A Novel FYVE Domain-Containing Protein Kinase, PsZFPK1, Plays a Critical Role in Vegetative Growth, Sporangium Formation, Oospore Production, and Virulence in Phytophthora sojae
Abstract
:1. Introduction
2. Materials and Methods
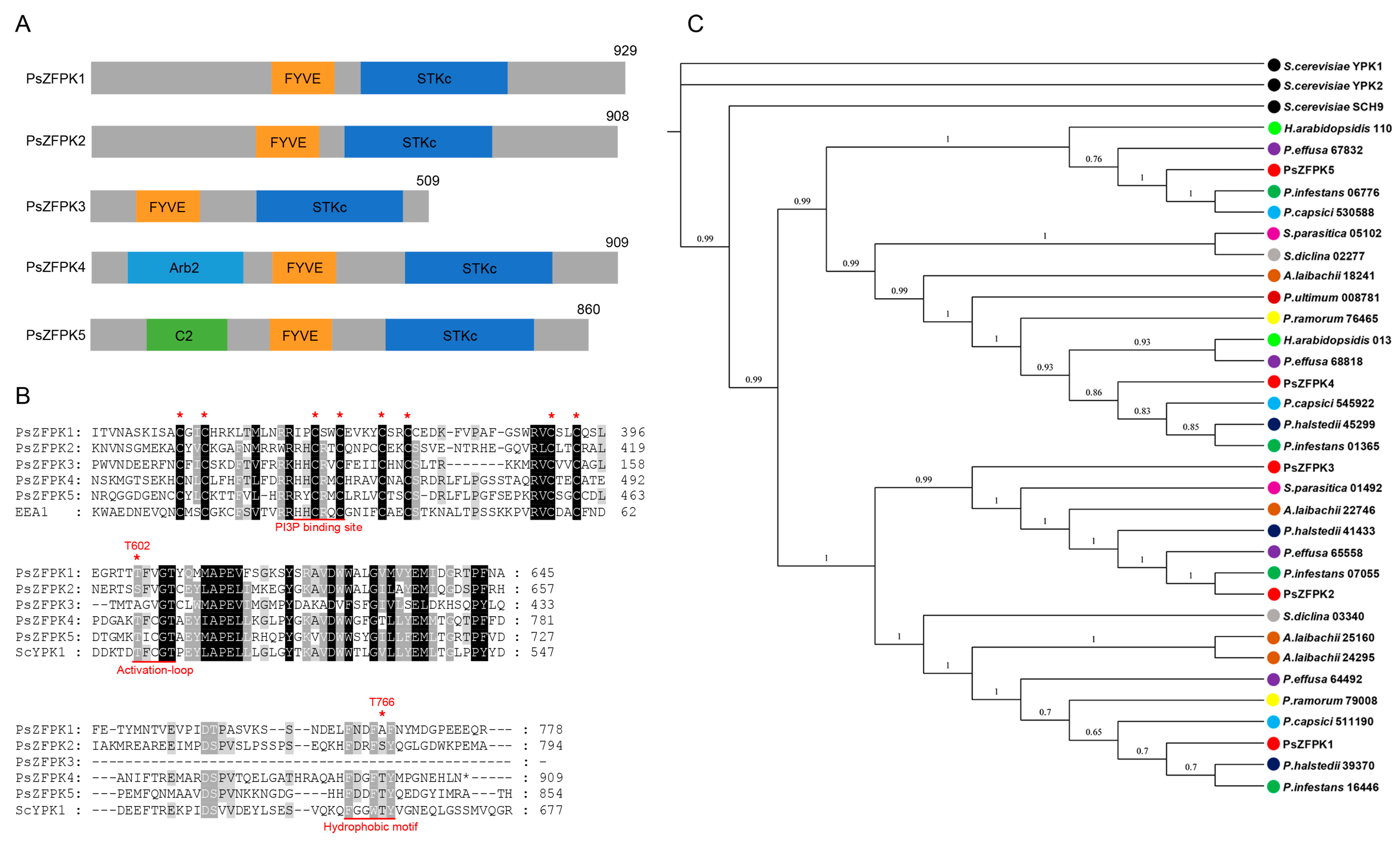
2.1. Sequence and Phylogenetic Analyses of PsZFPK1-5
2.2. P. sojae Strains and Culture Conditions
2.3. RNA Extraction and Gene Expression Analysis of PsZFPK1
2.4. Plasmid Construction
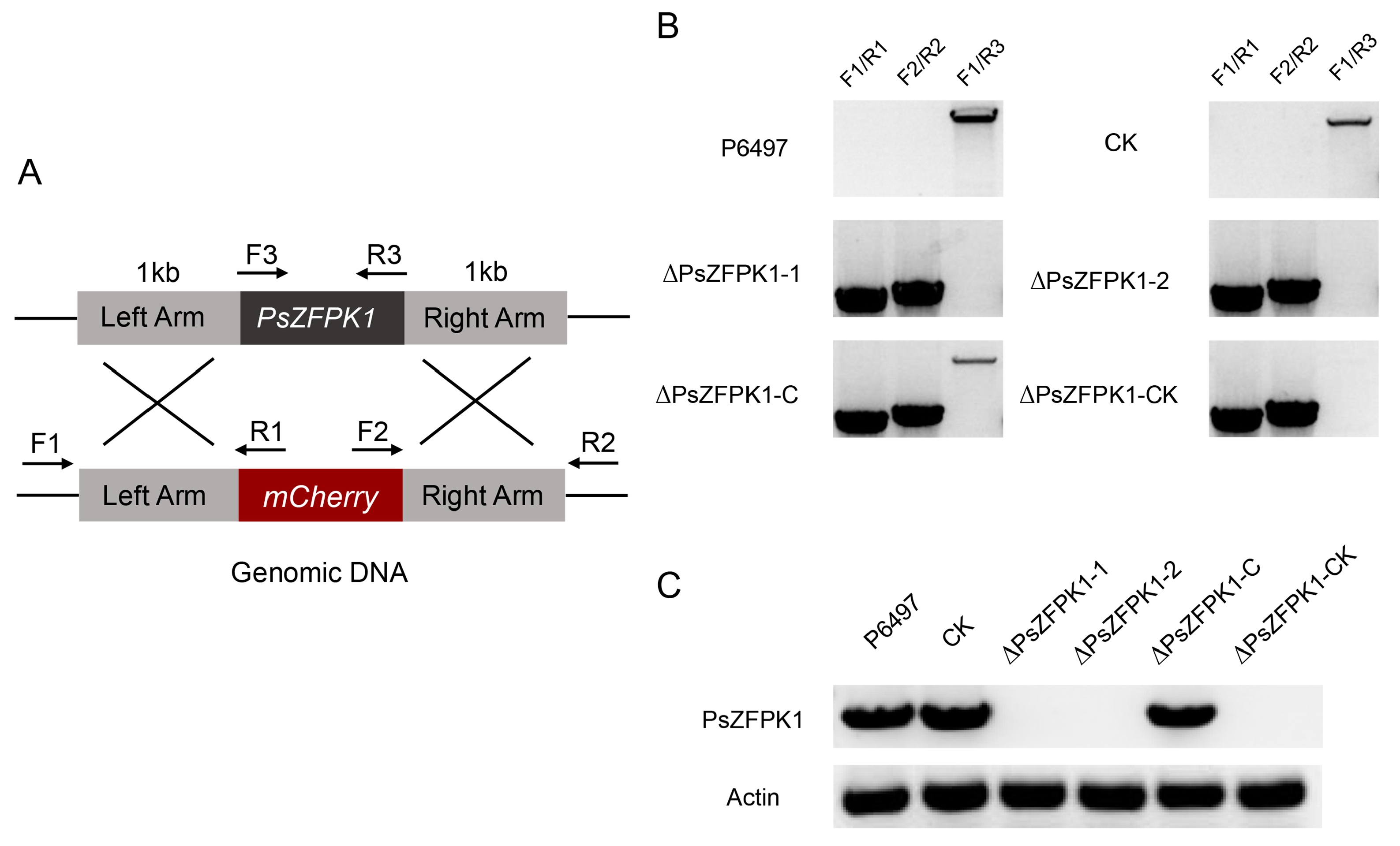
2.5. Generation of P. sojae Transformants
2.6. Phenotypic Analysis of Transformants
2.7. Subcellular Localization
3. Results
3.1. Sequence and Phylogenetic Analyses of Five Putative PsZFPKs
3.2. Expression Pattern Analysis of PsZFPK1
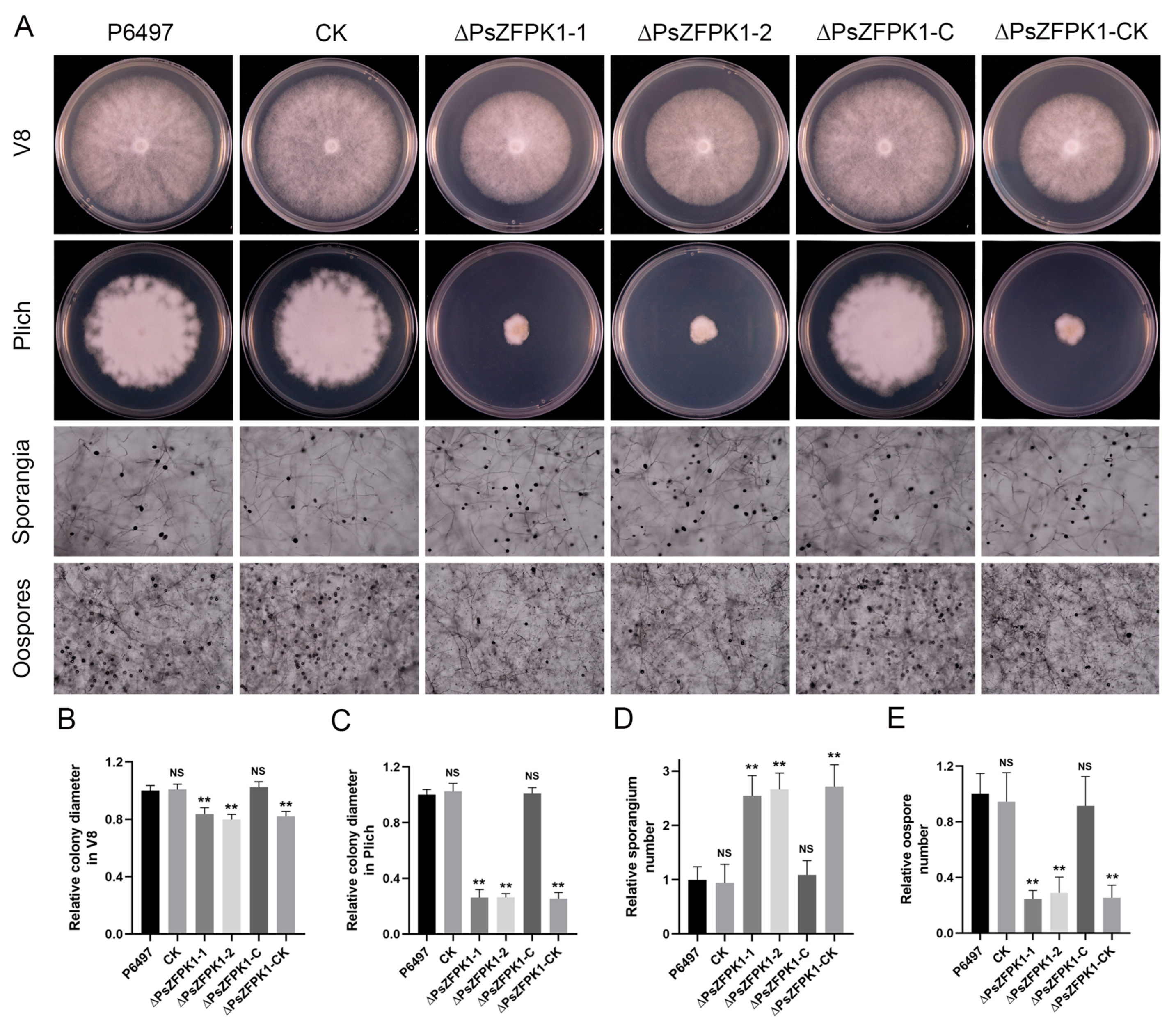
3.3. PsZFPK1 Plays a Critical Role in Vegetative Growth, Sporangium Formation, Oospore Production, and Virulence in P. sojae
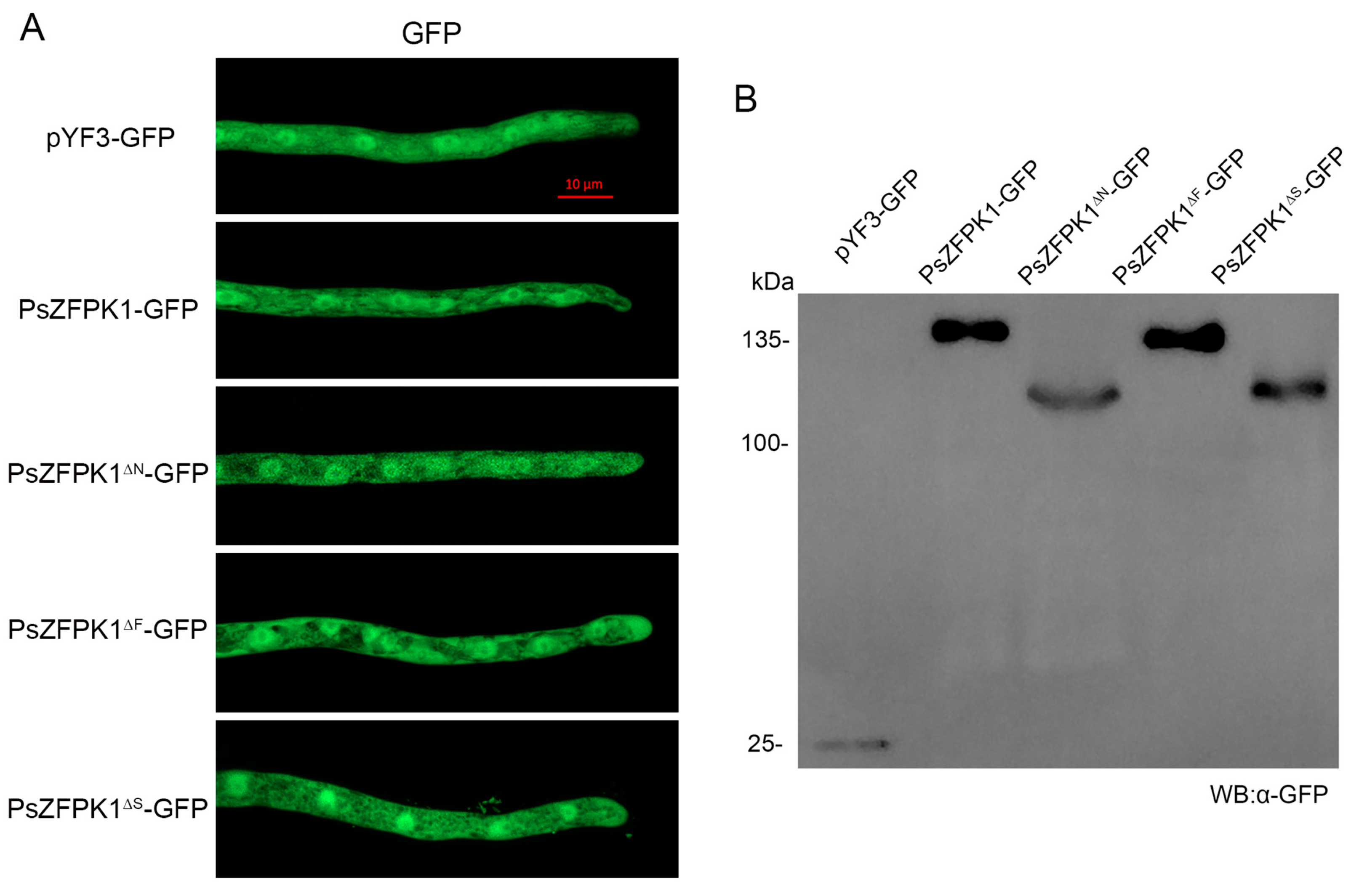
3.4. The N-Terminal Region, FYVE and STKc Domains, and T602 Phosphorylation Site Are Important for the Function of PsZFPK1
3.5. PsZFPK1 Is Not Necessary for the Stress Response
3.6. The Cytoplasmic Localization of PsZFPK1 Is Not Affected by the N-Terminal Region or the FYVE/STKc Domains
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Miao, J.; Liu, X.; Li, G.; Du, X.; Liu, X. Multiple point mutations in PsORP1 gene conferring different resistance levels to oxathiapiprolin confirmed using CRISPR-Cas9 in Phytophthora sojae. Pest Manag. Sci. 2020, 76, 2434–2440. [Google Scholar] [CrossRef]
- Tyler, B.M. Phytophthora sojae: Root rot pathogen of soybean and model oomycete. Mol. Plant Pathol. 2007, 8, 1–8. [Google Scholar] [CrossRef]
- Kamoun, S.; Furzer, O.; Jones, J.D.G.; Judelson, H.S.; Ali, G.S.; Dalio, R.J.D.; Roy, S.G.; Schena, L.; Zambounis, A.; Panabières, F.; et al. The Top 10 oomycete pathogens in molecular plant pathology. Mol. Plant Pathol. 2015, 16, 413–434. [Google Scholar] [CrossRef]
- Tyler, B.M.; Tripathy, S.; Zhang, X.; Dehal, P.; Jiang, R.H.Y.; Aerts, A.; Arredondo, F.D.; Baxter, L.; Bensasson, D.; Beynon, J.L.; et al. Phytophthora genome sequences uncover evolutionary origins and mechanisms of pathogenesis. Science 2006, 313, 1261–1266. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ye, W.; Wang, X.; Tao, K.; Lu, Y.; Dai, T.; Dong, S.; Dou, D.; Gijzen, M.; Wang, Y. Digital gene expression profiling of the Phytophthora sojae transcriptome. Mol. Plant-Microbe Interact. 2011, 24, 1530–1539. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fang, Y.; Tyler, B.M. Efficient disruption and replacement of an effector gene in the oomycete Phytophthora sojae using CRISPR/Cas9. Mol. Plant Pathol. 2016, 17, 127–139. [Google Scholar] [CrossRef]
- Fang, Y.; Cui, L.; Gu, B.; Arredondo, F.; Tyler, B.M. Efficient Genome Editing in the Oomycete Phytophthora sojae Using CRISPR/Cas9. Curr. Protoc. Microbiol. 2017, 44, 21A.1.1–21A.1.26. [Google Scholar] [CrossRef] [PubMed]
- Pearce, L.R.; Komander, D.; Alessi, D.R. The nuts and bolts of AGC protein kinases. Nat. Rev. Mol. Cell Biol. 2010, 11, 9–22. [Google Scholar] [CrossRef]
- Arencibia, J.M.; Pastor-Flores, D.; Bauer, A.F.; Schulze, J.O.; Biondi, R.M. AGC protein kinases: From structural mechanism of regulation to allosteric drug development for the treatment of human diseases. Biochim. Biophys. Acta 2013, 1834, 1302–1321. [Google Scholar] [CrossRef] [PubMed]
- Pokorny, D.; Truebestein, L.; Fleming, K.D.; Burke, J.E.; Leonard, T.A. In vitro reconstitution of Sgk3 activation by phosphatidylinositol 3-phosphate. J. Biol. Chem. 2021, 297, 100919. [Google Scholar] [CrossRef]
- Gu, Q.; Zhang, C.; Yu, F.; Yin, Y.; Shim, W.-B.; Ma, Z. Protein kinase FgSch9 serves as a mediator of the target of rapamycin and high osmolarity glycerol pathways and regulates multiple stress responses and secondary metabolism in Fusarium graminearum. Environ. Microbiol. 2015, 17, 2661–2676. [Google Scholar] [CrossRef]
- Wang, Y.; Deng, Y.Z.; Cui, G.; Huang, C.; Zhang, B.; Chang, C.; Jiang, Z.; Zhang, L.H. The AGC Kinase SsAgc1 Regulates Sporisorium scitamineum Mating/Filamentation and Pathogenicity. mSphere 2019, 4, e00259-19. [Google Scholar] [CrossRef] [Green Version]
- Wang, C.; Shen, D.; Wang, J.; Chen, Y.; Dong, Y.; Tang, Z.; Xia, A. An AGC kinase, PgAGC1 regulates virulence in the entomopathogenic oomycete Pythium guiyangense. Fungal Biol. 2019, 123, 87–93. [Google Scholar] [CrossRef]
- Qiu, M.; Li, Y.; Zhang, X.; Xuan, M.; Zhang, B.; Ye, W.; Zheng, X.; Govers, F.; Wang, Y. G protein α subunit suppresses sporangium formation through a serine/threonine protein kinase in Phytophthora sojae. PLoS Pathog. 2020, 16, e1008138. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gaullier, J.-M.; Simonsen, A.; D’Arrigo, A.; Bremnes, B.; Stenmark, H.; Aasland, R. FYVE fingers bind PtdIns(3)P. Nature 1998, 394, 432–433. [Google Scholar] [CrossRef] [PubMed]
- Stenmark, H.; Aasland, R.; Driscoll, P.C. The phosphatidylinositol 3-phosphate-binding FYVE finger. FEBS Lett. 2002, 513, 77–84. [Google Scholar] [CrossRef] [Green Version]
- Patki, V.; Lawe, D.C.; Corvera, S.; Virbasius, J.V.; Chawla, A. A functional PtdIns(3)P-binding motif. Nature 1998, 394, 433–434. [Google Scholar] [CrossRef] [PubMed]
- Gillooly, D.J.; Simonsen, A.; Stenmark, H. Cellular functions of phosphatidylinositol 3-phosphate and FYVE domain proteins. Biochem. J. 2001, 355, 249–258. [Google Scholar] [CrossRef]
- Banerjee, S.; Basu, S.; Sarkar, S. Comparative genomics reveals selective distribution and domain organization of FYVE and PX domain proteins across eukaryotic lineages. BMC Genom. 2010, 11, 83. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guetta, D.; Langou, K.; Grunwald, D.; Klein, G.; Aubry, L. FYVE-dependent endosomal targeting of an arrestin-related protein in amoeba. PLoS ONE 2010, 5, e15249. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, J.; Du, X.; Zhou, X.; Jin, D.; Miao, J.; Liu, X. An FYVE-Domain-Containing Protein, PsFP1, Is Involved in Vegetative Growth, Oxidative Stress Response and Virulence of Phytophthora sojae. Int. J. Mol. Sci. 2021, 22, 6601. [Google Scholar] [CrossRef] [PubMed]
- Lu, X.; Yang, Z.; Song, W.; Miao, J.; Zhao, H.; Ji, P.; Li, T.; Si, J.; Yin, Z.; Jing, M.; et al. The Phytophthora sojae effector PsFYVE1 modulates immunity-related gene expression by targeting host RZ-1A protein. Plant Physiol. 2022, 191, 925–945. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Yang, K.; Yan, Q.; Wang, X.; Cheng, M.; Si, J.; Xue, X.; Shen, D.; Jing, M.; Tyler, B.M.; et al. Targeting of anti-microbial proteins to the hyphal surface amplifies protection of crop plants against Phytophthora pathogens. Mol. Plant 2021, 14, 1391–1403. [Google Scholar] [CrossRef] [PubMed]
- Vassella, E.; Krämer, R.; Turner, C.M.R.; Wankell, M.; Modes, C.; van den Bogaard, M.; Boshart, M. Deletion of a novel protein kinase with PX and FYVE-related domains increases the rate of differentiation of Trypanosoma brucei. Mol. Microbiol. 2001, 41, 33–46. [Google Scholar] [CrossRef] [Green Version]
- Judelson, H.S.; Ah-Fong, A.M. The kinome of Phytophthora infestans reveals oomycete-specific innovations and links to other taxonomic groups. BMC Genom. 2010, 11, 700. [Google Scholar] [CrossRef] [Green Version]
- Thompson, J.D.; Gibson, T.J.; Plewniak, F.; Jeanmougin, F.; Higgins, D.G. The CLUSTAL_X windows interface: Flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Res. 1997, 25, 4876–4882. [Google Scholar] [CrossRef] [Green Version]
- Ronquist, F.; Teslenko, M.; van der Mark, P.; Ayres, D.L.; Darling, A.; Höhna, S.; Larget, B.; Liu, L.; Suchard, M.A.; Huelsenbeck, J.P. MrBayes 3.2: Efficient Bayesian phylogenetic inference and model choice across a large model space. Syst. Biol. 2012, 61, 539–542. [Google Scholar] [CrossRef] [Green Version]
- Sheng, Y.; Lin, L.; Chen, H.; Pu, T.; Liu, X.; Dong, S.; Ye, W.; Wang, Y. The bZIP transcription factor PsBZP32 is involved in cyst germination, oxidative stress response, and pathogenicity of Phytophthora sojae. Phytopathol. Res. 2021, 3, 1. [Google Scholar] [CrossRef]
- Xue, Z.; Wang, W.; Shen, J.; Zhang, J.; Zhang, X.; Liu, X. A Patched-Like Protein PsPTL Is Not Essential for the Growth and Response to Various Stresses in Phytophthora sojae. Front. Microbiol. 2021, 12, 673784. [Google Scholar] [CrossRef]
- Zhang, X.; Zhai, C.; Hua, C.; Qiu, M.; Hao, Y.; Nie, P.; Ye, W.; Wang, Y. PsHint1, associated with the G-protein α subunit PsGPA1, is required for the chemotaxis and pathogenicity of Phytophthora sojae. Mol. Plant Pathol. 2016, 17, 272–285. [Google Scholar] [CrossRef]
- Qiu, M.; Li, Y.; Ye, W.; Zheng, X.; Wang, Y. A CRISPR/Cas9-mediated in situ complementation method for Phytophthora sojae mutants. Mol. Plant Pathol. 2021, 22, 373–381. [Google Scholar] [CrossRef] [PubMed]
- Beebe, S.J. The cAMP-dependent protein kinases and cAMP signal transduction. Semin. Cancer Biol. 1994, 5, 285–294. [Google Scholar]
- Kaffarnik, F.; Müller, P.; Leibundgut, M.; Kahmann, R.; Feldbrügge, M. PKA and MAPK phosphorylation of Prf1 allows promoter discrimination in Ustilago maydis. EMBO J. 2003, 22, 5817–5826. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.; Jung, W.H.; Kronstad, J.W. The cAMP/protein kinase A signaling pathway in pathogenic basidiomycete fungi: Connections with iron homeostasis. J. Microbiol. 2015, 53, 579–587. [Google Scholar] [CrossRef] [Green Version]
- Attwood, M.M.; Fabbro, D.; Sokolov, A.V.; Knapp, S.; Schiöth, H.B. Trends in kinase drug discovery: Targets, indications and inhibitor design. Nat. Rev. Drug Discov. 2021, 20, 839–861. [Google Scholar] [CrossRef]
- Kroemer, G.; Mariño, G.; Levine, B. Autophagy and the integrated stress response. Mol. Cell 2010, 40, 280–293. [Google Scholar] [CrossRef] [Green Version]
- Mizushima, N.; Komatsu, M. Autophagy: Renovation of cells and tissues. Cell 2011, 147, 728–741. [Google Scholar] [CrossRef] [Green Version]
- Luo, Q.; Wang, F.-X.; Zhong, N.-Q.; Wang, H.-Y.; Xia, G.-X. The role of autophagy during development of the oomycete pathogen Phytophthora infestans. J. Genet. Genom. 2014, 41, 225–228. [Google Scholar] [CrossRef]
- Wang, J.; Zhou, G.; Huang, W.; Li, W.; Feng, D.; Liu, L.; Xi, P.; Jiang, Z.; Kong, G. Autophagy-Related Gene PlATG6a Is Involved in Mycelial Growth, Asexual Reproduction and Tolerance to Salt and Oxidative Stresses in Peronophythora litchii. Int. J. Mol. Sci. 2022, 23, 1839. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Zhang, X.; Wang, W.; Geng, X.; Shi, Y.; Na, R.; Dou, D.; Li, H. Network and role analysis of autophagy in Phytophthora sojae. Sci. Rep. 2017, 7, 1879. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kamada, Y.; Funakoshi, T.; Shintani, T.; Nagano, K.; Ohsumi, M.; Ohsumi, Y. Tor-mediated induction of autophagy via an Apg1 protein kinase complex. J. Cell Biol. 2000, 150, 1507–1513. [Google Scholar] [CrossRef] [Green Version]
- Wullschleger, S.; Loewith, R.; Hall, M.N. TOR signaling in growth and metabolism. Cell 2006, 124, 471–484. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, D.H.; Eu, Y.-J.; Yoo, C.M.; Kim, Y.-W.; Pih, K.T.; Jin, J.B.; Kim, S.J.; Stenmark, H.; Hwang, I. Trafficking of phosphatidylinositol 3-phosphate from the trans-Golgi network to the lumen of the central vacuole in plant cells. Plant Cell 2001, 13, 287–301. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kale, S.D.; Gu, B.; Capelluto, D.G.; Dou, D.; Feldman, E.; Rumore, A.; Arredondo, F.D.; Hanlon, R.; Fudal, I.; Rouxel, T.; et al. External lipid PI3P mediates entry of eukaryotic pathogen effectors into plant and animal host cells. Cell 2010, 142, 284–295. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dumas, J.J.; Merithew, E.; Sudharshan, E.; Rajamani, D.; Hayes, S.; Lawe, D.; Corvera, S.; Lambright, D.G. Multivalent endosome targeting by homodimeric EEA1. Mol. Cell 2001, 8, 947–958. [Google Scholar] [CrossRef]
- Kutateladze, T.G. Phosphatidylinositol 3-phosphate recognition and membrane docking by the FYVE domain. Biochim. Biophys. Acta 2006, 1761, 868–877. [Google Scholar] [CrossRef] [Green Version]
- Musacchio, A.; Gibson, T.; Rice, P.; Thompson, J.; Saraste, M. The PH domain: A common piece in the structural patchwork of signalling proteins. Trends Biochem. Sci. 1993, 18, 343–348. [Google Scholar] [CrossRef] [PubMed]
- Chandra, M.; Collins, B.M. The Phox Homology (PX) Domain. In Protein Reviews—Purinergic Receptors; Atassi, M., Ed.; Springer: Cham, Switzerland, 2019; pp. 1–17. [Google Scholar]
- Bae, H.; Viennet, T.; Park, E.; Chu, N.; Salguero, A.; Eck, M.J.; Arthanari, H.; Cole, P.A. PH domain-mediated autoinhibition and oncogenic activation of Akt. Elife 2022, 11, e80148. [Google Scholar] [CrossRef]
- Maffucci, T.; Falasca, M. Specificity in pleckstrin homology (PH) domain membrane targeting: A role for a phosphoinositide-protein co-operative mechanism. FEBS Lett. 2001, 506, 173–179. [Google Scholar] [CrossRef] [Green Version]
- Singh, N.; Reyes-Ordoñez, A.; Compagnone, M.A.; Moreno, J.F.; Leslie, B.J.; Ha, T.; Chen, J. Redefining the specificity of phosphoinositide-binding by human PH domain-containing proteins. Nat. Commun. 2021, 12, 4339. [Google Scholar] [CrossRef]
- Kanai, F.; Liu, H.; Field, S.J.; Akbary, H.; Matsuo, T.; Brown, G.E.; Cantley, L.C.; Yaffe, M.B. The PX domains of p47phox and p40phox bind to lipid products of PI(3)K. Nat. Cell Biol. 2001, 3, 675–678. [Google Scholar] [CrossRef] [PubMed]
- Ellson, C.D.; Andrews, S.; Stephens, L.R.; Hawkins, P.T. The PX domain: A new phosphoinositide-binding module. J. Cell Sci. 2002, 115, 1099–1105. [Google Scholar] [CrossRef] [PubMed]
- Alessi, D.R.; Deak, M.; Casamayor, A.; Caudwell, F.B.; Morrice, N.; Norman, D.G.; Gaffney, P.; Reese, C.B.; MacDougall, C.N.; Harbison, D.; et al. 3-Phosphoinositide-dependent protein kinase-1 (PDK1): Structural and functional homology with the Drosophila DSTPK61 kinase. Curr. Biol. 1997, 7, 776–789. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Du, K.; Tsichlis, P.N. Regulation of the Akt kinase by interacting proteins. Oncogene 2005, 24, 7401–7409. [Google Scholar] [CrossRef] [Green Version]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ru, B.; Hao, X.; Li, W.; Peng, Q.; Miao, J.; Liu, X. A Novel FYVE Domain-Containing Protein Kinase, PsZFPK1, Plays a Critical Role in Vegetative Growth, Sporangium Formation, Oospore Production, and Virulence in Phytophthora sojae. J. Fungi 2023, 9, 709. https://doi.org/10.3390/jof9070709
Ru B, Hao X, Li W, Peng Q, Miao J, Liu X. A Novel FYVE Domain-Containing Protein Kinase, PsZFPK1, Plays a Critical Role in Vegetative Growth, Sporangium Formation, Oospore Production, and Virulence in Phytophthora sojae. Journal of Fungi. 2023; 9(7):709. https://doi.org/10.3390/jof9070709
Chicago/Turabian StyleRu, Binglu, Xinchang Hao, Wenhao Li, Qin Peng, Jianqiang Miao, and Xili Liu. 2023. "A Novel FYVE Domain-Containing Protein Kinase, PsZFPK1, Plays a Critical Role in Vegetative Growth, Sporangium Formation, Oospore Production, and Virulence in Phytophthora sojae" Journal of Fungi 9, no. 7: 709. https://doi.org/10.3390/jof9070709





