Continuous Flow UV-C Irradiation Effects on the Physicochemical Properties of Aloe vera Gel and Pitaya (Stenocereus spp.) Blend
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Materials and Reagents
2.2. Aloe vera–Pitaya Blend
2.3. UV-C Light Processing
2.4. Physicochemical and Optical Properties of the Aloe vera-Pitaya Blend
2.5. Analytical Methods
2.6. Aloin A Content
2.7. Betalain Quantification
2.8. Betacyanin Retention
2.9. Simple Sugar Content
2.10. Color Characteristics
2.11. NMR Analysis of Concentrated Betacyanins
2.12. Experimental Design and Statistical Analysis
3. Results and Discussion
3.1. Chemical Characterization and Optical Properties of Aloe vera–Pitaya (Stenocereus spp.) Blend
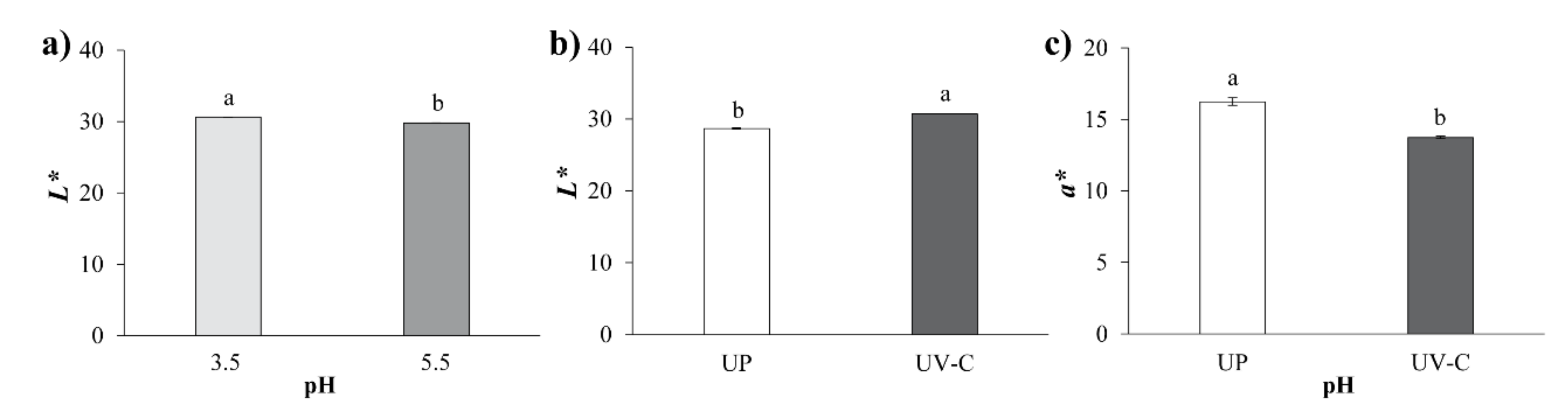
3.2. Color
3.3. Effect of UV-C Procesing on Betalains, Betacyanins, and Betaxanthines
3.4. NMR Analysis of UV-C Processed and Unprocessed Betalains Samples
3.5. Total Polyphenols
3.6. Total Antioxidant Activity
3.7. Total Sugars
3.8. Free Sugar Content
3.9. Aloin A
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Nazir, M.; Arif, S.; Rao Khan, R.S.; Nazir, W.; Khalid, N.; Maqsood, S. Opportunities and challenges for functional and medicinal beverages: Current and future trends. Trends Food Sci. Technol. 2019, 88, 513–526. [Google Scholar] [CrossRef]
- Domínguez-Fernández, R.N.; Arzate-Vazquez, I.; Chanona-Perez, J.J.; Welti-Chanes, J.S.; Alvarado-González, J.S.; Calderon-Dominguez, G.; Garybay-Febles, V.; Gutierrez-Lopez, G.F. El gel de Aloe vera: Estructura, composición química, procesamiento, actividad biológica e importancia en la industria farmacéutica y alimentaria. Rev. Mex. Ing. Quim. 2012, 11, 23–43. [Google Scholar]
- Rodríguez Rodriguez, E.; Darias Martín, J.; Diaz Romero, C. Aloe vera as a functional ingredient in foods. Crit. Rev. Food Sci. 2010, 50, 305–326. [Google Scholar] [CrossRef]
- García-Cruz, L.; Valle-Guadarrama, S.; Salinas-Moreno, Y.; Joaquín-Cruz, E. Physical, Chemical, and Antioxidant Activity Characterization of Pitaya (Stenocereus pruinosus) Fruits. Plant Foods Hum. Nutr. 2013, 68, 403–410. [Google Scholar] [CrossRef] [PubMed]
- Roobab, U.; Aadil, R.M.; Madni, G.M.; Bekhit, A.E.-D. The Impact of Nonthermal Technologies on the Microbiological Quality of Juices: A Review. Compr. Rev. Food Sci. Food Saf. 2018, 17, 437–457. [Google Scholar] [CrossRef]
- Femenia, A.; Garcia-Pascual, P.; Simal, S.; Rosello, C. Effects of heat treatment and dehydration on bioactive polysaccharide acemannan and cell wall polymers from Aloe barbadensis Miller. Carbohydr. Polym. 2003, 51, 397–405. [Google Scholar] [CrossRef]
- Rodriguez-Gonzalez, V.M.; Femenia, A.; González-Laredo, R.F.; Rocha-Guzman, N.E.; Gallegos-Infante, J.A.; Candelas-Cadillo, M.G.; Ramirez-Baca, P.; Simal, S.; Rosello, C. Effects of pasteurization on bioactive polysaccharide acemannan and cell wall polymers from Aloe barbadensis Miller. Carbohydr. Polym. 2011, 86, 1675–1683. [Google Scholar] [CrossRef]
- Rodríguez-Rodríguez, M.Z.; Meléndez-Pizarro, C.O.; Espinoza-Hicks, J.C.; Quintero-Ramos, A.; Sánchez-Madrigal, M.Á.; Meza-Velázquez, J.A.; Jiménez-Castro, J.A. Effects of UV-C irradiation and traditional thermal processing on acemannan contained in Aloe vera gel blends. Carbohydr. Polym. 2019, 222, 114998. [Google Scholar] [CrossRef]
- Güneşer, O. Pigment and color stability of beetroot betalains in cow milk during thermal treatment. Food Chem. 2016, 196, 220–227. [Google Scholar] [CrossRef]
- Ding, W.J.; Wu, X.F.; Zhong, J.S.; Wan, J.Z. Effects of temperature, pH and light on the stability of aloin A and characterization of its major degradation products. Int. J. Food Sci. Technol. 2014, 49, 1773–1779. [Google Scholar] [CrossRef]
- Rodríguez-Rodríguez, M.Z.; Quintero-Ramos, A.; Meléndez-Pizarro, C.O.; Meza-Velázquez, J.A.; Jimé>nez-Castro, J.A.; Sánchez-Madrigal, M.Á.; Espinoza-Hicks, J.C. Microbial and physicochemical properties of UV-C processed Aloe vera gel blends at different pHs using a continuous flow UV system Aloe vera gel blends at different pHs using a continuous flow UV system. Rev. Mex. Ing. Quim. 2020, 19, 175–188. [Google Scholar] [CrossRef] [Green Version]
- Worobo, R.W. Efficacy of the CiderSure 3500 ultraviolet light unit in apple cider. In Proceedings of the Apple Cider Food Safety Workshop, Ithaca, NY, USA, 15–16 July 1999; Cornell University, U.S. Food and Drug Administration. Center for Food Safety and Applied Nutrition, Department of Food Science and Technology: Washington, DC, USA.
- Lopez-Malo, A.; Palou, E. Ultraviolet light and food preservation. In Novel Food Processing Technologies; Barbosa-Canovas, G., Tapia, M.S., Cano, P.M., Eds.; CRC press: Boca Raton, FL, USA, 2005; Volume 4, pp. 1357–1372. [Google Scholar]
- Gayán, E.; Condón, S.; Álvarez, I. Continuous-flow UV liquid food pasteurization: Engineering aspects. Food Bioprocess Technol. 2014, 7, 2813–2827. [Google Scholar] [CrossRef]
- Koutchma, T. Advances in ultraviolet light technology for non-thermal processing of liquid foods. Food Bioprocess Technol. 2009, 2, 138–155. [Google Scholar] [CrossRef]
- Gopisetty, V.V.S.; Patras, A.; Kilonzo-Nthenge, A.; Yannam, S.; Bansode, R.R.; Sasges, M.; Xiao, H. Impact of UV-C irradiation on the quality, safety, and cytotoxicity of cranberry-flavored water using a novel continuous flow UV system. LWT Food Sci. Technol. 2018, 95, 230–239. [Google Scholar] [CrossRef]
- Reyes, J.E.; Guanoquiza, M.I.; Tabilo-Munizaga, G.; Vega-Galvez, A.; Miranda, M.; Pérez-Won, M. Microbiological stabilization of Aloe vera (Aloe barbadensis Miller) gel by high hydrostatic pressure treatment. Int. J. Food Microbiol. 2012, 158, 218–224. [Google Scholar] [CrossRef] [PubMed]
- Quintero-Ramos, A.; Churey, J.J.; Hartman, P.; Barnard, J.; Worobo, R.W. Modeling of Escherichia coli inactivation by UV irradiation at different pH values in apple cider. J. Food Prot. 2004, 67, 1153–1156. [Google Scholar] [CrossRef]
- Koutchma, T.; Keller, S.; Chirtel, S.; Parisi, B. Ultraviolet disinfection of juice products in laminar and turbulent flow reactors. Inn. Food Sci. Emerg. Technol. 2004, 5, 179–189. [Google Scholar] [CrossRef]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventós, R.M. Analysis of total phenols and other oxidation substrates and antioxidants by means of folin-ciocalteu reagent. Methods Enzym. 1999, 299, 152–178. [Google Scholar] [CrossRef]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a free radical method to evaluate antioxidant activity. LWT Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Dubois, M.; Gilles, K.A.; Hamilton, J.K.; Rebers, P.T.; Smith, F. Colorimetric method for determination of sugars and related substances. Anal. Chem. 1956, 28, 350–356. [Google Scholar] [CrossRef]
- Bozzi, A.; Perrin, C.; Austin, S.; Vera, F.A. Quality and authenticity of commercial aloe vera gel powders. Food Chem. 2007, 103, 22–30. [Google Scholar] [CrossRef]
- Castellanos-Santiago, E.; Yahia, E.M. Identification and quantification of betalains from the fruits of 10 Mexican prickly pear cultivars by high-performance liquid chromatography and electrospray ionization mass spectrometry. J. Agric. Food Chem. 2008, 56, 5758–5764. [Google Scholar] [CrossRef]
- Cai, Y.; Sun, M.; Wu, H.; Huang, R.; Corke, H. Characterization and quantification of betacyanin pigments from diverse Amaranthus species. J. Agric. Food Chem. 1998, 46, 2063–2070. [Google Scholar] [CrossRef]
- Stintzing, F.C.; Schieber, A.; Carle, R. Betacyanins in fruits from red-purple pitaya, Hylocereus polyrhizus (Weber) Britton & Rose. Food Chem. 2002, 77, 101–106. [Google Scholar] [CrossRef]
- Minitab. Statistical Software 16; Minitab Inc.: State College, PA, USA, 2010. [Google Scholar]
- Koutchma, T.; Parisi, B.; Patazca, E. Validation of UV coiled tube reactor for fresh juices. J. Environ. Eng. Sci. 2007, 6, 319–328. [Google Scholar] [CrossRef]
- Koutchma, T.; Popovi´c, V.; Ros-Polski, V.; Popielarz, A. Effects of ultraviolet light and high-pressure processing on quality and health-related constituents of fresh juice products. Compr. Rev. Food Sci. Food Saf. 2016, 15, 844–867. [Google Scholar] [CrossRef] [Green Version]
- Stintzing, F.C.; Conrad, J.; Klaiber, I.; Beifuss, U.; Carle, R. Structural investigations on betacyanin pigments by LC NMR and 2D NMR spectroscopy. Phytochemistry 2004, 65, 415–422. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.I. Stabilization of betalains: A review. Food Chem. 2016, 197, 1280–1285. [Google Scholar] [CrossRef] [PubMed]
- Woo, K.K.; Ngou, F.H.; Ngo, L.S.; Soong, W.K.; Tang, P.Y. Stability of betalain pigment from red dragon fruit (Hylocereus polyrhizus). Am. J. Food Technol. 2011, 6, 140–148. [Google Scholar] [CrossRef] [Green Version]
- Mello, F.R.; Bernardo, C.; Dias, C.O.; Gonzaga, L.; Amante, E.R.; Fett, R.; Candido, L.M.B. Antioxidant properties, quantification and stability of betalains from pitaya (Hylocereus undatus) peel. Ciência Rural. 2015, 45, 323–328. [Google Scholar] [CrossRef]
- Tang, C.S.; Norziah, M.H. Stability of betacyanin pigments from red purple pitaya fruit (Hylocereus polyrhizus): Influence of pH, temperature, metal ions and ascorbic acid. Indones. J. Chem. 2007, 7, 327–331. [Google Scholar] [CrossRef]
- Wong, Y.M.; Siow, L.F. Effects of heat, pH, antioxidant, agitation and light on betacyanin stability using red-fleshed dragon fruit (Hylocereus polyrhizus) juice and concentrate as models. J. Food Sci. Technol. 2015, 52, 3086–3092. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Herbach, K.M.; Stintzing, F.C.; Carle, R. Thermal degradation of betacyanins in juices from purple pitaya [Hylocereus polyrhizus (Weber) Britton & Rose] monitored by high-performance liquid chromatography–tandem mass spectrometric analyses. Eur. Food Res. Technol. 2004, 219, 377–385. [Google Scholar] [CrossRef]
- Cai, Y.; Sun, M.; Schliemann, W.; Corke, H. Chemical stability and colorant properties of betaxanthin pigments from Celosia Argentea. J. Agric. Food Chem. 2001, 49, 4429–4435. [Google Scholar] [CrossRef]
- Martins, N.; Roriz, C.L.; Morales, P.; Barros, L.; Ferreira, I.C. Coloring attributes of betalains: A key emphasis on stability and future applications. Food Funct. 2017, 8, 1357–1372. [Google Scholar] [CrossRef]
- Slimen, I.B.; Najar, T.; Abderrabba, M. Chemical and antioxidant properties of betalains. J. Agric. Food Chem. 2017, 65, 675–689. [Google Scholar] [CrossRef]
- Chethan, S.; Malleshi, N.G. Finger millet polyphenols: Optimization of extraction and the effect of pH on their stability. Food Chem. 2007, 105, 862–870. [Google Scholar] [CrossRef]
- Friedman, M.; Jürgens, H.S. Effect of pH on the stability of plant phenolic compounds. J. Agric. Food Chem. 2000, 48, 2101–2110. [Google Scholar] [CrossRef]
- Pala, Ç.U.; Toklucu, A.K. Effect of UV-C light on anthocyanin content and other quality parameters of pomegranate juice. J. Food Comp. Anal. 2011, 24, 790–795. [Google Scholar] [CrossRef]
- Pala, Ç.U.; Toklucu, A.K. Microbial, physicochemical and sensory properties of UV-C processed orange juice and its microbial stability during refrigerated storage. LWT Food Sci. Technol. 2013, 50, 426–431. [Google Scholar] [CrossRef]
- Caminiti, I.M.; Palgan, I.; Muñoz, A.; Noci, F.; Whyte, P.; Morgan, D.J.; Lyng, J.G. The effect of ultraviolet light on microbial inactivation and quality attributes of apple juice. Food Bioprocess Technol. 2012, 5, 680–686. [Google Scholar] [CrossRef]
- Caminiti, I.M.; Noci, F.; Morgan, D.J.; Cronin, D.A.; Lyng, J.G. The effect of pulsed electric fields, ultraviolet light or high intensity light pulses in combination with manothermosonication on selected physico-chemical and sensory attributes of an orange and carrot juice blend. Food Bioprod. Process. 2012, 90, 442–448. [Google Scholar] [CrossRef]
- Ferrario, M.; Schenk, M.; Carrillo, M.G.; Guerrero, S. Development and quality assessment of a turbid carrot-orange juice blend processed by UV-C light assisted by mild heat and addition of Yerba Mate (Ilex paraguariensis) extract. Food Chem. 2018, 269, 567–576. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ray, A.; Dutta Gupta, S.; Ghosh, S. Isolation and characterization of potent bioactive fraction with antioxidant and UV absorbing activity from Aloe barbadensis Miller gel. J. Plant Biochem. Biotechnol. 2013, 22, 483–487. [Google Scholar] [CrossRef]
- Li, X.; Li, M.; Ji, N.; Jin, P.; Zhang, J.; Zheng, Y.; Li, F. Cold plasma treatment induces phenolic accumulation and enhances antioxidant activity in fresh-cut pitaya (Hylocereus undatus) fruit. LWT Food Sci. Technol. 2019, 115, 108447. [Google Scholar] [CrossRef]
- Minjares-Fuentes, J.R.; Femenia, A. Effect of processing on the bioactive polysaccharides and phenolic compounds from Aloe vera (Aloe barbadensis Miller). In Dietary Fibre Functionality in Food and Nutraceuticals: From Plant to Gut; Hosseinnian, F., Oomah, D., Campos-Vega, R., Eds.; Wiley & Sons Ltd.: Oxford, UK, 2017; pp. 263–287. [Google Scholar]
- Islam, M.S.; Patras, A.; Pokharel, B.; Wu, Y.; Vergne, M.J.; Shade, L. UV-C irradiation as an alternative disinfection technique: Study of its effect on polyphenols and antioxidant activity of apple juice. Inn. Food Sci. Emerg. Technol. 2016, 34, 344–351. [Google Scholar] [CrossRef] [Green Version]
- Esquivel, P.; Stintzing, F.C.; Carle, R. Comparison of morphological and chemical fruit traits from different pitaya genotypes (Hylocereus sp.) grown in Costa Rica. J. Appl. Bot. Food Qual. 2007, 81, 7. [Google Scholar]
- Salinas, C.; Handford, M.; Pauly, M.; Dupree, P.; Cardemil, L. Structural modifications of fuctans in Aloe barbadensis Miller (Aloe vera) grown under water stress. PLoS ONE 2016, 11, e0159819. [Google Scholar] [CrossRef] [Green Version]
- Quezada, M.P.; Salinas, C.; Gotteland, M.; Cardemil, L. Acemannan and fructans from Aloe vera (Aloe barbadensis Miller) plants as novel prebiotics. J. Agric. Food Chem. 2017, 65, 10029–10039. [Google Scholar] [CrossRef]




| Source | DF | Sum of Square | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| TP | Aloin A | TAA | L * | a * | b * | Glucose | Fructose | Sucrose | TS | BT | BC% | BX% | ||
| Model | 7 | 536.767 | 10.702 | 44422.98 * | 16.318 * | 30.857 * | 7.698 * | 45.847 * | 30.744 * | 360.263 * | 6272.48 * | 1492.320 * | 2106.460 * | 3678.577 * |
| pH | 1 | 251.368 * | 1.272 | 304.105 * | 2.5149 * | 7.617 * | 3.875 * | 8.2661 * | 4.89459 * | 65.771 * | 876.38 * | 772.490 * | 385.926* | 1238.512 |
| UP vs UV-C | 1 | 61.111 | 0.492 | 33038.14 * | 12.434 * | 18.884 * | 2.816 * | 25.703 * | 13.2101 * | 234.548 * | 124.790 | 366.807 * | 1459.048 * | 1786.446 * |
| pH *UP vs UV-C | 1 | 166.288 * | 0.809 | 7675.694 * | 0.719 | 3.279 | 0.906 * | 0.8752 * | 0.5102 * | 23.614 * | 2870.04 * | 193.601 * | 128.67 | 412.837 * |
| D | 2 | 51.535 | 6.114 | 562.874 * | 0.338 | 0.840 | 0.014 | 6.6157 * | 9.2903 * | 30.297 * | 24.088 | 94.847 | 130.441 | 165.546 |
| pH*D | 2 | 6.464 | 2.016 | 2842.172 * | 0.312 | 0.236 | 0.088 | 4.3878 * | 2.8383 * | 6.033 * | 2377.17 * | 64.573 | 2.402 | 75.236 |
| Error | 8 | 220.604 | 9.784 | 392.659 | 1.632 | 8.305 | 0.373 | 0.0201 | 0.2542 | 0.809 | 482.102 | 307.833 | 343.083 | 227.379 |
| Treatment/pH | L * | a * | b * |
|---|---|---|---|
| UP/3.5 | 28.71 ± 0.54 b | 16.34 ± 0.62 a | 7.59 ± 0.15 b |
| UP/5.5 | 28.65 ± 0.18 b | 16.15 ± 0.14 ab | 7.43 ± 0.07 b |
| D1-UV/3.5 | 31.24 ± 0.32 a | 12.97 ± 1.44 ab | 8.97 ± 0.33 a |
| D1-UV/5.5 | 30.37 ± 0.08 ab | 14.56 ± 0.20 ab | 7.94 ± 0.01 b |
| D2-UV/3.5 | 31.61 ± 0.86 a | 12.26 ± 1.91b | 9.18 ± 0.38 a |
| D2-UV/5.5 | 30.13 ± 0.15 ab | 14.53 ± 0.12 ab | 7.74 ± 0.02 b |
| D3-UV/3.5 | 30.86 ± 0.61 a | 13.11 ± 1.45 ab | 9.19 ± 0.24 b |
| D3-UV/5.5 | 30.11 ± 0.25 ab | 14.97 ± 0.01 ab | 7.87 ± 0.18 b |
| Treatment/pH | Total Sugars (mg/g dm) | Glucose (mg/g dm) | Fructose (mg/g dm) | Sucrose (mg/g dm) | Aloin A (mg/L) |
|---|---|---|---|---|---|
| UP/3.5 | 839.11 ± 11.78 ab | 25.31 ± 0.04 c | 27.17 ± 0.10 c | 128.92 ± 0.32 e | 2.84 ± 0.85 a |
| UP/5.5 | 807.52 ± 1.93 cd | 23.07 ± 0.05 e | 25.44 ± 0.02 d | 120.66 ± 0.58 f | 2.95 ± 0.23 a |
| D1-UV/3.5 | 789.57 ± 5.17 d | 27.50 ± 0.03 b | 28.28 ± 0.21 b | 132.28 ± 0.19 cd | 3.99 ± 0.12 a |
| D1-UV/5.5 | 846.91 ± 15.03 a | 24.63 ± 0.03 d | 26.06 ± 0.18 d | 131.08 ± 0.15 d | 3.05 ± 0.31 a |
| D2-UV/3.5 | 796.43 ± 3.55 d | 27.68 ± 0.08 b | 28.84 ± 0.19 ab | 134.75 ± 0.30 b | 3.36 ± 0.12 a |
| D2-UV/5.5 | 838.44 ± 1.47 ab | 27.48 ± 0.08 b | 28.93 ± 0.19 ab | 132.56 ± 0.23 c | 2.95 ± 0.71 a |
| D3-UV/3.5 | 819.19 ± 2.34 abcd | 27.92 ± 0.03 a | 29.45 ± 0.15 a | 137.86 ± 0.28 a | 2.58 ± 0.46 a |
| D3-UV/5.5 | 810.64 ± 8.16 bcd | 27.48 ± 0.03 b | 28.87 ± 0.27 ab | 133.28 ± 0.28 c | 2.67 ± 0.44 a |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meléndez-Pizarro, C.O.; Calva-Quintana, A.; Espinoza-Hicks, J.C.; Sánchez-Madrigal, M.Á.; Quintero-Ramos, A. Continuous Flow UV-C Irradiation Effects on the Physicochemical Properties of Aloe vera Gel and Pitaya (Stenocereus spp.) Blend. Foods 2020, 9, 1068. https://doi.org/10.3390/foods9081068
Meléndez-Pizarro CO, Calva-Quintana A, Espinoza-Hicks JC, Sánchez-Madrigal MÁ, Quintero-Ramos A. Continuous Flow UV-C Irradiation Effects on the Physicochemical Properties of Aloe vera Gel and Pitaya (Stenocereus spp.) Blend. Foods. 2020; 9(8):1068. https://doi.org/10.3390/foods9081068
Chicago/Turabian StyleMeléndez-Pizarro, Carmen O., Arlet Calva-Quintana, José C. Espinoza-Hicks, Miguel Á. Sánchez-Madrigal, and Armando Quintero-Ramos. 2020. "Continuous Flow UV-C Irradiation Effects on the Physicochemical Properties of Aloe vera Gel and Pitaya (Stenocereus spp.) Blend" Foods 9, no. 8: 1068. https://doi.org/10.3390/foods9081068







