Lignans Extract from Knotwood of Norway Spruce—A Possible New Weapon against GTDs
Abstract
:1. Introduction
2. Materials and Methods
2.1. Fungal Strains
2.2. Preparation of the GE
2.3. In Vitro Antifungal Activity of GE
2.4. In Vitro Antifungal Activity of Pure HMR vs. GE
2.5. Plant Material and Treatments
2.6. Extraction of the DNA and DNA Pooling
2.7. Library Preparation and Sequencing
2.8. Bioinformatic and Data Evaluation
2.9. Statistical Analyses of In Vitro Experiment
2.10. Analyses of HTS Data
3. Results
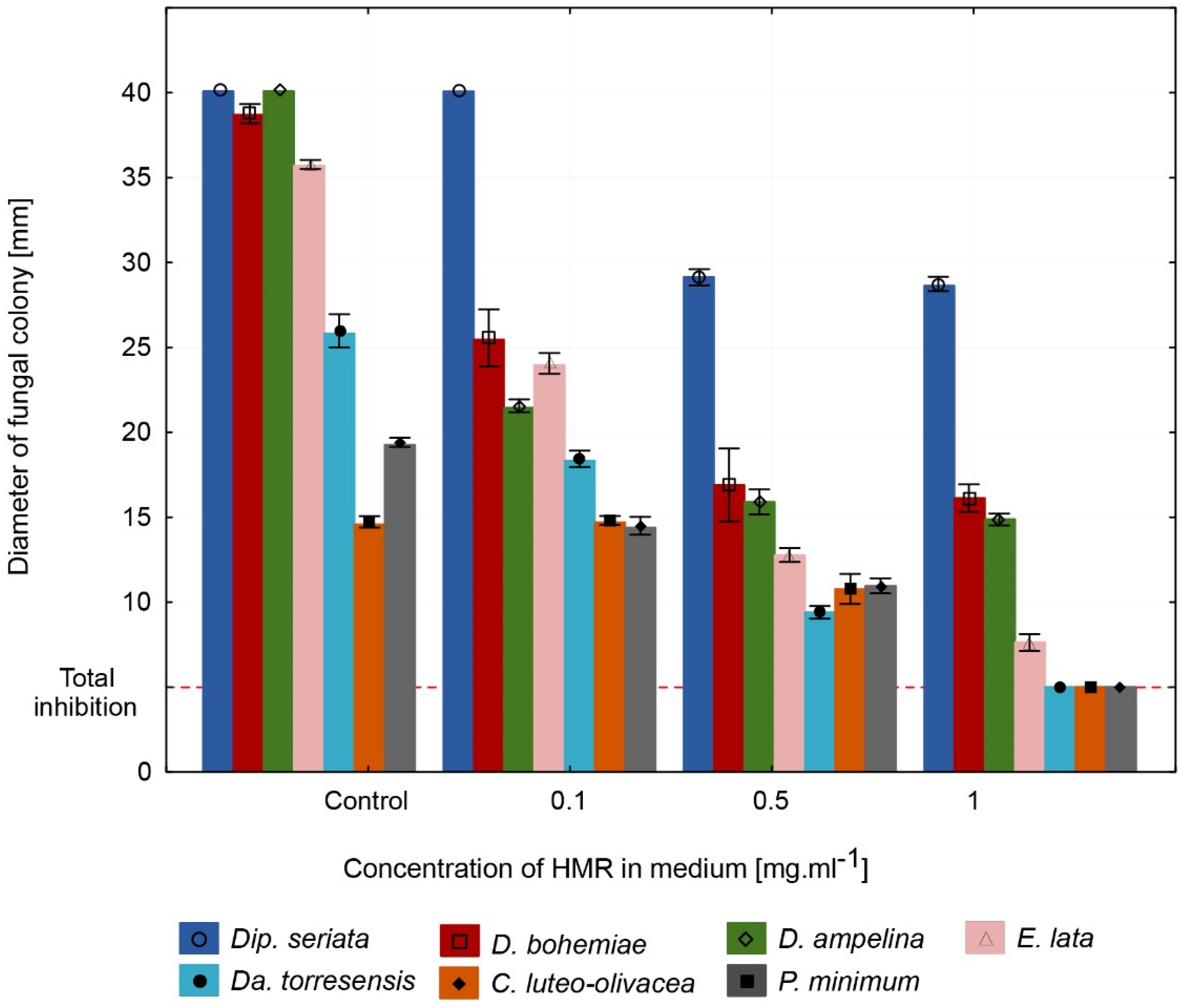
3.1. GE Exhibits Strong Antifungal Effects against GTD Fungi In Vitro
3.2. In Vitro Trials Reveal Stronger Antifungal Capacity of GE Compared to Pure HMRTM
3.3. Sequencing Depth and Community Diversity
3.4. Taxonomic Distribution of Fungi Identified by HTAS
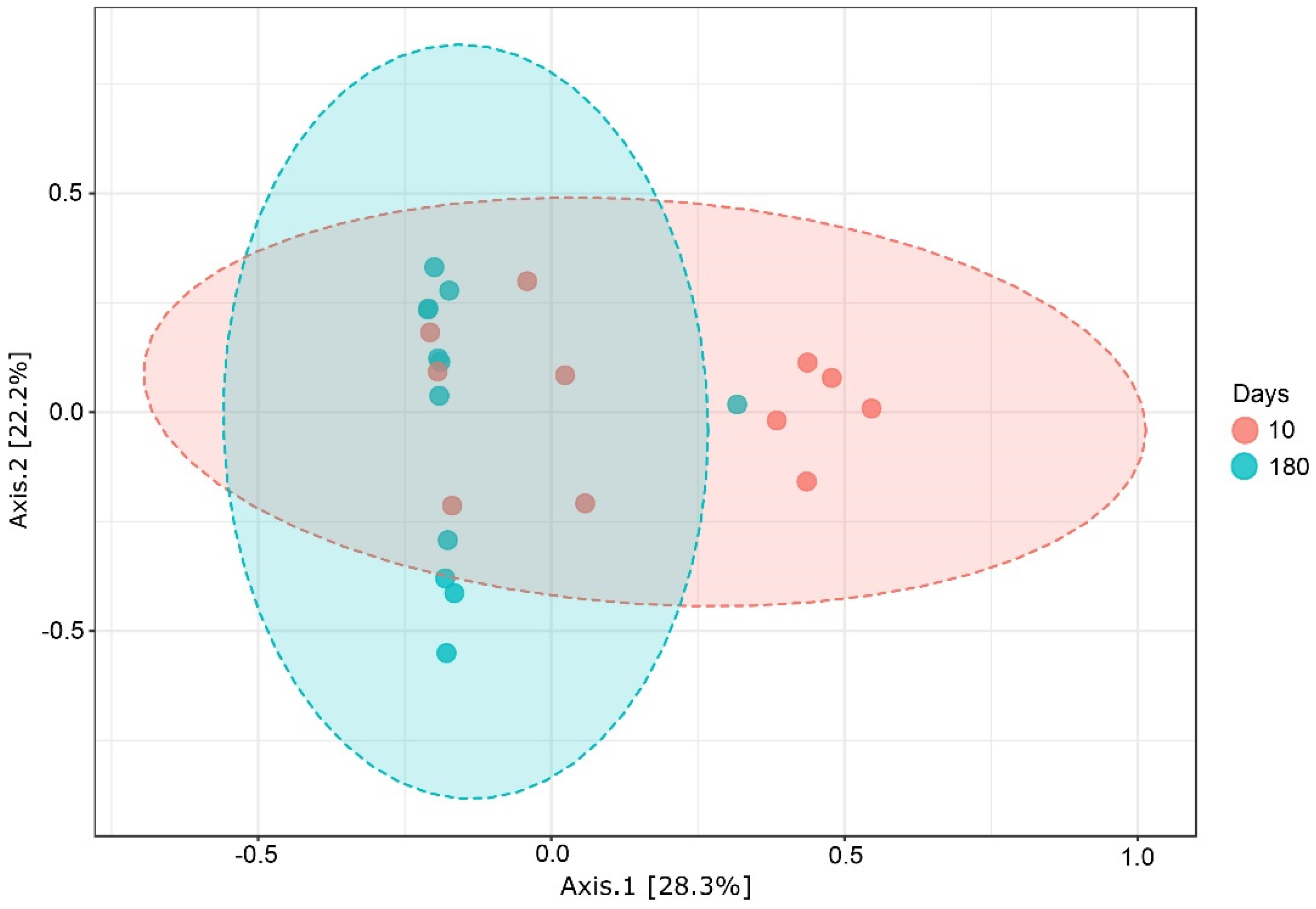
3.5. Alpha Diversity Metrics Did Not Differ among Treated × Untreated Plants
3.6. Shared Fungal Assemblages and Unique OTUs
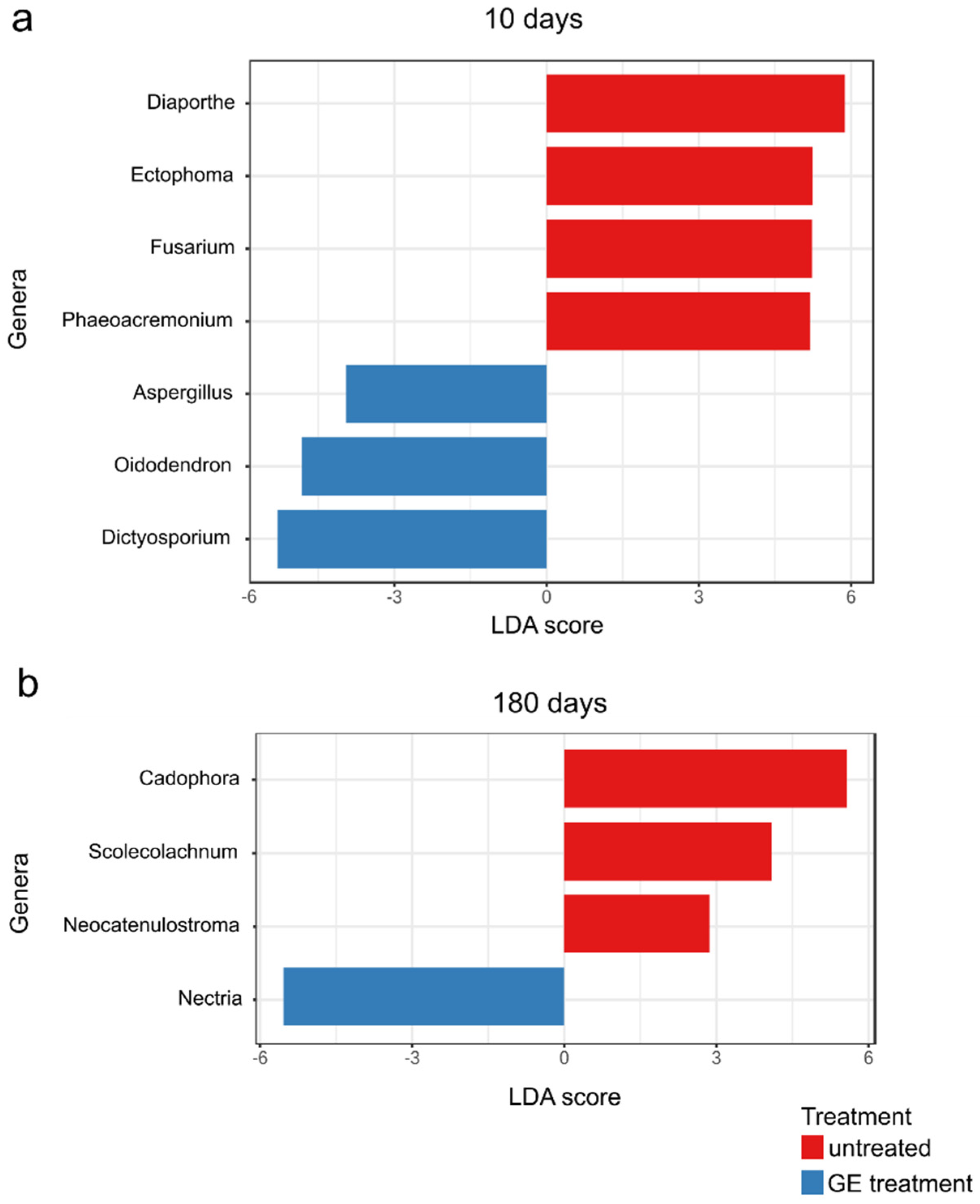
3.7. The Natural Infection Rates Caused by Fungal Trunk Pathogens Differed between Treated Untreated Plants
3.8. The Non-GTD Mycobiota Varied among Treated × Untreated Plants
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- OICV. Statistical Report on World Vitiviniculture; International Organisation of Vine and Wine, Intergovernmental Organisation: Paris, France, 2019. [Google Scholar]
- Fontaine, F.; Pinto, C.; Vallet, J.; Clément, C.; Gomes, A.C.; Spagnolo, A. The effects of grapevine trunk diseases (GTDs) on vine physiology. Eur. J. Plant Pathol. 2016, 144, 707–721. [Google Scholar] [CrossRef]
- Hofstetter, V.; Buyck, B.; Croll, D.; Viret, O.; Couloux, A.; Gindro, K. What if esca disease of grapevine were not a fungal disease? Fungal Divers. 2012, 54, 51–67. [Google Scholar] [CrossRef] [Green Version]
- Marion, C.; Martin, N.; Florence, F.; Jacques, W. Current knowledge on grapevine trunk diseases with complex etiology: A systemic approach. Phytopathol. Mediterr. 2020, 59, 29–53. [Google Scholar] [CrossRef]
- Lucia, G.-D.; Florence, F.; Laura, M. Grapevine trunk disease in European and Mediterranean vineyards: Occurrence, distribution and associated disease-affecting cultural factors. Phytopathol. Mediterr. 2019, 58, 49–71. [Google Scholar] [CrossRef]
- Gramaje, D.; Armengol, J. Fungal trunk pathogens in the grapevine propagation process: Potential inoculum sources, detection, identification, and management strategies. Plant Dis. 2011, 95, 1040–1055. [Google Scholar] [CrossRef] [Green Version]
- Gramaje, D.; Úrbez-Torres, J.R.; Sosnowski, M.R. Managing grapevine trunk diseases with respect to etiology and epidemiology: Current strategies and future prospects. Plant Dis. 2018, 102, 12–39. [Google Scholar] [CrossRef] [Green Version]
- Úrbez-Torres, J.R.; Peduto, F.; Smith, R.J.; Gubler, W.D. Phomopsis dieback: A grapevine trunk disease caused by Phomopsis viticola in California. Plant Dis. 2013, 97, 1571–1579. [Google Scholar] [CrossRef] [Green Version]
- Guarnaccia, V.; Groenewald, J.Z.; Woodhall, J.; Armengol, J.; Cinelli, T.; Eichmeier, A.; Ezra, D.; Florence, F.; Gramaje, D.; Gutierrez-Aguirregabiria, A.; et al. Diaporthe diversity and pathogenicity revealed from a broad survey of grapevine diseases in Europe. Pers. -Mol. Phylogeny Evol. Fungi 2018, 40, 135–153. [Google Scholar] [CrossRef] [Green Version]
- Mondello, V.; Songy, A.; Battiston, E.; Pinto, C.; Coppin, C.; Trotel-Aziz, P.; Clément, C.; Mugnai, L.; Fontaine, F. Grapevine trunk diseases: A review of fifteen years of trials for their control with chemicals and biocontrol agents. Plant Dis. 2018, 102, 1189–1217. [Google Scholar] [CrossRef] [Green Version]
- Vek, V.; Keržič, E.; Poljanšek, I.; Eklund, P.; Humar, M.; Oven, P. Wood extractives of silver fir and their antioxidant and antifungal properties. Molecules 2021, 26, 6412. [Google Scholar] [CrossRef]
- Kawamura, F.; Ramle, S.F.M.; Sulaiman, O.; Hashim, R.; Ohara, S. Antioxidant and antifungal activities of extracts from 15 selected hardwood species of Malaysian timber. Eur. J. Wood Wood Prod. 2011, 69, 207–212. [Google Scholar] [CrossRef]
- Salem, M.Z.M.; Elansary, H.O.; Elkelish, A.A.; Zeidler, A.; Ali, H.M.; EL-Hefny, M.; Yessoufou, K. In vitro bioactivity and antimicrobial activity of Picea abies and Larix decidua wood and bark extracts. BioResources 2016, 11, 17. [Google Scholar] [CrossRef] [Green Version]
- Minova, S.; Sešķēna, R.; Voitkāne, S.; Metla, Z.; Daugavietis, M.; Jankevica, L. Impact of pine (L.) and spruce ((L.) Karst.) bark extracts on important strawberry pathogens. Proc. Latv. Acad. Sci. Sect. B. Nat. Exact Appl. Sci. 2015, 69, 62–67. [Google Scholar] [CrossRef] [Green Version]
- Latva-Mäenpää, H.; Laakso, T.; Sarjala, T.; Wähälä, K.; Saranpää, P. Root neck of Norway spruce as a source of bioactive lignans and stilbenes. Holzforschung 2014, 68, 1–7. [Google Scholar] [CrossRef]
- Shain, L.; Hillis, E.W. Phenolic extractives in Norway spruce and their effects on Fomes annosus. Phytopathology 1971, 61, 841–845. [Google Scholar] [CrossRef]
- Widad, M.K.A.-A.; Fitua, M.A. Antimicrobial activity of hydroxymatairesinol (HMR) lignan. Iraqi J. Pharm. Sci. 2013, 22, 30–34. [Google Scholar] [CrossRef]
- Blanchette, R.A.; Biggs, A.R. Defense mechanisms of woody plants against fungi. In Springer Series in Wood Science; Springer: Berlin/Heidelberg, Germany, 1992. [Google Scholar]
- Balík, J.; Híc, P.; Tříska, J.; Vrchotová, N.; Smetana, P.; Smutek, L.; Rohlik, B.-A.; Houška, M. Beer and beer-based beverage contain lignans. J. Food Sci. Technol. 2021, 58, 581–585. [Google Scholar] [CrossRef]
- Brennan, M.; Hentges, D.; Cosgun, S.; Dumarcay, S.; Colin, F.; Gérardin, C.; Gérardin, P. Intraspecific variability of quantity and chemical composition of ethanolic knotwood extracts along the stems of three industrially important softwood species: Abies alba, Picea abies and Pseudotsuga menziesii. Holzforschung 2021, 75, 168–179. [Google Scholar] [CrossRef]
- Eichmeier, A.; Pečenka, J.; Peňázová, E.; Baránek, M.; Català-García, S.; León, M.; Armengol, J.; Gramaje, D. High-throughput amplicon sequencing-based analysis of active fungal communities inhabiting grapevine after hot-water treatments reveals unexpectedly high fungal diversity. Fungal Ecol. 2018, 36, 26–38. [Google Scholar] [CrossRef]
- Bruez, E.; Vallance, J.; Gautier, A.; Laval, V.; Compant, S.; Maurer, W.; Sessitsch, A.; Lebrun, M.-H.; Rey, P. Major changes in grapevine wood microbiota are associated with the onset of esca, a devastating trunk disease. Environ. Microbiol. 2020, 22, 5189–5206. [Google Scholar] [CrossRef]
- White, T.J.; Bruns, T.; Lee, S.; Taylor, J. Amplification and direct sequencing of fungal ribosomal RNA Genes for phylogenetics. In PCR Protocols; Innis, M.A., Gelfand, D.H., Sninsky, J.J., White, T.J., Eds.; Academic Press: San Diego, CA, USA, 1990; pp. 315–322. [Google Scholar]
- Andrews, S. A Quality Control Tool for High Throughput Sequence Data. 2010. Available online: http://www.bioinformatics.babraham.ac.uk/projects/fastqc (accessed on 30 August 2021).
- Větrovský, T.; Baldrian, P.; Morais, D. SEED 2: A user-friendly platform for amplicon high-throughput sequencing data analyses. Bioinformatics 2018, 34, 2292–2294. [Google Scholar] [CrossRef]
- Aronesty, E. EA-Utils: Command-Line Tools for Processing Biological Sequencing Data; Erik Aronesty: Durham, NC, USA, 2011. [Google Scholar]
- Bengtsson-Palme, J.; Ryberg, M.; Hartmann, M.; Branco, S.; Wang, Z.; Godhe, A.; De Wit, P.; Sánchez-García, M.; Ebersberger, I.; de Sousa, F.; et al. Improved software detection and extraction of ITS1 and ITS2 from ribosomal ITS sequences of fungi and other eukaryotes for analysis of environmental sequencing data. Methods Ecol. Evol. 2013, 4, 914–919. [Google Scholar] [CrossRef]
- Edgar, R.C. UPARSE: Highly accurate OTU sequences from microbial amplicon reads. Nat. Methods 2013, 10, 996–998. [Google Scholar] [CrossRef]
- Abarenkov, K.; Henrik Nilsson, R.; Larsson, K.-H.; Alexander, I.J.; Eberhardt, U.; Erland, S.; Høiland, K.; Kjøller, R.; Larsson, E.; Pennanen, T.; et al. The UNITE database for molecular identification of fungi—recent updates and future perspectives. New Phytol. 2010, 186, 281–285. [Google Scholar] [CrossRef]
- Glynou, K.; Nam, B.; Thines, M.; Maciá-Vicente, J.G. Facultative root-colonizing fungi dominate endophytic assemblages in roots of nonmycorrhizal Microthlaspi species. New Phytol. 2018, 217, 1190–1202. [Google Scholar] [CrossRef] [Green Version]
- Dhariwal, A.; Chong, J.; Habib, S.; King, I.L.; Agellon, L.B.; Xia, J. MicrobiomeAnalyst: A web-based tool for comprehensive statistical, visual and meta-analysis of microbiome data. Nucleic Acids Res. 2017, 45, W180–W188. [Google Scholar] [CrossRef]
- Bertsch, C.; Ramírez-Suero, M.; Magnin-Robert, M.; Larignon, P.; Chong, J.; Abou-Mansour, E.; Spagnolo, A.; Clément, C.; Fontaine, F. Grapevine trunk diseases: Complex and still poorly understood. Plant Pathol. 2013, 62, 243–265. [Google Scholar] [CrossRef] [Green Version]
- Fontaine, F.; Gramaje, D.; Armengol, J.; Smart, R.; Nagy, Z.A.; Borgo, M.; Rego, C.; Corio-Costet, M.-F. Grapevine Trunk Diseases. A Review; OIV Publications: Paris, France, 2016; 24p. [Google Scholar]
- Komárek, M.; Čadková, E.; Chrastný, V.; Bordas, F.; Bollinger, J.-C. Contamination of vineyard soils with fungicides: A review of environmental and toxicological aspects. Environ. Int. 2010, 36, 138–151. [Google Scholar] [CrossRef]
- Milićević, T.; Relić, D. Environmental and human health risk assessment in vineyards based on potentially toxic elements in soil-grapevine-air system. In Agricultural Research Updates; Nova Science Publishers: New York, NY, USA, 2021; Volume 36, pp. 1–75. [Google Scholar]
- Vek, V.; Balzano, A.; Poljanšek, I.; Humar, M.; Oven, P. Improving fungal decay resistance of less durable sapwood by impregnation with scots pine knotwood and black locust heartwood hydrophilic extractives with antifungal or antioxidant properties. Forests 2020, 11, 1024. [Google Scholar] [CrossRef]
- Vek, V.; Poljanšek, I.; Humar, M.; Willför, S.; Oven, P. In vitro inhibition of extractives from knotwood of Scots pine (Pinus sylvestris) and black pine (Pinus nigra) on growth of Schizophyllum commune, Trametes versicolor, Gloeophyllum trabeum and Fibroporia vaillantii. Wood Sci. Technol. 2020, 54, 1645–1662. [Google Scholar] [CrossRef]
- Välimaa, A.L.; Honkalampi-Hämäläinen, U.; Pietarinen, S.; Willför, S.; Holmbom, B.; von Wright, A. Antimicrobial and cytotoxic knotwood extracts and related pure compounds and their effects on food-associated microorganisms. Int. J. Food Microbiol. 2007, 115, 235–243. [Google Scholar] [CrossRef]
- Gramaje, D.; Baumgartner, K.; Halleen, F.; Mostert, L.; Sosnowski, M.R.; Úrbez-Torres, J.R.; Armengol, J. Fungal trunk diseases: A problem beyond grapevines? Plant Pathol. 2016, 65, 355–356. [Google Scholar] [CrossRef]
- Mostert, L.; Groenewald, J.Z.; Summerbell, R.C.; Gams, W.; Crous, P.W. Taxonomy and pathology of Togninia (Diaporthales) and its Phaeoacremonium anamorphs. Stud. Mycol. 2006, 54, 1–113. [Google Scholar] [CrossRef] [Green Version]
- Gramaje, D.; Mostert, L.; Groenewald, J.Z.; Crous, P.W. Phaeoacremonium: From esca disease to phaeohyphomycosis. Fungal Biol. 2015, 119, 759–783. [Google Scholar] [CrossRef]
- Aigoun-Mouhous, W.; Eichmeier, A.; Armengol, J.; Gramaje, D.; León, M.; Chaouia, C.; Zitouni, A.; Mahamedi, A.E.; Barankova, K.; Berraf-Tebbal, A. Cadophora sabaouae sp. nov. and Phaeoacremonium species associated with Petri disease on grapevine propagation material and young grapevines in Algeria. Plant Dis. 2021, 105, 3657–3668. [Google Scholar] [CrossRef]
- David, G.; Lizel, M.; Josep, A. Characterization of Cadophora luteo-olivacea and C. melinii isolates obtained from grapevines and environmental samples from grapevine nurseries in Spain. Phytopathol. Mediterr. 2011, 50, S112–S126. [Google Scholar] [CrossRef]
- Halleen, F.; Mostert, L.; Crous, P.W. Pathogenicity testing of lesser-known vascular fungi of grapevines. Australas. Plant Pathol. 2007, 36, 277–285. [Google Scholar] [CrossRef]
- Úrbez-Torres, J.R.; Haag, P.; Bowen, P.; O’Gorman, D.T. Grapevine trunk diseases in British Columbia: Incidence and characterization of the fungal pathogens associated with esca and petri diseases of grapevine. Plant Dis. 2013, 98, 469–482. [Google Scholar] [CrossRef] [Green Version]
- Overton, B.; Stewart, E.; Wenner, N. Molecular phylogenetics of grapevine decline fungi from Pennsylvania and New York. Phytopathol. Mediterr. 2005, 44, 90–91. [Google Scholar]
- Travadon, R.; Lawrence, D.P.; Rooney-Latham, S.; Gubler, W.D.; Wilcox, W.F.; Rolshausen, P.E.; Baumgartner, K. Cadophora species associated with wood-decay of grapevine in North America. Fungal Biol. 2015, 119, 53–66. [Google Scholar] [CrossRef]
- Antonia, C.; Francesco, L.; Lizel, M.; Francois, H.; Maria, R. Occurrence fungi causing black foot on young grapevines and nursery rootstock plants in Italy. Phytopathol. Mediterr. 2017, 56, 10–39. [Google Scholar] [CrossRef]
- Reis, P.; Cabral, A.; Nascimento, T.; Oliveira, H.; Rego, C. Diversity of Ilyonectria species in a young vineyard affected by black foot disease. Phytopathol. Mediterr. 2013, 52, 335–346. [Google Scholar] [CrossRef]
- Berlanas, C.; López-Manzanares, B.; Gramaje, D. Estimation of viable propagules of black-foot disease pathogens in grapevine cultivated soils and their relation to production systems and soil properties. Plant Soil 2017, 417, 467–479. [Google Scholar] [CrossRef]
- Pečenka, J.; Eichmeier, A.; Peňázová, E.; Baránek, M.; León, M.; Armengol, J. First report of Dactylonectria torresensis causing black-foot disease on grapevines in the Czech Republic. Plant Dis. 2018, 102, 2038. [Google Scholar] [CrossRef]
- Agustí-Brisach, C.; Gramaje, D.; García-Jiménez, J.; Armengol, J. Detection of black-foot disease pathogens in the grapevine nursery propagation process in Spain. Eur. J. Plant Pathol. 2013, 137, 103–112. [Google Scholar] [CrossRef]
- Güngör-Savaş, N.; Akgül, D.S.; Özarslandan, M.; Yıldız, M. First report of Dactylonectria alcacerensis and Dactylonectria torresensis associated with black Foot disease of grapevine in Turkey. Plant Dis. 2020, 104, 2027. [Google Scholar] [CrossRef] [Green Version]
- Berlanas, C.; Ojeda, S.; López-Manzanares, B.; Andrés-Sodupe, M.; Bujanda, R.; del Pilar Martínez-Diz, M.; Díaz-Losada, E.; Gramaje, D. Occurrence and diversity of black-foot disease fungi in symptomless grapevine nursery stock in Spain. Plant Dis. 2020, 104, 94–104. [Google Scholar] [CrossRef]
- Eichmeier, A.; Kiss, T.; Penazova, E.; Pecenka, J.; Berraf-Tebbal, A.; Baranek, M.; Pokluda, R.; Cechova, J.; Gramaje, D.; Grzebelus, D. MicroRNAs in Vitis vinifera cv. Chardonnay are differentially expressed in response to Diaporthe species. Genes 2019, 10, 905. [Google Scholar] [CrossRef] [Green Version]
- Mundy, D.C.; Brown, A.; Jacobo, F.; Tennakoon, K.; Woolley, R.H.; Vanga, B.; Tyson, J.; Johnston, P.; Ridgway, H.J.; Bulman, S. Pathogenic fungi isolated in association with grapevine trunk diseases in New Zealand. N. Z. J. Crop Hortic. Sci. 2020, 48, 84–96. [Google Scholar] [CrossRef]
- Lawrence, D.P.; Travadon, R.; Baumgartner, K. Diversity of Diaporthe species associated with wood cankers of fruit and nut crops in northern California. Mycologia 2015, 107, 926–940. [Google Scholar] [CrossRef]
- Baumgartner, K.; Fujiyoshi, P.T.; Travadon, R.; Castlebury, L.A.; Wilcox, W.F.; Rolshausen, P.E. Characterization of species of Diaporthe from wood cankers of grape in Eastern North American vineyards. Plant Dis. 2013, 97, 912–920. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pitt, W.M.; Huang, R.; Trouillas, F.P.; Steel, C.C.; Savocchia, S. Evidence that Eutypa lata and other diatrypaceous species occur in New South Wales vineyards. Australas. Plant Pathol. 2010, 39, 97–106. [Google Scholar] [CrossRef]
- Rolshausen, P.E.; Greve, L.C.; Labavitch, J.M.; Mahoney, N.E.; Molyneux, R.J.; Gubler, W.D. Pathogenesis of Eutypa lata in grapevine: Identification of virulence factors and biochemical characterization of cordon dieback. Phytopathology 2008, 98, 222–229. [Google Scholar] [CrossRef] [Green Version]
- Trouillas, F.P.; Gubler, W.D. Host range, biological variation, and phylogenetic diversity of Eutypa lata in California. Phytopathology 2010, 100, 1048–1056. [Google Scholar] [CrossRef] [Green Version]
- Živković, S.; Vasić, T.; Ivanović, M.; Jevremović, D.; Marković, J.; Trkulja, V. Morphological and molecular identification of Eutypa lata on grapevine in Serbia. J. Plant Dis. Prot. 2019, 126, 479–483. [Google Scholar] [CrossRef]
- Lardner, R.; Stummer, B.E.; Sosnowski, M.R.; Scott, E.S. Molecular identification and detection of Eutypa lata in grapevine. Mycol. Res. 2005, 109, 799–808. [Google Scholar] [CrossRef]
- Moisy, C.; Berger, G.; Flutre, T.; Le Cunff, L.; Péros, J.-P. Quantitative assessment of grapevine wood colonization by the dieback fungus Eutypa lata. J. Fungi 2017, 3, 21. [Google Scholar] [CrossRef] [Green Version]
- Elena, G.; Garcia-Figueres, F.; Reigada, S.; Luque, J. Intraspecific variation in Diplodia seriata isolates occurring on grapevines in Spain. Plant Pathol. 2015, 64, 680–689. [Google Scholar] [CrossRef]
- Mohammadi, H.; Gramaje, D.; Banihashemi, Z.; Armengol, J. Characterization of Diplodia seriata and Neofusicoccum parvum associated with grapevine decline in Iran. J. Agric. Sci. Technol. 2013, 15, 603–616. [Google Scholar]
- Phillips, A.J.L.; Crous, P.W.; Alves, A. Diplodia seriata, the anamorph of “Botryosphaeria” obtusa. Fungal Divers. 2007, 25, 141–155. [Google Scholar]
- Linaldeddu, B.T.; Deidda, A.; Scanu, B.; Franceschini, A.; Serra, S.; Berraf-Tebbal, A.; Zouaoui Boutiti, M.; Ben Jamâa, M.L.; Phillips, A.J.L. Diversity of Botryosphaeriaceae species associated with grapevine and other woody hosts in Italy, Algeria and Tunisia, with descriptions of Lasiodiplodia exigua and Lasiodiplodia mediterranea sp. nov. Fungal Divers. 2015, 71, 201–214. [Google Scholar] [CrossRef]
- Sosnowski, M.R.; Ayres, M.R.; McCarthy, M.G.; Scott, E.S. Winegrape cultivars (Vitis vinifera) vary in susceptibility to the grapevine trunk pathogens Eutypa lata and Diplodia seriata. Aust. J. Grape Wine Res. 2022, 28, 166–174. [Google Scholar] [CrossRef]
- Karličić, V.; Jovičić-Petrović, J.; Marojević, V.; Zlatković, M.; Orlović, S.; Raičević, V. Potential of Trichoderma spp. and Pinus sylvestris bark extracts as biocontrol agents against fungal pathogens residing in the Botryosphaeriales. Environ. Sci. Proc. 2021, 3, 99. [Google Scholar] [CrossRef]
- Karličić, V.; Zlatković, M.; Jovičić-Petrović, J.; Nikolić, M.P.; Orlović, S.; Raičević, V. Trichoderma spp. from pine bark and pine bark extracts: Potent biocontrol agents against Botryosphaeriaceae. Forests 2021, 12, 1731. [Google Scholar] [CrossRef]
- Piispanen, R.; Willför, S.; Saranpää, P.; Holmbom, B. Variation of lignans in Norway spruce (Picea abies [L.] Karst.) knotwood: Within-stem variation and the effect of fertilisation at two experimental sites in Finland. Trees 2008, 22, 317–328. [Google Scholar] [CrossRef]
- Valette, N.; Perrot, T.; Sormani, R.; Gelhaye, E.; Morel-Rouhier, M. Antifungal activities of wood extractives. Fungal Biol. Rev. 2017, 31, 113–123. [Google Scholar] [CrossRef]
- Kusumoto, N.; Zhao, T.; Swedjemark, G.; Ashitani, T.; Takahashi, K.; Borg-Karlson, A.-K. Antifungal properties of terpenoids in Picea abies against Heterobasidion parviporum. For. Pathol. 2014, 44, 353–361. [Google Scholar] [CrossRef]
- Rudman, P. Literaturberichte. Holzforschung 1965, 19, 58–63. [Google Scholar] [CrossRef]
- Wei, Y.-J.; Wu, Y.; Yan, Y.-Z.; Zou, W.; Xue, J.; Ma, W.-R.; Wang, W.; Tian, G.; Wang, L.-Y. High-throughput sequencing of microbial community diversity in soil, grapes, leaves, grape juice and wine of grapevine from China. PLoS ONE 2018, 13, e0193097. [Google Scholar] [CrossRef] [Green Version]
- Martínez-Diz, M.d.P.; Andrés-Sodupe, M.; Bujanda, R.; Díaz-Losada, E.; Eichmeier, A.; Gramaje, D. Soil-plant compartments affect fungal microbiome diversity and composition in grapevine. Fungal Ecol. 2019, 41, 234–244. [Google Scholar] [CrossRef]
- Coller, E.; Cestaro, A.; Zanzotti, R.; Bertoldi, D.; Pindo, M.; Larger, S.; Albanese, D.; Mescalchin, E.; Donati, C. Microbiome of vineyard soils is shaped by geography and management. Microbiome 2019, 7, 140. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carbone, M.J.; Alaniz, S.; Mondino, P.; Gelabert, M.; Eichmeier, A.; Tekielska, D.; Bujanda, R.; Gramaje, D. Drought influences fungal community dynamics in the grapevine rhizosphere and root microbiome. J. Fungi 2021, 7, 686. [Google Scholar] [CrossRef] [PubMed]
- Castañeda, L.E.; Barbosa, O. Metagenomic analysis exploring taxonomic and functional diversity of soil microbial communities in Chilean vineyards and surrounding native forests. PeerJ 2017, 5, e3098. [Google Scholar] [CrossRef]
- Knapp, D.G.; Lázár, A.; Molnár, A.; Vajna, B.; Karácsony, Z.; Váczy, K.Z.; Kovács, G.M. Above-ground parts of white grapevine Vitis vinifera cv. Furmint share core members of the fungal microbiome. Environ. Microbiol. Rep. 2021, 13, 509–520. [Google Scholar] [CrossRef]
- Swift, J.F.; Hall, M.E.; Harris, Z.N.; Kwasniewski, M.T.; Miller, A.J. Grapevine microbiota reflect diversity among compartments and complex interactions within and among root and shoot systems. Microorganisms 2021, 9, 92. [Google Scholar] [CrossRef]
- Deyett, E.; Rolshausen, P.E. Endophytic microbial assemblage in grapevine. FEMS Microbiol. Ecol. 2020, 96, fiaa053. [Google Scholar] [CrossRef]
- Dissanayake, A.J.; Purahong, W.; Wubet, T.; Hyde, K.D.; Zhang, W.; Xu, H.; Zhang, G.; Fu, C.; Liu, M.; Xing, Q.; et al. Direct comparison of culture-dependent and culture-independent molecular approaches reveal the diversity of fungal endophytic communities in stems of grapevine (Vitis vinifera). Fungal Divers. 2018, 90, 85–107. [Google Scholar] [CrossRef]
- Jayawardena, R.S.; Purahong, W.; Zhang, W.; Wubet, T.; Li, X.; Liu, M.; Zhao, W.; Hyde, K.D.; Liu, J.; Yan, J. Biodiversity of fungi on Vitis vinifera L. revealed by traditional and high-resolution culture-independent approaches. Fungal Divers. 2018, 90, 1–84. [Google Scholar] [CrossRef] [Green Version]
- Del Frari, G.; Gobbi, A.; Aggerbeck, M.R.; Oliveira, H.; Hansen, L.H.; Ferreira, R.B. Characterization of the wood mycobiome of Vitis vinifera in a vineyard affected by Esca. spatial distribution of fungal communities and their putative relation with leaf symptoms. Front. Plant Sci. 2019, 10, 910. [Google Scholar] [CrossRef] [Green Version]
- Martínez-Diz, M.d.P.; Eichmeier, A.; Spetik, M.; Bujanda, R.; Díaz-Fernández, Á.; Díaz-Losada, E.; Gramaje, D. Grapevine pruning time affects natural wound colonization by wood-invading fungi. Fungal Ecol. 2020, 48, 100994. [Google Scholar] [CrossRef]
- Niem, J.M.; Billones-Baaijens, R.; Stodart, B.; Savocchia, S. Diversity profiling of grapevine microbial endosphere and antagonistic potential of endophytic Pseudomonas against grapevine trunk diseases. Front. Microbiol. 2020, 11, 477. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Diz, M.d.P.; Díaz-Losada, E.; Andrés-Sodupe, M.; Bujanda, R.; Maldonado-González, M.M.; Ojeda, S.; Yacoub, A.; Rey, P.; Gramaje, D. Field evaluation of biocontrol agents against black-foot and Petri diseases of grapevine. Pest Manag. Sci. 2021, 77, 697–708. [Google Scholar] [CrossRef] [PubMed]
- Del Frari, G.; Gobbi, A.; Aggerbeck, M.R.; Oliveira, H.; Hansen, L.H.; Ferreira, R.B. Fungicides and the grapevine wood mycobiome: A case study on tracheomycotic ascomycete Phaeomoniella chlamydospora reveals potential for two novel control strategies. Front. Plant Sci. 2019, 10, 1405. [Google Scholar] [CrossRef] [PubMed]
- Rudman, P. The causes of natural durability in timber—part XI. some tests on the fungi toxicity of wood extractives and related compounds. Holzforschung 1963, 17, 54–57. [Google Scholar] [CrossRef]
- Gramaje, D.; Eichmeier, A.; Spetik, M.; Carbone, M.J.; Bujanda, R.; Vallance, J.; Rey, P. Exploring the temporal dynamics of the fungal microbiome in rootstocks, the lesser-known half of the grapevine crop. Environ. Microbiome 2021. [Google Scholar] [CrossRef]
- Almeida, A.B.d.; Concas, J.; Campos, M.D.; Materatski, P.; Varanda, C.; Patanita, M.; Murolo, S.; Romanazzi, G.; Félix, M.d.R. Endophytic fungi as potential biological control agents against grapevine trunk diseases in Alentejo region. Biology 2020, 9, 420. [Google Scholar] [CrossRef]
- Ziedan, E.-S.H.; Embaby, E.-S.M.; Farrag, E.S. First record of Fusarium vascular wilt on grapevine in Egypt. Arch. Phytopathol. Plant Prot. 2011, 44, 1719–1727. [Google Scholar] [CrossRef]
- Highet, A.S.; Nair, N.G. Fusarium oxysporum associated with grapevine decline in the Hunter Valley, NSW, Australia. Aust. J. Grape Wine Res. 1995, 1, 48–50. [Google Scholar] [CrossRef]
- Omer, A.D.; Granett, J.; Wakeman, R.J. Pathogenicity of Fusarium oxysporum on different Vitis rootstocks. J. Phytopathol. 1999, 147, 433–436. [Google Scholar] [CrossRef]
- Grasso, S. Infection of Fusarium oxysporum and Cylindrocarpon destructans, associated with a grapevine decline in a vineyard of eastern Sicily. Inf. Fitopatol. 1984, 36, 59–63. [Google Scholar]
- Hemida, K.A.R.; Ziedan, E.H.E.; El-Saman, M.G.M.; El-Naggar, M.A.; Mostafa, H.M. Etiology of fungi associated with grapevine decline and their pathological potential. Arab. Univ. J. Agric. Sci. 2017, 25, 355–365. [Google Scholar] [CrossRef]
- Vincenzo, M.; Alessandro, S.; Philippe, L.; Christophe, C.; Florence, F. Phytoprotection potential of Fusarium proliferatum for control of Botryosphaeria dieback pathogens in grapevine. Phytopathol. Mediterr. 2019, 58, 293–306. [Google Scholar] [CrossRef]
- McMahan, G.; Yeh, W.; Marshall, M.N.; Olsen, M.; Sananikone, S.; Wu, J.Y.; Block, D.E.; VanderGheynst, J.S. Characterizing the production of a wild-type and benomyl-resistant Fusarium lateritium for biocontrol of Eutypa lata on grapevine. J. Ind. Microbiol. Biotechnol. 2001, 26, 151–155. [Google Scholar] [CrossRef] [PubMed]
- Billones-Baaijens, R.; Savocchia, S. A review of Botryosphaeriaceae species associated with grapevine trunk diseases in Australia and New Zealand. Australas. Plant Pathol. 2019, 48, 3–18. [Google Scholar] [CrossRef]
- Rego, C.; Nascimento, T.; Cabral, A.; Silva, M.J.; Oliveira, H. Control of grapevine wood fungi in commercial nurseries. Phytopathol. Mediterr. 2009, 48, 128–135. [Google Scholar]
- Fourie, P.H.; Halleen, F. Occurrence of grapevine trunk disease pathogens in rootstock mother plants in South Africa. Australas. Plant Pathol. 2004, 33, 313–315. [Google Scholar] [CrossRef]
- Varela, C.; Redondo, V.; Costas Imbernon, D.; Aguín, O.; Mansilla, P. Fungi associated with grapevine trunk diseases in nursery-produced Vitis vinifera plants. Phytopathol. Mediterr. 2019, 57, 407–424. [Google Scholar] [CrossRef]
- Lade, S.B.; Štraus, D.; Oliva, J. Variation in fungal community in grapevine (Vitis vinifera) nursery stock depends on nursery, variety and rootstock. J. Fungi 2022, 8, 47. [Google Scholar] [CrossRef]
- Navarrete, F.; Abreo, E.; Martínez, S.; Bettucci, L.; Lupo, S. Pathogenicity and molecular detection of Uruguayan isolates of Greeneria uvicola and Cadophora luteo-olivacea associated with grapevine trunk diseases. Phytopathol. Mediterr. 2011, 50, S166–S175. [Google Scholar]
- Rühl, E.H.; Manty, F.; Konrad, H.; Bleser, E. The importance of pathogen-free grapevine propagation material in regards to clonal selection and rootstock breeding in Germany. Int. J. Hortic. Sci. 2011, 17, 11–13. [Google Scholar] [CrossRef]
- John, S.; Wicks, T.J.; Hunt, J.S.; Lorimer, M.F.; Oakey, H.; Scott, E.S. Protection of grapevine pruning wounds from infection by Eutypa lata using Trichoderma harzianum and Fusarium lateritium. Australas. Plant Pathol. 2005, 34, 569–575. [Google Scholar] [CrossRef]
- Blundell, R.; Eskalen, A. Biological and chemical pruning wound protectants reduce infection of grapevine trunk disease pathogens. Calif. Agric. 2021, 75, 128–134. [Google Scholar] [CrossRef]
- Martinez-Diz, M.D.; Diaz-Losada, E.; Diaz-Fernandez, A.; Bouzas-Cid, Y.; Gramaje, D. Protection of grapevine pruning wounds against Phaeomoniella chlamydospora and Diplodia seriata by commercial biological and chemical methods. Crop Prot. 2021, 143, 105465. [Google Scholar] [CrossRef]
- Yacoub, A.; Haidar, R.; Gerbore, J.; Masson, C.; Dufour, M.C.; Guyoneaud, R.; Rey, P. Pythium oligandrum induces grapevine defence mechanisms against the trunk pathogen Neofusicoccum parvum. Phytopathol. Mediterr. 2020, 59, 565–580. [Google Scholar] [CrossRef]
- Ayres, M.R.; Wicks, T.J.; Scott, E.S.; Sosnowski, M.R. Developing pruning wound protection strategies for managing Eutypa dieback. Aust. J. Grape Wine Res. 2017, 23, 103–111. [Google Scholar] [CrossRef]
- Pitt, W.M.; Sosnowski, M.R.; Huang, R.J.; Qiu, Y.; Steel, C.C.; Savocchia, S. Evaluation of fungicides for the management of botryosphaeria canker of grapevines. Plant Dis. 2012, 96, 1303–1308. [Google Scholar] [CrossRef]
- Kotze, C.; Van Niekerk, J.; Mostert, L.; Halleen, F.; Fourie, P. Evaluation of biocontrol agents for grapevine pruning wound protection against trunk pathogen infection. Phytopathol. Mediterr. 2011, 50, S247–S263. [Google Scholar]
- Rolshausen, P.E.; Urbez-Torres, J.R.; Rooney-Latham, S.; Eskalen, A.; Smith, R.J.; Gubler, W.D. Evaluation of Pruning Wound Susceptibility and Protection Against Fungi Associated with Grapevine Trunk Diseases. Am. J. Enol. Vitic. 2010, 61, 113–119. [Google Scholar]
- Alvarez-Perez, J.M.; Gonzalez-Garcia, S.; Cobos, R.; Olego, M.A.; Ibanez, A.; Diez-Galan, A.; Garzon-Jimeno, E.; Coque, J.J.R. Use of endophytic and rhizospheric actinobacteria from grapevine plants to reduce fungal graft infections in nurseries that lead to young grapevine decline. Appl. Environ. Microbiol. 2017, 83, e01564-17. [Google Scholar] [CrossRef] [Green Version]
- Cobos, R.; Mateos, R.M.; Alvarez-Perez, J.M.; Olego, M.A.; Sevillano, S.; Gonzalez-Garcia, S.; Garzon-Jimeno, E.; Coque, J.J.R. Effectiveness of natural antifungal compounds in controlling infection by grapevine trunk disease pathogens through pruning wounds. Appl. Environ. Microbiol. 2015, 81, 6474–6483. [Google Scholar] [CrossRef] [Green Version]
- Rolshausen, P.E.; Gubler, W.D. Use of boron for the control of Eutypa dieback of grapevines. Plant Dis. 2005, 89, 734–738. [Google Scholar] [CrossRef] [PubMed]




Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Špetík, M.; Balík, J.; Híc, P.; Hakalová, E.; Štůsková, K.; Frejlichová, L.; Tříska, J.; Eichmeier, A. Lignans Extract from Knotwood of Norway Spruce—A Possible New Weapon against GTDs. J. Fungi 2022, 8, 357. https://doi.org/10.3390/jof8040357
Špetík M, Balík J, Híc P, Hakalová E, Štůsková K, Frejlichová L, Tříska J, Eichmeier A. Lignans Extract from Knotwood of Norway Spruce—A Possible New Weapon against GTDs. Journal of Fungi. 2022; 8(4):357. https://doi.org/10.3390/jof8040357
Chicago/Turabian StyleŠpetík, Milan, Josef Balík, Pavel Híc, Eliška Hakalová, Kateřina Štůsková, Lucie Frejlichová, Jan Tříska, and Aleš Eichmeier. 2022. "Lignans Extract from Knotwood of Norway Spruce—A Possible New Weapon against GTDs" Journal of Fungi 8, no. 4: 357. https://doi.org/10.3390/jof8040357





