The Effect of Co-Doping on the Structural and Magnetic Properties of Single-Domain Crystalline Copper Ferrite Nanoparticles
Abstract
:1. Introduction
2. Materials and Methods
3. Results and Discussion
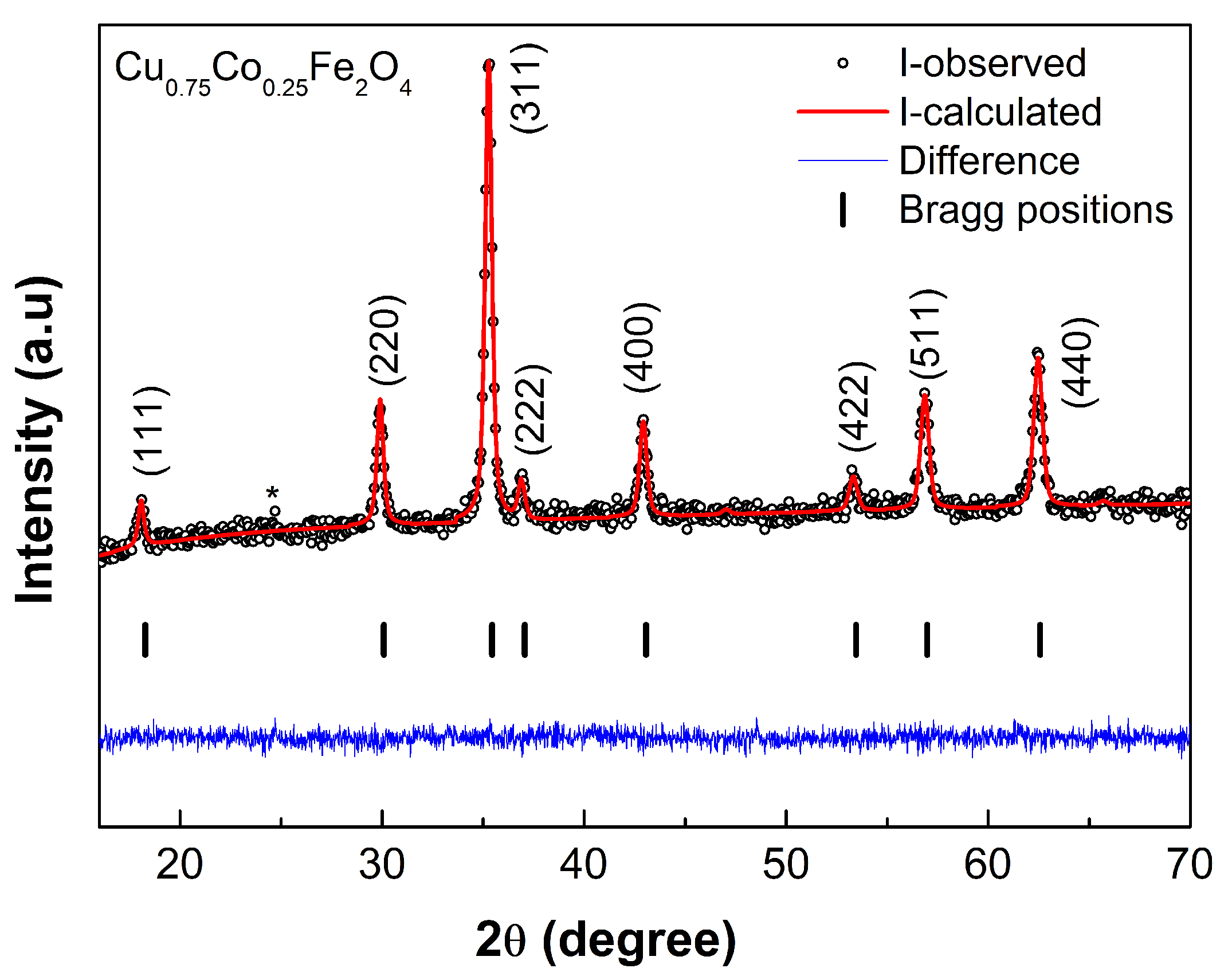
3.1. XRD Analysis
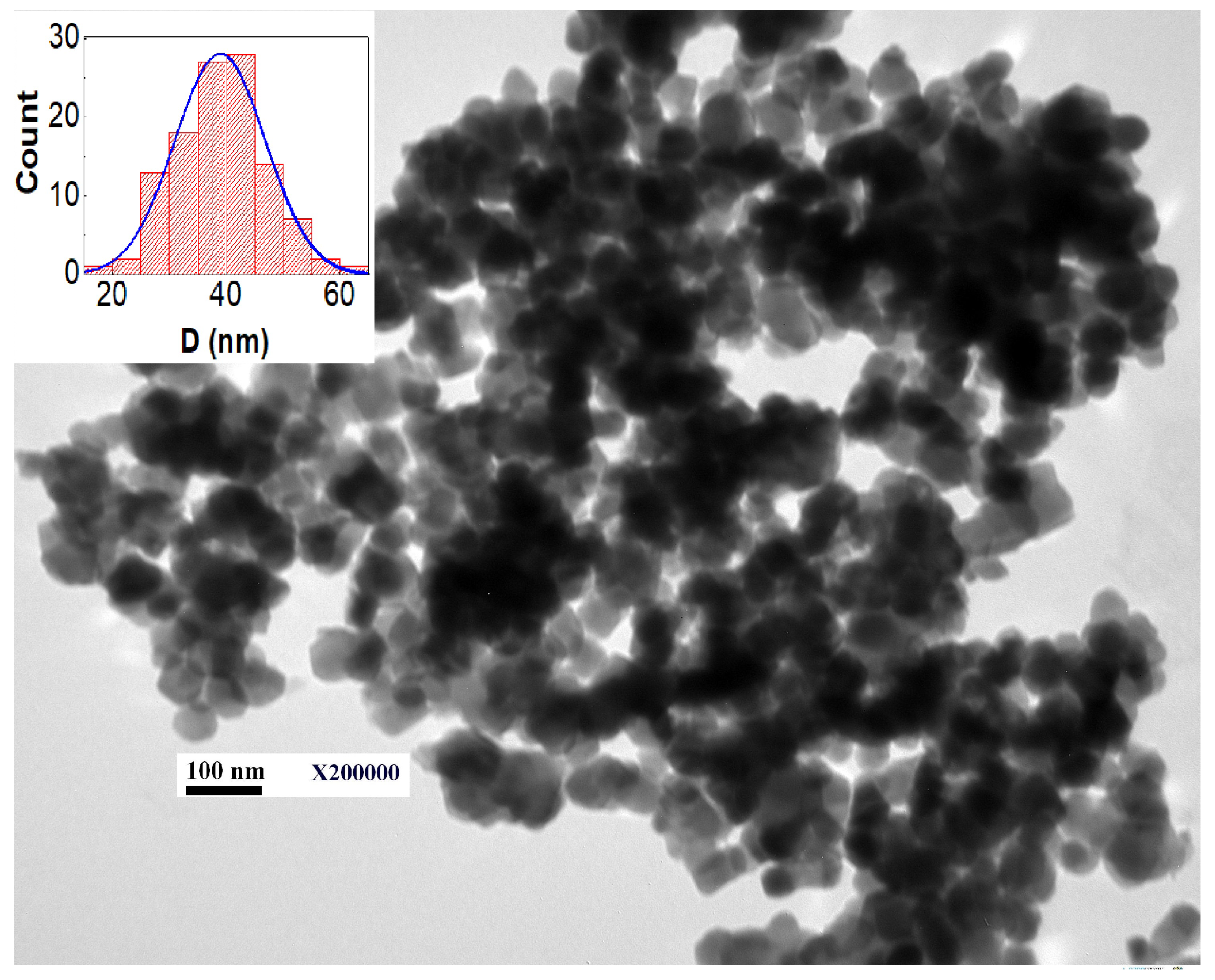
3.2. TEM Analysis
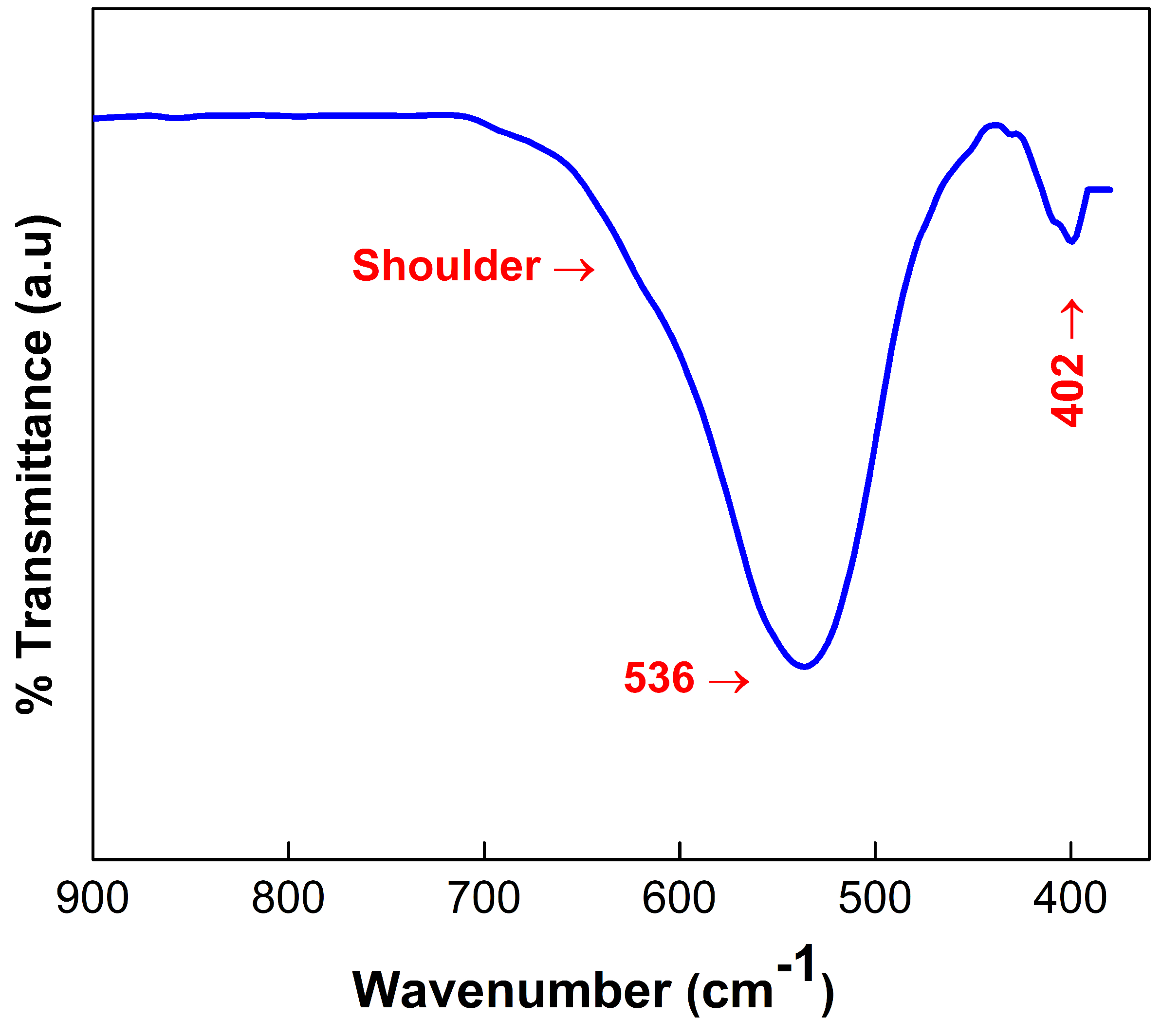
3.3. FT-IR Analysis
3.4. Magnetic Properties
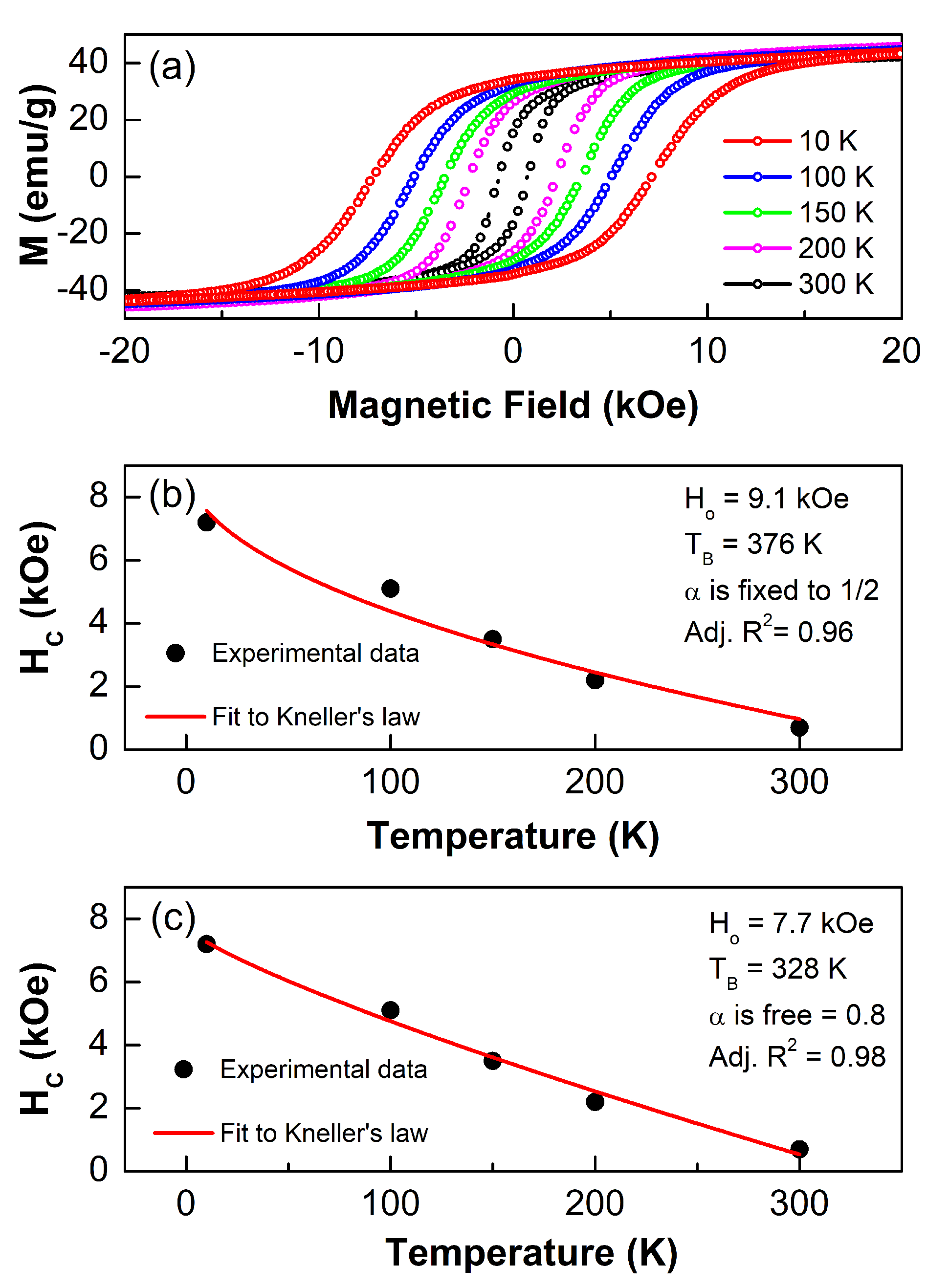
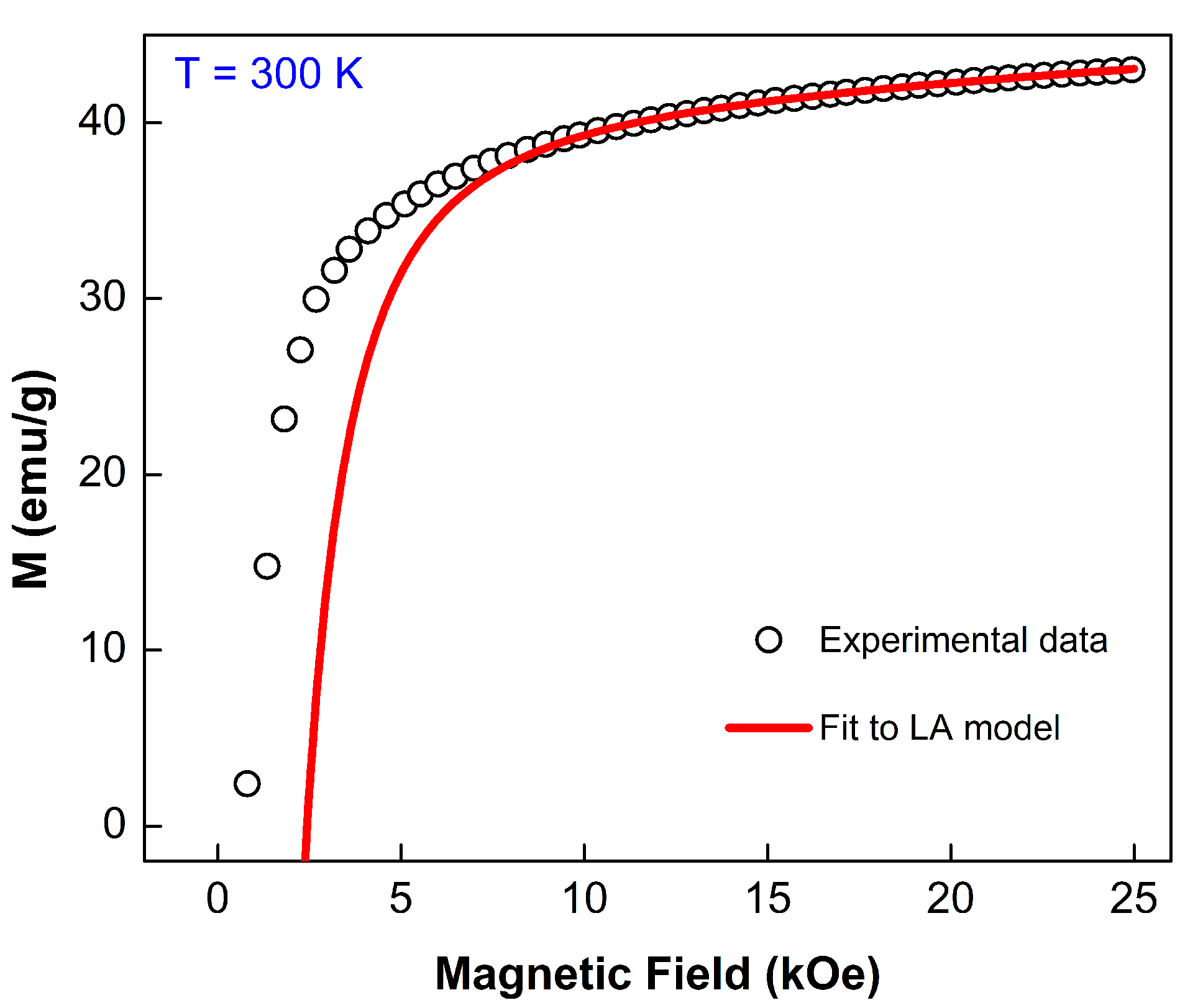
3.4.1. M vs. H Curves Analysis
3.4.2. Law of Approach to Saturation
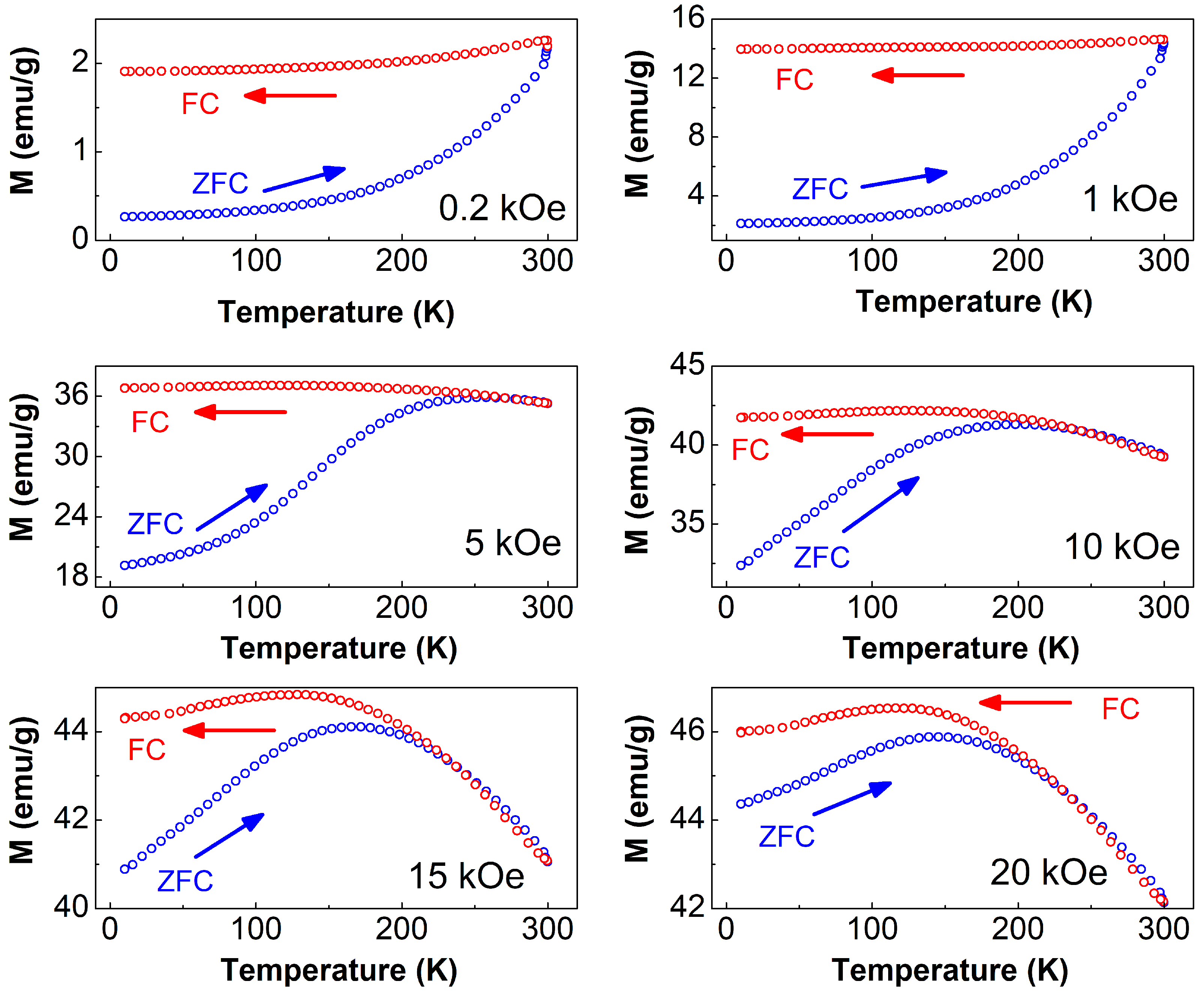
3.4.3. M vs. T Analysis
4. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Pirhashemi, M.; Habibi-Yangjeh, A.; Pouran, S.R. Review on the criteria anticipated for the fabrication of highly efficient ZnO-based visible-light-driven photocatalysts. J. Ind. Eng. Chem. 2018, 62, 1–25. [Google Scholar] [CrossRef]
- Masunga, N.; Mmelesi, O.K.; Kefeni, K.K.; Mamba, B.B. Recent advances in copper ferrite nanoparticles and nanocomposites synthesis, magnetic properties and application in water treatment. J. Environ. Chem. Eng. 2019, 7, 103179. [Google Scholar] [CrossRef]
- Wang, J.; Deng, Q.; Li, M.; Jiang, K.; Zhang, J.; Hu, Z.; Chu, J. Copper ferrites@ reduced graphene oxide anode materials for advanced lithium storage applications. Sci. Rep. 2017, 7, 8903. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ponhan, W.; Maensiri, S. Fabrication and magnetic properties of electrospun copper ferrite (CuFe2O4) nanofibers. Solid State Sci. 2009, 11, 479–484. [Google Scholar] [CrossRef]
- Nikolić, V.N.; Vasić, M.; Milić, M.M. Observation of low-and high-temperature CuFe2O4 phase at 1100 °C: The influence of Fe3+ ions on CuFe2O4 structural transformation. Ceram. Int. 2018, 44, 21145–21152. [Google Scholar] [CrossRef]
- Desai, M.; Prasad, S.; Venkataramani, N.; Samajdar, I.; Nigam, A.; Krishnan, R. Annealing induced structural change in sputter deposited copper ferrite thin films and its impact on magnetic properties. J. Appl. Phys. 2002, 91, 2220–2227. [Google Scholar] [CrossRef] [Green Version]
- Hammad, T.M.; Kuhn, S.; Amsha, A.A.; Hempelmann, R. Investigation of structural, optical, and magnetic properties of Co2+ ions substituted CuFe2O4 spinel ferrite nanoparticles prepared via precipitation approach. J. Aust. Ceram. Soc. 2021, 57, 543–553. [Google Scholar] [CrossRef]
- Alzoubi, G.M. Observation of Spin-Glass-like Behavior over a Wide Temperature Range in Single-Domain Nickel-Substituted Cobalt Ferrite Nanoparticles. Nanomaterials 2022, 12, 1113. [Google Scholar] [CrossRef]
- Hashim, M.; Shirsath, S.E.; Meena, S.; Kotnala, R.; Kumar, S.; Bhatt, P.; Jotania, R.; Kumar, R. Study of structural and magnetic properties of (Co–Cu) Fe2O4/PANI composites. Mater. Chem. Phys. 2013, 141, 406–415. [Google Scholar] [CrossRef]
- Alzoubi, G.M.; Albiss, B.; Shatnawi, M.; Bsoul, I.; Alsmadi, A.; Salameh, B.; Alna’washi, G. Influence of High-Temperature Annealing on Structural and Magnetic Properties of Crystalline Cobalt Ferrite Nanoparticles in the Single-Domain Regime. J. Supercond. Nov. Magn. 2020, 33, 3179–3188. [Google Scholar] [CrossRef]
- Kumar, N.; Singh, D.; Nigam, A.; Rajpoot, O.; Yadav, M.; Singh, Y.P.; Prakash, P.S.; Singh, S. Structural and magnetic properties of zinc doped copper ferrite synthesized by sol-gel and hydrothermal route. Mater. Phys. Mech. 2021, 47, 306–314. [Google Scholar]
- Hammad, T.M.; Salem, J.K.; Amsha, A.A.; Hejazy, N.K. Optical and magnetic characterizations of zinc substituted copper ferrite synthesized by a co-precipitation chemical method. J. Alloys Compd. 2018, 741, 123–130. [Google Scholar] [CrossRef]
- Sang, C.; Jin, S.; Li, G.; Luo, Y. Preparation of copper ferrite by sol–gel method and the synergistic catalytic for the thermal decomposition of ammonium perchlorate. J. Sol-Gel Sci. Technol. 2021, 98, 559–567. [Google Scholar] [CrossRef]
- Surashe, V.; Waghule, N.; Raut, A.; Pandit, A.; Dorik, R.; Jadhav, K. Ceramic synthesis and X-ray diffraction characterization of copper ferrite. AIP Conf. Proc. 2021, 2369, 020209. [Google Scholar]
- Rajini, R.; Ferdinand, A.C. Structural, morphological and magnetic properties of (c-ZnFe2O4 and t-CuFe2O4) ferrite nanoparticle synthesized by reactive ball milling. Chem. Data Collect. 2022, 38, 100825. [Google Scholar] [CrossRef]
- Mathew, D.S.; Juang, R.S. An overview of the structure and magnetism of spinel ferrite nanoparticles and their synthesis in microemulsions. Chem. Eng. J. 2007, 129, 51–65. [Google Scholar] [CrossRef]
- Dippong, T.; Levei, E.A.; Deac, I.G.; Neag, E.; Cadar, O. Influence of Cu2+, Ni2+, and Zn2+ Ions Doping on the Structure, Morphology, and Magnetic Properties of Co-Ferrite Embedded in SiO2 Matrix Obtained by an Innovative Sol-Gel Route. Nanomaterials 2020, 10, 580. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alzoubi, G.M.; Alsmadi, A.; Alna’washi, G.; Salameh, B.; Shatnawi, M.; Alnemrat, S.; Albiss, B.; Bsoul, I. Coexistence of superparamagnetism and spin-glass like behavior in zinc-substituted cobalt ferrite nanoparticles. Appl. Phys. A 2020, 126, 512. [Google Scholar] [CrossRef]
- Alzoubi, G.M. Probing the structural and magnetic properties of small crystalline nickel ferrite nanoparticles near the upper size limit of the single-domain regime. Adv. Appl. Ceram. 2020, 119, 224–232. [Google Scholar] [CrossRef]
- Chatterjee, B.K.; Bhattacharjee, K.; Dey, A.; Ghosh, C.K.; Chattopadhyay, K.K. Influence of spherical assembly of copper ferrite nanoparticles on magnetic properties: Orientation of magnetic easy axis. Dalton Trans. 2014, 43, 7930–7944. [Google Scholar] [CrossRef] [PubMed]
- Djerdj, I. Rietveld Refinement in the Characterization of Crystalline Materials; MDPI: Basel, Switzerland, 2019. [Google Scholar]
- Young, R.A. The Rietveld Method; International Union of Crystallography: Chester, UK, 1993; Volume 5. [Google Scholar]
- Sickafus, K.E.; Hughes, R. Spinel compounds: Structure and property relations. J. Am. Ceram. Soc. 1999, 82, 3277–3278. [Google Scholar]
- Samavati, A.; Mustafa, M.; Ismail, A.; Othman, M.; Rahman, M. Copper-substituted cobalt ferrite nanoparticles: Structural, optical and antibacterial properties. Mater. Express 2016, 6, 473–482. [Google Scholar] [CrossRef]
- Mahmood, N.B.; Saeed, F.R.; Gbashi, K.R.; Hamodi, A.; Jaffar, Z.M. Structural properties of CoxCu1- xFe2O4 solid solution. J. Mech. Behav. Mater. 2021, 30, 220–227. [Google Scholar] [CrossRef]
- Nikam, D.S.; Jadhav, S.V.; Khot, V.M.; Bohara, R.; Hong, C.K.; Mali, S.S.; Pawar, S. Cation distribution, structural, morphological and magnetic properties of Co1- xZnxFe2O4 (x = 0–1) nanoparticles. RSC Adv. 2015, 5, 2338–2345. [Google Scholar] [CrossRef]
- Nayek, C.; Manna, K.; Bhattacharjee, G.; Murugavel, P.; Obaidat, I. Investigating size-and temperature-dependent coercivity and saturation magnetization in PEG coated Fe3O4 nanoparticles. Magnetochemistry 2017, 3, 19. [Google Scholar] [CrossRef] [Green Version]
- Zhang, H.; Zeng, D.; Liu, Z. The law of approach to saturation in ferromagnets originating from the magnetocrystalline anisotropy. J. Magn. Magn. Mater. 2010, 322, 2375–2380. [Google Scholar] [CrossRef]
- Nairan, A.; Khan, M.; Khan, U.; Iqbal, M.; Riaz, S.; Naseem, S. Temperature-dependent magnetic response of antiferromagnetic doping in cobalt ferrite nanostructures. Nanomaterials 2016, 6, 73. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nadeem, K.; Krenn, H.; Traußnig, T.; Würschum, R.; Szabó, D.; Letofsky-Papst, I. Effect of dipolar and exchange interactions on magnetic blocking of maghemite nanoparticles. J. Magn. Magn. Mater. 2011, 323, 1998–2004. [Google Scholar] [CrossRef]
| Quantity | Refined Value |
|---|---|
| D(nm) | 20 |
| 5.39 | |
| R-factors (%) | |
| 1.4755 | |
| 1.5232 | |
| () | 1.06 |
| Cell parameters (Å) | |
| 8.3708 | |
| u | 0.2599 |
| Temperature | ||||
|---|---|---|---|---|
| (K) | (emu/g) | (kOe) | (emu/g) | (erg/cm) |
| 300 | ||||
| 200 | − | − | ||
| 150 | − | − | ||
| 100 | − | − | ||
| 10 | − | − |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alzoubi, G.M. The Effect of Co-Doping on the Structural and Magnetic Properties of Single-Domain Crystalline Copper Ferrite Nanoparticles. Magnetochemistry 2022, 8, 164. https://doi.org/10.3390/magnetochemistry8120164
Alzoubi GM. The Effect of Co-Doping on the Structural and Magnetic Properties of Single-Domain Crystalline Copper Ferrite Nanoparticles. Magnetochemistry. 2022; 8(12):164. https://doi.org/10.3390/magnetochemistry8120164
Chicago/Turabian StyleAlzoubi, Gassem M. 2022. "The Effect of Co-Doping on the Structural and Magnetic Properties of Single-Domain Crystalline Copper Ferrite Nanoparticles" Magnetochemistry 8, no. 12: 164. https://doi.org/10.3390/magnetochemistry8120164
APA StyleAlzoubi, G. M. (2022). The Effect of Co-Doping on the Structural and Magnetic Properties of Single-Domain Crystalline Copper Ferrite Nanoparticles. Magnetochemistry, 8(12), 164. https://doi.org/10.3390/magnetochemistry8120164






