Abstract
Scytalidiosis in humans primarily causes feet and nail infections, with systemic infections rarely reported. In dogs, only one systemic infection of Scytalidium spp. has been reported to date. A 3-year-old giant schnauzer presented with loss of appetite, lethargy, and hind limb lameness. A complete clinical examination was performed, along with hematobiochemical tests, radiography, CT, MRI, and cytological and microbiological analyses of it enlarged lymph nodes. Hyperglobulinemia, vertebral osteolysis, and generalized lymphadenomegaly were diagnosed. Cytopathological and molecular investigations confirmed Scytalidium. Although treated with itraconazole, the dog’s condition worsened after a premature discontinuation of therapy, leading to euthanasia. A post-mortem and histopathological examination revealed widespread infection. This case highlights the need to consider fungal infections in cases of elevated β-2 protein.
1. Introduction
Fungal infections are often insidious, diagnosed late, and sometimes only detected during necropsy and they pose a significant threat to animal health due to their high mortality and morbidity rates [1]. These infections can be classified as superficial, subcutaneous, or systemic. Among these, systemic mycoses are rarely reported in animals and are mainly observed in those with a compromised immune system or underlying disorders, with only one case in the literature attributed to Scytalidium spp. [2]. Scytalidium infections typically affect the feet and nails of individuals who walk barefoot on contaminated soil in tropical regions [3]. Here, we describe a fatal case of systemic scytalidiosis in an apparently immunocompetent giant schnauzer dog, characterized by hind limb pain, spinal pain, and generalized lymphadenomegaly. Clinico-pathologically, the dog presented with severe hyperbetaglobulinemia, which was reduced following antifungal therapy.
2. Case Presentation
A three-year old female giant schnauzer dog was referred to the veterinary hospital due to a loss of appetite, reluctance to move, kyphosis, and left hind leg lameness. The dog resided in a rural area of the Po Valley in Northern Italy, had regular access to the outdoors and was consistently fed with commercial food.
At the time of the initial evaluation by the referring veterinarian (day 0), complete hematological and biochemical examinations were conducted. The Complete Blood Count (CBC) examination (ADVIA 2120, Siemens, Munich, Germany) revealed mild leukopenia (4.7 × 103/µL; reference range 5.45–12.98 × 103/µL). Reactive lymphocytes were observed in blood smears. Serum biochemical abnormalities included hyperproteinaemia (8.7 g/dL; reference range 5.2–8.5 g/dL) (AU 680, Beckman coulter, Brea, CA, USA), with an albumin/globulin ratio of 0.30. Capillary electrophoresis of serum protein (Capillarys, Sebia, Lisses, France) showed a peak in the β-2 fraction (2.48 g/dL; reference intervals 0.37–1.62 g/dL). The dog was treated with a broad-spectrum antimicrobial (cephalexin) and NSAIDs for three weeks and showed slight improvement. On day +90, due to a progressive worsening of the dog’s clinical condition, the owner returned to the practitioner.
Physical examination showed that the dog was alert and responsive with a normal respiratory rate, heart rate, and rectal temperature. At physical examination, mild lumbar discomfort was elicited by palpation and the dog showed left hind limb lameness.
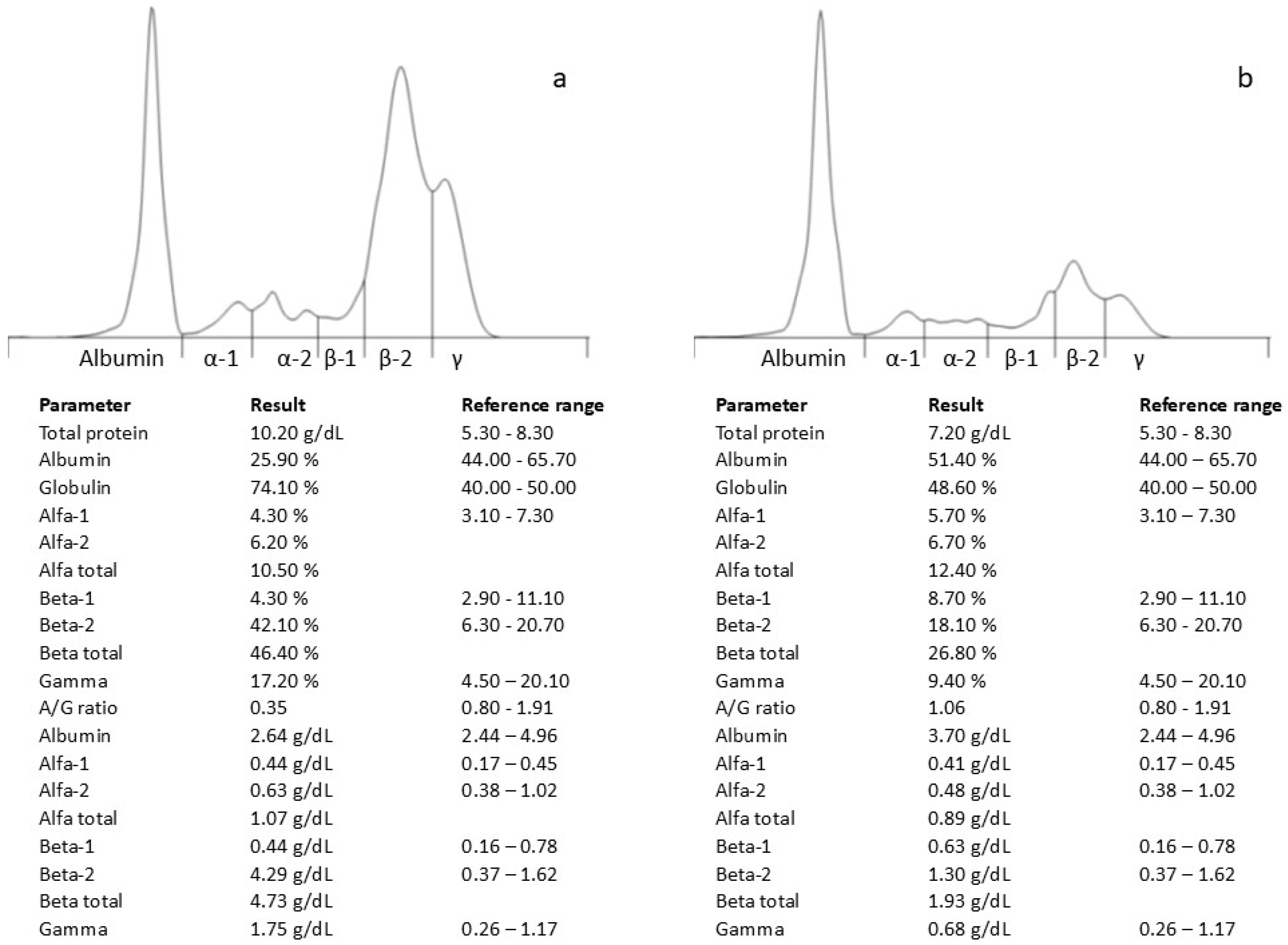
Hematological and biochemical analyses revealed a further increased plasma protein concentration (10.2 g/dL; reference range 5.2–8.2 g/dL) and hypoalbuminemia (2.36 g/dL; reference range 2.6–3.80 g/dL), with an electrophoretic pattern similar to the previous findings (Figure 1a). An ELISA test against the Leishmania infantum antibody was negative (VetLine Leishmania, Novatec Germany, Leinfelden-Echterdingen, Germany).
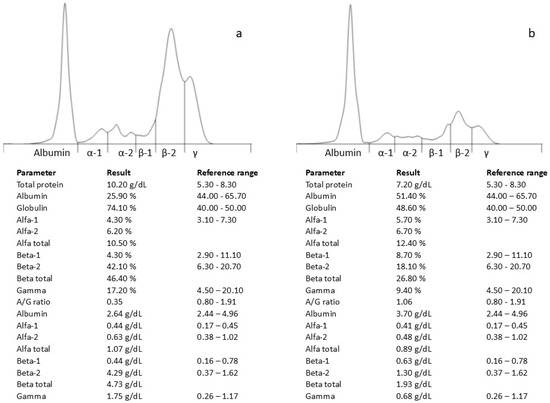
Figure 1.
Serum protein electrophoresis profile before (a) and after antifungal therapy (b).
2.1. Imaging Evaluations
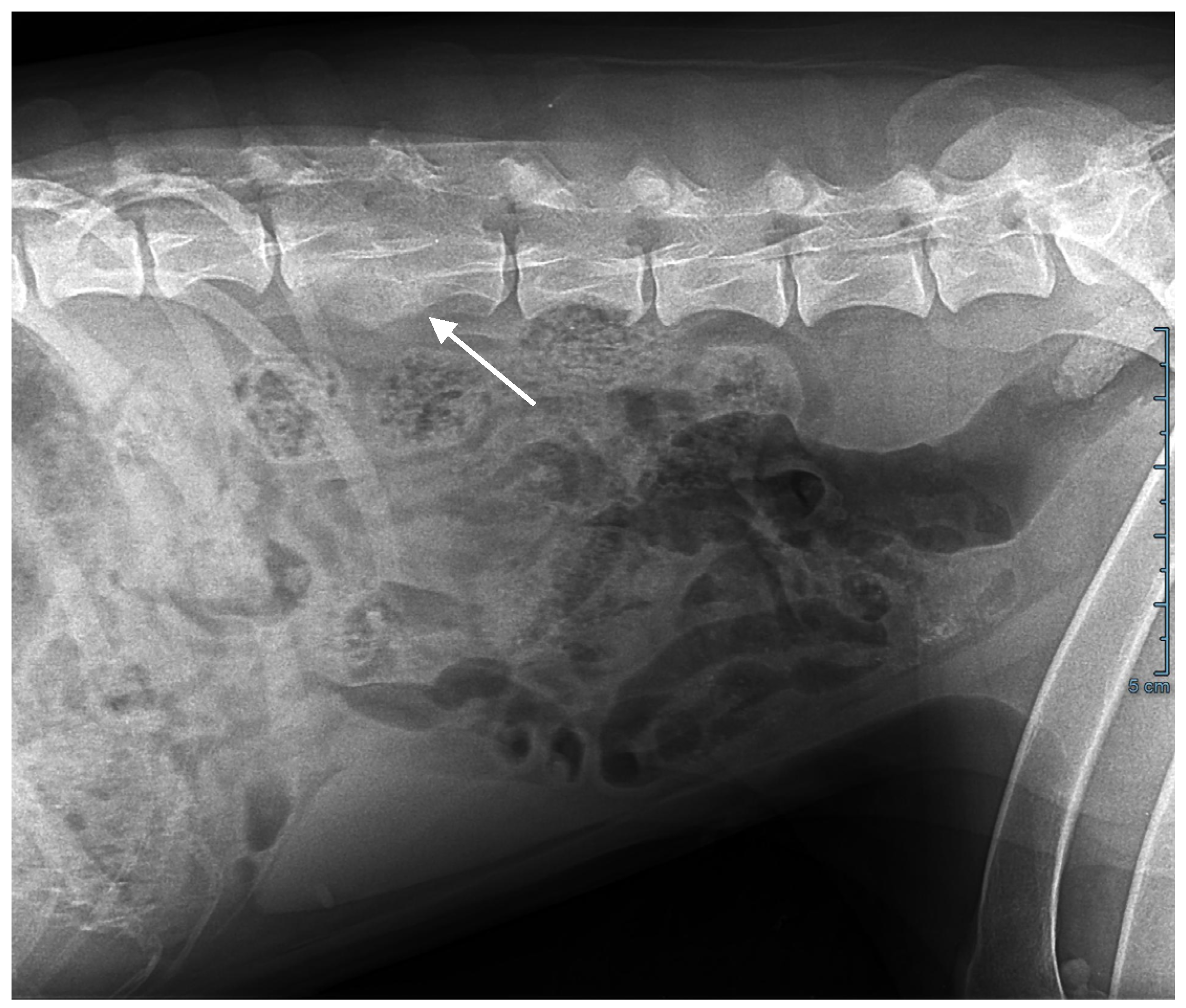
Abdominal ultrasonography, computed tomography, and an MRI scan showed T3-T4 thoracic and L2-L3 lumbar discospondylitis with osteolysis and non-compressive ankylosing hyperostosis (Figure 2); generalized lymphadenomegaly with a five-fold increase in lymph node volume and a loss of structure was also observed.
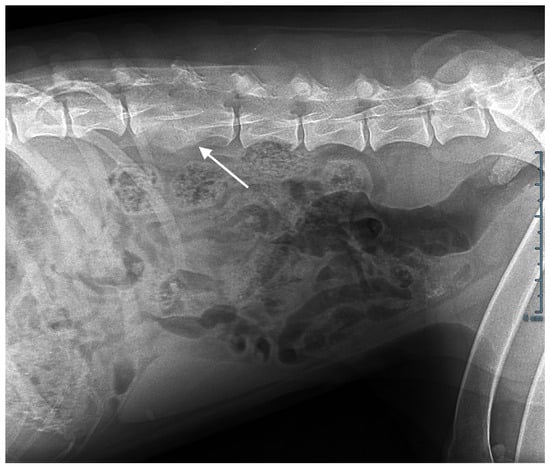
Figure 2.
Abdominal X-ray. L2-L3 osteolysis and bone remodelling with fusion of the vertebral bodies (arrow).
2.2. Cytological Examination
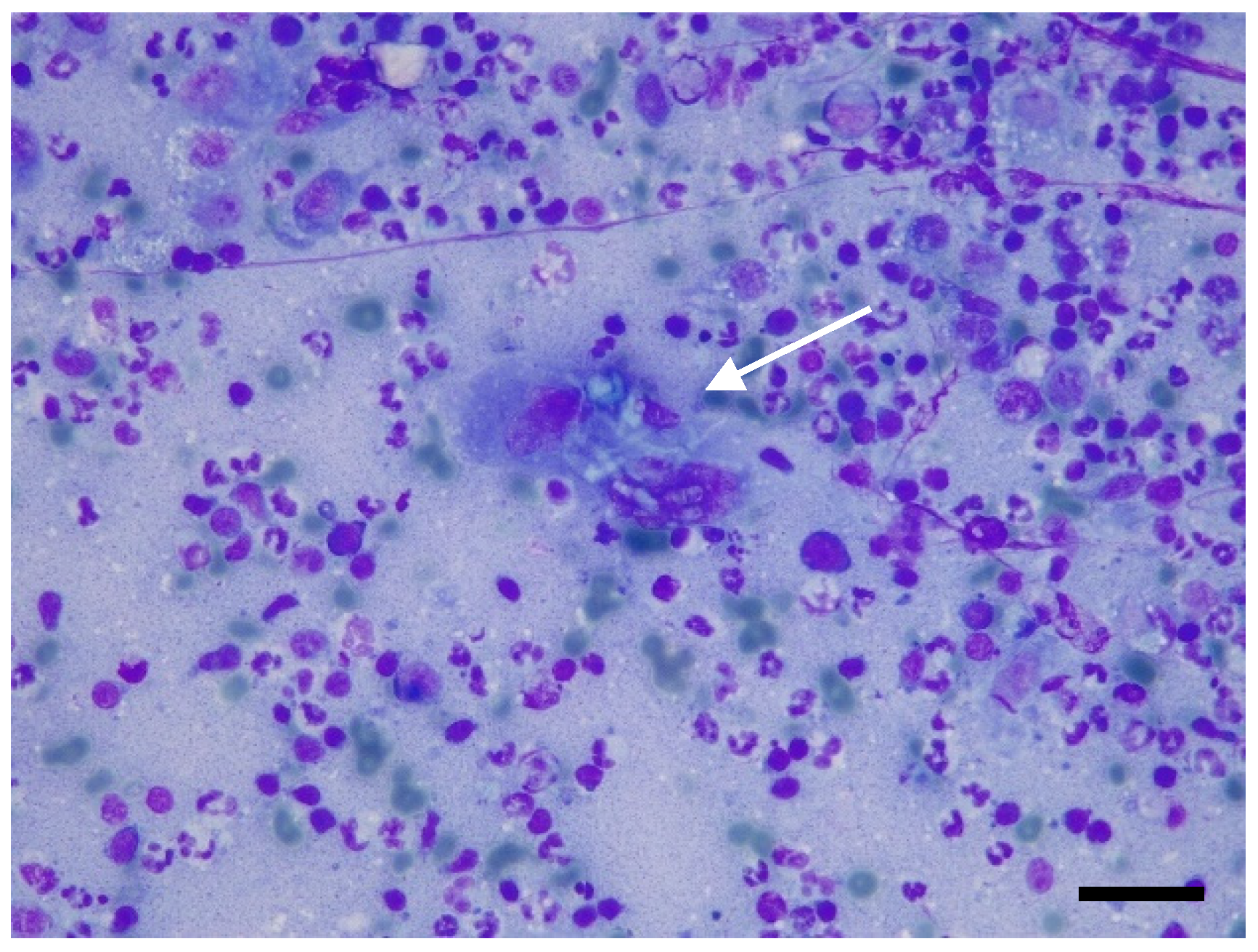
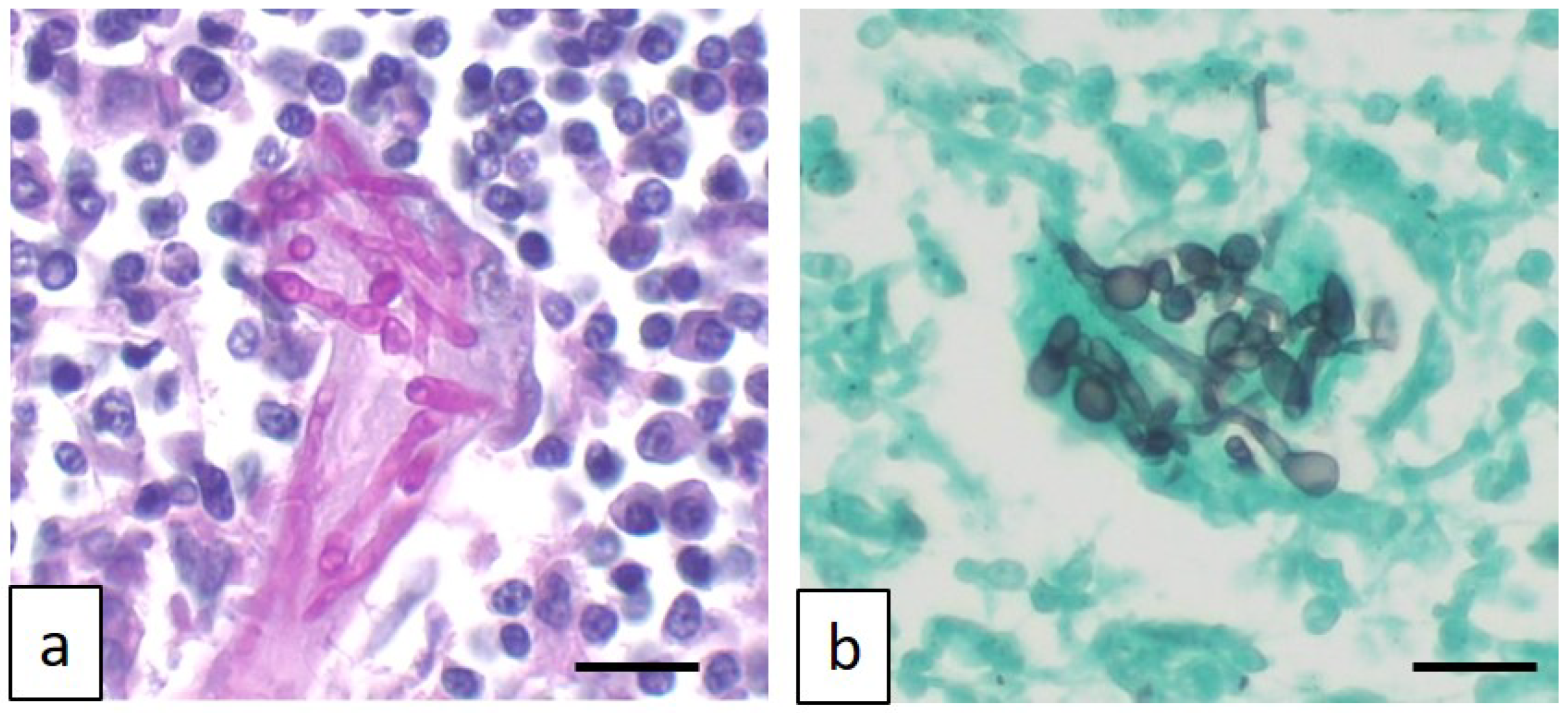
Based on the imaging results, fine-needle aspiration was requested and carried out from the popliteal lymph node. Smears were air-dried and stained with Romanowsky stain (May–Grunwald Giemsa, Merck KGaA, Frankfurter- Darmstadt, Germany). Neutrophilic–macrophagic cell infiltrates were present, with occasionally segmented septate hyphae and, more rarely, lymphocytes, plasma cells, and eosinophilic granulocytes (Figure 3).
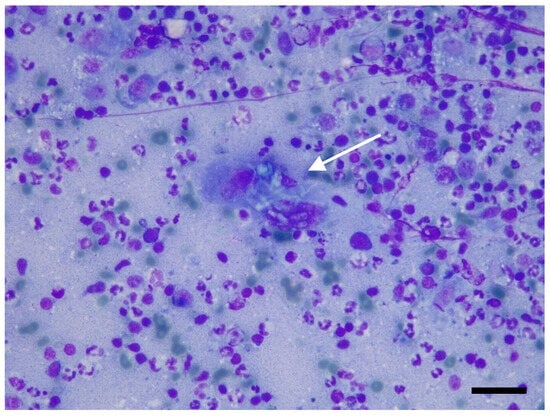
Figure 3.
Fine-needle aspiration from popliteal lymph node: neutrophilic lymphadenitis with multinucleated giant cell containing sinuous septate fungal hyphae (arrow). May–Grünwald Giemsa stain, original magnification 200×. Scale bar: 50 μm.
A diagnosis of systemic mycosis was reached and an antifungal treatment with itraconazole was initiated at 5 mg/kg orally BID. Gabapentin at 5 mg/kg PO TID was added to manage pain. After two weeks (day +105) of antifungal treatment, the owner reported that the dog was in good overall health, active, and had an improved appetite.
On day +135 (one month after the initiation of therapy), the patient’s clinical condition had significantly improved, including their total protein (7.2 g/dL) and β-2 fraction of serum proteins (1.3 g/dL) (Figure 1b). Despite the veterinarian’s recommendation to continue the therapy, the owner decided to discontinue its administration.
On day +165 (one month after the discontinuation of the therapy), the dog’s health condition deteriorated again, and she presented with neurological signs. The owner returned to the veterinary clinic, where the hematological and biochemical tests were repeated, revealing a new increase in the β-2 fraction (1.66 g/dL) and C-reactive protein (14.3 mg/dL). Following the onset of neurological clinical signs, a new treatment plan was implemented, with fluconazole at a dosage of 8 mg/kg BID. Fluconazole was chosen for its ability to cross the blood–brain barrier, although the drug is more commonly indicated for infections caused by yeasts. However, on day +175, the clinical and neurological condition of the dog had continued to worsen despite the antifungal treatment being resumed and the owner decided to proceed with euthanasia.
2.3. Necropsy
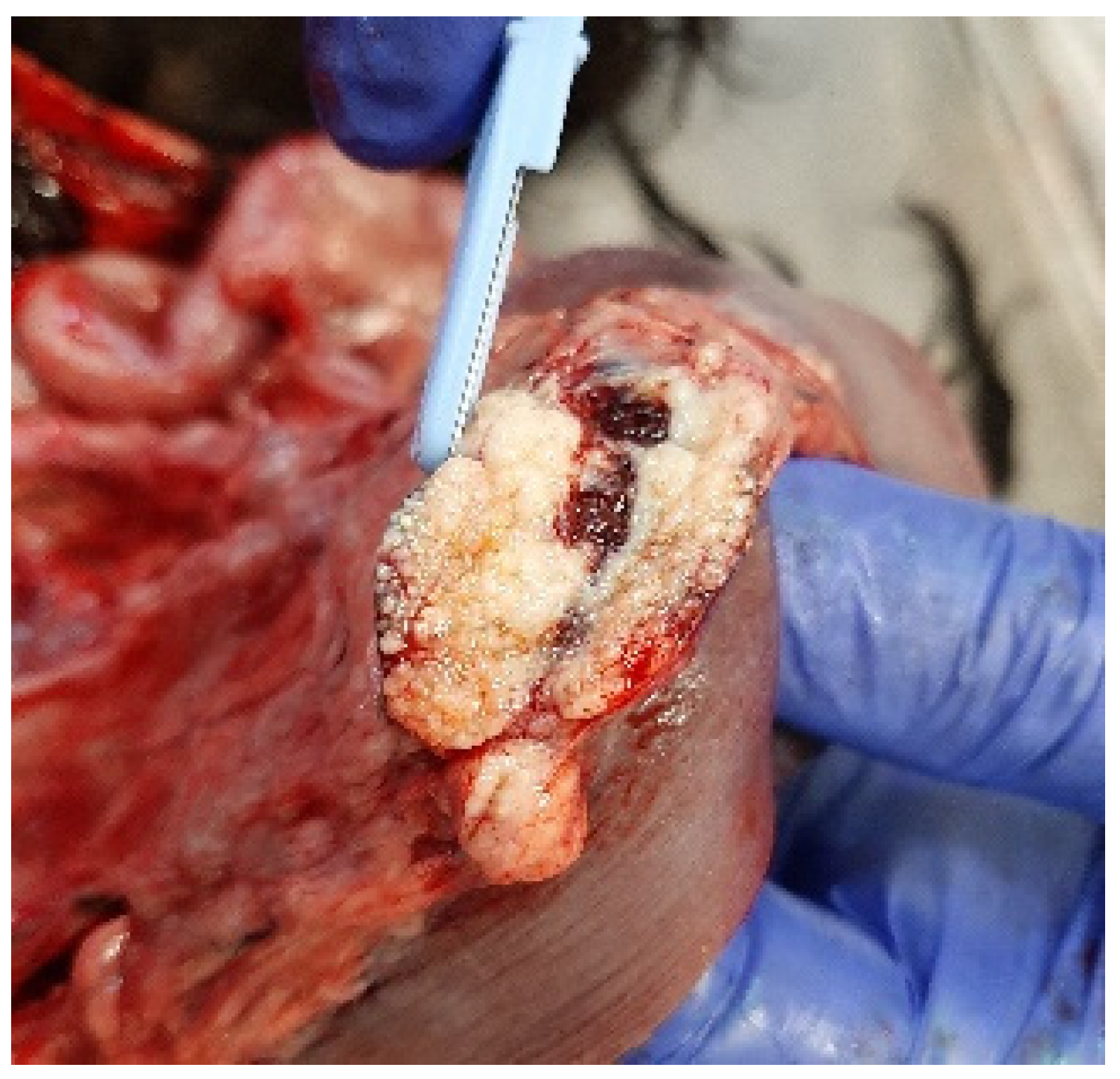
At necropsy, the dog was in poor physical condition, exhibiting widespread skeletal muscle hypotrophy. Gross examination revealed liver congestion with an enhanced lobular pattern, dilation of the right heart accompanied by a thinning of the right ventricular wall, and diffuse and irregular lymphadenomegaly that was most prominent in the periportal, epigastric, mesenteric, splenic, and retroperitoneal lymph nodes (Figure 4). The cross-section of the affected lymph nodes displayed irregular effacement due to miliary to coalescing white-yellowish lesions, occasionally with necrotic centres.
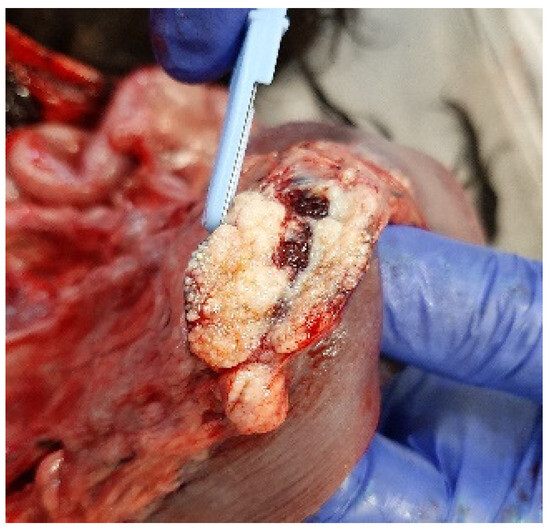
Figure 4.
Mesenteric lymph node effaced by miliary to coalescing white-yellowish lesions.
2.4. Histopathological Examination
The histopathological evaluation revealed diffuse vacuolar degeneration of the hepatocellular cords, with multifocal centrilobular necrosis in the liver; in the lymph nodes, characterized by cortical follicular and paracortical hyperplasia, severe, chronic, necrotizing, and granulomatous lymphadenitis was observed. The nodal lesions contained intralesional hyphae, ascospores, and asci within necrotic foci or within the cytoplasm of giant multinucleated histiocytes, consistent with ascomycetes (Figure 5a,b).
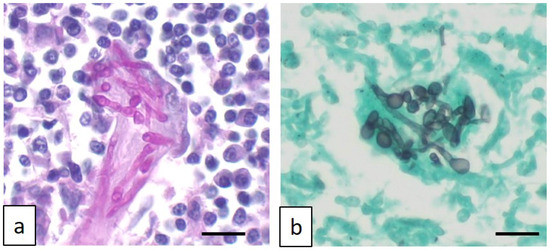
Figure 5.
Photomicrograph of affected lymph nodes characterized by multinucleated giant histiocytes engulfing fungal sinuous hyphae, as highlighted by Periodic acid–Schiff stain (a) and Grocott methenamine silver stain (b). Original magnification 400×. Scale bar: 50 μm.
2.5. Microbiological Investigation
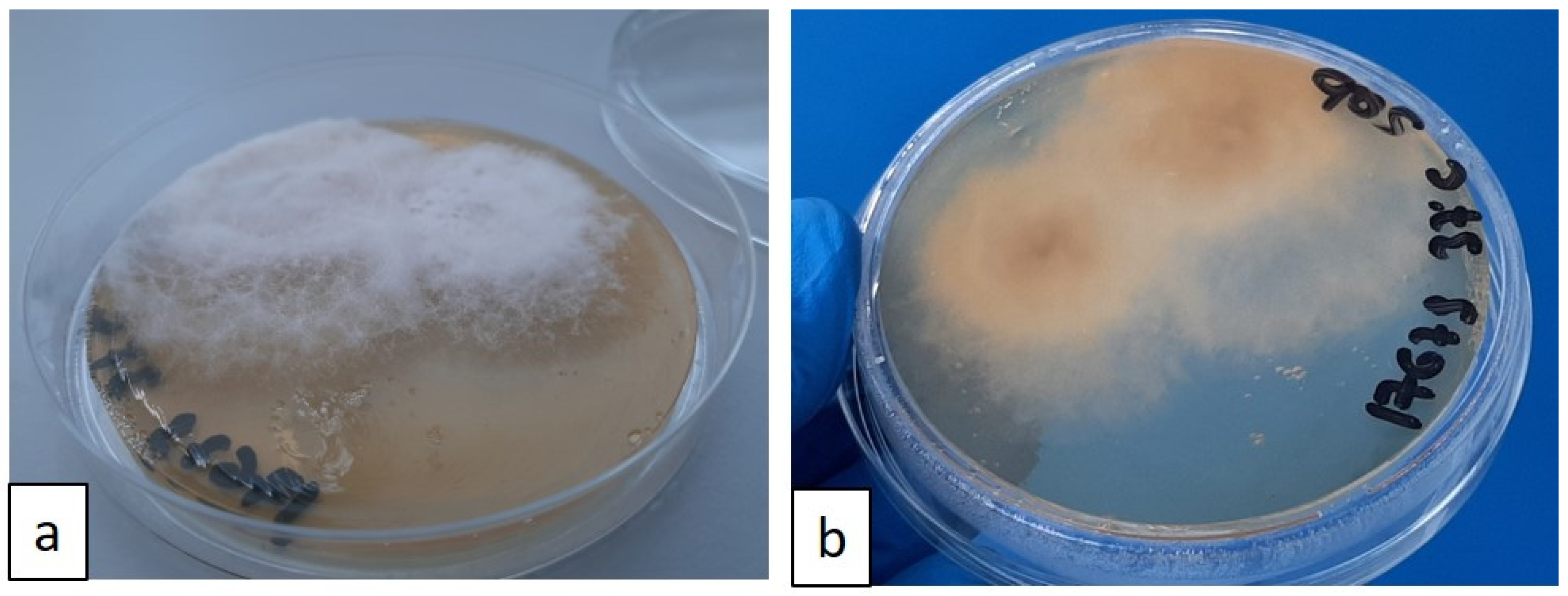
For the microbiological examination, a needle aspirate was obtained from the popliteal lymph nodes in vivo, and a post-mortem sample was taken from the mesenteric lymph nodes. The samples were preserved in e-Swab® (Copan Italia S.p.A-Brescia, Italy) and used for fungal culture analysis. The samples were plated on Sabouraud plate agar and soy agar with 5% sheep blood (BD Diagnostic Systems, Becton Dickinson GmbH-Heidelberg, Germany). The plates were incubated at 25 °C and 37 °C. After 72 h, a pure culture of a fine, filamentous white fungal growth was observed at both incubation temperatures. By the fifth day of incubation, the culture plates were completely covered by a grey woolly growth (Figure 6a,b).
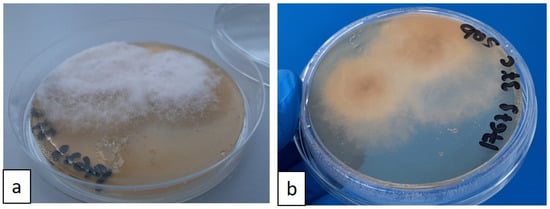
Figure 6.
(a,b) Sabouraud agar plate, recto–verso: growth of white, cottony fungal colonies after 5 days of incubation at 37 °C; fungal colonies tend to pigment over time.
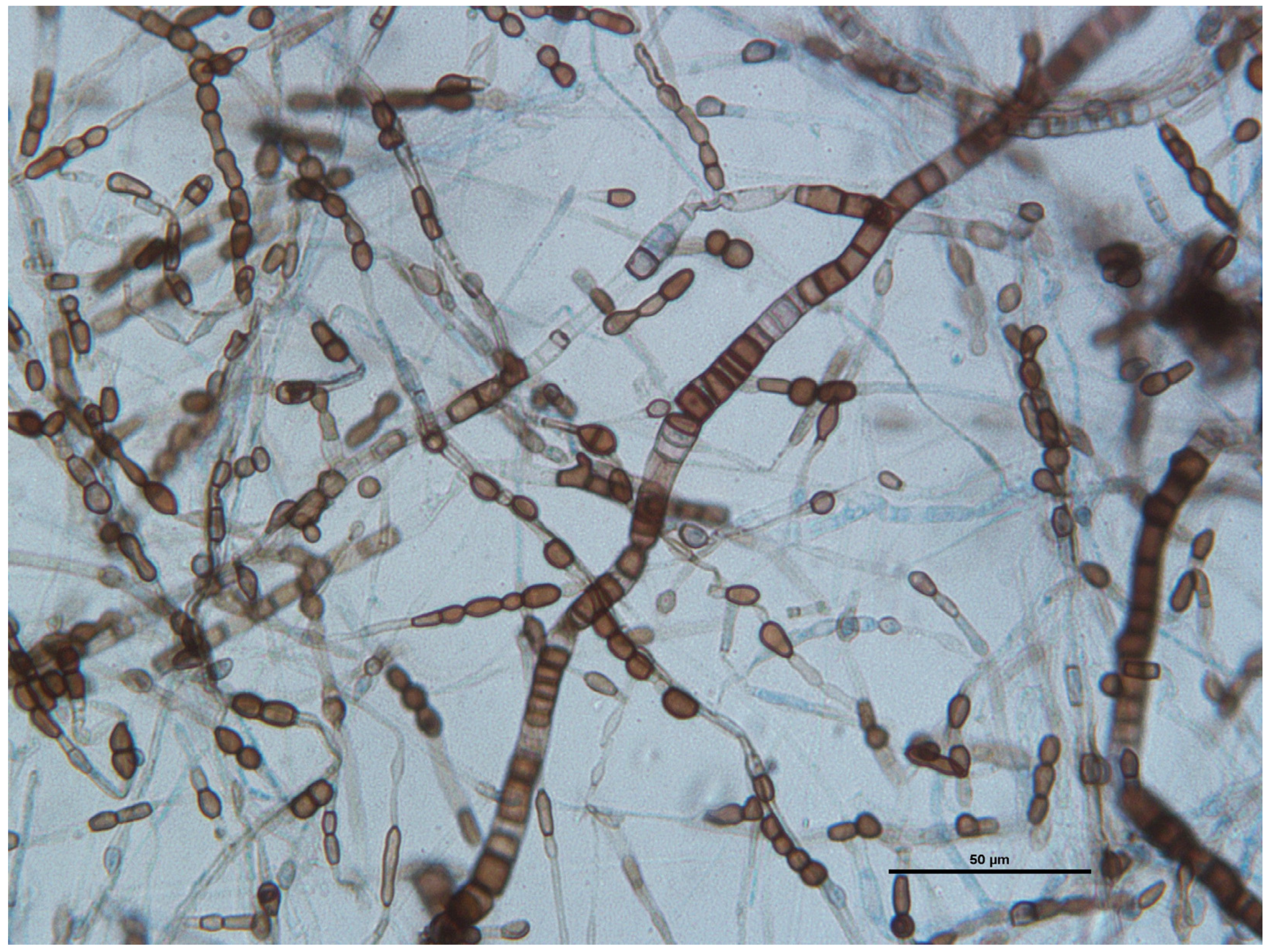
Microscopic examination of an adhesive tape preparation showed many wide and septate brown and hyaline hyphae and abundant melanized one-to-two-celled globose or barrel-shaped arthroconidia produced on the aerial mycelium (Figure 7).
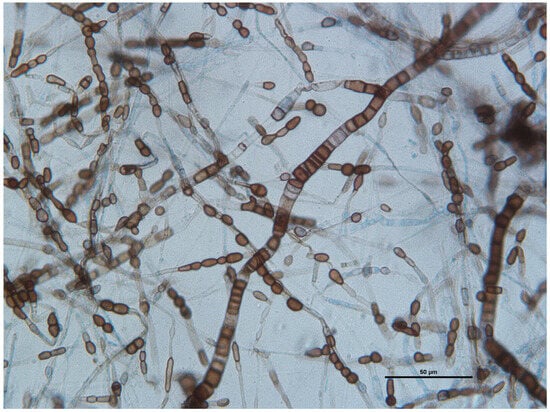
Figure 7.
Wet-mount preparations stained with lactophenol cotton blue: septate and branched hyaline to dark brown hyphae ranging from 2 to 8 mm, with uni- or bicellular arthroconidia measuring 4 μm by 8 μm. Scale bar: 50 μm.
The in vitro antifungal sensitivity testing was performed according to the Clinical and Laboratory Standards Institute—CLSI-M-38A2 microdilution method [4]. Antifungal activity was determined for amphotericin B, itraconazole, voriconazole and posaconazole. The minimum inhibitory concentration (MIC) for each of the four antifungal drugs tested in the present study were as follows: amphotericin B: 1 mg/L, itraconazole 4 mg/L, voriconazole 0.5 mg/L, and posaconazole 2 mg/L.
In order to characterize them from a molecular point of view, one colony was picked from the plate with a flocked swab and then DNA was extracted using a commercial kit (Maxwell RSC Blood DNA Kit, Promega Corporation, Madison, WI 53711, USA), with automated instrumentation (Maxwell RSC, Promega) following the manufacturer’s instructions and slight modifications in the form of a prolonged lysis incubation time. A negative sample, consisting of only reagents, was extracted simultaneously as a control for environmental contamination.
The DNA was then PCR-amplified using primers targeting the intergenic transcribed spacer (ITS) 1 and 2 regions [2,3]. The PCR products were then Sanger-sequenced and electrophoretically separated on an automated sequencer (ABI 310 Genetic Analyzer, Thermo Fisher Scientific, Waltham, MA 02451, USA). The sequences obtained were manually reviewed and blasted in the Genebank on the NCBI website (http://www.ncbi.nlm.nih.gov/BLAST/ (accessed on 18 May 2021)), revealing 95.46% homology with Scytalidium lignicola. Therefore, the isolated sample falls within the Scytalidium genus, but ITS region or genome sequencing is needed to improve this characterization. The negative sample did not reveal any sequences referable to Scytalidium lignicola.
3. Discussion
This report describes the clinicopathological changes, management, and outcome of a systemic Scytalidium spp. infection in a dog without known immunodeficiency. Scytalidium spp. are ascomycete fungi that are widely distributed phytopathogens primarily found in plants, fruit trees (such as lemon or banana trees), and soil [1]. The genus comprises over 15 species. Neoscytalidium dimidiatum and Scytalidium hyalinum have significant implications for human pathology [3].
N. dimidiatum and S. hyalinum can cause superficial and, less commonly, deep infections that resemble dermatophytosis, referred to as scytalidiosis. These filamentous fungi are endemic in tropical and subtropical regions like Asia, India, Africa, South America and the Caribbean, where they account for approximately 40% of dermatomycoses [5,6,7,8].
Scytalidium primarily affects the feet and nails of individuals walking barefoot on contaminated soils. Typically, infections remain localized to these areas; however, systemic forms characterized by central nervous system abscesses, sinusitis, osteomyelitis, mycetoma, and subcutaneous or disseminated lesions have been reported on rare occasions, especially in immunocompromised patients [3].
In cases of deep infections, N. dimidiatum is the most frequently implicated causative agent, while S. hyalinum has been found in only a few cases [9]. The prognosis of these infections is death in almost 50% of patients. There is no standardized therapy for Scytalidium spp. infections. Indeed, isolated fungal strains have shown high resistance to antifungal drugs commonly used in human and veterinary medicine [3]. In the present case, the fungal strain also demonstrated resistance to most antifungal agents used in human therapy.
Protein electrophoresis revealed a peak in the β-2 fraction. Proteins known to migrate to the β-2 fraction of serum include transferrin, β-2 lipoprotein, immunoglobulin M, immunoglobulin A, and positive acute-phase proteins such as C-reactive protein, complement factor 3a, and hemopexin [10]. Hypoalbuminemia can be considered a consequence of acute and/or chronic infection, considering that albumin is a negative acute-phase protein. Increased β-globulins can be observed in cases of acute or chronic inflammation [11,12]. Similar electrophoretic patterns have been observed in psittacine birds and penguins affected by aspergillosis [13,14], as well as in some case reports of infection caused by fungi other than Aspergillus [15].
Analogous electrophoretic alterations have been observed in some dogs affected by Dirofilaria immitis [16], Mesocestoides [17], and Angiostrongylus vasorum [18]. This dog had no history of travel to subtropical or endemic regions for scytalidiosis, no concomitant pathologies, and no history of immunosuppression, prolonged antibiotic use, or corticosteroid therapy. Additionally, the breed was not considered predisposed to opportunistic fungal infections [19]. The route of infection has not been identified, although the presence of more marked lymphadenomegaly in the intra-abdominal lymph nodes might suggest that the pathogen entered through the digestive tract. In other reported human cases, traumatic inoculation with plant material contaminated with the fungus has been suspected. To the best of the authors’ knowledge, this is the second report of a systemic infection caused by Scytalidium spp. in veterinary medicine. In the first report, which also had a fatal outcome [2], the infection affected an immunocompetent German shepherd, a dog breed known for its predisposition to systemic fungal infections [20].
4. Conclusions
Fungal diseases in animals are often insidious, diagnosed late, and sometimes only detected through specific diagnostic investigations. Systemic fungal infections should be considered possible differential diagnoses in dogs presenting with hyperbetaglobulinemia in their β-2 fraction. Early identification and prompt treatment of these infections are crucial to improving prognosis, as delayed diagnosis often leads to poor clinical outcomes.
Author Contributions
A.G.: Writing—review and editing, Writing—original draft, Conceptualization, Formal analysis. M.E.T.: Writing—review and editing, Writing—original draft, Conceptualization, Formal analysis. M.P.: Writing—original draft, Formal Analysis. F.G.: Writing—review and editing, Supervision. E.O.: Writing—review and editing. C.S.: Formal Analysis. S.N.: Writing—review and editing, Formal analysis. M.G.: Writing—review and editing, Writing—original draft, Formal analysis. F.M.: Writing—review and editing, Supervision. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Ethical review was not required for the study involving the animal, in accordance with local legislation and institutional requirements. The study was conducted for diagnostic purposes with the owner’s agreement. Written informed consent was obtained from the owner for the use of the animal’s samples in this study.
Informed Consent Statement
Not applicable.
Data Availability Statement
The original contributions presented in this study are included in the article. Further inquiries can be directed to the corresponding author.
Acknowledgments
The authors would like to express their gratitude to Marina Almeoni for her meticulous patient care, George Lubas for his support, and the laboratory technicians at I-Vet for facilitating the completion of this report.
Conflicts of Interest
Author Maria Elena Turba was employed by the company Genefast srl. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
- Fisher, M.C.; Henk, D.A.; Briggs, C.J.; Brownstein, J.S.; Madoff, L.C.; McCraw, S.L.; Gurr, S.J. Emerging fungal threats to animal, plant and ecosystem health. Nature 2012, 484, 186–194. [Google Scholar] [CrossRef] [PubMed]
- Dunlap, A.E.; Swinford, A.K.; Wells, K.L. Disseminated Scytalidium infection in a German shepherd dog. Med. Mycol. Case Rep. 2015, 7, 20–22. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Machouart, M.; Menir, P.; Helenon, R.; Quist, D.; Desbois, N. Scytalidium and scytalidiosis: Hat’s new in 2012. Med. Mycol. 2013, 23, 40–46. [Google Scholar] [CrossRef] [PubMed]
- CLSI M38-A2; Reference Method for Broth Dilution Antifungal Susceptibility Testing of Filamentous Fungi; Approved Standard-Second Edition. Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2008.
- Fujita, S.I.; Senda, Y.; Nakaguchi, S.; Hashimoto, T. Multiplex PCR using internal transcribed spacer 1 and 2 regions for rapid detection and identification of yeast strains. J. Clin. Microbiol. 2001, 39, 3617–3622. [Google Scholar] [CrossRef]
- Bellemain, E.; Carlsen, T.; Brochmann, C.; Coissac, E.; Taberlet, P.; Kauserud, H. ITS as an environmental DNA barcode for fungi: An in silico approach reveals potential PCR biases. BMC Microbiol. 2010, 10, 189. [Google Scholar] [CrossRef]
- De Hoog, G.S.; Guarro, J.; Gene, J.F.M.J.; Figueras, M.J. Atlas of Clinical Fungi, 2nd ed.; Centraalbureau Voor Schimmelcultures: Utrecht, The Netherlands, 2004. [Google Scholar]
- Hay, R.J. Scytalidium infections. Curr. Opin. Infect. Dis. 2002, 15, 99. [Google Scholar] [CrossRef] [PubMed]
- Zaatari, G.S.; Reed, R.; Morewessel, R. Subcutaneous Hyphomycosis Caused by Scytalidium hyalinum. Am. J. Clin. Pathol. 1984, 82, 252–256. [Google Scholar] [CrossRef] [PubMed]
- Ritzmann, S.E.; Daniels, J.C. Serum Protein Abnormalities: Diagnostic and Clinical Aspects; Little, Brown: New York, NY, USA, 1982. [Google Scholar]
- Ceron, J.J.; Eckersall, P.D.; Martínez-Subiela, S. Acute phase proteins in dogs and cats: Current knowledge and future perspectives. Vet. Clin. Pathol. 2005, 34, 85–998. [Google Scholar] [CrossRef]
- Moore, A.R.; Avery, P.R. Protein characterization using electrophoresis and immunofixation; a case-based review of dogs and cats. Vet. Clin. Pathol. 2019, 48, 29–44. [Google Scholar] [CrossRef] [PubMed]
- Cray, C.; Reavill, D.; Romagnano, A.; Van Sant, F.; Champagne, D.; Stevenson, R.; Rolfe, V.; Griffin, C.; Clubb, S. Galactomannan assay and plasma protein electrophoresis findings in psittacine birds with aspergillosis. J. Avian Med. Surg. 2009, 23, 125–135. [Google Scholar] [CrossRef] [PubMed]
- Desoubeaux, G.; Cray, C.; Chesnay, A. Challenges to establish the diagnosis of aspergillosis in non-laboratory animals: Looking for alternatives in veterinary medicine and demonstration of feasibility through two concrete examples in penguins and dolphins. Front. Cell. Infect. Microbiol. 2022, 12, 757200. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Erne, J.B.; Walker, M.C.; Strik, N.; Alleman, A.R. Systemic infection with Geomyces organisms in a dog with lytic bone lesions. J. Am. Vet. Med. Assoc. 2007, 230, 537–540. [Google Scholar] [CrossRef] [PubMed]
- Cavalera, M.A.; Paltrinieri, S.; Giordano, A.; Iatta, R.; Gernone, F.; Mendoza-Roldan, J.A.; Gusatoaia, O.; Otranto, D.; Zatelli, A. Serum protein electrophoresis in Dirofilaria immitis naturally infected dogs: Latest news and a systematic literature review. Vet. Parasitol. 2022, 305, 109720. [Google Scholar] [CrossRef] [PubMed]
- Spina, F.; Rossi, S.; Spattini, G. Cestodiasi peritoneale in un cane del centro Italia. Veterinaria 2016, 30, 307–310. [Google Scholar]
- Bertazzolo, W.; Didier, M.; Ridolfi, M.; Venco, L. Detection of a characteristic beta-2 peak in serum by capillary zone electrophoresis in dogs with Angiostrongylus vasorum. Vet. Clin. Pathol. 2022, 51, 70–76. [Google Scholar] [CrossRef] [PubMed]
- HSVMA. Congenital and Heritable Disorders Guide; HSVMA: Davis, CA, USA, 2012. [Google Scholar]
- Elad, D. Disseminated canine mold infections. Vet. J. 2019, 243, 82–90. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).