Comprehensive Changes in Nutrient Constituents and Antioxidant Activity during Food Processing of Isoflavone-Enriched Soybean Leaf by Mycelia of Tricholoma matsutake
Abstract
1. Introduction
2. Materials and Methods
2.1. Experiment with Plants and Microorganisms
2.2. Reagents and Analytical Instruments
2.3. Preparation of IESL by Processing Stages
2.4. pH, Acidity, Brix, Reducing Sugars, and Soluble Proteins
2.5. Analysis of Fatty Acids
2.6. Analysis of Free Amino Acids
2.7. Analysis of Minerals
2.8. Analysis of Water-Soluble Vitamins
2.9. Metabolic Derivatization and Gas Chromatography-Mass Spectrometry Analysis
2.10. Processing of Data
2.11. Extraction and Analysis of Antioxidant Activities
2.11.1. Preparation of Sample Extracts for Analysis of Antioxidant Activities
2.11.2. Analysis of Antioxidant Activities
2.12. Statistical Analysis
3. Results and Discussion
3.1. Changes in pH, Acidity, Brix, Reducing Sugars, and Soluble Proteins
3.2. Changes in Fatty Acid Compositions
3.3. Changes in Free Amino Acid Compositions
3.4. Changes in Mineral Compositions
3.5. Changes in Water-Soluble Vitamin Compositions
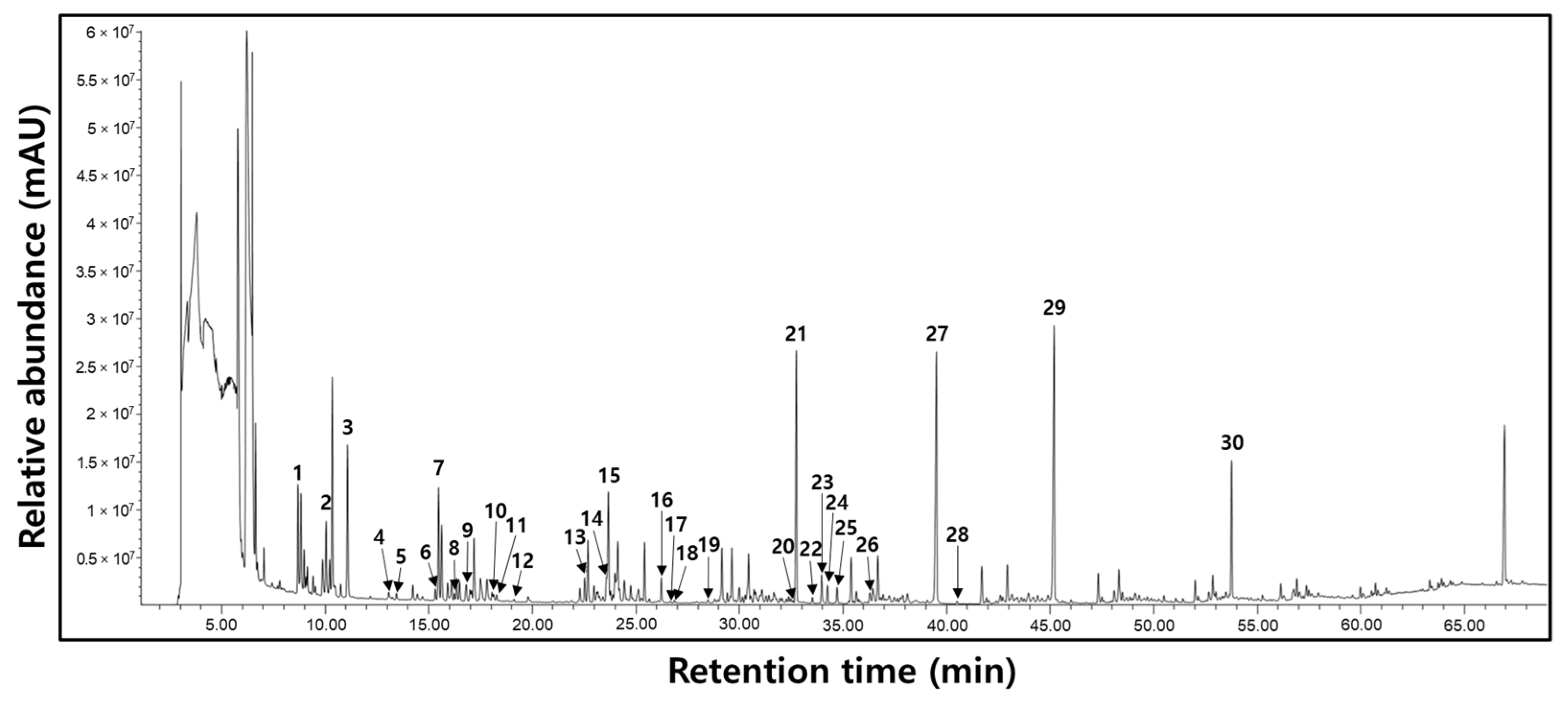
3.6. Changes in Metabolite Compositions
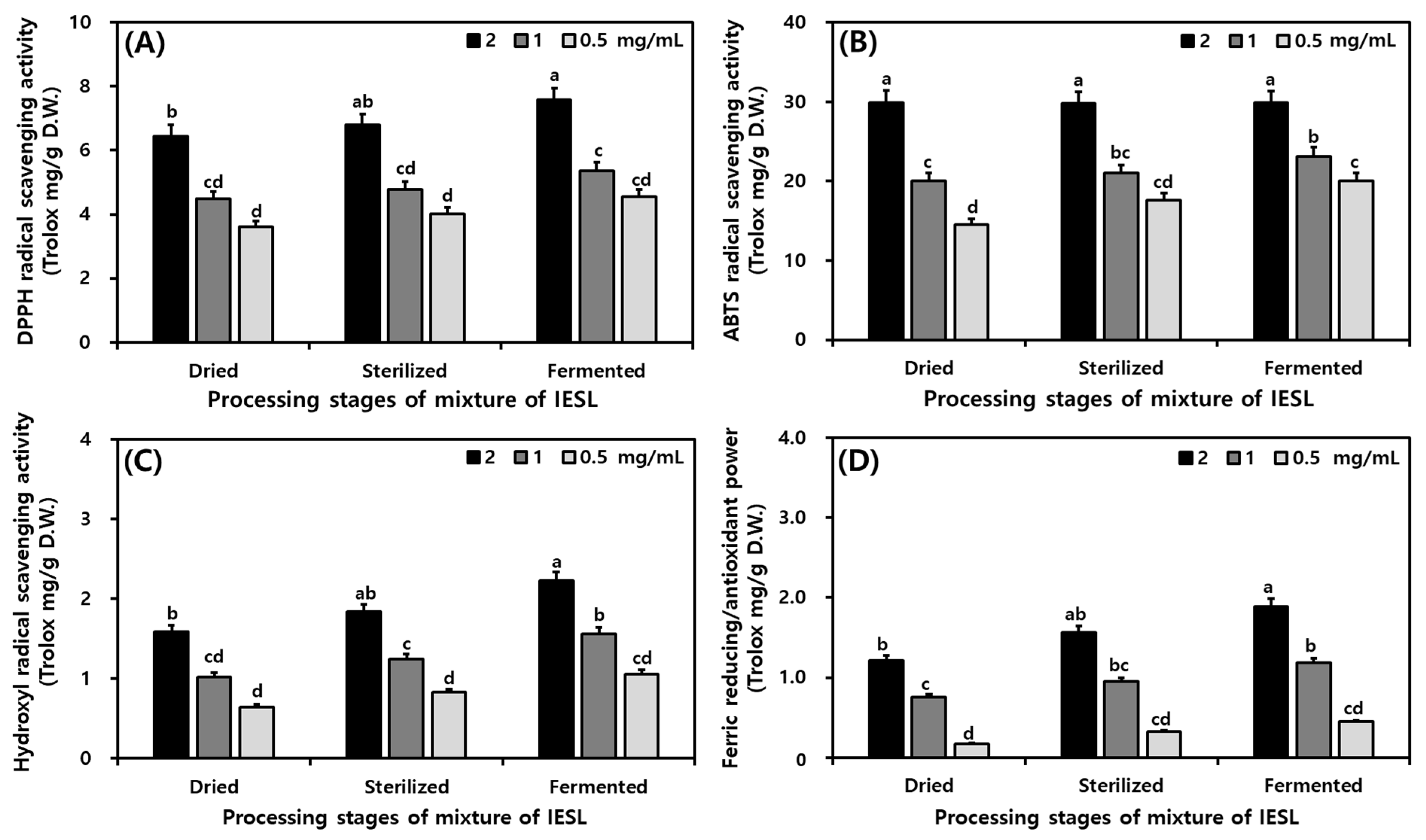
3.7. Changes in Antioxidant Activities
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ban, Y.J.; Song, Y.H.; Kim, J.Y.; Cha, J.Y.; Ali, I.; Baiseitova, A.; Shah, A.B.; Kim, W.Y.; Park, K.H. A significant change in free amino acids of soybean (Glycine max L. Merr) through Ethylene Application. Molecules 2021, 26, 1128. [Google Scholar] [CrossRef] [PubMed]
- Yuk, H.J.; Curtis-Long, M.J.; Ryu, H.W.; Jang, K.C.; Seo, W.D.; Kim, J.Y.; Kang, K.Y.; Park, K.H. Pterocarpan profiles for soybean leaves at different growth stages and investigation of their glycosidase inhibitions. J. Agric. Food Chem. 2011, 59, 12683–12690. [Google Scholar] [CrossRef] [PubMed]
- Yuk, H.J.; Lee, J.H.; Curtis-Long, M.J.; Lee, J.W.; Kim, Y.S.; Ryu, H.W.; Park, C.G.; Jeong, T.S.; Park, K.H. The most abundant polyphenol of soy leaves, coumestrol, displays potent α-glucosidase inhibitory activity. Food Chem. 2011, 126, 1057–1063. [Google Scholar] [CrossRef]
- Kim, J.E.; Jeon, S.M.; Park, K.H.; Lee, W.S.; Jeong, T.S.; McGregor, R.A.; Choi, M.S. Does Glycine max leaves or Garcinia Cambogia promote weight-loss or lower plasma cholesterol in overweight individuals: A randomized control trial. Nutr. J. 2011, 10, 94. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Ji, H.S.; Kang, J.H.; Shin, D.H.; Park, H.Y.; Choi, M.S.; Lee, C.H.; Lee, I.K.; Yun, B.S.; Jeong, T.S. Soy leaf extract containing kaempferol glycosides and pheophorbides improves glucose homeostasis by enhancing pancreatic β-Cell function and suppressing hepatic lipid accumulation in db/db mice. J. Agric. Food Chem. 2015, 63, 7198–7210. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Kang, J.H.; Han, J.M.; Cho, M.H.; Chung, Y.J.; Park, K.H.; Shin, D.H.; Park, H.Y.; Choi, M.S.; Jeong, T.S. Anti-obesity effects of soy leaf via regulation of adipogenic transcription factors and fat oxidation in diet-induced obese mice and 3T3-L1 adipocytes. J. Med. Food. 2015, 18, 899–908. [Google Scholar] [CrossRef]
- Yuk, H.J.; Song, Y.H.; Curtis-Long, M.J.; Kim, D.W.; Woo, S.G.; Lee, Y.B.; Uddin, Z.; Kim, C.Y.; Park, K.H. Ethylene induced a high accumulation of dietary isoflavones and expression of isoflavonoid biosynthetic genes in soybean (Glycine max) leaves. J. Agric. Food Chem. 2016, 64, 7315–7324. [Google Scholar] [CrossRef]
- Xie, C.L.; Kang, S.S.; Cho, K.M.; Park, K.H.; Lee, D.H. Isoflavone-enriched soybean (Glycine max) leaves prevents ovariectomy-induced obesity by enhancing fatty acid oxidation. J. Funct. Foods. 2018, 43, 165–172. [Google Scholar] [CrossRef]
- Xie, C.L.; Park, K.H.; Kang, S.S.; Cho, K.M.; Lee, D.H. Isoflavone-enriched soybean leaves attenuate ovariectomy-induced osteoporosis in rats by anti-inflammatory activity. J. Sci. Food Agric. 2020, 101, 1499–1506. [Google Scholar] [CrossRef]
- Jung, S.; Chae, S.W.; Shin, D.H. Fermented foods of Korea and their functionalities. Fermentation 2022, 8, 645. [Google Scholar] [CrossRef]
- Li, S.; Jin, Z.; Hu, D.; Yang, W.; Yan, Y.; Nie, X.; Lin, J.; Zhang, Q.; Gai, D.; Ji, Y.; et al. Effect of solid-state fermentation with Lactobacillus casei on the nutritional value, isoflavones, phenolic acids and antioxidant activity of whole soybean flour. LWT 2020, 125, 109264. [Google Scholar] [CrossRef]
- Alhomodi, A.F.; Zavadil, A.; Berhow, M.; Gibbons, W.R.; Karki, B. Application of cocultures of fungal mycelium during solid-state fermentation of canola meal for potential feed application. J. Am. Oil Chem. Soc. 2021, 98, 509–517. [Google Scholar] [CrossRef]
- You, L.; Gao, Q.; Feng, M.; Yang, B.; Ren, J.; Gu, L.; Cui, C.; Zhao, M. Structural haracterization of polysaccharides from Tricholoma matsutake and their antioxidant and antitumour activities. Food Chem. 2013, 138, 2242–2249. [Google Scholar] [CrossRef]
- Onuma, H.; Hara, K.; Sugita, K.; Kano, A.; Fukuta, Y.; Shirasaka, N. Purification and characterization of a glycoside hydrolase family 5 endoglucanase from Tricholoma matsutake grown on barley based solid-state medium. J. Biosci. Bioeng. 2019, 128, 669–676. [Google Scholar] [CrossRef]
- Kusuda, M.; Ueda, M.; Konishi, Y.; Yamanaka, K.; Terashita, T.; Miyatake, K. Effects of carbohydrate substrate on the vegetative mycelial growth of an ectomycorrhizal mushroom, Tricholoma matsutake, isolated from Quercus. Mycoscience 2007, 48, 358–364. [Google Scholar] [CrossRef]
- Kusuda, M.; Ueda, M.; Miyatake, K.; Terashita, T. Characterization of the carbohydrase productions of an ectomycorrhizal fungus, Tricholoma matsutake. Mycoscience 2008, 49, 291–297. [Google Scholar] [CrossRef]
- Xu, M.; Qu, Y.; Li, H.; Tang, S.; Chen, C.; Wang, Y.; Wang, H. Improved extraction yield, water solubility, and antioxidant activity of lentinan from Lentinula edodes via Bacillus subtilis natto fermentation. Fermentation 2023, 9, 333. [Google Scholar] [CrossRef]
- Lee, J.H.; Hwang, C.E.; Son, K.S.; Cho, K.M. Comparisons of nutritional constituents in soybeans during solid state fermentation times and screening for their glucosidase enzymes and antioxidant properties. Food Chem. 2019, 272, 362–371. [Google Scholar] [CrossRef]
- Lee, S.; Lee, S.; Singh, D.; Oh, J.Y.; Jeon, E.J.; Ryu, H.S.; Lee, D.W.; Kim, B.S.; Lee, C.H. Comparative evaluation of microbial diversity and metabolite profiles in doenjang, a fermented soybean paste, during the two different industrial manufacturing processes. Food Chem. 2017, 221, 1578–1586. [Google Scholar] [CrossRef]
- Lee, H.Y.; Cho, D.Y.; Jang, K.J.; Lee, J.H.; Jung, J.G.; Kim, M.J.; Jeong, J.B.; Haque, M.A.; Cho, K.M. Changes of γ-aminobutyric acid, phytoestrogens, and biofunctional properties of the isoflavone-enriched soybean (Glycine max) leaves during solid lactic acid fermentation. Fermentation 2022, 8, 525. [Google Scholar] [CrossRef]
- Hwang, C.E.; Kim, S.C.; Kim, D.H.; Lee, H.Y.; Suh, H.K.; Cho, K.M.; Lee, J.H. Enhancement of isoflavone aglycone, amino acid, and CLA contents in fermented soybean yogurts using different strains: Screening of antioxidant and digestive enzyme inhibition properties. Food Chem. 2021, 340, 128199. [Google Scholar] [CrossRef]
- Zhang, Y.Y.; Stockmann, R.; Ng, K.; Ajlouni, S. Bioprocessing of pea protein can enhance fortified Fe but reduce Zn in vitro bioaccessibility. J. Agric. Food Chem. 2022, 70, 1241–1251. [Google Scholar] [CrossRef]
- Datta, S.; Sinha, B.K.; Bhattacharjee, S.; Seal, T. Nutritional composition, mineral content, antioxidant activity and quantitative estimation of water soluble vitamins and phenolics by RP-HPLC in some lesser used wild edible plants. Heliyon 2019, 5, 14–31. [Google Scholar] [CrossRef]
- Muzdalifah, D.; Athaillah, Z.A.; Nugrahani, W.; Devi, A.F. Colour and pH changes of tempe during extended fermentation. AIP Conf. Proc. 2017, 1803, 020036. [Google Scholar]
- Lee, S.Y.; Lee, S.; Lee, S.; Oh, J.Y.; Jeon, E.J.; Ryu, S.H.; Lee, C.H. Primary and secondary metabolite profiling of doenjang, a fermented soybean paste during industrial processing. Food Chem. 2014, 165, 157–166. [Google Scholar] [CrossRef]
- Sharma, R.; Garg, P.; Kumar, P.; Bhatia, S.K.; Kulshrestha, S. Microbial fermentation and its role in quality improvement of fermented foods. Fermentation 2020, 6, 106. [Google Scholar] [CrossRef]
- Ruiz-Teran, F.; Owens, J.D. Chemical and enzymic changes during the fermentation of bacteria-free soya bean tempe. J. Sci. Food Agric. 1996, 71, 523–530. [Google Scholar] [CrossRef]
- Park, M.K.; Cho, I.H.; Lee, S.; Choi, H.K.; Kwon, D.Y.; Kim, Y.S. Metabolite profiling of Cheonggukjang, a fermented soybean paste, during fermentation by gas chromatography-mass spectrometry and principal component analysis. Food Chem. 2010, 122, 1313–1319. [Google Scholar] [CrossRef]
- Shi, H.; Yang, E.; Li, Y.; Chen, X.; Zhang, J. Effect of solid-state fermentation on nutritional quality of leaf flour of the drumstick tree (Moringa oleifera Lam.). Front. Bioeng. Biotechnol. 2021, 9, 626628. [Google Scholar] [CrossRef]
- Wang, J.; Jiang, O.; Huang, Z.; Wang, Y.; Roubik, H.; Yang, K.; Cai, M.; Peilong Sun, P. Solid-state fermentation of soybean meal with edible mushroom mycelium to improve its nutritional, antioxidant capacities and physicochemical properties. Fermentation 2023, 9, 322. [Google Scholar] [CrossRef]
- Ali, M.W.; Ilays, M.Z.; Saeed, M.T.; Shin, D.H. Comparative assessment regarding antioxidative and nutrition potential of Moringa oleifera leaves by bacterial fermentation. J. Food Sci. Technol. 2019, 57, 1110–1118. [Google Scholar] [CrossRef] [PubMed]
- Samtiya, M.; Aluko, R.E.; Puniya, A.K.; Dhawa, T. Enhancing micronutrients bioavailability through fermentation of plant-based foods: A concise review. Fermentation 2021, 7, 63. [Google Scholar] [CrossRef]
- Cai, J.S.; Feng, J.Y.; Ni, Z.J.; Ma, R.H.; Thakur, K.; Wang, S.; Hu, F.; Zhang, J.G.; Wei, Z.J. An update on the nutritional, functional, sensory characteristics of soy products, and applications of new processing strategies. Trends Food Sci. Technol. 2021, 112, 676–689. [Google Scholar] [CrossRef]
- Olanipekun, B.; Adelakun, O. Nutritional and microbiological attributes of soybean (Glycine max) during fermentation with Rhizopus oligosporus. Food Sci. Qual. Manag. 2015, 39, 111–118. [Google Scholar]
- Yaman, M.; Çatak, J.; Uğur, H.; Gürbüz, M.; Belli, İ.; Tanyıldız, S.N.; Yıldırım, H.; Cengiz, S.; Yavuz, B.B.; Kişmiroğlu, C.; et al. The bioaccessibility of water-soluble vitamins: A review. Trends Food Sci. Technol. 2021, 09, 552–563. [Google Scholar] [CrossRef]
- Zhu, Y.Y.; Thakur, K.; Feng, J.Y.; Cai, J.S.; Zhang, J.G.; Hu, F.; Russo, P.; Spano, G.; Wei, Z.J. Riboflavin-overproducing lactobacilli for the enrichment of fermented soymilk: Insights into improved nutritional and functional attributes. Appl. Microbiol. Biotechnol. 2020, 104, 5759–5772. [Google Scholar] [CrossRef]
- Jaworska, G.; Pogon, K.; Bernas, E.; Duda-Chodak, A. Nutraceuticals and antioxidant activity of prepared for consumption commercial mushrooms Agaricus bisporus and Pleurotus ostreatus. J. Food Qual. 2015, 38, 111–122. [Google Scholar] [CrossRef]
- Jaworska, G.; Pogoń, K.; Bernaś, E.; Skrzypczak, A.; Kapusta, I. Vitamins, phenolics and antioxidant activity of culinary prepared Suillus luteus (L.) Roussel mushroom. LWT 2014, 59, 701–706. [Google Scholar] [CrossRef]
- Razali, S.; Khan, A.Y.F.; Khatib, A.; Ahmed, Q.U.; Wahab, R.A.; Zakaria, Z.A. An in vitro anticancer activity evaluation of Neolamarckia cadamba (Roxb.) bosser leaves’ extract and its metabolite profile. Front. Pharmacol. 2021, 12, 741683. [Google Scholar] [CrossRef]
- Dumschott, K.; Richter, A.; Loescher, W.; Merchant, A. Post photosynthetic carbon partitioning to sugar alcohols and consequences for plant growth. Phytochemistry 2017, 144, 243–252. [Google Scholar] [CrossRef]
- Gupta, R.K.; Gangoliya, S.S.; Singh, N.K. Reduction of phytic acid and enhancement of bioavailable micronutrients in food grains. J. Food Sci. Technol. 2015, 52, 676–684. [Google Scholar] [CrossRef]
- Stratil, P.; Klejdus, B.; Kubáň, V. Determination of phenolic compounds and their antioxidant activity in fruits and cereals. Talanta 2007, 71, 1741–1751. [Google Scholar] [CrossRef]
- Floegel, A.; Kim, D.O.; Chung, S.J.; Koo, S.I.; Chun, O.K. Comparison of ABTS/DPPH assays to measure antioxidant capacity in popular antioxidant-rich US foods. J. Food Compost. Anal. 2011, 24, 1043–1048. [Google Scholar] [CrossRef]
- Bai, Y.; Xu, Y.; Wang, B.; Li, S.; Guo, F.; Hua, H.; Zhao, Y.; Yu, Z. Comparison of phenolic compounds, antioxidant and antidiabetic activities between selected edible beans and their different growth periods leaves. J. Funct. Foods 2017, 35, 694–702. [Google Scholar] [CrossRef]
- Benzie, I.F.F.; Strain, J.J. The Ferric Reducing Ability of Plasma (FRAP) as a Measure of “Antioxidant Power”: The FRAP Assay. Anal. Biochem. 1996, 239, 70–76. [Google Scholar] [CrossRef]

| Index 1 | Processing Stages 2 | ||
|---|---|---|---|
| Dried IESL | Sterilized IESL | Fermented IESL | |
| pH | 5.76 ± 0.09 b | 5.83 ± 0.09 b | 6.33 ± 0.03 a |
| Acidity (%, as lactic acid) | 0.09 ± 0.00 a | 0.08 ± 0.00 ab | 0.07 ± 0.01 b |
| Brix (%) | 18.5 ± 0.03 a | 18.5 ± 0.02 a | 18.6 ± 0.03 a |
| Reducing sugar (mg/mL) | 0.8 ± 0.02 c | 1.3 ± 0.03 b | 1.5 ± 0.04 a |
| Soluble protein (mg/mL) | 2.26 ± 0.06 a | 2.17 ± 0.03 b | 2.09 ± 0.02 b |
| Contents 1 (mg/100 g) | Processing Stages 2 | ||
|---|---|---|---|
| Dried IESL | Sterilized IESL | Fermented IESL | |
| Saturated fatty acids | |||
| Lauric acid (C12:0) | 2.9 ± 0.02 b | 4.4 ± 0.02 a | nd 3 |
| Palmitic acid (C16:0) | 176.5 ± 1.83 b | 177.5 ± 2.88 b | 303.6 ± 2.18 a |
| Stearic acid (C18:0) | 69.7 ± 0.49 b | 65.2 ± 0.26 c | 98.1 ± 0.91 a |
| Arachidic acid (C20:0) | 6.2 ± 0.01 b | 6.6 ± 0.03 b | 10.5 ± 0.03 a |
| Behenic acid (C22:0) | 6.2 ± 0.01 b | 6.8 ± 0.04 b | 14.0 ± 0.07 a |
| Lignoceric acid (C24:0) | 7.7 ± 0.09 b | 8.3 ± 0.04 b | 18.7 ± 0.09 a |
| Total | 269.2 | 268.8 | 444.9 |
| Unsaturated fatty acids | |||
| Palmitoleic acid (C16:1) | 9.6 ± 0.08 b | 10.1 ± 0.10 b | 13.5 ± 0.08 a |
| Oleic acid (C18:1c) | 29.1 ± 0.46 b | 32.0 ± 0.60 b | 77.7 ± 0.89 a |
| Linoleic acid (C18:2c) | 56.2 ± 0.81 b | 56.7 ± 0.84 b | 274.9 ± 1.75 a |
| α-Linolenic acid (C18:3n3) | 46.9 ± 0.35 b | 47.2 ± 0.36 b | 87.8 ± 0.39 a |
| Ecosadienoic acid (C20:2) | nd | 2.5 ± 0.13 a | 2.3 ± 0.12 b |
| Arachidonic acid (C20:4n6) | 2.8 ± 0.04 b | 3.0 ± 0.05 b | 6.1 ± 0.01 a |
| Nervonic acid (C24:1n9) | nd | nd | 2.2 ± 0.01 a |
| Total | 144.6 | 151.5 | 464.5 |
| Total fatty acids | 413.8 | 420.3 | 909.4 |
| Contents 1 (mg/100 g) | Processing Stages 2 | ||
|---|---|---|---|
| Dried IESL | Sterilized IESL | Fermented IESL | |
| Non-essential amino acids | |||
| Proline | 157.34 ± 2.90 a | 135.45 ± 1.17 b | 24.10 ± 0.11 c |
| Hydroxyproline | 1.07 ± 0.01 b | 2.58 ± 0.03 ab | 3.24 ± 0.06 a |
| Aspartic acid | 176.84 ± 2.66 a | 161.28 ± 3.11 b | 71.76 ± 0.69 c |
| Serine | 96.82 ± 2.84 a | 80.27 ± 1.81 b | 54.61 ± 0.43 c |
| Aspartic acid—NH2 | 653.12 ± 11.06 a | 529.22 ± 6.16 b | 50.05 ± 0.51 c |
| Glutamic acid | 121.53 ± 0.08 b | 65.57 ± 1.28 c | 211.65 ± 2.58 a |
| Sarcosine | nd 3 | nd | 23.13 ± 1.16 a |
| Aminoadipic acid | 30.60 ± 0.93 b | 27.52 ± 0.78 b | 67.54 ± 0.18 a |
| Glycine | 17.28 ± 0.18 b | 16.88 ± 0.48 b | 36.34 ± 0.28 a |
| Alanine | 87.71 ± 0.99 a | 71.11 ± 0.96 c | 77.63 ± 0.98 b |
| Citrulline | 7.57 ± 0.08 b | 4.30 ± 0.02 c | 11.88 ± 0.05 a |
| α-aminobutyric acid | 8.56 ± 0.14 a | 6.76 ± 0.13 b | 3.11 ± 0.02 c |
| Cystine | 19.58 ± 0.78 b | 6.31 ± 0.12 c | 30.06 ± 1.05 a |
| Cystathionine | nd | nd | 12.07 ± 0.06 a |
| Tyrosine | 51.50 ± 1.85 b | 31.91 ± 0.76 c | 67.83 ± 1.93 a |
| β-alanine | 26.95 ± 0.25 a | 18.06 ± 0.09 c | 22.79 ± 0.94 b |
| β-aminoisobutyric acid | 37.73 ± 0.90 b | 11.62 ± 0.28 c | 54.74 ± 1.84 a |
| γ-aminobutyric acid | 134.66 ± 2.30 a | 121.61 ± 1.80 b | 25.93 ± 0.40 c |
| Aminoethanol | 17.04 ± 0.15 b | 11.56 ± 0.18 c | 24.31 ± 0.22 a |
| Hydroxylysine | nd | 3.60 ± 0.02 a | nd |
| Ornithine | 4.29 ± 0.01 b | 3.82 ± 0.01 c | 24.92 ± 0.05 a |
| Anserine | 4.79 ± 0.09 b | 2.47 ± 0.08 c | 17.73 ± 0.12 a |
| Arginine | 183.57 ± 2.38 a | 135.14 ± 0.88 b | 57.60 ± 0.67 c |
| Total | 1838.55 | 1447.04 | 973.02 |
| Essential amino acids | |||
| Threonine | 41.90 ± 0.21 b | 32.49 ± 0.26 c | 48.73 ± 1.01 a |
| Valine | 128.83 ± 2.11 a | 106.32 ± 3.21 b | 58.35 ± 1.09 c |
| Methionine | 10.54 ± 0.12 b | 3.79 ± 0.03 c | 25.27 ± 0.16 a |
| Isoleucine | 72.74 ± 1.41 a | 61.22 ± 2.60 b | 57.47 ± 0.08 c |
| Leucine | 71.95 ± 0.42 b | 50.39 ± 0.25 c | 80.95 ± 1.50 a |
| Phenylalanine | 145.76 ± 2.29 a | 106.66 ± 3.33 b | 89.30 ± 1.47 c |
| Lysine | 48.54 ± 0.43 b | 30.50 ± 0.53 c | 82.02 ± 0.10 a |
| Histidine | 30.90 ± 0.55 a | 22.49 ± 0.12 b | 19.14 ± 0.26 c |
| Total | 551.16 | 413.86 | 461.23 |
| Total amino acids | 2389.71 | 1860.90 | 1434.25 |
| Ammonia | 19.13 | 23.20 | 36.82 |
| Contents 1 (mg/100 g) | Processing Stages 2 | ||
|---|---|---|---|
| Dried IESL | Sterilized IESL | Fermented IESL | |
| Phosphorus (P) | 2.92 ± 0.02 b | 3.02 ± 0.03 b | 4.33 ± 0.04 a |
| Sulfur (S) | 2.42 ± 0.03 c | 2.78 ± 0.04 b | 3.87 ± 0.03 a |
| Potassium (K) | 19.96 ± 0.40 b | 20.23 ± 0.61 b | 26.44 ± 0.28 a |
| Calcium (Ca) | 11.35 ± 0.17 c | 11.73 ± 0.29 b | 15.45 ± 0.37 a |
| Magnesium (Mg) | 2.58 ± 0.13 b | 2.57 ± 0.13 b | 3.36 ± 0.17 a |
| Iron (Fe) | 0.18 ± 0.00 b | 0.19 ± 0.01 b | 0.23 ± 0.01 a |
| Zinc (Zn) | 0.07 ± 0.01 c | 0.08 ± 0.00 b | 0.10 ± 0.00 a |
| Manganese (Mn) | 0.11 ± 0.01 b | 0.11 ± 0.00 b | 0.15 ± 0.00 a |
| Aluminium (Al) | 0.23 ± 0.00 b | 0.24 ± 0.00 b | 0.30 ± 0.00 a |
| Boron (B) | 0.09 ± 0.00 b | 0.10 ± 0.01 b | 0.13 ± 0.00 a |
| Silicon (Si) | 0.07 ± 0.00 c | 0.26 ± 0.01 b | 0.39 ± 0.01 a |
| Sodium (Na) | 0.32 ± 0.02 c | 0.41 ± 0.02 b | 0.57 ± 0.01 a |
| Total | 40.30 | 41.72 | 55.32 |
| Contents 1 (mg/100 g) | Processing Stages 2 | ||
|---|---|---|---|
| Dried IESL | Sterilized IESL | Fermented IESL | |
| B2 (Riboflavin) | 0.4 ± 0.01 c | 1.3 ± 0.05 b | 10.8 ± 0.32 a |
| B3 (Niacin) | 8.0 ± 0.29 c | 11.1 ± 0.45 b | 21.4 ± 0.85 a |
| B5 (Pantothenic acid) | nd 3 | nd | nd |
| B9 (Folic acid) | nd | nd | nd |
| C (Ascorbic acid) | nd | nd | nd |
| Total | 8.4 | 12.4 | 32.2 |
| Contents 1 (μg/g) | Processing Stages 2 | ||
|---|---|---|---|
| Dried IESL | Sterilized IESL | Fermented IESL | |
| Amino acids | |||
| Alanine | 10.68 ± 0.56 a | 7.00 ± 0.65 b | 4.97 ± 0.23 c |
| Valine | 11.35 ± 1.09 a | 5.42 ± 0.21 b | 5.42 ± 0.18 b |
| Leucine | 5.34 ± 0.49 a | 2.91 ± 0.11 b | 2.91 ± 0.11 b |
| Isoleucine | 5.33 ± 0.33 a | 2.57 ± 0.10 b | 2.57 ± 0.10 b |
| Proline | 14.02 ± 0.68 a | 6.10 ± 0.24 b | 6.12 ± 0.26 b |
| Serine | 3.70 ± 0.34 a | 1.74 ± 0.14 c | 1.79 ± 0.11 b |
| Threonine | 2.12 ± 0.04 a | 1.29 ± 0.06 b | 1.12 ± 0.06 c |
| Aspartic acid | 1.45 ± 0.05 a | 0.76 ± 0.04 b | 0.44 ± 0.04 c |
| Pyroglutamic acid | 2.53 ± 0.16 a | 1.61 ± 0.10 b | 1.60 ± 0.08 b |
| Phenylalanine | 3.57 ± 0.16 a | 1.48 ± 0.07 b | 1.45 ± 0.12 c |
| Asparagine | 4.52 ± 0.35 a | 1.10 ± 0.05 b | 1.09 ± 0.06 b |
| Total | 64.61 | 31.98 | 29.48 |
| Organic acids | |||
| Lactic acid | 6.67 ± 0.55 a | 6.28 ± 0.04 b | 5.96 ± 0.09 c |
| Oxalic acid | 99.21 ± 7.19 c | 108.10 ± 7.01 b | 119.58 ± 7.50 a |
| Malonic acid | 10.18 ± 0.52 a | 9.05 ± 0.53 b | 3.58 ± 0.14 c |
| Succinic acid | 3.87 ± 0.14 a | 2.80 ± 0.11 b | 2.20 ± 0.04 c |
| Fumaric acid | 1.70 ± 0.13 b | 2.07 ± 0.03 b | 1.01 ± 0.09 c |
| Malic acid | 12.78 ± 0.07 a | 8.76 ± 0.51 b | 8.67 ± 0.53 b |
| Cyanuric acid | 6.10 ± 0.14 c | 6.38 ± 0.56 b | 6.73 ± 0.56 a |
| Glutamic acid | 1.04 ± 0.06 a | 0.37 ± 0.00 c | 0.39 ± 0.01 b |
| Citric acid | 4.24 ± 0.18 a | 2.74 ± 0.20 c | 2.89 ± 0.17 b |
| Total | 145.79 | 146.55 | 151.01 |
| Carbohydrates | |||
| Pinitol | 118.60 ± 7.12 a | 67.97 ± 4.13 b | 67.57 ± 4.59 c |
| Fructose | 11.42 ± 0.74 c | 12.98 ± 0.96 a | 12.81 ± 0.90 b |
| Glucose | 2.76 ± 0.24 c | 8.19 ± 0.51 a | 8.09 ± 0.66 b |
| Myo-inositol | 1.80 ± 0.04 a | 1.36 ± 0.13 b | 1.35 ± 0.12 b |
| Sucrose | 18.23 ± 0.53 a | 0.73 ± 0.00 b | 0.69 ± 0.06 c |
| Total | 152.81 | 91.23 | 90.51 |
| Fatty acids | |||
| Myristic acid | 1.21 ± 0.05 a | 1.11 ± 0.09 b | 1.05 ± 0.10 b |
| Palmitic acid | 66.30 ± 1.02 c | 75.17 ± 6.89 a | 74.39 ± 4.12 b |
| Stearic acid | 78.99 ± 5.57 b | 80.26 ± 7.52 a | 77.97 ± 3.65 b |
| Total | 146.5 | 156.54 | 153.41 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cho, D.-Y.; Lee, H.-Y.; Jeong, J.-B.; Lee, J.-H.; Lee, G.-Y.; Jang, M.-Y.; Lee, J.-H.; Lee, J.-H.; Haque, M.A.; Cho, K.-M. Comprehensive Changes in Nutrient Constituents and Antioxidant Activity during Food Processing of Isoflavone-Enriched Soybean Leaf by Mycelia of Tricholoma matsutake. Fermentation 2023, 9, 677. https://doi.org/10.3390/fermentation9070677
Cho D-Y, Lee H-Y, Jeong J-B, Lee J-H, Lee G-Y, Jang M-Y, Lee J-H, Lee J-H, Haque MA, Cho K-M. Comprehensive Changes in Nutrient Constituents and Antioxidant Activity during Food Processing of Isoflavone-Enriched Soybean Leaf by Mycelia of Tricholoma matsutake. Fermentation. 2023; 9(7):677. https://doi.org/10.3390/fermentation9070677
Chicago/Turabian StyleCho, Du-Yong, Hee-Yul Lee, Jong-Bin Jeong, Ji-Ho Lee, Ga-Young Lee, Mu-Yeon Jang, Jin-Hwan Lee, Ji-Hyun Lee, Md. Azizul Haque, and Kye-Man Cho. 2023. "Comprehensive Changes in Nutrient Constituents and Antioxidant Activity during Food Processing of Isoflavone-Enriched Soybean Leaf by Mycelia of Tricholoma matsutake" Fermentation 9, no. 7: 677. https://doi.org/10.3390/fermentation9070677
APA StyleCho, D.-Y., Lee, H.-Y., Jeong, J.-B., Lee, J.-H., Lee, G.-Y., Jang, M.-Y., Lee, J.-H., Lee, J.-H., Haque, M. A., & Cho, K.-M. (2023). Comprehensive Changes in Nutrient Constituents and Antioxidant Activity during Food Processing of Isoflavone-Enriched Soybean Leaf by Mycelia of Tricholoma matsutake. Fermentation, 9(7), 677. https://doi.org/10.3390/fermentation9070677







