, F.F. Meeting the consumer challenge through genetically customized wine-yeast strains. Trends Biotechnol. 2002, 20, 426–432. [Google Scholar] [CrossRef] [PubMed]
- Pretorius, I.S.; Curtin, C.D.; Chambers, P.J. The winemaker’s bug: From ancient wisdom to opening new vistas with frontier yeast science. Bioeng. Bugs 2012, 3, 147–156. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.R.; Hong, J.L.; Xu, J.X.; Li, L.; Guo, W.L.; Pan, Y.Y.; Chen, S.J.; Bai, W.D.; Rao, P.F.; Ni, L.; et al. Exploring core functional microbiota responsible for the production of volatile flavour during the traditional brewing of Wuyi Hong Qu glutinous rice wine. Food Microbiol. 2018, 76, 487–496. [Google Scholar] [CrossRef] [PubMed]
- Canonico, L.; Solomon, M.; Comitini, F.; Ciani, M.; Varela, C. Volatile profile of reduced alcohol wines fermented with selected non-Saccharomyces yeasts under different aeration conditions. Food Microbiol. 2019, 84, 103247. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.P.; Mao, J.; Liu, Y.Y.; Meng, X.Y.; Ji, Z.W.; Zhou, Z.L.; Ai-lati, A. Bacterial succession and the dynamics of volatile compounds during the fermentation of Chinese rice wine from Shaoxing region. World J. Microbiol. Biotechnol. 2015, 31, 1907–1921. [Google Scholar] [CrossRef] [PubMed]
- Lv, X.-C.; Huang, R.-L.; Chen, F.; Zhang, W.; Rao, P.-F.; Ni, L. Bacterial community dynamics during the traditional brewing of Wuyi Hong Qu glutinous rice wine as determined by culture-independent methods. Food Control 2013, 34, 300–306. [Google Scholar] [CrossRef]
- Cai, H.; Zhang, T.; Zhang, Q.; Luo, J.; Cai, C.; Mao, J. Microbial diversity and chemical analysis of the starters used in traditional Chinese sweet rice wine. Food Microbiol. 2018, 73, 319–326. [Google Scholar] [CrossRef]
- Liu, H.; Sun, B. Effect of Fermentation Processing on the Flavor of Baijiu. J. Agric. Food Chem. 2018, 66, 5425–5432. [Google Scholar] [CrossRef]
- Zhang, J.; Fang, L.; Huang, X.; Ding, Z.; Wang, C. Evolution of polyphenolic, anthocyanin, and organic acid components during coinoculation fermentation (simultaneous inoculation of LAB and yeast) and sequential fermentation of blueberry wine. J. Food Sci. 2022, 87, 4878–4891. [Google Scholar] [CrossRef]
- Li, P.P.; Su, R.; Wang, Q.; Liu, K.Y.; Yang, H.; Du, W.; Li, Z.A.; Chen, S.; Xu, B.; Yang, W. Comparison of fungal communities and nonvolatile flavor components in black Huangjiu formed using different inoculation fermentation methods. Front. Microbiol. 2022, 13, 955825. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.P.; Yu, J.X.; Wei, X.L.; Ji, Z.W.; Zhou, Z.L.; Meng, X.Y.; Mao, J. Sequencing-based screening of functional microorganism to decrease the formation of biogenic amines in Chinese rice wine. Food Control 2016, 64, 98–104. [Google Scholar] [CrossRef]
- Lv, X.C.; Jiang, Y.J.; Liu, J.; Guo, W.L.; Liu, Z.B.; Zhang, W.; Rao, P.F.; Ni, L. Evaluation of different PCR primers for denaturing gradient gel electrophoresis (DGGE) analysis of fungal community structure in traditional fermentation starters used for Hong Qu glutinous rice wine. Int. J. Food Microbiol. 2017, 255, 58–65. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Mao, J.; Meng, X.; Li, X.; Liu, Y.; Feng, H. Changes in flavour characteristics and bacterial diversity during the traditional fermentation of Chinese rice wines from Shaoxing region. Food Control 2014, 44, 58–63. [Google Scholar] [CrossRef]
- Qingtao, L.; Xinhui, Y.; Qixing, L.; Jianghua, L.; Fang, F.; Guocheng, D.; Zhen, K. Molecular Engineering of Bacillus paralicheniformis Acid Urease To Degrade Urea and Ethyl Carbamate in Model Chinese Rice Wine. J. Agric. Food Chem. 2018, 66, 13011–13019. [Google Scholar]
- Xiaole, X.; Yi, L.; Qingwen, Z.; Yang, H.; Bin, Z. Mixed Starter Culture Regulates Biogenic Amines Formation via Decarboxylation and Transamination during Chinese Rice Wine Fermentation. J. Agric. Food Chem. 2018, 66, 6348–6356. [Google Scholar]
- Wu, P.; Cai, C.; Shen, X.; Wang, L.; Zhang, J.; Tan, Y.; Jiang, W.; Pan, X. Formation of ethyl carbamate and changes during fermentation and storage of yellow rice wine. Food Chem. 2014, 152, 108–112. [Google Scholar] [CrossRef] [PubMed]
- Shalamitskiy, M.Y.; Tanashchuk, T.N.; Cherviak, S.N.; Vasyagin, E.A.; Ravin, N.V.; Mardanov, A.V. Ethyl Carbamate in Fermented Food Products: Sources of Appearance, Hazards and Methods for Reducing Its Content. Foods 2023, 12, 3816. [Google Scholar] [CrossRef] [PubMed]
- Wei, T.Y.; Jiao, Z.H.; Hu, J.J.; Lou, H.H.; Chen, Q.H. Chinese Yellow Rice Wine Processing with Reduced Ethyl Carbamate Formation by Deleting Transcriptional Regulator Dal80p in Saccharomyces cerevisiae. Molecules 2020, 25, 3580. [Google Scholar] [CrossRef]
- Pashangeh, S.; Berizi, E.; Majlesi, M.; Ghaderi, S.; Nizet, V.; Dahesh, S. Effect of eliminating hdcA gene of Staphylococcus epidermidis TYH1 on Histamine production. Iran. J. Microbiol. 2022, 14, 529–534. [Google Scholar] [CrossRef]
- Rodríguez-Lucas, C.; Ladero, V. Enterococcal Phages: Food and Health Applications. Antibiotics 2023, 12, 842. [Google Scholar] [CrossRef]
- Del Rio, B.; Ladero, V.; Redruello, B.; Linares, D.M.; Fernandez, M.; Martin, M.C.; Alvarez, M.A. Lactose-mediated carbon catabolite repression of putrescine production in dairy Lactococcus lactis is strain dependent. Food Microbiol. 2015, 48, 163–170. [Google Scholar] [CrossRef]
- Luo, Y.; Huang, Y.; Xu, R.X.; Qian, B.; Zhou, J.W.; Xia, X.L. Primary and Secondary Succession Mediate the Accumulation of Biogenic Amines during Industrial Semidry Chinese Rice Wine Fermentation. Appl. Environ. Microbiol. 2020, 86, e01177-20. [Google Scholar] [CrossRef]
- Wojtowicz, J.S. Long-Term Health Outcomes of Regular, Moderate Red Wine Consumption. Cureus 2023, 15, e46786. [Google Scholar] [CrossRef] [PubMed]
- Perrone, B.; Giacosa, S.; Rolle, L.; Cocolin, L.; Rantsiou, K. Investigation of the dominance behavior of Saccharomyces cerevisiae strains during wine fermentation. Int. J. Food Microbiol. 2013, 165, 156–162. [Google Scholar] [CrossRef]
- Cappello, M.S.; Zapparoli, G.; Logrieco, A.; Bartowsky, E.J. Linking wine lactic acid bacteria diversity with wine aroma and flavour. Int. J. Food Microbiol. 2017, 243, 16–27. [Google Scholar] [CrossRef] [PubMed]
- Mateo, E.; Torija, M.J.; Mas, A.; Bartowsky, E.J. Acetic acid bacteria isolated from grapes of South Australian vineyards. Int. J. Food Microbiol. 2014, 178, 98–106. [Google Scholar] [CrossRef] [PubMed]
- Testa, B.; Lombardi, S.J.; Tremonte, P.; Succi, M.; Tipaldi, L.; Pannella, G.; Sorrentino, E.; Iorizzo, M.; Coppola, R. Biodiversity of Lactobacillus plantarum from traditional Italian wines. World J. Microbiol. Biotechnol. 2014, 30, 2299–2305. [Google Scholar] [CrossRef]
- Zhang, S.W.; Chen, X.; Zhong, Q.D.; Zhuang, X.L.; Bai, Z.H. Microbial Community Analyses Associated with Nine Varieties of Wine Grape Carposphere Based on High-Throughput Sequencing. Microorganisms 2019, 7, 668. [Google Scholar] [CrossRef]
- Maicas, S. The use of alternative technologies to develop malolactic fermentation in wine. Appl. Microbiol. Biotechnol. 2001, 56, 35–39. [Google Scholar] [CrossRef]
- Sumby, K.M.; Bartle, L.; Grbin, P.R.; Jiranek, V. Measures to improve wine malolactic fermentation. Appl. Microbiol. Biotechnol. 2019, 103, 2033–2051. [Google Scholar] [CrossRef]
- Sun, J.; Ge, Y.; Gu, X.; Li, R.; Ma, W.; Jin, G. Identification and Characterization of Malolactic Bacteria Isolated from the Eastern Foothills of Helan Mountain in China. Foods 2022, 11, 2455. [Google Scholar] [CrossRef] [PubMed]
- Michlmayr, H.; Nauer, S.; Brandes, W.; Schümann, C.; Kulbe, K.D.; del Hierro, A.M.; Eder, R. Release of wine monoterpenes from natural precursors by glycosidases from Oenococcus oeni. Food Chem. 2012, 135, 80–87. [Google Scholar] [CrossRef]
- Gagné, S.; Lucas, P.M.; Perello, M.C.; Claisse, O.; Lonvaud-Funel, A.; de Revel, G. Variety and variability of glycosidase activities in an Oenococcus oeni strain collection tested with synthetic and natural substrates. J. Appl. Microbiol. 2011, 110, 218–228. [Google Scholar] [CrossRef] [PubMed]
- Ferner, M.J.; Müller, G.; Schumann, C.; Shaikh, Y.; Kmapeis, P.; Ulber, R.; Raddatz, H. Immobilisation of glycosidases from commercial preparation on magnetic beads. Part 2: Aroma enhancement in wine using immobilised glycosidases. Vitis 2018, 57, 129–136. [Google Scholar]
- Tavernini, L.; Aburto, C.; Romero, O.; Illanes, A.; Wilson, L. Encapsulation of Combi-CLEAs of Glycosidases in Alginate Beads and Polyvinyl Alcohol for Wine Aroma Enhancement. Catalysts 2021, 11, 866. [Google Scholar] [CrossRef]
- Ahumada, K.; Urrutia, P.; Illanes, A.; Wilson, L. Production of combi-CLEAs of glycosidases utilized for aroma enhancement in wine. Food Bioprod. Process. 2015, 94, 555–560. [Google Scholar] [CrossRef]
- González-Pombo, P.; Fariña, L.; Carrau, F.; Batista-Viera, F.; Brena, B.M. Aroma enhancement in wines using co-immobilized Aspergillus niger glycosidases. Food Chem. 2014, 143, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Bartowsky, E.J.; Henschke, P.A. Acetic acid bacteria spoilage of bottled red wine—A review. Int. J. Food Microbiol. 2008, 125, 60–70. [Google Scholar] [CrossRef]
- Bartowsky, E.J.; Xia, D.; Gibson, R.L.; Fleet, G.H.; Henschke, P.A. Spoilage of bottled red wine by acetic acid bacteria. Lett. Appl. Microbiol. 2003, 36, 307–314. [Google Scholar] [CrossRef]
- Gupta, A.; Singh, V.K.; Qazi, G.N.; Kumar, A. Gluconobacter oxydans: Its biotechnological applications. J. Mol. Microbiol. Biotechnol. 2001, 3, 445–456. [Google Scholar] [PubMed]
- Nurgel, C.; Pickering, G.J.; Inglis, D.L. Sensory and chemical characteristics of Canadian ice wines. J. Sci. Food Agric. 2004, 84, 1675–1684. [Google Scholar] [CrossRef]
- Izquierdo-Canas, P.M.; Lopez-Martin, R.; Garcia-Romero, E.; Gonzalez-Arenzana, L.; Minguez-Sanz, S.; Chatonnet, P.; Palacios-Garcia, A.; Puig-Pujol, A. Effect of kaolin silver complex on the control of populations of Brettanomyces and acetic acid bacteria in wine. J. Food Sci. Technol. 2018, 55, 1823–1831. [Google Scholar] [CrossRef] [PubMed]
- Bae, S.; Fleet, G.H.; Heard, G.M. Occurrence and significance of Bacillus thuringiensis on wine grapes. Int. J. Food Microbiol. 2004, 94, 301–312. [Google Scholar] [CrossRef] [PubMed]
- Hranilovic, A.; Gambetta, J.M.; Jeffery, D.W.; Grbin, P.R.; Jiranek, V. Lower-alcohol wines produced by Metschnikowia pulcherrima and Saccharomyces cerevisiae co-fermentations: The effect of sequential inoculation timing. Int. J. Food Microbiol. 2020, 329, 108651. [Google Scholar] [CrossRef] [PubMed]
- Romano, P.; Fiore, C.; Paraggio, M.; Caruso, M.; Capece, A. Function of yeast species and strains in wine flavour. Int. J. Food Microbiol. 2003, 86, 169–180. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, E.S.; Cardello, H.M.A.B.; Jeronimo, E.M.; Souza, E.L.R.; Serra, G.E. The influence of different yeasts on the fermentation, composition and sensory quality of cachaca. World J. Microbiol. Biotechnol. 2005, 21, 707–715. [Google Scholar] [CrossRef]
- Domizio, P.; Romani, C.; Lencioni, L.; Comitini, F.; Gobbi, M.; Mannazzu, I.; Ciani, M. Outlining a future for non-Saccharomyces yeasts: Selection of putative spoilage wine strains to be used in association with Saccharomyces cerevisiae for grape juice fermentation. Int. J. Food Microbiol. 2011, 147, 170–180. [Google Scholar] [CrossRef] [PubMed]
- Dutraive, O.; Benito, S.; Fritsch, S.; Beisert, B.; Patz, C.D.; Rauhut, D. Effect of Sequential Inoculation with Non-Saccharomyces and Saccharomyces Yeasts on Riesling Wine Chemical Composition. Fermentation 2019, 5, 79. [Google Scholar] [CrossRef]
- Ge, Q.; Guo, C.F.; Zhang, J.; Yan, Y.; Zhao, D.Q.; Li, C.H.; Sun, X.Y.; Ma, T.T.; Yue, T.L.; Yuan, Y.H. Effects of Simultaneous Co-Fermentation of Five Indigenous Non-Saccharomyces Strains with S. cerevisiae on Vidal Icewine Aroma Quality. Foods 2021, 10, 1452. [Google Scholar] [CrossRef]
- Fedrizzi, B.; Caramia, G.; Cipriani, M.; Finato, F.; Simonato, B.; Tosi, E.; Zapparoli, G. Changes in Wine Aroma Composition According to Botrytized Berry Percentage: A Preliminary Study on Amarone Wine. Food Technol. Biotechnol. 2011, 49, 529–535. [Google Scholar]
- Tosi, E.; Fedrizzi, B.; Azzolini, M.; Finato, F.; Simonato, B.; Zapparoli, G. Effects of noble rot on must composition and aroma profile of Amarone wine produced by the traditional grape withering protocol. Food Chem. 2012, 130, 370–375. [Google Scholar] [CrossRef]
- Bamforth, C.W. Progress in Brewing Science and Beer Production. Annu. Rev. Chem. Biomol. Eng. 2017, 8, 161–176. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Cai, G.L.; Li, X.M.; Lu, J. Development of a novel SRAP-SCAR marker for rapid identification of lager and ale types in brewer’s yeast. Mol. Biol. Rep. 2023, 50, 1943–1948. [Google Scholar] [CrossRef] [PubMed]
- Kato, T.; Takahashi, T. Studies on the Genetic Characteristics of the Brewing Yeasts Saccharomyces: A Review. J. Am. Soc. Brew. Chem. 2023, 81, 199–210. [Google Scholar] [CrossRef]
- Pereira, G.M.D.; Ramos, C.L.; Galvao, C.; Dias, E.S.; Schwan, R.F. Use of specific PCR primers to identify three important industrial species of Saccharomyces genus: Saccharomyces cerevisiae, Saccharomyces bayanus and Saccharomyces pastorianus. Lett. Appl. Microbiol. 2010, 51, 131–137. [Google Scholar]
- Monerawela, C.; Bond, U. Recombination sites on hybrid chromosomes in Saccharomyces pastorianus share common sequence motifs and define a complex evolutionary relationship between group I and II lager yeasts. Fems Yeast Res. 2017, 17, fox047. [Google Scholar]
- Suzuki, K.; Iijima, K.; Sakamoto, K.; Sami, M.; Yamashita, H. A Review of Hop Resistance in Beer Spoilage Lactic Acid Bacteria. J. Inst. Brew. 2006, 112, 173–191. [Google Scholar] [CrossRef]
- Hucker, B.; Christophersen, M.; Vriesekoop, F. The influence of thiamine and riboflavin on various spoilage microorganisms commonly found in beer. J. Inst. Brew. 2017, 123, 24–30. [Google Scholar] [CrossRef]
- Soto, J.B.; Fernandez-Franzon, M.; Ruiz, M.J.; Juan-Garcia, A. Presence of Ochratoxin A (OTA) Mycotoxin in Alcoholic Drinks from Southern European Countries: Wine and Beer. J. Agric. Food Chem. 2014, 62, 7643–7651. [Google Scholar] [CrossRef]
- Dysvik, A.; La Rosa, S.L.; Liland, K.H.; Myhrer, K.S.; Østlie, H.M.; De Rouck, G.; Rukke, E.O.; Westereng, B.; Wicklund, T. Co-fermentation Involving Saccharomyces cerevisiae and Lactobacillus Species Tolerant to Brewing-Related Stress Factors for Controlled and Rapid Production of Sour Beer. Front. Microbiol. 2020, 11, 279. [Google Scholar] [CrossRef]
- Chen, L.H.; Ren, L.X.; Li, D.N.; Ma, X. Analysis of microbiomes in three traditional starters and volatile components of the Chinese rice wines. Food Sci. Biotechnol. 2021, 30, 87–96. [Google Scholar] [CrossRef]
- Huang, Z.R.; Guo, W.L.; Zhou, W.B.; Li, L.; Xu, J.X.; Hong, J.L.; Liu, H.P.; Zeng, F.; Bai, W.D.; Liu, B.; et al. Microbial communities and volatile metabolites in different traditional fermentation starters used for Hong Qu glutinous rice wine. Food Res. Int. 2019, 121, 593–603. [Google Scholar] [CrossRef]
- Jiang, L.; Su, W.; Mu, Y.C.; Mu, Y. Major Metabolites and Microbial Community of Fermented Black Glutinous Rice Wine with Different Starters. Front. Microbiol. 2020, 11, 523789. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Zhang, Q.C.; Yang, H.; Sun, L.J.; Xia, H.C.; Sun, W.J.; Wang, Z.; Zhang, J.X. Bacterial Communities Related to Aroma Formation during Spontaneous Fermentation of ‘Cabernet Sauvignon’ Wine in Ningxia, China. Foods 2022, 11, 2775. [Google Scholar] [CrossRef]
- Pons, A.; Mouakka, N.; Deliere, L.; Crachereau, J.C.; Davidou, L.; Sauris, P.; Guilbault, P.; Darriet, P. Impact of Plasmopara viticola infection of Merlot and Cabernet Sauvignon grapes on wine composition and flavor. Food Chem. 2018, 239, 102–110. [Google Scholar] [CrossRef]
- Zhang, L.; Tao, Y.S.; Wen, Y.; Wang, H. Aroma Evaluation of Young Chinese Merlot Wines with Denomination of Origin. S. Afr. J. Enol. Vitic. 2013, 34, 46–53. [Google Scholar] [CrossRef]
- Ma, Y.W.; Peng, S.; Mi, L.; Li, M.; Jiang, Z.Z.; Wang, J. Correlation between fungi and volatile compounds during different fermentation modes at the industrial scale of Merlot wines. Food Res. Int. 2023, 174, 113638. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.W.; Wang, Y.; Chen, X.; Cui, B.J.; Bai, Z.H.; Zhuang, G.Q. Variety features differentiate microbiota in the grape leaves. Can. J. Microbiol. 2020, 66, 653–663. [Google Scholar] [CrossRef]
- Leveau, J.; Tech, J.J. Grapevine microbiomics: Bacterial diversity on grape leaves and berries revealed by high-throughput sequence analysis of 16S rRNA amplicons. Acta Hortic. 2011, 905, 31–42. [Google Scholar] [CrossRef]
- Coetzee, C.; du Toit, W.J. A comprehensive review on Sauvignon blanc aroma with a focus on certain positive volatile thiols. Food Res. Int. 2012, 45, 287–298. [Google Scholar] [CrossRef]
- Lund, C.M.; Thompson, M.K.; Benkwitz, F.; Wohler, M.W.; Triggs, C.M.; Gardner, R.; Heymann, H.; Ncolau, L. New Zealand Sauvignon blanc Distinct Flavor Characteristics: Sensory, Chemical, and Consumer Aspects. Am. J. Enol. Vitic. 2009, 60, 1–12. [Google Scholar] [CrossRef]
- Schmidt, C.V.; Olsen, K.; Mouritsen, O.G. Umami potential of fermented beverages: Sake, wine, champagne, and beer. Food Chem. 2021, 360, 128971. [Google Scholar] [CrossRef]
- Slaghenaufi, D.; Luzzini, G.; Borgato, M.; Boscaini, A.; Dal Cin, A.; Zandonà, V.; Ugliano, M. Characterization of the Aroma Profile of Commercial Prosecco Sparkling Wines. Appl. Sci. 2023, 13, 3609. [Google Scholar] [CrossRef]
- Kallitsounakis, G.; Catarino, S. An overview on botrytized wines. Ciência Técnica Vitivinícola 2020, 35, 76–106. [Google Scholar] [CrossRef]
- Li, H.; James, A.; Shen, X.M.; Wang, Y.S. Roles of microbiota in the formation of botrytized grapes and wines. Cyta J. Food 2021, 19, 656–667. [Google Scholar] [CrossRef]
- Chen, Y.; Zhang, W.; Yi, H.; Wang, B.; Xiao, J.; Zhou, X.Y.; Xu, J.K.; Jiang, L.; Shi, X.W. Microbial community composition and its role in volatile compound formation during the spontaneous fermentation of ice wine made from Vidal grapes. Process Biochem. 2020, 92, 365–377. [Google Scholar] [CrossRef]
- Durán-Guerrero, E.; Castro-Mejías, R.; García-Moreno, M.D.; Rodríguez-Dodero, M.D.; Schwarz, M.; Guillén-Sánchez, D. Aroma of Sherry Products: A Review. Foods 2021, 10, 753. [Google Scholar] [CrossRef]
- Prata-Sena, M.; Castro-Carvalho, B.M.; Nunes, S.; Amaral, B.; Silva, P. The terroir of Port wine: Two hundred and sixty years of history. Food Chem. 2018, 257, 388–398. [Google Scholar] [CrossRef]
- De Roos, J.; De Vuyst, L. Microbial acidification, alcoholization, and aroma production during spontaneous lambic beer production. J. Sci. Food Agric. 2019, 99, 25–38. [Google Scholar] [CrossRef]
- Bonatto, D. The diversity of commercially available ale and lager yeast strains and the impact of brewer’s preferential yeast choice on the fermentative beer profiles. Food Res. Int. 2021, 141, 110125. [Google Scholar] [CrossRef] [PubMed]
- De Roos, J.; De Vuyst, L. Acetic acid bacteria in fermented foods and beverages. Curr. Opin. Biotechnol. 2018, 49, 115–119. [Google Scholar] [CrossRef] [PubMed]
- Vaughan, A.; O’Sullivan, T.; van Sinderen, D. Enhancing the microbiological stability of malt and beer—A review. J. Inst. Brew. 2005, 111, 355–371. [Google Scholar] [CrossRef]
- Vion, C.; Peltier, E.; Bernard, M.; Muro, M.; Marullo, P. Marker Assisted Selection of Malic-Consuming Saccharomyces cerevisiae Strains for Winemaking. Efficiency and Limits of a QTL’s Driven Breeding Program. J. Fungi 2021, 7, 304. [Google Scholar] [CrossRef] [PubMed]
- Bellon, J.R.; Ford, C.M.; Borneman, A.R.; Chambers, P.J. A Novel Approach to Isolating Improved Industrial Interspecific Wine Yeasts Using Chromosomal Mutations as Potential Markers for Increased Fitness. Front. Microbiol. 2018, 9, 1442. [Google Scholar] [CrossRef] [PubMed]
- Jung, J.Y.; Kang, M.J.; Hwang, H.S.; Baek, K.R.; Seo, S.O. Reduction of Ethyl Carbamate in an Alcoholic Beverage by CRISPR/Cas9-Based Genome Editing of the Wild Yeast. Foods 2022, 12, 102. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Coello, M.S.; Briones Pérez, A.I.; Ubeda Iranzo, J.F.; Martin Alvarez, P.J. Characteristics of wines fermented with different Saccharomyces cerevisiae strains isolated from the La Mancha region. Food Microbiol. 1999, 16, 563–573. [Google Scholar] [CrossRef]
- Zheng, H.; Meng, K.; Liu, J.; Lin, Z.; Peng, Q.; Xie, G.; Wu, P.; Elsheery, N.I. Identification and expression of bifunctional acid urea-degrading enzyme/urethanase from Enterobacter sp. R-SYB082 and its application in degradation of ethyl carbamate in Chinese rice wine (Huangjiu). J. Sci. Food Agric. 2022, 102, 4599–4608. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, T.C.; Lalor, E.; Hanniffy, O.; Savage, A.V.; Tuohy, M.G. Comparison of wild-type and UV-mutant beta-glucanase-producing strains of Talaromyces emersonii with potential in brewing applications. J. Ind. Microbiol. Biotechnol. 2005, 32, 125–134. [Google Scholar] [CrossRef]
- Chandel, A.K.; Kapoor, R.K.; Singh, A.; Kuhad, R.C. Detoxification of sugarcane bagasse hydrolysate improves ethanol production by Candida shehatae NCIM 3501. Bioresour. Technol. 2007, 98, 1947–1950. [Google Scholar] [CrossRef]
- Laughery, M.F.; Plummer, D.A.; Wilson, H.E.; Vandenberg, B.N.; Mitchell, D.; Mieczkowski, P.A.; Roberts, S.A.; Wyrick, J.J. Genome-wide maps of UVA and UVB mutagenesis in yeast reveal distinct causative lesions and mutational strand asymmetries. Genetics 2023, 224, iyad086. [Google Scholar] [CrossRef] [PubMed]
- Yi, S.; Zhang, X.; Li, H.X.; Du, X.X.; Liang, S.W.; Zhao, X.H. Screening and Mutation of Saccharomyces cerevisiae UV-20 with a High Yield of Second Generation Bioethanol and High Tolerance of Temperature, Glucose and Ethanol. Ind. J. Microbiol. 2018, 58, 440–447. [Google Scholar] [CrossRef] [PubMed]
- Yang, T.; Zhang, S.; Li, L.; Tian, J.; Li, X.; Pan, Y. Screening and transcriptomic analysis of the ethanol-tolerant mutant Saccharomyces cerevisiae YN81 for high-gravity brewing. Front. Microbiol. 2022, 13, 976321. [Google Scholar] [CrossRef] [PubMed]
- Munekazu, K. Fermentation characteristics of hybrids between the cryophilic wine yeast Saccharomyces bayanus and the mesophilic wine yeast Saccharomyces cerevisiae. J. Ferment. Bioeng. 1994, 77, 432–435. [Google Scholar]
- Shinohara, T.; Saito, K.; Yanagida, F.; Goto, S. Selection and hybridization of wine yeasts for improved winemaking properties: Fermentation rate and aroma productivity. J. Ferment. Bioeng. 1994, 77, 428–431. [Google Scholar] [CrossRef]
- Skała, J.; Kotylak, Z. Protoplast fusion in Saccharomyces cerevisiae. Acta Microbiol. Pol. 1984, 33, 25–35. [Google Scholar] [PubMed]
- Morgan, A.J. Yeast strain improvement by protoplast fusion and transformation. Experientia. Suppl. 1983, 46, 155–166. [Google Scholar] [PubMed]
- Kavanagh, K.; Walsh, M.; Whittaker, P.A. Enhanced intraspecific protoplast fusion in yeast. FEMS Microbiol. Lett. 1991, 65, 283–286. [Google Scholar] [CrossRef] [PubMed]
- Xin, Y.; Yang, M.; Yin, H.; Yang, J. Improvement of Ethanol Tolerance by Inactive Protoplast Fusion in Saccharomyces cerevisiae. BioMed Res. Int. 2020, 2020, 1979318. [Google Scholar] [CrossRef]
- Wang, Z.; Xu, K.; Cai, R.; Yue, T.; Yuan, Y.; Gao, Z. Construction of recombinant fusant yeasts for the production of cider with low alcohol and enhanced aroma. Eur. Food Res. Technol. 2020, 246, 745–757. [Google Scholar] [CrossRef]
- Yang, Y.; Xia, Y.; Lin, X.; Wang, G.; Zhang, H.; Xiong, Z.; Yu, H.; Yu, J.; Ai, L. Improvement of flavor profiles in Chinese rice wine by creating fermenting yeast with superior ethanol tolerance and fermentation activity. Food Res. Int. 2018, 108, 83–92. [Google Scholar] [CrossRef] [PubMed]
- Kuzma, J. Policy: Reboot the debate on genetic engineering. Nature 2016, 531, 165–167. [Google Scholar] [CrossRef] [PubMed]
- Liang, Z.; Zhi, H.; Fang, Z.; Zhang, P. Genetic engineering of yeast, filamentous fungi and bacteria for terpene production and applications in food industry. Food Res. Int. 2021, 147, 110487. [Google Scholar] [CrossRef] [PubMed]
- Matthews, A.; Grbin, P.R.; Jiranek, V. Biochemical characterisation of the esterase activities of wine lactic acid bacteria. Appl. Microbiol. Biotechnol. 2007, 77, 329–337. [Google Scholar] [CrossRef] [PubMed]
- Lilly, M.; Lambrechts, M.G.; Pretorius, I.S. Effect of increased yeast alcohol acetyltransferase activity on flavor profiles of wine and distillates. Appl. Environ. Microbiol. 2000, 66, 744–753. [Google Scholar] [CrossRef] [PubMed]
- Mason, A.B.; Dufour, J.P. Alcohol acetyltransferases and the significance of ester synthesis in yeast. Yeast 2000, 16, 1287–1298. [Google Scholar] [CrossRef] [PubMed]
- Fujii, T.; Nagasawa, N.; Iwamatsu, A.; Bogaki, T.; Tamai, Y.; Hamachi, M. Molecular cloning, sequence analysis, and expression of the yeast alcohol acetyltransferase gene. Appl. Environ. Microbiol. 1994, 60, 2786–2792. [Google Scholar] [CrossRef]
- Lilly, M.; Bauer, F.F.; Lambrechts, M.G.; Swiegers, J.H.; Cozzolino, D.; Pretorius, I.S. The effect of increased yeast alcohol acetyltransferase and esterase activity on the flavour profiles of wine and distillates. Yeast 2006, 23, 641–659. [Google Scholar] [CrossRef] [PubMed]
- Kang, C.; Yu, X.W.; Xu, Y. Cloning and expression of a novel prolyl endopeptidase from Aspergillus oryzae and its application in beer stabilization. J. Ind. Microbiol. Biotechnol. 2015, 42, 263–272. [Google Scholar] [CrossRef]
- Stemmer, W.P. DNA shuffling by random fragmentation and reassembly: In vitro recombination for molecular evolution. Proc. Natl. Acad. Sci. USA 1994, 91, 10747–10751. [Google Scholar] [CrossRef]
- Zhang, Y.X.; Perry, K.; Vinci, V.A.; Powell, K.; Stemmer, W.P.; del Cardayré, S.B. Genome shuffling leads to rapid phenotypic improvement in bacteria. Nature 2002, 415, 644–646. [Google Scholar] [CrossRef] [PubMed]
- Gong, J.; Zheng, H.; Wu, Z.; Chen, T.; Zhao, X. Genome shuffling: Progress and applications for phenotype improvement. Biotechnol. Adv. 2009, 27, 996–1005. [Google Scholar] [CrossRef] [PubMed]
- Shi, D.J.; Wang, C.L.; Wang, K.M. Genome shuffling to improve thermotolerance, ethanol tolerance and ethanol productivity of Saccharomyces cerevisiae. J. Ind. Microbiol. Biotechnol. 2009, 36, 139–147. [Google Scholar] [CrossRef] [PubMed]
- Snoek, T.; Picca Nicolino, M.; Van den Bremt, S.; Mertens, S.; Saels, V.; Verplaetse, A.; Steensels, J.; Verstrepen, K.J. Large-scale robot-assisted genome shuffling yields industrial Saccharomyces cerevisiae yeasts with increased ethanol tolerance. Biotechnol. Biofuels 2015, 8, 32. [Google Scholar] [CrossRef] [PubMed]
- Jetti, K.D.; Gns, R.R.; Garlapati, D.; Nammi, S.K. Improved ethanol productivity and ethanol tolerance through genome shuffling of Saccharomyces cerevisiae and Pichia stipitis. Int. Microbiol. Off. J. Span. Soc. Microbiol. 2019, 22, 247–254. [Google Scholar] [CrossRef] [PubMed]
- Gu, C.; Wang, G.; Mai, S.; Wu, P.; Wu, J.; Wang, G.; Liu, H.; Zhang, J. ARTP mutation and genome shuffling of ABE fermentation symbiotic system for improvement of butanol production. Appl. Microbiol. Biotechnol. 2017, 101, 2189–2199. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Chen, X.; Dong, C.; Zhao, F.; Tang, L.; Mao, Z. Combining genome shuffling and interspecific hybridization among Streptomyces improved ε-poly-L-lysine production. Appl. Biochem. Biotechnol. 2013, 169, 338–350. [Google Scholar] [CrossRef] [PubMed]
- John, R.P.; Gangadharan, D.; Madhavan Nampoothiri, K. Genome shuffling of Lactobacillus delbrueckii mutant and Bacillus amyloliquefaciens through protoplasmic fusion for L-lactic acid production from starchy wastes. Bioresour. Technol. 2008, 99, 8008–8015. [Google Scholar] [CrossRef] [PubMed]
- Thornton, R.J. The introduction of flocculation into a homothallic wine yeast. A practical example of the modification of winemaking properties by the use of genetic techniques. Am. J. Enol. Vitic. 1985, 36, 47–49. [Google Scholar] [CrossRef]
- Fogel, S.; Welch, J.W.; Cathala, G.; Karin, M. Gene amplification in yeast: CUP1 copy number regulates copper resistance. Curr. Genet. 1983, 7, 347–355. [Google Scholar] [CrossRef]
- Kutyna, D.R.; Varela, C.; Stanley, G.A.; Borneman, A.R.; Henschke, P.A.; Chambers, P.J. Adaptive evolution of Saccharomyces cerevisiae to generate strains with enhanced glycerol production. Appl. Microbiol. Biotechnol. 2012, 93, 1175–1184. [Google Scholar] [CrossRef]
- Moneke, A.; Okolo, B.; Nweke, A.; Ezeogu, L.; Ire, F. Selection and characterisation of high ethanol tolerant Saccharomyces yeasts from orchard soil. Afr. J. Biotechnol. 2010, 7. [Google Scholar]
- Jones, R.M.; Russell, I.; Stewart, G.G. The use of catabolite derepression as a means of improving the fermentation rate of brewing yeast strains. J. Am. Soc. Brew. Chem. 1986, 44, 161–166. [Google Scholar] [CrossRef]
- Fukuda, H.; Kizaki, Y.; Tsukihashi, T.; Wakabayashi, S. Shochu brewing characteristics and properties of a trichothecin-resistant shochu yeast mutant. Biotechnol. Lett. 2001, 23, 2009–2013. [Google Scholar] [CrossRef]
- Mukai, N.; Nishimori, C.; Fujishige, I.W.; Mizuno, A.; Takahashi, T.; Sato, K. Beer brewing using a fusant between a sake yeast and a brewer’s yeast. J. Biosci. Bioeng. 2001, 91, 482–486. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, C.; Qi, Y.; Dai, L.; Ma, H.; Guo, X.; Xiao, D. Acetate ester production by Chinese yellow rice wine yeast overexpressing the alcohol acetyltransferase-encoding gene ATF2. Genet. Mol. Res. 2014, 13, 9735–9746. [Google Scholar] [CrossRef] [PubMed]
- Guangfa, X.; Zhiqiang, Z.; Jin, M.A.; Lan, W.; Jianwei, F.U. Screening of yeast strains forquick fermentation in Chinese rice wine brewing. China Brew. 2010. Available online: https://api.semanticscholar.org/CorpusID:88292344 (accessed on 26 April 2024).
- Yang, L.; Jiang, Y.; Li, Y. Screening, identification and fermentation characteristics of a Chinese rice wine yeast strain with high stress tolerance. J. Chin. Inst. Food Sci. Technol. 2013, 13, 71–77. [Google Scholar]
- Hirooka, K.; Yamamoto, Y.; Tsutsui, N.; Tanaka, T. Improved production of isoamyl acetate by a sake yeast mutant resistant to an isoprenoid analog and its dependence on alcohol acetyltransferase activity, but not on isoamyl alcohol production. J. Biosci. Bioeng. 2005, 99, 125–129. [Google Scholar] [CrossRef]
- Inoue, T.; Iefuji, H.; Katsumata, H. Characterization and isolation of mutants producing increased amounts of isoamyl acetate derived from hygromycin B-resistant sake yeast. Biosci. Biotechnol. Biochem. 2012, 76, 60–66. [Google Scholar] [CrossRef]
- Takahashi, T.; Ohara, Y.; Sueno, K. Breeding of a sake yeast mutant with enhanced ethyl caproate productivity in sake brewing using rice milled at a high polishing ratio. J. Biosci. Bioeng. 2017, 123, 707–713. [Google Scholar] [CrossRef]
- Pires, E.J.; Teixeira, J.A.; Brányik, T.; Vicente, A.A. Yeast: The soul of beer’s aroma—A review of flavour-active esters and higher alcohols produced by the brewing yeast. Appl. Microbiol. Biotechnol. 2014, 98, 1937–1949. [Google Scholar] [CrossRef]
- Krogerus, K.; Magalhães, F.; Vidgren, V.; Gibson, B. New lager yeast strains generated by interspecific hybridization. J. Ind. Microbiol. Biotechnol. 2015, 42, 769–778. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Zhao, G.A.; Wang, L.P. Controlled formation of volatile components in cider making using a combination of Saccharomyces cerevisiae and Hanseniaspora valbyensis yeast species. J. Ind. Microbiol. Biotechnol. 2006, 33, 192–196. [Google Scholar] [CrossRef] [PubMed]
- Valles, B.S.; Bedriñana, R.P.; Tascón, N.F.; Simón, A.Q.; Madrera, R.R. Yeast species associated with the spontaneous fermentation of cider. Food Microbiol. 2007, 24, 25–31. [Google Scholar] [CrossRef] [PubMed]
- Bellon, J.R.; Eglinton, J.M.; Siebert, T.E.; Pollnitz, A.P.; Rose, L.; de Barros Lopes, M.; Chambers, P.J. Newly generated interspecific wine yeast hybrids introduce flavour and aroma diversity to wines. Appl. Microbiol. Biotechnol. 2011, 91, 603–612. [Google Scholar] [CrossRef]
- Bellon, J.R.; Yang, F.; Day, M.P.; Inglis, D.L.; Chambers, P.J. Designing and creating Saccharomyces interspecific hybrids for improved, industry relevant, phenotypes. Appl. Microbiol. Biotechnol. 2015, 99, 8597–8609. [Google Scholar] [CrossRef] [PubMed]
- Peng, Q.; Meng, K.; Zheng, H.; Yu, H.; Zhang, Y.; Yang, X.; Lin, Z.; Xie, G. Metabolites comparison in post-fermentation stage of manual (mechanized) Chinese Huangjiu (yellow rice wine) based on GC-MS metabolomics. Food Chem. X 2022, 14, 100324. [Google Scholar] [CrossRef]
- Yang, Y.; Xia, Y.; Wang, G.; Tao, L.; Yu, J.; Ai, L. Effects of boiling, ultra-high temperature and high hydrostatic pressure on free amino acids, flavor characteristics and sensory profiles in Chinese rice wine. Food Chem. 2019, 275, 407–416. [Google Scholar] [CrossRef]
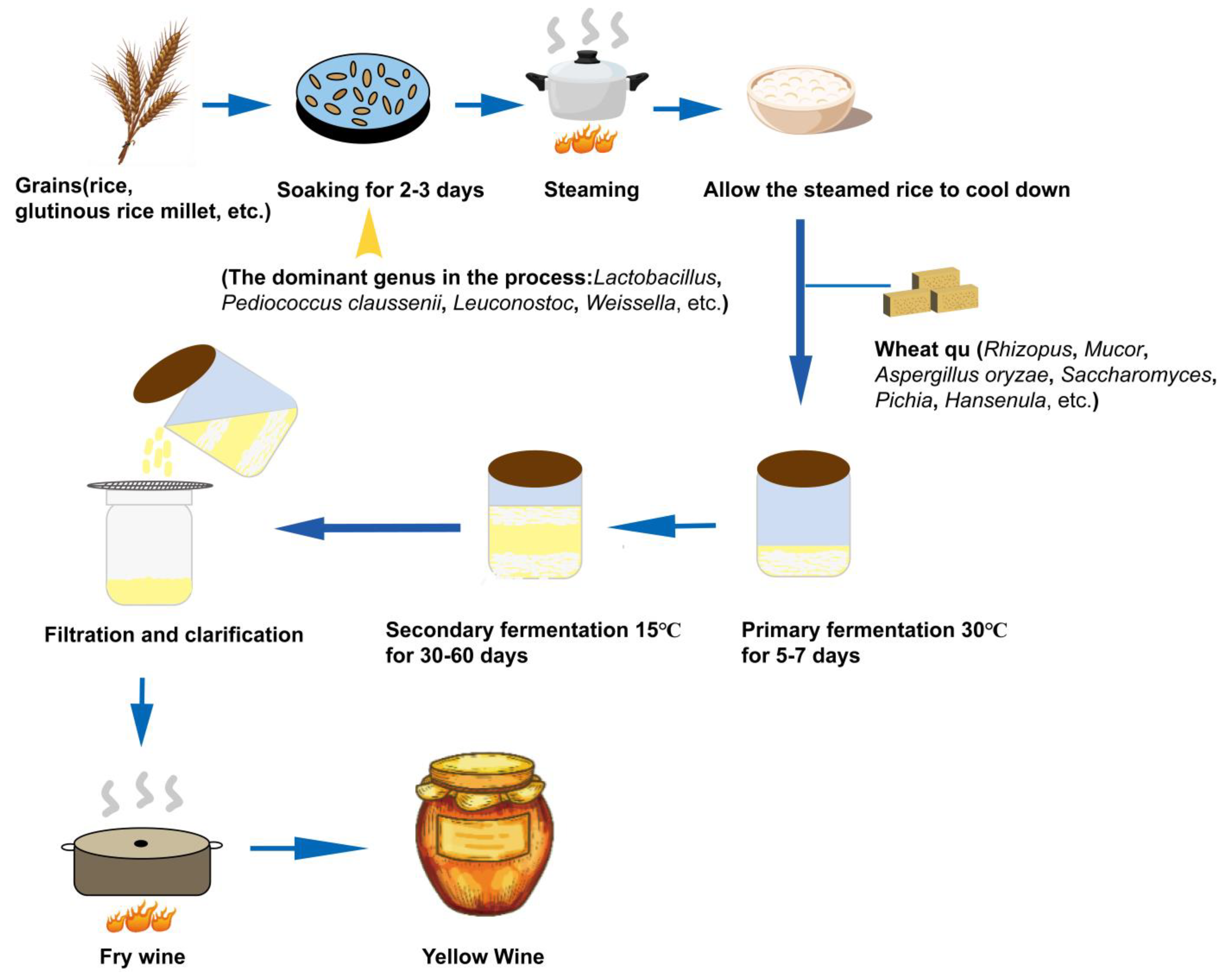
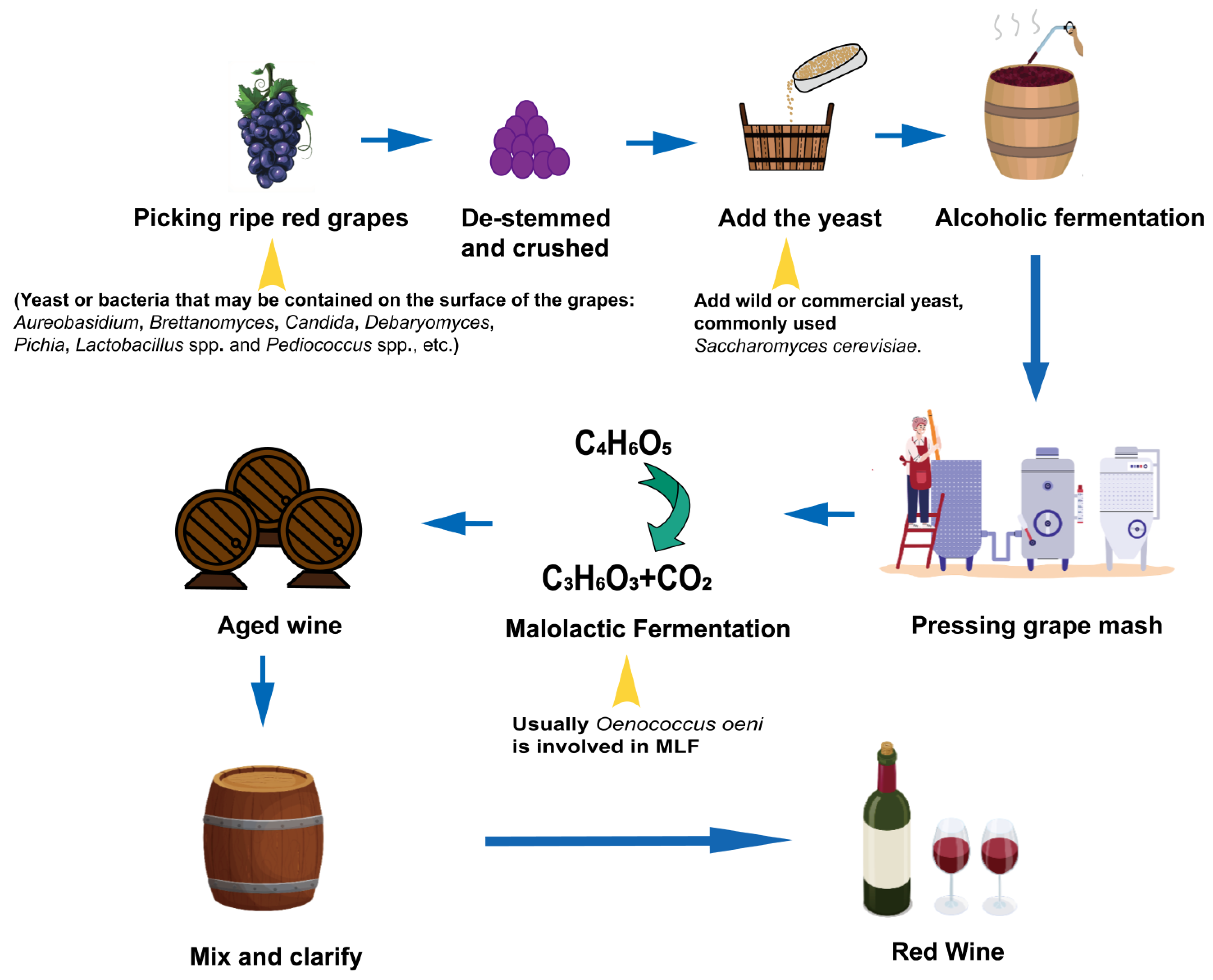
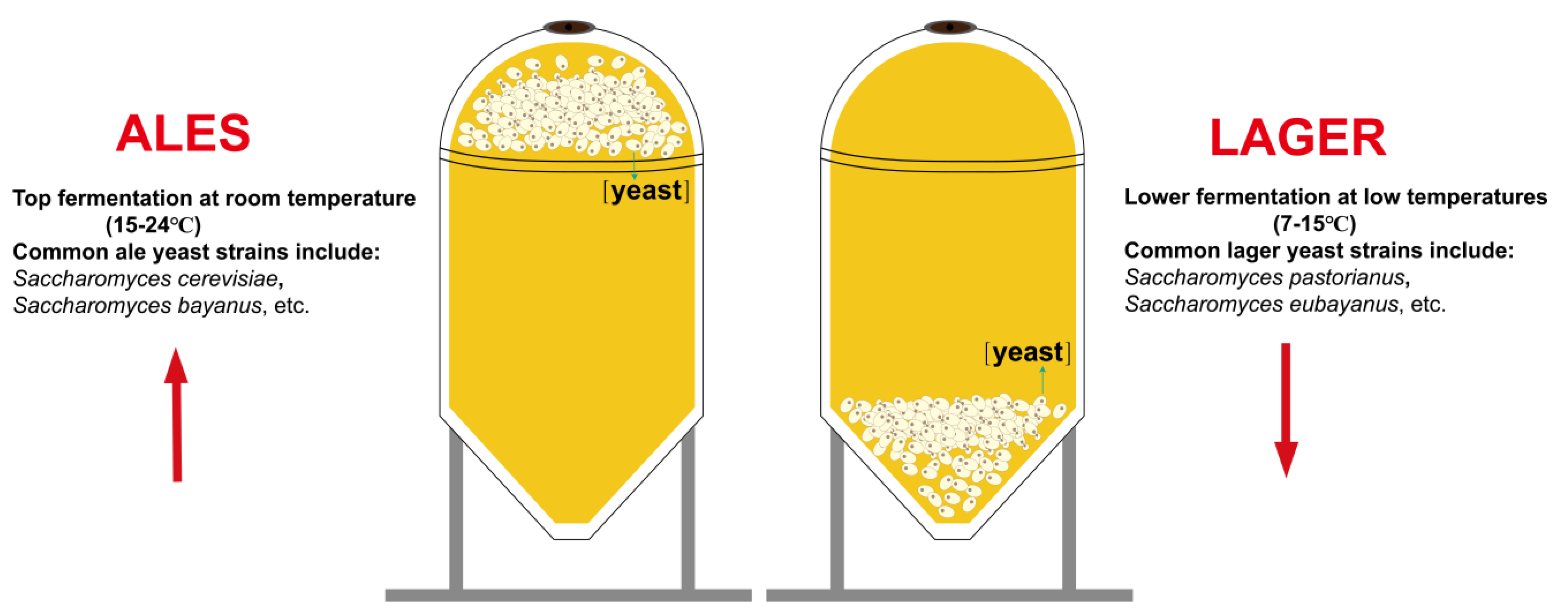
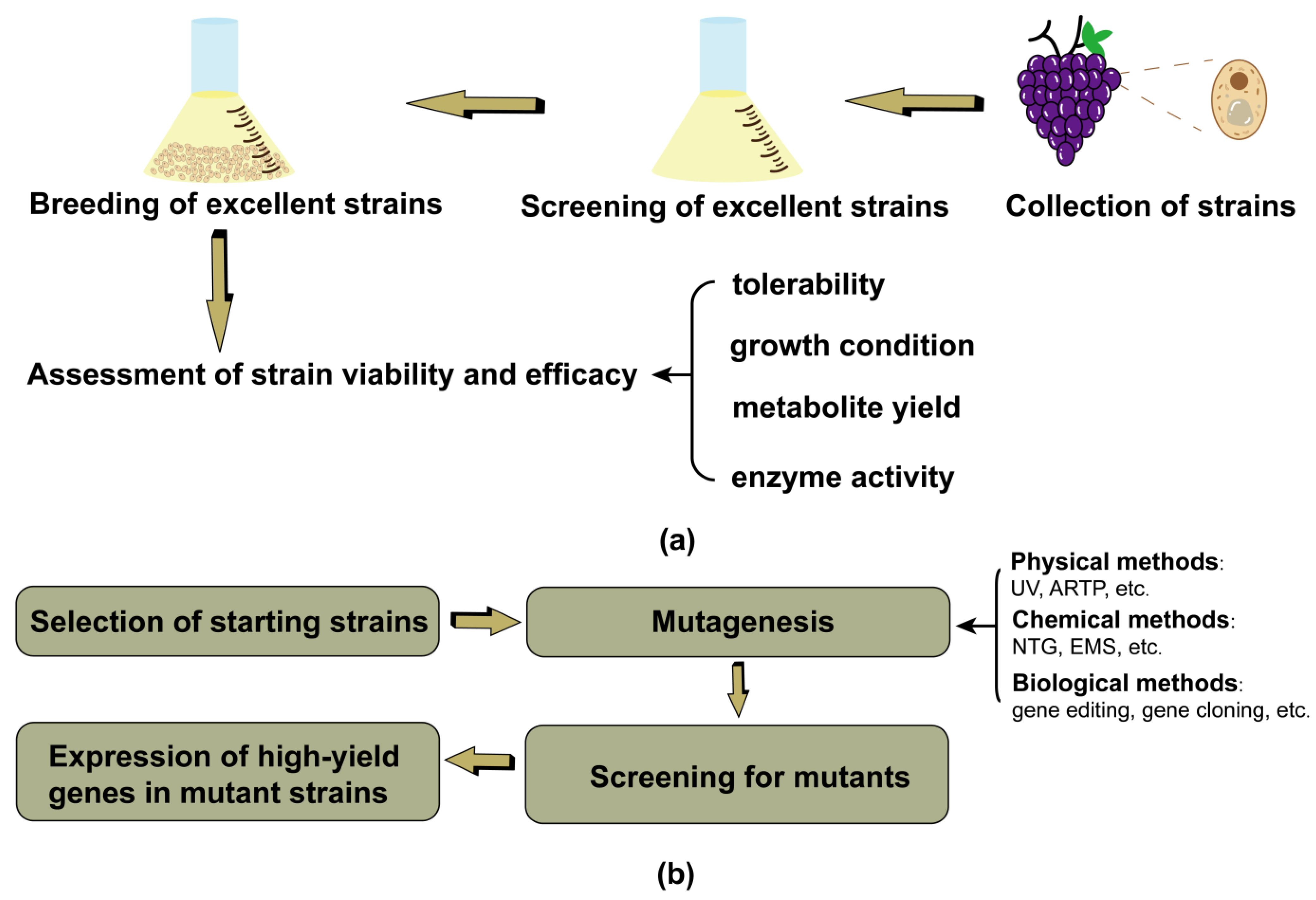

| Alcoholic Beverage | Types | Dominant Microorganisms | Sensorial Properties | References | |
|---|---|---|---|---|---|
| Yellow wine | Semi-dry Shaoxing Yellow Wine | Bacillus, Lactobacillus, Leuconostoc, Lactococcus, Thermoactinomyces | It has a pronounced rice aroma and light floral and fruity notes, as well as a distinctive earthiness from the fermentation process. | [36,42] | |
| CMQ (from Chongming, Shanghai) | Pantoea, Bacillus, Rhizopus, Candida | Soft, full-bodied, malty, and fruity on the palate. | [92] | ||
| NBQ (from Ningbo, Zhejiang) | Pediococcus, Lactobacillus, Acetobacter, Weissella, Bacillus, Rhizopus, Candida, Aspergillus | Pale golden or orange-yellow in color, with a strong wheat and yeast aroma. | |||
| YCQ (from Yichang, Hubei) | Pediococcus, Lactobacillus, Leuconostoc, Weissella, Lactococcus, Ochrobactrum, Rhizopus, Mucor | Long aftertaste, often with a light sweetness in the aftertaste, refreshing taste. | |||
| Hong Qu glutinous yellow wine | Bacillus, ginsengihumi, Pantoea sp., Elizabethkingia sp., Streptococcus sp. | Reddish-brown, usually sweeter, with a distinctive hong qu aroma. | [93] | ||
| Black glutinous yellow wine | Pediococcus, Leuconostoc, Rhizopus, Saccharomycopsis | Black or purple-black in color, with a sweet and sour taste and a complex aroma. | [94] | ||
| Wine | Red wine | Cabernet Sauvignon | Pantoea, Lactobacillus, Rhodococcus, Fructobacillus, and Komagataeibacter | Red wines typically have flavors of dark fruits like blackberry, cherry, and plum. They may also exhibit notes of chocolate, tobacco, and leather. Aromas can include earthy tones, spices, and sometimes a smoky character. | [95,96] |
| Merlot | Starmerella, Kazachstania | [97,98] | |||
| Shiraz | Pseudomonas, Alternaria sp. | [99] | |||
| Pinot Noir | Bacillus | [99] | |||
| White wine | Chardonnay | Pseudomonas, Bacillus, Leuconostoc, Erwinia | White wines usually have lighter, fresher flavors such as apples, pears, citrus, and tropical fruits. Floral and mineral aromas are also common. | [99,100] | |
| Riesling | Pseudomonas | [99] | |||
| Sauvignon Blanc | / | [101,102] | |||
| Sparkling wine | Champagne | / | Sparkling wine flavors often include green apple, pear, citrus, and sometimes toasty or nutty notes in aged varieties. | [103] | |
| Prosecco | / | [104] | |||
| Dessert Wine | Noble Rot Wine | Botrytis cinerea | Dessert wine with flavors of dried fruit, honey, caramel, and nuts. | [105,106] | |
| Ice Wine | Hanseniaspora uvarum, Metschnikowia fructicola, Saccharomyces cerevisiae, Lactococcus lactis and Leuconostoc spp. | [107] | |||
| Fortified Wine | Sherry | / | Fortified wines have a high alcohol content and intense flavors, including nutty, sweet, or spicy. | [108] | |
| Port | / | [109] | |||
| Beer | Lager beer, Pilsner beer | lager yeast | With a smooth and refreshing flavor. | [110] | |
| Ales, stouts, and porters | ale yeast | Often rich and complex with fruity, malty flavors, etc. | [111] | ||
| Belgian Lambic Beer | Saccharomyces cerevisiae, Lactobacillus | With a distinctive sour and fruity flavor. | [110,112] | ||
| Methods | Roles | Characteristics | |
|---|---|---|---|
| Natural selection | In the natural environment, strains with better tolerance and adaptability are screened and bred through the process of natural selection, based on the genetic variation and adaptability of the species, without human intervention. | The presence of stochastic and temporal evolution with environmental dependence. | |
| Crossing techniques | By crossing two different strains or lines of bacteria and yeast, their affinities and genetic variations are utilized to produce progeny strains with superior traits. | Can increase the genetic diversity of strains, be more stringent in the operation of tests and the choice of instruments, and have a high efficiency of selection and breeding. | |
| Physical mutagenesis | UV mutagenesis | UV light causes base transitions, inversions, shifted mutations, or deletions, which can lead to mutagenesis of the strain. | Classic method, good results, simple equipment, and easy operation. |
| Laser irradiation mutagenesis | When lasers irradiate organisms, their energy is directly or indirectly absorbed by biomolecules, which can lead to molecular stimulation of photodissociation, catabolism, and free radical reactions in biomolecules, resulting in aberrations in DNA molecules or chromosomes. | High energy density, relatively concentrated, good monochromaticity, and directionality; genetic mutation upon mutagenesis. | |
| Microwave mutagenesis | Can stimulate rapid vibration of polar molecules (e.g., water, proteins, nucleotides, fats, and carbohydrates), disrupting the hydrogen bonding and base accumulation of DNA molecules and leading to changes in the structure of the DNA, resulting in genetic variation. | Simple equipment, low cost, easy method, safe operation, and good mutation effect. | |
| High Static Pressure Mutagenesis | High hydrostatic pressure is a special processing technology for materials using hydrostatic pressure of more than 100MPa. It can not only change the volume, morphology, and cellular composition of microbial cells, but also alter the nucleic acid structure of microorganisms and their biological functions and gene expression. | Simple method, simple equipment, and good mutagenic effect. | |
| Ultrasound mutagenesis | Under the action of sound waves, the tiny bubbles in the liquid will oscillate, expand, contract, and even collapse. Cavitation bubble adiabatic contraction leads to the collapse of the moment, presenting more than 5000 °C temperatures and thousands of atmospheres of pressure, accompanied by powerful shock waves or jet streams, enough to change the cell wall membrane structure and cause the exchange of substances inside and outside the cells, and even mutation. | Simple equipment, safe operation, simple operation methods, and a higher mutation rate for mutagenesis. | |
| Chemical mutagenesis | Techniques for selecting new mutagenic strains by inducing genetic variation in microorganisms using chemical mutagens | Operation of toxic and hazardous chemical substances for personal and environmental safety. | |
| Protoplast fusion | The technique of artificially fusing two protoplasts with different genetic characteristics to obtain stable recombinants with parental genetic characteristics, and strains with good fermentation performance can be used directly as parental strains for the fermentation performance required for protoplast fusion breeding. | Overcoming the deficiencies of distant integration, simplicity of operation, integrity of genetic information, and a high frequency of recombination. | |
| Genetic engineering techniques | Biogenetic traits targeted and directly modified at the DNA molecular level | Can lead to changes in the species’ orientation and relatively short selection cycles. | |
| Genome shuffling | Through recursive recombination at the genomic level, the targeted evolution of the entire organism is efficiently realized, breaking through the limitations of traditional microbial strain improvement methods [146,147,148]. | Phenotypic improvement of microbial strains can be accomplished by modifying the whole genome of multiple parents, without the need to know much about the genetic background of the modified strains. | |
| Field | Work | Results | References |
|---|---|---|---|
| Wine | Spore hybridization | Improvement of fermentation efficiency and SO2 tolerance | [149] |
| Wine | Insertion of the CUP1 gene at multiple loci to improve copper tolerance | Enhances antibacterial resistance | [150] |
| Wine | Adaptive evolution screening of novel wine yeast strains with improved characteristics | Enhances glycerol production; improves the taste of wine | [151] |
| Wine | Selection of yeast from orchard soil | High ethanol tolerance and improved fermentation performance | [152] |
| Beer | Mutagenesis screening of 2-DOG-resistant yeast | Improves the utilization and fermentation efficiency of polysaccharides | [153] |
| Shochu | Screening of trichothecene-resistant yeast | Improves fermentation efficiency | [154] |
| Sake | Fusion of sake yeast and beer yeast | Accelerated fermentation rate, high ester yield, and hypertonic resistance | [155] |
| Yellow wine | Removal of transcription regulators. | Low production of urea and ethyl carbamate | [49] |
| Yellow wine | Overexpression of ATF2 in industrial yellow wine yeast strain (RY1) by homologous recombination | Increases the acetate ester | [156] |
| Fruit wine | Protoplast fusion using Saccharomyces cerevisiae and Candida ethanolica as starting strains | Obtaining a new fusion yeast for aroma production and ethanol reduction | [130] |
| Rice wine | Saccharomyces cerevisiae hybrids created by directed evolution and protoplast fusion strategies | Screening of Saccharomyces cerevisiae with high fermentation efficiency and high stress resistance | [157,158] |
| Brewed Wine | Main Raw Material | Ways to Improve Flavor Quality | References |
|---|---|---|---|
| Sake | Rice | Breeding of sake yeast with high production of isoamyl acetate or ethyl hexanoate | [159,160,161] |
| Beer | Malt | Breeding of aroma-producing beer yeast | [162,163] |
| Fruit wine | Juice | Selection of yeast strains by protoplast fusion to regulate flavor substance production | [164,165] |
| Wine | Grapes | Selection of aroma-producing yeast by interspecific crossing | [11,166,167] |
| Yellow wine | Grain | Enrichment of microbial diversity in yellow wine brewing and improvement of brewing process | [15,168,169] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, X.; Song, C.; Zhao, J.; Xiong, Z.; Peng, L.; Zou, L.; Shen, C.; Li, Q. Application of Strain Selection Technology in Alcoholic Beverages: A Review. Foods 2024, 13, 1396. https://doi.org/10.3390/foods13091396
Chen X, Song C, Zhao J, Xiong Z, Peng L, Zou L, Shen C, Li Q. Application of Strain Selection Technology in Alcoholic Beverages: A Review. Foods. 2024; 13(9):1396. https://doi.org/10.3390/foods13091396
Chicago/Turabian StyleChen, Xiaodie, Chuan Song, Jian Zhao, Zhuang Xiong, Lianxin Peng, Liang Zou, Caihong Shen, and Qiang Li. 2024. "Application of Strain Selection Technology in Alcoholic Beverages: A Review" Foods 13, no. 9: 1396. https://doi.org/10.3390/foods13091396








