The use of glycerol, which is a renewable resource, as starting material for the biotechnological production of 3HP, is a common strategy. It can be obtained as a by-product of biodiesel production. Biodiesel is produced from the transesterification of oils with a short-chain alcohol (mainly methanol) in the presence of a catalyst [
5]. During biodiesel production, crude glycerol is generated as by-product in a ratio of about 100 kg glycerol to 1 ton of biodiesel produced [
6]. As the worldwide production of biodiesel is tending to increase, the amounts of crude glycerol that are available will follow the same trend in the future. It is estimated that the production of glycerol will reach 4.2 × 10
9 L in 2020, and that the world glycerol market will have a value of about 2.52 billion US dollars (USD) [
7,
8]. Moreover, the oversupply of glycerol leads to a decrease in its price [
9]. During 2013, the prices of refined glycerol varied from 900 USD/ton to 965 USD/ton, with the prices of the unrefined crude glycerol being as low as 240 USD/ton [
7]. The high quantities of crude glycerol that are produced require its treatment or further use, which, together with the low price, creates an ideal situation for the incorporation of crude glycerol into the biotechnological production of a variety of fuels and chemicals [
8,
9]. Moreover, upgrading of biodiesel-derived glycerol to high added-value products will boost the economy of the biodiesel production sector [
10]. Conversion of glycerol to 3HP is one of the available options for the utilization of glycerol, and much research effort is being made towards this conversion. Several bacterial strains, such as
Klebsiella sp., are capable of natural uptake of glycerol through oxidative or reductive pathways. In the oxidative pathway, this leads to the formation of pyruvate through the Embden–Meyerhof–Parnas pathway, whereas the reductive pathway results in the formation of 1,3-propanediol [
11]. Genetic manipulation of the natural glycerol consuming strains can result in the conversion of glycerol to 3HP, through 3-hydroxypropionaldehyde as an intermediate. To achieve this conversion, the genes of either the
dha operon or the
pdu operon are commonly used, as summarized in the following sections.
2.1. Production Using Genes That Are Part of the Dha Operon
The naturally occurring
dha operon encodes the necessary enzymes for the conversion of glycerol to 1,3-propanediol. A detailed description of the genes in the
dha operon and their action is given in our previous review article [
3]. Briefly, the operon mainly consists of genes encoding (1) a glycerol dehydratase, which catalyzes the conversion of glycerol to 3-hydroxypropionaldehyde; (2) a reactivase of the glycerol dehydratase; and (3) a 1,3-propanediol oxidoreductase, which catalyzes the conversion of 3-hydroxypropionaldehyde to 1,3-propanediol, with some structural differences in different microorganisms. Conversion of glycerol to 3HP using the
dha operon is performed in two stages, whereby glycerol is initially converted to 3-hydroxypropionaldehyde through the action of the enzyme glycerol dehydratase, followed by conversion of the 3-hydroxypropionaldehyde to 3HP by the action of an aldehyde dehydrogenase (ald) enzyme (
Figure 1). A strategy that is commonly employed is to use a microorganism that already possess the
dha operon, and genetically modify it to produce 3HP. In that case, the gene encoding the 1,3-propanediol oxidoreductase should be deleted or underexpressed if the co-production of 1,3-propanediol is not wanted, but special care should be taken to maintain a balance between NADH and NAD
+. Regeneration of NAD
+ is important, as it is required for the action of the ald enzyme. NAD
+ can be regenerated in the electron transport chain, which requires increased aeration, but the presence of oxygen inhibits the synthesis of coenzyme B
12, which is also involved in the process [
12]. Moreover, as will be discussed later, oxygen also inactivates the glycerol dehydratase. Regeneration of NAD
+ can also take place during the conversion of 3-hydroxypropionaldehyde to 1,3-propanediol by the action of the 1,3-propanediol oxidoreductase, which consumes NADH [
13]. If the 1,3-propanediol oxidoreductase gene is knocked out, another pathway for NAD
+ should be found in order to maintain the balance between NADH and NAD
+, such as lactate or acetate formation [
12]. On the other hand, co-production of 3HP and 1,3-propanediol can also be desirable, as 1,3-propanediol also has significant applications and these two molecules can be separated easily, due to the presence of different functional groups on the molecules [
14].
Another consideration when the
dha operon is used is that the first step of the reaction requires coenzyme B
12, which is produced naturally de novo by some bacterial strains, such as
Klebsiella pneumoniae [
15,
16], whereas other bacteria, such as
Escherichia coli, cannot produce it de novo [
16]. When the host microorganism is not capable of producing this coenzyme, external addition of it should be included in the cultivation—which will affect the cost of the process. An effort is therefore being made to create new bacterial strains that are capable of producing coenzyme B
12. Another consideration when using the
dha operon is the inactivation of the glycerol dehydratase during the catalysis of glycerol conversion [
17,
18], so proper expression of the glycerol dehydratase reactivase is important to maintain the activity of glycerol dehydratase and, in turn, 3HP production. Glycerol dehydratase can also be inactivated by oxygen [
19], which on the other hand, is necessary for efficient regeneration of the NAD
+ [
20] that is required for the action of the ald enzymes.
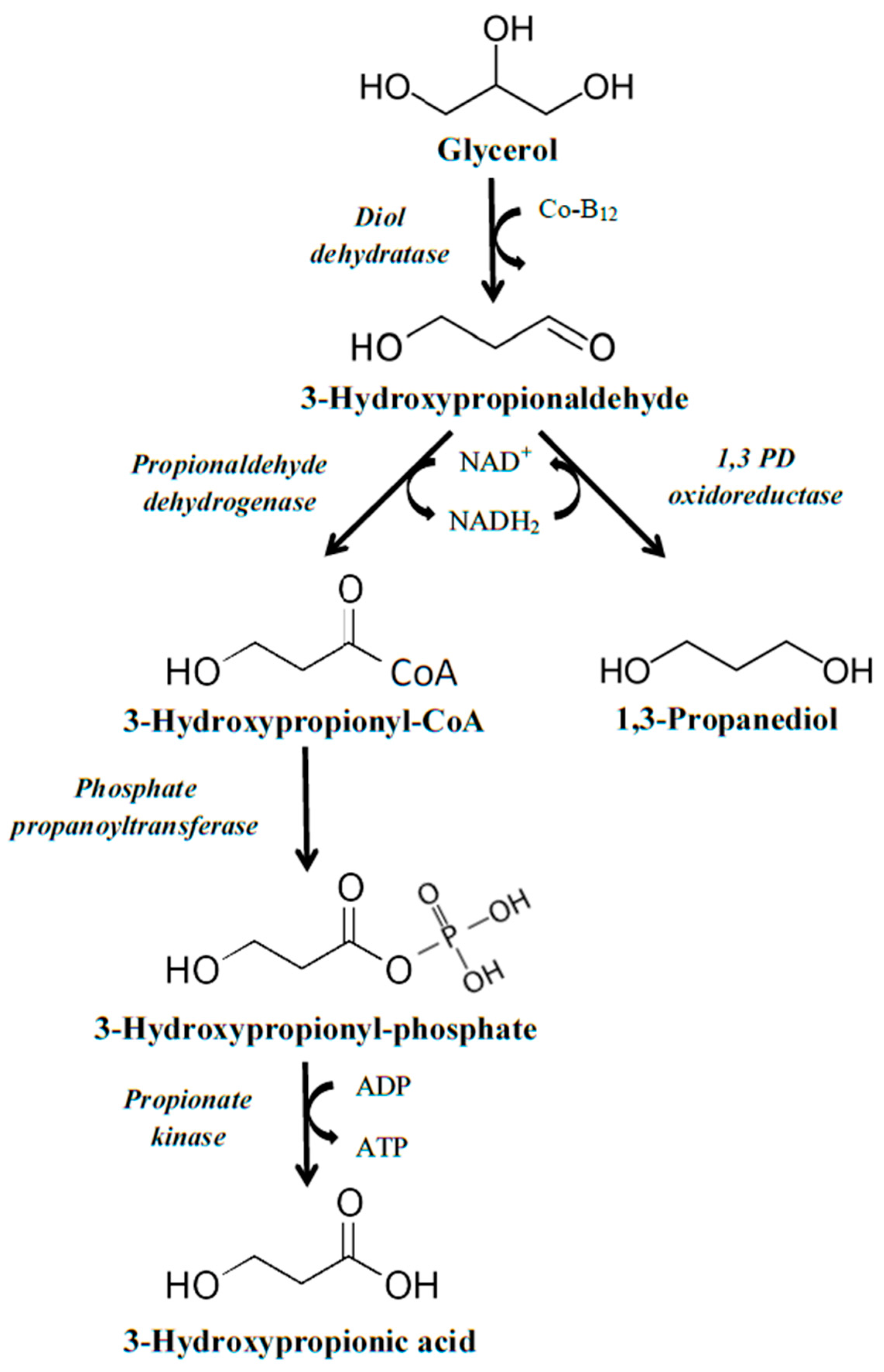
2.2. Production Using Genes That Are Part of the Pdu Operon
The second operon that can be used for the construction of a strain capable of converting glycerol to 3HP is the
pdu operon. In nature, the
pdu operon is required for microbial growth on 1,2-propanediol and it can be found in
Salmonella and
Lactobacillus species [
21,
22]. First, 1,2-propanediol is converted to propionaldehyde by the action of an AdoCbI-dependent diol dehydratase; this is further converted to propionic acid and propanol by the action of the enzymes CoA-dependent aldehyde dehydrogenase, phosphotransacylase, propionate kinase, and alcohol dehydrogenase [
22]. Regarding the conversion of glycerol to 3HP by the enzymes encoded in the
pdu operon, more intermediate steps are required than when using the
dha operon. Initially, glycerol is converted to 3-hydroxypropionaldehyde by the action of the diol dehydratase, which is followed by its transformation to 3-hydroxypropionyl-CoA by the action of propionaldehyde dehydrogenase. The third step consists of phosphorylation of 3-hydroxypropionyl-CoA to 3-hydroxypropionyl phosphate by phosphate propanoyltransferase. Finally, dephosphorylation of 3-hydroxypropionyl phosphate, by the enzyme propionate kinase, leads to the formation of 3HP (
Figure 2).
Similarly to the
dha pathway, the use of the
pdu pathway presents some challenges that should be taken into account when intending to use it for industrial production of 3HP. As NAD
+ is required during the second step of the pathway (conversion of 3-hydroxypropionaldehyde to 3-hydroxypropionyl-CoA), an adequate supply of NAD
+ is necessary. Similar strategies and considerations as described before apply here also, with the co-production of 1,3-propanediol serving as a promising solution, as the action of 1,3-propanediol oxidoreductase requires NADH, and therefore, NAD
+ is regenerated [
23,
24], maintaining the equilibrium between NAD
+ and NADH in the process.
2.3. Engineering of K. pneumoniae Cells for the Conversion of Glycerol to 3HP
K. pneumoniae is an important candidate for use in the conversion of glycerol to 3HP.
K. pneumoniae can naturally produce the required coenzyme B
12 de novo, which is important for the economics of the process, as there is no need for external addition of coenzyme B
12. Moreover, the
dha operon is endogenous in
K. pneumoniae, thus minimizing the transfer of required genes when constructing a strain capable of producing 3HP, and has excellent glycerol fermentation capability [
25]. On the other hand, as 1,3-propanediol is produced through the
dha operon, the genes encoding the 1,3-propanediol oxidoreductase should be deleted, unless co-production of 1,3-propanediol is desired. On the downside, use of
K. pneumoniae can potentially give rise to public health concerns. Bacteria of the
Klebsiella genus can cause human nosocomial infection, due to their ability to spread rapidly in hospital environments.
K. pneumoniae, in particular, is the most important of the
Klebsiella genus, from a medical point of view, as it can cause a broad variety of infections, such as urinary tract infections, soft tissue infections, septicemias, and pneumonia in hospital environments [
26].
As was discussed before, the balance of the cofactors NADH and NAD
+ is important during the conversion of glycerol to 3HP. A strategy to maintain this balance is the co-production of 3HP and 1,3-propanediol. For example, Su et al. [
27] constructed a strain through the heterologous expression of
DhaS, a putative ald from
Bacillus subtilis, which showed higher specificity toward 3-hydroxypropionic acid than toward other aldehydes (propionaldehyde, benzaldehyde, valeraldehyde, butyraldehyde, and acetaldehyde). The recombinant
K. pneumoniae strain was capable of producing 3HP at 18.0 g/L, and 1,3-propanediol at 27 g/L in 24 h under non-optimized bioreactor conditions. During the cultivation, a significant amount of lactic acid (36.1 g/L) was also produced (
Table 1). The authors suggested that some possible reasons for the low 3HP concentration might be the competition for NAD
+ between 3HP and biomass formation, the toxicity of 3HP toward the host cells, and the non-optimal activity of the ald toward 3-hydroxypropionaldehyde. It is obvious from the above that an adequate supply of NAD
+ is necessary to achieve a high degree of 3HP production, and the discovery of more efficient alds is also important.
The strategy of co-production was also followed by Huang et al. [
28], who expressed the
aldH gene from
E. coli in
K. pneumoniae cells. The authors suggested that the relative amounts of 3HP and 1,3-propanediol could be controlled by controlling the aeration levels in the culture. Cell growth and production of 3HP were enhanced by increasing the aeration rate, but 1,3-propanediol production was reduced. Under fully aerobic conditions, however, the
dha operon was repressed—resulting in no production of either 3HP or 1,3-propanediol. The highest 3HP concentration (48.9 g/L) was reached under microaerobic conditions (1.5 vvm aeration, which resulted in decrease of the dissolved oxygen from 100% to 0% in 2.7 h of culture), with the simultaneous production of 25.3 g/L 1,3-propanediol after 28 h of cultivation in a fed-batch bioreactor. The overall yield of 1,3-propanediol and 3HP was 0.66 mol/mol. The authors also demonstrated that the formation of other by-products was also affected by the level of aeration under micro-anaerobic conditions, with the production of lactic acid and acetic acid being enhanced, whereas ethanol and succinic acid production was reduced by increasing the aeration level. Formate production initially increased with increasing levels of aeriation, followed by a sharp decrease when aeration was higher than 0.6 vvm. Ko et al. [
12] also attempted to control the production of other metabolic by-products, especially of acetic acid, with the aim of improving co-production of 3HP and 1,3-propanediol from the recombinant strain of
K. pneumoniae (J2B), which overexpressed the ald gene (encoding alpha-ketoglutaric semialdehyde dehydrogenase—
kgsadh). The methods that they evaluated for acetate reduction were reduction of the glycerol assimilation through the glycolytic pathway, increasing the glycerol flow towards 3HP and 1,3-propanediol formation, and finally controlling the aeration levels. To improve the co-production, the authors evaluated the deletion and overexpression of several genes. The best results were obtained when the genes encoding lactate dehydrogenase (
ldhA), succinate dehydrogenase (
frdA), and alcohol dehydrogenase (
adhE) were deleted, which—together with the overexpression of
dhaB and
gdrAB—resulted in 3HP at 43 g/L and 1,3-propanediol at 21 g/L during fed-batch bioreactor cultivation, with an overall yield of 0.49 mol/mol. Although these genetic manipulations reduced the amount of acetate, a considerable amount of acetate (>150 mM) had accumulated in the bioreactor by the end of the cultivation.
In another study conducted by Li et al. [
29], a systematic optimization of glycerol metabolism took place in order to improve the 3HP production titers. During this work, different promoters for the overexpression of
PuuC (a native ald of
K. pneumoniae), which is a key enzyme for 3HP formation, were investigated. Among the promoters tested (
tac and
lac), the IPTG-induced
tac was found to be the most efficient for overexpression of
PuuC. Moreover, 3HP production significantly increased when the synthesis of lactic acid and acetic acid was blocked. Finally, optimization of cultivation parameters, such as aeration (microaerobic conditions), pH (7.0), and IPTG concentration (0.02 mM) improved the production of 3HP even further, resulting in a concentration of 83.8 g/L with a yield 52 g/g after 72 h of cultivation. To the best of our knowledge, this concentration is the highest reported from glycerol. Although addition of IPTG is not optimal from an economic point of view, the amount required during this work was relatively low compared to what is commonly used (0.5–2 mM IPTG was used for
tac-driven gene expression in
E. coli, and up to 5 mM IPTG was used for
Zymomonas mobilis or
Pseudomonas putida) [
29]. The high concentration of 3HP demonstrated is an important step towards industrial application.
The most frequent approach for the genetic engineering of novel strains is plasmid insertion (of the required genes) into the host strain. However, as strains containing plasmids are not genetically stable and they require inducers and antibiotics to maintain the selection pressure during cultivation, Wang and Tian [
25] tried a different approach for construction of the host strain. More specifically, they constructed a plasmid-free
K. pneumoniae strain through chromosomal engineering by replacing the IS1 region in the chromosome with the AD DNA cassette containing the
aldH gene from
E. coli through homologous recombination. This strain was able to produce 3HP at 0.9 g/L (
Table 1) in flask cultures when glycerol, at 40 g/L, was added. Although the concentration of the 3HP produced was low, this work demonstrated a new approach for the construction of host strains without any need for inducers and antibiotics, which could be very useful for the development of new strains in the future.
Finally, a totally different approach to production of 3HP from glycerol was studied by Zhao et al. [
30]. In their process, they used a two-step approach, where glycerol was first converted to 1,3-propanediol by
K. pneumoniae, followed by conversion of 1,3-propanediol to 3HP by
Gluconobacter oxydans—a bacterium that incompletely oxidizes a wide range of ketones, organic acids, and aldehydes. The final concentration of 3HP was 60.5 g/L, and the conversion rate of glycerol to 3HP was 0.5 g/g. Moreover, it was the first time that acrylic acid production was reported as a by-product of 3HP production from 1,3-propanediol, at a concentration of approximately 1 g/L—another high added-value chemical with several applications, such as plastics, adhesives, and coatings.
2.4. Engineering of E. coli Cells for the Conversion of Glycerol to 3HP
E. coli is commonly used as a host microorganism for genetic modifications, and there is a wide range of commercial genetic tools available, making it easy to handle with efficient control of gene transfer. In addition, E. coli has a large number of alds in its genome—genes that could be studied and overexpressed for the conversion of glycerol to 3HP. However, the disadvantage of E. coli is that it is not able to produce coenzyme B12, leading to the necessity of external addition of this expensive compound, thus affecting process economics and industrial applications.
As when
K. pneumoniae is used as the host strain, when using
E. coli, one common strategy is to use the genes of the
dha operon. As has been discussed, glycerol dehydratase undergoes rapid inactivation, which can result in shutdown of 3HP production. Niu et al. [
31] demonstrated that when the glycerol dehydratase reactivase gene was cloned together with the glycerol dehydratase gene, the 3HP concentration increased fivefold, due to the prevention of glycerol dehydratase inactivation. In an attempt to increase the 3HP production yield, the authors also expressed the gene encoding the NAD
+-regenerating enzyme, glycerol-3-phosphate dehydrogenase (
Table 2). However, the concentration of 3HP was reduced, with increased production of malic acid, due to the lack of NAD
+. Enhancement of 3HP production by regulation of glycerol metabolism and minimizing of by-product formation was attempted by Jung et al. [
4]. During their work, they tried to eliminate the formation of major by-products, such as acetate and 1,3-propanediol, and to increase the metabolic flow of glycerol towards 3HP by upregulating the glycerol kinase (
glpK) and the glycerol facilitator (
glpF), and by deleting the regulatory factor that repressed the use of glycerol (
glpR). After these modifications, the generation of by-products was minimized, and the uptake of glycerol was improved, resulting in a production of 3HP as high as 42.1 g/L. The average yield was 0.268 g/g.
When aiming to produce 3HP through the 3-hydroxypropionaldehyde intermediate, proper balancing of the steps should be attempted, to avoid accumulation of the 3-hydroxypropionaldehyde. 3-Hydroxypropionaldehyde is very toxic for microorganisms, even at concentrations of 15–30 mM, and its accumulation can result in inhibition of 3HP production, with concentrations as low as 10 mM or even lower having a significant negative effect on growth and enzyme activity [
32,
33]. One solution to the problem could be the selection of an efficient aldehyde dehydrogenase that would rapidly act on 3-hydroxypropionaldehyde and convert it to 3HP. Based on this strategy, Chu et al. [
34] tried 17 candidate aldehyde dehydrogenases for their activity against 3-hydroxypropionaldehyde, with the
gabD4 from
Cupriavidus necator turning out to be the most effective one. In an attempt to further improve the aldehyde dehydrogenase selected, the authors performed site-directed and saturation mutagenesis, based on homologous modeling. The mutant enzyme obtained had 1.4-fold higher activity compared to the wild type one, and a high 3HP production of 71.9 g/L (with a productivity of 1.8 g/L·h) was achieved in fed-batch bioreactor culture, which, to the best of our knowledge, is the highest reported concentration reported with
E. coli as a host growing on glycerol.
Another strategy to avoid 3-hydroxypropionaldehyde accumulation is the use of promoters with different strength in controlling the expression of the enzymes [
3], although the number of genetic elements can limit the control over gene expression [
33]. The use of the in silico design tool “UTR Designer” has also been proposed; it can provide precise predictions of the translation initiation efficiency [
33]. This tool was employed by Lim et al. [
33] in their effort to prevent the accumulation of 3-hydroxypropionaldehyde by fine-tuning the expression levels of aldehyde dehydrogenase and glycerol dehydratase. Moreover, they deleted the by-product formation genes,
yghD and
ackA-pta, in order to improve metabolic flow towards the formation of 3HP. During flask culture, 3HP formation reached 17.9 g/L with a yield of 0.61 g/g. Following on from these results, the authors tried a fed-batch cultivation with the addition of glucose together with the glycerol. Under these conditions, the production of 3HP increased to 40.5 g/L with a yield of 0.97 g/g. This increase in the yield of 3HP formation could be explained by the better carbon flow of glycerol towards 3HP formation, as glucose could cover the needs of cell growth. A different approach for balancing the pathway enzymes to prevent 3-hydroxypropionaldehyde accumulation, and in turn, improve the 3HP production yields, was used by Sankaranarayanan et al. [
35]. In their work, they used a synthetic regulatory cassette comprised of varying-strength promoters and bicistronic ribosome-binding sites (RBSs) to control the expression of the genes. Fine-tuning of the levels of expression between the two genes could result in no secretion of 3-hydroxypropionaldehyde, which was achieved when aldehyde dehydrogenase had an expression that was 8-fold higher than that of glycerol dehydratase. This strategy resulted in the engineering of an
E. coli strain capable of producing up to 56.4 g/L 3HP in a fed-batch bioreactor, with the addition of glucose together with glycerol. The addition of glucose as a co-substrate was also found to improve the activity of the aldehyde dehydrogenase gene, and in turn, improve the 3HP production yields [
36]. Niu et al. [
36] reported a 3.5-fold increase in the activity of the enzyme, which improved the 3HP production in flasks from 3.39 g/L (control—no addition of glucose) to 6.80 g/L. Optimization of the glucose concentration and feeding strategy improved the 3HP production to up to 17.2 g/L during fed-batch cultivation. The authors also suggested that addition of glucose reduced the imbalance between the activities of glycerol dehydratase and aldehyde dehydrogenase, as it led to an increase in the activity of aldehyde dehydrogenase.
Not only the presence of 3-hydroxypropionaldehyde, but also high concentrations of 3HP can inhibit the growth of the host microorganisms, thus hindering the production of 3HP. Aiming to identify a 3HP-tolerant
E. coli strain, Sankaranarayanan et al. [
37] studied nine acid-tolerant strains that efficiently produced various organic acids at high titers. Construction of the 3HP producing strains was achieved by expressing the
dha operon, and an ald from
Azospirillum brasilense. Of all the strains tested, two showed a high degree of growth in the presence of 25 g/L 3HP, and one of them,
E. coli W, outperformed the rest, and produced 41.5 g/L 3HP with a yield of 0.31 g/g. During this work, it was found that there are significant differences in 3HP tolerance among
E. coli strains, and this should be taken into account when selecting an appropriate host microorganism.
To improve the metabolic flow of glycerol towards 3HP, other genes of glycerol catabolism are often deleted, which might result in strains with poor growth [
38]. To avoid this and at the same time improve the metabolic flow towards 3HP, Tsuruno et al. [
38] suggested the use of a metabolic toggle switch (MTS), and tested the conditional repression of the following genes:
glpK (encoding glycerol kinase),
tpiA (encoding triosephosphate isomerase), and
gapA (encoding glyceraldehyde-3-phosphate dehydrogenase). After testing the three different strains for their efficiency in 3HP production, it was found that only the strain with the MTS for
gapA improved the production of 3HP to 4.88 g/L. Deletion of the gene
yqhD (whose protein product is responsible for the conversion of 3-hydroxypropionaldehyde to 1,3-propanediol) further improved the concentration of 3HP to 6.06 g/L, with a yield of 0.515 mol/mol.
Finally, another strategy for genetically modifying
E. coli was proposed by Honjo et al. [
39], who used a dual synthetic pathway for the construction of
E. coli strains. More specifically, they transferred the genes from both the
dha and the
pdu operons for the construction of one strain, and compared with the results obtained using a strain with only the genes from the
dha operon. The strain with both pathways produced 3HP at 5.05 g/L, whereas the strain with only the
dha operon produced only 2.98 g/L 3HP. The yield was 0.54 mol/mol. When the strain only had the
pdu operon, it produced 1.41 g/L, thus underpinning the synergistic action of the two pathways in the conversion of glycerol to 3HP.
2.5. Other Microorganisms
It is clear that most of the research is focused mainly on either
E. coli or
K. pneumoniae as host microorganism for the construction of a cell factory capable of producing 3HP from glycerol. However, there are other microorganisms that could be suitable and efficient cell factories for efficient production of 3HP. For example,
Lactobacillus reuteri is a good candidate, due to its ability to naturally produce the coenzyme B
12 and to its high acid tolerance—both of which are required when producing 3HP. Moreover, the
pdu operon is endogenous in that specific species [
40], minimizing the need for gene transfer. The use of a recombinant
L. reuteri strain with a mutation in the catabolite repression element (CRE) was investigated by Dishisha et al. [
23], to improve the metabolic flux of glycerol to 3HP and 1,3-propanediol. During the flux analysis for the different steps of the
pdu operon, it was found that the glycerol dehydration to 3-hydroxypropionaldehyde was ten times faster than the subsequent oxidation and reduction of 3-hydroxypropionaldehyde to 1,3-propanediol and 3HP. Thus, establishment of an optimal feeding rate of glycerol was crucial to avoid 3-hydroxypropionaldehyde accumulation and direct the flux of glycerol towards 3HP and 1,3-propanediol formation. The final titers were 10.6 g/L 3HP and 9.0 g/L 1,3-propanediol under anaerobic conditions in the fed-batch bioreactor when resting cells of
L. reuteri were used (
Table 3).
The same recombinant strain of
L. reuteri was also used for the preparation of crosslinked, cryostructured monoliths that could be used as a biocatalyst for the conversion of glycerol to 3-hydroxypropionaldehyde, 3HP, and 1,3-propanediol [
14]. Different crosslinkers were tested for the preparation of monolith columns of resting
L. reuteri cells, with only the mixture of synthetic macromolecular structures of activated polyethyleneimine and modified polyvinyl alcohol (Cryo-PEI/PVA) demonstrating enhanced biocatalytic activity, mechanical stability, and sustained viability. Under optimal conditions, 3.3 g/L 3HP was produced during fed-batch feeding of the immobilized cells. Finally, a two-step process involving the cultivation of
L. reuteri and
G. oxydans has also been proposed [
41]. More specifically, in the first step, the anaerobic cultivation of
L. reuteri in fed-batch mode resulted in the production of equimolar quantities of 3HP and 1,3-propanediol, whereas in the second step, the 1,3-propanediol in the cell-free supernatant was selectively oxidized to 3HP by
G. oxydans under aerobic batch cultivation. At the end of the first step, 14 g/L 3HP and 12 g/L 1,3-propanediol were produced, with the corresponding conversion yields being 0.48 g/g and 0.42 g/g, respectively. Finally, the oxidation in the second step resulted in the quantitative conversion of 1,3-propanediol to 3HP, with a final 3HP concentration of 23.6 g/L and an overall conversion yield of glycerol to 3HP of approximately 1 mol/mol.
Another host strain that has been used is
Bacillus subtilis, which shows high growth rates, and is a non-pathogenic microorganism classified as GRAS (generally recognized as safe) [
42]. The first attempt to use this host microorganism for 3HP was described by Kalantari et al. [
42]. The constructed strain performed well in shaker flasks, producing up to 10 g/L with average yield 0.79 g/g, and showing good tolerance towards the 3HP produced. One drawback of the use of this strain as host is the lack of any native ability to produce the coenzyme B
12 (as with
E. coli); the authors suggested that this can be solved by transferring the necessary genes from
Bacillus megaterium. This work demonstrated that
Bacillus strains can also be used as cell factories for 3HP production, and more work using these cells will probably be conducted in the future.