1. Introduction
Li-ion batteries (LIB) are promising energy-storage devices for portable consumer electronics as well as energy sources for electric vehicles. However, Li-ion batteries are susceptible to high temperatures, which can lead to ignition and even the explosion of these batteries. In the event of improper operation, gas and heat may be generated inside the battery as a result of electrode, electrolyte or solvent degradation. This gas mixture can be very explosive or even self-igniting if it is released from the cell, mixed with air or through oxygen-release reactions from the electrodes (especially the cathode). Physical damage by crushing or puncturing can also cause rapid ignition and the destruction of the battery [
1,
2].
Many efforts have been made to improve the safety of Li-ion batteries. There have been developments to obtain safer electrolytes: for instance, by the addition of flame retardants (FRs) to electrolytes, or by using less-flammable electrolyte solvents such as ionic liquids (ILs) and hydrofluoroethers (HFEs) instead of organic electrolytes [
1]. Fluorinated solvents are also a possibility for enhancing the battery’s safety characteristics [
3]. Safety devices incorporated into battery cells and modules, such as a shutdown separator, cell vent, and current interrupt devices, etc., are further options for managing battery safety [
2,
4].
Several different materials can be used as anode materials [
5]. Typically, graphite, LTO or Si–graphite blend materials are used in commercial cells. They are processed as a layer together with a binder and carbon black. Accordingly, the combustible fraction involved in the thermal runaway fire (expressed as carbon) is correspondingly high. To our knowledge, no flame retardants are currently processed directly into the anode material.
Standard organic, carbonate-based electrolytes are sensitive to increases in local temperature and overcharge, even if the temperature does not overcome 100 °C [
6]. Such abusive conditions can cause exothermic reactions inside the cell and lead to thermal runaway. The main strategies to improve LIB safety on a cell level are the development of less/non-flammable electrolytes, the use of electrolyte additives or the addition of fire retardants [
7,
8,
9,
10]. ILs are of major interest due to their high thermal, chemical and electrochemical stability. However, their high viscosity and respectively poor ionic conductivity hamper the application of ILs in batteries. Therefore, the addition of organic solvents to such IL electrolytes is often applied to improve the ionic conductivity and/or reduce the viscosity of pure IL electrolytes [
11,
12]. Solid-state electrolytes, namely, inorganic solid electrolytes and polymer electrolytes, are often described as more thermally stable; therefore, they can replace liquid electrolytes, overcoming the issue of their low ionic conductivity [
13]. Additionally, organic phosphorous compounds can be used as fire retardants. They improve the thermal stability of the electrolyte, reduce its flammability by interrupting decomposition reactions, and can even diminish the ageing process [
14,
15,
16,
17,
18,
19,
20,
21,
22]. At the same time, however, organic phosphates can act as electrolytes in the battery itself (e.g., trimethyl phosphate). Non-flammable phosphate electrolytes improve the thermal stability of the battery and suppress gas generation during charging and discharging [
17,
23].
Lithium bis(trifluoromethanesulfonyl)imide (LiTFSI) is an alternative conducting salt. It is related to the standardly used, high reactive LiPF
6 due to its higher thermal and electrochemical stability [
24]. Its main disadvantage is its corrosivity towards aluminum (a cathode current collector) [
24]. Electrolyte additives such as lithium difluoro(oxalato)borate (LiDFOB), in combination with fluoroethylene carbonate (FEC), can prevent Al corrosion in presence of LiTFSI and improve cell cycling and cell aging [
24].
The formation of solid electrolyte interface layers (SEIs) appears on an anode surface during the first charging cycles as a result of electrolyte decomposition, This protecting the electrolytes from further decomposition. Thus, the thermal stability of the SEI is crucial for battery safety. The SEI decomposition temperature can be increased if a combination of thermally stable Li salts and high-boiling electrolytes is used. Some extra additives (e.g., organic phosphorous compounds) can also strengthen the SEI and improve its thermal stability [
25,
26]. Jiang et al. proposed a composite electrolyte additive consisting of perfluoro-2-methyl-3-pentanone (PFMP) and N,N-dimethylacetamide (DMAC), which doubled the protection mechanism. DMAC improved the thermal stability of the electrolyte and PFMP served as a self-cooling component [
27].
The separator in the battery cell also plays a crucial role in battery safety. It prevents physical contact between the cathode and anode while simultaneously allowing ion transport between both electrodes [
4]. Damage caused by external puncturing, dendrite growth or shrinkage by overheating or overcharging leads to an internal short circuit and thus accelerates thermal runaway in the battery. Separators with a so-called “shutdown behavior” are favored because of their ability to “close pores” if the temperature increases [
2]. Single- or three-layer separators composed of polyolefins are the most widely used separators in batteries [
9,
28,
29]. They exhibit a high porosity, low thickness and demonstrate a low ionic resistivity. A crucial disadvantage of such separators, however, is their narrow melting temperature range, which is between 135 °C and 165 °C [
4]. Additionally, a shrinkage of the separator occurs at even lower temperatures. Composite separators made of ceramics such as LiAlO
2, Al
2O
3, MgO, etc., coated onto polyolefins were developed with the aim of improving the melting point of the separator and thus the cell safety [
4,
9,
29,
30,
31]. There are still many efforts being made with respect to the thermal-stability improvements of separators. On one hand, different polymers and ceramics are intensively investigated with respect to their higher thermal stability. For example, fluorinated polyimide nanofibers with improved flame-retardant properties were proposed [
32]. Luo et al. demonstrated that no visible changes occurred when the separator was heated up to 160 °C. This was additionally confirmed through differential scanning calorimetry and thermogravimetric analysis [
32]. Liu et al. developed a polyphenylene sulfide separator which displayed a high porosity, high wettability and high thermal stability (up to 280 °C) [
33]. A composite, polyvinyl-alcohol-based separator with a wider shutdown temperature window of 155 °C was fabricated and successfully tested [
34]. Cellulose-based separators are a good option for both battery safety improvement and the environment. In addition to demonstrating negligible shrinkage at an elevated temperature, they can also improve rate capability and enhance capacity retention and cycling stability [
35,
36]. The incorporation of MoO
3 and Al-doped Li
6.75La
3Zr
1.75Ta
0.25O
12 into poly(vinylidene fluoride hexafluoropropylene) demonstrates superior safety to flame events. Only approximately 5% of shrinkage was observed after heating the separator at 160 °C for 4 h [
31].
Another possible avenue to improving the flame-retardant effect of a separator is to include additives with known flame-retardant properties as surface coatings or even introduce them into the separator structure. Liao et al. proposed an environmentally friendly separator consisting of bacterial cellulose, attapulgite rod and ammonium polyphosphate which displayed self-extinguishing characteristics after ignition with a low heat and gas contribution [
36]. Lee et al. significantly improved the thermal stability of a tri-layer separator by coating it with brominated poly(2,6-dimethyl-1,4-phenylene oxide) [
37]. Peng et al. soaked ceramic separators with phenol-formaldehyde resin, which is known for its insulating properties and is already widely used in electrical equipment [
38].
Li dendrite growth is an adverse process inside the battery which can lead to separator damage and a short circuit [
39]. Dendrite penetration through the separator can be suppressed by coating a polyolefin film with an aramid resin. The small pores of this separator do not allow dendrites to grow into or through it, leading to granular or spherical deposits along the plane of the separator [
39]. Good separation between the cathode and anode can be achieved by coating both electrodes with an α-Al
2O
3 slurry. Such a coating is very thin, less sensitive toward high temperatures and more mechanically and dimensionally stable when compared to commercial separators [
40]. Flame retardants can even be added to the current collector foils. Thus, Ye et al. proposed an ultralight, polyimide-coated Cu current collector with the addition of flame-retardant triphenyl phosphate, thus improving the cell safety [
41].
Another potential approach to reduce the incidence of fires is to use substances that can release fire-retardant gases. It is known that oxalates, formates, fumarates and malonates can release CO
2 under thermal exposure. The exact decomposition depends strongly on the surrounding atmosphere and the reaction conditions. Nevertheless, it is conceivable that the released CO
2 has a fire-retardant effect or can influence the gas composition limits for explosion. Lithium oxalate in particular has already been investigated mechanistically; therefore, it is known that it decomposes into the corresponding lithium carbonate at approximately 550 °C, with the CO being split off [
42,
43,
44,
45]. The CO immediately reacts again in the presence of oxygen, forming CO
2.
In this study, we have incorporated various flame retardants directly into the anode material to answer the open question of how to best introduce flame retardants into the cell and whether they have the desired safety-enhancing effect. This allows for the corresponding substances to act directly in the cell so that they can react appropriately (well and quickly) in the case of cell abuse or cell failure. Since this function is embodied directly in the cell, failures due to electrical issues or time delays from activating external safety measures can also be overcome. At the same time, the ion transport and cell chemistry are not or are only marginally affected. Due to the high flammability of the carbonate mixtures typically used as electrolytes, we have considered the influence of the electrolytes to address the overall safety.
In the present study, three organic salts (namely, lithium oxalate, sodium fumarate and sodium malonate) with two groups of -COO- in their structure were introduced into the Li battery anode. The intent was to achieve a CO2 release during thermal decomposition at elevated temperatures, thus slowing down fire extension or even preventing battery ignition. For that purpose, full coin cells as well as pouch-bag cells with modified anodes vs. NMC111 (NMC—lithium nickel manganese cobalt oxide) cathodes were assembled and filled with three different electrolytes:
- (1)
An LP30-standard Li-ion battery electrolyte containing ethylene carbonate (EC) and dimethyl carbonate (DMC) in equivalent volumetric parts as solvents and 1 M LiPF6 as a conductive salt;
- (2)
Ethylene carbonate (EC) and propylene carbonate (PC) + 1 M LiDFOB;
- (3)
1,2-butylene carbonate (1,2-BC) and fluoroethylene carbonate (FEC) + 1 M LiTFSI.
The performance of these battery cells (within 122 cycles) and the cell aging (within 1024 cycles) was investigated. Finally, the pouch cells were overcharged, and the released gases were studied using gas chromatography (GC). Postmortem analyses on coin cells using liquid GC-MS (MS—mass spectrometry) to investigate the decomposition reactions taking place in the presence of flame retardants are also provided.
4. Discussion
In this study, three flame retardants incorporated into the anode layer (namely, lithium oxalate, sodium fumarate and sodium malonate) were investigated together with three electrolytes (namely, 1 M LiPF
6 in EC/DMC, 1 M LiTFSI in 12-BC/FEC and LiDFOB in EC/PC). It was shown that the electrolytes were optimized in terms of flammability (especially with respect to flash point,
Table 2) and the flame retardants were selected with respect to their potential for CO
2 release.
Based on the measurements, the gas release of the FRs can be discussed in more detail. The release of CO
2 in the air atmosphere, neglecting energy balancing, can be estimated from TGA measurements that exploited revealed information about mass losses. Assuming an ideal gas behavior and under atmospheric conditions (which are present after release) in a temperature range between 300 °C and 600 °C, respectively (as the temperature range in which thermal runaway of a battery usually occurs), and assuming the direct formation of CO from CO
2 (1:1 mol), the calculated values of
Table 6 were obtained. From this point of view, lithium oxalate is more favorable as a flame retardant than sodium fumarate or malonate.
The fabrication of the anode layers was specifically investigated and described. Thus, the rheological behavior of the anode slurry was studied in detail. The slurry containing 10 wt.% and more of lithium oxalate can be considered a mixture of graphite, active carbon, and undissolved FR crystals in a saturated Li-oxalate-Na-CMC/SBR-water solution. In contrast, a major part of sodium fumarate and sodium malonate were dissolved in the slurry, even at high concentrations. Adding a water-soluble FR to a slurry containing a Na-CMC-water solution can also cause changes in the Na-CMC polymer structure and thus in the entire graphite/active carbon/Na-CMC network.
Mandrel bending tests around a metal cylinder showed no significant changes in the adhesion of the self-made anode sheets up to an FR content of 10%. For lithium oxalate, even at a 35 wt.% FR content, almost the same adhesive strength could be shown. SEM, microscopy and conductivity tests on the electrodes showed that the anode layers containing FRs were very similar up to approximately a 20 wt.% FR content in the case of lithium oxalate and up to a 10 wt.% FR in the cases of sodium fumarate and sodium malonate. This was confirmed by the adhesion tests and suggests that the FRs are well-fabricated into the layers.
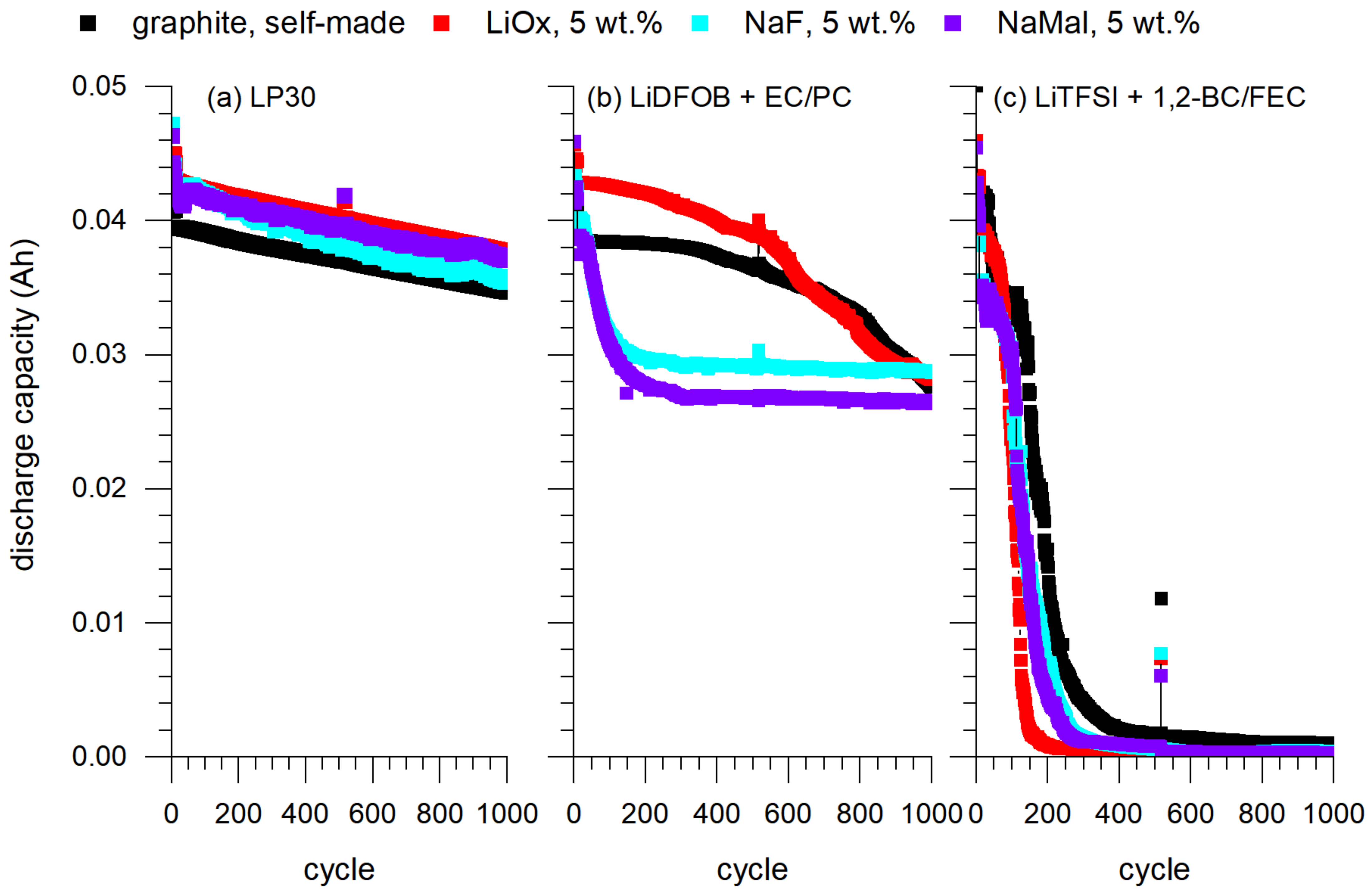
The FRs were investigated in coin cells to evaluate their impact on cell capacity, especially at higher current rates. A significant capacity loss was observed for higher C-rates when the FR concentration increased. This can be explained by the fact that FRs themselves are a source of additional resistivity, meaning that the mobility of the electrons through the anode layer at elevated current rates was hampered since their intrinsic electrical conductivity was lower than that of the surrounding electrode layer. Another reason for the capacity loss at high C-rates may be changes in the SEI layer caused by the high amount of the FR. Possibly, the transport of Li ions was slowed down during charging and discharging as a result of changes in the SEI caused by FR decomposition. At the same time, there was no noticeable difference in the rate capability for cells with Li oxalate and Na fumarate. On the contrary, discharge capacities of the cells with Na malonate were remarkably smaller. Higher C-rates intensified this effect. The influence of FRs on discharge capacity was more pronounced at high C-rates (>5 C). Sodium malonate featured the slightest effect on concentration-dependent capacity losses and was highly dependent on the electrolyte used. The influence for sodium fumarate had a moderate impact, and lithium oxalate had the largest impact on capacity losses.
Cell tests provide an indication that, at low current rates, the flame retardants have no negative effect on performance and cell capacity, even at high FR concentrations. Higher currents above 5 C, on the other hand, lead to a reduction in discharge capacity with an increasing FR content due to deteriorated conductivity in the layers. While lithium oxalate and sodium fumarate both exhibited similar behaviors, sodium malonate showed the weakest performance and exhibited a significant capacity loss at low-current rates. Gas-releasing tests demonstrated that the evolved CO2 amount was higher when flame retardants were present in the anode layer.