1. Introduction
Beef tripe is particularly popular in the Asian market due to its unique flavor and nutritional value [
1]. With the rise of the plant-based market, non-meat protein products are also showing potential in the field of tripe [
2,
3]. However, relatively little research has been conducted on edible non-meat protein alternative to tripe [
4], especially those products that accurately mimic the texture, color, and sensory experience of natural tripe. If a non-meat protein product could be developed to closely mimic the texture and appearance of real tripe, it would not only satisfy consumer demand for healthy and environmentally friendly plant-based food products, but also provide an edible experience similar to that of traditional tripe.
Gel simulation bionics is an advanced technology that uses polymer network systems to mimic the structure and function of biological tissues [
5]. This technique has been widely used in the medical field for skin simulation for wound repair [
6], cosmetic surgery [
7], and in vitro release experiments [
8]. For example, Wei et al. [
9] developed a skin simulation material with moisturizing and anti-freezing properties using polyacrylic acid (PAA) and betaine; Zhang et al. [
10] simulated the viscoelasticity of porcine skin using agarose gel; and Beatrice et al. [
8] used a stabilized hydrogel to simulate the vascular flow pool of the cardiac diaphragm to study the release and distribution of drugs in vitro, and these studies have fully shown the wide practicality of gel simulation technology. If gel simulation bionic technology can be applied to the production of food products, it will help to develop new types of food to meet the diversified needs of the market and may have a positive impact on the food industry.
At present, most materials utilized in gel bionic technology include polyacrylamide [
11], PAA [
9], nanofibers [
12], and other non-edible substances. This limitation hinders the application of gel bionic technology in the food industry. KGM is a natural polymer polysaccharide derived from konjac. KGM is safe for consumption, and it possesses remarkable thickening and gelling attributes; thus, KGM is extensively used in the fields of food and medicine [
13,
14]. However, current research is mostly focused on the KGM single system [
14] or the complex polysaccharide system [
15]. This system lacks the toughness and chewiness required to effectively simulate the oral chewing experience of meat. Moreover, this type of system is nutritionally simple. The ingredients are relatively simple. It has been found that protein/polysaccharide composite gel systems can make up for the limitations of single-component gels and enrich the nutrition of the gel through synergistic interactions between ingredients. Soy-protein isolate (SPI), pea-protein isolate (PPI), and whey-protein isolate (WPI) are commonly used proteins in biomimetic meat processing [
16,
17,
18], and many studies have also combined them with KGM to simulate meat products. For example, Ran et al. [
19] simulated the texture and structure of fish-ball by using a hybrid gel of KGM and SPI; Wang et al. [
20] structured the gel network of PPI and KGM in order to mimic animal proteins; and Du et al. [
21] applied a hybrid gel of WPI and KGM in 3D printing. But few studies have compared these three proteins’ properties to the macroscopic effect of KGM and apply them to the field of bionic tripe.
In addition, different processing conditions also have an effect on the properties of composite gels. It has been shown that the movement of water molecules in the hydrogel structure is restricted during freezing, which at the same time promotes polymer chain alignment or assembly, thus affecting the structure and texture of the gel [
22,
23,
24]. Although this property is used in many applications such as the food, medicine, biology, and chemical industries [
25,
26], many of these studies were on single polysaccharide gels [
27], konjac glucomannan/xanthan gum/sodium alginate composite gel [
23], or soy protein-dextran conjugate gels [
28]. There are few studies and applications on the effect of freezing on protein/KGM hybrid gels.
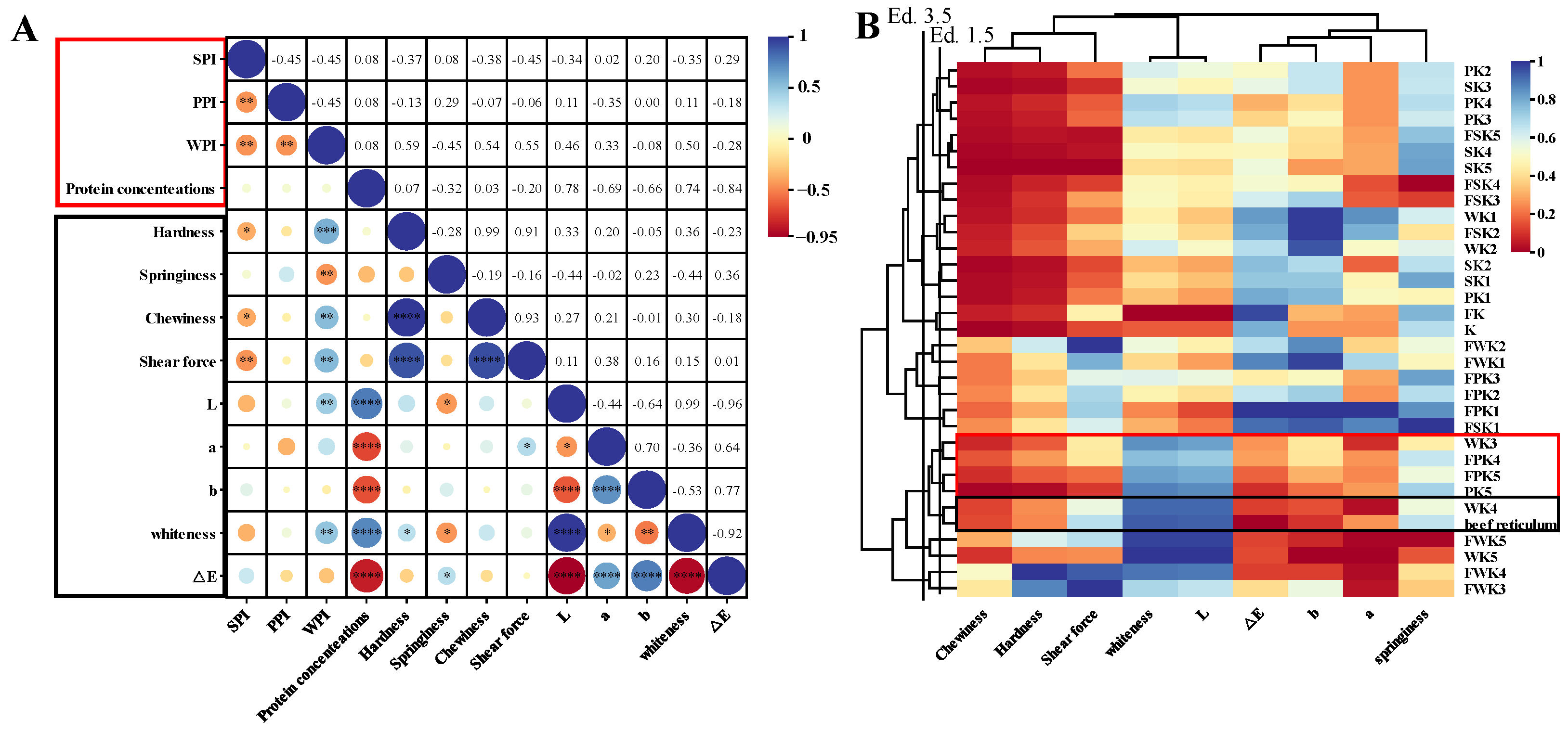
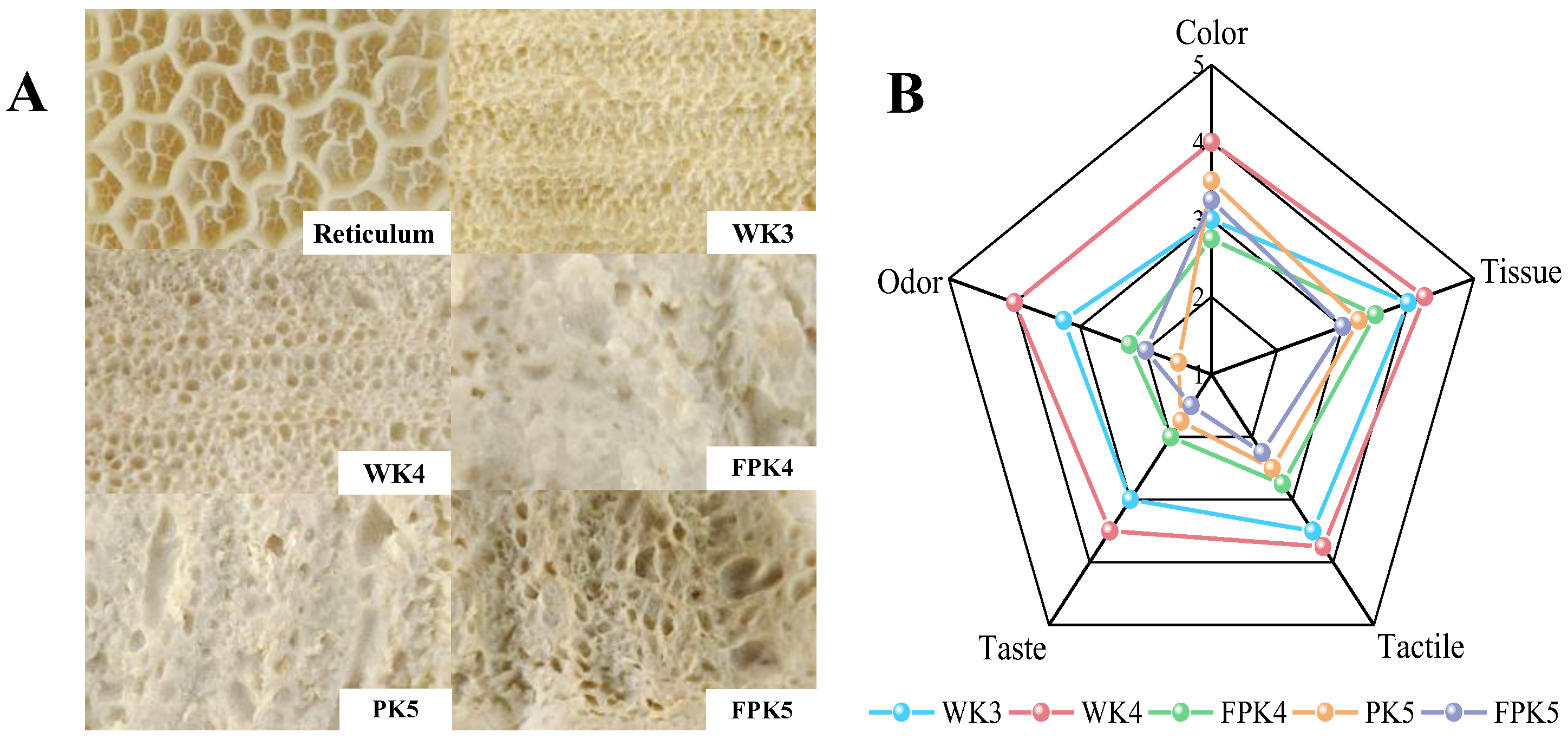
In this study, we compared the effects of three non-meat proteins (SPI, WPI, and PPI) on the texture and color of KGM composite gels at different protein levels and processing conditions. And using cluster analysis and sensory evaluation, these composite gel simulants were compared and screened with natural tripe stomach to obtain an edible composite gel formulation that simulated the texture, appearance, and taste of natural tripe. On this basis, the screened optimum gel formulation was subjected to rheological analysis, powder X-ray diffraction analysis, FTIR spectral analysis, contribution of intermolecular forces, and scanning electron microscopy analysis to preliminary explore its gel-forming mechanism in order to provide some help for the processing of non-meat protein products in the future.
3. Conclusions
In this study, we compared the effects of adding SPI, PPI, and WPI on the gel properties of KGM under cooling and freezing conditions. The addition of proteins enhanced the gel properties of KGM, improving hardness and chewiness, and the effect of WPI on the texture and chroma of the KGM composite gels was particularly significant; freezing facilitated the polymerization of the konjac/protein composite gels, which improved the gel properties, but also lowered the sensory scores. The best protein-polysaccharide gel formulation, WK4, which more accurately simulated tripe in terms of texture, appearance, and sensory experience, was screened by cluster analysis and sensory evaluation, and its characterization properties were investigated. The addition of WPI greatly affected the rheological properties, intermolecular forces, crystallinity, and microstructure of the KGM complexes. When the concentration of WPI was 8%, the energy storage modulus (G′) was enhanced compared to the single KGM gel, which was consistent with the results of the textural study, the strength of the hydrogen bonding and hydrophobic interactions was enhanced, and the crystallinity was increased, and the hydrophobic interactions and disulfide bonding interactions were the key to maintain the strength of the WK4 gel. The SEM images in the WK4 group showed that the gels formed a dense and continuous gel network. This study provides a theoretical basis for the development of biomimetic meat products using composite gels with different exogenous proteins and KGM.
4. Materials and Methods
4.1. Materials
Fresh beef reticulum (one of the tripe) was obtained from the meat market behind the North Gate of Chengdu University (Chengdu, China). KGM was provided by Hubei Johnson Konjac Technology Co., Ltd. (Ezhou, China); SPI was provided by Linyi Shan song Biological Products Co., Ltd. (Linyi, China); PPI was provided by Yantai Shuang ta Foods Co., Ltd. (Zhaoyuan, China); WPI was obtained from Volac Co., Ltd. (Felinfach, Lampeter, UK); Calcium hydroxide (Ca(OH)2) was obtained from Henan Wan bang Chemical Technology Co., Ltd. (Zhoukou, China).
4.2. Processing of Beef Reticulum
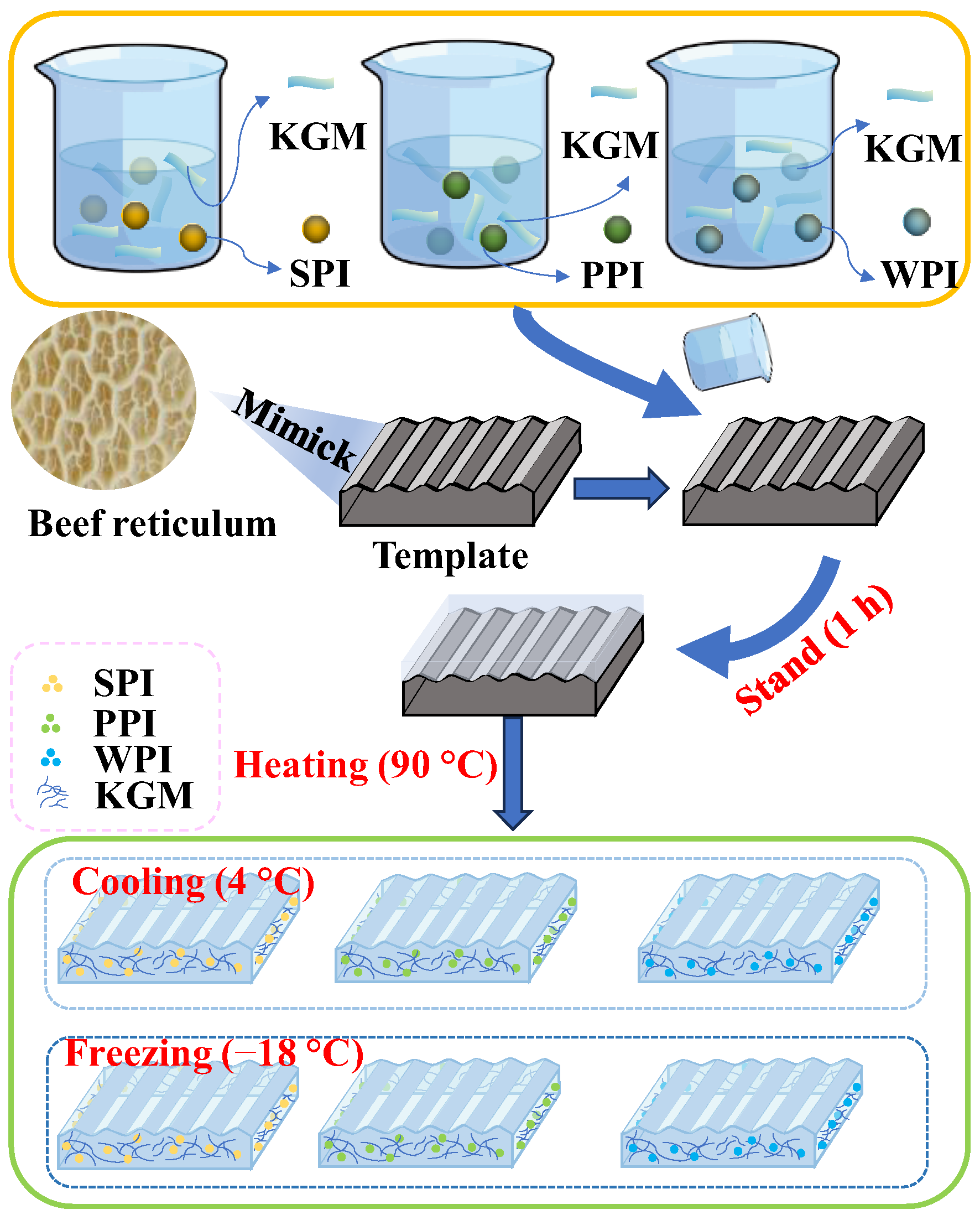
The fresh beef reticulum was washed with water to remove surface impurities (grass clippings, dust) and excess tissue (broken edges) with a knife and then was placed in a cooking pot and boiled in boiling water at 100 °C for 20 min. Take a part of it and cut it into regular pieces of 20 mm × 20 mm × 5 mm with a knife, and then cool it down to 25 °C and prepare it for use.
4.3. Preparation of Composite Gels
The preparation of composite gels was performed according to the previous method, improving on it with pre-experiments [
19]. PPI, WPI, and SPI were hydrated at different concentrations (0–10%,
w/
w) with 50% deionized water for 30 min. The proteins were dissolved completely. Then, the protein solution was added with 5% KGM and mixed evenly. (The reason why the protein concentration was fixed in the range of 0–10% and the KGM concentration was fixed at 5% was because after a large number of pre-experiments, it was proved that the protein/KGM mixtures in this range had a certain degree of fluidity, which could be conveniently molded. And they can both form stable solid gels after heating). Simultaneously, a certain amount of Ca(OH)
2 (4 wt% of KGM mass) was added to make up for the mass difference with water (
Figure 8 and
Table 6). The mixture was placed into a silicone mold with a diameter of 20 mm × 20 mm × 5 mm, let stand for 1 h, and then placed in a water bath for heating and shaping (90 °C, 1 h). After the samples were recovered to 25 °C, the prepared samples were equally divided into two groups: one group was kept in the refrigerator at 4 °C for 12 h, which was the heating–cooling processing method, and the other group was kept in the refrigerator at −18 °C for 12 h, which was the heating–freezing processing method.
4.4. Texture Profile Analysis (TPA)
The prepared composite gel sample and beef reticulum were removed from the refrigerator and after recovery to 25 °C. The gel strength was measured using a TA-XT. plus texture analyzer (Stable Micro Systems Co., Ltd. (Godalming, Surrey, UK)). The testing conditions were as follows [
2]: pre-test speed of 2 mm/s, test speed of 1 mm/s, post-test speed of 2 mm/s, cylindrical P/20 probe, trigger force of 15.0 g, and pressing depth accounting for 50% of the sample height. The results of the hardness, springiness, and chewiness were analyzed using the Exponent Connect software (Version 8, Stable Micro Systems Co., Ltd. (Godalming, Surrey, UK)), which is compatible with the texture meter. Each sample was measured 5 times in parallel. The reticulum was measured in the same way.
4.5. Shear Force
The shear force of the sample was measured using the A/MORS Meat of the texture analyzer (TA-XT. plus, Stable Micro Systems, UK). The experimental parameters were set as follows: A/MORS probe, pre-test speed of 1.0 mm/s, test speed of 1.0 mm/s, post-test speed of 10.0 mm/s, measured strain of 80%, trigger force of 15.0 g. The results of the shear force were analyzed using the Exponent Connect software (Stable Micro Systems, UK). Each sample was measured 5 times in parallel. The reticulum was measured in the same way.
4.6. Color
Referring to the method of Yao Li [
37], a colorimeter (CS-220, Hangzhou Cai pu Technology Co., Ltd. (Hangzhou, China)) was used to measure the color of beef reticulum and composite gel simulants. The results are expressed in L* (lightness), a* (redness/greenness), b* (yellowness/blueness), whiteness value, and color difference value (△E). Each sample was measured 5 times in parallel. Reticulum was measured in the same way.
The formula for calculating the whiteness value is presented as follows:
The calculation formula of color difference value (ΔE) is presented as follows:
4.7. Sensory Evaluation
During a 5-day training session with 15 teachers and students (7 female and 8 male, mean age 29.5 ± 13.59 years) of the food program according to the International Standard method (ISO 8586-1 2012 [
49]), the participants were familiarized with the odors of H
2S and NH3 as well as the texture of tripe to understand the criteria for the assessment of odor and texture [
50]. At the end of the training, a sensory evaluation panel evaluated the acceptability of samples screened using cluster analysis for color, tissue morphology, tactile sensation, taste sensation, and odor. At the completion of the training, each member received five de-identified samples placed in white plastic trays for sensory evaluation of color, histomorphology, taste, and odor, with effects between samples eliminated by referencing purified water. Acceptability of color, tissue morphology, tactile sensation, taste sensation, and odor was assessed using the 5-point descriptive scale, where score 1 corresponds to completely unacceptable and score 5 corresponds to very acceptable. The study was conducted at Chengdu University with the approval of the Ethics Committee, and all participants gave informed verbal consent for the analytical tests and product contents, including allergens. Participants had the right to withdraw at any time during the process.
4.8. Rheological Property
The textural properties of food products are usually associated with rheological properties [
51]. The rheological properties of the composite gels were determined using an Anton Paar MCR302 rheometer using a PP 25 parallel plate fixture with a test gap of 5 mm. After loading the samples, the excess sample was removed and a layer of silicone oil was covered around the edges of the samples to prevent evaporation of water during the test. Strain scans were first carried out at 25 °C with a frequency of 1 Hz, an initial strain of 0.01%, a final strain of 100%, and a point count of 30 to determine the Linear Viscoelastic Range (LVR) and gel properties in this range. The LVR can help to pre-set consistent strain and stress values for further measurements. Subsequently, frequency scans were performed to characterize the viscoelastic properties. Frequency scanning: Frequency scanning (100–0.1 rad/s) was performed in the gel state at 25 °C, and a strain of 1% was chosen to ensure that it was within the linear viscoelastic region [
52]. The samples were sealed with silicone oil to prevent evaporation.
4.9. Fourier Transform Infrared Spectroscopy (FTIR)
The molecular structure of the single KGM gel group K versus the screened optimum gel formulation group WK4 was analyzed using an infrared spectrometer (Spectrum 3, Agilent Technologies Inc., Santa Clara, CA, USA). About 2 mg of lyophilized sample was weighed and mixed thoroughly with 200 mg of potassium bromide powder, and the infrared spectrograms were obtained by scanning at 25 °C over a range of 4000–400 cm
−1 with a resolution of 4 cm
−1 and 32 scans [
53].
4.10. X-ray Diffraction Analysis (XRD)
XRD is a technique to determine the crystalline structure of materials [
47]. A powder X-ray diffractometer (D8, Bruker Co., Ltd., Karlsruhe, Germany) was used to test freeze-dried gels of a single KGM gel—group K—against a screened optimal gel formulation—group WK4. The test step size was 0.2°, the scanning rate was 2°/min, and the scanning angle range was 10°–60°.
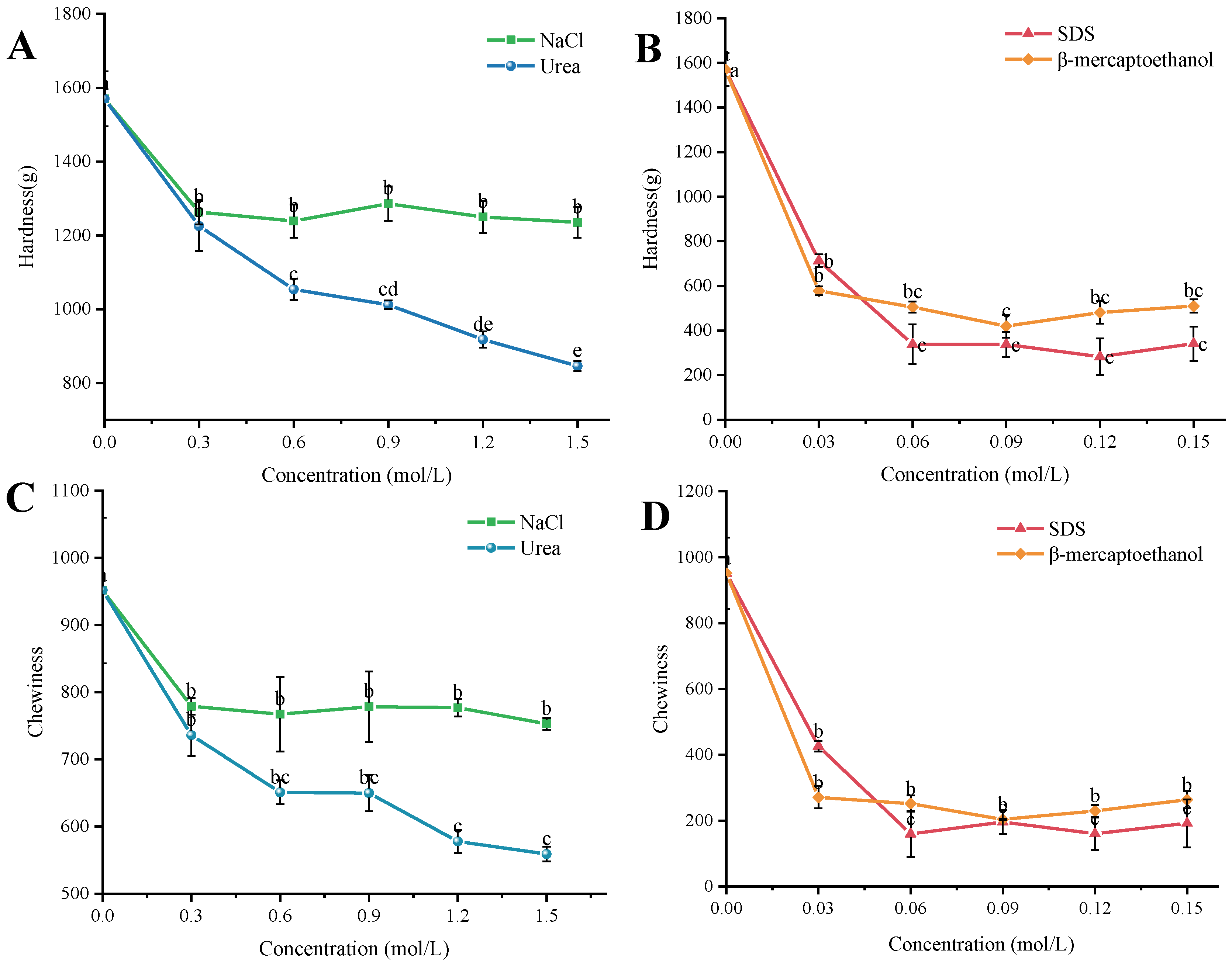
4.11. Degree of Contribution of Intermolecular Forces
In order to investigate the contribution of intermolecular forces in the formulation of the best simulants, experiments were carried out with reference to Yu [
54]. The gels were prepared using the preparation method of WK4 in
Table 6, and the prepared WK4 was placed in different concentration gradients of denaturants at 4 °C for 12 h. The control group was soaked in distilled water. Changes in hardness and chewiness of the gel samples after soaking with different disruptors were measured using a TA-XT. plus texture analyzer (the operating parameters of the texture analyzer are the same as in 2.4) to investigate the extent to which electrostatic, hydrogen, hydrophobic, and disulfide bonds contribute to the intermolecular forces in the composite gel system. Each sample was measured five times in parallel. The concentrations of the disruptive agents are shown below:
- (1)
Electrostatic interactions: NaCl was used to break the electrostatic interactions in the gel system, and NaCl was added at concentrations of 0.3 mol/L, 0.6 mol/L, 0.9 mol/L, 1.2 mol/L, and 1.5 mol/L. The electrostatic interactions in the gel system were analyzed by using NaCl.
- (2)
Hydrogen-bonding interactions: Urea was used to destroy the hydrogen-bonding interactions in the gel system, and the urea was added at concentrations of 0.3 mol/L, 0.6 mol/L, 0.9 mol/L, 1.2 mol/L, and 1.5 mol/L.
- (3)
Hydrophobic interactions: Sodium dodecyl sulfate (SDS) was used to destroy the hydrophobic interactions in the gel system, and the concentrations of SDS were 0.03 mol/L, 0.06 mol/L, 0.09 mol/L, 0.12 mol/L, and 0.15 mol/L. The hydrophobic interactions in the gel system were also destroyed by the use of SDS.
- (4)
Disulfide bonding interactions: β-mercaptoethanol was used to destroy the disulfide bonding interactions in the gel system, and β-mercaptoethanol was added at concentrations of 0.03 mol/L, 0.06 mol/L, 0.09 mol/L, 0.12 mol/L, and 0.15 mol/L.
4.12. Scanning Electron Microscope (SEM) Observation
An S-4800 scanning electron microscope (HITACHI Co., Ltd., Tokyo, Japan) was utilized in this study. Freeze-dried samples of K and WK4 were fractured to reveal their cross-sectional areas and were subsequently coated with gold for 60 s prior to observation. The microstructure was then examined at an accelerating voltage of 3.0 kV and a magnification of 500 times.
4.13. Statistical Analysis
The results of the experiments are reported as the mean ± standard deviation. A one-way ANOVA was performed using SPSS 26 statistical software (IBM Corp., Armonk, NY, USA) to determine significant differences. A correlation analysis and cluster analysis of tripe and 32 gels prepared under different conditions were performed using Chiplot. Plots were plotted using Origin software (version 2018).