Varicocele in an Adult Ram: Histopathological Examination and Sperm Quality Evaluation
Abstract
1. Introduction
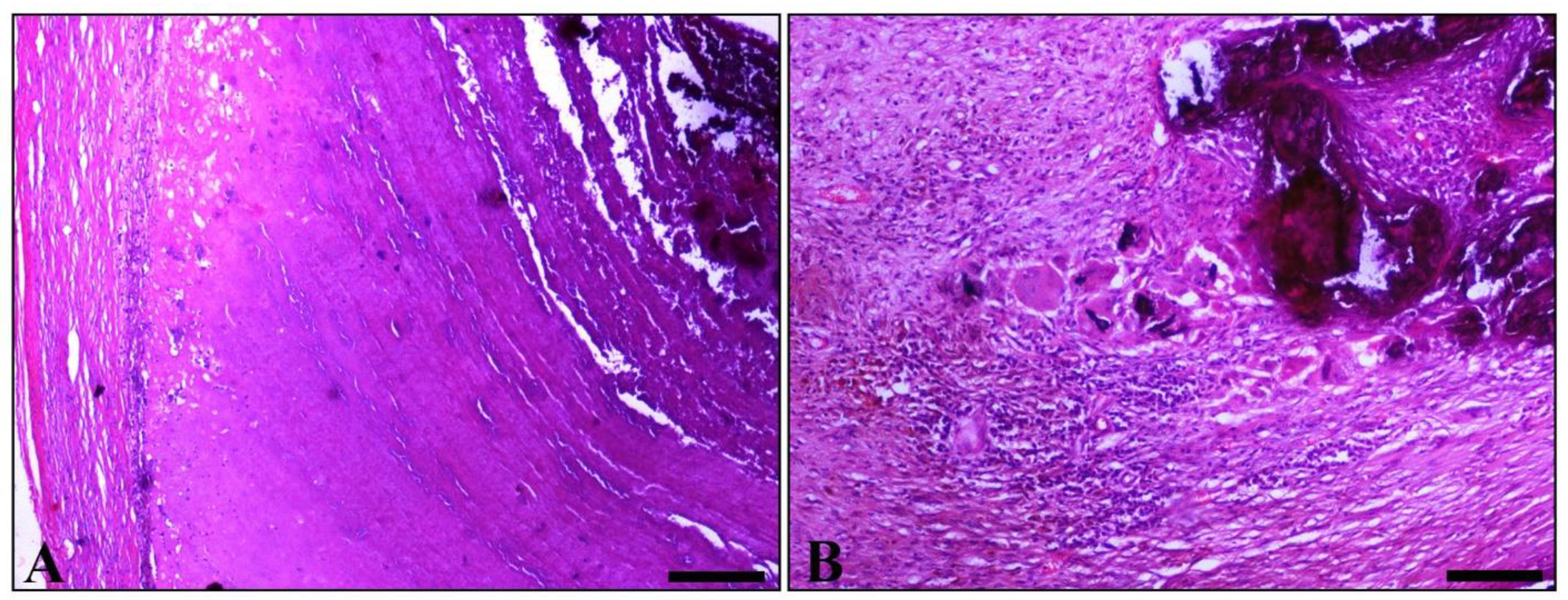
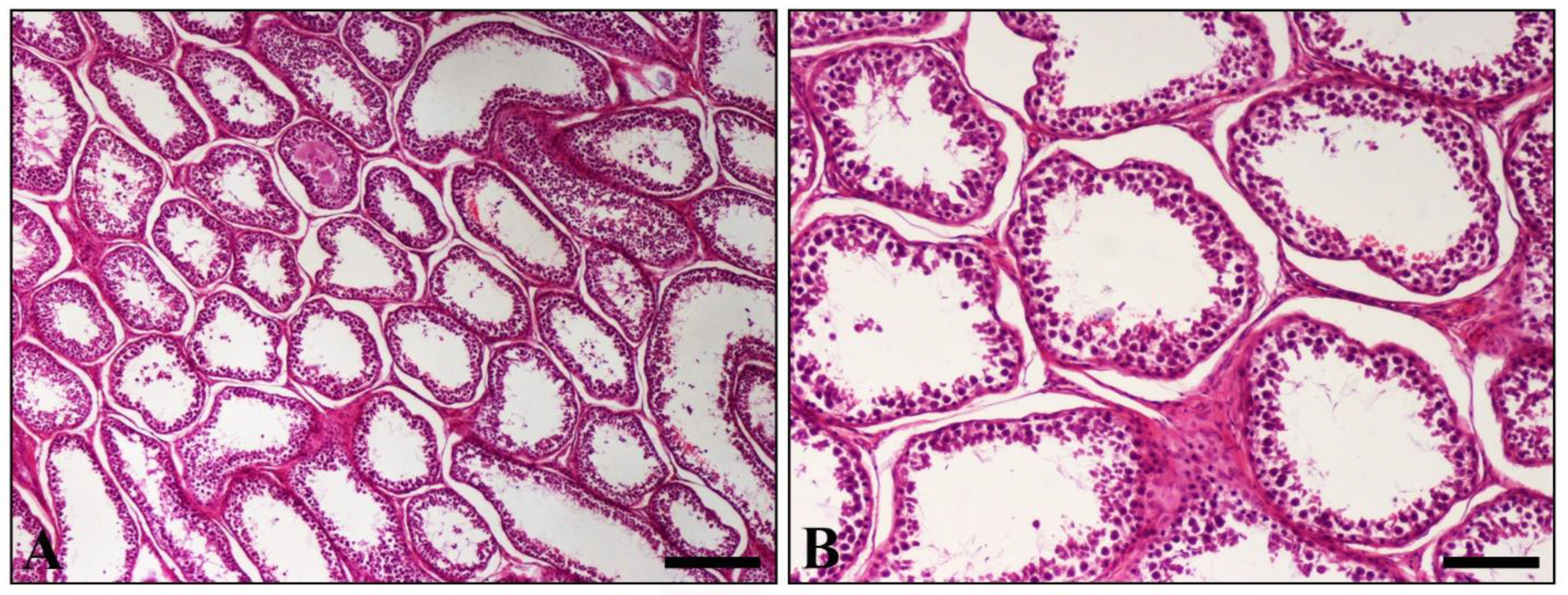
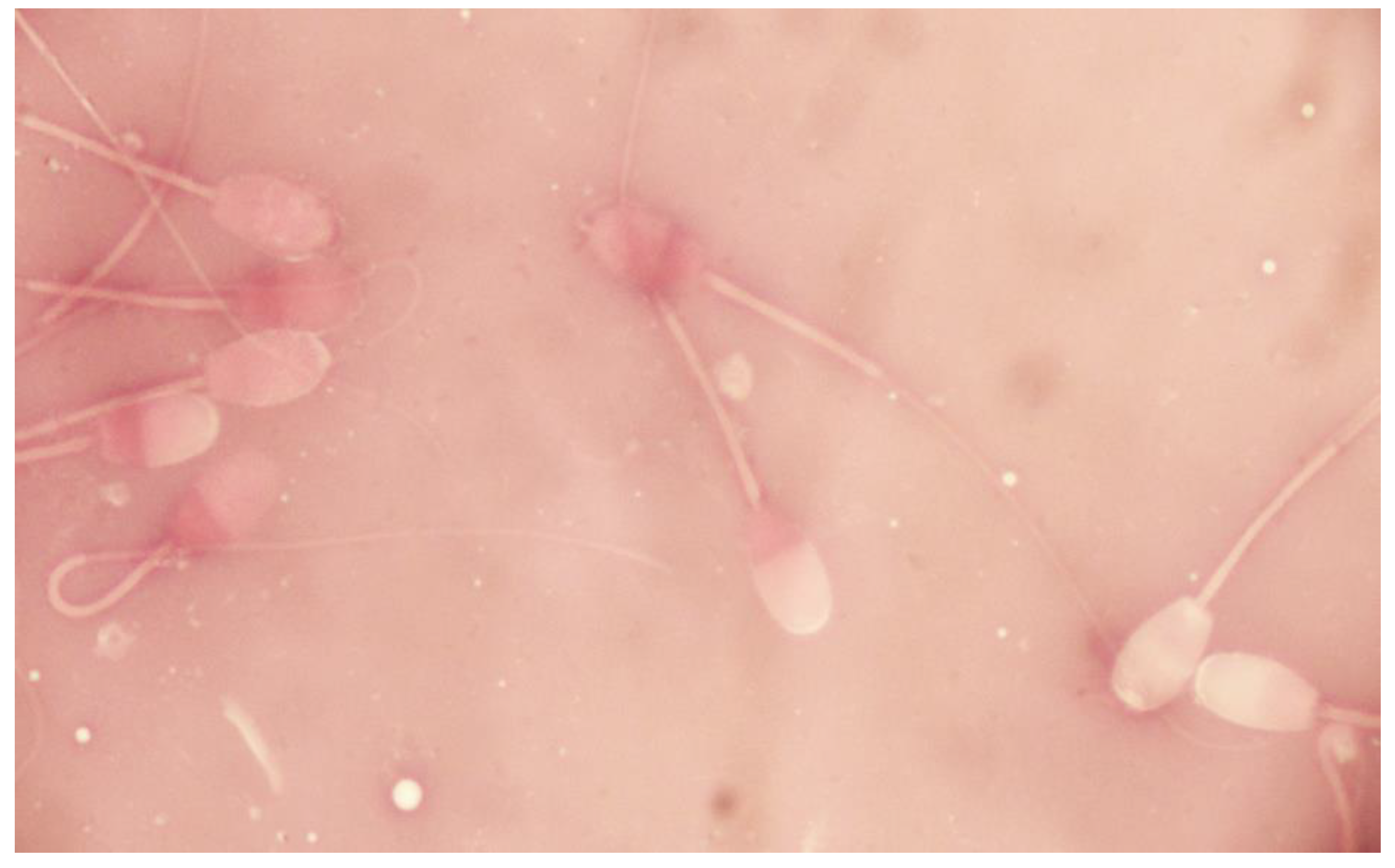
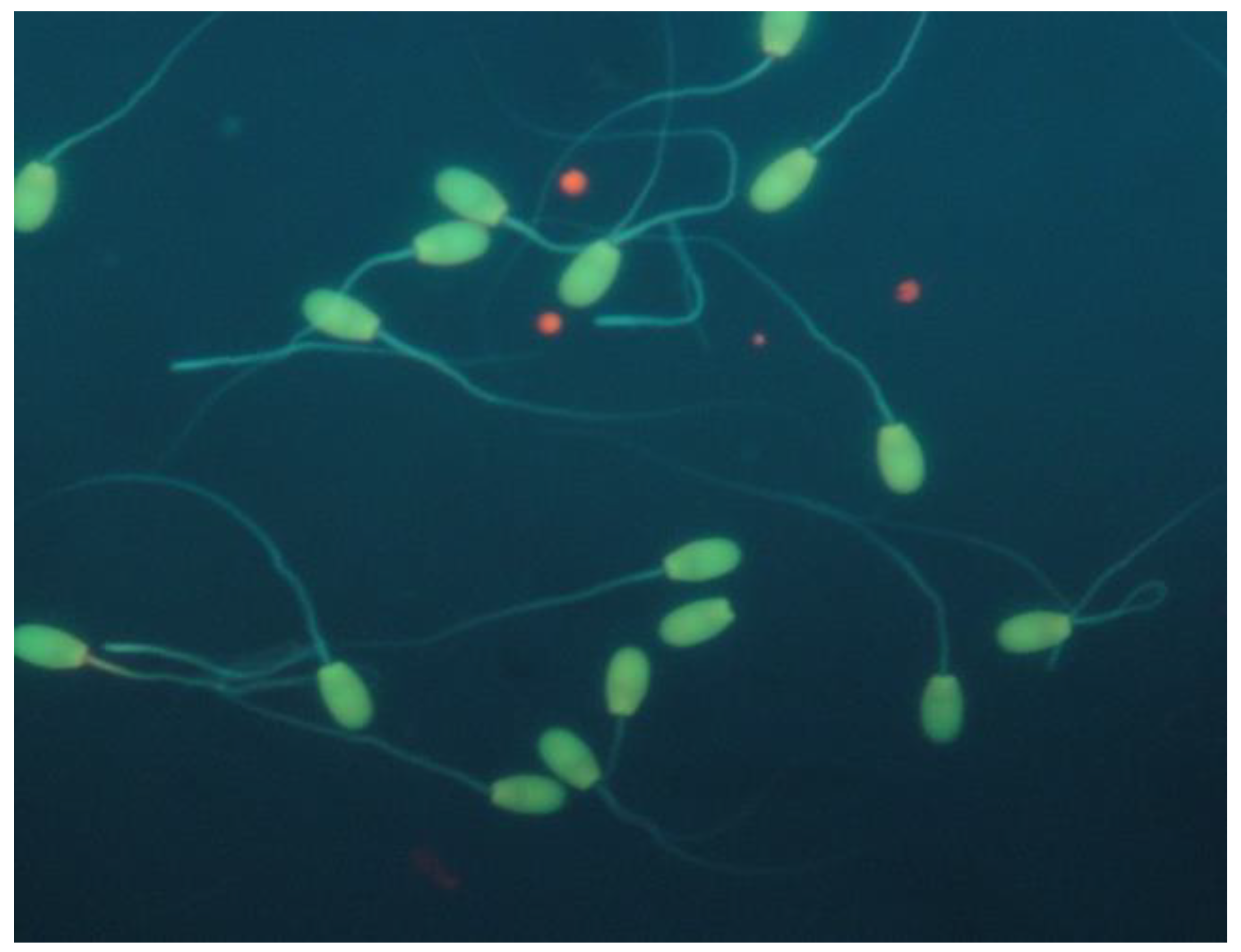
2. Case Presentation
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Punjani, N.; Wald, G.; Gaffney, C.D.; Goldstein, M.; Kashanian, J.A. Predictors of varicocele-associated pain and its impact on semen parameters following microsurgical repair. Andrologia 2021, 53, 8. [Google Scholar] [CrossRef] [PubMed]
- Kantartzi, P.D.; Goulis, C.D.; Goulis, G.D.; Papadimas, I. Male infertility and varicocele: Myths and reality. Hippokratia 2007, 11, 99–104. [Google Scholar] [PubMed]
- Khodamoradi, K.; Golan, R.; Dullea, A.; Ramasamy, R. Exosomes as Potential Biomarkers for Erectile Dysfunction, Varicocele, and Testicular Injury. Sex. Med. Rev. 2021. [Google Scholar] [CrossRef] [PubMed]
- Marmar, J.L. The evolution and refinements of varicocele surgery. Asian J. Androl. 2016, 18, 171–178. [Google Scholar] [CrossRef]
- Naughton, C.K.; Nangia, A.K.; Agarwal, A. Pathophysiology of varicoceles in male infertility. Hum. Reprod. Update 2001, 7, 473–481. [Google Scholar] [CrossRef]
- Koivisto, M.B.; Luvizotto, M.C.R.; Nogueira, G.P.; Vicente, W.R.R.; Costa, M.T.A. Testosterone concentration in a bovine Bos indicus with bilateral varicocele: Case report. Braz. J. Vet. Res. Anim. Sci. 2002, 39, 27–31. [Google Scholar] [CrossRef]
- Bousmaha, F.; Benchaib Khoudja, F. Comparative and pathological study of the testis and epididymis in rams. Bucks and bulls of Algeria. Asian J. Anim. Vet. Adv. 2012, 7, 950–959. [Google Scholar] [CrossRef][Green Version]
- Ezzi, A.; Ladds, P.W.; Hoffmann, D.; Foster, R.A.; Briggs, G.D. Pathology of varicocele in the ram. Aust. Vet. J. 1988, 65, 11–15. [Google Scholar] [CrossRef] [PubMed]
- Ott, R.S.; Heath, E.H.; Bane, A. Abnormal spermatozoa, testicular degeneration, and varicocele in a ram. Am. J. Vet. Res. 1982, 43, 241–245. [Google Scholar]
- Braun, W.F.; Cole, W.J. Unilateral scrotal hernia repair in a ram lamb. J. Am. Vet. Med. Assoc. 1985, 187, 500. [Google Scholar] [PubMed]
- Roberts, S.J. Scrotal hernia in rams. A case report. Cornell Vet. 1988, 78, 351–352. [Google Scholar] [PubMed]
- Pastuszak, A.W.; Wang, R. Varicocele and testicular function. Asian J. Androl. 2015, 17, 659–667. [Google Scholar] [CrossRef]
- Tejada, R.I.; Mitchell, J.C.; Norman, A.; Marik, J.J.; Friedman, S. A test for the practical evaluation of male fertility by acridine orange (AO) fluorescence. Fertil. Steril. 1984, 42, 87–91. [Google Scholar] [CrossRef]
- Janett, F.; Thun, R. Case report: Varicocele in a ram. Schweiz. Arch. Tierheilkd. 1995, 137, 386–388. [Google Scholar] [PubMed]
- Lacasta, D.; Ferrer, L.M.; Ramos, J.J.; Ochoa, P.G. Ultrasonographic diagnosis of unilateral scrotal hernia, varicocele and sperm granuloma in a ram. Acta Vet. Brno 2009, 78, 657–659. [Google Scholar] [CrossRef][Green Version]
- Blom, E. Pathological conditions in genital organs and sperm as a cause for the rejection of breeding bulls for import into and export from Denmark (an andrologic retrospective, 1958–1982). Nord. Vet. Med. 1983, 35, 105–130. [Google Scholar]
- Callaghan, M.J.; McAuliffe, P.; Rodgers, R.J.; Hernandez-Medrano, J.; Perry, V.E.A. Subacute ruminal acidosis reduces sperm quality in beef bulls. J. Anim. Sci. 2016, 94, 3215–3228. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Barth, A.D.; Bowman, P.A. The sequential appearance of sperm abnormalities after scrotal insulation or dexamethasone treatment in bulls. Can. Vet. J. 1994, 35, 93. [Google Scholar] [PubMed]

| Sperm Kinematics | Epididymal Spermatozoa | |
|---|---|---|
| Variable | LE | RE |
| Total (%) | 30.70 | 42.60 |
| Progr. (%) | 0.90 | 2.50 |
| Rapid (%) | 3.30 | 9.60 |
| Medium (%) | 2.60 | 5.50 |
| Slow (%) | 24.80 | 27.50 |
| VCL (μm/s) | 34.40 | 50.60 |
| VSL (μm/s) | 8.10 | 13.50 |
| VAP (μm/s) | 15.70 | 28.50 |
| ALH (μm) | 2.40 | 2.50 |
| BCF (Hz) | 10.60 | 13.20 |
| LIN (%) | 23.50 | 26.60 |
| STR (%) | 51.40 | 47.30 |
| WOB (%) | 45.70 | 56.30 |
| Sperm Morphology | Epididymal Spermatozoa | |
|---|---|---|
| Variable | LE | RE |
| Normal morphology (%) | 86.00 | 78.50 |
| Head (%) | 0.75 | 0.50 |
| Midpiece (%) | 0.75 | 3.00 |
| Tail (%) | 12.50 | 18.00 |
| Cytoplasmic droplets (%) | 0.00 | 0.00 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Basioura, A.; Brellou, G.D.; Tsakmakidis, I.; Vlemmas, I.; Boscos, C.M.; Giadinis, N. Varicocele in an Adult Ram: Histopathological Examination and Sperm Quality Evaluation. Vet. Sci. 2022, 9, 86. https://doi.org/10.3390/vetsci9020086
Basioura A, Brellou GD, Tsakmakidis I, Vlemmas I, Boscos CM, Giadinis N. Varicocele in an Adult Ram: Histopathological Examination and Sperm Quality Evaluation. Veterinary Sciences. 2022; 9(2):86. https://doi.org/10.3390/vetsci9020086
Chicago/Turabian StyleBasioura, Athina, Georgia D. Brellou, Ioannis Tsakmakidis, Ioannis Vlemmas, Constantin M. Boscos, and Nektarios Giadinis. 2022. "Varicocele in an Adult Ram: Histopathological Examination and Sperm Quality Evaluation" Veterinary Sciences 9, no. 2: 86. https://doi.org/10.3390/vetsci9020086
APA StyleBasioura, A., Brellou, G. D., Tsakmakidis, I., Vlemmas, I., Boscos, C. M., & Giadinis, N. (2022). Varicocele in an Adult Ram: Histopathological Examination and Sperm Quality Evaluation. Veterinary Sciences, 9(2), 86. https://doi.org/10.3390/vetsci9020086







