Meta-Transcriptomic Analysis Reveals Novel RNA Viruses in Polychaetes Perinereis
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Collection and Processing
2.2. RNA Library Construction and Meta-Transcriptomic Sequencing
2.3. Sequence Assembly and RNA Virus Discovery
2.4. Genome Structure Prediction and Phylogenetic Analysis
3. Results
3.1. Composition and Diversity of the Perinereis Virome
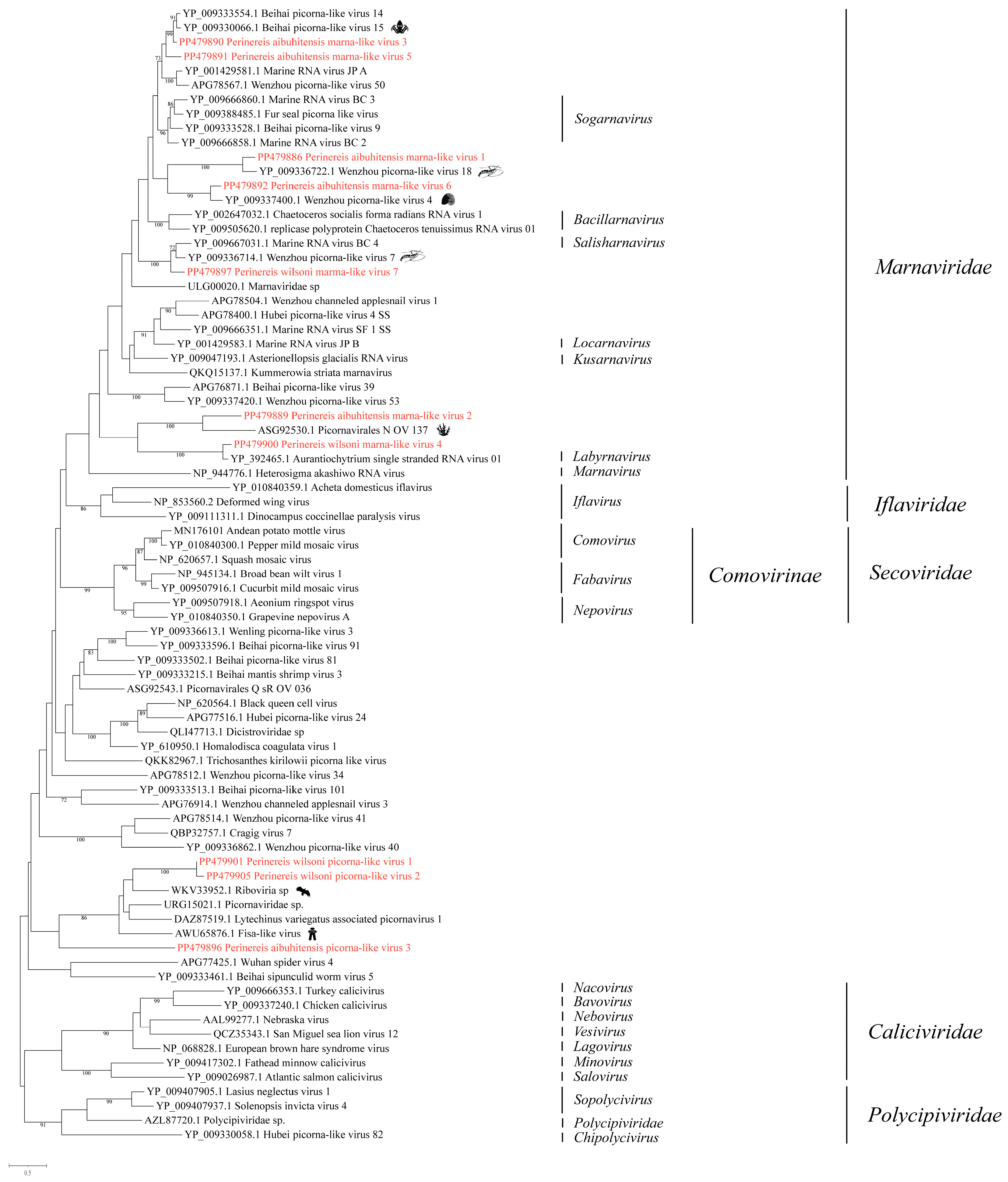
3.2. Phylogenetic Analysis of the Novel Viruses in the Order Picornavirales
3.2.1. Phylogenetic Analysis of the Novel Viruses in the Family Marnaviridae
3.2.2. Phylogenetic Analysis of the Novel Viruses in Unclassified Picornavirales
3.3. Phylogenetic Analysis of the Novel Viruses in the Family Picobirnaviridae
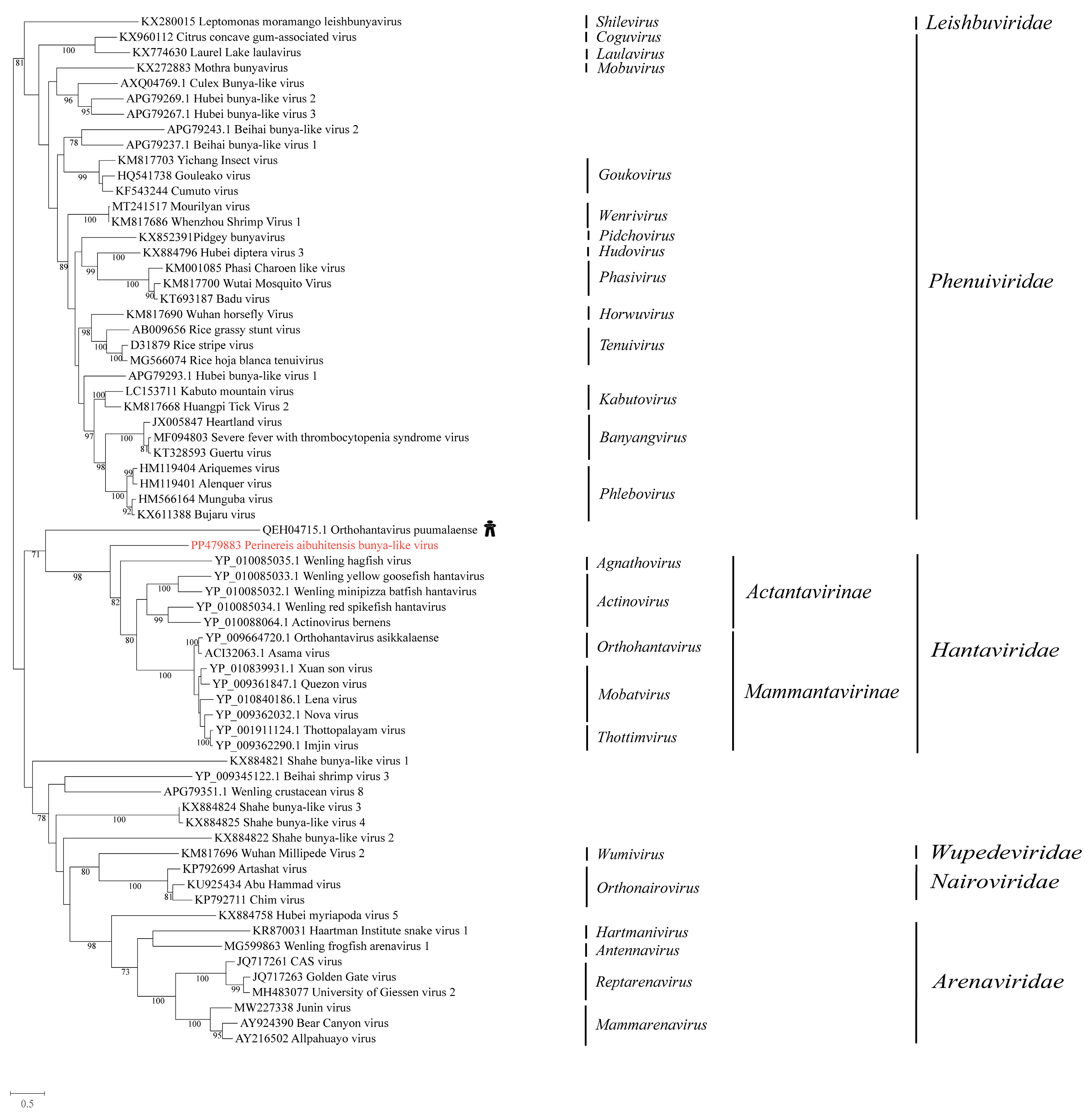
3.4. Phylogenetic Analysis of the Novel Viruses in Unclassified Bunyavirales
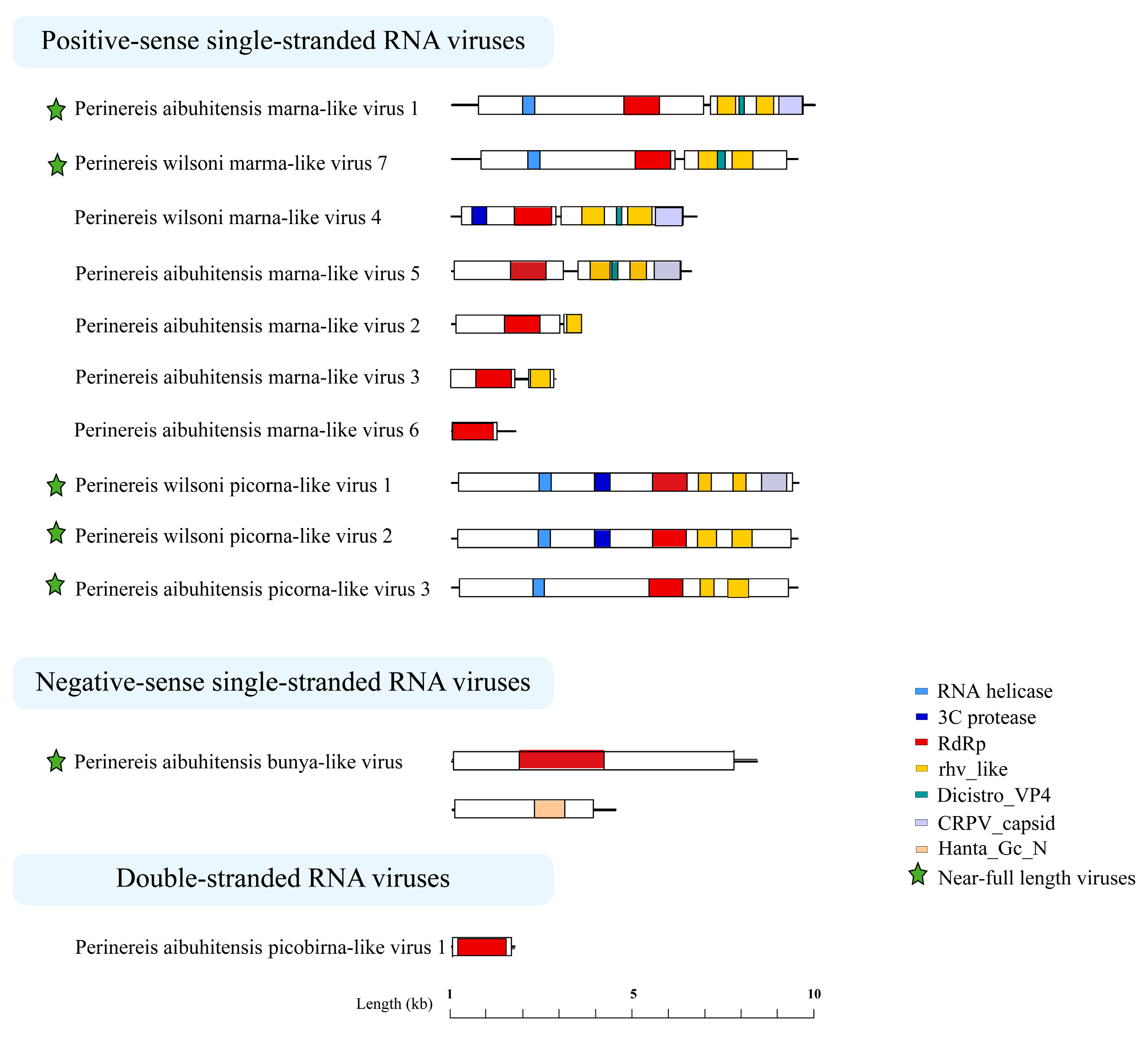
3.5. Genome Structures of the Perinereis Viruses
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wilson, R.S.; Glasby, C.J.; Bakken, T. The Nereididae (Annelida)—Diagnoses, descriptions, and a key to the genera. ZooKeys 2023, 1182, 35. [Google Scholar] [CrossRef] [PubMed]
- Villalobos-Guerrero, T.F. Redescription of ttwo overlooked species of the Perinereis Nuntia complex and morphological delimitation of P. Nuntia (Savigny in Lamarck, 1818) from the Red Sea (Annelida, Nereididae). Zoosystema 2019, 41, 465–496. [Google Scholar] [CrossRef]
- Elgetany, A.H.; Struck, T.H.; Glasby, C.J. Three new species of the genus Perinereis (Annelida, Nereididae) from egyptian coasts. ZooKeys 2022, 1132, 163–188. [Google Scholar] [CrossRef] [PubMed]
- Villalobos-Guerrero, T.F.; Park, T.; Idris, I. Review of some Perinereis Kinberg, 1865 (Annelida: Nereididae) species of group 2 sensu Hutchings, Reid & Wilson, 1991 from the eastern and south-eastern Asian seas. J. Mar. Biol. Assoc. UK 2021, 101, 279–307. [Google Scholar] [CrossRef]
- Conde-Vela, V.M. Reinstatement of Perinereis Bairdii (Webster, 1884) and description of P. Websteri Sp. Nov. from Bermuda, including the reproductive morphology of two Atlantic Perinereis Species (Annelida: Errantia: Phyllodocida). Eur. J. Taxon. 2021, 787, 104–145. [Google Scholar] [CrossRef]
- Sakurai, T.; Kobayashi, J.; Ito, N.; Serizawa, S.; Shiraishi, H.; Yabe, T.; Ishii, Y.; Suzuki, N. Respiratory uptake and depuration kinetics of perfluorooctanesulfonate (PFOS) in a marine sandworm species. Bull. Environ. Contam. Toxicol. 2017, 99, 203–207. [Google Scholar] [CrossRef] [PubMed]
- Fang, J.; Zhang, J.; Jiang, Z.; Du, M.; Liu, Y.; Mao, Y.; Gao, Y.; Fang, J. Environmental remediation potential of Perinereis aibuhitensis (Polychaeta) based on the effects of temperature and feed types on its carbon and nitrogen budgets. Mar. Biol. Res. 2016, 12, 583–594. [Google Scholar] [CrossRef]
- Wouters, R.; Lavens, P.; Nieto, J.; Sorgeloos, P. Penaeid shrimp broodstock nutrition: An updated review on research and development. Aquaculture 2001, 202, 1–21. [Google Scholar] [CrossRef]
- Moussa Dorgham, M.; Hamdy, R.; Hassan, H.; Rasheedy, A.; Mohamed Atta, M. Biochemical composition of the nereidid Perinereis cultrifera from the Egyptian Mediterranean coast. Rev. Biol. Mar. Oceanogr. 2015, 50, 535–543. [Google Scholar] [CrossRef]
- Techaprempreecha, S.; Khongchareonporn, N.; Chaicharoenpong, C.; Aranyakananda, P.; Chunhabundit, S.; Petsom, A. Nutritional composition of farmed and wild sandworms, Perinereis Nuntia. Fuel Energy Abstr. 2011, 169, 265–269. [Google Scholar] [CrossRef]
- Lytle, J.S.; Lytle, T.F.; Ogle, J.T. Polyunsaturated fatty acid profiles as a comparative tool in assessing maturation diets of Penaeus Vannamei. Aquaculture 1990, 89, 287–299. [Google Scholar] [CrossRef]
- Lim, J.A.; Loo, P.L.; Tan, K.S.; Ng, N.K. Fish culture waste improves growth and fatty acid composition of the Polychaete Namalycastis sp. (Nereididae) and its potential use as feed for mud crabs. Aquac. Res. 2021, 52, 2622–2639. [Google Scholar] [CrossRef]
- Olive, P. Polychaeta as a World Resource: A review of patterns of exploitation as sea angling baits and the potential for aquaculture based production. Mem. Mus. Natn. Hist. Nat. 1994, 162, 23–27. [Google Scholar]
- Vijayan, K.; Raj, V.S.; Balasubramanian, C.; Alavandi, S.; Sekhar, V.T.; Santiago, T. Polychaete Worms—A vector for white spot syndrome virus (WSSV). Dis. Aquat. Organ. 2005, 63, 107–111. [Google Scholar] [CrossRef] [PubMed]
- Desrina, H. On the Role of the Polychaete Dendronereis Spp. in the Transmission of White Spot Syndrome Virus in Shrimp Ponds; Wageningen University: Wageningen, The Netherlands, 2014. [Google Scholar]
- Flegel, T.; Fegan, D.F. Strategies for preventing the spread of fish and shellfish diseases. Fish. Sci. 2002, 68, 776–788. [Google Scholar] [CrossRef] [PubMed]
- Flegel, T.W. RNA-Seq data analysis in galaxy. ScienceAsia 2006, 32, 215. [Google Scholar] [CrossRef]
- Thitamadee, S.; Prachumwat, A.; Srisala, J.; Jaroenlak, P.; Salachan, P.V.; Sritunyalucksana, K.; Flegel, T.W.; Itsathitphaisarn, O. Review of current disease threats for cultivated penaeid shrimp in Asia. Aquaculture 2016, 452, 69–87. [Google Scholar] [CrossRef]
- WOAH. Report of the Meeting of WOAH Aquatic Animal Health Standards Commission. Available online: https://www.woah.org/app/uploads/2022/11/a-aac-sep-2022-report.pdf (accessed on 22 May 2024).
- Haryadi, D.; Verreth, J.a.J.; Verdegem, M.C.J.; Vlak, J.M. Transmission of white spot syndrome sirus (WSSV) from Dendronereis Spp. (Peters) (Nereididae) to Penaeid shrimp. J. Fish Dis. 2015, 38, 419–428. [Google Scholar] [CrossRef] [PubMed]
- NACA. Disease Advisory: Decapod Iridescent Virus 1 (DIV1): An Emerging Threat to the Shrimp Industry. Available online: https://enaca.org/?id=1098&title=decapod-iridescent-virus-1-an-emerging-threat-to-the-shrimpindustry (accessed on 22 May 2024).
- Qiu, L.; Chen, M.M.; Wan, X.Y.; Li, C.; Zhang, Q.L.; Wang, R.Y.; Cheng, D.Y.; Dong, X.; Yang, B.; Wang, X.H.; et al. Characterization of a new member of Iridoviridae, shrimp hemocyte iridescent virus (SHIV), found in white leg shrimp (Litopenaeus vannamei). Sci. Rep. 2017, 7, 11834. [Google Scholar] [CrossRef]
- Qiu, L.; Chen, M.M.; Wan, X.Y.; Zhang, Q.L.; Li, C.; Dong, X.; Yang, B.; Huang, J. Detection and quantification of shrimp hemocyte iridescent virus by TaqMan probe based real-time PCR. J. Invertebr. Pathol. 2018, 154, 95–101. [Google Scholar] [CrossRef]
- Zhao, W.X.; Wan, X.Y.; Xia, J.T.; Yao, L.; Xv, R.D.; Wang, W.; Yu, X.T.; Zhang, Q.L. Investigation of the prevalence of covert mortality nodavirus (CMNV) in shrimp from 2021 to 2022. Prog. Fish. Sci. 2024, 45, 1–10. [Google Scholar] [CrossRef]
- Devauchelle, G. Ultrastructural characterization of an iridovirus from the marine worm Nereis diversicolor (O.F. Müller). Virology 1977, 81, 237–246. [Google Scholar] [CrossRef] [PubMed]
- Fidalgo E Costa, P.; Gil, J.; Passos, A.M.; Pereira, P.; Melo, P.; Batista, F.; Cancela Da Fonseca, L. The market features of imported non-indigenous Polychaetes in Portugal and consequent ecological concerns. Sci. Mar. 2006, 70, 287–292. [Google Scholar] [CrossRef]
- Haditomo, A.H.C.; Chilmawati, D. The white sport syndrome virus (WSSV) load in Dendronereis spp. 2012. [Google Scholar]
- Prastowo, B.W.; Ariawan, K.; Nur, E.M.; Rahardianti, R. Identification of Polychaete worm, Nereis Sp. as a white spot syndrome virus (WSSV) vector in their habitat and challenge test in the laboratory. J. Fish. Sci. 2009, 11, 183–191. [Google Scholar]
- Yang, M.; Zeng, C.; Xia, J.; Liu, Q.; Fang, J.; Zhang, Q. Disinfection of Perinereis aibuhitensis eggs with peroxymonosulfate to eliminate covert mortality nodavirus (CMNV). Aquaculture 2023, 572, 739539. [Google Scholar] [CrossRef]
- Palacios, G.; Druce, J.; Du, L.; Tran, T.; Birch, C.; Briese, T.; Conlan, S.; Quan, P.-L.; Hui, J.; Marshall, J.; et al. A new arenavirus in a cluster of fatal transplant-associated diseases. N. Engl. J. Med. 2008, 358, 991–998. [Google Scholar] [CrossRef] [PubMed]
- Shi, M.; Lin, X.D.; Tian, J.H.; Chen, L.J.; Chen, X.; Li, C.X.; Qin, X.C.; Li, J.; Cao, J.P.; Eden, J.S.; et al. Redefining the invertebrate RNA virosphere. Nature 2016, 540, 539–543. [Google Scholar] [CrossRef] [PubMed]
- Bikel, S.; Valdez-Lara, A.; Cornejo-Granados, F.; Rico, K.; Canizales-Quinteros, S.; Soberón, X.; Del Pozo-Yauner, L.; Ochoa-Leyva, A. Combining metagenomics, metatranscriptomics and viromics to explore novel Microbial Interactions: Towards a Systems-Level Understanding of Human Microbiome. Comput. Struct. Biotechnol. J. 2015, 13, 390–401. [Google Scholar] [CrossRef] [PubMed]
- Shi, M.; Zhang, Y.Z.; Holmes, E.C. Meta-transcriptomics and the evolutionary biology of RNA viruses. Virus Res. 2018, 243, 83–90. [Google Scholar] [CrossRef]
- Dong, X.; Hu, T.; Liu, Q.; Li, C.; Sun, Y.; Wang, Y. A novel hepe-like virus from farmed giant freshwater prawn Macrobrachium Rosenbergii. Viruses 2020, 3, 323. [Google Scholar] [CrossRef]
- Dong, X.; Li, C.; Wang, Y.; Hu, T.; Zhang, F.; Meng, F.; Gao, M.; Han, X.; Wang, G.; Qin, J.; et al. Diversity and connectedness of brine shrimp viruses in global hypersaline ecosystems. Sci. China Life Sci. 2024, 67, 188–203. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Zhou, Y.; Chen, Y.; Gu, J. Fastp: An ultra-fast all-in-one FASTQ preprocessor. Bioinformatics 2018, 34, i884–i890. [Google Scholar] [CrossRef] [PubMed]
- Langmead, B.; Salzberg, S.L. Fast gapped-read alignment with Bowtie 2. Nat. Methods 2012, 9, 357–359. [Google Scholar] [CrossRef] [PubMed]
- NCBI. ORFfinder Home. Available online: https://www.ncbi.nlm.nih.gov/orffinder (accessed on 23 March 2024).
- NCBI. Conserved Domain Search. Available online: https://www.ncbi.nlm.nih.gov/Structure/cdd/wrpsb.cgi (accessed on 23 March 2024).
- Mistry, J.; Chuguransky, S.; Williams, L.; Qureshi, M.; Salazar, G.A.; Sonnhammer, E.L.L.; Tosatto, S.C.E.; Paladin, L.; Raj, S.; Richardson, L.J.; et al. Pfam: The protein families database in 2021. Nucleic Acids Res. 2021, 49, D412–D419. [Google Scholar] [CrossRef]
- InterPro. Available online: https://www.ebi.ac.uk/interpro/ (accessed on 23 March 2024).
- Edgar, R.C.; Taylor, B.; Lin, V.; Altman, T.; Barbera, P.; Meleshko, D.; Lohr, D.; Novakovsky, G.; Buchfink, B.; Al-Shayeb, B.; et al. Petabase-scale sequence alignment catalyses viral discovery. Nature 2022, 602, 142–147. [Google Scholar] [CrossRef] [PubMed]
- Katoh, K.; Standley, D.M. MAFFT multiple sequence alignment software version 7: Improvements in performance and usability. Mol. Biol. Evol. 2013, 30, 772–780. [Google Scholar] [CrossRef]
- Capella-Gutiérrez, S.; Silla-Martínez, J.M.; Gabaldón, T. trimAl: A tool for automated alignment trimming in large-scale Phylogenetic analyses. Bioinformatics 2009, 25, 1972–1973. [Google Scholar] [CrossRef]
- Nguyen, L.-T.; Schmidt, H.A.; von Haeseler, A.; Minh, B.Q. IQ-TREE: A fast and effective stochastic algorithm for estimating maximum-likelihood phylogenies. Mol. Biol. Evol. 2015, 32, 268–274. [Google Scholar] [CrossRef] [PubMed]
- Lang, A.S.; Vlok, M.; Culley, A.I.; Suttle, C.A.; Takao, Y.; Tomaru, Y. ICTV report consortium ICTV virus taxonomy profile: Marnaviridae 2021. J. Gen. Virol. 2021, 102, 001633. [Google Scholar] [CrossRef]
- Picornavirales. ICTV. Available online: https://ictv.global/report_9th/RNApos/Picornavirales (accessed on 1 February 2024).
- Siqueira, J.D.; Dominguez-Bello, M.G.; Contreras, M.; Lander, O.; Caballero-Arias, H.; Xutao, D.; Noya-Alarcon, O.; Delwart, E. Complex virome in feces from Amerindian children in isolated Amazonian villages. Nat. Commun. 2018, 9, 4270. [Google Scholar] [CrossRef]
- Laenen, L.; Vergote, V.; Calisher, C.; Klempa, B.; Klingström, J.; Kuhn, J.; Maes, P. Hantaviridae: Current classification and future perspectives. Viruses 2019, 11, 788. [Google Scholar] [CrossRef] [PubMed]
- Suttle, C.A. Marine viruses—Major players in the global ecosystem. Nat. Rev. Microbiol. 2007, 5, 801–812. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Wu, W.; Tian, X.; Hou, X.; Cui, X.; Xiao, Y.; Jiao, Q.; Zhou, P.; Liu, L.; Shi, W.; et al. A total infectome approach to understand the etiology of infectious disease in pigs. Microbiome 2022, 10, 73. [Google Scholar] [CrossRef] [PubMed]
- WoRMS. World Register of Marine Species—Perinereis Wilsoni Glasby & Hsieh, 2006. Available online: https://www.marinespecies.org/aphia.php?p=taxdetails&id=390131 (accessed on 18 March 2024).
- WoRMS. World Register of Marine Species—Perinereis Aibuhitensis (Grube, 1878). Available online: https://www.marinespecies.org/aphia.php?p=taxdetails&id=716016 (accessed on 18 March 2024).
- CRAB. Live Saltwater Bait and the Introduction of Non-Native Species into California. Available online: https://www.opc.ca.gov/webmaster/ftp/project_pages/AIS/AIS_LiveBait.pdf (accessed on 18 March 2024).


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Luo, J.; Zhang, F.; Zhou, C.; Meng, F.; Wang, G.; Qiu, L.; Shi, W.; Huang, J.; Dong, X. Meta-Transcriptomic Analysis Reveals Novel RNA Viruses in Polychaetes Perinereis. Vet. Sci. 2024, 11, 273. https://doi.org/10.3390/vetsci11060273
Luo J, Zhang F, Zhou C, Meng F, Wang G, Qiu L, Shi W, Huang J, Dong X. Meta-Transcriptomic Analysis Reveals Novel RNA Viruses in Polychaetes Perinereis. Veterinary Sciences. 2024; 11(6):273. https://doi.org/10.3390/vetsci11060273
Chicago/Turabian StyleLuo, Jingfei, Fan Zhang, Chengyan Zhou, Fanzeng Meng, Guohao Wang, Liang Qiu, Weifeng Shi, Jie Huang, and Xuan Dong. 2024. "Meta-Transcriptomic Analysis Reveals Novel RNA Viruses in Polychaetes Perinereis" Veterinary Sciences 11, no. 6: 273. https://doi.org/10.3390/vetsci11060273





