1. Introduction
Central venous catheterization (CVC) is a routine procedure in intensive care medicine and anesthesiology, performed over 6 million times annually in the United States and Europe [
1,
2]. The internal jugular vein (IJV) is the most utilized insertion site [
3], with clinical indications including administration of medication, recording of central venous pressure or other haemodynamic parameters [
4], haemodialysis [
5], and accessing the right atrium [
6]. Traditionally, CVC placement via the IJV is performed using landmark techniques based on knowledge of anatomic structures and palpation of the carotid artery (CA) [
7,
8]. However, this approach does not account for anatomic variations or the presence of venous thrombosis at the CVC insertion site [
7,
8,
9], resulting in accidental puncture of the CA with incidence of up to 9.4% [
1] and leading to devastating events including hematoma, stroke and death [
10,
11].
Interpatient anatomic variations [
12,
13] and the presence of venous thrombosis can generally not be identified using a landmark technique. However, ultrasound (US) can be used to visualize anatomic structures and confirm patency of the vein to help avoid unintended arterial puncture or unsuccessful cannulation [
8]. The ability of US-guidance to reduce the number of complications and to increase the safety and quality of CVC placement has been demonstrated in several systematic reviews and meta-analyses [
7,
8]. The use of real-time US for guiding the puncture of the vein and confirming the correct needle, wire and catheter position in the vein is now the recommended approach endorsed by many professional societies [
9,
14,
15,
16].
The US images used for needle guidance can be captured with the US probe oriented transversely to the vein, showing the short axis (SAX) image such that the needle is inserted “out-of-plane”, or parallel to the vein [
14,
17]. Alternatively, the long axis (LAX) can be captured such that the needle is inserted “in-plane”, or at an oblique orientation relative to the US image [
14,
17]. While there are no data to suggest which approach is superior [
8], SAX with the out-of-plane approach is the preferred option [
16] because it is easier to learn [
18] and results in higher success rates with the first attempt for CVC placement in the IJV [
19]. However, in this approach, the needle tip and shaft both appear as hyperechoic spots in the US image, making it challenging to distinguish one from the other. The uncertainty surrounding the exact location of the needle tip in US images is associated with inadvertent arterial, posterior IJV, and pleural punctures [
11,
20,
21]. As a result, acute adverse events still occur under US-guided CVC, with an adverse event rate as high as 19.7% being reported in the literature [
22] (or a complication rate of 4.6% as reported by [
23]). The key challenge for US-guided CVC is maintaining visibility of the needle tip in the US image throughout the entire insertion [
8]. The success of vein puncture may be affected by the inability to visualize all relevant anatomy and the needle tip within the limited 2D US field of view (FoV). Additionally, the clinician obtains the visual information to guide the insertion from the US console while simultaneously performing the insertion on the patient. This results in a decoupling of their visual and motor fields, which may affect insertion success.
To address these limitations of US-guided CVC, several mixed-reality (MR) systems were developed to provide 3D visualizations for needle-insertion. Here, the term MR is employed to refer to visualization techniques that fall on the reality-virtuality spectrum between fully real and fully virtual environments [
24]. Rochlen, Levin, and Tait developed a training system for central-line insertion using augmented reality (AR) glasses [
25], the use of which allows participants to place the needle in the mannequin using a “first-person” point-of-view AR system, where the relevant internal anatomical landmarks are rendered within the AR glasses and perceived by the users in their anatomically correct positions. While the technical details were lacking in [
25], users participating in their study (
) reported the first-person perspective AR view of the internal anatomy was helpful in guiding needle-insertion. Huang et al. [
26] also introduced an AR-glass system for central-line simulation and teaching. Distinct from [
25], this system displayed a concise CVC instructional slideshow as a checklist for participants to follow as they carried out the procedure instead of displaying the underlying anatomical structure as a visual aid. As these two systems were designed for teaching and training purposes, feasibility of clinical translation of these systems was not demonstrated.
Ameri et al. [
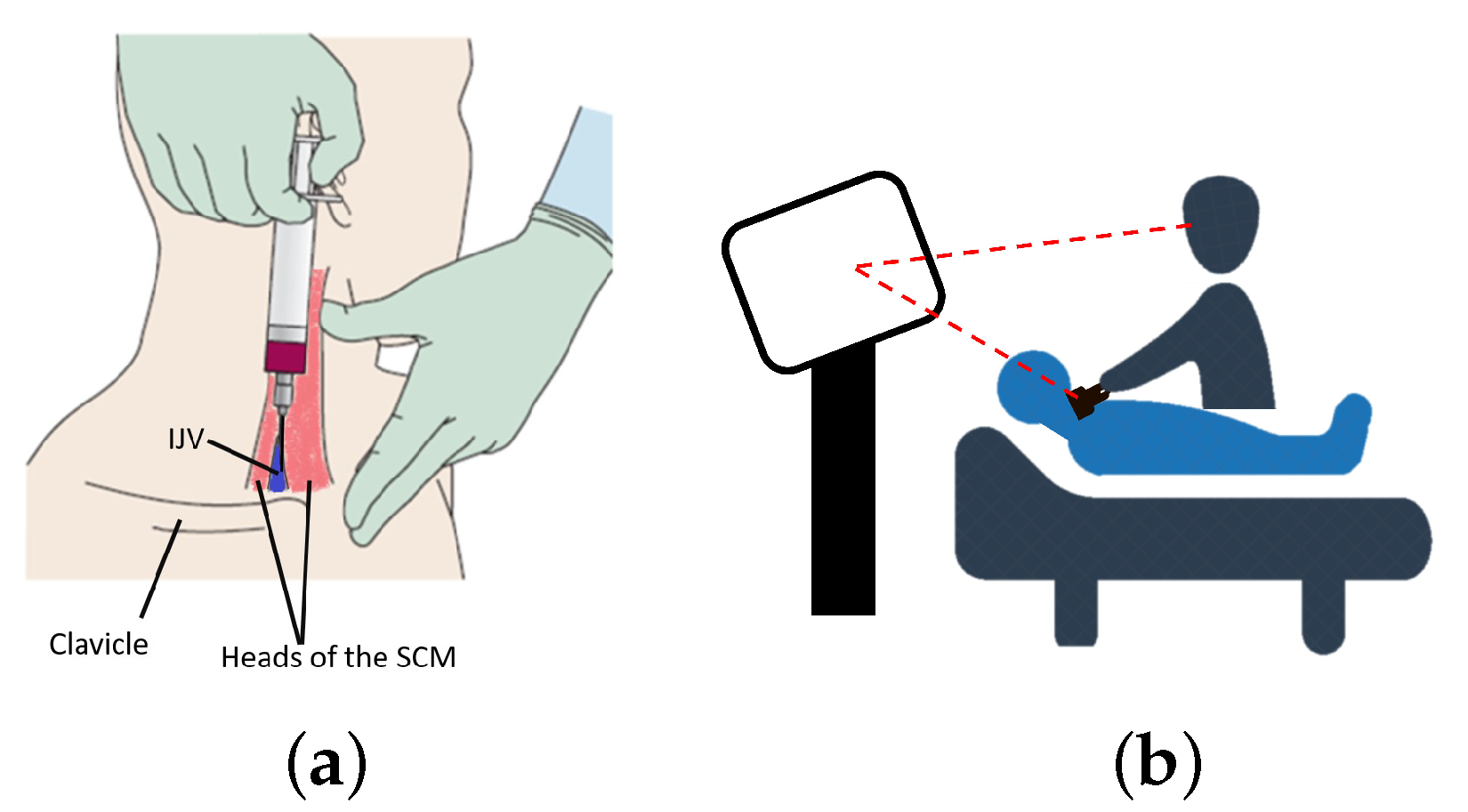
27] developed a surgical navigation system for guiding CVC puncture that employed a magnetic tracking system (MTS) to render virtual representations of the US transducer, surgical needle, and needle trajectory using a standard 2D monitor. Their visualization comprised the real-time position and orientation (pose) of the tracked needle relative to a stationary, front-facing US image to maintain consistency with the traditional US-guided procedure (
Figure 1b). This system did not result in a significant reduction of the complications associated with the needle-insertion when compared to that experienced by expert users employing the US-only technique [
27]. The performance of this visualization paradigm may have been influenced by both the fixed front-facing US visualization, as the user still relied on a 2D visualization to perform a 3D procedure, and the visual and motor disparity produced when the visualization device is exterior to the visual field of the phantom where the needle-insertion is performed.
In this paper, we investigate the efficacy of a first-person immersive MR system for the navigation of needles for central venous catheterization (CVC). While the long-term goal of this work is clinical deployment, we first must understand how the visualization paradigm associated with surgical information affects the complication rate during US-guided CVC. Similar to [
27], we propose a needle guidance system using an MTS to provide, in real-time, the spatial relationship between surgical instruments relative to the patient anatomy. We compare the standard-of-care US-only guidance to the use of our MR guidance system, where the images are displayed both on a 2D monitor and also using a first-person perspective view available with a head-mounted display (HMD). We hypothesize that employing an HMD for visualization will improve the success of the needle-insertions compared to those that use US-only guidance or a conventional 2D monitor. Via a user study involving 31 clinicians and 20 medical students, this work highlights the importance of providing coherent visual and motor fields for image-guided surgical applications.
4. Discussion
For the 2D monitor-based MR guidance, our data showed there was no significant improvement in targeting accuracy over US-only guidance for either the expert clinicians or the novice medical students. In our monitor-based MR guidance system, the virtual representation of a tracked needle, needle trajectory, US transducer, and streaming US video was visualized in a common coordinate system (
Figure 4). The angle of view for US video was unconstrained, i.e., participants were free to adjust the vantage point, with most participants preferring an oblique viewing angle for improved 3D perception. These findings are consistent with the conclusions of Ameri et al. [
27], who also found no significant improvements of the expert cohort when monitor-based MR guidance was used compared to the US-only mode of visualization. The key difference between their system and ours is that theirs used a fixed front-facing US image (retaining the familiar view of US images on a US console) [
27], while ours allowed the user to choose an arbitrary 3D vantage point to view the 2D image from. In both systems, virtual representations of tracked surgical instruments supplemented the 3D view of the surgical scene, with the intention to improve the visualization and success rates of the needle-insertion. The lack of improvement in our system and that proposed by Ameri et al. is likely due to the decoupling between the clinician’s motor and visual fields when the US console or a monitor was used as the visualization device.
In contrast, the use of HMD, which delivered an MR guidance system using a first-person immersion vantage point, significantly improved the outcome of CVC insertion. The CVC insertion success rate and accuracy (measured as the distance between the final tip location to the center line of the vessel and from the vessel wall) were improved when compared to US-only guidance. These results emphasize the importance of using an HMD to ensure a coherent visual and motor field during needle guidance. This is achieved in the HMD by bringing the needle guidance information directly into the line-of-sight of the clinician. While the monitor-based system would be more readily integrated into a clinical workflow, our results suggest there are benefits for using an HMD for needle guidance, promoting the continued pursuit of research related to the use of HMDs in similar clinical scenarios.
The US-only case resulted in a 68% success rate for the expert cohort, which is low despite the simplicity of the phantom. We believe there were several factors affecting the outcome of this guidance approach. During analysis, it was observed that clinicians may not use the US-guidance information effectively to follow the needle tip throughout the insertion, as in most US-only cases the US image remained fairly stationary. In addition, we observed a high reliance on muscle memory as the user would align the needle with the center of the probe and in one swift motion perform the insertion based on intuition. The user would verify that the needle was in the US image based on its reflection. However, in many cases the users captured the shaft instead of the tip of the needle, leading the user to erroneously believe that they had performed the insertion successfully. Additionally, as this is the approach with which they are most familiar, there is a potential that they were overconfident in the accuracy of their needle placement given the simplicity of the phantom.
In terms of complication rates, there was no significant difference in the rates of CA puncture between these three modes of visualization implemented for our experiment, as the overall rates of CA puncture were low. One expert and two novices punctured the CA, and all punctures occurred under US-guidance. The observed low complication rate of CA puncture is likely due to the simplicity of our anthropomorphic neck phantom as the IJV and CA had a simple orientation with limited overlap as it was laterally positioned to the IJV (
Figure 2). Additional experiments using a range of neck phantoms with diverse anatomical variations are planned future projects. As depicted in
Figure 2, the appearance and configuration of the neck vasculature are variable. The utilization of US to guide needle-insertion in the SAX/out-of-plane approach has the inherent inability to track the position of needle tip once it is traversed beyond the US image plane. In a realistic clinical scenario, the inadvertent posterior IJV wall punctures could result in damage to critical anatomical structures adjacent to the IJV, including CA, but this was not the case using our phantom. While our study did not show that the first-person immersive MR system had a significant effect on the rates of CA puncture, the HMD system has indeed resulted in higher rates of successful insertions as the participants positioned the needle closer to the center of the vessel with fewer posterior wall punctures than US-only guidance.
The feasibility of using an HMD in a clinical setting is a critical concern for using the first-person immersive MR for needle guidance. Results of the questionnaire responses suggested that the first-person immersive MR HMD system may be more feasible when used for training rather than for clinical deployment, as on average these clinicians ranked the clinician viability of the system a compared to a for usefulness for training. Most clinicians indicated that they would consider using advanced visual guidance on an ad-hoc basis, with the belief that the more complicated cases could benefit from the advantages offered by the advanced visualization system. We are currently employing the HTC VIVE Pro as a virtual-reality display device even though it has stereo cameras and can be used as an augmented reality video-pass-through display. Incorporation of the stereo camera feeds for visual guidance may facilitate the clinical acceptability of this first-person immersive technology as the clinician would be able to visualize the guidance information while maintaining a direct view of the real surrounding environment. However, such an approach is not feasible with current devices as the stereo camera image resolution and fidelity are low. Alternatively, mixed-reality optical-see-through HMD devices such as the Microsoft Hololens could be used, but extensive evaluation of the accuracy tracking (i.e., camera hand-eye calibration) and careful integration with optical- or magnetic-tracking systems would be required.
Our results, in conjunction with those presented by Ameri et al. [
27], suggest that visualizing the 3D spatial relationship of surgical instruments using a 2D display does not provide sufficient guidance for a user to perceive the 3D context of the surgical scene, regardless of whether the US image is viewed in a front-facing or an oblique perspective. In contrast, a 2D monitor-based MR system has been developed and successfully applied to focal liver tumor ablation [
37], prostate brachytherapy [
38], and breast biopsy [
39], suggesting there is a potential pathway to implement a clinically feasible monitor-based guidance system for CVC insertion. Developing and incorporating advanced 3D visualization to improve 3D perception using a 2D monitor is a planned future project. Alternatively, a tablet-based display, which could be situated closer to the surgical site, could facilitate greater coherence of the visual and motor fields.