Hydrogel-Transformable Antioxidant Poly-γ-Glutamic Acid/Polyethyleneimine Hemostatic Powder for Efficient Wound Hemostasis
Abstract
1. Introduction
2. Results and Discussion
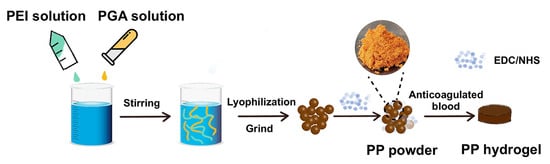
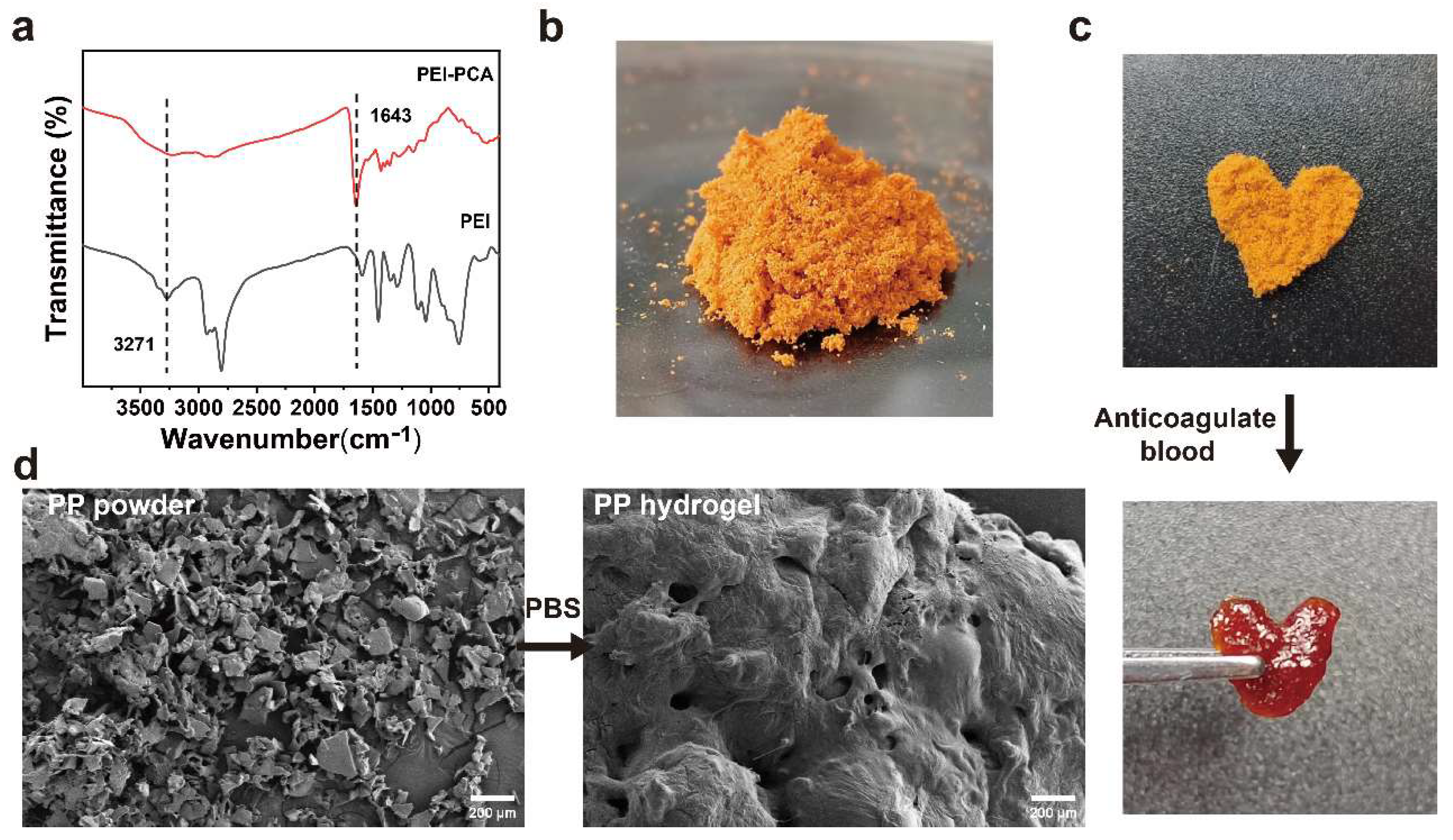
2.1. Preparation and Characterization of the PP Hemostatic Powder
2.2. Mechanical and Rheological Properties
2.3. Swelling and Degradation
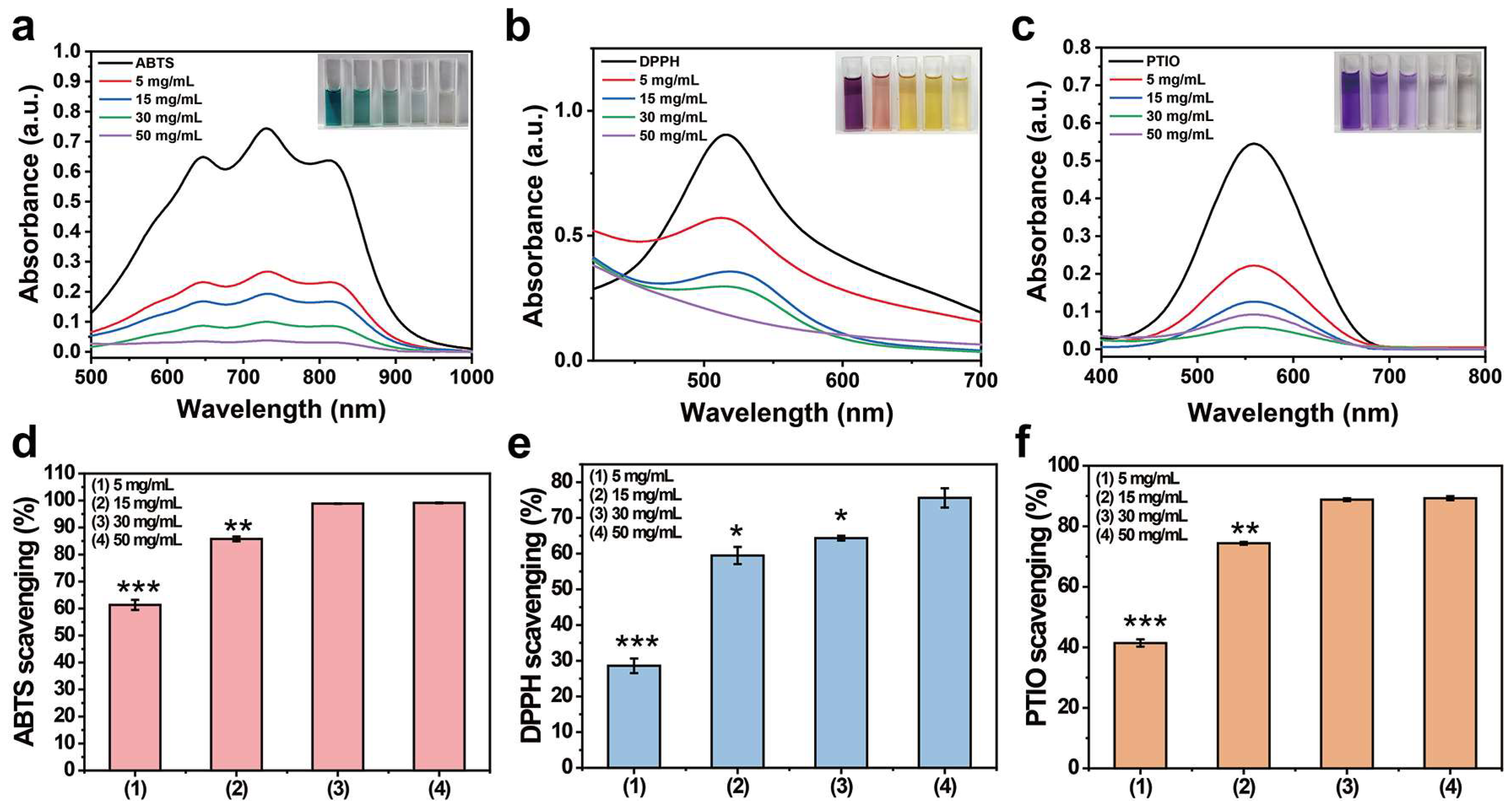
2.4. Antioxidant Properties
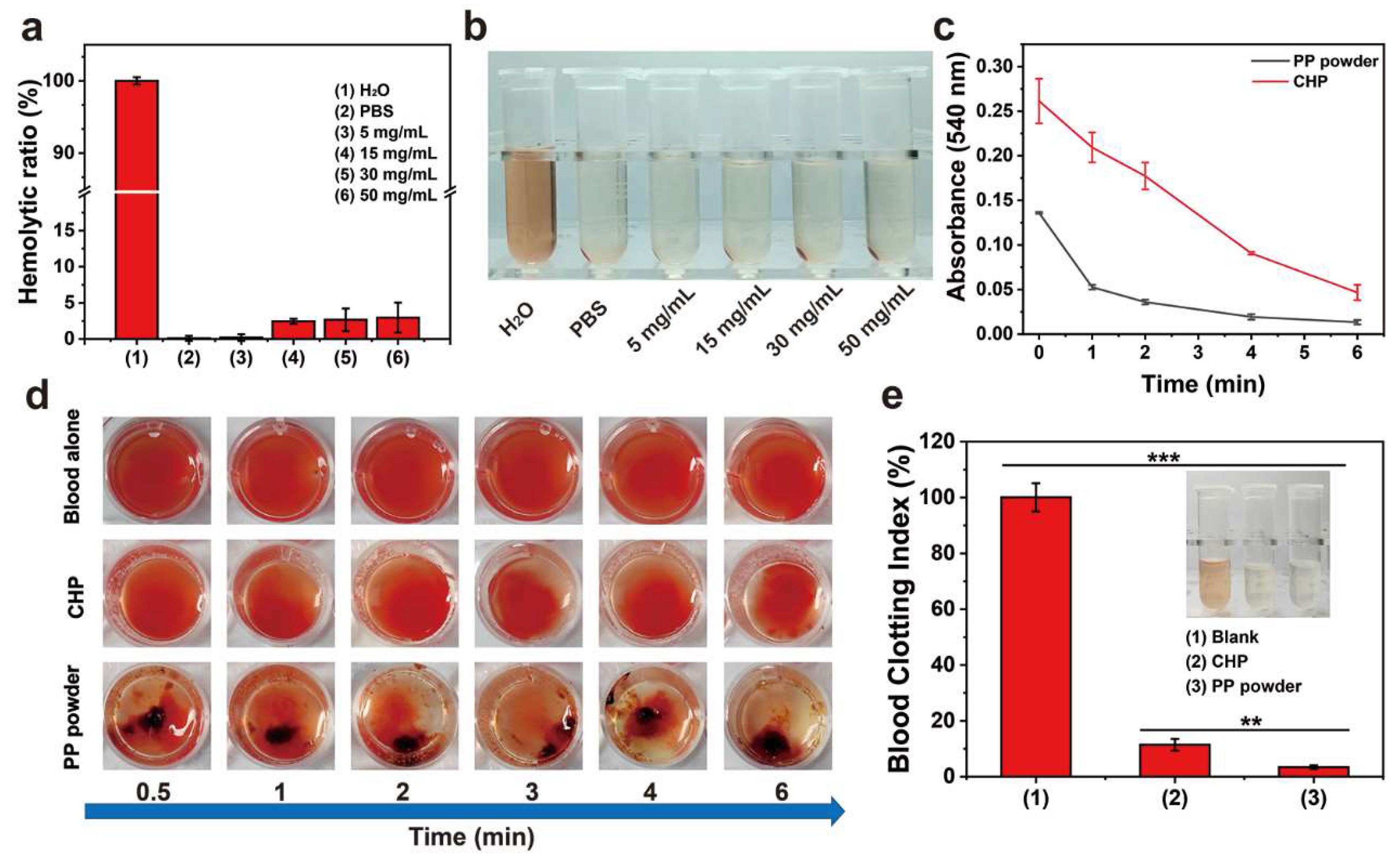
2.5. In Vitro Safety
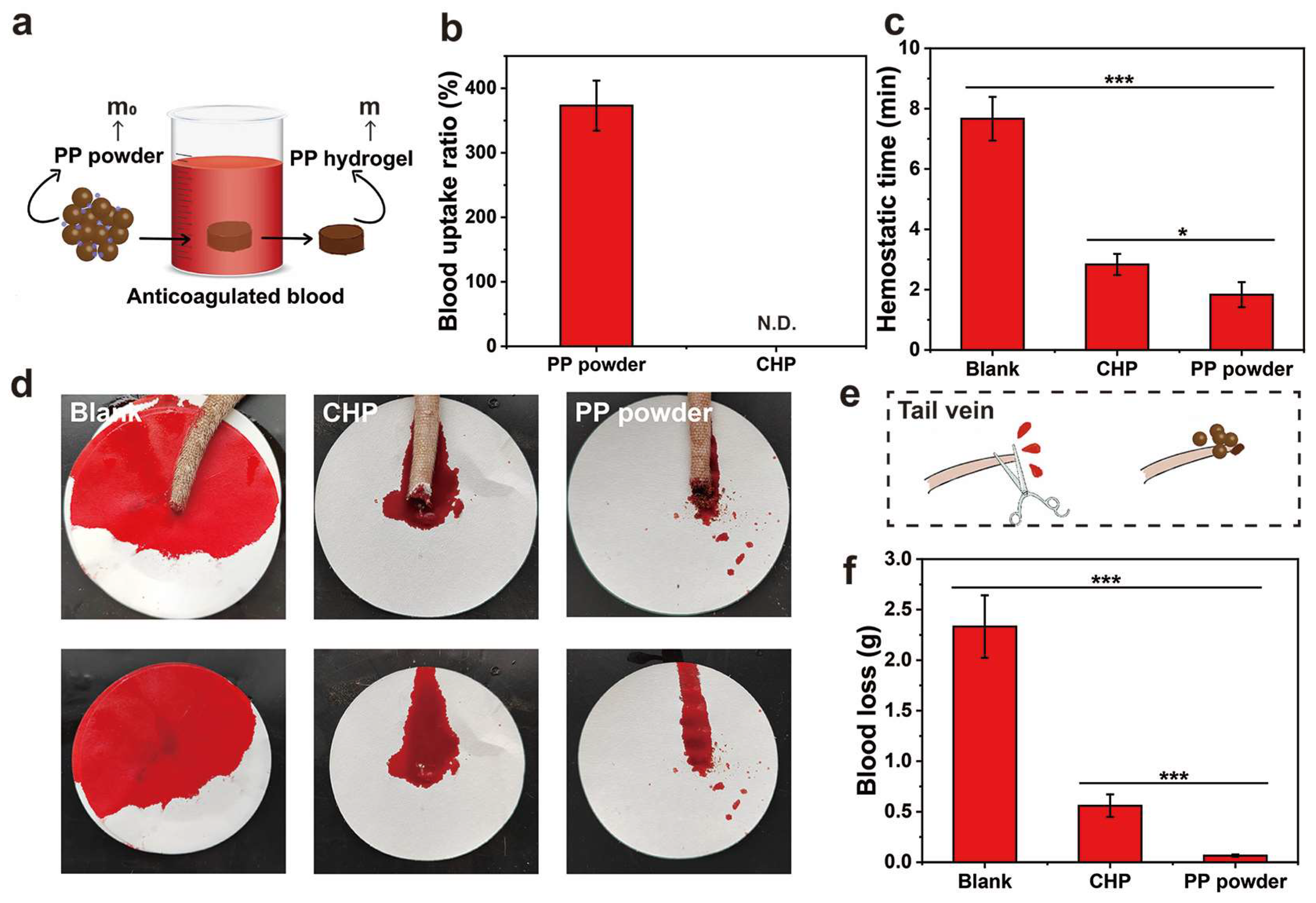
2.6. In Vitro Hemostasis Experiments
2.7. In Vivo Hemostasis Experiments
3. Conclusions
4. Materials and Methods
4.1. Materials
4.2. Preparation of the PP Hemostatic Powder
4.3. Characterization
4.4. Mechanical Testing
4.5. Study of Swelling Behaviors
4.6. In Vitro Degradation
4.7. Antioxidant Properties
4.7.1. ABTS· Scavenging Experiment
4.7.2. DPPH· Scavenging Experiment
4.7.3. PTIO· Scavenging Experiment
4.8. Blood Compatibility
4.9. In Vitro Hemostasis Test
4.10. In Vivo Hemostasis Study
4.11. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cui, T.; Yu, J.; Wang, C.; Chen, S.; Li, Q.; Guo, K.; Qing, R.; Wang, G.; Ren, J. Micro-Gel Ensembles for Accelerated Healing of Chronic Wound via pH Regulation. Adv. Sci. 2022, 9, e2201254. [Google Scholar] [CrossRef] [PubMed]
- Nie, L.; Wei, Q.; Sun, M.; Ding, P.; Wang, L.; Sun, Y.; Ding, X.; Okoro, O.V.; Jiang, G.; Shavandi, A. Injectable, self-healing, transparent, and antibacterial hydrogels based on chitosan and dextran for wound dressings. Int. J. Biol. Macromol. 2023, 233, 123494. [Google Scholar] [CrossRef] [PubMed]
- Altenschmidt, L.; Sanchez-Paradinas, S.; Lubkemann, F.; Zambo, D.; Abdelmonem, A.M.; Bradtmuller, H.; Masood, A.; Morales, I.; de la Presa, P.; Knebel, A.; et al. Aerogelation of Polymer-Coated Photoluminescent, Plasmonic, and Magnetic Nanoparticles for Biosensing Applications. ACS Appl. Nano Mater. 2021, 4, 6678–6688. [Google Scholar] [CrossRef]
- Long, M.; Zhang, B.; Peng, S.; Liao, J.; Zhang, Y.; Wang, J.; Wang, M.; Qin, B.; Huang, J.; Huang, J.; et al. Interactions between two-dimensional nanoclay and blood cells in hemostasis. Mater. Sci. Eng. C 2019, 105, 110081. [Google Scholar] [CrossRef] [PubMed]
- Dai, Z.; Zhang, Y.; Chen, C.; Yu, F.; Tian, J.; Cai, H.; Jiang, X.; Zhang, L.; Zhang, W. An Antifouling and Antimicrobial Zwitterionic Nanocomposite Hydrogel Dressing for Enhanced Wound Healing. ACS Biomater. Sci. Eng. 2021, 7, 1621–1630. [Google Scholar] [CrossRef]
- Liu, C.; Liu, C.; Liu, Z.; Shi, Z.; Liu, S.; Wang, X.; Wang, X.; Huang, F. Injectable thermogelling bioadhesive chitosan-based hydrogels for efficient hemostasis. Int. J. Biol. Macromol. 2023, 224, 1091–1100. [Google Scholar] [CrossRef]
- Hu, B.; Ouyang, Y.; Zhao, T.; Wang, Z.; Yan, Q.; Qian, Q.; Wang, W.; Wang, S. Antioxidant Hydrogels: Antioxidant Mechanisms, Design Strategies, and Applications in the Treatment of Oxidative Stress-Related Diseases. Adv. Healthc. Mater. 2024. [Google Scholar] [CrossRef]
- Xiao, L.; Hui, F.; Tian, T.; Yan, R.; Xin, J.; Zhao, X.; Jiang, Y.; Zhang, Z.; Kuang, Y.; Li, N.; et al. A Novel Conductive Antibacterial Nanocomposite Hydrogel Dressing for Healing of Severely Infected Wounds. Front. Chem. 2021, 9, 787886. [Google Scholar] [CrossRef]
- Gan, D.; Huang, Z.; Wang, X.; Jiang, L.; Wang, C.; Zhu, M.; Ren, F.; Fang, L.; Wang, K.; Xie, C.; et al. Graphene Oxide-Templated Conductive and Redox-Active Nanosheets Incorporated Hydrogels for Adhesive Bioelectronics. Adv. Funct. Mater. 2019, 30, 1907678. [Google Scholar] [CrossRef]
- Jing, X.; Mi, H.-Y.; Lin, Y.-J.; Enriquez, E.; Peng, X.-F.; Turng, L.-S. Highly Stretchable and Biocompatible Strain Sensors Based on Mussel-Inspired Super-Adhesive Self-Healing Hydrogels for Human Motion Monitoring. ACS Appl. Mater. Interfaces 2018, 10, 20897–20909. [Google Scholar] [CrossRef]
- Luo, X.; Ao, F.; Huo, Q.; Liu, Y.; Wang, X.; Zhang, H.; Yang, M.; Ma, Y.; Liu, X. Skin-inspired injectable adhesive gelatin/HA biocomposite hydrogel for hemostasis and full-thickness dermal wound healing. Biomater. Adv. 2022, 139, 35882139. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Dai, C.; Fan, L.; Jiang, Y.; Liu, C.; Zhou, Z.; Guan, P.; Tian, Y.; Xing, J.; Li, X.; et al. Injectable Self-Healing Natural Biopolymer-Based Hydrogel Adhesive with Thermoresponsive Reversible Adhesion for Minimally Invasive Surgery. Adv. Funct. Mater. 2021, 31, 2007457. [Google Scholar] [CrossRef]
- Prasad, A.S.; Wilson, J.; Thomas, L.V. Designer injectable matrices of photocrosslinkable carboxymethyl cellulose methacrylate based hydrogels as cell carriers for gel type autologous chondrocyte implantation (GACI). Int. J. Biol. Macromol. 2023, 224, 465–482. [Google Scholar] [CrossRef]
- Kim, H.; Kim, J.; Choi, J.; Park, Y.; Ki, C. Characterization of silk hydrogel formed with hydrolyzed silk fibroin-methacrylate via photopolymerization. Polymer 2018, 153, 232–240. [Google Scholar] [CrossRef]
- Wu, C.; Jiao, Q.; Wang, C.; Zheng, Y.; Pan, X.; Zhong, W.; Xu, K. Nanofibrillar peptide hydrogels for self-delivery of lonidamine and synergistic photodynamic therapy. Acta Biomater. 2023, 155, 139–153. [Google Scholar] [CrossRef] [PubMed]
- Zeng, L.; Chang, Y.; Wu, Y.; Yang, J.; Xu, J.-F.; Zhang, X. Charge-reversal surfactant antibiotic material for reducing microbial corrosion in petroleum exploitation and transportation. Sci. Adv. 2020, 6, eaba7524. [Google Scholar] [CrossRef] [PubMed]
- Oleyaei, S.A.; Razavi, S.M.A.; Mikkonen, K.S. Novel nanobiocomposite hydrogels based on sage seed gum-laponite: Physico-chemical and rheological characterization. Carbohydr. Polym. 2018, 192, 282–290. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Garcia, F.D.; van Dongen, J.A.; Burgess, J.K.; Harmsen, M.C. Matrix Metalloproteases from Adipose Tissue-Derived Stromal Cells Are Spatiotemporally Regulated by Hydrogel Mechanics in a 3D Microenvironment. Bioengineering 2022, 9, 340. [Google Scholar] [CrossRef]
- Ahn, D.; Sun, J.; Han, S.; Lee, J.; Jeong, S.; Cha, S.; Noh, S.; Choi, H.; Ren, B.; Yoon, H.; et al. Controllable Physical Synergized Triboelectricity, Shape Memory, Self-Healing, and Optical Sensing with Rollable Form Factor by Zn cluster. Adv. Sci. 2022, 9, e2200441. [Google Scholar] [CrossRef]
- Pei, X.; Wang, J.; Cong, Y.; Fu, J. Recent progress in polymer hydrogel bioadhesives. J. Polym. Sci. 2021, 59, 1312–1337. [Google Scholar] [CrossRef]
- Zhou, Q.; Zhou, X.; Mo, Z.; Zeng, Z.; Wang, Z.; Cai, Z.; Luo, L.; Ding, Q.; Li, H.; Tang, S. A PEG-CMC-THB-PRTM hydrogel with antibacterial and hemostatic properties for promoting wound healing. Int. J. Biol. Macromol. 2023, 224, 370–379. [Google Scholar] [CrossRef] [PubMed]
- Jung, D.H.; Park, C.H.; Choi, S.I.; Kim, H.R.; Lee, M.; Moon, H.S.; Park, J.C. Comparison of a Polysaccharide Hemostatic Powder and Conventional Therapy for Peptic Ulcer Bleeding. Clin. Gastroenterol. Hepatol. 2023, 21, 2844–2853. [Google Scholar] [CrossRef] [PubMed]
- Peng, X.; Xu, X.; Deng, Y.; Xie, X.; Xu, L.; Xu, X.; Yuan, W.; Yang, B.; Yang, X.; Xia, X.; et al. Ultrafast Self-Gelling and Wet Adhesive Powder for Acute Hemostasis and Wound Healing. Adv. Funct. Mater. 2021, 31, 2102583. [Google Scholar] [CrossRef]
- Zhang, W.; Liu, W.; Long, L.; He, S.; Wang, Z.; Liu, Y.; Yang, L.; Chen, N.; Hu, C.; Wang, Y. Responsive multifunctional hydrogels emulating the chronic wounds healing cascade for skin repair. J. Control. Release 2023, 354, 821–834. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Li, S.; Yuan, X.; Zhao, J.; Hou, X. An in situ catechol functionalized ε-polylysine/polyacrylamide hydrogel formed by hydrogen bonding recombination with high mechanical property for hemostasis. Int. J. Biol. Macromol. 2021, 191, 714–726. [Google Scholar]
- Zheng, Y.; Shariati, K.; Ghovvati, M.; Vo, S.; Origer, N.; Imahori, T.; Kaneko, N.; Annabi, N. Hemostatic patch with ultra-strengthened mechanical properties for efficient adhesion to wet surfaces. Biomaterials 2023, 301, 122240. [Google Scholar] [CrossRef]
- Xia, L.; Wang, S.; Jiang, Z.; Chi, J.; Yu, S.; Li, H.; Zhang, Y.; Li, L.; Zhou, C.; Liu, W.; et al. Hemostatic performance of chitosan-based hydrogel and its study on biodistribution and biodegradability in rats. Carbohydr. Polym. 2021, 264, 117965. [Google Scholar] [CrossRef]
- Zhou, C.; Xu, R.; Han, X.; Tong, L.; Xiong, L.; Liang, J.; Sun, Y.; Zhang, X.; Fan, Y. Protocatechuic acid-mediated injectable antioxidant hydrogels facilitate wound healing. Composites 2022, 250 Pt B, 110451. [Google Scholar] [CrossRef]
- Li, X.F.; Lu, P.; Jia, H.R.; Li, G.; Zhu, B.; Wang, X.; Wu, F.G. Emerging materials for hemostasis. Coord. Chem. Rev. 2022, 475, 214823. [Google Scholar] [CrossRef]
- Guo, B.; Dong, R.; Liang, Y.; Li, M. Haemostatic materials for wound healing applications. Nat. Rev. Chem. 2021, 5, 773–791. [Google Scholar] [CrossRef]
- Puertas-Bartolomé, M.; Benito-Garzón, L.; Fung, S.; Kohn, J.; Vázquez-Lasa, B.; Román, J.S. Bioadhesive functional hydrogels: Controlled release of catechol species with antioxidant and antiinflammatory behavior. Mater. Sci. Eng. C Mater. Biol. Appl. 2019, 105, 110040. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Chen, M.; Luo, M.; Li, T.; Hu, C.; Xie, C.; Li, S.; Leng, T.; Tian, J.; Xu, P. Bioactive glass ions hydrogels with antiinflammation antioxidant capacity for treating inflammation-related diseases. Mater. Des. 2023, 227, 111669. [Google Scholar] [CrossRef]
- Dulong, V.; Kouassi, M.-C.; Labat, B.; Le Cerf, D.; Picton, L. Antioxidant properties and bioactivity of Carboxymethyl pullulan grafted with ferulic acid and of their hydrogels obtained by enzymatic reaction. Food Chem. 2018, 262, 21–29. [Google Scholar] [CrossRef] [PubMed]
- Komeri, R.; Thankam, F.G.; Muthu, J. Free radical scavenging injectable hydrogels for regenerative therapy. Biomater. Adv. 2016, 71, 100–110. [Google Scholar] [CrossRef]
- Li, X. 2-Phenyl-4,4,5,5-tetramethylimidazoline-1-oxyl 3-Oxide (PTIO•) Radical Scavenging: A New and Simple Antioxidant Assay In Vitro. J. Agric. Food Chem. 2017, 65, 6288–6297. [Google Scholar] [CrossRef]
- Tan, Z.; Li, X.; Yu, C.; Yao, M.; Zhao, Z.; Guo, B.; Liang, L.; Wei, Y.; Yao, F.; Zhang, H.; et al. A self-gelling powder based on polyacrylic acid/polyacrylamide/quaternate chitosan for rapid hemostasis. Int. J. Biol. Macromol. 2023, 232, 123449. [Google Scholar] [CrossRef]
- Wang, Z.; Lyu, T.; Xie, Q.; Zhang, Y.; Sun, H.; Wan, Y.; Tian, Y. Shape-adapted self-gelation hydrogel powder for high-performance hemostasis and wound healing. Appl. Mater. Today 2023, 35, 101948. [Google Scholar] [CrossRef]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, X.; Han, W.; He, G.; Yang, J.; Li, J.; Ma, H.; Wang, S. Hydrogel-Transformable Antioxidant Poly-γ-Glutamic Acid/Polyethyleneimine Hemostatic Powder for Efficient Wound Hemostasis. Gels 2024, 10, 68. https://doi.org/10.3390/gels10010068
Li X, Han W, He G, Yang J, Li J, Ma H, Wang S. Hydrogel-Transformable Antioxidant Poly-γ-Glutamic Acid/Polyethyleneimine Hemostatic Powder for Efficient Wound Hemostasis. Gels. 2024; 10(1):68. https://doi.org/10.3390/gels10010068
Chicago/Turabian StyleLi, Xiang, Wenli Han, Gao He, Jiahao Yang, Jing Li, Hongxia Ma, and Shige Wang. 2024. "Hydrogel-Transformable Antioxidant Poly-γ-Glutamic Acid/Polyethyleneimine Hemostatic Powder for Efficient Wound Hemostasis" Gels 10, no. 1: 68. https://doi.org/10.3390/gels10010068
APA StyleLi, X., Han, W., He, G., Yang, J., Li, J., Ma, H., & Wang, S. (2024). Hydrogel-Transformable Antioxidant Poly-γ-Glutamic Acid/Polyethyleneimine Hemostatic Powder for Efficient Wound Hemostasis. Gels, 10(1), 68. https://doi.org/10.3390/gels10010068