Studies on Loading Salicylic Acid in Xerogel Films of Crosslinked Hyaluronic Acid
Abstract
1. Introduction
2. Results and Discussion
2.1. Determination of SA Saturated Solubility
2.2. UV-Vis Spectrophotometry
2.2.1. Calculation of λmax
2.2.2. Determination of Drug Loading
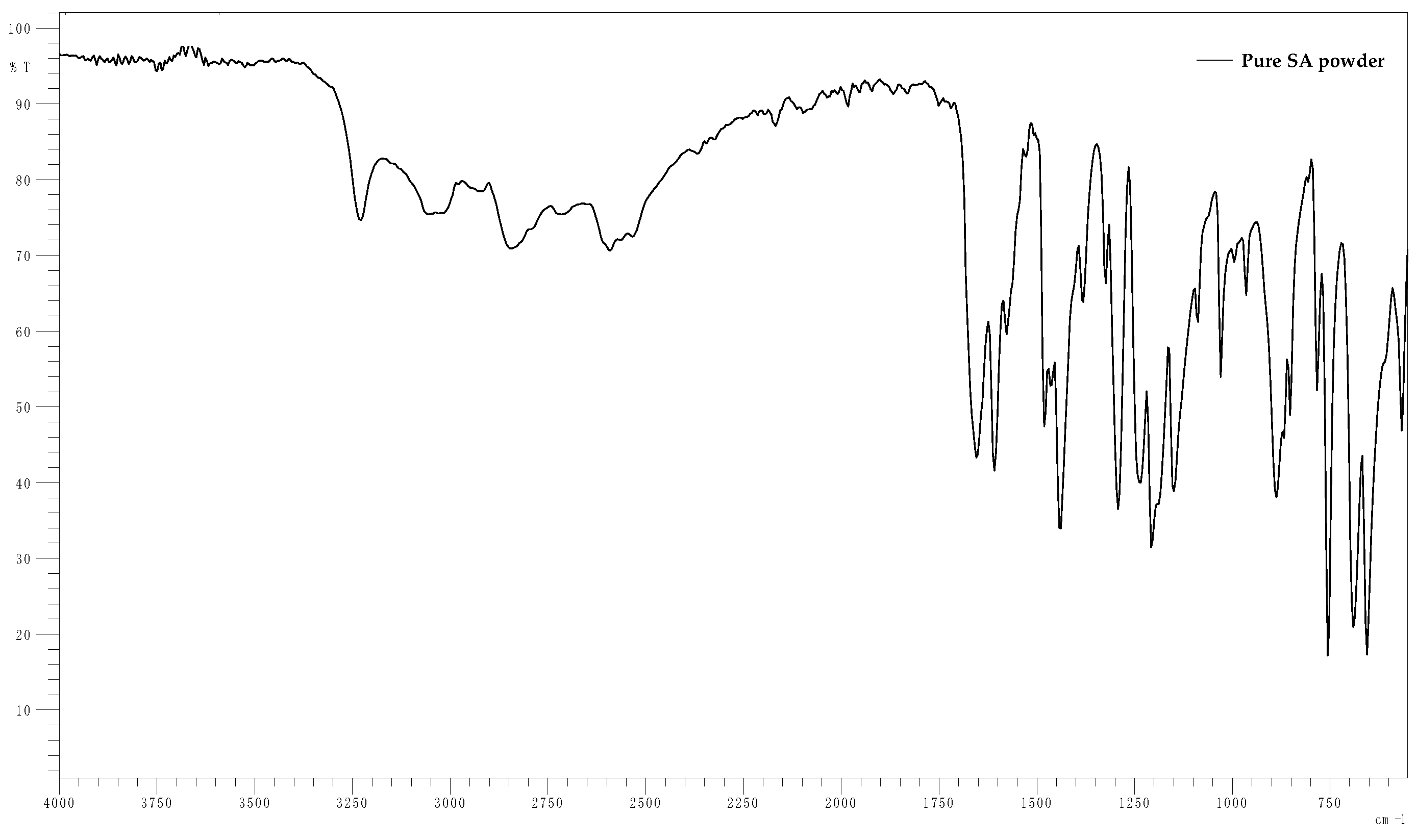
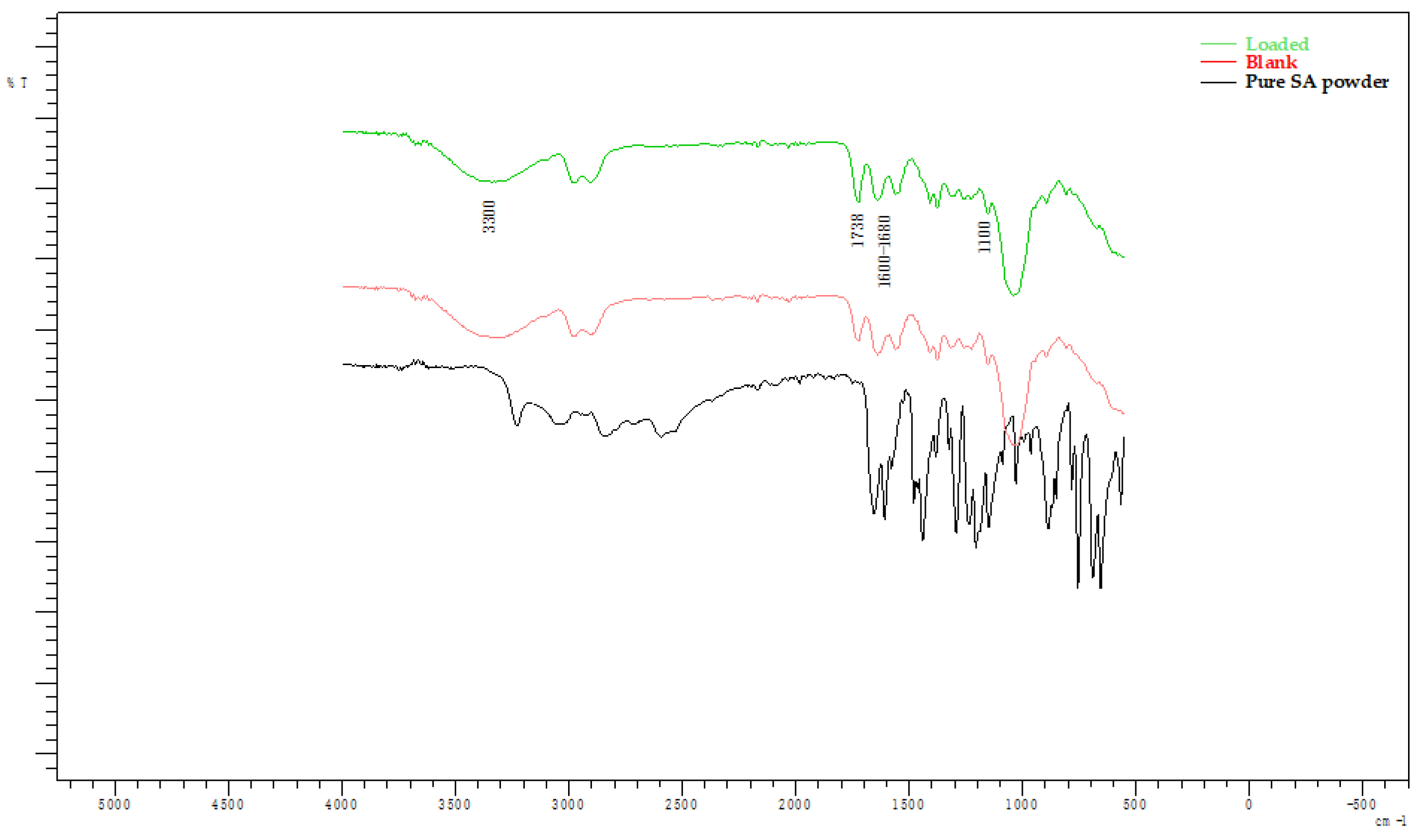
2.3. Fourier-Transform Infra-Red (FT-IR)
2.4. Weight Studies
2.5. Film Thickness
2.6. Discussion
3. Conclusions
4. Materials and Methods
4.1. Materials
4.2. Methods
4.2.1. Drug Loading Method
4.2.2. Determination of SA Saturated Solubility
4.2.3. UV-Vis Spectrophotometry
Determination of λmax
Plotting of Calibration Curves
Determination of Drug Loading
4.2.4. Fourier-Transform Infra-Red (FT-IR)
- Films swelled in water of different pH values (2.1, 7.8 and 11);
- Films loaded with SA solutions of different pH values (2.1, 7.8 and 11);
- SA powder.
4.2.5. Weight Studies
4.2.6. Film Thickness
4.2.7. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Pastore, M.N.; Kalia, Y.N.; Horstmann, M.; Roberts, M.S. Transdermal patches: History, development and pharmacology. Br. J. Pharmacol. 2015, 172, 2179–2209. [Google Scholar] [CrossRef] [PubMed]
- Monou, P.K.; Mamaligka, A.M.; Tzimtzimis, E.K.; Tzetzis, D.; Vergkizi-Nikolakaki, S.; Vizirianakis, I.S.; Andriotis, E.G.; Eleftheriadis, G.K.; Fatouros, D.G. Fabrication and Preliminary In Vitro Evaluation of 3D-Printed Alginate Films with Cannabidiol (CBD) and Cannabigerol (CBG) Nanoparticles for Potential Wound-Healing Applications. Pharmaceutics 2022, 14, 1637. [Google Scholar] [CrossRef]
- Cacciotti, I.; Ciocci, M.; Di Giovanni, E.; Nanni, F.; Melino, S. Hydrogen Sulfide-Releasing Fibrous Membranes: Potential Patches for Stimulating Human Stem Cells Proliferation and Viability under Oxidative Stress. Int. J. Mol. Sci. 2018, 19, 2368. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Park, K. Polymeric Micelles and Alternative Nanonized Delivery Vehicles for Poorly Soluble Drugs. Int. J. Pharm. 2013, 453, 198–214. [Google Scholar] [CrossRef] [PubMed]
- Teoh, J.H.; Tay, S.M.; Fuh, J.; Wang, C.-H. Fabricating Scalable, Personalized Wound Dressings with Customizable Drug Loadings via 3D Printing. J. Control. Release 2022, 341, 80–94. [Google Scholar] [CrossRef]
- Nayak, A.K.; Das, B. 1-Introduction to polymeric gels. In Polymeric Gels: Characterization, Properties and Biomedical Applications; Woodhead Publishing Series in Biomaterials; Pal, K., Banerjee, B., Eds.; Woodhead Publishing: Cambridge, UK, 2018; pp. 3–27. [Google Scholar] [CrossRef]
- Chronopoulou, L.; Cacciotti, I.; Amalfitano, A.; Nitto, A.D.; Arienzo, V.D.; Nocca, G.; Palocci, C. Biosynthesis of innovative calcium phosphate/hydrogel composites: Physicochemical and biological characterization. Nanotechnology 2020, 32, 095102. [Google Scholar] [CrossRef]
- Ciocci, M.; Cacciotti, I.; Seliktar, D.; Melino, S. Injectable silk fibroin hydrogels functionalized with microspheres as adult stem cells-carrier systems. Int. J. Biol. Macromol. 2018, 108, 960–971. [Google Scholar] [CrossRef]
- Thang, N.H.; Chien, T.B.; Cuong, D.X. Polymer-Based Hydrogels Applied in Drug Delivery: An Overview. Gels 2023, 9, 523. [Google Scholar] [CrossRef] [PubMed]
- Peppas, N.A.; Bures, P.; Leobandung, W.; Ichikawa, H. Hydrogels in pharmaceutical formulations. Eur. J. Pharm. Biopharm. 2000, 50, 27–46. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, E.M. Hydrogel: Preparation, characterization, and applications: A review. J. Adv. Res. 2015, 6, 105–121. [Google Scholar] [CrossRef]
- Rashid, M.A.M.; Rahman, M.; Mahmud, A.O.; Morshed, A.S.M.; Haque, M.M.; Hossain, M.M. UV-Vis Spectrophotometer as an Alternative Technique for the Determination of Hydroquinone in Vinyl Acetate Monomer. Photochem 2022, 2, 435–447. [Google Scholar] [CrossRef]
- Mummaleti, G.; Kong, F. Fabrication, properties and applications of Xerogels in food processing. J. Agric. Food Res. 2023, 11, 100506. [Google Scholar] [CrossRef]
- Kemas, C.U.; Ngwuluka, N.C.; Ochekpe, N.A.; Nep, E.I. Starch-based xerogels: Effect of acetylation on Physicochemical and rheological properties. Int. J. Biol. Macromol. 2017, 98, 94–102. [Google Scholar] [CrossRef] [PubMed]
- Holthoff, E.L.; Bright, F.V. Molecularly imprinted xerogels as platforms for sensing. Acc. Chem. Res. 2007, 40, 756–767. [Google Scholar] [CrossRef] [PubMed]
- Awadallah-F, A.; Al-Muhtaseb, S.A. Influence of chitosan addition on resorcinol–formaldehyde xerogel structure. Appl. Sci. 2019, 9, 4582. [Google Scholar] [CrossRef]
- Rodríguez-Dorado, R.; López-Iglesias, C.; García-González, C.A.; Auriemma, G.; Aquino, R.P.; Del Gaudio, P. Design of Aerogels, Cryogels and Xerogels of Alginate: Effect of Molecular Weight, Gelation Conditions and Drying Method on Particles’ Micromeritics. Molecules 2019, 24, 1049. [Google Scholar] [CrossRef]
- Groult, S.; Buwalda, S.; Budtova, T. Pectin hydrogels, aerogels, cryogels and xerogels: Influence of drying on structural and release properties. Eur. Polym. J. 2021, 149, 110386. [Google Scholar] [CrossRef]
- Sakuma, W.; Yamasaki, S.; Fujisawa, S.; Kodama, T.; Shiomi, J.; Kanamori, K.; Saito, T. Mechanically Strong, scalable, mesoporous xerogels of nanocellulose featuring light permeability, thermal insulation, and flame self-extinction. ACS Nano 2021, 15, 1436–1444. [Google Scholar] [CrossRef]
- Idumah, C.I. Emerging advancements in xerogel polymeric bionanoarchitectures and applications. JCIS Open 2023, 9, 100073. [Google Scholar] [CrossRef]
- Abdul Khalil, H.P.S.; Yahya, E.B.; Tajarudin, H.A.; Balakrishnan, V.; Nasution, H. Insights into the Role of Biopolymer-Based Xerogels in Biomedical Applications. Gels 2022, 8, 334. [Google Scholar] [CrossRef]
- Czarnobaj, K. Preparation and characterization of silica xerogels as carriers for drugs. Drug Deliv. 2008, 15, 485–492. [Google Scholar] [CrossRef] [PubMed]
- Figueira, D.R.; Miguel, S.P.; de Sá, K.D.; Correia, I.J. Production and characterization of polycaprolactone- hyaluronic acid/chitosan- zein electrospun bilayer nanofibrous membrane for tissue regeneration. Int. J. Biol. Macromol. 2016, 93, 1100–1110. [Google Scholar] [CrossRef] [PubMed]
- Li, N.N.; Fu, C.P.; Zhang, L.M. Using casein and oxidized hyaluronic acid to form biocompatible composite hydrogels for controlled drug release. Mater. Sci. Eng. C 2014, 36, 287–293. [Google Scholar] [CrossRef] [PubMed]
- Bhatnagar, D.; Xu, D.; Gersappe, D.; Rafailovich, M. Hyaluronic acid and gelatin clay composite hydrogels: Substrates for cell adhesion and controlled drug delivery. J. Chem. Biol. Interfaces 2014, 2, 34–44. [Google Scholar] [CrossRef]
- Huang, A.C.; Chuang, Y.K.; Huang, C.F.; Shu, C.M. Thermokinetic analysis of the stability of malic and salicylic acids in cosmeceutical formulations containing metal oxides. J. Therm. Anal. Calorim. 2018, 132, 165–172. [Google Scholar] [CrossRef]
- Musleh, C.; Dodou, K. How Dissolving HA Microneedles Change Before/After Application. Cosmetics and Toiletries. 2023. October Issue. Available online: https://www.cosmeticsandtoiletries.com/testing/efficacy/article/22870788/how-dissolving-ha-microneedles-change-beforeafter-application (accessed on 11 December 2023).
- Gura, E.; Hückel, M.; Müller, P.-J. Specific degradation of hyaluronic acid and its rheological properties. Polym. Degrad. Stab. 1998, 59, 297–302. [Google Scholar] [CrossRef]
- Mero, A.; Campisi, M. Hyaluronic Acid Bioconjugates for the Delivery of Bioactive Molecules. Polymers 2014, 6, 346–369. [Google Scholar] [CrossRef]
- Chen, M.; Gupta, V.; Anselmo, A.C.; Muraski, J.A.; Mitragotri, S. Topical delivery of hyaluronic acid into skin using SPACE-peptide carriers. J. Control. Release 2014, 173, 67–74. [Google Scholar] [CrossRef]
- Bukhari, S.N.A.; Roswandi, N.L.; Waqas, M.; Habib, H.; Hussain, F.; Khan, S.; Sohail, M.; Ramli, N.A.; Thu, H.E.; Hussain, Z. Hyaluronic acid, a promising skin rejuvenating biomedicine: A review of recent updates and pre-clinical and clinical investigations on cosmetic and nutricosmetic effects. Int. J. Biol. Macromol. 2018, 120, 1682–1695. [Google Scholar] [CrossRef]
- Rashid, F.; Albayati, M.; Dodou, K. Studies on Novel Methods for Formulating Novel Cross-Linked Hydrogel Films of Hyaluronic Acid. Cosmetics 2019, 6, 59. [Google Scholar] [CrossRef]
- Chang, W.H.; Liu, P.Y.; Lin, M.H.; Lu, C.J.; Chou, H.Y.; Nian, C.Y.; Jiang, Y.T. Applications of hyaluronic acid in ophthalmology and contact lenses. Molecules 2021, 26, 2485. [Google Scholar] [CrossRef]
- Ohri, R.; Hahn, S.K.; Stayton, P.S.; Hoffman, A.S.; Giachelli, M. Hyaluronic acid grafting mitigates calcification of glutaraldehyde-fixed bovine pericardium. J. Biomed. Mater. Res. 2004, 70A, 328–334. [Google Scholar] [CrossRef] [PubMed]
- Kang, H.; Zuo, Z.; Lin, R.; Yao, M.; Han, Y.; Han, J. The most promising microneedle device: Present and future of hyaluronic acid microneedle patch. Drug Deliv. 2022, 29, 3087–3110. [Google Scholar] [CrossRef]
- Pluda, S.; Salvagnini, C.; Fontana, A.; Marchetti, A.; Di Lucia, A.; Galesso, D.; Guarise, C. Investigation of Crosslinking Parameters and Characterization of Hyaluronic Acid Dermal Fillers: From Design to Product Performances. Gels 2023, 9, 733. [Google Scholar] [CrossRef] [PubMed]
- Ilyin, S.O.; Kulichikhin, V.G.; Malkin, A.Y. The rheological characterisation of typical injection implants based on hyaluronic acid for contour correction. Rheol. Acta 2016, 55, 223–233. [Google Scholar] [CrossRef]
- Hahn, S.K.; Kim, S.J.; Kim, M.J.; Chung, C.H.; Moon, D.B.; Lee, Y. Development of a novel sustained release formulation of recombinant human growth hormone using sodium hyaluronate microparticles. J. Control. Release 2005, 104, 323–335. [Google Scholar] [CrossRef]
- Kurczewska, J. Recent Reports on Polysaccharide-Based Materials for Drug Delivery. Polymers 2022, 14, 4189. [Google Scholar] [CrossRef]
- Khunmanee, S.; Jeong, Y.; Park, H. Crosslinking method of hyaluronic-based hydrogel for biomedical applications. J. Tissue Eng. 2017, 8, 1–16. [Google Scholar] [CrossRef]
- Maleki, A.; Kjøniksen, A.L.; Nyström, B. Characterization of the chemical degradation of hyaluronic acid during chemical gelation in the presence of different cross-linker agents. Carbohydr. Res. 2007, 342, 2776–2792. [Google Scholar] [CrossRef]
- Gatej, J.; Popa, M.; Rinaudo, M. Role of the pH on Hyaluronan Behavior in Aqueous Solution. Biomacromolecules 2005, 6, 61–67. [Google Scholar] [CrossRef]
- Maleki, A.; Kjøniksen, A.L.; Nyström, B. Effect of pH on the Behavior of Hyaluronic Acid in Dilute and Semidilute Aqueous Solutions. Macromol. Symp. 2008, 274, 131–140. [Google Scholar] [CrossRef]
- Arif, T. Salicylic acid as a peeling agent: A comprehensive review. Clin. Cosmet. Investig. Dermatol. 2015, 8, 455–461. [Google Scholar] [CrossRef] [PubMed]
- Madan, R.K.; Levitt, J. A review of toxicity from topical salicylic acid preparations. JAAD 2014, 70, 788–792. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Jiang, R.; Zhou, J.; Xu, X.; Sun, Z.; Li, J.; Chen, X.; Li, Z.; Yan, X.; Zhao, D.; et al. Salicylic acid in ginseng root alleviates skin hyperpigmentation disorders by inhibiting melanogenesis and melanosome transport. Eur. J. Pharmacol. 2021, 910, 174458. [Google Scholar] [CrossRef]
- Lee, T.-W.; Kim, J.-C.; Hwang, S.-J. Hydrogel patches containing Triclosan for acne treatment. Eur. J. Pharm. Biopharm. 2003, 56, 407–412. [Google Scholar] [CrossRef]
- Otto, D.P.; Combrinck, J.; Otto, A.; Tiedt, L.R.; de Villiers, M.M. Dissipative Particle Dynamics Investigation of the Transport of Salicylic Acid through a Simulated In Vitro Skin Permeation Model. Pharmaceuticals 2018, 11, 134. [Google Scholar] [CrossRef]
- Shalmashi, A.; Eliassi, A. Solubility of Salicylic Acid in Water, Ethanol, Carbon Tetrachloride, Ethyl Acetate, and Xylene. J. Chem. Eng. Data 2008, 53, 199–200. [Google Scholar] [CrossRef]
- Fan, C.H.; Zhang, Y.C.; Wang, J.H. Effect of pH on the Ultraviolet Spectra and Fluorescence Characteristics of Dissolved Organic Matter in the Process of Straw Humification. Spectrosc. Spectr. Anal. 2015, 35, 1933–1937. [Google Scholar]
- Singh, S.; Rai, K.A.; Tewari, P.T. Recent advancement in hyaluronic acid-based hydrogel for biomedical engineering application: A mini-review. Mater. Today Proc. 2023, 78, 138–144. [Google Scholar] [CrossRef]
- Li, Y.; Yang, L. Driving forces for drug loading in drug carriers. J. Microencapsul. 2015, 32, 255–272. [Google Scholar] [CrossRef]
- Jiao, L.Y.; Yuan, G.; Qi, Y.P.; Li, L.L. Synthesis and Properties of Polyacrylate Emulsion Modified by Pentaerythritol Tetraacrylate. Adv. Mater. Res. 2013, 781–784, 550–553. [Google Scholar] [CrossRef]
- Caló, E.; Barros, J.M.S.D.; Fernández-Gutiérrez, M.; San Román, J.; Ballamy, L.; Khutoryanskiy, V.V. Antimicrobial hydrogels based on autoclaved poly(vinyl alcohol) and poly(methyl vinyl ether-alt-maleic anhydride) mixtures for wound care applications. RSC Adv. 2016, 6, 55211–55219. [Google Scholar] [CrossRef]
- Mahindrakar, J.N.; Patil, Y.; Salunkhe, P.; Ankushrao, S.; Kadam, V.N. Optically transparent, organosoluble poly(ether-amide)s bearing triptycene unit; synthesis and characterization. J. Macromol. Sci. 2018, 55, 658–667. [Google Scholar] [CrossRef]
- Saraswathy, M.; Sreenivasan, K. Conjugation of curcumin onto hyaluronic acid enhances its aqueous solubility and stability. J. Colloid Interface Sci. 2011, 359, 318–325. [Google Scholar] [CrossRef]
- Polák, J.; Morávek, A.; Brkljača, Z.; Vazdar, M.; Cibulka, I.; Heyda, J. Computation and volumetric insight into (P,T) effect on aqueous guanidinium chloride. J. Chem. Thermodyn. 2021, 158, 106450. [Google Scholar] [CrossRef]
- Maharjan, A.S.; Pilling, D.; Gomer, R.H. High and low molecular weight hyaluronic acid differentially regulate human fibrocyte differentiation. PLoS ONE 2011, 6, e26078. [Google Scholar] [CrossRef] [PubMed]
- Mondek, J.; Kalina, M.; Simulescu, V.; Pekař, M. Thermal degradation of high molar mass hyaluronan in solution and in powder; comparison with BSA. Polym. Degrad. Stab. 2015, 120, 107–113. [Google Scholar] [CrossRef]
- Sudha, P.N.; Rose, M.H. Beneficial effects of hyaluronic acid. Adv. Food Nutr. Res. 2014, 72, 137–176. [Google Scholar] [CrossRef] [PubMed]
- Hubbe, M.; Ayoub, A.; Daystar, J.; Venditti, R.; Pawlak, J. Enhanced Absorbent Products Incorporating Cellulose and Its Derivatives: A Review. BioResources 2013, 8, 6556–6629. [Google Scholar] [CrossRef]
- Burke, S.E.; Barrett, C.J. Swelling behavior of hyaluronic acid/polyallylamine hydrochloride multilayer films. Biomacromolecules 2005, 6, 1419–1428. [Google Scholar] [CrossRef]
- Le, H.V.; Dulong, V.; Picton, L.; Cerf, D.L. Polyelectrolyte complexes of hyaluronic acid and diethylaminoethyl dextran: Formation, stability and hydrophobicity. Colloids Surf. A Physicochem. Eng. Asp. 2021, 629, 127485. [Google Scholar] [CrossRef]
- Wong, R.S.; Ashton, M.; Dodou, K. Effect of Crosslinking Agent Concentration on the Properties of Unmedicated Hydrogels. Pharmaceutics 2015, 7, 305–319. [Google Scholar] [CrossRef] [PubMed]
- Wong, R.S.H.; Dodou, K. Effect of Drug Loading Method and Drug Physicochemical Properties on the Material and Drug Release Properties of Poly (Ethylene Oxide) Hydrogels for Transdermal Delivery. Polymers 2017, 9, 286. [Google Scholar] [CrossRef]
- Drug Bank. Available online: https://go.drugbank.com/drugs/DB00936 (accessed on 11 December 2023).
- Po, H.N.; Senozan, N.M. The Henderson-Hasselbalch equation: Its history and limitations. J. Chem. Educ. 2001, 78, 1499. [Google Scholar] [CrossRef]
- Rattana, M.; Paradee, N.; Sirivat, A.; Niamlang, S. Porcine and Fish Gelatin Hydrogels for Controlled Release of Salicylic Acid and 5-sulfosalicylic Acid. Int. J. Drug Dev. Res. 2015, 7, 107–117. [Google Scholar]
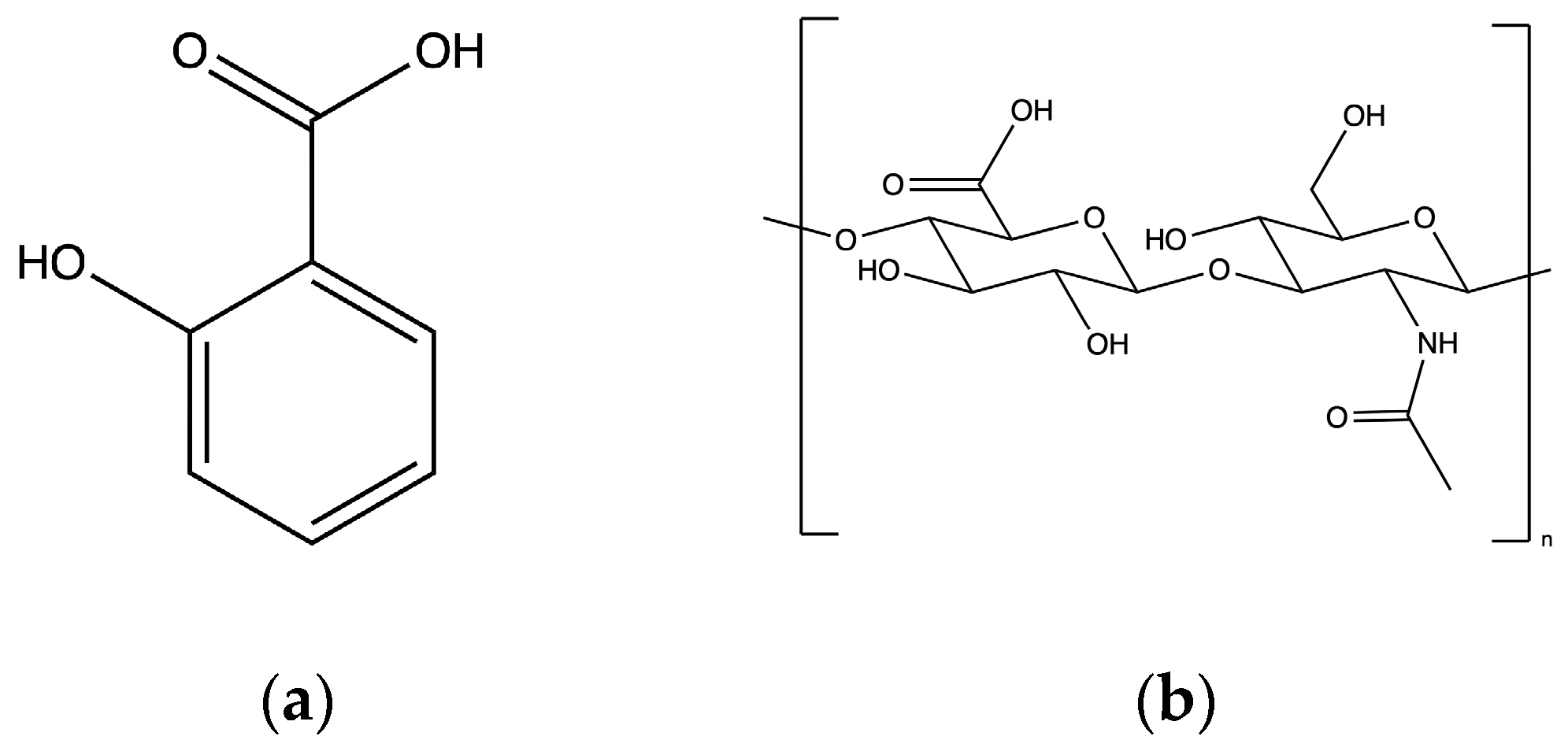












| pH | Solubility (mg/mL) | Ionized Salicylic Acid (%) | Temperature (°C) |
|---|---|---|---|
| 2.00 | 15.3 ± 0.9 | 9.9 | 37 |
| 5.00 | >34.9 ± 3.2 | 99.1 |
| pH | Saturated Solubility (mg SA/mL H2O) |
|---|---|
| 2.1 | 1.97 |
| 7.8 | 2.47 |
| 11 | 2.47 |
| pH | Ionization (%) |
|---|---|
| 2.1 | 11.18 |
| 7.8 | 99.99 |
| 11 | 99.94 |
| Sample | pH | Average Film Weight (g) (±SD) | |||
|---|---|---|---|---|---|
| Xerogel Pre-Loading | Swelled Film Post-loading | Xerogel Post-Loading A | Xerogel Post-Loading B | ||
| Blank | 2.1 | 0.0055 (±0.022) | 0.0166 (±0.007) | 0.0039 (±0.002) | 0.0042 (±0.002) |
| Blank | 7.8 | 0.0063 (±0.001) | 0.0255 (±0.005) | 0.0046 (±0.001) | 0.0052 (±0.001) |
| Blank | 11 | 0.0065 (±0.003) | 0.0269 (±0.002) | 0.0047 (±0.000) | 0.0053 (±0.000) |
| SA | 2.1 | 0.0060 (±0.001) | 0.0187 (±0.003) | 0.0044 (±0.001) | 0.0046 (±0.001) |
| SA | 7.8 | 0.0049 (±0.000) | 0.0199 (±0.002) | 0.0042 (±0.001) | 0.0043 (±0.000) |
| SA | 11 | 0.0050 (±0.001) | 0.0317 (±0.010) | 0.0033 (±0.000) | 0.0040 (±0.000) |
| Sample | pH | Degree of Swelling (%) (±SD) | Gel Fraction A1 (%) (±SD) | Gel Fraction B2 (%) (±SD) | Weight Loss A1 (%) (±SD) | Weight Loss B2 (%) (±SD) |
|---|---|---|---|---|---|---|
| Blank | 2.1 | 198.70 (±7.79) | 71.43 (±1.29) | 77.92 (±3.90) | 28.57 (±1.30) | 22.08 (±3.39) |
| Blank | 7.8 | 301.89 (±19.28) | 72.81 (±3.11) | 81.66 (±0.73) | 27.19 (±3.12) | 18.34 (±0.73) |
| Blank | 11 | 312.86 (±4.79) | 71.66 (±2.54) | 80.76 (±0.12) | 28.34 (±2.54) | 19.24 (±0.12) |
| SA | 2.1 | 212.63 (±16.29) | 72.98 (±4.24) | 76.80 (±5.72) | 27.02 (±4.24) | 23.20 (±5.73) |
| SA | 7.8 | 310.32 (±32.41) | 86.76 (±4.35) | 89.06 (±2.76) | 13.24 (±4.35) | 10.94 (±2.76) |
| SA | 11 | 520.12 (±121.64) | 65.10 (±3.37) | 80.60 (±5.49) | 34.90 (±3.37) | 19.40 (±5.49) |
| Sample | pH | Difference WLA-WLB (%) (±SD) | Difference GFA-GFB (%) (±SD) |
|---|---|---|---|
| Blank | 2.1 | −23.03 (±10.15) | 9.05 (±3.47) |
| Blank | 7.8 | −31.76 (±8.75) | 12.33 (±5.33) |
| Blank | 11 | −31.74 (±6.56) | 12.80 (±4.14) |
| SA | 2.1 | −19.89 (±8.36) | 7.13 (±2.20) |
| SA | 7.8 | −28.75 (±12.37) | 5.28 (±4.32) |
| SA | 11 | −44.62 (±13.33) | 23.85 (±6.75) |
| Sample | pH | Film Thickness (mm) (n = 3) | Increase of Film Thickness (%) | |
|---|---|---|---|---|
| Xerogel | 0.150 | N/A | ||
| Swollen unmedicated film (Blank) | Blank 1 | 2.1 | 0.213 | 86.667 |
| Blank 2 | 7.8 | 0.320 | 106.667 | |
| Blank 3 | 11 | 0.293 | 93.333 | |
| Loaded Film (SA) | SA 1 | 2.1 | 0.310 | 106.667 |
| SA 2 | 7.8 | 0.290 | 93.333 | |
| SA 3 | 11 | 0.323 | 113.333 |
| Solution | pH | Blanking Solution |
|---|---|---|
| SA Solution 1 | 2.1 | Water at pH 2.1 |
| SA Solution 2 | 7.8 | Water at pH 7.8 |
| SA Solution 3 | 11 | Water at pH 11 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mamaligka, A.M.; Dodou, K. Studies on Loading Salicylic Acid in Xerogel Films of Crosslinked Hyaluronic Acid. Gels 2024, 10, 54. https://doi.org/10.3390/gels10010054
Mamaligka AM, Dodou K. Studies on Loading Salicylic Acid in Xerogel Films of Crosslinked Hyaluronic Acid. Gels. 2024; 10(1):54. https://doi.org/10.3390/gels10010054
Chicago/Turabian StyleMamaligka, Anastasia Maria, and Kalliopi Dodou. 2024. "Studies on Loading Salicylic Acid in Xerogel Films of Crosslinked Hyaluronic Acid" Gels 10, no. 1: 54. https://doi.org/10.3390/gels10010054
APA StyleMamaligka, A. M., & Dodou, K. (2024). Studies on Loading Salicylic Acid in Xerogel Films of Crosslinked Hyaluronic Acid. Gels, 10(1), 54. https://doi.org/10.3390/gels10010054









