Abstract
Members of the fungal order Diaporthales are sac fungi that include plant pathogens (the notorious chestnut blight fungus), as well as saprobes and endophytes, and are capable of colonizing a wide variety of substrates in different ecosystems, habitats, and hosts worldwide. However, many Diaporthales species remain unidentified, and various inconsistencies within its taxonomic category remain to be resolved. Here, we aimed to identify and classify new species of Diaporthales by using combined morphological and molecular characterization and coupling this information to expand our current phylogenetic understanding of this order. Fungal samples were obtained from dead branches and diseasedleaves of Camellia (Theaceae) and Castanopsis (Fagaceae) in Fujian Province, China. Based on morphological characteristics and molecular phylogenetic analyses derived from the combined nucleotide sequences of loci of the internal transcribed spacer regions with the intervening 5.8S nrRNA gene (ITS), the 28S large subunit of nuclear ribosomal RNA gene (LSU), the translation elongation factor 1-α gene (tef1), the partial beta-tubulin gene (tub2), and partial RNA polymerase II second-largest subunit gene (rpb2), three new species of Diaporthales were identified and characterized. They are as follows: Chrysofolia camelliae sp. nov., Dendrostoma castanopsidis sp. nov., and Pseudoplagiostoma wuyishanense sp. nov. They are described and illustrated. This study extends our understanding of species diversity within the Diaporthales.
1. Introduction
The fungal order Diaporthales consists of 32 families [1,2], and includes several important plant pathogens, e.g., the chestnut blight fungus, Cryphonectria parasitica, the soybean stem canker-causing fungus, Diaporthe phaseolorum, and the peach canker and citrus fruit stem-end rot fungi, Diaporthe amygdali (Phomopsis amygdali) and Diaporthe citri, respectively [3]. A number of asexual diaporthalean plant pathogens have also been described including the causative agent of grape bitter rot, Greeneria uvicola, and the dogwood anthracnose fungus, Discula destructiva. Diaporthales are sac fungi, characterized by ascomata with short or long necks, usually having a central column structure, immersed in host tissue, and unitunicate asci, with a refractive ring at maturity [4,5]. Members of the Diaporthales are widely distributed geographically and have many hosts, inhabiting or colonizing a range of plant, animal, soil, and water substrates and habitats [6,7,8]. Plant-associated members are often endophytic, inhabiting plant tissues, or parasitic, causing disease in plants, with others possibly acting as saprobes [9]. Endophytic associations have been reported between Diaporthe endophytica (Diaporthaceae)and leaves of Schinus terebinthifolius in Thailand [10]. Phaeoappendicospora thailandensis (Phaeoappendicosporaceae) was isolated from dead branch of Quercus sp. in Thailand [6], and Pyrispora castaneae (Pyrisporaceae) was identifiedas a pathogen or saprobe from Castanea mollissima leaves in China [8]. Additional plant pathogens within this order include Cytospora jilongensis (Cytosporaceae),which causes stem cankers on Prunus davidiana, [11] and Aurifilum cerciana (Cryphonectriaceae),responsible for foliage blight on Terminalia mantaly [12].
The genus Chrysofolia Crous & M.J. Wingf. (Cryphonectriaceae) was erected based on the type species C. colombiana by Crous et al. [13]. Chrysofolia is mainly characterized by separate-to-aggregated pycnidial conidiomata with globose pycnidia [13,14], conidiophores reduced to conidiogenous cells [15], hyaline, smooth, aggregated, and cylindrical-to-ampulliform conidiogenous cells [13,16], and possessing conidia with hyaline, smooth, guttulate, straight-to-allantoid base with flattened hilum. Species within Chrysofolia have been reported to inhabit or colonize plant tissues as endophytes and pathogens [16]. Currently, Chrysofolia includes six species: C. barringtoniae [16], C. colombiana [13], C. coriariae [15], C. fructicola [17], C. galloides [18], and C. kunmingensis [14].
Dendrostoma X.L. Fan & C.M. Tian (Erythrogloeaceae) was established with the type species Dendrostoma mali, with D. osmanthi and D. quercinum subsequently described by Fan et al. [1]. The asexual morph of Dendrostoma is characterized by clavate asci with fusoid-to-cylindrical, bicellular, multiguttulate ascospores [1,19]. Based on morphological characteristics and multi-locus (ITS–LSU–rpb2–tef1) data, Jiang et al. identified ten new species of Dendrostoma: D. aurorae, D. castaneae, D. castaneicola, D. chinense, D. dispersum, D. parasiticum, D. qinlingense, D. quercus, D. shaanxiense, and D. shandongense in China [20].
Pseudoplagiostoma eucalypti is the type species of Pseudoplagiostoma Cheew., M.J. Wingf. & Crous (Pseudoplagiostomataceae) as introduced by Cheewangkoon et al. [21]. Currently, Pseudoplagiostomataceae comprises the single genus of Pseudoplagiostoma. Species of Pseudoplagiostoma include plant pathogens, endophytes, and saprophytes [21,22,23]. It is worth noting that most of the species Pseudoplagiostoma have been found in Asia, indicating the high likelihood of species in this area [23].
Here we identify three new species within the Diaporthales, with Chrysofolia camelliae sp. nov. in Cryphonectriaceae, Dendrostoma castanopsidis sp. nov. in Erythrogloeaceae, and Pseudoplagiostoma wuyishanense sp. nov. in the Pseudoplagiostomataceae of this order. Characterization included a combined morphological and molecular phylogenetic analyses, with the latter including data generated from multi-locus nucleotide sequencing that included the internal transcribed spacer regions with the intervening 5.8S nrRNA gene (ITS), the 28S large subunit of nuclear ribosomal RNA gene (LSU), the translation elongation factor 1-α gene (tef1), the partial beta-tubulin gene (tub2), and partial RNA polymerase II second-largest subunit gene (rpb2). These data establish new members of the Diaporthales, helping improve placement of various genera within this order.
2. Materials and Methods
2.1. Sample Collection, Isolation, and Morphological Observations
Plant samples of Camellia, Castanopsis fargesii, and an unidentified tree were collected from Sanming, Quanzhou, and Wuyishan City of Fujian Province, China, in September 2022. Samples were treated as described in Fu et al. [24]. Fungal isolates were single colonies purified by culturing on potato dextrose agar (PDA) in a light incubator at 25 °C and under a 12 h light/dark regime. To promote sporulation, some strains were specially removed to synthetic low nutrient agar (SNA) containing sterilized pine needles [15]. Dried holotype specimens were deposited in the Herbarium Mycologicum Academiae Sinicae, Institute of Microbiology, Chinese Academy of Sciences, Beijing, China (HMAS). Ex-type living cultures were conserved in the China General Microbiological Culture Collection Center (CGMCC). Colony morphologies were captured by camera (Canon EOS 6D MarkII, Tokyo, Japan) at 7 and 14 d after inoculation [23]. Micromorphological characteristics of strains were recorded and observed and photographed using a stereo microscope (Nikon SMZ745, Tokyo, Japan) and biological microscope (Ni-U, Tokyo, Japan) with a digital camera (Olympus, Tokyo, Japan). Conidia of some fungal isolates were observed by a scanning electron microscope (Hitachi TM3030 PLUS, Tokyo, Japan) with measurements of micromorphological structures, carried out using Digimizer 5.4.4 software (https://www.digimizer.com, accessed on 2 April 2024) [25]. All fungal strains were stored in 10% sterilized glycerin and sterile water at 4 °C in 2.0 mL tubes. Taxonomic information of the new taxa were registered in MycoBank (http://www.mycobank.org, accessed on 15 April 2024).
2.2. DNA Extraction, PCR Amplification, and Sequencing
Fungal genomic DNA was extracted from growing mycelia using the Fungal DNA Mini Kit (OMEGA-D3390, Feiyang Biological Engineering Co., Ltd., Guangzhou, China) following the manufacturer’s instructions. A total of five gene loci were examined by polymerase chain reaction (PCR) amplification of nucleotide sequences that included regions of ITS, LSU, tef1, tub2, and rpb2 genes. The PCR thermal cycle program and primer pairs are listed in Table 1. The PCR reaction volume was 25 µL, containing 12.5 μL of 2 × Rapid Taq Master Mix (Vazyme, Nanjing, China), 1 μL of each forward and reverse primer (10 μM) (Sangon, Shanghai, China), 1 μL of template genomic DNA, and 9.5 µL of double-distilled water (ddH2O), and amplification was performed using a Bio-Rad Thermocycler (Hercules, CA, USA). The integrity and sizes of all the PCR products were checked on 1% agarose gel electrophoresis and products were sequenced using a commercial company (Tsingke Co., Ltd., Fuzhou, China). Both forward and reverse sequences of PCR products for each loci were obtained and processed by MEGA 7.0.20 software [26]. New sequences generated in this study have been deposited in GenBank (https://www.ncbi.nlm.nih.gov, accessed on 15 April 2024, Table 2, Table 3 and Table 4).

Table 1.
The primer pairs, sequences, and programs in this study.

Table 2.
Species names, voucher or culture codes, hosts or substrate, locations, and corresponding GenBank accession numbers of DNA sequences used in the molecular phylogenetic analyses of Chrysofolia and allied genera.

Table 3.
Species names, voucher or culture codes, hosts or substrate, locations, and corresponding GenBank accession numbers of DNA sequences used in the molecular phylogenetic analyses of Dendrostoma.

Table 4.
Species names, voucher or culture codes, hosts or substrate, locations, and corresponding GenBank accession numbers of DNA sequences used in the molecular phylogenetic analyses of Pseudoplagiostoma.
2.3. Phylogenetic Analyses
NCBI-Blast searches using sequence data generated from fungal samples were used to identify and download data from GenBank for multi-locus phylogenetic analyses (Table 2, Table 3 and Table 4). Gene sequences were initially aligned with MAFFTv.7 and optimized manually with MEGA 7.0.20 software and trimAL v1.2 (http://trimal.cgenomics.org, accessed on 3 April 2024) [14,26,33]. Multi-locus phylogenetic analyses of the concatenated aligned dataset were obtained by maximum likelihood (ML) and Bayesian inference (BI) methods. They were inferred using IQtree 1.6.8 [34] and MrBayes 3.2.6 [35] with Phylosuite software v1.2.3 (https://dongzhang0725.github.io/, accessed on 3 April 2024) [36]. IQtree was run under an edge-linked partition model for 5000 ultrafast bootstraps [37]. For Bayesian inference analyses, PartitionFinder2 was used to select the best-fit partition model [38]. The best evolutionary models were used (2 parallel runs, 2,000,000 generations, sample frequency = 100), in which the first 1/4 of trees were discarded as burn-in and the remaining 3/4 of trees were used to calculate Bayesian Posterior Probabilities (BYPP). The phylogenetic trees were visualized using FigTree1.4.4 (http://tree.bio.ed.ac.uk/software/figtree, accessed on 3 April 2024) and embellished with Adobe Illustrator CS 6.0 (Adobe Systems Inc., San Jose, CA, USA).
3. Results
3.1. Phylogenetic Analyses
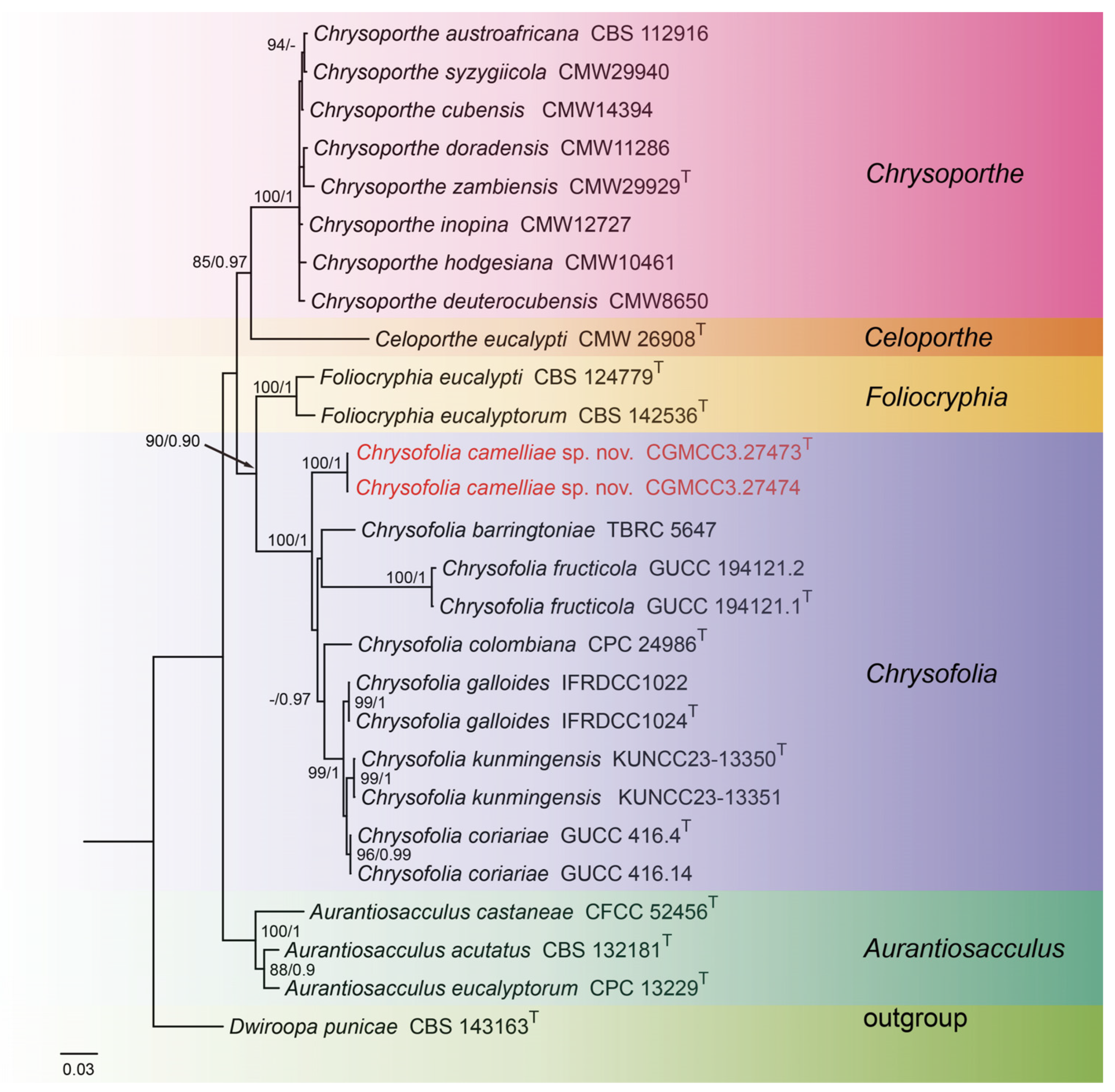
For construction of the phylogenetic tree of isolates matching Chrysofolia, the concatenated sequence dataset of ITS, LSU, tef1, and tub2 was used which included 27 taxa with Dwiroopa punicae (CBS 143163) as the outgroup (Figure 1). The aligned four-locus datasets had an aligned length of 2633 total characters (i.e., ITS: 1–674, LSU: 675–1517, tef1: 1518–2032, tub2: 2033–2633) including gaps, of which 1906 characters were constant, 253 were variable and parsimony-uninformative, and 474 were parsimony-informative. The best model for the dataset was estimated by PartitionFinder2, and selected in the Bayesian analysis were SYM + I + G for ITS (Lset nst = 6, rates = invgamma), GTR + I + G for LSU (Lset nst = 6, rates = invgamma), HKY + G for tef1 (Lset nst = 2, rates = gamma), and GTR + G for tub2 (Lset nst = 6, rates = gamma). Bayesian analyses resulted in an average standard deviation of split frequencies = 0.004399. The topology of the ML tree was similar to Bayesian analyses; thus, the Bayesian tree is shown. Maximum-likelihood bootstrap support (ML-BS) values (≥80%) and Bayesian posterior probabilities (BI-PP) values (≥0.90) are provided as the first and second positions, respectively, above the nodes.
Figure 1.
Phylogenetic tree of Chrysofolia and allied genera inferred from Bayesian inference analyses based on a combined ITS, LSU, tef1, and tub2 sequence dataset, with Dwiroopa punicae (CBS 143163) as outgroup. The ML and BI bootstrap support values above 80% and 0.90 BYPP are indicated at the nodes. Strains marked with “T” are ex-type, ex-epitype, or ex-neotype. The strains from this study are indicated in red.
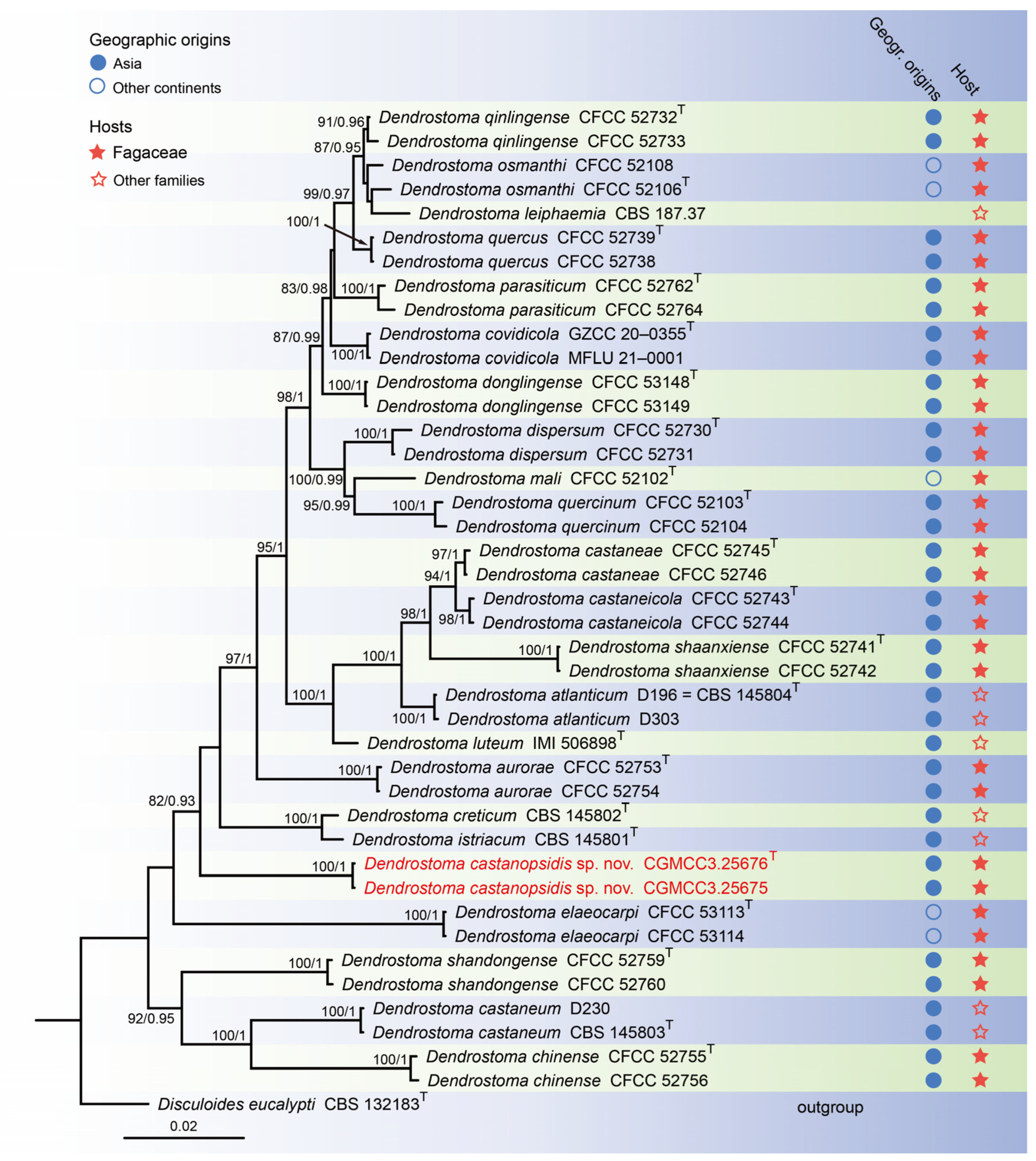
For construction of the phylogenetic tree for analysis of the Dendrostoma-matching isolates, the concatenated sequence dataset of ITS, LSU, rpb2, and tef1 was analyzed using sequences from 42 taxa with Disculoides eucalypti (CBS 132183) as an outgroup (Figure 2). The aligned four-locus datasets had an aligned length of 2804 total characters (i.e., ITS: 1–503, LSU: 504–1344, rpb2: 1345–2419, tef1: 2420–2804) including gaps, of which 2208 characters were constant, 137 were variable and parsimony-uninformative, and 459 were parsimony-informative. The best model for the dataset was estimated by PartitionFinder2, and selected in the Bayesian analysis were SYM + I + G for ITS (Lset nst = 6, rates = invgamma), GTR + I + G for LSU and rpb2 (Lset nst = 6, rates = invgamma), and GTR + G for tef1 (Lset nst = 6, rates = gamma). Bayesian analyses resulted in an average standard deviation of split frequencies = 0.002505. The topology of the ML tree was similar to Bayesian analyses; thus, the Bayesian tree is displayed. Maximum-likelihood bootstrap support (ML-BS) values (≥80%) and Bayesian posterior probabilities (BI-PP) values (≥0.90) are provided as the first and second positions, respectively, above the nodes.
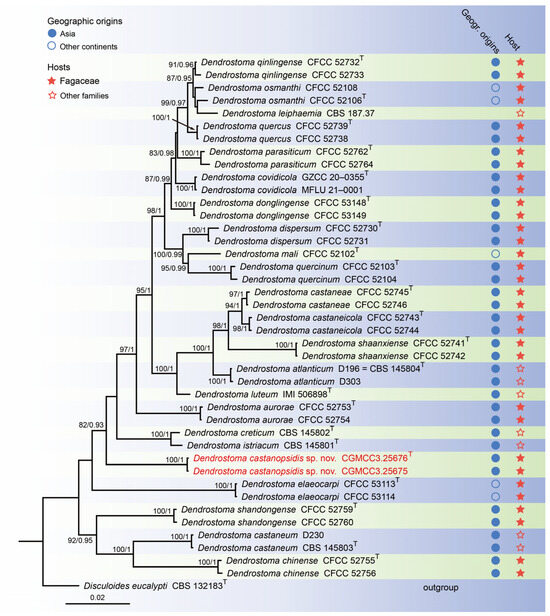
Figure 2.
Phylogenetic tree of Dendrostoma inferred from Bayesian inference analyses based on a combined ITS, LSU, rpb2, and tef1 sequence dataset, with Disculoides eucalypti (CBS 132183) as outgroup. The ML and BI bootstrap support values above 80% and 0.90 BYPP are shown at the nodes. Strains marked with “T” are ex-type, ex-epitype, or ex-neotype. Thestrains from this study are indicated in red.
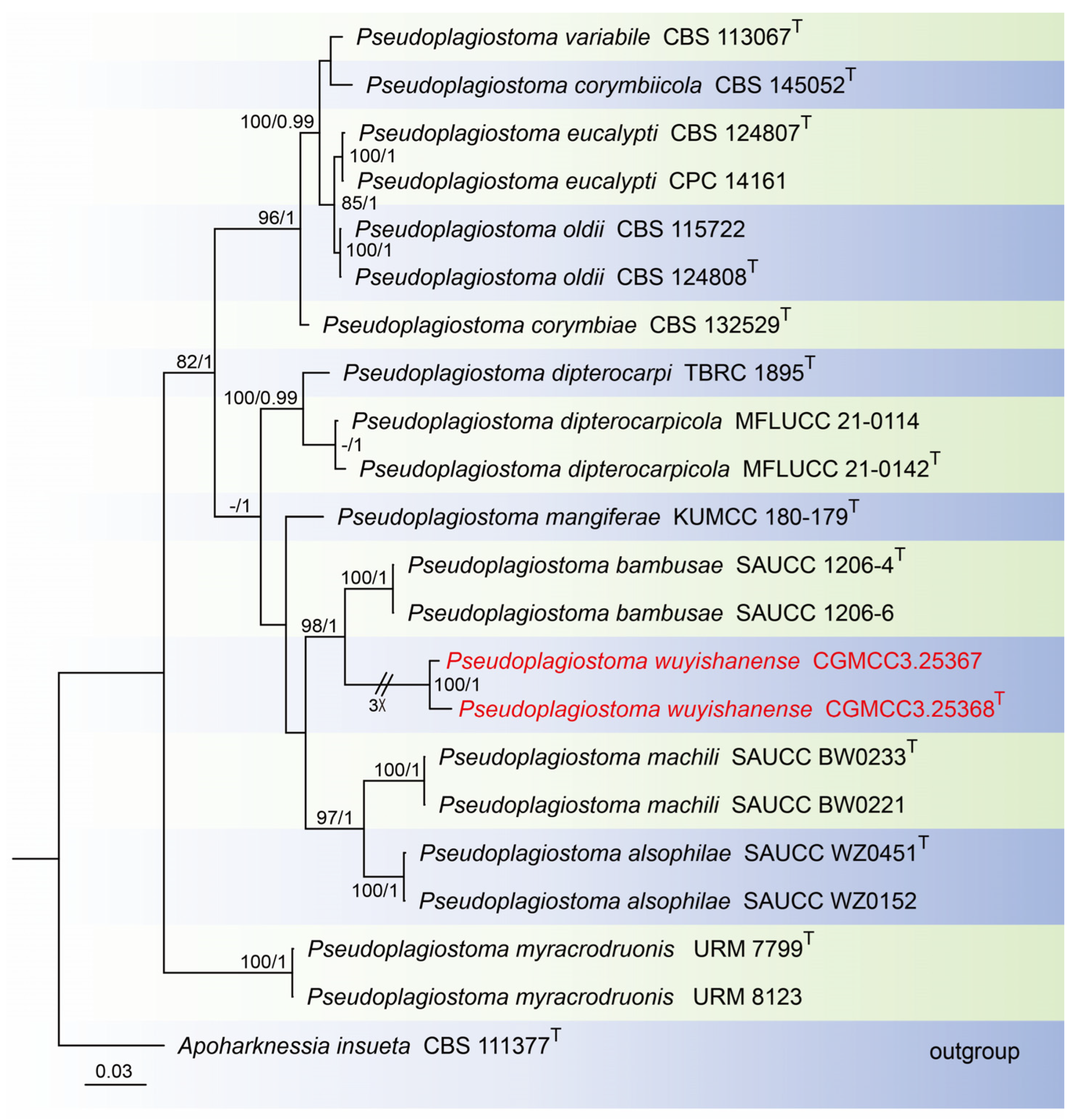
For construction of the phylogenetic tree for analysis of the Pseudoplagiostoma-matching isolates, the concatenated sequence dataset of ITS, LSU, rpb2, tef1, and tub2 was analyzed using 22 taxa with Apoharknessia insueta (CBS 111377) as the outgroup (Figure 3). The aligned five-locus datasets had an aligned length of 3060 total characters (i.e., ITS: 1–649, LSU: 650–1505, rpb2: 1506–2081, tef1: 2082–2570, tub2: 2571–3060) including gaps, of which 1984 characters were constant, 169 were variable and parsimony-uninformative, and 907 were parsimony-informative. The best model for the dataset was estimated by PartitionFinder2, and selected in the Bayesian analysis were GTR + I + G for ITS, LSU and tef1 (Lset nst = 6, rates = invgamma), and GTR + G for rpb2 and tub2 (Lset nst = 6, rates = gamma). Bayesian analyses resulted in an average standard deviation of split frequencies = 0.000884. The Maximum-likelihood resulted in a similar topology to Bayesian inference; thus, the Bayesian tree is provided. Maximum-likelihood bootstrap support (ML-BS) values (≥80%) and Bayesian posterior probabilities (BI-PP) values (≥0.90) are shown as the first and second positions, respectively, above the nodes.
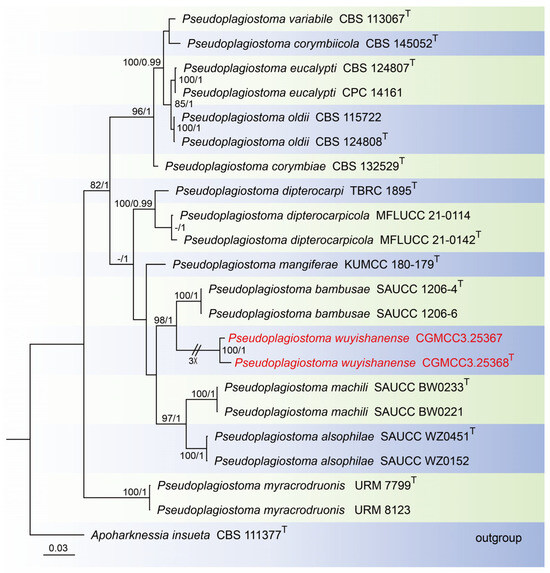
Figure 3.
Phylogenetic tree of Pseudoplagiostoma inferred from Bayesian inference analyses based on a combined ITS, LSU, rpb2, tef1, and tub2 sequence dataset, with Apoharknessia insueta (CBS 111377) as outgroup. The ML and BI bootstrap support values above 80% and 0.90 BYPP are shown at the nodes. Strains marked with “T” are ex-type, ex-epitype, or ex-neotype. The strains from this study are indicated in red.
3.2. Taxonomy
3.2.1. Chrysofolia camelliae T.C. Mu and J.Z. Qiu, sp. nov., Figure 4
MycoBank Number: MB853510
Etymology: the epithet “camelliae” refers to the host genus, Camellia.
Holotype: China: Fujian Province, Sanming City, 26°39′28″ N, 117°51′16″ E, on diseased leaves of Camellia sp. (Theaceae), 9 September 2022, holotype HMAS 352951; ex-holotype living culture CGMCC3.27473.
Description: Leaf spots irregular and fawn or umber. Asexual morphs developed on PDA. Conidiomata pycnidial, separated to aggregated, globose to subglobose, orange conidial droplets exuded from ostioles. Conidiophores reduced to conidiogenous cells. Conidiophores cells hyaline, smooth, cylindrical to ampulliform, 4.0–6.0 μm × 1.0–4.0 μm. Conidia hyaline, smooth, aseptate, subfusoid, elongate ellipsoidal, 5.0–9.0 μm × 2.0–4.0 μm, mean = 8.0 μm × 3.0 μm, L/W ratio = 2.0, n = 30. Sexual morph not observed.
Culture characteristics: Colonies on PDA were flat with irregular margin, aerial mycelium orange in the center and white at the edge. Surface white initially, becoming orange with age, reverse golden-orange. Growth on PDA attained 57.3–64.7 mm in diameter after 1 week at 25 °C, growth rate 8.2–9.2 mm/day. Growth on PDA attained 78.7–81.3 mm in diameter after 2 weeks at 25 °C, growth rate 5.6–5.8 mm/day.
Material examined: China: Fujian Province, Sanming City, 26°39′28″ N, 117°51′16″ E, on diseased leaves of Camellia sp. (Theaceae), 9 September 2022, paratype HMAS 352952; ex-paratype living culture CGMCC3.27474.

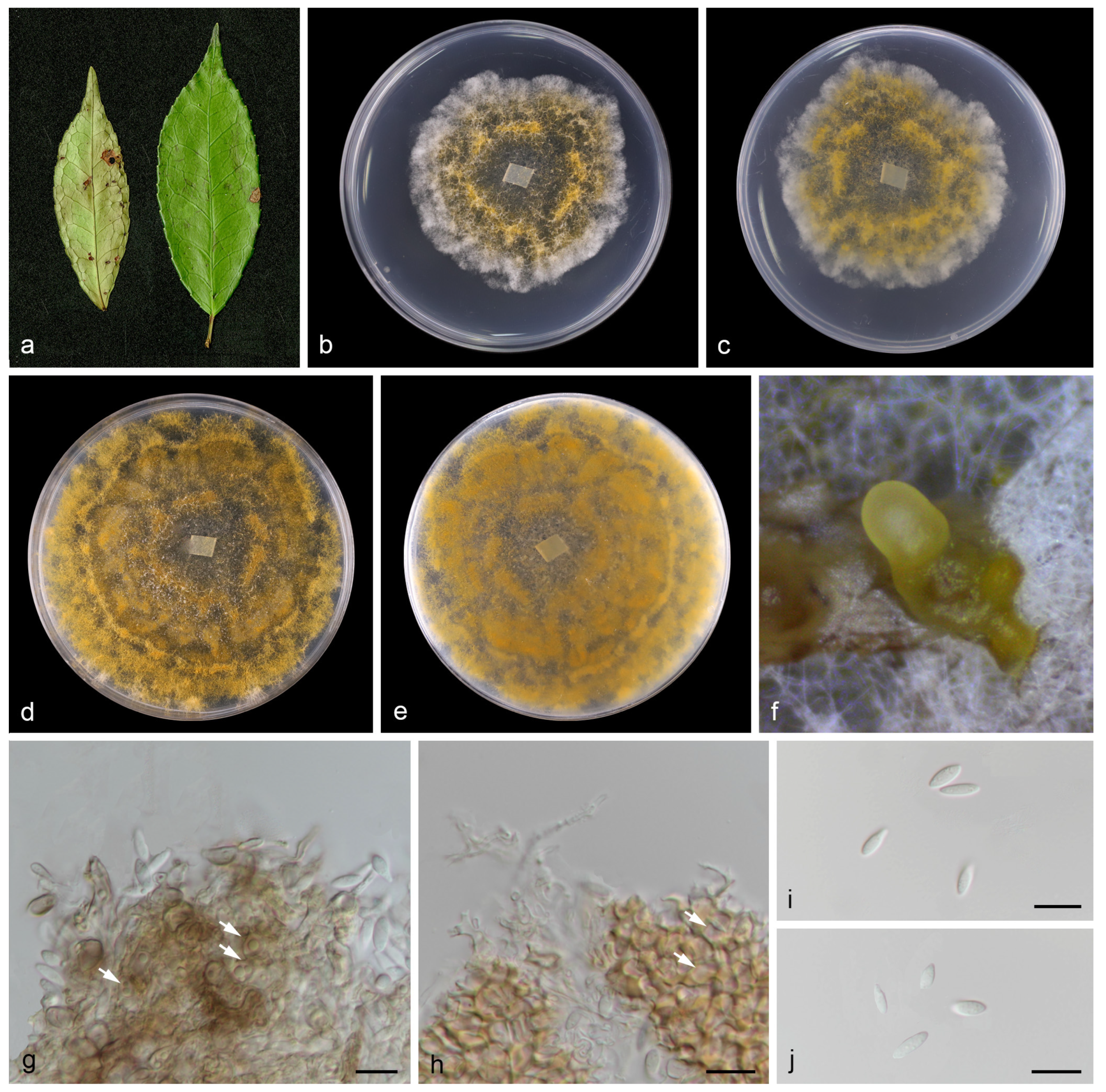
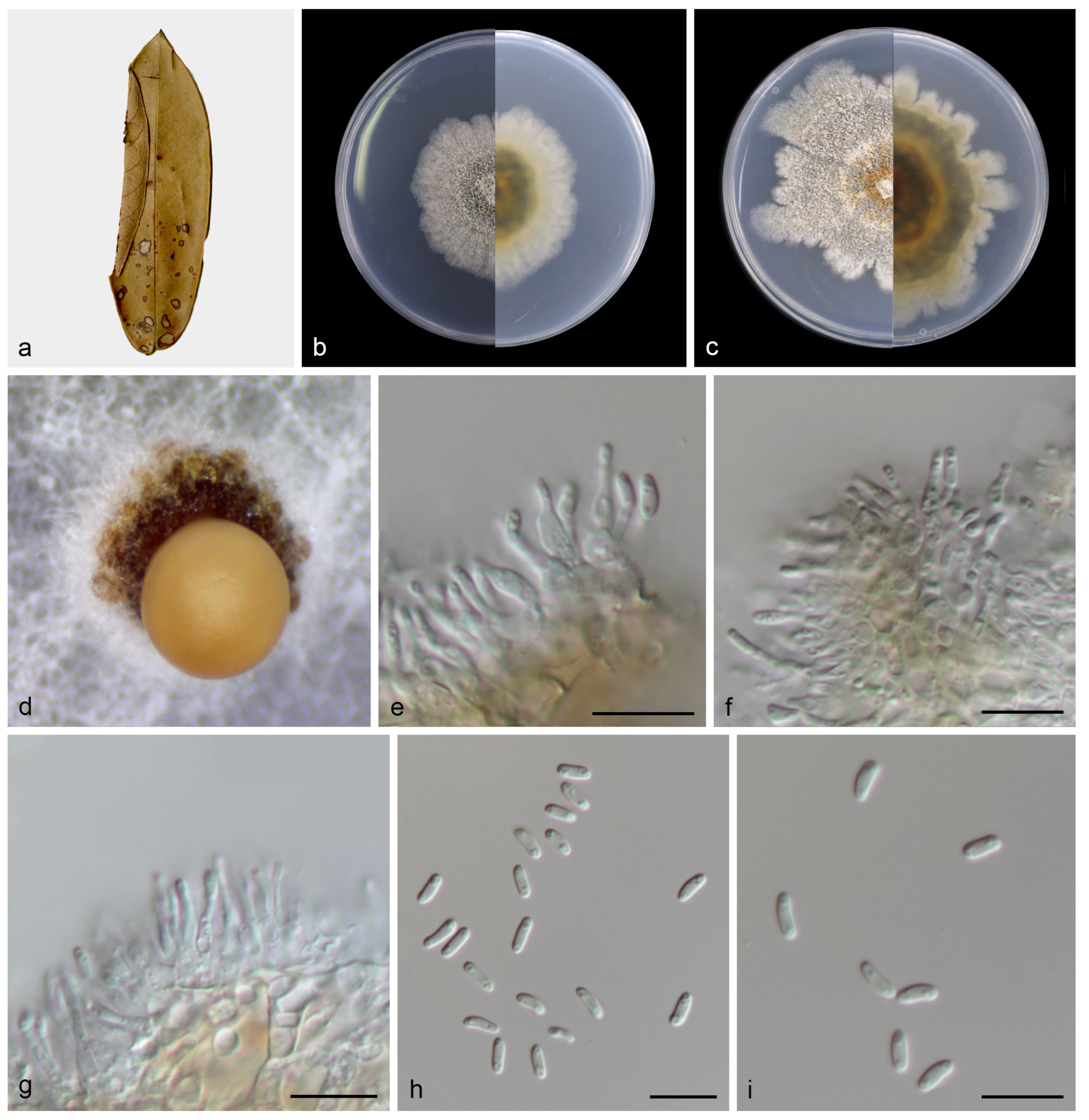
Figure 4.
Chrysofolia camelliae (holotype HMAS 352951). (a) Diseased leaves of Camellia sp.; (b,c) surface and reverse sides of colony after 7 days on PDA (d,e) and 14 days; (f) mass of conidia; (g,h) conidiogenous cells and conidia; (i,j) conidia. Scale bars: (g–j) 10 µm.
Figure 4.
Chrysofolia camelliae (holotype HMAS 352951). (a) Diseased leaves of Camellia sp.; (b,c) surface and reverse sides of colony after 7 days on PDA (d,e) and 14 days; (f) mass of conidia; (g,h) conidiogenous cells and conidia; (i,j) conidia. Scale bars: (g–j) 10 µm.
Notes: In the current study, to the best of our knowledge, this is the first instance of Chrysofolia on leaves of Camellia sp. in China, and Chrysofolia camelliae sp. nov. is described. Based on the multi-gene phylogenetic analyses of ITS, LSU, tef1, and tub2, the new species is strongly supported (100% ML/1 PP, Figure 4) with C. barringtoniae, C. colombiana, C. coriariae, C. fructicola, C. galloides, and C. kunmingensis. BLASTn analysis of C. camelliae (CGMCC3.27473) and C. barringtoniae (TBRC 5647) revealed 28 bp (28/608 bp, 4.6%) nucleotide differences in ITS. BLASTn analysis of C. camelliae (CGMCC3.27473) and C. colombiana (CPC 24986) revealed 84 bp (84/461 bp, 18.3%) nucleotide differences in tef1. BLASTn analysis of C. camelliae (CGMCC3.27473) and C. coriariae (GUCC 416.4) revealed 81 bp (81/320 bp, 25.3%) nucleotide differences in tef1 and 55 bp (55/527 bp, 10.4%) nucleotide differences in tub2. BLASTn analysis of C. camelliae (CGMCC3.27473) and C. fructicola (GUCC 194121.1) revealed 60 bp (60/609 bp, 9.9%) nucleotide differences in ITS and 118 bp (118/492 bp, 24.0%) nucleotide differences in tef1. BLASTn analysis of C. camelliae (CGMCC3.27473) and C. galloides (IFRDCC1024) revealed 85 bp (85/466 bp, 18.2%) nucleotide differences in tef1 and 54 bp (54/523 bp, 10.3%) nucleotide differences in tub2. BLASTn analysis of C. camelliae (CGMCC3.27473) and C. kunmingensis (KUNCC23-13350) revealed 51 bp (51/434 bp, 11.8%) nucleotide differences in tub2. Morphologically, the conidia of C. camelliae are large than C. barringtoniae (5.0–9.0 μm × 2.0–4.0 μm vs. 3.0–7.5 μm × 2.0–3.0 μm). Therefore, we describe this fungus as a new species.
3.2.2. Dendrostoma castanopsidis T.C. Mu and J.Z. Qiu, sp. nov., Figure 5
MycoBank Number: MB853511
Etymology: the epithet “castanopsidis” refers to the host genus, Castanopsis.
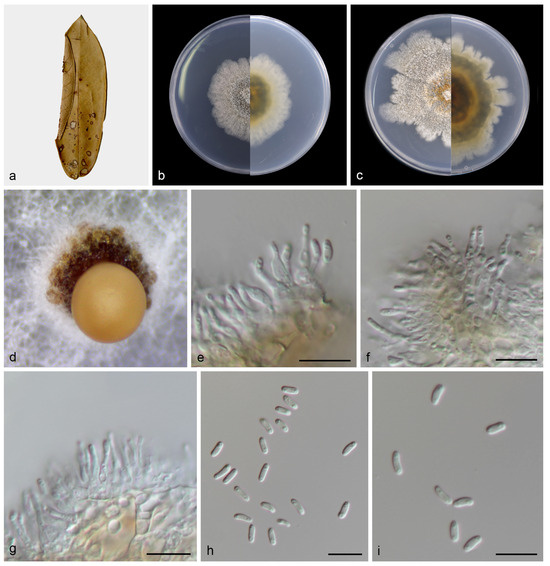
Figure 5.
Dendrostoma castanopsidis (holotype HMAS 352711). (a) Diseased leaves of Castanopsis fargesii; (b) surface and reverse sides of colony after 7 days on PDA (c) and 14 days; (d) mass of conidia; (e–g) conidiogenous cells with conidia; (h,i) conidia. Scale bars: (e–i) 10 µm.
Figure 5.
Dendrostoma castanopsidis (holotype HMAS 352711). (a) Diseased leaves of Castanopsis fargesii; (b) surface and reverse sides of colony after 7 days on PDA (c) and 14 days; (d) mass of conidia; (e–g) conidiogenous cells with conidia; (h,i) conidia. Scale bars: (e–i) 10 µm.
Holotype: China: Fujian Province, Quanzhou City, 24°54′38″ N, 117°13′46″ E, on diseased leaves of Castanopsis fargesii (Fagaceae), 17 September 2022, holotype HMAS 352711; ex-holotype living culture CGMCC3.25676.
Description: Leaf spots subcircular and irregular, gray to brown. Asexual morphs developed on PDA. Conidiomata pycnidial, separated, conical, orange conidial droplets exuded from ostioles. Conidiophores reduced to conidiogenous cells. Conidiophores cells hyaline, smooth, cylindrical, subcylindrical to ampulliform, 5.0–12.0 μm × 1.0–2.0 μm. Conidia hyaline, smooth, aseptate, biguttulate, cylindric-clavate, 4.0–6.0 μm × 1.0–2.0 μm, mean = 5.0 μm × 2.0 μm, L/W ratio = 3.0, n = 30. Sexual morph not observed.
Culture characteristics: Colonies on PDA were flat and fluffy with pale gray aerial mycelium. Surface white and pale gray initially, then becoming gray and fawnby age, reverse brown. PDA attaining 47.1–48.6 mm in diameter after 1 week at 25 °C, growth rate 6.7–6.9 mm/day. PDA attaining 53.9–72.3 mm in diameter after 2 weeks at 25 °C, growth rate 3.9–5.2 mm/day.
Material examined: China: Fujian Province, Quanzhou City, 24°54′38″ N, 117°13′46″ E, on diseased leaves of Castanopsis fargesii (Fagaceae), 17 September 2022, paratype HMAS 352710; ex-paratype living culture CGMCC3.25675.
Notes: To the best of our knowledge this is the first description of Castanopsis on leaves of Castanopsis fargesii in China, and Dendrostoma castanopsidis sp. nov. is depicted. Based on the multigene phylogenetic analyses of ITS, LSU, tef1, and rpb2, our two strains were closer to D. creticum, D. elaeocarpi, and D. istriacum with medium support in their clade (82% ML/0.93 PP, Figure 5). BLASTn analysis of D. castanopsidis (CGMCC3.25676) and D. creticum (CBS 145802) revealed 33 bp (33/615 bp, 5.4%) nucleotide differences in ITS, 78 bp (78/203 bp, 38.4%) nucleotide differences in tef1, and 44 bp (44/912 bp, 4.9%) nucleotide differences in rpb2. BLASTn analysis of D. castanopsidis (CGMCC3.25676) and D. elaeocarpi (CFCC 53113) revealed 31 bp (31/466 bp, 6.7%) nucleotide differences in ITS, 84 bp (84/209 bp, 40.2%) nucleotide differences in tef1, and 56 bp (56/711 bp, 7.9%) nucleotide differences in rpb2. BLASTn analysis of D. castanopsidis (CGMCC3.25676) and D. istriacum (CBS 145801) revealed 31 bp (31/616 bp, 5.0%) nucleotide differences in ITS, 90 bp (90/212 bp, 42.5%) nucleotide differences in tef1, and 49 bp (49/942 bp, 5.2%) nucleotide differences in rpb2. In morphology, the conidia and conidiogenous cells of D. castanopsidis are narrower than D. istriacum (1.0–2.0 vs. 1.9–2.7 μm; 1.0–2.0 vs. 1.8–5.3 μm). Therefore, we introduce the new fungal species: Dendrostoma castanopsidis sp. nov.
3.2.3. Pseudoplagiostoma wuyishanense T.C. Mu and J.Z. Qiu, sp. nov., Figure 6
MycoBank Number: MB853512
Etymology: the epithet “wuyishanense” refers to the locality, Wuyi Mountain National Nature Reserve.
Holotype: China: Fujian Province, Wuyi Mountain National Nature Reserve, 27°40′13″ N, 117°39′23″ E, on dead branches of an unidentified tree, 6 September 2022, holotype HMAS 352519; ex-holotype living culture CGMCC3.25368.
Description: Saprobic on dead branches of broadleaf tree. Asexual morphs developed on PDA. Conidiomata were pycnidial, globose to subglobose, superficial in culture, black orbrown, solitary or aggregated, exuding cream to pale luteous conidial masses from ostioles. Conidiophores reduced to conidiogenous cells. Conidiophores cells hyaline, smooth, cylindrical to clavate, phialidic, 12.0–18.0 μm × 3.0–4.0 μm. Conidia hyaline, smooth, aseptate, long-oval, subcylindrical, 20.0–26.0 μm × 10.0–14.0 μm, mean = 23.0 μm × 12.0 μm, L/W ratio = 1.8, n = 30, base with inconspicuous to conspicuous hilum (1.0–2.0 µm diam). Sexual morph not observed.
Culture characteristics: Colonies on PDA flat and fluffy with gray aerial mycelium. Surface white and pale gray initially, then becoming gray by age, reverse brown to black in the center and gray margin. Colonies on SNA (containing pine needle) wavy hyphae, aerial mycelium white to grayish. PDA attaining 25.1–29.5 mm in diameter after 1 week at 25 °C, growth rate 3.6–4.2 mm/day. PDA attaining 60.2–67.5 mm in diameter after 2 weeks at 25 °C, growth rate 4.3–4.8 mm/day. SNA attaining 58.7–65.5 mm in diameter after 2 weeks at 25 °C, growth rate 4.2–4.7 mm/day.
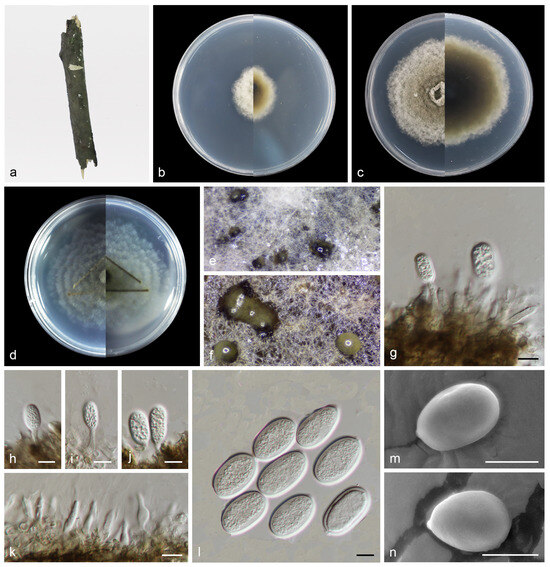
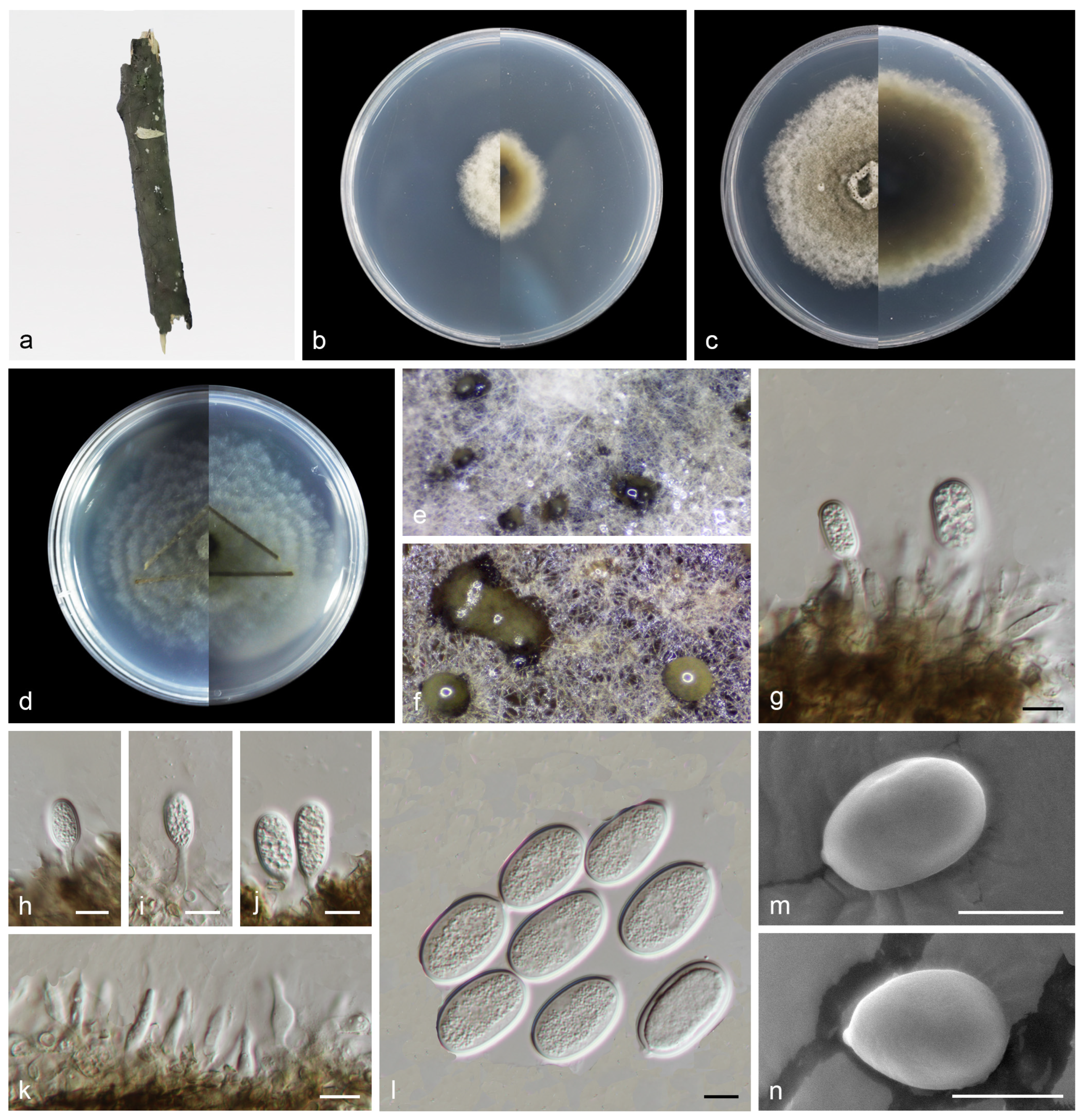
Figure 6.
Pseudoplagiostoma wuyishanense (holotype HMAS 352519). (a) Dead branches; (b) surface and reverse sides of colony after 7 days on PDA (c) and 14 days; (d) surface and reverse sides of colony after 14 days on SNA; (e,f) mass of conidia; (g–j) conidiogenous cells with conidia; (k) conidiogenous cells; (l–n) conidia. Scale bars: (g–n) 10 µm.
Figure 6.
Pseudoplagiostoma wuyishanense (holotype HMAS 352519). (a) Dead branches; (b) surface and reverse sides of colony after 7 days on PDA (c) and 14 days; (d) surface and reverse sides of colony after 14 days on SNA; (e,f) mass of conidia; (g–j) conidiogenous cells with conidia; (k) conidiogenous cells; (l–n) conidia. Scale bars: (g–n) 10 µm.
Material examined: China: Fujian Province, Wuyi Mountain National Nature Reserve, 27°40′13″ N, 117°39′23″ E, on dead branches of an unidentified tree, 6 September 2022, paratype HMAS 352518; ex-paratype living culture CGMCC3.25367.
Notes: In this study, Pseudoplagiostoma wuyishanense sp. nov. is described from dead branches of an unidentified broadleaf tree in China. Tang et al. found P. dipterocarpicola from dead twigs of Dipterocarpus sp. in northern Thailand [39]. Based on the multigene phylogenetic analyses of ITS, LSU, tub2, tef1, and rpb2, the new species is well supported (100% ML/1 PP, Figure 6) within Pseudoplagiostoma and close to P. bambusae. P. bambusae was described from the diseased leaves of Bambusoideae sp. in China, Fujian Province, Fujian Wuyi Mountain National Nature Reserve [23]. However, BLASTn analysis of P. wuyishanense (CGMCC3.25368) and P. bambusae (SAUCC 1206-4) revealed 29 bp (29/600 bp, 4.8%) nucleotide differences in ITS, 194 bp (194/835 bp, 23.2%) nucleotide differences in LSU, 63 bp (63/188 bp, 33.5%) nucleotide differences in tef1, and 46 bp (46/474 bp, 9.7%) nucleotide differences in tub2. Morphologically, the conidia and conidiogenous cells of P. wuyishanense are large than P. bambusae (20.0–26.0 μm × 10.0–14.0 μm vs. 13–20 μm × 5.7–7.6 μm; 12.0–18.0 μm × 3.0–4.0 μm vs. 5.0–13.0 μm × 1.5–2.5 μm). Therefore, we describe this fungus as a new species. Two species of Pseudoplagiostoma (P. bambusae, P. wuyishanense) were found in the same place (Fujian Wuyi Mountain National Nature Reserve, Fujian Province, China). This indicates that there may be a high species diversity of Pseudoplagiostoma in this area.
4. Discussion
The Diaporthales constitute a cosmopolitan, highly diverse group of sac fungi that includes saprophytic, endophytic, and phytopathogenic members; however, important aspects of their interfamilial taxonomic relationships remain poorly characterized. A number of Diaporthales species were resolved based on morphological characteristics and multi-locus phylogenetic analyses [15]. In this study, three new species (Chrysofolia camelliae sp. nov., Dendrostoma castanopsidis sp. nov., and Pseudoplagiostoma wuyishanense sp. nov.) belonging to three families (Cryphonectriaceae, Erythrogloeaceae, and Pseudoplagiostomataceae, respectively) in Diaporthales from China were identified and described.
Previously, the identification of genera and species of Diaporthales depended largely on the host specificity and morphology, which led to several incorrect placements (and naming) [10]. However, with the development of molecular phylogenic approaches, more recent comprehensive methods have been applied to resolving issues within the taxonomy of Diaporthales. For example, Cytosporaceae, Diaporthaceae, Gnomoniaceae, and Melanconidaceae were accepted in Diaporthales by phylogenetic analyses of LSU sequences in 2002 [4]. Cryphonectriaceae was established to accommodate Cryphonectria, Chrysoporthe, Endothia, and allied genera by using ITS, LSU, tub1, and tub2 sequence data in 2006 [40]. Cheewangkoon et al. introduced Pseudoplagiostomaceae by using LSU in 2010 [21]. Based on phylogenetic analyses of combined loci of ITS, LSU, tef1, and rpb2, Senanayake et al. introduced Auratiopycnidiellaceae, Coryneaceae, and Erythrogloeaceae [6]. Recently, Wijayawardene et al. accepted 32 families in Diaporthales [2]. Combined, these advances suggest increased characterization and diversity within Diaporthales.
Presently, six species of Chrysofolia are accepted and have been reported as plant pathogens and endophytes. Chrysofolia barringtoniae, Chrysofolia fructicola, and Chrysofolia kunmingensis were isolated as endophytes from leaves of Barringtonia acutangula, Rosa roxburghii, and Coriaria nepalensis, respectively, whereas Chrysofolia coriariae appears to be a pathogen of the Coriaria nepalensis plant [14,16,17,18]. Thus, the same species can play multiple roles and may develop into a plant pathogen from initial endophytic interactions under some conditions, e.g., stress or changes to host plant health [41,42].
Twenty-two species of Dendrostoma have been reported as tree pathogens, with Dendrostoma infecting branches, twigs, and stems of Fagaceae plants, including beeches, chestnuts, and oaks, resulting in production losses important in both agriculture and forestry [20]. The identification of Dendrostoma castanopsidis in this study indicated its associated with the evergreen Castanopsis fargesii (Fagaceae). Interestingly, Fagaceae is an important flowering plant family (~930 species) with temperate members, mainly deciduous, whereas tropical varieties are evergreens and shrubs [43,44]. With increased collection, fungal Fagaceae plant pathogens, particularly those of Dendrostoma,are likely to be found in China and beyond. Although this is the first report on the description of Dendrostoma castanopsidis from Castanopsis fargesii (Fagaceae), this finding is in line with an earlier study [19] which reported Dendrostoma collected from Fagaceae plants, namely Castanea mollissima (harboring D. aurorae, D. castaneae, D. castaneicola, D. chinense, D. shaanxiense, and D. shandongense), C. sativa (D. atlanticum, D. castaneum, and D. luteum), Fagus sylvatica (D. covidicola), Quercus acutissima (D. quercinum), Q. aliena (D. parasiticum and D. qinlingense), Q. coccifera (D. creticum), Q. ilex (D. istriacum), Q. mongolica (D. donglingense), Quercus sp. (D. dispersum, D. leiphaemia, and D.quercus), and Q. wutaishanica (D. parasiticum and D. qinlingense).
The identification of a new species of Pseudoplagiostoma, Pseudoplagiostoma wuyishanense sp. nov., from dead branches reveals that members of the Pseudoplagiostoma could potentially contribute to key ecological functions in plant biomass turnover as saprobes [39]. Several studies have reported the abundance of Pseudoplagiostoma in Asia. For example, P. eucalypti was found from leaf spots in Viet Nam [21]. P. dipterocarpi and P. dipterocarpicola were also collected from Dipterocarpus in Thailand [22,39]. Phookamsak et al. found P. mangiferae from leaves of Mangifera in China [45]. Zhang et al. described three species of Pseudoplagiostoma in China, viz., P. alsophilae, P. bambusae, and P. machili. Through the study and analysis historical biogeography, Asia is probably the ancestral area of Pseudoplagiostoma [23]. Despite this, additional studies of Pseudoplagiostoma are required to reveal species and ecological diversity.
5. Conclusions
In this study, we have identified three new species of Diaporthales isolated in Fujian Province, China, namely Chrysofolia camelliae sp. nov. (Cryphonectriaceae), Dendrostoma castanopsidis sp. nov. (Erythrogloeaceae), and Pseudoplagiostoma wuyishanense sp. nov. (Pseudoplagiostomataceae). Identification was based on morphological and multi-locus phylogenetic analyses, which strongly supported their designations. Our data contribute to the characterization of the ecological diversity of Diaporthales. Identifying the attributes and functional consequences of these fungi is warranted.
Author Contributions
Conceptualization, T.M., X.G. and J.Q.; methodology, Y.L.; software, H.P.; validation, Z.L., C.L. and J.X.; formal analysis, X.C.; investigation, X.Z.; resources, T.M. and Z.Z.; data curation, T.M., Y.L., N.O.K., H.P., Z.L., C.L., J.X., X.C., X.Z., Z.Z., H.L., M.Y.J.-S., P.J., J.W. and S.H.; writing—original draft preparation, T.M. and M.Y.J.-S.; writing—review and editing, N.O.K. and J.Q.; visualization, H.L., P.J. and S.H.; supervision, X.G. and J.Q.; project administration, X.G. and J.Q.; funding acquisition, J.W., X.G. and J.Q. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the National Natural Science Foundation of China (Nos. 32270029, U1803232, 31670026), the National Key R & D Program of China (No. 2017YFE0122000), a Social Service Team Support Program Project (No. 11899170165), Science and Technology Innovation Special Fund (Nos. KFB23084, CXZX2019059S, CXZX2019060G) of Fujian Agriculture and Forestry University, a Fujian Provincial Major Science and Technology Project (No. 2022NZ029017), an Investigation and evaluation of biodiversity in the Jiulong River Basin (No. 082·23259-15), and Macrofungal and microbial resource investigation project in Longqishan Nature Reserve (No. SMLH2024(TP)-JL003#).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
All sequences generated in this study were submitted to the NCBI database.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Fan, X.L.; Bezerra, J.D.P.; Tian, C.M.; Crous, P.W. Families and genera of diaporthalean fungi associated with canker and dieback of tree hosts. Persoonia 2018, 40, 119–134. [Google Scholar] [CrossRef] [PubMed]
- Wijayawardene, N.N.; Hyde, K.D.; Dai, D.Q.; Sánchez-García, M.; Goto, B.T.; Saxena, R.K.; Erdoğdu, M.; Selçuk, F.; Rajeshkumar, K.C.; Aptroot, A.; et al. Outline of Fungi and fungus-like taxa—2021. Mycosphere 2022, 13, 53–453. [Google Scholar] [CrossRef]
- Xiao, X.E.; Liu, Y.D.; Zheng, F.; Xiong, T.; Zeng, Y.T.; Wang, W.; Zheng, X.L.; Wu, Q.; Xu, J.P.; Crous, P.W.; et al. High species diversity in Diaporthe associated with citrus diseases in China. Persoonia 2023, 51, 229–256. [Google Scholar] [CrossRef] [PubMed]
- Castlebury, L.A.; Rossman, A.Y.; Jaklitsch, W.J.; Vasilyeva, L.N. A preliminary overview of the Diaporthales based on large subunit nuclear ribosomal DNA sequences. Mycologia 2002, 94, 1017–1031. [Google Scholar] [CrossRef] [PubMed]
- Rossman, A.Y.; Farr, D.F.; Castlebury, L.A. A review of the phylogeny and biology of the Diaporthales. Mycoscience 2007, 48, 135–144. [Google Scholar] [CrossRef]
- Senanayake, I.C.; Crous, P.W.; Groenewald, J.Z.; Maharachchikumbura, S.S.N.; Jeewon, R.; Phillips, A.J.L.; Bhat, J.D.; Perera, R.H.; Li, Q.R.; Li, W.J.; et al. Families of Diaporthales based on morphological and phylogenetic evidence. Stud. Mycol. 2017, 86, 217–296. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.K.; Hyde, K.D.; Mapook, A.; Maharachchikumbura, S.S.N.; Bhat, J.D.; McKenzie, E.H.C.; Jeewon, R.; Wen, T.C. Taxonomic studies of some often over-looked Diaporthomycetidae and Sordariomycetidae. Fungal Divers. 2021, 111, 443–572. [Google Scholar] [CrossRef]
- Jiang, N.; Fan, X.L.; Tian, C.M. Identification and Characterization of Leaf-Inhabiting Fungi from Castanea Plantations in China. J. Fungi 2021, 7, 64. [Google Scholar] [CrossRef] [PubMed]
- Luo, Z.L.; Hyde, K.D.; Liu, J.K.; Maharachchikumbura, S.S.N.; Jeewon, R.; Bao, D.F.; Bhat, D.J.; Lin, C.G.; Li, W.L.; Yang, J.; et al. Freshwater Sordariomycetes. Fungal Divers. 2019, 99, 451–660. [Google Scholar] [CrossRef]
- Gomes, R.R.; Glienke, C.; Videira, S.I.; Lombard, L.; Groenewald, J.Z.; Crous, P.W. Diaporthe: A genus of endophytic, saprobic and plant pathogenic fungi. Persoonia 2013, 31, 1–41. [Google Scholar] [CrossRef]
- Wang, S.; Jiang, N.; Ma, R. Morphology and Phylogeny Reveal Three New Species of Cytospora Associated with Tree Cankers in China. J. Fungi 2024, 10, 139. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Chen, S. Identification and pathogenicity of Aurifilum species (Cryphonectriaceae, Diaporthales) on Terminalia species in Southern China. MycoKeys 2023, 98, 37–58. [Google Scholar] [CrossRef] [PubMed]
- Crous, P.W.; Wingfield, M.J.; Guarro, J.; Hernández-Restrepo, M.; Sutton, D.A.; Acharya, K.; Barber, P.A.; Boekhout, T.; Dimitrov, R.A.; Dueñas, M.; et al. Fungal Planet description sheets: 320–370. Persoonia 2015, 34, 167–266. [Google Scholar] [CrossRef] [PubMed]
- Lei, Y.; Yang, E.; Dao, C.; Li, X.; Karunarathna, S.C.; Tibpromma, S.; Yan, K. Endophytic fungi associated with Coriaria nepalensis in an abandoned open-pit phosphate mine in Yunnan, P.R. China. Phytotaxa 2023, 616, 27–46. [Google Scholar] [CrossRef]
- Wang, S.Y.; McKenzie, E.H.C.; Phillips, A.J.L.; Li, Y.; Wang, Y. Taxonomy and Multigene Phylogeny of Diaporthales in Guizhou Province, China. J. Fungi 2022, 8, 1301. [Google Scholar] [CrossRef] [PubMed]
- Suwannarach, N.; Kumla, J.; Sri-Ngernyuang, K.; Lumyong, S. A new endophytic fungus, Chrysofolia barringtoniae sp. nov., from Thailand. Mycoscience 2016, 57, 361–365. [Google Scholar] [CrossRef]
- Zhang, H.; Mao, Y.T.; Ma, M.X.; Tao, G.C.; Wei, T.P.; Jiang, Y.L. Culturable endophytic Sordariomycetes from Rosa roxburghii: New species and lifestyles. J. Syst. Evol. 2023. [Google Scholar] [CrossRef]
- Ma, T.; Yang, Z. First Report of a New Species, Chrysofolia galloides sp. nov., Causing Black Spots on Gallnuts of Rhus punjabensis var. sinica in China. Plant Disease 2024, 108, 796. [Google Scholar] [CrossRef] [PubMed]
- Samarakoon, M.C.; Promputtha, I.; Liu, J.K. Dendrostoma covidicola sp. nov. (Erythrogloeaceae, Diaporthales) on Fagussylvatica from Sichuan Province, China. Phytotaxa 2021, 483, 85–94. [Google Scholar] [CrossRef]
- Jiang, N.; Fan, X.L.; Crous, P.W.; Tian, C.M. Species of Dendrostoma (Erythrogloeaceae, Diaporthales) associated with chestnut and oak canker diseases in China. MycoKeys 2019, 48, 67–96. [Google Scholar] [CrossRef]
- Cheewangkoon, R.; Groenewald, J.Z.; Verkley, G.J.M.; Hyde, K.D.; Wingfield, M.J.; Gryzenhout, M.; Summerell, B.A.; Denman, S.; Toanun, C.; Crous, P.W. Re-evaluation of Cryptosporiopsis eucalypti and Cryptosporiopsis-like species occurring on Eucalyptus leaves. Fungal Divers. 2010, 44, 89–105. [Google Scholar] [CrossRef]
- Suwannarach, N.; Kumla, J.; Lumyong, S. Pseudoplagiostoma dipterocarpi sp. nov., a new endophytic fungus from Thailand. Mycoscience 2016, 57, 118–122. [Google Scholar] [CrossRef]
- Zhang, Z.; Liu, X.; Tao, M.; Liu, X.; Xia, J.; Zhang, X.; Meng, Z. Taxonomy, Phylogeny, Divergence Time Estimation, and Biogeography of the Family Pseudoplagiostomataceae (Ascomycota, Diaporthales). J. Fungi 2023, 9, 82. [Google Scholar] [CrossRef] [PubMed]
- Fu, M.; Crous, P.W.; Bai, Q.; Zhang, P.F.; Xiang, J.; Guo, Y.S.; Zhao, F.F.; Yang, M.M.; Hong, N.; Xu, W.X.; et al. Colletotrichum species associated with anthracnose of Pyrus spp. in China. Persoonia 2019, 42, 1–35. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Zhang, J.; Li, D.; Xia, J.; Zhang, X. Morphological and Phylogenetic Analyses Reveal Three New Species of Pestalotiopsis (Sporocadaceae, Amphisphaeriales) from Hainan, China. Microorganisms 2023, 11, 1627. [Google Scholar] [CrossRef]
- Kumar, S.; Stecher, G.; Tamura, K. Mega7: Molecular Evolutionary Genetics Analysis Version 7.0 for Bigger Datasets. Mol. Biol. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef]
- White, T.J.; Bruns, T.D.; Lee, S.B.; Taylor, J.W. Amplification and direct sequencing offungal ribosomal RNA genes for phylogenetics. In PCR Protocols: A Guide to Methods and Applications; Innis, M.A., Gelfand, D.H., Sninsky, J.J., White, T.J., Eds.; Academic Press: New York, NY, USA, 1990; pp. 315–322. [Google Scholar]
- Rehner, S.A.; Samuels, G.J. Taxonomy and phylogeny of Gliocladium analysed from nuclear large subunit ribosomal DNAsequences. Mycol. Res. 1994, 98, 625–634. [Google Scholar] [CrossRef]
- Vilgalys, R.; Hester, M. Rapid genetic identification and mapping of enzymatically amplified ribosomal DNA from several Cryptococcus species. J. Bacteriol. 1990, 172, 4238–4246. [Google Scholar] [CrossRef]
- Liu, Y.J.; Whelen, S.; Hall, B.D. Phylogenetic Relationships among Ascomycetes: Evidence from an RNA polymerse II subunit. Mol. Biol. Evol. 1999, 16, 1799–1808. [Google Scholar] [CrossRef]
- Carbone, I.; Kohn, L.M. A method for designing primer sets for speciation studies in filamentous ascomycetes. Mycologia 1999, 91, 553–556. [Google Scholar] [CrossRef]
- Glass, N.L.; Donaldson, G.C. Development of primer sets designed for use with the PCR to amplify conserved genes fromfilamentous ascomycetes. Appl. Environ. Microbiol. 1995, 61, 1323–1330. [Google Scholar] [CrossRef] [PubMed]
- Katoh, K.; Rozewicki, J.; Yamada, K.D. Mafft online service: Multiple sequence alignment, interactive sequence choice and visualization. Brief. Bioinform. 2019, 20, 1160–1166. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, L.T.; Schmidt, H.A.; von Haeseler, A.; Minh, B.Q. Iq-Tree: A Fast and Effective Stochastic Algorithm for Estimating Maximum-Likelihood Phylogenies. Mol. Biol. Evol. 2015, 32, 268–274. [Google Scholar] [CrossRef] [PubMed]
- Ronquist, F.; Teslenko, M.; van der Mark, P.; Ayres, D.L.; Darling, A.; Höhna, S.; Larget, B.; Liu, L.; Suchard, M.A.; Huelsenbeck, J.P. Mrbayes 3.2: Efficient Bayesian Phylogenetic Inference and Model Choice across a Large Model Space. Syst. Biol. 2012, 61, 539–542. [Google Scholar] [CrossRef] [PubMed]
- Xiang, C.Y.; Gao, F.; Jakovlić, I.; Lei, H.P.; Hu, Y.; Zhang, H.; Zou, H.; Wang, G.T.; Zhang, D. Using Phylosuite for molecular phylogeny and tree-based analyses. iMeta 2023, 2, e87. [Google Scholar] [CrossRef]
- Hoang, D.T.; Chernomor, O.; von Haeseler, A.; Minh, B.Q.; Vinh, L.S. Ufboot2: Improving the Ultrafast Bootstrap Approximation. Mol. Biol. Evol. 2017, 35, 518–522. [Google Scholar] [CrossRef] [PubMed]
- Lanfear, R.; Frandsen, P.B.; Wright, A.M.; Senfeld, T.; Calcott, B. Partitionfinder 2: New Methods for Selecting Partitioned Models of Evolution for Molecular and Morphological Phylogenetic Analyses. Mol. Biol. Evol. 2016, 34, 772–773. [Google Scholar] [CrossRef] [PubMed]
- Tang, X.; Jayawardena, R.S.; Stephenson, S.L.; Kang, J.C. A new species Pseudoplagiostoma dipterocarpicola (Pseudoplagiostomataceae, Diaporthales) found in northern Thailand on members of the Dipterocarpaceae. Phytotaxa 2022, 543, 233–243. [Google Scholar] [CrossRef]
- Gryzenhout, M.; Myburg, H.; Wingfield, B.D.; Wingfield, M.J. Cryphonectriaceae (Diaporthales), a new family including Cryphonectria, Chrysoporthe, Endothiaand allied genera. Mycologia 2006, 98, 239–249. [Google Scholar] [CrossRef]
- Promputtha, I.; Hyde, K.D.; McKenzie, E.H.C.; Peberdy, J.F.; Lumyong, S. Can leaf degrading enzymes provide evidence that endophytic fungi becoming saprobes? Fungal Divers. 2010, 41, 89–99. [Google Scholar] [CrossRef]
- Sahu, N.; Indic, B.; Wong-Bajracharya, J.; Merényi, Z.; Ke, H.M.; Ahrendt, S.; Monk, T.L.; Kocsubé, S.; Drula, E.; Lipzen, A.; et al. Vertical and horizontal gene transfer shaped plant colonization and biomass degradation in the fungal genus Armillaria. Nat. Microbiol. 2023, 8, 1668–1681. [Google Scholar] [CrossRef] [PubMed]
- Jiang, N.; Voglmayr, H.; Bian, D.R.; Piao, C.G.; Wang, S.K.; Li, Y. Morphology and Phylogeny of Gnomoniopsis (Gnomoniaceae, Diaporthales) from Fagaceae Leaves in China. J. Fungi 2021, 7, 792. [Google Scholar] [CrossRef] [PubMed]
- Jiang, N.; Voglmayr, H.; Xue, H.; Piao, C.G.; Li, Y. Morphology and Phylogeny of Pestalotiopsis (Sporocadaceae, Amphisphaeriales) from Fagaceae Leaves in China. Microbiol. Spectr. 2022, 10, 1–28. [Google Scholar] [CrossRef] [PubMed]
- Phookamsak, R.; Hyde, K.D.; Jeewon, R.; Bhat, D.J.; Jones, E.B.G.; Maharachchikumbura, S.S.N.; Raspé, O.; Karunarathna, S.C.; Wanasinghe, D.N.; Hongsanan, S.; et al. Fungal diversity notes 929–1035: Taxonomic and phylogenetic contributions on genera and species of fungi. Fungal Divers. 2019, 95, 1–273. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).