Improved Cartilage Protection with Low Molecular Weight Hyaluronic Acid Hydrogel
Abstract
:1. Introduction
2. Materials and Methods
2.1. Experimental Design
2.2. Hyaluronic Acid Methacrylation and Characterization
2.3. Explant Dissection and Processing
2.4. Time-Zero MeHA Integration and Cartilage Fortification
2.4.1. Serial Mechanical Testing
2.4.2. Diffusion and Integration with Cartilage Tissue
2.5. Living Explant Degenerative Culture
2.5.1. DMMB Assay
2.5.2. Mechanical Testing
2.5.3. Histology and Immunofluorescence
2.6. Statistical Analysis and Experimental Rigor
3. Results
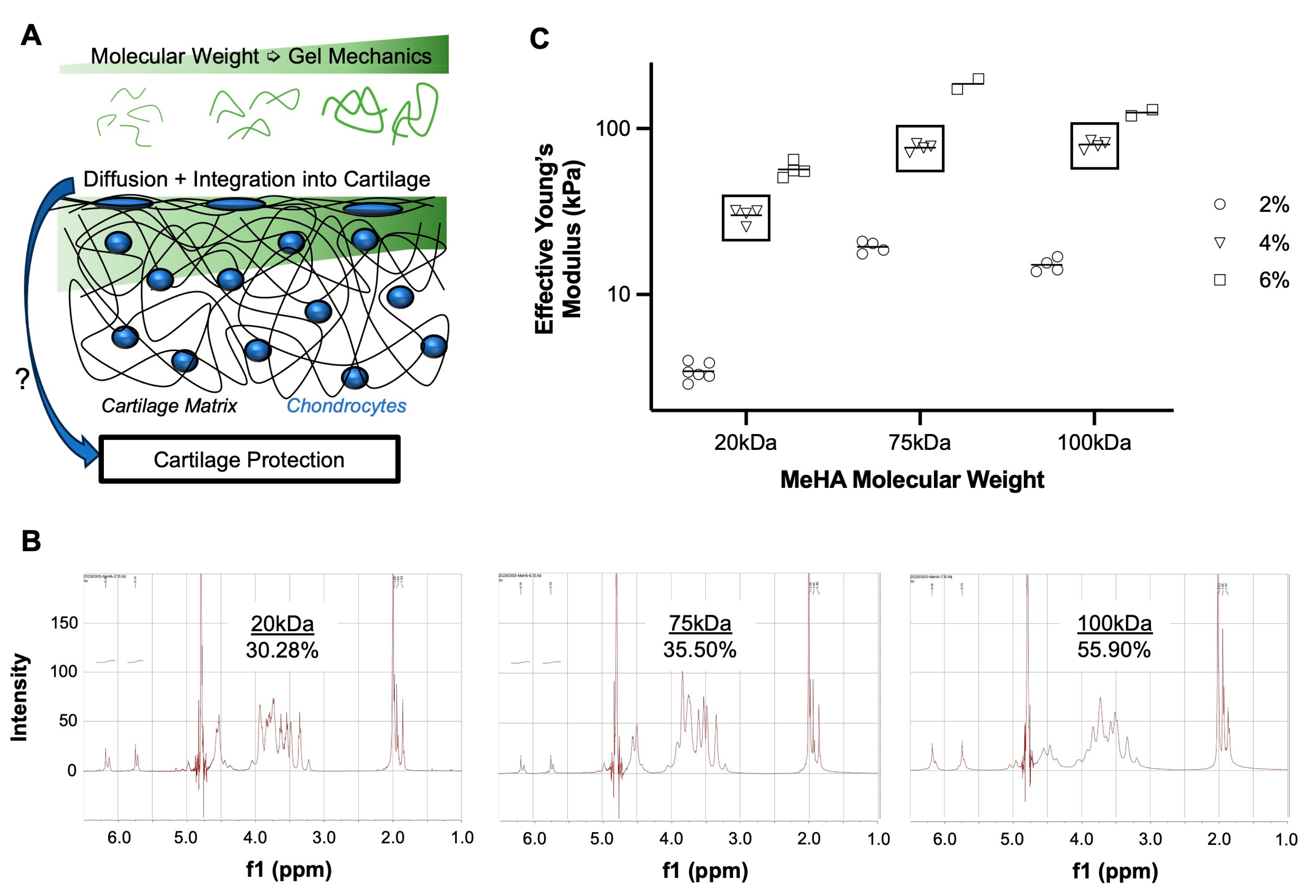
3.1. Higher MW MeHA Forms Stiffer Hydrogel
3.2. Lower MW MeHA Integrates Better Than Higher MW MeHA
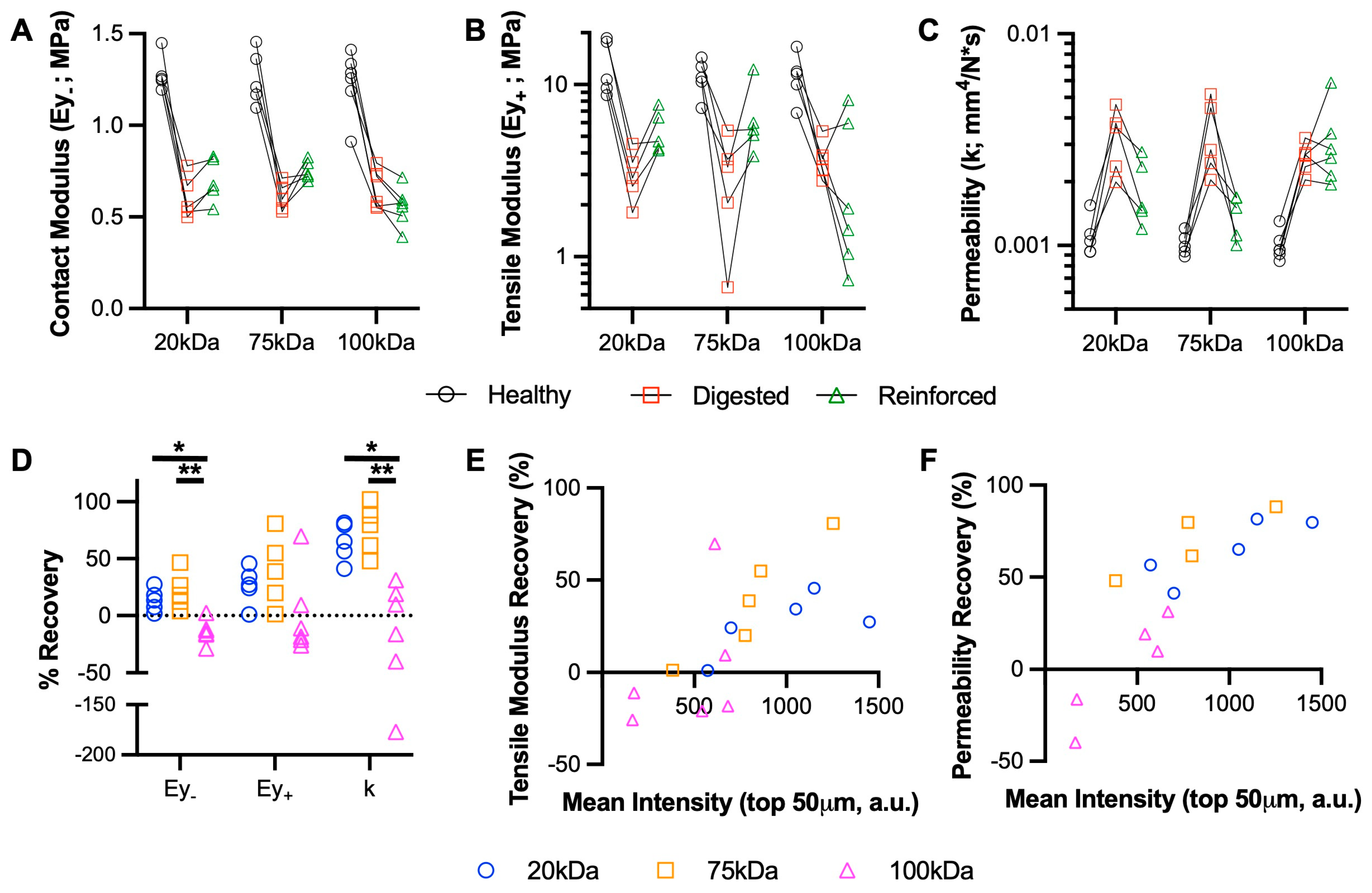
3.3. Medium MW MeHA Provides Greatest Restoration of Mechanical Properties
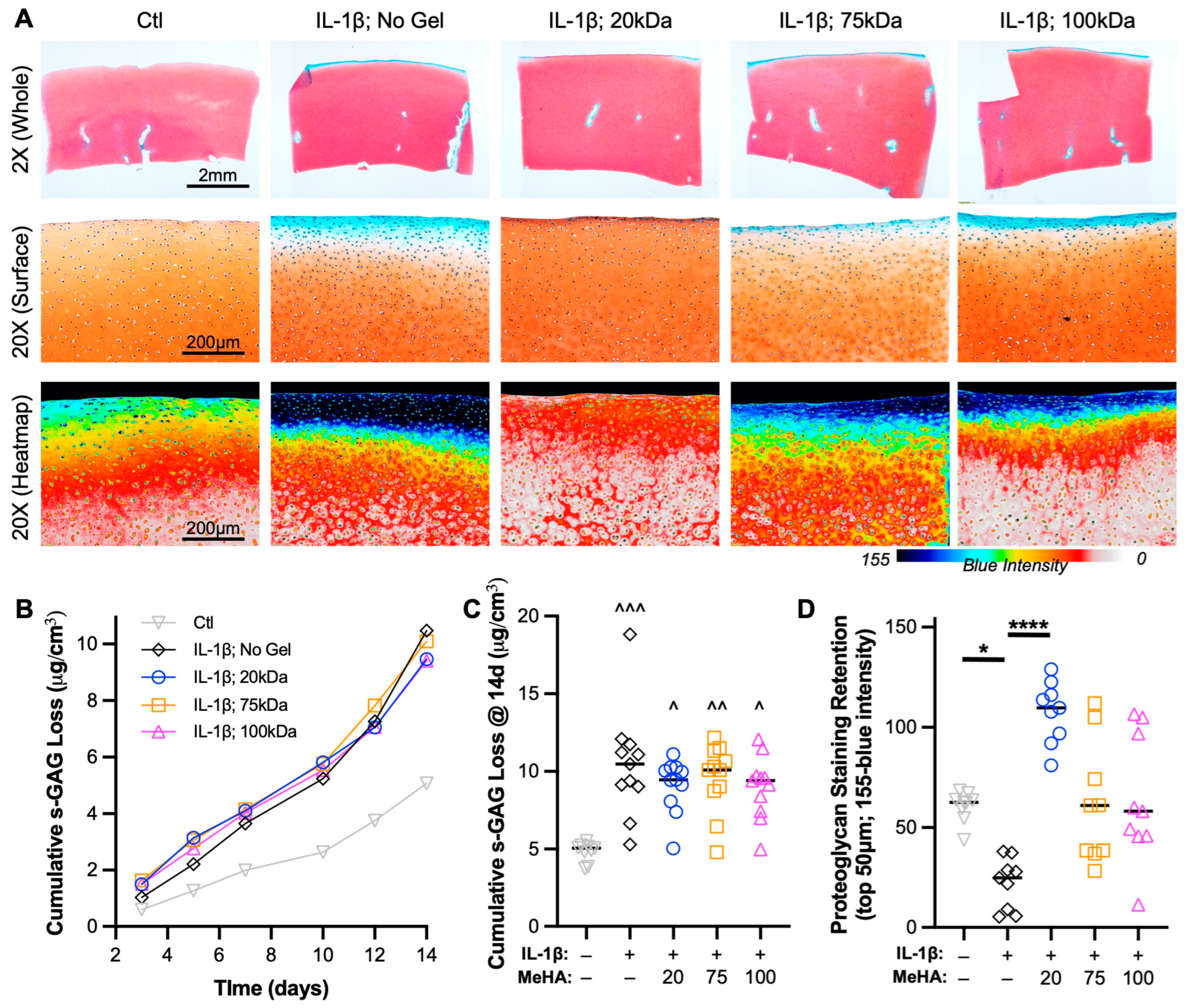
3.4. Low MW MeHA Provides Greatest Biomechanical Protection during Catabolic Culture
3.5. Low MW MeHA Prevents Superficial Proteoglycan Loss and Matrix Catabolism
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sanders, T.L.; Maradit Kremers, H.; Bryan, A.J.; Larson, D.R.; Dahm, D.L.; Levy, B.A.; Stuart, M.J.; Krych, A.J. Incidence of Anterior Cruciate Ligament Tears and Reconstruction: A 21-Year Population-Based Study. Am. J. Sport. Med. 2016, 44, 1502–1507. [Google Scholar] [CrossRef] [PubMed]
- Baker, B.E.; Peckham, A.C.; Pupparo, F.; Sanborn, J.C. Review of meniscal injury and associated sports. Am. J. Sport. Med. 1985, 13, 1–4. [Google Scholar] [CrossRef]
- Domnick, C.; Raschke, M.J.; Herbort, M. Biomechanics of the anterior cruciate ligament: Physiology, rupture and reconstruction techniques. World J. Orthop. 2016, 7, 82–93. [Google Scholar] [CrossRef]
- Pozzi, A.; Tonks, C.A.; Ling, H.Y. Femorotibial contact mechanics and meniscal strain after serial meniscectomy. Vet. Surg. 2010, 39, 482–488. [Google Scholar] [CrossRef] [PubMed]
- Luc, B.; Gribble, P.A.; Pietrosimone, B.G. Osteoarthritis prevalence following anterior cruciate ligament reconstruction: A systematic review and numbers-needed-to-treat analysis. J. Athl. Train. 2014, 49, 806–819. [Google Scholar] [CrossRef]
- Cinque, M.E.; Dornan, G.J.; Chahla, J.; Moatshe, G.; LaPrade, R.F. High Rates of Osteoarthritis Develop After Anterior Cruciate Ligament Surgery: An Analysis of 4108 Patients. Am. J. Sport. Med. 2018, 46, 2011–2019. [Google Scholar] [CrossRef] [PubMed]
- Bitton, R. The economic burden of osteoarthritis. Am. J. Manag. Care 2009, 15, S230–S235. [Google Scholar]
- Inoue, M.; Muneta, T.; Ojima, M.; Nakamura, K.; Koga, H.; Sekiya, I.; Okazaki, M.; Tsuji, K. Inflammatory cytokine levels in synovial fluid 3, 4 days postoperatively and its correlation with early-phase functional recovery after anterior cruciate ligament reconstruction: A cohort study. J. Exp. Orthop. 2016, 3, 30. [Google Scholar] [CrossRef]
- Jacobs, C.A.; Hunt, E.R.; Conley, C.E.; Johnson, D.L.; Stone, A.V.; Huebner, J.L.; Kraus, V.B.; Lattermann, C. Dysregulated Inflammatory Response Related to Cartilage Degradation after ACL Injury. Med. Sci. Sport. Exerc. 2020, 52, 535–541. [Google Scholar] [CrossRef]
- Irie, K.; Uchiyama, E.; Iwaso, H. Intraarticular inflammatory cytokines in acute anterior cruciate ligament injured knee. Knee 2003, 10, 93–96. [Google Scholar] [CrossRef] [PubMed]
- Bigoni, M.; Turati, M.; Sacerdote, P.; Gaddi, D.; Piatti, M.; Castelnuovo, A.; Franchi, S.; Gandolla, M.; Pedrocchi, A.; Omeljaniuk, R.J.; et al. Characterization of synovial fluid cytokine profiles in chronic meniscal tear of the knee. J. Orthop. Res. Off. Publ. Orthop. Res. Soc. 2017, 35, 340–346. [Google Scholar] [CrossRef] [PubMed]
- King, J.D.; Rowland, G.; Villasante Tezanos, A.G.; Warwick, J.; Kraus, V.B.; Lattermann, C.; Jacobs, C.A. Joint Fluid Proteome after Anterior Cruciate Ligament Rupture Reflects an Acute Posttraumatic Inflammatory and Chondrodegenerative State. Cartilage 2020, 11, 329–337. [Google Scholar] [CrossRef] [PubMed]
- Hunt, E.R.; Jacobs, C.A.; Conley, C.E.; Ireland, M.L.; Johnson, D.L.; Lattermann, C. Anterior cruciate ligament reconstruction reinitiates an inflammatory and chondrodegenerative process in the knee joint. J. Orthop. Res. Off. Publ. Orthop. Res. Soc. 2021, 39, 1281–1288. [Google Scholar] [CrossRef]
- Troeberg, L.; Nagase, H. Proteases involved in cartilage matrix degradation in osteoarthritis. Biochim. Biophys. Acta 2012, 1824, 133–145. [Google Scholar] [CrossRef] [PubMed]
- Knudson, W.; Ishizuka, S.; Terabe, K.; Askew, E.B.; Knudson, C.B. The pericellular hyaluronan of articular chondrocytes. Matrix Biol. 2019, 78–79, 32–46. [Google Scholar] [CrossRef]
- Fischenich, K.M.; Button, K.D.; Coatney, G.A.; Fajardo, R.S.; Leikert, K.M.; Haut, R.C.; Haut Donahue, T.L. Chronic Changes in the Articular Cartilage and Meniscus Following Traumatic Impact to the Lapine Knee. J. Biomech. 2015, 48, 246–253. [Google Scholar] [CrossRef]
- Chery, D.R.; Han, B.; Li, Q.; Zhou, Y.; Heo, S.J.; Kwok, B.; Chandrasekaran, P.; Wang, C.; Qin, L.; Lu, X.L.; et al. Early changes in cartilage pericellular matrix micromechanobiology portend the onset of post-traumatic osteoarthritis. Acta Biomater. 2020, 111, 267–278. [Google Scholar] [CrossRef]
- Orozco, G.A.; Eskelinen, A.S.A.; Kosonen, J.P.; Tanaka, M.S.; Yang, M.; Link, T.M.; Ma, B.; Li, X.; Grodzinsky, A.J.; Korhonen, R.K.; et al. Shear strain and inflammation-induced fixed charge density loss in the knee joint cartilage following ACL injury and reconstruction: A computational study. J. Orthop. Res. Off. Publ. Orthop. Res. Soc. 2022, 40, 1505–1522. [Google Scholar] [CrossRef]
- Testa, G.; Giardina, S.M.C.; Culmone, A.; Vescio, A.; Turchetta, M.; Cannavo, S.; Pavone, V. Intra-Articular Injections in Knee Osteoarthritis: A Review of Literature. J. Funct. Morphol. Kinesiol. 2021, 6, 15. [Google Scholar] [CrossRef]
- Edsman, K.; Hjelm, R.; Larkner, H.; Nord, L.I.; Karlsson, A.; Wiebensjo, A.; Hoglund, A.U.; Kenne, A.H.; Nasstrom, J. Intra-articular Duration of Durolane after Single Injection into the Rabbit Knee. Cartilage 2011, 2, 384–388. [Google Scholar] [CrossRef]
- Jackson, D.W.; Simon, T.M. Intra-articular distribution and residence time of Hylan A and B: A study in the goat knee. Osteoarthr. Cartil. 2006, 14, 1248–1257. [Google Scholar] [CrossRef]
- Selig, D.J.; Kress, A.T.; Horton, I.M.; Livezey, J.R.; Sadik, E.J.; DeLuca, J.P. Pharmacokinetics, safety and efficacy of intra-articular non-steroidal anti-inflammatory drug injections for the treatment of osteoarthritis: A narrative review. J. Clin. Pharm. Ther. 2022, 47, 1122–1133. [Google Scholar] [CrossRef]
- Bajpayee, A.G.; De la Vega, R.E.; Scheu, M.; Varady, N.H.; Yannatos, I.A.; Brown, L.A.; Krishnan, Y.; Fitzsimons, T.J.; Bhattacharya, P.; Frank, E.H.; et al. Sustained intra-cartilage delivery of low dose dexamethasone using a cationic carrier for treatment of post traumatic osteoarthritis. Eur. Cells Mater. 2017, 34, 341–364. [Google Scholar] [CrossRef]
- Mehta, S.; Boyer, T.L.; Akhtar, S.; He, T.; Zhang, C.; Vedadghavami, A.; Bajpayee, A.G. Sustained intra-cartilage delivery of interleukin-1 receptor antagonist using cationic peptide and protein-based carriers. Osteoarthr. Cartil. 2023, 31, 780–792. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Adjei, I.M.; Brown, S.B.; Liseth, O.; Sharma, B. Manganese dioxide nanoparticles protect cartilage from inflammation-induced oxidative stress. Biomaterials 2019, 224, 119467. [Google Scholar] [CrossRef] [PubMed]
- Bonitsky, C.M.; McGann, M.E.; Selep, M.J.; Ovaert, T.C.; Trippel, S.B.; Wagner, D.R. Genipin crosslinking decreases the mechanical wear and biochemical degradation of impacted cartilage in vitro. J. Orthop. Res. Off. Publ. Orthop. Res. Soc. 2017, 35, 558–565. [Google Scholar] [CrossRef]
- McGann, M.E.; Bonitsky, C.M.; Jackson, M.L.; Ovaert, T.C.; Trippel, S.B.; Wagner, D.R. Genipin crosslinking of cartilage enhances resistance to biochemical degradation and mechanical wear. J. Orthop. Res. Off. Publ. Orthop. Res. Soc. 2015, 33, 1571–1579. [Google Scholar] [CrossRef] [PubMed]
- Cooper, B.G.; Lawson, T.B.; Snyder, B.D.; Grinstaff, M.W. Reinforcement of articular cartilage with a tissue-interpenetrating polymer network reduces friction and modulates interstitial fluid load support. Osteoarthr. Cartil. 2017, 25, 1143–1149. [Google Scholar] [CrossRef]
- Makela, J.T.A.; Cooper, B.G.; Korhonen, R.K.; Grinstaff, M.W.; Snyder, B.D. Functional effects of an interpenetrating polymer network on articular cartilage mechanical properties. Osteoarthr. Cartil. 2018, 26, 414–421. [Google Scholar] [CrossRef]
- Kahle, E.R.; Han, B.; Chandrasekaran, P.; Phillips, E.R.; Mulcahey, M.K.; Lu, X.L.; Marcolongo, M.S.; Han, L. Molecular Engineering of Pericellular Microniche via Biomimetic Proteoglycans Modulates Cell Mechanobiology. ACS Nano 2022, 16, 1220–1230. [Google Scholar] [CrossRef]
- Phillips, E.R.; Prudnikova, K.; Bui, T.; Taylor, A.J.; Galindo, D.A.; Huneke, R.B.; Hou, J.S.; Mulcahey, M.K.; Marcolongo, M.S. Biomimetic proteoglycans can molecularly engineer early osteoarthritic cartilage in vivo. J. Orthop. Res. Off. Publ. Orthop. Res. Soc. 2019, 37, 403–411. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Qiu, Y.; Qu, L.; Wang, Q.; Zhou, Q. Hydrogels for Treatment of Different Degrees of Osteoarthritis. Front. Bioeng. Biotechnol. 2022, 10, 858656. [Google Scholar] [CrossRef] [PubMed]
- Wei, W.; Ma, Y.; Yao, X.; Zhou, W.; Wang, X.; Li, C.; Lin, J.; He, Q.; Leptihn, S.; Ouyang, H. Advanced hydrogels for the repair of cartilage defects and regeneration. Bioact. Mater. 2021, 6, 998–1011. [Google Scholar] [CrossRef] [PubMed]
- Kowalski, M.A.; Fernandes, L.M.; Hammond, K.E.; Labib, S.; Drissi, H.; Patel, J.M. Cartilage-penetrating hyaluronic acid hydrogel preserves tissue content and reduces chondrocyte catabolism. J. Tissue Eng. Regen. Med. 2022, 16, 1138–1148. [Google Scholar] [CrossRef] [PubMed]
- DiDomenico, C.D.; Lintz, M.; Bonassar, L.J. Molecular transport in articular cartilage—What have we learned from the past 50 years? Nat. Rev. Rheumatol. 2018, 14, 393–403. [Google Scholar] [CrossRef] [PubMed]
- van Lent, P.L.; van den Berg, W.B.; Schalkwijk, J.; van de Putte, L.B.; van den Bersselaar, L. The impact of protein size and charge on its retention in articular cartilage. J. Rheumatol. 1987, 14, 798–805. [Google Scholar]
- Tsanaktsidou, E.; Kammona, O.; Kiparissides, C. On the synthesis and characterization of biofunctional hyaluronic acid based injectable hydrogels for the repair of cartilage lesions. Eur. Polym. J. 2019, 114, 47–56. [Google Scholar] [CrossRef]
- Ma, W.; Suh, W.H. Cost-Effective Cosmetic-Grade Hyaluronan Hydrogels for ReNcell VM Human Neural Stem Cell Culture. Biomolecules 2019, 9, 515. [Google Scholar] [CrossRef]
- Patel, J.M.; Wise, B.C.; Bonnevie, E.D.; Mauck, R.L. A Systematic Review and Guide to Mechanical Testing for Articular Cartilage Tissue Engineering. Tissue Eng. Part. C Methods 2019, 25, 593–608. [Google Scholar] [CrossRef]
- Moore, A.C.; DeLucca, J.F.; Elliott, D.M.; Burris, D.L. Quantifying Cartilage Contact Modulus, Tension Modulus, and Permeability With Hertzian Biphasic Creep. J. Tribol. 2016, 138, 0414051–0414057. [Google Scholar] [CrossRef]
- Grenier, S.; Bhargava, M.M.; Torzilli, P.A. An in vitro model for the pathological degradation of articular cartilage in osteoarthritis. J. Biomech. 2014, 47, 645–652. [Google Scholar] [CrossRef]
- Lv, M.; Zhou, Y.; Polson, S.W.; Wan, L.Q.; Wang, M.; Han, L.; Wang, L.; Lu, X.L. Identification of Chondrocyte Genes and Signaling Pathways in Response to Acute Joint Inflammation. Sci. Rep. 2019, 9, 93. [Google Scholar] [CrossRef] [PubMed]
- Bansal, S.; Miller, L.M.; Patel, J.M.; Meadows, K.D.; Eby, M.R.; Saleh, K.S.; Martin, A.R.; Stoeckl, B.D.; Hast, M.W.; Elliott, D.M.; et al. Transection of the medial meniscus anterior horn results in cartilage degeneration and meniscus remodeling in a large animal model. J. Orthop. Res. Off. Publ. Orthop. Res. Soc. 2020, 38, 2696–2708. [Google Scholar] [CrossRef] [PubMed]
- Majda, D.; Bhattarai, A.; Riikonen, J.; Napruszewska, B.D.; Zimowska, M.; Michalik-Zym, A.; Tӧyrӓs, J.; Lehto, V.P. New approach for determining cartilage pore size distribution: NaCl-thermoporometry. Microporous Mesoporous Mater. 2017, 241, 238–245. [Google Scholar] [CrossRef]
- Bajpayee, A.G.; Wong, C.R.; Bawendi, M.G.; Frank, E.H.; Grodzinsky, A.J. Avidin as a model for charge driven transport into cartilage and drug delivery for treating early stage post-traumatic osteoarthritis. Biomaterials 2014, 35, 538–549. [Google Scholar] [CrossRef] [PubMed]
- Kuehl, C.; Zhang, T.; Kaminskas, L.M.; Porter, C.J.; Davies, N.M.; Forrest, L.; Berkland, C. Hyaluronic Acid Molecular Weight Determines Lung Clearance and Biodistribution after Instillation. Mol. Pharm. 2016, 13, 1904–1914. [Google Scholar] [CrossRef]
- Grenier, S.; Donnelly, P.E.; Gittens, J.; Torzilli, P.A. Resurfacing damaged articular cartilage to restore compressive properties. J. Biomech. 2015, 48, 122–129. [Google Scholar] [CrossRef]
- Liu, Z.; Lin, W.; Fan, Y.; Kampf, N.; Wang, Y.; Klein, J. Effects of Hyaluronan Molecular Weight on the Lubrication of Cartilage-Emulating Boundary Layers. Biomacromolecules 2020, 21, 4345–4354. [Google Scholar] [CrossRef]
- Homandberg, G.A.; Ummadi, V.; Kang, H. High molecular weight hyaluronan promotes repair of IL-1 beta-damaged cartilage explants from both young and old bovines. Osteoarthr. Cartil. 2003, 11, 177–186. [Google Scholar] [CrossRef]
- Tavianatou, A.G.; Caon, I.; Franchi, M.; Piperigkou, Z.; Galesso, D.; Karamanos, N.K. Hyaluronan: Molecular size-dependent signaling and biological functions in inflammation and cancer. FEBS J. 2019, 286, 2883–2908. [Google Scholar] [CrossRef]
- Quero, L.; Klawitter, M.; Schmaus, A.; Rothley, M.; Sleeman, J.; Tiaden, A.N.; Klasen, J.; Boos, N.; Hottiger, M.O.; Wuertz, K.; et al. Hyaluronic acid fragments enhance the inflammatory and catabolic response in human intervertebral disc cells through modulation of toll-like receptor 2 signalling pathways. Arthritis Res. Ther. 2013, 15, R94. [Google Scholar] [CrossRef] [PubMed]
- Knudson, W.; Casey, B.; Nishida, Y.; Eger, W.; Kuettner, K.E.; Knudson, C.B. Hyaluronan oligosaccharides perturb cartilage matrix homeostasis and induce chondrocytic chondrolysis. Arthritis Rheum. 2000, 43, 1165–1174. [Google Scholar] [CrossRef] [PubMed]
- Bian, L.; Lima, E.G.; Angione, S.L.; Ng, K.W.; Williams, D.Y.; Xu, D.; Stoker, A.M.; Cook, J.L.; Ateshian, G.A.; Hung, C.T. Mechanical and biochemical characterization of cartilage explants in serum-free culture. J. Biomech. 2008, 41, 1153–1159. [Google Scholar] [CrossRef]
- Geiger, B.C.; Wang, S.; Padera, R.F., Jr.; Grodzinsky, A.J.; Hammond, P.T. Cartilage-penetrating nanocarriers improve delivery and efficacy of growth factor treatment of osteoarthritis. Sci. Transl. Med. 2018, 10, eaat8800. [Google Scholar] [CrossRef] [PubMed]
- Patel, J.M.; Loebel, C.; Saleh, K.S.; Wise, B.C.; Bonnevie, E.D.; Miller, L.M.; Carey, J.L.; Burdick, J.A.; Mauck, R.L. Stabilization of Damaged Articular Cartilage with Hydrogel-Mediated Reinforcement and Sealing. Adv. Healthc. Mater. 2021, 10, e2100315. [Google Scholar] [CrossRef]




Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Brackin, R.B.; McColgan, G.E.; Pucha, S.A.; Kowalski, M.A.; Drissi, H.; Doan, T.N.; Patel, J.M. Improved Cartilage Protection with Low Molecular Weight Hyaluronic Acid Hydrogel. Bioengineering 2023, 10, 1013. https://doi.org/10.3390/bioengineering10091013
Brackin RB, McColgan GE, Pucha SA, Kowalski MA, Drissi H, Doan TN, Patel JM. Improved Cartilage Protection with Low Molecular Weight Hyaluronic Acid Hydrogel. Bioengineering. 2023; 10(9):1013. https://doi.org/10.3390/bioengineering10091013
Chicago/Turabian StyleBrackin, Riley B., Gail E. McColgan, Saitheja A. Pucha, Michael A. Kowalski, Hicham Drissi, Thanh N. Doan, and Jay M. Patel. 2023. "Improved Cartilage Protection with Low Molecular Weight Hyaluronic Acid Hydrogel" Bioengineering 10, no. 9: 1013. https://doi.org/10.3390/bioengineering10091013
APA StyleBrackin, R. B., McColgan, G. E., Pucha, S. A., Kowalski, M. A., Drissi, H., Doan, T. N., & Patel, J. M. (2023). Improved Cartilage Protection with Low Molecular Weight Hyaluronic Acid Hydrogel. Bioengineering, 10(9), 1013. https://doi.org/10.3390/bioengineering10091013






