Removal of Malachite Green by Poly(acrylamide-co-acrylic acid) Hydrogels: Analysis of Coulombic and Hydrogen Bond Donor–Acceptor Interactions
Abstract
:1. Introduction
2. Results and Discussion
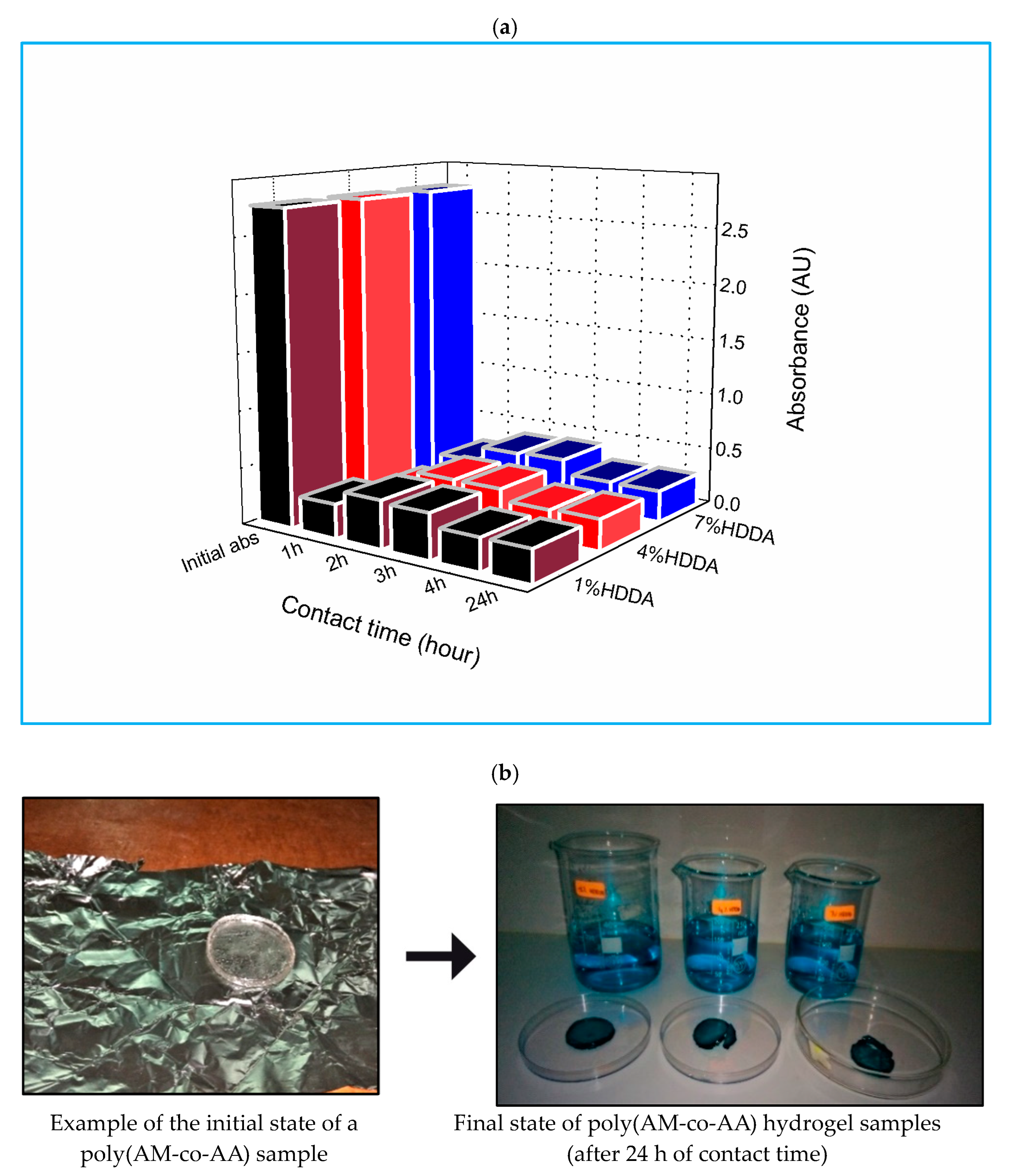
2.1. MG Absorbance on Crosslinked Poly(AM-co-AA)
2.2. Ionic Interaction in the Poly(AM-co-AA)/MG Hydrogel
2.3. Effect of Initial Monomer Concentration on Equilibrium Swelling
2.4. Molecular Docking Analysis
2.4.1. Donor and Acceptor Sites to Build the Hydrogen Bond
2.4.2. Hydrogen Bonding Interaction
2.5. Discussion of the Results with Other Work and Effects of Other Parameters
2.5.1. Removal Percentage
2.5.2. pH Effect
2.5.3. Water Quality Effect
2.5.4. Temperature Effect and Recycling of the Hydrogel
3. Conclusions
4. Materials and Methods
4.1. Materials
4.2. Preparation of Photochemically Crosslinked Copolymers
4.3. Study of the Swelling of the Copolymer Hydrogels in the MG Solution
4.4. Model Proposition and Software
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- de Moraes, K.R.; Souza, A.T.; Bartoň, D.; Blabolil, P.; Muška, M.; Prchalová, M.; Randák, T.; Říha, M.; Vašek, M.; Turek, J.; et al. Can a Protected Area Help Improve Fish Populations under Heavy Recreation Fishing? Water 2023, 15, 632. [Google Scholar] [CrossRef]
- Zou, J.; Chen, X.; Liu, F.; Wang, F.; Du, M.; Wu, B.; Yang, N. The Effects of Economic Policy Instruments of Diffuse Water Pollution from Agriculture: A Comparative Analysis of China and the UK. Water 2023, 15, 637. [Google Scholar] [CrossRef]
- Yang, X.; Xia, J.; Liu, J.; Li, J.; Wang, M.; Li, Y. Evolutionary Characteristics of Runoff in a Changing Environment: A Case Study of Dawen River, China. Water 2023, 15, 636. [Google Scholar] [CrossRef]
- Winarto; Yamamoto, E.; Yasuoka, K. Water Molecules in a Carbon Nanotube under an Applied Electric Field at Various Temperatures and Pressures. Water 2017, 9, 473. [Google Scholar] [CrossRef]
- Flores Alarcón, M.A.D.; Revilla Pacheco, C.; Garcia Bustos, K.; Tejada Meza, K.; Terán-Hilares, F.; Pacheco Tanaka, D.A.; Colina Andrade, G.J.; Terán-Hilares, R. Efficient Dye Removal from Real Textile Wastewater Using Orange Seed Powder as Suitable Bio-Adsorbent and Membrane Technology. Water 2022, 14, 4104. [Google Scholar] [CrossRef]
- Vanitha, K.; Jibrail, K.; Sie, Y.L. Efficiency of various recent wastewater dye removal methods: A review. J. Env. Chem. Eng. 2018, 6, 4676–4697. [Google Scholar] [CrossRef]
- Wong, S.; Ghafar, N.A.; Ngadi, N.; Razmi, F.A.; Inuwa, I.M.; Mat, R.; Amin, N.A.S. Effective removal of anionic textile dyes using adsorbent synthesized from coffee waste. Sci. Rep. 2020, 10, 2928. [Google Scholar] [CrossRef]
- Mojiri, A. Removal of Dye from Aquatic Environments: State-of-the-Art and Future Perspectives. Separations 2022, 9, 375. [Google Scholar] [CrossRef]
- Ledakowicz, S.; Paździor, K. Recent Achievements in Dyes Removal Focused on Advanced Oxidation Processes Integrated with Biological Methods. Molecules 2021, 26, 870. [Google Scholar] [CrossRef]
- Ihaddaden, S.; Aberkane, D.; Boukerroui, A.; Robert, D. Removal of methylene blue (basic dye) by coagulation-flocculation with biomaterials (bentonite and Opuntia ficus indica). J. Water Proc. Eng. 2022, 49, 102952. [Google Scholar] [CrossRef]
- Yang, J.; Shojaei, S.; Shojaei, S. Removal of drug and dye from aqueous solutions by graphene oxide: Adsorption studies and chemometrics methods. Npj Clean Water 2022, 5, 5. [Google Scholar] [CrossRef]
- Wei, Q.; Zhang, Y.; Zhang, K.; Mwasiagi, J.I.; Zhao, X.; Chow, C.W.K.; Tang, R. Removal of direct dyes by coagulation: Adaptability and mechanism related to the molecular structure. Korean J. Chem. Eng. 2022, 39, 1850–1862. [Google Scholar] [CrossRef]
- Mustafa, T.Y.; Tushar, K.S.; Sharmeen, A.; Ang, H.M. Dye and its removal from aqueous solution by adsorption: A review. Adv. Colloid Interface Sci. 2014, 209, 172–184. [Google Scholar] [CrossRef]
- Enas, M.A. Hydrogel: Preparation, characterization, and applications: A review. J. Adv. Res. 2015, 6, 105–121. [Google Scholar] [CrossRef]
- Ali, I.; Alharbi, O.M.L.; ALOthman, Z.A.; Alwarthan, A.; Al-Mohaimeed, A.M. Preparation of a carboxymethylcellulose-iron composite for uptake of atorvastatin in water. Int. J. Biol. Macromol. 2019, 132, 244–253. [Google Scholar] [CrossRef]
- Ali, I.; Alharbi, O.M.L.; ALOthman, Z.A.; Al-Mohaimeed, A.M.; Alwarthan, A. Modeling of fenuron pesticide adsorption on CNTs for mechanistic insight and removal in water. Environ. Res. 2019, 170, 389–397. [Google Scholar] [CrossRef] [PubMed]
- Azam, M.; Wabaidur, S.M.; Khan, M.R.; Al-Resayes, S.I.; Islam, M.S. Heavy Metal Ions Removal from Aqueous Solutions by Treated Ajwa Date Pits: Kinetic, Isotherm, and Thermodynamic Approach. Polymers 2022, 14, 914. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.A.; Otero, M.; Kazi, M.; Alqadami, A.A.; Wabaidur, S.M.; Siddiqui, M.R.; Alothman, Z.A.; Sumbul, S. Unary and binary adsorption studies of lead and malachite green onto a nanomagnetic copper ferrite/drumstick pod biomass composite. J. Hazard. Mater. 2019, 365, 759–770. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.A.; Wabaidur, S.M.; Siddiqui, M.R.; Alqadami, A.A.; Khan, A.H. Silico-manganese fumes waste encapsulated cryogenic alginate beads for aqueous environment de-colorization. J. Clean. Prod. 2020, 244, 118867. [Google Scholar] [CrossRef]
- Mansri, A.; Mahroug, H.; Dergal, F. In situ preparation of hydroxyapatite composites into hydrolyzed polyacrylamide solution and methylene blue dye retention. Turk. J. Chem. 2019, 43, 213–228. [Google Scholar] [CrossRef]
- Şolpan, D.; Sibel Duran, S.; Torun, M. Removal of cationic dyes by poly(acrylamide-co-acrylic acid) hydrogels in aqueous solutions. Radiat. Phys. Chem. 2008, 4, 447–452. [Google Scholar] [CrossRef]
- Hamri, S.; Bouchaour, T.; Lerari, D.; Bouberka, Z.; Supiot, P.; Maschke, U. Cleaning of Wastewater Using Crosslinked Poly(Acrylamide-co-Acrylic Acid) Hydrogels: Analysis of Rotatable Bonds, Binding Energy and Hydrogen Bonding. Gels 2022, 8, 156. [Google Scholar] [CrossRef] [PubMed]
- Bendahma, Y.H.; Hamri, S.; Merad, M.; Bouchaour, T.; Maschke, U. Conformational modeling of the system pollutant/three-dimensional poly (2-hydroxyethyl methacrylate) (PHEMA) in aqueous medium: A new approach. Polym. Bull. 2019, 76, 1517–1530. [Google Scholar] [CrossRef]
- Shivaji, S.; Ranjana, S.; Roy, D. Toxicological effects of malachite green: Review. Aquat. Toxicol. 2004, 3, 319–329. [Google Scholar] [CrossRef]
- Aldert, A.B.; Peter, S. Determination of residues of malachite green in aquatic animals. J. Chromatogr. B 2003, 2, 351–359. [Google Scholar] [CrossRef]
- Al-Tohamy, R.; Ali, S.S.; Li, F.; Okasha, K.M.; Mahmoud, Y.A.-G.; Elsamahy, T.; Jiao, H.; Fu, Y.; Sun, J. A critical review on the treatment of dye-containing wastewater: Ecotoxicological and health concerns of textile dyes and possible remediation approaches for environmental safety. Ecotoxicol. Environ. Saf. 2022, 231, 113160. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Oh, S.; Rakwal, R.; Liu, C.; Zhang, Z. Environmental and Health Impacts of Textile Dyeing Industry. Int. J. Environ. Sci. Dev. 2013, 5, 456–460. [Google Scholar] [CrossRef]
- Peighambardoust, S.J.; Aghamohammadi-Bavil, O.; Foroutan, R.; Arsalani, N. Removal of malachite green using carboxymethyl cellulose-g-polyacrylamide/montmorillonite nanocomposite hydrogel. Int. J. Biol. Macromol. 2020, 159, 1122–1131. [Google Scholar] [CrossRef]
- Kaur, K.; Khushbu; Vaid, V.; Anupama; Anshul; Ankush; Jindal, R. Efficient removal of Rose Bengal and Malachite Green dyes using Green and sustainable Chitosan/CMC/Bentonite-based hydrogel materials. Polym. Bull. 2023, 80, 6609. [Google Scholar] [CrossRef]
- Layth, S.J.; Nadher, D.R.; Hayder, O.J. Synthesis and Characterization of Poly (Acryl Amide–Maleic Acid) Hydrogel: Adsorption Kinetics of a Malachite Green from Aqueous Solutions. Eurasian J. Anal. Chem. 2018, 13, em74. [Google Scholar] [CrossRef]
- Benchohra, M.; Bouazzaoui, F.; Karapinar, E.; Salim, A. Controllability of Second Order Functional Random Differential Equations with Delay. Mathematics 2022, 10, 1120. [Google Scholar] [CrossRef]
- Aziz, K.B.; Salh, M.D.; Kaufhold, S.; Bertier, P. The High Efficiency of Anionic Dye Removal Using Ce-Al13/Pillared Clay from Darbandikhan Natural Clay. Molecules 2019, 24, 2720. [Google Scholar] [CrossRef] [PubMed]
- Chauhdary, Y.; Hanif, M.A.; Rashid, U.; Bhatti, I.A.; Anwar, H.; Jamil, Y.; Alharthi, F.A.; Kazerooni, E.A. Effective Removal of Reactive and Direct Dyes from Colored Wastewater Using Low-Cost Novel Bentonite Nanocomposites. Water 2022, 14, 3604. [Google Scholar] [CrossRef]
- Reddy, A.V.B.; Rafiq, R.; Ahmad, A.; Maulud, A.S.; Moniruzzaman, M. Cross-Linked Ionic Liquid Polymer for the Effective Removal of Ionic Dyes from Aqueous Systems: Investigation of Kinetics and Adsorption Isotherms. Molecules 2022, 27, 7775. [Google Scholar] [CrossRef]
- Gul, S.; Kanwal, M.; Qazi, R.A.; Gul, H.; Khattak, R.; Khan, M.S.; Khitab, F.; Krauklis, A.E. Efficient Removal of Methyl Red Dye by Using Bark of Hopbush. Water 2022, 14, 2831. [Google Scholar] [CrossRef]
- Silva, M.E.S.; Dutra, E.R.; Mano, V.M.; Machado, J.C. Preparation and thermal study of polymers derived from acrylamide. Polym. Degrad. Stab. 2000, 3, 491–495. [Google Scholar] [CrossRef]
- Nicholas, P.; Cheremisinoff, M. Cheremisinoff, Condensed Encyclopedia of Polymer Engineering Terms; Nicholas, P., Ed.; Butterworth-Heinemann: Oxford, UK, 2001; pp. 165–182. ISBN 9780080502823. [Google Scholar]
- Crivello, J.V.; Reichmanis, E. Photopolymer Materials and Processes for Advanced Technologies. Chem. Mater. 2014, 26, 533–548. [Google Scholar] [CrossRef]
- Kim, D.; Seo, K.; Park, K. Polymer composition and acidification effects on the swelling and mechanical properties of poly(acrylamide-co-acrylic acid) superporous hydrogels. J. Biomater. Sci. Polym. Ed. 2004, 15, 189–199. [Google Scholar] [CrossRef]
- International Union of Pure and Applied Chemistry. ‘Ionic Bond’ in IUPAC Compendium of Chemical Terminology, 3rd ed.; International Union of Pure and Applied Chemistry: North Carolina, USA, 2006; Online version 3.0.1, 2019. [Google Scholar] [CrossRef]
- Schneider, H.J. Ionic Interactions in Supramolecular Complexes. In Ionic Interactions in Natural and Synthetic Macromolecules; Wiley Online Library: Hoboken, NJ, USA, 2012; pp. 35–47. ISBN 9781118165850. [Google Scholar] [CrossRef]
- Hua, Y.; Xu, D.; Liu, Z.; Zhou, J.; Han, J.; Lin, Z.; Xu, D.; Chen, G.; Huang, X.; Chen, J.; et al. Effective adsorption and removal of malachite green and Pb2+ from aqueous samples and fruit juices by pollen–inspired magnetic hydroxyapatite nanoparticles/hydrogel beads. J. Clean. Prod. 2023, 411, 137233. [Google Scholar] [CrossRef]
- Alothman, Z.A.; Bahkali, A.H.; Khiyami, M.A.; Alfadul, S.M.; Wabaidur, S.M.; Alam, M.; Alfarhan, B.Z. Low cost biosorbents from fungi for heavy metals removal from wastewater. Sep. Sci. Technol. 2020, 55, 1766–1775. [Google Scholar] [CrossRef]
- Kenawy, E.R.; Ghfar, A.A.; Wabaidur, S.M.; Khan, M.A.; Siddiqui, M.R.; Alothman, Z.A.; Alqadami, A.A.; Hamid, M. Cetyltrimethylammonium bromide intercalated and branched polyhydroxystyrene functionalized montmorillonite clay to sequester cationic dyes. J. Environ. Manag. 2018, 219, 285–293. [Google Scholar] [CrossRef]
- Yinuo Li, Y.; Ran, T.; Yang, H.; Dong, Z.; Shi, Y. Ir-reversible on/off switching hydrogel for efficient dye capture and release. J. Env. Chem. Eng. 2023, 11, 109829. [Google Scholar] [CrossRef]
- Seddiki, N.; Aliouche, D. Synthesis, characterization and rheological behavior of pH sensitive poly(acrylamide-co-acrylic acid) hydrogels. Arab. J. Chem. 2017, 10, 539–547. [Google Scholar] [CrossRef]
- Imran Ali, I.; Peng, C.; Ye, T.; Naz, I. Sorption of cationic malachite green dye on phytogenic magnetic nanoparticles functionalized by 3-marcaptopropanic acid. RSC Adv. 2018, 8, 8878–8897. [Google Scholar] [CrossRef]
- Alqadami, A.A.; Wabaidur, S.M.; Jeon, B.H.; Khan, M.A. Co-hydrothermal valorization of food waste: Process optimization, characterization, and water decolorization application. Biomass. Conv. Bioref. 2023, 1–12. [Google Scholar] [CrossRef]
- Betraoui, A.; Seddiki, N.; Souag, R.; Guerfi, N.; Semlali, A.; Aouak, T.; Aliouche, D. Synthesis of New Hydrogels Involving Acrylic Acid and Acrylamide Grafted Agar-Agar and Their Application in the Removal of Cationic Dyes from Wastewater. Gels 2023, 9, 499. [Google Scholar] [CrossRef]
- Zhenyu, Y.; Jie, W.; Yiming, W.; Qian, L.; Yujie, Z.; Yu, W.; Li, L.; Stephen, F.L.; Xuhong, G. Preparation of a poly(acrylic acid) based hydrogel with fast adsorption rate and high adsorption capacity for the removal of cationic dyes. RSC Adv. 2019, 9, 21075–21085. [Google Scholar] [CrossRef]
- Mousavi, H.Z.; Khaligh, A.; Behzad, M.; Rahchamani, J. Application of Polyacrylamide for Methylene Blue Removal from Aqueous Solutions. J. Appl. Sol. Chem. Model. 2014, 3, 39–47. [Google Scholar] [CrossRef]
- Zhao, Z.; Li, L.; Geleta, G.S.; Ma, L.; Wang, Z. Polyacrylamide-Phytic Acid-Polydopamine Conducting Porous Hydrogel for Efficient Removal of Water-Soluble Dyes. Sci. Rep. 2017, 7, 7878. [Google Scholar] [CrossRef] [PubMed]
- Mihoub, M.; Hamri, S.; Bouchaour, T.; Popa, M.; Popa, D.M.; Bedjaoui Alachaher, L.; Hamcerencu, M. An Interpenetrating Polymer Network Hydrogel Based on Cellulose, Applied to Remove Colorant Traces from the Water Medium: Electrostatic Interactions Analysis. Polymers 2022, 14, 5090. [Google Scholar] [CrossRef]
- Hamri, S.; Lerari, D.; Sehailia, M.; Dali-Youcef, B.; Bouchaour, T.; Bachari, K. Prediction of equilibrium swelling ratio on synthesized polyacrylamide hydrogel using central composite design modeling. Int. J. Plast. Technol. 2018, 22, 247–261. [Google Scholar] [CrossRef]










| Copolymer Network | MG Removal (%) |
|---|---|
| Poly(AMC2-co-AA)/4 wt % HDDA | 96.4 |
| Poly(AMC2-co-AA)/7 wt % HDDA | 95.7 |
| Poly(AMC2-co-AA)/1 wt % HDDA | 95.5 |
| Poly(AMC1-co-AA)/4 wt % HDDA | 93.1 |
| Poly(AMC1-co-AA)/7 wt % HDDA | 91.0 |
| Poly(AMC1-co-AA)/1 wt % HDDA | 89.0 |
| Poly(AMC3-co-AA)/7 wt % HDDA | 85.3 |
| Poly(AMC3-co-AA)/1 wt % HDDA | 84.5 |
| Poly(AMC3-co-AA)/4 wt % HDDA | 83.7 |
| Equilibrium Swelling (ES) (%) | |||
|---|---|---|---|
| Poly(AMC1-co-AA) | Poly(AMC2-co-AA) | Poly(AMC3-co-AA) | |
| 1 wt % HDDA | 96.5 | 89.1 | 97.9 |
| 4 wt % HDDA | 72.7 | 40.6 | 72.9 |
| 7 wt % HDDA | 67.6 | 20.1 | 51.9 |
| Models | Hydrogen Bond Acceptor (HBA) | Hydrogen Bond Donor (HBD) | Rotatable Bond |
|---|---|---|---|
| Model 1 | 16 | 7 | 121 |
| Model 2 | 16 | 12 | 91 |
| Model 3 | 16 | 16 | 50 |
| MG | 0 | 2 | 6 |
| Models | Hydrogen Bond | Distance (Å) |
|---|---|---|
| Model 1 | N……H | 4.19 |
| 6.25 | ||
| 6.50 | ||
| 6.20 | ||
| Model 2 | N……H | 5.21 |
| 9.64 | ||
| Model 3 | N……H | 3.74 |
| 4.65 | ||
| 6.29 |
| Models | Mode | Affinity (kcal/mol) | Dist. from Rmsd l.b. (Å) |
|---|---|---|---|
| Model 1 | |||
| 1 | −4.9 | 0.000 | |
| 2 | −3.7 | 14.657 | |
| 3 | −3.5 | 14.084 | |
| 4 | −3.5 | 13.731 | |
| 5 | −3.4 | 6.865 | |
| 6 | −3.3 | 6.035 | |
| 7 | −3.3 | 9.936 | |
| 8 | −3.3 | 2.641 | |
| 9 | −3.2 | 12.364 | |
| Model 2 | |||
| 1 | −4.2 | 0.000 | |
| 2 | −4.1 | 14.630 | |
| 3 | −4.1 | 21.335 | |
| 4 | −3.3 | 18.793 | |
| 5 | −3.2 | 5.862 | |
| 6 | −3.2 | 35.362 | |
| 7 | −3.2 | 7.697 | |
| 8 | −2.9 | 33.130 | |
| 9 | −2.7 | 31.404 | |
| Model 3 | |||
| 1 | −4.3 | 0.000 | |
| 2 | −3.7 | 5.015 | |
| 3 | −3.4 | 12.720 | |
| 4 | −3.3 | 22.798 | |
| 5 | −3.2 | 14.812 | |
| 6 | −2.9 | 8.349 | |
| 7 | −2.9 | 15.384 | |
| 8 | −2.8 | 17.327 | |
| 9 | −2.7 | 22.691 |
| Copolymer Network * | Solution AM (mg/mL) | Darocur 1173 (wt %) | HDDA (wt %) |
|---|---|---|---|
| PAMC1-co-AA | 134 | 0.5 | 1 |
| 4 | |||
| 7 | |||
| PAMC2-co-AA | 100 | 0.5 | 1 |
| 4 | |||
| 7 | |||
| PAMC3-co-AA | 67 | 0.5 | 1 |
| 4 | |||
| 7 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hamri, S.; Bouzi, B.; Lerari, D.; Dergal, F.; Bouchaour, T.; Bachari, K.; Bouberka, Z.; Maschke, U. Removal of Malachite Green by Poly(acrylamide-co-acrylic acid) Hydrogels: Analysis of Coulombic and Hydrogen Bond Donor–Acceptor Interactions. Gels 2023, 9, 946. https://doi.org/10.3390/gels9120946
Hamri S, Bouzi B, Lerari D, Dergal F, Bouchaour T, Bachari K, Bouberka Z, Maschke U. Removal of Malachite Green by Poly(acrylamide-co-acrylic acid) Hydrogels: Analysis of Coulombic and Hydrogen Bond Donor–Acceptor Interactions. Gels. 2023; 9(12):946. https://doi.org/10.3390/gels9120946
Chicago/Turabian StyleHamri, Salah, Bouchra Bouzi, Djahida Lerari, Fayçal Dergal, Tewfik Bouchaour, Khaldoun Bachari, Zohra Bouberka, and Ulrich Maschke. 2023. "Removal of Malachite Green by Poly(acrylamide-co-acrylic acid) Hydrogels: Analysis of Coulombic and Hydrogen Bond Donor–Acceptor Interactions" Gels 9, no. 12: 946. https://doi.org/10.3390/gels9120946





