Japonamides A and B, Two New Cyclohexadepsipeptides from the Marine-Sponge-Derived Fungus Aspergillus japonicus and Their Synergistic Antifungal Activities
Abstract
:1. Introduction
2. Materials and Methods
2.1. General Experimental Procedures
2.2. Fungal Material, Fermentation, and Extraction
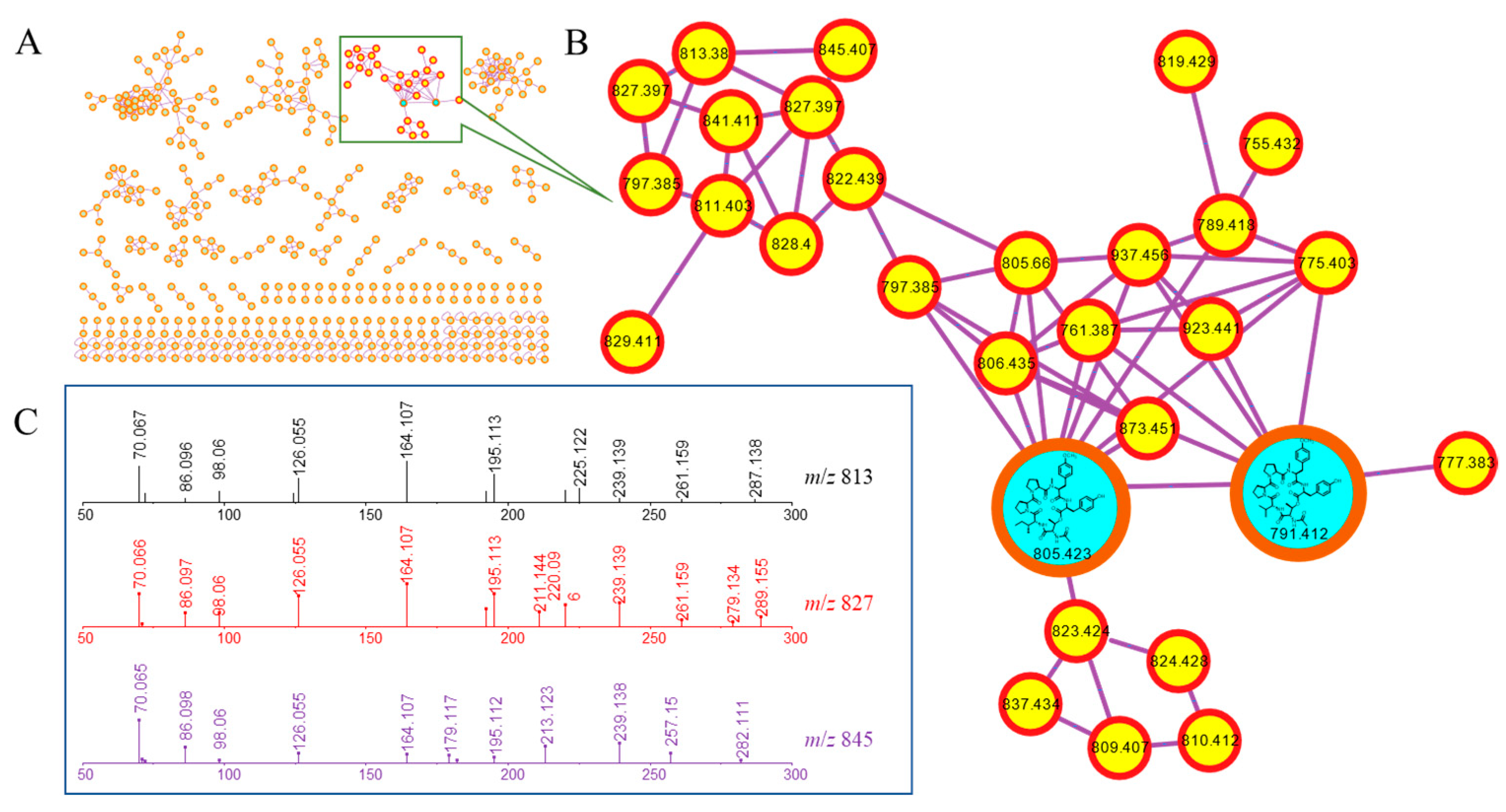
2.3. Molecular Networking Analyses and Compound Isolation
2.4. Absolute Configurations of Amino Acids by the Advanced Marfey’s Analysis
2.5. In Vitro Activities of Compounds 1 and 2 in Combination with Antibiotics against Candida albicans SC5314
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Du, M.; Xuan, W.; Zhen, X.; He, L.; Lan, L.; Yang, S.; Wu, N.; Qin, J.; Zhao, R.; Qin, J.; et al. Antimicrobial photodynamic therapy for oral Candida infection in adult AIDS patients: A pilot clinical trial. Photodiagnosis Photodyn. Ther. 2021, 34, 102310. [Google Scholar] [CrossRef] [PubMed]
- Park, M.; Ho, D.Y.; Wakelee, H.A.; Neal, J.W. Opportunistic invasive fungal infections mimicking progression of non-small-cell lung cancer. Clin. Lung Cancer 2021, 22, e193–e200. [Google Scholar] [CrossRef]
- Phoompoung, P.; Villalobos, A.P.C.; Jain, S.; Foroutan, F.; Orchanian-Cheff, A.; Husain, S. Risk factors of invasive fungal infections in lung transplant recipients: A systematic review and meta-analysis. J. Heart Lung Transplant. 2022, 41, 255–262. [Google Scholar] [CrossRef] [PubMed]
- Mishra, P.; Agrawal, N.; Bhurani, D.; Agarwal, N.B. Invasive Fungal infections in patients with acute myeloid leukemia undergoing intensive chemotherapy. Indian J. Hematol. Blood Transfus. 2020, 36, 64–70. [Google Scholar] [CrossRef]
- Bertolini, M.; Ranjan, A.; Thompson, A.; Diaz, P.I.; Sobue, T.; Maas, K.; Dongari-Bagtzoglou, A. Candida albicans induces mucosal bacterial dysbiosis that promotes invasive infection. PLoS Pathog. 2019, 15, e1007717. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lone, S.A.; Ahmad, A. Candida auris-the growing menace to global health. Mycoses. 2019, 62, 620–637. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yáñez, A.; Murciano, C.; Gil, M.L.; Gozalbo, D. Immune Response to Candida albicans Infection. In Encyclopedia of Mycology; Zaragoza, Ó., Casadevall, A., Eds.; Elsevier: Oxford, UK, 2021; Volume 1, pp. 556–575. [Google Scholar] [CrossRef]
- Xiao, X.F.; Wu, J.X.; Xu, Y.C. Treatment of invasive fungal disease: A case report. World J. Clin. Cases. 2019, 7, 2374–2383. [Google Scholar] [CrossRef]
- Yin, S.; Li, L.; Su, L.; Li, H.; Zhao, Y.; Wu, Y.; Liu, R.; Zou, F.; Ni, G. Synthesis and in vitro synergistic antifungal activity of analogues of Panax stipulcanatus saponin against fluconazole-resistant Candida albicans. Carbohydr. Res. 2022, 517, 108575. [Google Scholar] [CrossRef] [PubMed]
- Yang, Q.; Liu, Z.; Wang, Y.; Xie, J.; Zhang, K.; Dong, Y.; Wang, Y.F. In vitro synergistic antifungal activities with caspofungin plus fluconazole or voricanazole against Candida species determined by Etest method. Int. J. Infect. Dis. 2022, 122, 982–990. [Google Scholar] [CrossRef]
- Zhou, J.; Zou, Y.; Cai, Y.; Chi, F.; Huang, W.; Shi, W.; Qian, H. A designed cyclic peptide based on trastuzumab used to construct peptide-drug conjugates for its HER2-targeting ability. Bioorg. Chem. 2021, 117, 105453. [Google Scholar] [CrossRef] [PubMed]
- Larnaudie, S.C.; Sanchis, J.; Nguyen, T.H.; Peltier, R.; Catrouillet, S.; Brendel, J.C.; Porter, C.J.H.; Jolliffe, K.A.; Perrier, S. Cyclic peptide-poly (HPMA) nanotubes as drug delivery vectors: In vitro assessment, pharmacokinetics and biodistribution. Biomaterials 2018, 178, 570–582. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mao, B.; Liu, C.; Zheng, W.; Li, X.; Ge, R.; Shen, H.; Guo, X.; Lian, Q.; Shen, X.; Li, C. Cyclic cRGDfk peptide and Chlorin e6 functionalized silk fibroin nanoparticles for targeted drug delivery and photodynamic therapy. Biomaterials 2018, 161, 306–320. [Google Scholar] [CrossRef]
- Tian, J.; Shen, Y.; Yang, X.; Liang, S.; Shan, L.; Li, H.; Liu, R.; Zhang, W. Antifungal cyclic peptides from Psammosilene tunicoides. J. Nat. Prod. 2010, 73, 1987–1992. [Google Scholar] [CrossRef]
- Han, J.; Wang, H.; Zhang, R.; Dai, H.; Chen, B.; Wang, T.; Sun, J.; Wang, W.; Song, F.; Li, E.; et al. Cyclic tetrapeptides with synergistic antifungal activity from the fungus Aspergillus westerdijkiae using LC-MS/MS-Based molecular networking. Antibiotics 2022, 11, 166. [Google Scholar] [CrossRef]
- Wu, W.; Dai, H.; Bao, L.; Ren, B.; Lu, J.; Luo, Y.; Guo, L.; Zhang, L.; Liu, H. Isolation and structural elucidation of proline-containing cyclopentapeptides from an endolichenic Xylaria sp. J. Nat. Prod. 2011, 74, 1303–1308. [Google Scholar] [CrossRef] [PubMed]
- Luo, M.; Chang, S.; Li, Y.; Xi, X.; Chen, M.; He, N.; Wang, M.; Zhao, W.; Xie, Y. Molecular networking-based screening led to the discovery of a cyclic heptadepsipeptide from an endolichenic Xylaria sp. J. Nat. Prod. 2022, 85, 972–979. [Google Scholar] [CrossRef] [PubMed]
- Watrous, J.; Roach, P.; Alexandrov, T.; Heath, B.S.; Yang, J.Y.; Kersten, R.D.; van der Voort, M.; Pogliano, K.; Gross, H.; Raaijmakers, J.M.; et al. Mass spectral molecular networking of living microbial colonies. Proc. Natl. Acad. Sci. USA 2012, 109, E1743–E1752. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ding, Y.; Gruschow, S.; Greshock, T.J.; Finefield, J.M.; Sherman, D.H.; Williams, R.M. Detection of VM55599 and preparaherquamide from Aspergillus japonicus and Penicillium fellutanum: Biosynthetic implications. J. Nat. Prod. 2008, 71, 1574–1578. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hayashi, H.; Nishimoto, Y.; Akiyama, K.; Nozaki, H. New paralytic alkaloids, asperparalines A, B and C, from Aspergillus japonicus JV-23. Biosci. Biotechnol. Biochem. 2000, 64, 111–115. [Google Scholar] [CrossRef] [PubMed]
- Han, J.; Chen, B.; Zhang, R.; Zhang, J.; Dai, H.; Wang, T.; Sun, J.; Zhu, G.; Li, W.; Li, E.; et al. Exploring verrucosidin derivatives with glucose-uptake-stimulatory activity from Penicillium cellarum using MS/MS-based molecular networking. J. Fungi 2022, 8, 143. [Google Scholar] [CrossRef] [PubMed]
- Tong, Y.; Zhang, J.; Wang, L.; Wang, Q.; Huang, H.; Chen, X.; Zhang, Q.; Li, H.; Sun, N.; Liu, G.; et al. Hyper-synergistic antifungal activity of rapamycin and peptide-like compounds against Candida albicans orthogonally via Tor1 Kinase. ACS Infect. Dis. 2021, 7, 2826–2835. [Google Scholar] [CrossRef] [PubMed]
- Siemion, I.Z.; Wieland, T.; Pook, K.H. Influence of the distance of the proline carbonyl from the beta and gamma carbon on the 13C chemical shifts. Angew. Chem. Int. Ed. Engl. 1975, 14, 702–703. [Google Scholar] [CrossRef] [PubMed]
- Tang, W.Z.; Liu, J.T.; Hu, Q.; He, R.J.; Guan, X.Q.; Ge, G.B.; Han, H.; Yang, F.; Lin, H.W. Pancreatic lipase inhibitory cyclohexapeptides from the marine sponge-derived fungus Aspergillus sp. 151304. J. Nat. Prod. 2020, 83, 2287–2293. [Google Scholar] [CrossRef] [PubMed]
- Jegorov, A.; Paizs, B.; Kuzma, M.; Zabka, M.; Landa, Z.; Sulc, M.; Barrow, M.P.; Havlicek, V. Extraribosomal cyclic tetradepsipeptides beauverolides: Profiling and modeling the fragmentation pathways. J. Mass Spectrom. 2004, 39, 949–960. [Google Scholar] [CrossRef] [PubMed]
- Paizs, B.; Suhai, S. Towards understanding the tandem mass spectra of protonated oligopeptides. 1: Mechanism of amide bond cleavage. J. Am. Soc. Mass. Spectrom. 2004, 15, 103–113. [Google Scholar] [CrossRef] [Green Version]
- Polce, M.J.; Ren, D.; Wesdemiotis, C. Dissociation of the peptide bond in protonated peptides. J Mass Spectrom. 2000, 35, 1391–1398. [Google Scholar] [CrossRef]





| 1 | 2 | ||||
|---|---|---|---|---|---|
| Position | Units | δC | δH (mult, J in Hz) | δC | δH (mult, J in Hz) |
| l-Pro-1 | |||||
| 1 | CO | 169.3 | 169.3 | ||
| 2 | α-CH | 57.3 | 4.62 (dd, 8.8, 3.5) | 57.2 | 4.62 (dd, 8.8, 3.4) |
| 3 | β-CH2 | 27.4 | 1.71 (m) | 27.5 | 1.72 (m) |
| 2.16 (m) | 2.15 (m) | ||||
| 4 | γ-CH2 | 23.9 | 1.85 (m) | 23.9 | 1.84 (m) |
| 1.89 (m) | 1.90 (m) | ||||
| 5 | δ-CH2 | 47.1 | 3.57 (dt, 14.0, 8.0) | 47.2 | 3.61 (m) |
| 3.82 (dt, 14.0, 8.0) | 3.85(ddd,10.0,8.1,5.2) | ||||
| l-Pro-2 | |||||
| 6 | CO | 172.2 | 172.2 | ||
| 7 | α-CH | 54.5 | 4.31 (dd, 8.0, 5.4) | 54.5 | 4.31 (dd, 8.0, 5.4) |
| 8 | β-CH2 | 28.0 | 0.73 (m) | 28.0 | 0.72 (m) |
| 1.01 (m) | 1.01 (m) | ||||
| 9 | γ-CH2 | 25.0 | 1.63 (m) | 25.0 | 1.62 (m) |
| 1.88 (m) | 1.89 (m) | ||||
| 10 | δ-CH2 | 46.8 | 3.44 (dt, 14.0, 7.0) | 46.8 | 3.44 (dt, 10.0, 7.1) |
| 3.64 (dt, 14.0, 7.0) | 3.63 (m) | ||||
| N-Me-O-Me-l-Tyr | |||||
| 11 | CO | 169.6 | 169.7 | ||
| 12 | α-CH | 61.8 | 4.79 (dd, 11.6, 3.5) | 61.8 | 4.80 (dd, 11.5, 3.5) |
| 13 | β-CH2 | 32.2 | 2.73 (dd, 14.5, 11.6) | 32.2 | 2.73 (dd, 14.5, 11.5) |
| 2.99 (dd, 14.5, 3.5) | 2.99 (dd, 14.5, 3.5) | ||||
| 14 | γ-C | 129.8 | 129.8 | ||
| 15 or 19 | δ-CH | 130.4 | 7.10 (d, 8.9) | 130.4 | 7.10 (d, 8.5) |
| 16 or 18 | ε-CH | 113.9 | 6.85 (d, 8.9) | 113.9 | 6.84 (d, 8.5) |
| 17 | ζ-C | 158.1 | 158.1 | ||
| 20 | N-CH3 | 28.2 | 2.23 (s) | 28.2 | 2.23 (s) |
| 21 | OCH3 | 55.1 | 3.70 (s) | 55.1 | 3.70 (s) |
| l-Tyr | |||||
| 22 | CO | 170.7 | 170.7 | ||
| 23 | α-CH | 55.7 | 4.25 (td, 8.7, 6.0) | 55.7 | 4.26(ddd,10.0,8.5,5.0) |
| 24 | β-CH2 | 36.3 | 2.84 (m) | 36.3 | 2.86 (m) |
| 25 | γ-C | 127.8 | 127.9 | ||
| 26 or 30 | δ-CH | 130.1 | 6.95 (d, 8.5) | 130.1 | 6.95 (d, 8.5) |
| 27 or 29 | ε-CH | 115.0 | 6.65 (d, 8.5) | 115.1 | 6.61 (d, 8.5) |
| 28 | ζ-C | 155.9 | 155.9 | ||
| NH | 8.66 (d, 8.7) | 8.67 (d, 8.5) | |||
| OH | 9.22 (s) | 9.23 (s) | |||
| N-Ac-l-Thr | |||||
| 31 | CO | 167.7 | 167.6 | ||
| 32 | α-CH | 54.5 | 4.71 (dd, 9.1, 4.5) | 54.4 | 4.72 (dd, 9.1, 4.5) |
| 33 | β-CH | 69.9 | 4.96 (qd, 6.6, 4.5) | 69.9 | 4.94 (dd, 6.6, 4.5) |
| 34 | γ-CH3 | 15.2 | 1.21 (d, 6.6) | 15.2 | 1.21 (d, 6.6) |
| 35 | CO | 170.0 | 169.9 | ||
| 36 | CH3 | 22.7 | 1.98 (s) | 22.7 | 1.98 (s) |
| NH | 7.78 (d, 9.1) | 7.78 (d, 9.1) | |||
| d-Ile or Val | |||||
| 37 | CO | 169.8 | 169.8 | ||
| 38 | α-CH | 54.5 | 4.42 (t, 8.9) | 55.8 | 4.32 (d, 8.8) |
| 39 | β-CH | 36.3 | 1.70 (m) | 29.8 | 1.90 (m) |
| 40 | γ1-CH2 or γ1-CH3 | 25.2 | 1.07 (m) | 18.7 | 0.85 (d, 5.2) |
| 1.31 (m) | |||||
| 41 | γ2-CH3 | 14.7 | 0.82 (d, 6.7) | 18.6 | 0.86 (d, 5.2) |
| 42 | δ-CH3 | 11.6 | 0.85 (t, 7.4) | ||
| NH | 7.35 (d, 8.9) | 7.40 (d, 8.8) | |||
| Drugs | Antifungal MICs (μM) | Synergistic Antifungal MICs (μM) | FICI a | Definition b |
|---|---|---|---|---|
| Rapamycin | 0.5 | 0.002 | − | − |
| 1 | >100 | 3.125 | 0.03 | HS |
| 2 | >100 | 3.125 | 0.03 | HS |
| Fluconazole | 0.25 | 0.063 | − | − |
| 1 | >100 | 12.5 | 0.25 | S |
| 2 | >100 | 12.5 | 0.25 | S |
| Ketoconazole | 0.016 | 0.002 | − | − |
| 1 | >100 | 6.25 | 0.125 | S |
| 2 | >100 | 6.25 | 0.125 | S |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, H.; Zhang, R.; Ma, B.; Wang, W.; Yu, C.; Han, J.; Zhu, L.; Zhang, X.; Dai, H.; Liu, H.; et al. Japonamides A and B, Two New Cyclohexadepsipeptides from the Marine-Sponge-Derived Fungus Aspergillus japonicus and Their Synergistic Antifungal Activities. J. Fungi 2022, 8, 1058. https://doi.org/10.3390/jof8101058
Wang H, Zhang R, Ma B, Wang W, Yu C, Han J, Zhu L, Zhang X, Dai H, Liu H, et al. Japonamides A and B, Two New Cyclohexadepsipeptides from the Marine-Sponge-Derived Fungus Aspergillus japonicus and Their Synergistic Antifungal Activities. Journal of Fungi. 2022; 8(10):1058. https://doi.org/10.3390/jof8101058
Chicago/Turabian StyleWang, Haifeng, Rui Zhang, Ben Ma, Wenzhao Wang, Chong Yu, Junjie Han, Lingjuan Zhu, Xue Zhang, Huanqin Dai, Hongwei Liu, and et al. 2022. "Japonamides A and B, Two New Cyclohexadepsipeptides from the Marine-Sponge-Derived Fungus Aspergillus japonicus and Their Synergistic Antifungal Activities" Journal of Fungi 8, no. 10: 1058. https://doi.org/10.3390/jof8101058







