Abstract
Plants in the Oxytropis genus can live with the endophytic fungi Alternaria sect. Undifilum. Swainsonine, the mycotoxin produced by the endophyte render the host plant toxic and this has been detrimental to grazing livestock in both China and U.S.A. Despite previous efforts, many questions remain to be solved, such as the transmission mode and life cycle of host–endophyte symbiont, the biosynthesis pathway of swainsonine, and in particular the ecological role and evolution of such symbiosis. In this review, we compile the literature to synthesize ideas on the diversity of the symbiosis and propagation of the endophyte. We further compare the previous work from both Alternaria sect. Undifilum and other swainsonine producing fungi to orchestrate a more comprehensive biosynthesis pathway of swainsonine. We also connect swainsonine biosynthesis pathway with that of its precursor, lysine, and link this to a potential role in modulating plant stress response. Based on this we hypothesize that this host–endophyte co-evolution originated from the needs for host plant to adapt for stress. Validation of this hypothesis will depend on future research on endophytic symbiosis in Oxytropis and help in better understanding the roles of plant–endophyte symbiosis in non-Poaceae species.
1. Introduction
Locoweeds are forbs from the Oxytropis and Astragalus genus that are poisonous to grazing animals. The name (“loco” means crazy in Spanish) is historical as the consumption of locoweeds was observed to cause abnormal behavior in sheep and horses on rangelands of the U.S.A. [1,2,3]. Later on, several studies determined that the main toxic agent in locoweeds is swainsonine, an indolizidine alkaloid, which inhibits alpha-mannosidase II and causes neurotoxic symptoms and other pathogenic changes in animals [4,5,6,7]. Currently, locoweeds are the most problematic weeds on the rangelands in the U.S.A. and China [8,9]. An outbreak of locoweeds not only causes losses to the livestock industry, but also creates ecological problems because they can easily become dominant in rangeland plant communities [10,11,12]. This is particularly true in China as overstocking has been increasing over years due to the demand for meat consumption. Despite of extensive research into locoweeds focusing on biological, ecological, and toxicological aspects, currently there is no effective way to prevent locoweeds outbreaks.
Toxins in poisonous plants are not always produced by the plants themselves. Secondary metabolites caused animal poisoning by Lolium perenne aroused the research interests in plant–endophyte symbiosis. Endophyte symbiosis was previously considered to be mutualistic. This arose from early studies carried out in Poaceae, in which the symbionts formed with Epichloë (anamorph Neotyphodium) species produce toxic alkaloids and help the hosts deter herbivores and increase stress tolerance [13,14,15]. Numerous studies have focused on understanding the production and biosynthesis of swainsonine in locoweeds. Although initially recognized as a plant derived toxin, swainsonine was proved to be produced by endophytic fungi in locoweeds [16]. Different varieties of swainsonine-producing endophytes were isolated from locoweeds in the U.S.A. and China, and phylogenetic studies showed that they all belong to a redefined lineage as Alternaria sect. Undifilum [17,18,19]. The symbiosis between locoweeds and A. sect. Undifilum were considered most likely to be commensal (reviewed by Creamer and Baucom [20]), as despite toxins are produced, locoweeds do not have obvious herbivore deterrence to insects and grazing animals. In addition, the symbiosis also does not provide remarkable stress tolerance or growth promoting effects to the hosts. However, recent studies showed that A. sect. Undifilum might help their locoweed hosts against pathogens [21], or regulate the endophytic fungi assemblage [22]. Therefore, the ecological role of locoweed symbiosis awaits further investigation.
In this paper, we reviewed the classic studies of locoweeds to consider the latest development in our understanding in the relationships between host, endophyte, and swainsonine in locoweeds. We synthesized and compiled previous efforts with a focus to understand the diversity, and evolutionary and ecological significance of such symbiosis. As most locoweeds in China are Oxytropis species, and there have been many studies from the U.S.A. focusing on Astragalus locoweeds, we have particularly emphasized on research in Oxytropis locoweeds. This will also bring to an international audience locoweed papers that have been published in Chinese.
1.1. Distribution, Diversity, and Abundance of Oxytropis spp. and Alternaria sect. Undifilum
Oxytropis species are perennial forbs or semi-shrubs which belong to Fabaceae. There are approximately 310 Oxytropis species worldwide, mainly in Eurasia and America, within which 133 species (74 endemic) were documented in China [23]. Common Oxytropis locoweeds include O. sericea and O. lambertii in the U.S.A., which spread extensively in rangelands in western America, and causes animal poisoning in grazing livestock [24]. In China, Oxytropis locoweeds are more diverse, with the common ones being O. glabra, O. ochrocephala, O. kansuensis, O. glacialis, O. sericopetata, O. deflexa, O. hirta, O. falcate, etc. (Table 1). Those species can be found in all the four types of grasslands in China spanning Xinjiang, Gansu, Ningxia, Qinghai, Tibet, and Inner Mongolia (representing meadow steppe, typical steppe, desert steppe, and alpine steppe terrains [25]).

Table 1.
Species and distribution of poisonous Oxytropis spp. in the natural grasslands of the Western United States and China.
Until recently, the taxonomic classification of the swainsonine producing endophytes in Oxytropis locoweeds was ambiguous. Braun et al. first isolated an endophyte from three locoweed populations in New Mexico and named it Alternaria sp. (Pleosporaceae) according to their morphological characteristics but rectified it to Embellisia sp. [19]. Wang et al. isolated a strain from O. kansuensis in China and found it to be similar to the one described by Braun, and named it Embellisia oxytropis [31]. In 2009, Pryor et al. combined Embellisia sp. and Helminthosporium bornmuellerii into Undifilum sp. [18]. In 2013, Woudenberg et al. constructed a phylogenetic tree across three loci (GAPDH, RPB2 and TEF1), and moved Undifilum to a section of Alternaria [17]. Therefore, U. oxytropis, the most widely used name for the endophyte in Oxytropis locoweeds, was changed to Alternaria sect. Undifilum and this designation had been used thereafter (Table 2). The genetic variation of the A. sect. Undifilum conspecies is shown in Figure 1.

Table 2.
Formally recognized Oxytropis A. sect. Undifilum associations and their typical location.

Figure 1.
Maximum Likelihood phylogenetic tree constructed based on ITS sequences of 24 strains representing the swainsonine producing endophytes. The purple dots indicate fungi were isolated from the Oxytropis genus, and the green ones indicate the Astragalus genus. Metarhizium robertsii and Arthroderma otae, which also produce swainsonine, were used as the outgroup and were indicated in blue and orange, respectively. Bootstrap (1000 replicates) values were displayed.
Endophyte colonization was found in various parts of the hosts in Oxytropis, and particularly in leaves and stems compared to roots during vegetative growth [37]. Eight Embellisia spp. were isolated from 11 locoweed populations in the U.S.A. The infection rates range were between 100% and 93.1% in flowers and seeds, and between 72.9% and 97.2% from leaves and stems [19]. During host reproduction, the endophyte is enriched in flowers, and this aids next generation transmission generation via the seed. Within the seed the endophyte is localized to the inside the seed coat (testa), and specifically in the endothelium parenchyma and aleurone layer [42]. No endophytes have been reported within the embryo and endosperm. Thus, the endophyte is transmitted through maternal material only, presumably from the integument in the ovary. Outside the seed, mycelium has been found between the intercellular spaces, and is most abundant in meristem rather than mature tissues [43,44]. This might be due to an efficiency in utilizing xylose and other polysaccharides [45]. In this context, it is also worth noticing that endophyte levels decrease in underground compared to aboveground tissues [46] (Figure 2). Therefore, it was speculated that locoweed endophytes may prefer to live in tissues with stronger photosynthetic activity and higher lysine levels [34], similar to the evidence demonstrated in grass and other plant species [47,48]. It is possible that the endophyte may preferentially infect certain tissue types or are distributed in a gradient as the plant grows from a seed to maturity plant.
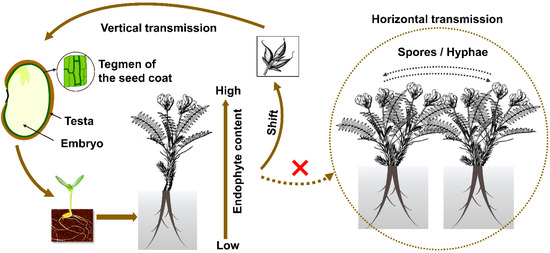
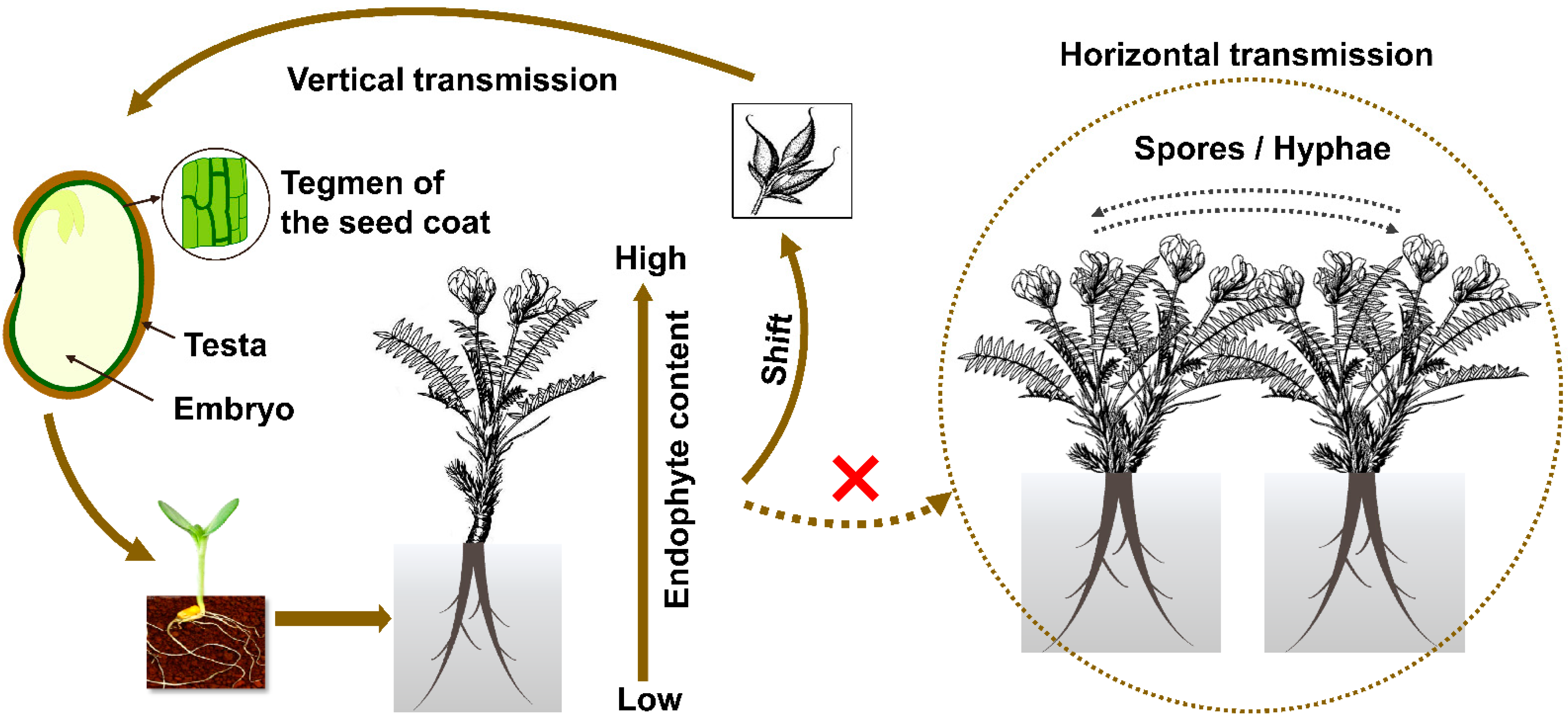
Figure 2.
Model of transmission in Oxytropis locoweeds. Dotted line and the cross mark in red indicated horizontal transmission has not been found in the field for such symbiosis.
Swainsonine is produced only by A. sect. Undifilum conspecies and many studies have aimed to identify biotic and abiotic factors that influence swainsonine production in the hosts. To address this, it is crucial to have accurate and effective detection methods of locoweed endophytes, both qualitatively and quantitatively. For qualitative detection, traditional approaches are based on plant culturing followed by fungal isolation from aseptic plant tissues [49,50] and remain effective for the morphological and molecular identification of the abundant and culturable endophyte taxon. Endophytes, including A. sect. Undifilum could be successfully cultured from plant leaf material from locoweeds [21]. This strategy can be supplemented by microscopic approaches to observe endophyte in planta. Endophytes can be viewed by aniline blue staining or electron microscopy [51]. Reyna et al. [44] later used scanning electron microscopy (SEM) and transmission electron microscopy (TEM) to localize and characterize the growth patterns of the fungus Undifilum oxytropis in O. lambertii and O. sericea. In their work, green fluorescence protein (GFP) transformed fungal strains were also successfully used to observe the colonization. Alternatively, endophytes could be cultured from plant material and PCR were employed for further identification of A. sect. Undifilum. For example, the ribosomal internal transcribed spacer (ITS) and β-tubulin were used as targets for amplification and identification A. sect. Undifilum endophyte in O. sericea and O. lambertii [19].
However, as many fungi could not be successfully cultured in vitro, PCR-based molecular identification without fungal culture has become more popular in laboratories. Gene specific primers can amplify regions targeting a specific fungus. This was first used successfully in Festuca by Doss et al. [52]. Based on the ITS region, which was the basis of locoweed endophyte specific primers OR1/ITS5 [27], Cook et al. developed a quantitative read-time (qPCR) method for the determination of the relative amount of endophytes and tested the method across three locoweeds in U.S.A [35] with a detection limit of 0.2 pg/ng (endophyte/plant DNA). This method has been used effectively in many locoweed species in both U.S.A. and China. A newly designed primer set, Omtssu F/Omtssu R was also tested and compared with OR1/ITS5 showed higher specificity and only amplified from locoweed endophyte DNA [53]. In our group, qPCR testing 19 Oxytropis species showed that the OR1/ITS5 had specificity in 17 species (data not shown) but not in O. ochrocephala and O. kansuensis, two morphologically close species. In O. ochrocephala OR1/ITS5 annealed to a chloroplast sequence with similar size, which will skew the data [54]. Thus, we suggest that using all the available qPCR methods after validation in the Oxytropis species being tested to avoid a lack of specificity. New primers could be developed based on genes on the swainsonine biosynthesis pathway (as in the SwnK cluster, see below) [55]. Alternatively, high-throughput sequencing could offer another way of measuring the relative abundance of locoweed endophyte based on distinguishing reads and counting reads hits. The full genome of A. sect. Undifilum has been studied that could help in providing a reference genome in amplicon sequencing.
1.2. Propagation and Reproduction of A. sect. Undifilum
Studies of the lifestyle of plant endophytes were greatly facilitated by the advance of microscopy. In locoweeds studied so far in U.S. and China, the precise location of A. sect. Undifilum within leaves and petioles is vascular bundle and pith portion, respectively. The hyphae live within cell walls but do not penetrate host cells, and no haustoria were observed. This suggests that A. sect. Undifilum is not pathogenic but most likely commensal because there are neither marked advantages when this symbiosis is formed comparing with the endophyte free plants [20,43].
Rodriguez and coworkers [56] classified endophytes into four groups based on their transmission and ecological interactions. Class 1 endophytes are those that infect a narrow host range, are generally vertically transmitted, and produce mycotoxins. Grass endophytes Epichloe festuca and Neotyphodium sp. fit within that grouping. Class 2 endophytes have a broad host range and are vertically and horizontally transmitted. Class 2 endophytes include Phoma, Colletotrichum sp., Fusarium sp., and Curvularia sp. Class 3 and Class 4 endophytes infect broad host ranges, are horizontally transmitted, and infect shoots and roots, respectively [20]. Unlike many other saprophytic endophytes that can generate spores, conidia of A. sect. Undifilum were never observed in Oxytropis plants in the past. A. sect. Undifilum do not generate gamobia with stroma and are vertically transmitted via seeds (Figure 2). The vertical transmission rate is usually not 100%, indicating the loss of infection through generations [32,57]. Horizontal transmission has never been recorded in the field for the last 20 years, and therefore A. sect. Undifilum are presumed to be Class I endophytes.
In the laboratory surface-sterilized seeds following seed coat removal could be inoculated with an endophyte culture, but conidia from A. sect. Undifilum produced in culture do not infect plants. This could be attributed to that the plant does not recognize the fungus as a pathogen, nor does the fungus act like a pathogen, or puncture cell walls. This contrasts with the pathogenic A. astragali (A. gansuense comb. nov.) isolated from standing milkvetch (Astragalus adsurgens) which cause yellow stunt and root rot [53]. The different life history suggests that swainsonine producing A. sect. Undifilum in Oxytropis plants is not a pathogen. Recently, some exploratory work from Li’s group in Lanzhou University showed evidence of some previously unidentified life strategy of A. sect. Undifilum [58]. Firstly, in O. glabra and O. ochrocepala, hyphae grew out from petiole that was mechanically wounded, and the hyphae covered leaflets later in all out of the 26 plant leaves. Secondly, spores were observed from the plant stem cutting tissue cultured on plates. Nevertheless, whether those results could represent an unidentified phenomenon in the field is still unknown.
Beyond such board classifications, it is difficult to explain why the Oxytropis genus contain both toxic and non-toxic species. It is not difficult to understand that toxic and non-toxic accessions/landraces/individuals were found in the same species, such as in O. lambertii [36], because random loss of the endophyte during transmission occurs, as we discussed above. However, considering the speciation event under the genus, several species were fixed with no symbiotic A. sect. Undifilum whereas the other toxic species do. Indeed, toxin producing and non-producing species can be located in the same geographical area (personal observation). Since no vertical transmission was observed, naturally the non-toxic species would not be able to acquire such symbiosis and become toxic. This must indicate that in the non-toxic species, the endophyte was lost due to either selection in the common ancestor or genetic drift happened in parallel. In either case, genetic variation at certain loci might have significant impact on the symbiotic colonization. Nonetheless, it could not be ruled out that A. sect. Undifilum used to be saprophytic in evolution. On the other hand, whether A. sect. Undifilum is present in the soil remain unambiguous. Lu et al. [21] isolated several fungi from the soil locoweeds grew, and although those fungi produce swainsonine they did not belong to A. sect. Undifilum. It would be more precise to confirm the presence or absence of A. sect. Undifilum in the rhizosphere soil of Oxytropis plants using metagenomics or amplicon sequencing.
1.3. The Endophyte and the Biosynthesis of Swainsonine
Studies indicate that swainsonine is produced by the A. sect. Undifilum endophytes rather than the host plants of Oxytropis [19,57]. Toxic Oxytropis species all contain A. sect. Undifilum conspecies, and in vitro culture of those A. sect. Undifilum was able to produce swainsonine [19,27,30,57]. Swainsonine could not be detected (nil or below 0.001% w/w) in Oxytropis plants from seeds with the seed coat removed or whole seeds treated with fungicide [42,59]. In line with this, the aseptic Oxytropis seeds, which do not contain detectable swainsonine, when inoculated with A. sect. Undifilum, produced swainsonine (>0.01% w/w) [59]. In some cases, the concentration of swainsonine in the plants was comparable to the amount of the A. sect. Undifilum endophyte. Indeed, “SD mice” showed similar clinical and pathological symptoms after being fed with A. sect. Undifilum containing feed and as with dried Oxytropis plants [33].
This showed that the relationship between the A. sect. Undifilum endophyte and swainsonine is complex. For example, whilst A. sect. Undifilum was detected by qPCR in Oxytropis [54] or Astragalus samples [27], no detectable swainsonine was detected. Conversely, the endophyte could not be isolated from swainsonine-containing O. latibracteata Jurtz. samples (unpublished data). Additionally, A. sect. Unidifilum minus swainsonine producing Oxytropis samples could be reflecting the contributions by other fungal species. Swainsonine producing fungal species other that A. sect. Undifilum include Metarhizium robertsii (and M. anisopliae), Slafractonia leguminicola (previously known as Rhizoctonia leguminicola), and Arthroderma otae [60]. The above findings suggest that the biosynthesis of swainsonine in fungi is probably conserved in some lineage of symbiosis relationship.
Such observations naturally pose the question of the role of swainsonine for fungal species. In both plants and animals, the infectivity of fungi to their hosts could not be related to swainsonine production. In fact, swainsonine producing strains showed slightly less virulence than the knockout mutant. Inoculation experiments showed that by M. robertsii infection did not produce detectable swainsonine in maize (Zea mays), soybean (Glycine max), and milkvetch (Astragalus sinicus) [61]. Thus, the production of swainsonine is not necessary for colonization of those characterized fungi. It is worth noticing that although Astragalus and Oxytropis species are often colonized by A. sect. Undifilum and yield swainsonine, the infection of M. robertsii, which also produce swainsonine, did not trigger the swainsonine production in A. sinicus. This suggests that the fungi–host symbiosis to produce swainsonine are species specific, regardless of the diverse fungal lineage to produce the toxin.
1.4. The Biosynthesis Pathway of Swainsonine
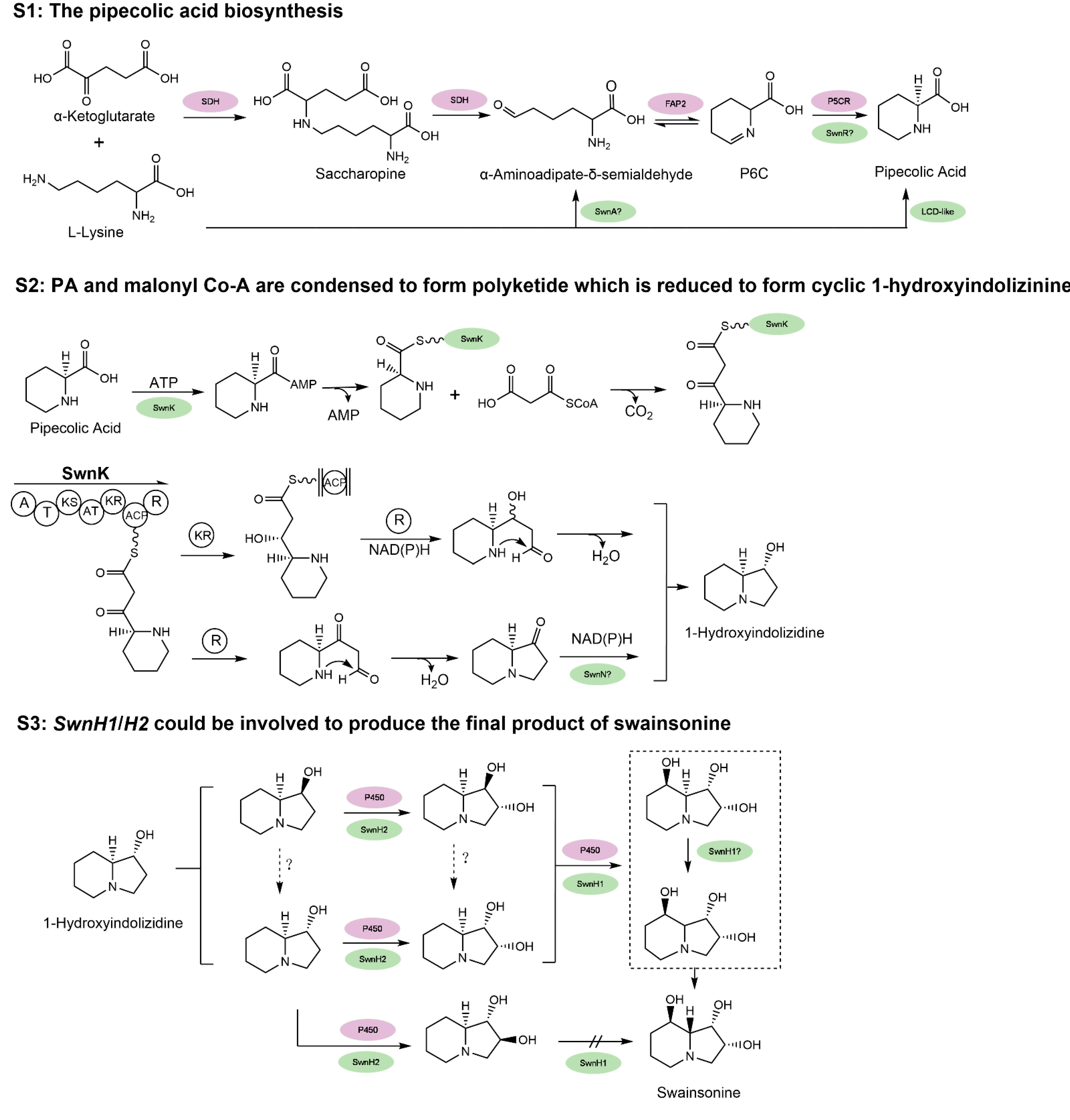
Despite its role as a locoweed toxin, the biosynthetic pathway of swainsonine has yet to be conclusively resolved. As swainsonine can be produced by several fungi that are not closely related phylogenetically, its biosynthetic pathway may have a deep evolutionary history. Based on previous work (see review by Tan et al. [62]), the pathway is arbitrarily divided into three stages (Figure 3). At stage 1 (S1), PA (pipecolic acid) is produced from lysine. At stage 2 (S2), PA and malonyl Co-A are condensed to form polyketide which is reduced to form cyclic 1-hydroxyindolizinine. Then, at stage 3 (S3), this direct precursor forms swainsonine after redox reactions. Several key steps in the biosynthesis pathway of swainsonine have been characterized in S. legunimicola and M. anisopliae. Additionally, a comparative genome sequencing revealed a multiple gene cluster, SWN and its orthologs as producing swainsonine [60]. The SWN cluster encodes seven putative genes, including swnA (an aromatic amino transferase gene), swnH1 and swnH2 (2-oxoglutarate- and Fe(II)-dependent dioxygenases gene), swnN (an NmrA-like, NADB Rossmann-fold reductase gene), swnR (an NADB Rossmann-fold reductase gene), swnT (a transmembrane transporter gene), and swnK (a multifunctional protein gene), which contribute to swainsonine production.
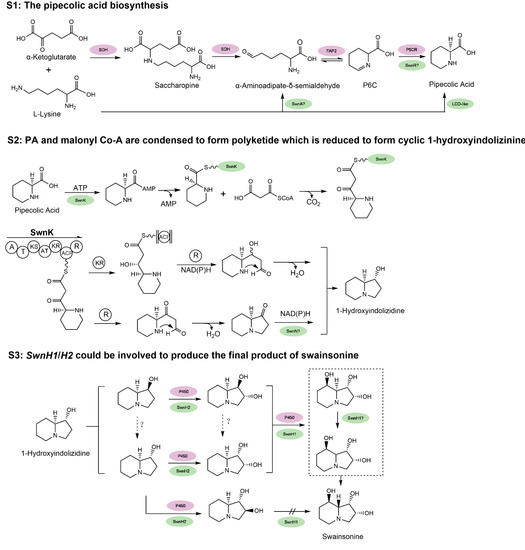
Figure 3.
The hypothesized biosynthesis pathway of swainsonine. S1: The multiple proposed biosynthesis pathways of pipecolic acid (PA). S2: PA and malonyl Co-A are condensed to form polyketide which is reduced to form cyclic 1-hydroxyindolizinine. S3: 1-hydroxyindolizinine forms SW after redox reactions. For comparison, genes in purple indicate all the annotated genes with proposed function and position from the locoweed endophytic fungus A. oxytropis, based on its genome sequencing [63]; genes in green represent the proposed and some verified genes in M. robertsii, and “?” indicates that the function and position of activity of the gene still need further characterization [60,61]. Refer to the text for the name of the genes. Certain chemical structures and reactions in the schematics were adapted from previous work by Cook et al. and Luo et al. [60,61].
At S1, the first crucial precursor for swainsonine production is PA. In prokaryotes, PA can be synthesized at least by four possible routes [64]. Three of them employ LKR/SDH (lysine-ketoglutarate reductase/saccharopine dehydrogenase, not necessarily linked), LAT (lys aminotransferase), and LysDH (lysine dehydrogenase), respectively, to produce α-aminoadipate-δ-semialdehyde (AASA). AASA can spontaneously convert to P6C, which is further reduced by a P5CR (1-pyrroline-5 carboxylate reductase). The fourth path is AASA independent, which uses racemase, aminotransferase, and reductase to produce Δ1-piperidine-2-carboxylate (P2C) and then to PA. Currently it is not clear if such multiple pathways are all present in fungi. It was suggested that in yeast, lysine can be degraded at least by transaminase(s) and lysine dehydrogenase [65]. For swainsonine biosynthesis, Wickwire et al. [66] suggested PA is produced via the lysine → saccharopine → P6C (1-piperideine-6-carboylate) → PA but not the P2C route.
Some progress has been made to help understand PA formation during swainsonine biosynthesis. Continuing from the previous identification of the SWN cluster, Luo et al. [61] further studied its function in M. robertsii. The disruption of swnA decreased PA by five-fold and therefore it should play an important role in the production of PA. Since swnA was annotated as an aminotransferase, it may function similar to the bacteria LAT (Lys aminotransferase) in PA formation, which catalyze the direct transamination between L-lysine and α-ketoglutarate to form α-aminoadipate-δ-semialdehyde (AASA) and L-glutamate. However, this would skip the intermediate saccharopine suggested by Wickwire et al. [66]. SwnR and SwnN both encode reductase, and Luo et al. showed that in the knockout mutant of swnR, a considerable amount (~3/4) of PA was still produced compared with that of the WT. In the case of a swnN knockout, more PA was produced than the WT. This would suggest that swnR is more likely to be working during PA formation. It is not known if it encodes a P5CR like gene. Rather swnN would act after PA formation and its malfunction caused PA unable to be converted to downstream metabolites. Since the disruption of swnA or swnR could still produce PA, it was thus believed that another PA biosynthesis pathways exists [61]. An LCD (lysine cyclodeaminase)—similar to the RapL from the rapamycin gene cluster in Streptomyces hygroscopicus [67]—involved pathway was verified by that one copy of the two LCD genes in M. robertsii could produce substantial PA [61]. It is worth mentioning that swnA mutant almost abolished swainsonine production but given that PA was still there via a possible redundant route, we suggested that the route that swnA is involved is responsible for the main production of swainsonine and must also play a regulatory role to other downstream genes for swainsonine biosynthesis.
At S2, both Cook et al. [60] and Luo et al. [61] proved that interruption of the swnK gene resulted in no detectable swainsonine. The multifunctional swnK therefore plays a key role as a PKS (polyketide synthase) at S2 to form the first intermediate eight-carbon cyclic precursors containing a C3=N+4 iminium ion. Evidence from Luo et al. [61] suggested depending on whether the polyketide complex is reduced once (R) or twice (KR and R) by the swnK reductase domains, next, the formation of 1-hydroxyindolizine can be swnN dependent or independent. It is not clear how the C3=N+4 iminium ion could be reduced to form 1-hydroxyindolizidine. Since swnN significantly reduced swainsonine production it may work at this stage as suggested.
At S3, from 1-hydroxyindolizidine, SwnH2 and H1 may catalyze sequentially the two hydroxylation steps to produce the final product of swainsonine, as its production was abolished by the disruption of either of them. Since many isomeric intermediates and substrate specificity have been found during these steps, it remains unclear how exactly SwnH2 and H1 elaborate at S3. SwnT, as predicted, is indeed involved in mediating the secretion of SW. Since deletion of swnA, swnK, and swn H1&2 made no detectable SW, they represent the key genes in SW biosynthesis. SwnN and swnR knockouts also yielded significant deceased amount of SW (10-fold and 5-fold, respectively), and thus should function in the main swainsonine pathway. The exact function and position of the several SWN genes remain to be elucidated.
Despite the progress in S. legunimicola and M. robertsii, the swainsonine biosynthesis pathway is unclear in A. sect. Undifilum. Experimental evidence is scarce. Mukerjee et al. [68] cloned a saccharopine reductase gene from U. oxytropis, and found the deletion mutant had decreased levels of saccharopine and lysine but increased level of PA and swainsonine. However, when Li and Lu [69] knocked out the homologous saccharopine reductase gene in A. oxytropis there were decreased levels of swainsonine and the levels of lysine were unaffected. This equivocal situation has yet to be resolved and, to date, no other genes have been clearly established in the proposed swainsonine pathway.
The genome of A. sect. Undifilum conspecies has been sequenced by two groups. Lu et al. [63] performed a ~56X depth sequencing using the Illumina Hiseq platform and assembled a 70.05 megabases (Mb) draft genome of a U. oxytropis isolated from China. Cook et al. [60] used an Illumina MiSeq platform to establish a ~78X fold depth of sequencing and reported an assembly of a 112.67 Mb A. oxytropis genome. The differences of genome size may be due to the two conspecies in A. sect. Undifilum represent different species/strains or heterozygosity in A. oxytropis. Lu et al. [63] defined five SDH (saccharopine dehydrogenase), one FAP2 (saccharopine oxidase), and two P5CR (pyrroline-5-carboxylate reductase) genes in the genome of U. oxytropis, as well as 77 PKS (polyketide synthase genes) and 79 P450 genes. The multiple SDH and P5CR genes Lu et al. [63] identified may represent different genes in the two family. However, given the large number of PKS and P450 genes, it is unlikely all of those genes represent different singletons specific to this pathway. It would be worthwhile to compare the synteny of those SWN genes as defined by Luo et al. [61] with genes previously annotated by Lu et al. [63], as many of the genes such as the SDHs and P450s have not been well characterized in previous work in M. robertsii and may share the similar function. It is worth noticing that the swnA and swnT genes are absent in the A. oxytropis SWN cluster [60]. The absence of swnA may reflect that either they have not been identified in the genome, or they were lost during evolution. As the deletion of swnA could result in little production of swainsonine in M. robertsii, it seems likely that other routes for PA accumulation readily exist in A. oxytropis. The loss of swnT may be related to the secretion of swainsonine as in this endophyte. The above requires further investigation. Li and Lu [69] compared the transcriptomes of WT and saccharopine-reductase-gene knockout A. oxytropis mutant to indicate differential expression by several genes related to the swainsonine biosynthesis. However, these genes were not annotated nor characterized making it difficult to summarize their function. A P2C (1-piperrideine-2-carboxylate) pathway was tentatively suggested by the authors to produce PA in A. oxytropis but this needs confirmation as previous work in S. legunimicola suggested otherwise [66]. Undoubtedly, better genome sequencing assemblies are required to begin to address this issue and group, including our own, are producing such data.
1.5. The Speculated Ecological Role of the Symbiosis in Oxytropis and Its Evolution
To date, the exact ecological relevance of the symbiosis between Oxytropis spp. and A. sect. Undifilum is unclear. This is in marked contrast to other mutualistic symbionts where endophytic interactions have demonstrated benefits to their host, for example in nutrient uptake and stress tolerance. In contrast, the benefits to Oxytropis through interaction with A. sect. Undifilum appear to be marginal at best [20]. It is clear that, unlike the case in poaceae, swainsonine cannot deter livestock grazing but may provide other cryptic benefits to the hosts. As we have discussed in the previous section, swainsonine is synthesized via the lysine → saccharopine (or skipped) → AASA→ P6C → PA → SW route in the fungi [62,66]. It may be relevant that this pathway is reminiscent of lysine catabolism in plants, and this could be a key observation in defining an ecological role for the Oxytropis spp. and A. sect. Undifilum interaction.
To explore the possible ecological role of lysine-PA-swainsonine metabolism in the symbiosis of Oxytropis spp. and A. sect. Undifilum, we speculated that it could be retributed to stress response. Lysine catabolism was suggested to be related to stress tolerance in prokaryotes [64]. Many genes in the catabolic pathway were up-regulated by osmotic stress. Similarly, lysine supplement increased the growth of Silicibacter pomeroyito high salinity, and expression of lysine catabolic operon increased salt tolerance and accumulation of more PA in Escherichia coli. Lysine catabolism was also suggested to be involved in plant stress tolerance [70]. Plant lysine catabolism is diverse, encompassing the cadaverine pathway, the SACPATH (saccharopine), and the NHP (N-hydroxypipecolate) pathways [64,71,72,73,74]. Under stress, levels of free lysine increase due to protein hydrolysis and, as a result several key enzymes in lysine transport and SACPATH pathways are induced in a lysine dependent manner [64]. These include such as the LKR/SDH and AASADH (aminoadipate semialdehyde dehydrogenase) and these many coupled to P5CR activities in order to keep the AASA concentrations to below toxic levels [75]. In the case of halophytes at least, this could be an adaptive pathway that sees increased levels of osmolytes proline and PA under salinity [47]. Being able to produce swainsonine from AASA, A. sect. Undifilum could also facilitate as an outlet to reduce the AASA. On the other hand, the NHP route seems to be deployed differently in making use of lysine to produce PA to induce systemic acquired resistance (SAR) following pathogen attack [76,77]. The endophyte may facilitate the host to produce PA from lysine when needed for downstream plant reaction. This would agree with previous findings that the abundance of A. sect. Unidifilum could regulate the assemblage of pathogens to the host [21,22]. Whether shared lysine catabolism is an ecological determinant of the interaction or not needs future work.
2. Future Perspectives
The understanding regarding host–endophyte interaction, irrespective of tremendous previous efforts, is still limited. This is particularly the case in Oxytropis spp. but the recent study in Astragalus the work by Harrison et al. [22] shed some light on the potential regulatory function of such symbiosis. Future research would possibly need to continue the focus on unravelling the mechanism in the process of symbiont establishment, model of interaction and co-evolution of the host-endophyte, new possibilities of transmission, and swainsonine biosynthesis. The information is still lacking in how the endophyte recognizes a narrow range of the hosts and elaborately evades host defense as no symptoms have been observed in the Oxytropis hosts infected by A. sect. Undifilum. This must indicate that co-evolution has been functioning through either environmental selection or genetic drift. What benefits, if any, do the endophyte supply to the hosts are still unclear although we speculated the endophyte may cryptically defend the hosts against certain biotic attack via sharing some metabolic pathway. Delicate genomes of both the host and the endophyte can serve for the reference in identifying key genes in the said interaction. The endophyte may also facilitate the growth of Oxytropis spp., especially by strengthening phosphorous uptake by altering either signaling transduction or root architecture, as phosphorous is the biggest limiting growth factor in land plants, and such beneficial symbiosis have been seen in root endophyte Colletotrichum tofieldiae confers plant fitness benefits that are phosphate status dependent [78].
Although indicated as vertically transmitted, whether A. sect. Undifilum can be propagated via horizontal transmission in the field, is yet unknown. A better understanding of the pollination process in Oxytropis spp. will also help explain how the genus has evolved into both toxic and non-toxic lineage, due to the maintenance and loss of symbiosis with A. sect. Undifilum. A global assessment of the phylogenetic relationship in both the hosts and endophyte in Oxytropis genus is also remarkably interesting. Especially, given the medicinal potential of swainsonine in the treatment of tumor, strains with high levels of swainsonine production can be selected from natural population or via genetic manipulation.
Author Contributions
Conceptualization, H.G., W.H. and L.A.J.M.; writing—original draft preparation, H.G. and W.H.; writing—review and editing, L.A.J.M.; visualization, H.G., X.L. and Y.F.; supervision, W.H.; funding acquisition, W.H., J.W. and Y.W. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by National Natural Science Foundation of China (31402133), Special Aid Fund for Qinghai Province (2020-QY-210), Key Laboratory Research Fund of Department of Education of Shaanxi Province (18JS110), International Collaboration Fund of Department of Science and Technology of Shaanxi Province (2020KW-030), and Key Laboratory of Grassland Resources of Ministry of Education (2020) in Inner Mongolia Agricultural University to W.H.; Natural Science Foundation of China [31702159], Natural Science Foundation of Shaanxi Province of China [2018JQ3029], and Scientific Research from Shaanxi Provincial Department of Education [16JK1756] to Y.F.; Key Research and Development Plan Project of Shaanxi Province [2018ZDXM-SF-016] to Y.W.; and the UK Biotechnology and Biological Sciences Research Council (BBSRC) exchange grant (BB/R02118X/1) to L.A.J.M.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- James, L.F.; Foote, W. Estrogenic properties of locoweed (Astragalus lentiginosus). Can. J. Comp. Med. 1972, 36, 360–365. [Google Scholar]
- Marsh, C.D. The Loco-Weed Disease of the Plains; Government Printing Office: Washington, DC, USA, 1909; Volume 112. [Google Scholar]
- Cook, D.; Ralphs, M.H.; Welch, K.D.; Stegelmeier, B.L. Locoweed poisoning in livestock. Rangel. Ecol. Manag. 2009, 31, 16–21. [Google Scholar] [CrossRef]
- Tulsiani, D.R.; Broquist, H.P.; James, L.F.; Touster, O. The similar effects of swainsonine and locoweed on tissue glycosidases and oligosaccharides of the pig indicate that the alkaloid is the principal toxin responsible for the induction of locoism. Arch. Biochem. 1984, 232, 76–85. [Google Scholar] [CrossRef]
- Hao, B.; Song, X.; Gao, Y.; Wang, X.; Liu, Y.; Chen, K.; Liang, Y.; Hu, Y.; Xing, X.; Wen, F. Analysis of the biosynthesis pathway of produced SW by endophytic fungi from locoweed using nitrosoguanidine mutation and high throughput sequencing technology. Chin. J. Anim. Vet. Sci. 2019, 50, 169–182. [Google Scholar] [CrossRef]
- Colegate, S.M.; Dorling, P.R.; Huxtable, C.R. A spectroscopic investigation of swainsonine: An α-mannosidase inhibitor isolated from Swainsona canescens. Aust. J. Chem. 1979, 32, 2257–2264. [Google Scholar] [CrossRef]
- Dorling, P.R.; Huxtable, C.R.; Colegate, S.M. Inhibition of lysosomal alpha-mannosidase by swainsonine, an indolizidine alkaloid isolated from Swainsona canescens. Biochem. J. 1980, 191, 649–651. [Google Scholar] [CrossRef] [PubMed]
- James, L.F.; Nielsen, D.B.; Panter, K.E. Impact of poisonous plants on the livestock industry. J. Range Manag. 1992, 45, 3–8. [Google Scholar] [CrossRef]
- Zhao, M.; Gao, X.; Wang, J.; He, X.; Han, B. A review of the most economically important poisonous plants to the livestock industry on temperate grasslands of China. J. Appl. Toxicol. 2013, 33, 9–17. [Google Scholar] [CrossRef]
- Panter, K.E.; James, L.F.; Stegelmeier, B.L.; Ralphs, M.H.; Pfister, J.A. Locoweeds: Effects on reproduction in livestock. J. Nat. Toxins 1999, 8, 53–62. [Google Scholar]
- Panter, K.E.; Ralphs, M.H.; James, L.F.; Stegelmeier, B.L.; Molyneux, R.J. Effects of locoweed (Oxytropis sericea) on reproduction in cows with a history of locoweed consumption. Vet. Hum. Toxicol. 1999, 41, 282–286. [Google Scholar] [CrossRef]
- Zhou, Q.; Bai, S.; Lu, H.; Zhao, B.; Chen, J. Recent advances and issues on locoweed-fungal endophyte. Pratacult. Sci. 2012, 29, 1049–1057. [Google Scholar]
- Siegel, M.R.; Bush, L.P.; Carroll, G.; Tudzynski, P. Toxin Production in Grass/Endophyte Associations; Springer: Berlin/Heidelberg, Germany, 1997. [Google Scholar]
- Bush, L.P.; Wilkinson, H.H.; Schardl, C.L. Bioprotective alkaloids of grass-fungal endophyte symbioses. Plant Physiol. 1997, 114, 1–7. [Google Scholar] [CrossRef]
- Johnson, L.J.; Bonth, A.C.M.; Briggs, L.R.; Caradus, J.R.; Finch, S.C.; Fleetwood, D.J.; Fletcher, L.R.; Hume, D.E.; Johnson, R.D.; Popay, A.J. The exploitation of epichloae endophytes for agricultural benefit. Fungal Divers. 2013, 60, 171–188. [Google Scholar] [CrossRef]
- Cook, D.; Gardner, D.R.; Pfister, J.A. Swainsonine-containing plants and their relationship to endophytic fungi. J. Agric. Food Chem. 2014, 62, 7326–7334. [Google Scholar] [CrossRef] [PubMed]
- Woudenberg, J.H.C.; Groenewald, J.Z.; Binder, M.; Crous, P.W. Alternaria redefined. Stud. Mycol. 2013, 75, 171–212. [Google Scholar] [CrossRef] [PubMed]
- Pryor, B.M.; Creamer, R.; Shoemaker, R.A.; Mclain-Romero, J.; Hambleton, S. Undifilum, a new genus for endophytic Embellisia oxytropis and parasitic Helminthosporium bornmuelleri on legumes. Botany 2009, 87, 178–194. [Google Scholar] [CrossRef]
- Braun, K.; Romero, J.; Liddell, C.; Creamer, R. Production of swainsonine by fungal endophytes of locoweed. Mycol. Res. 2003, 107, 980–988. [Google Scholar] [CrossRef]
- Creamer, R.; Baucom, D. Fungal endophytes of locoweeds: A commensal relationship? J. Plant Physiol. Pathol. 2013, 1, 2. [Google Scholar] [CrossRef]
- Lu, H.; Quan, H.; Zhou, Q.; Ren, Z.; Xue, R.; Zhao, B.; Creamer, R. Endogenous fungi isolated from three locoweed species from rangeland in western China. Afr. J. Microbiol. Res. 2017, 11. [Google Scholar] [CrossRef]
- Harrison, J.G.; Parchman, T.L.; Cook, D.; Gardner, D.R.; Forister, M.L. A heritable symbiont and host-associated factors shape fungal endophyte communities across spatial scales. J. Ecol. 2018, 106, 2274–2286. [Google Scholar] [CrossRef]
- Zhu, X.; Welsh, S.L.; Ohashi, H. Oxytropis Candolle. In Flora of China; Wu, Z., Raven, P.H., Hong, D., Eds.; Science Press: Beijing, China; Missouri Botanical Garden Press: St. Louis, MO, USA, 2010; Volume 10, pp. 453–500. [Google Scholar]
- Ralphs, M.H.; Gardner, D.R.; Turner, D.L.; Thacker, E. Predicting toxicity of tall larkspur (Delphinium barbeyi): Measurement of the variation in alkaloid concentration among plants and among years. J. Chem. Ecol. 2002, 28, 2327–2341. [Google Scholar] [CrossRef] [PubMed]
- Kang, L.; Han, X.; Zhang, Z.; Sun, J. Grassland ecosystems in China: Review of current knowledge and research advancement. Philos. Trans. R. Soc. Lond. B. Biol. Sci. 2007, 362, 997–1008. [Google Scholar] [CrossRef]
- Cook, D.; Gardner, D.R.; Lee, S.T.; Pfister, J.A.; Stonecipher, C.A.; Welsh, S.L. A swainsonine survey of North American Astragalus and Oxytropis taxa implicated as locoweeds. Toxicon 2016, 118, 104–111. [Google Scholar] [CrossRef]
- Ralphs, M.H.; Creamer, R.; Baucom, D.; Gardner, D.R.; Welsh, S.L.; Graham, J.D.; Hart, C.; Cook, D.; Stegelmeier, B.L. Relationship between the endophyte Embellisia spp. and the toxic alkaloid swainsonine in major locoweed species (Astragalus and Oxytropis). J. Chem. Ecol. 2008, 34, 32–38. [Google Scholar] [CrossRef]
- Hu, Y.; Liu, L.; Liu, P.; Wang, J. An overview of ecological research on locoweed. Prog. Anim. Med. 2011, 032, 108–112. [Google Scholar] [CrossRef]
- Liu, L.; Feng, Y.; Jia, Z.; Zhao, B. Research progress on development and utilization of locoweed in China. Acta Ecol. Anim. Domastici 2010, 31, 106–109. [Google Scholar] [CrossRef]
- Yu, Y.; Zhao, Q.; Wang, J.; Wang, J.; Wang, Y.; Song, Y.; Geng, G.; Li, Q. Swainsonine-producing fungal endophytes from major locoweed species in China. Toxicon 2010, 56, 330–338. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Nagao, H.; Li, Y.L.; Wang, H.S.; Kakishima, M. Embellisia oxytropis, a new species isolated from Oxytropis kansuensis in China. Mycotaxon 2006, 95, 255–260. [Google Scholar] [CrossRef]
- Ralphs, M.H.; Cook, D.; Gardner, D.R.; Grum, D.S. Transmission of the locoweed endophyte to the next generation of plants. Fungal Ecol. 2011, 4, 251–255. [Google Scholar] [CrossRef]
- Mclain-Romero, J.; Creamer, R.; Zepeda, H.; Strickland, J.; Bell, G. The toxicosis of Embellisia fungi from locoweed (Oxytropis lambertii) is similar to locoweed toxicosis in rats. J. Anim. Sci. 2004, 82, 2169–2174. [Google Scholar] [CrossRef]
- Barillas, J.R.V.; Paschke, M.W.; Ralphs, M.H.; Child, R.D. White locoweed toxicity facilitated by a fungal endophyte and nitrogen-fixing bacteria. Ecology 2007, 88, 1850–1856. [Google Scholar] [CrossRef]
- Cook, D.; Gardner, D.R.; Welch, K.D.; Roper, J.M.; Ralphs, M.H.; Green, B.T. Quantitative PCR method to measure the fungal endophyte in locoweeds. J. Agric. Food Chem. 2009, 57, 6050–6054. [Google Scholar] [CrossRef] [PubMed]
- Gardner, D.R.; Romero, J.; Ralphs, M.H.; Creamer, R. Correlation of an endophytic fungus (Alternaria spp.) with the presence of swainsonine in Lambert locoweed (Oxytropis lambertii). In Poisonous Plants and Related Toxins; Acamovic, T., Stewart, C.S., Pennycott, T.W., Eds.; CABI: Walligford, UK, 2004. [Google Scholar]
- Wang, Z.; Cao, S.; Xu, N.; Xu, B.; Li, Y. Cultural characteristics of fungal endophyte from 3 locoweed species in China. Acta Pratacult. Sin. 2017, 26, 158–169. [Google Scholar]
- Zhang, L.; He, S.; Yu, Y.; Zhao, Q. Isolation and identification of swainsonine-producing fungal endophyte from Oxytropis ochrocephala Bunge in Ningxia. China Anim. Husb. Vet. Med. 2013, 40, 165–172. [Google Scholar]
- Chen, J.; Zhao, B.; Lu, H.; Ding, L.; Zhang, G.; Li, X.; Gao, Y.; Zhou, Q.; Yang, X. Study on isolation, identification and the antibacterial activity of endophytic fungi from the major locoweed in China. Acta Vet. Zootech. Sin. 2012, 43, 1471–1478. [Google Scholar]
- Huo, H.; Lu, P.; Niu, Y.; Lv, G.; Li, S.; Gao, J.; Qian, Y. Isolation, culture and identification the 11 endophytic fungus from Oxytropis glabra DC. J. Inner Mong. Univ. 2010, 41, 206–211. [Google Scholar] [CrossRef]
- Lu, P.; Child, D.; Zhao, M.; Gardener, D.R.; Lv, G.; Han, G. Culture and identification of endophytic fungi from Oxytropis glabra DC. Acta Ecol. Sin. 2009, 29, 53–58. [Google Scholar]
- Oldrup, E.; Mclain-Romero, J.; Padilla, A.; Moya, A.; Creamer, R. Localization of endophytic Undifilum fungi in locoweed seed and influence of environmental parameters on a locoweed in vitro culture system. Botanique 2010, 88, 512–521. [Google Scholar] [CrossRef]
- Noor, A.I.; Nava, A.; Cooke, P.; Cook, D.; Creamer, R. Evidence for nonpathogenic relationships of Alternaria section Undifilum endophytes within three host locoweed plant species. Botany 2018, 96. [Google Scholar] [CrossRef]
- Reyna, R.; Cooke, P.; Grum, D.; Cook, D. Detection and localization of the endophyte Undifilum oxytropis in locoweed tissues. Botany 2012, 90, 1229–1236. [Google Scholar] [CrossRef]
- White, J.F.J.; Morgan-Jones, G.; Morrow, A.C. Taxonomy, life cycle, reproduction and detection of Acremonium endophytes. Agric. Ecosyst. Environ. 1993, 44, 13–37. [Google Scholar] [CrossRef]
- Cook, D.; Gardner, D.R.; Ralphs, M.H.; Pfister, J.A.; Welch, K.D.; Green, B.T. Swainsoninine concentrations and endophyte amounts of Undifilum oxytropis in different plant parts of Oxytropis sericea. J. Chem. Ecol. 2009, 35, 1272–1278. [Google Scholar] [CrossRef] [PubMed]
- Arruda, P.; Barreto, P. Lysine catabolism through the saccharopine pathway: Enzymes and intermediates involved in plant responses to abiotic and biotic stress. Front. Plant Sci. 2020, 11, 587. [Google Scholar] [CrossRef]
- Mills, W.R.; Lea, P.J.; Miflin, B.J. Photosynthetic formation of the aspartate family of amino acids in isolated chloroplasts. Plant Physiol. 1980, 65, 1166–1172. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Currah, R.; Zelmer, C. A key and notes for the genera of fungi mycorrhizal with orchids and a new species in the genus epulorhiza. Rep. Tottori Mycol. Inst. 1992, 30, 43–59. [Google Scholar]
- Harvais, G.; Hadley, G. The development of Orchis purpurella in asymbiotic and inoculated cultures. New Phytol. 1967, 66, 217–230. [Google Scholar] [CrossRef]
- Panaccione, D.G.; Beaulieu, W.T.; Cook, D. Bioactive alkaloids in vertically transmitted fungal endophytes. Funct. Ecol. 2014, 28, 299–314. [Google Scholar] [CrossRef]
- Doss, R.; Welty, R. A polymerase chain reaction-based procedure for detection of Acremonium coenophialum in tall fescue. Phytopathology 1995, 85, 913–917. [Google Scholar] [CrossRef]
- Liu, J.; Li, Y.; Creamer, R. A re-examination of the taxonomic status of Embellisia astragali. Curr. Microbiol. 2016, 72, 404–409. [Google Scholar] [CrossRef]
- He, W.; Guo, L.; Wang, L.; Zhao, Q.; Guo, L.; Cao, W.; Mur, L.A.J.; Wei, Y. Host genotype and precipitation influence of fungal endophyte symbiosis and mycotoxin abundance in a locoweed. Int. J. Mol. Sci. 2019, 20, 5285. [Google Scholar] [CrossRef]
- Noor, A.I.; Neyaz, M.; Cook, D.; Creamer, R. Molecular characterization of a fungal ketide synthase gene among swainsonine-producing Alternaria species in the USA. Curr. Microbiol. 2020, 77, 2554–2563. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, R.J.; White Jr, J.F.; Arnold, A.E.; Redman, R.S. Fungal endophytes: Diversity and functional roles. New Phytol. 2010, 182, 314–330. [Google Scholar] [CrossRef]
- Cook, D.; Grum, D.S.; Gardner, D.R.; Welch, K.D.; Pfister, J.A. Influence of endophyte genotype on swainsonine concentrations in Oxytropis sericea. Toxicon 2013, 61, 105–111. [Google Scholar] [CrossRef]
- Liu, H. Study on the Charcteristics of Locoweed Endophyte in 3 Species of Oxytropis from Seeds to Plants. Ph.D. Thesis, Lanzhou University, Lanzhou, China, 2019. [Google Scholar]
- Grum, D.S.; Cook, D.; Gardner, D.R.; Roper, J.M.; Pfister, J.A.; Ralphs, M.H. Influence of seed endophyte amounts on swainsonine concentrations in Astragalus and Oxytropis locoweeds. J. Agric. Food Chem. 2012, 60, 8083. [Google Scholar] [CrossRef]
- Cook, D.; Donzelli, B.G.G.; Creamer, R.; Baucom, D.L.; Gardner, D.R.; Pan, J.; Moore, N.; Krasnoff, S.B.; Jaromczyk, J.W.; Schardl, C.L. Swainsonine biosynthesis genes in diverse symbiotic and pathogenic fungi. G3 2017, 7, 1791–1797. [Google Scholar] [CrossRef] [PubMed]
- Luo, F.; Hong, S.; Chen, B.; Yin, Y.; Wang, C. Unveiling of swainsonine biosynthesis via a multibranched pathway in fungi. ACS Chem. Biol. 2020, 15, 2476–2484. [Google Scholar] [CrossRef]
- Tan, X.; Chen, A.J.; Wu, B.; Zhang, G.; Ding, G. Advance of swainsonine biosynthesis. Chin. Chem. Lett. 2018, 29, 417–422. [Google Scholar] [CrossRef]
- Lu, H.; Quan, H.; Ren, Z.; Wang, S.; Xue, R.; Zhao, B. The genome of Undifilum oxytropis provides insights into swainsonine biosynthesis and locoism. Sci. Rep. 2016, 6, 30760. [Google Scholar] [CrossRef]
- Neshich, I.A.P.; Kiyota, E.; Arruda, P. Genome-wide analysis of lysine catabolism in bacteria reveals new connections with osmotic stress resistance. ISME J. 2013, 7, 2400–2410. [Google Scholar] [CrossRef]
- Zabriskie, T.M.; Jackson, M.D. Lysine biosynthesis and metabolism in fungi. Nat. Prod. Rep. 2000, 17, 85–97. [Google Scholar] [CrossRef] [PubMed]
- Wickwire, B.M.; Harris, C.M.; Harris, T.M.; Broquist, H.P. Pipecolic acid biosynthesis in Rhizoctonia leguminicola. I. The lysine saccharopine, delta 1-piperideine-6-carboxylic acid pathway. J. Biol. Chem. 1990, 265, 14742–14747. [Google Scholar] [CrossRef]
- Gatto, G.J.; Boyne, M.T.; Kelleher, N.L.; Walsh, C.T. Biosynthesis of pipecolic acid by RapL, a lysine cyclodeaminase encoded in the rapamycin gene cluster. J. Am. Chem. Soc. 2006, 128, 3838. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, S.; Dawe, A.L.; Creamer, R. Potential role for saccharopine reductase in swainsonine metabolism in endophytic fungus, Undifilum oxytropis. Fungal Biol. 2012, 116, 902–909. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Lu, P. Transcriptome profiles of Alternaria oxytropis provides insights into swainsonine biosynthesis. Sci. Rep. 2019, 9, 6021. [Google Scholar] [CrossRef] [PubMed]
- Galili, G.; Tang, G.; Zhu, X.; Gakiere, B. Lysine catabolism: A stress and development super-regulated metabolic pathway. Curr. Opin. Plant Biol. 2001, 4, 261–266. [Google Scholar] [CrossRef]
- Arruda, P.; Kemper, E.L.; Papes, F.; Leite, A. Regulation of lysine catabolism in higher plants. Trends Plant Sci. 2000, 5, 324–330. [Google Scholar] [CrossRef]
- Bunsupa, S.; Katayama, K.; Ikeura, E.; Oikawa, A.; Yamazaki, M. Lysine decarboxylase catalyzes the first step of quinolizidine alkaloid biosynthesis and coevolved with alkaloid production in Leguminosae. Plant Cell 2012, 24, 1202–1216. [Google Scholar] [CrossRef]
- Papes, F.; Kemper, E.L.; Cord-Neto, G.; Langone, F.; Arruda, P. Lysine degradation through the saccharopine pathway in mammals: Involvement of both bifunctional and monofunctional lysine-degrading enzymes in mouse. Biochem. J. 1999, 344, 555–563. [Google Scholar] [CrossRef] [PubMed]
- Struys, E.A.; Jakobs, C. Metabolism of lysine in alpha-aminoadipic semialdehyde dehydrogenase-deficient fibroblasts: Evidence for an alternative pathway of pipecolic acid formation. FEBS Lett. 2010, 584, 181–186. [Google Scholar] [CrossRef] [PubMed]
- Szabados, L.; Savouré, A. Proline: A multifunctional amino acid. Trends Plant Sci. 2010, 15, 89–97. [Google Scholar] [CrossRef] [PubMed]
- Michael, H.; Jürgen, Z. L-Lys metabolism to N-hydroxypipecolic acid: An integral immune-activating pathway in plants. Plant J. 2018, 96. [Google Scholar] [CrossRef]
- Hartmann, M.; Zeier, T.; Bernsdorff, F.; Reichel-Deland, V.; Kim, D.; Hohmann, M.; Scholten, N.; Schuck, S.; Schuck, S.; Bräutigam, A.; et al. Flavin monooxygenase-generated N-hydroxypipecolic acid is a critical element of plant systemic immunity. Cell 2018, 173, 456–469. [Google Scholar] [CrossRef] [PubMed]
- Hiruma, K.; Gerlach, N.; Sacristán, S.; Nakano, R.T.; Hacquard, S.; Kracher, B.; Neumann, U.; Ramírez, D.; Bucher, M.; O’Connell, R.J. Root endophyte colletotrichum tofieldiae confers plant fitness benefits that are phosphate status dependent. Cell 2016, 165, 464–474. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).