Mineral Composition of Skeletal Elements in Dorid Nudibranchia Onchidoris muricata (Gastropoda, Mollusca)
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Sample Preparation for EDX
2.3. EDX Analysis
2.4. Statistical Analysis
2.5. Raman Spectroscopy
3. Results
3.1. Internal Morphology of Spicules
3.2. Analysis of Spicule Elemental Composition Using EDX
3.3. Grouping of Spicules According to the Mineral Composition Based on Sections
3.4. Analysis of Element Ratios in the Clusters
3.5. Grouping of Spicules According to the Mineral Composition Based on Fractures
3.6. Raman Spectroscopy Data
4. Discussion
4.1. Methodological Aspects
4.2. Analysis of Spicule Mineral Composition
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Graham, A. The molluscan skin with special reference to prosobranchs. J. Molluscan Stud. 1957, 32, 135–144. [Google Scholar]
- Jones, W.C.; Jenkins, D.A. Calcareous sponge spicules: A study of magnesian calcites. Calcif. Tissue Res. 1969, 4, 314–329. [Google Scholar] [CrossRef]
- Carter, J.G.; Aller, R.C. Calcification in the bivalve periostracum. Lethaia 1975, 8, 315–320. [Google Scholar]
- Le Roy, N.; Jackson, D.J.; Marie, B.; Ramos-Silva, P.; Marin, F. The evolution of metazoan α-carbonic anhydrases and their roles in calcium carbonate biomineralization. Front. Zool. 2014, 11, 75. [Google Scholar]
- Lambert, G.; Lambert, C.C. Spicule formation in the New Zealand ascidian Pyura pachydermatina (Chordata, Ascidiacea). Connect. tissue Res. 1996, 34, 263–269. [Google Scholar] [CrossRef] [PubMed]
- Salvini-Plawen, L. Neue scandinavische Aplacophora (Mollusca, Aculifera). Sarsia 1967, 27, 1–63. [Google Scholar]
- Rieger, R.M.; Sterrer, W. New spicular skeletons in Turbellaria, and the occurrence of spicules in marine meiofauna. J. Zool. Syst. Evol. Res. 1975, 13, 207–278. [Google Scholar]
- Emlet, R.B. Echinoderm calcite: A mechanical analysis from larval spicules. Biol. Bull. 1982, 163, 264–275. [Google Scholar]
- Leise, E.M. Chiton integument: Metamorphic changes in Mopalia muscosa (Mollusca, Polyplacophora). Zoomorphology 1984, 104, 337–343. [Google Scholar]
- Sandford, F. Physical and chemical analysis of the siliceous skeletons in six sponges of two groups (Demospongiae and Hexactinellida). Microsc. Res. Tech. 2003, 62, 336–355. [Google Scholar]
- Rahman, M.A.; Oomori, T. Structure, crystallization and mineral composition of sclerites in the alcyonarian coral. J. Cryst. Growth 2008, 310, 3528–3534. [Google Scholar] [CrossRef]
- Simkiss, K.; Wilbur, K.M. Biomineralization; Elsevier: Amsterdam, The Netherlands, 1989. [Google Scholar]
- Boskey, A.L. Biomineralization: An overview. Connect. tissue Res. 2003, 44, 5–9. [Google Scholar] [CrossRef] [PubMed]
- Weiner, S.; Dove, P.M. An overview of biomineralization processes and the problem of the vital effect. Rev. Mineral. Geochem. 2003, 54, 1–29. [Google Scholar] [CrossRef]
- Beniash, E.; Aizenberg, J.; Addadi, L.; Weiner, S. Amorphous calcium carbonate transforms into calcite during sea urchin larval spicule growth. Proc. R. Soc. London. Ser. B Biol. Sci. 1997, 264, 461–465. [Google Scholar] [CrossRef]
- Uriz, M.J.; Turon, X.; Becerro, M.A. Silica deposition in Demosponges: Spiculogenesis in Crambe crambe. Cell Tissue Res. 2000, 301, 299–309. [Google Scholar] [CrossRef] [PubMed]
- Uriz, M.J.; Turon, X.; Becerro, M.A.; Agell, G. Siliceous spicules and skeleton frameworks in sponges: Origin, diversity, ultrastructural patterns, and biological functions. Microsc. Res. Tech. 2003, 62, 279–299. [Google Scholar] [CrossRef]
- Müller, W.E.; Rothenberger, M.; Boreiko, A.; Tremel, W.; Reiber, A.; Schröder, H.C. Formation of siliceous spicules in the marine demosponge Suberites domuncula. Cell Tissue Res. 2005, 321, 285–297. [Google Scholar] [CrossRef]
- Wilt, F.; Killian, C.E.; Croker, L.; Hamilton, P. SM30 protein function during sea urchin larval spicule formation. J. Struct. Biol. 2013, 183, 199–204. [Google Scholar] [CrossRef]
- Koga, H.; Morino, Y.; Wada, H. The echinoderm larval skeleton as a possible model system for experimental evolutionary biology. Genesis 2014, 52, 186–192. [Google Scholar] [CrossRef]
- Voigt, O.; Adamska, M.; Adamski, M.; Kittelmann, A.; Wencker, L.; Wörheide, G. Spicule formation in calcareous sponges: Coordinated expression of biomineralization genes and spicule-type specific genes. Sci. Rep. 2017, 7, 45658. [Google Scholar] [CrossRef]
- Murdock, D.J. The ‘biomineralization toolkit’and the origin of animal skeletons. Biol. Rev. 2020, 95, 1372–1392. [Google Scholar] [CrossRef] [PubMed]
- Voigt, O.; Fradusco, B.; Gut, C.; Kevrekidis, C.; Vargas, S.; Wörheide, G. Carbonic anhydrases: An ancient tool in calcareous sponge biomineralization. Front. Genet. 2021, 12, 624533. [Google Scholar]
- Shimizu, K.; Nishi, M.; Sakate, Y.; Kawanami, H.; Bito, T.; Arima, J.; Leria, L.; Maldonado, M. Silica-associated proteins from hexactinellid sponges support an alternative evolutionary scenario for biomineralization in Porifera. Nat. Commun. 2024, 15, 181. [Google Scholar]
- Foale, S.J.; Willan, R.C. Scanning and transmission electron microscope study of specialized mantle structures in dorid nudibranchs (Gastropoda: Opisthobranchia: Anthobranchia). Mar. Biol. 1987, 95, 547–557. [Google Scholar] [CrossRef]
- Nikitenko, E.; Ereskovsky, A.; Vortsepneva, E. Ontogenetic dynamics of the subepidermal spicule complex in Nudibranchia (Gastropoda): The case of Onchidoris muricata. Zoology 2021, 144, 125886. [Google Scholar]
- Ehrlich, H. Molluscs spicules. In Biological Materials of Marine Origin; Springer: Berlin/Heidelberg, Germany, 2010. [Google Scholar]
- Ehrlich, H. Spicular structures in Molluscs. Mar. Biol. Mater. Invertebr. Orig. 2019, 13, 133–157. [Google Scholar]
- Mann, K.; Wilt, F.H.; Poustka, A.J. Proteomic analysis of sea urchin (Strongylocentrotus purpuratus) spicule matrix. Proteome Sci. 2010, 8, 33. [Google Scholar]
- Flores, R.L.; Livingston, B.T. The skeletal proteome of the sea star Patiria miniata and evolution of biomineralization in echinoderms. BMC Evol. Biol. 2017, 17, 125. [Google Scholar] [CrossRef]
- Borromeo, L.; Zimmermann, U.; Andò, S.; Coletti, G.; Bersani, D.; Basso, D.; Gentile, P.; Schulz, B.; Garzanti, E. Raman spectroscopy as a tool for magnesium estimation in Mg-calcite. J. Raman Spectrosc. 2017, 48, 983–992. [Google Scholar]
- Welton, J.E. SEM Petrology Atlas; American Association of Petroleum Geologists: Tulsa, OK, USA, 1984. [Google Scholar]
- Bora, J.; Deka, P.; Bhuyan, P.; Sarma, K.P.; Hoque, R.R. Morphology and mineralogy of ambient particulate matter over mid-Brahmaputra Valley: Application of SEM–EDX, XRD, and FTIR techniques. SN Appl. Sci. 2021, 3, 137. [Google Scholar]
- Malek, M.A.; Eom, H.J.; Hwang, H.; Do Hur, S.; Hong, S.; Hou, S.; Ro, C. Single particle mineralogy of microparticles from Himalayan ice-cores using SEM/EDX and ATR-FTIR imaging techniques for identification of volcanic ash signatures. Chem. Geol. 2019, 504, 205–215. [Google Scholar] [CrossRef]
- Ryan, P.C.; Reynolds Jr, R.C. The chemical composition of serpentine/chlorite in the Tuscaloosa Formation, United States Gulf Coast: EDX vs. XRD determinations, implications for mineralogic reactions and the origin of anatase. Clays clay Miner. 1997, 45, 339–352. [Google Scholar] [CrossRef]
- Almedros, A.; Porcel, D. A structural and microanalytical (EDX) study of calcium granules in the hepatopancreas of Helix aspersa. Comp. Biochem. Physiol. Part A Physiol. 1992, 103, 757–762. [Google Scholar] [CrossRef]
- Cribb, B.W.; Lin, C.L.; Rintoul, L.; Rasch, R.; Hasenpusch, J.; Huang, H. Hardness in arthropod exoskeletons in the absence of transition metals. Acta Biomater. 2010, 6, 3152–3156. [Google Scholar] [CrossRef]
- Gaspard, D.; Nouet, J. Hierarchical architecture of the inner layers of selected extant rhynchonelliform brachiopods. J. Struct. Biol. 2016, 196, 197–205. [Google Scholar] [CrossRef] [PubMed]
- Chan, V.B.; Toyofuku, T.; Wetzel, G.; Saraf, L.; Thiyagarajan, V.; Mount, A.S. Characterization of calcification events using live optical and electron microscopy techniques in a marine tubeworm. J. Vis. Exp. JoVE 2017, 120, 55164. [Google Scholar]
- Vittori, M.; Srot, V.; Bussmann, B.; Predel, F.; van Aken, P.A.; Štrus, J. Structural optimization and amorphous calcium phosphate mineralization in sensory setae of a terrestrial crustacean (Isopoda: Oniscidea). Micron 2018, 112, 26–34. [Google Scholar] [CrossRef]
- Krings, W.L.; Brütt, J.O.; Gorb, S.N. Elemental analyses reveal distinct mineralization patterns in radular teeth of various molluscan taxa. Sci. Rep. 2022, 12, 7499. [Google Scholar] [CrossRef]
- Krings, W.; Matsumura, Y.; Brütt, J.O.; Gorb, S.N. Material gradients in gastropod radulae and their biomechanical significance: A combined approach on the paludomid Lavigeria grandis. Sci. Nat. 2022, 109, 52. [Google Scholar]
- Krings, W.; Wägele, H.; Neumann, C.; Gorb, S.N. 2023. Coping with abrasive food: Diverging composition of radular teeth in two Porifera-consuming nudibranch species (Mollusca, Gastropoda). J. R. Soc. Interface 2023, 20, 20220927. [Google Scholar] [CrossRef]
- Krings, W.; Gorb, S.N. Particle binding capacity of snail saliva. J. Chem. Phys. 2023, 159, 185101. [Google Scholar] [CrossRef] [PubMed]
- Krings, W.; Below, P.; Gorb, S.N. Mandible mechanical properties and composition of the larval Glossosoma boltoni (Trichoptera, Insecta). Sci. Rep. 2024, 14, 4695. [Google Scholar]
- Hackethal, S.; Schulz-Kornas, E.; Gorb, S.N.; Krings, W. Wear patterns of radular teeth in Loligo vulgaris (Cephalopoda; Mollusca) are related to their structure and mechanical properties. Interface Focus 2024, 14, 20230082. [Google Scholar] [CrossRef]
- Lehnert, M.S. Metals and Their Functional Role in the Structures of Invertebrates; Springer Nature: Berlin/Heidelberg, Germany, 2024; Volume 18. [Google Scholar]
- Nikitenko, E.; Vortsepneva, E. Ontogenetic dynamics of the nudibranch epithelium in Onchidoris muricata (OF Müller, 1776). Zoology 2023, 161, 126129. [Google Scholar]
- Woodland, W. Studies in spicule formation. J. Cell Sci. 1907, 2, 31–44. [Google Scholar]
- Odum, H.T. Nudibranch spicules made of amorphous calcium carbonate. Science 1951, 114, 395. [Google Scholar] [PubMed]
- Cattaneo-Vietti, R.; Angelini, S.; Gaggero, L.; Lucchetti, G. Mineral composition of nudibranch spicules. J. Molluscan Stud. 1995, 61, 331–337. [Google Scholar]
- Weiner, S.; Levi-Kalisman, Y.; Raz, S.; Addadi, L. Biologically formed amorphous calcium carbonate. Connect. Tissue Res. 2003, 44, 214–218. [Google Scholar]
- Goldstein, J.I.; Newbury, D.E.; Michael, J.R.; Ritchie, N.W.; Scott, J.H.J.; Joy, D.C. Scanning Electron Microscopy and X-Ray Microanalysis; Springer: Berlin/Heidelberg, Germany, 2017. [Google Scholar]
- Wickham, H.; François, R.; Henry, L.; Müller, K.; Vaughan, D. dplyr: A Grammar of Data Manipulation. R Package Version 1.1.4. 2023. Available online: https://CRAN.R-project.org/package=dplyr (accessed on 12 February 2025).
- Greenacre, M. Compositional Data Analysis in Practice; Chapman and Hall/CRC: Boca Raton, FL, USA, 2018. [Google Scholar]
- van den Boogaart, K.G.; Tolosana-Delgado, R.; Bren, M. Compositions: Compositional Data Analysis. R Package Version 2.0-8. 2024. Available online: https://CRAN.R-project.org/package=compositions (accessed on 12 February 2025).
- Kassambara, A.; Mundt, F. factoextra: Extract and Visualize the Results of Multivariate Data Analyses. R Package Version 1.0.7. 2020. Available online: https://CRAN.R-project.org/package=factoextra (accessed on 12 February 2025).
- Lenth, R. emmeans: Estimated Marginal Means, aka Least-Squares Means_. R Package Version 1.10.6. 2024. Available online: https://CRAN.R-project.org/package=emmeans (accessed on 12 February 2025).
- Aitchison, J. The statistical analysis of compositional data. J. R. Stat. Soc. Ser. B Methodol. 1982, 44, 139–160. [Google Scholar]
- van den Boogaart, K.G.; Tolosona-Delgado, R. Analyzing Compositional Data with R; Springer: Berlin/Heidelberg, Germany, 2013. [Google Scholar]
- Filzmoser, P.; Hron, K.; Templ, M. Applied Compositional Data Analysis; Springer: Cham, Switzerland, 2018. [Google Scholar]
- Garrone, R.; Simpson, T.L.; Pottu-Boumendil, J.; Volcani, B.E. Silicon and Siliceous Structures in Biological Systems; Springer: New York, NY, USA, 1981; pp. 495–525. [Google Scholar]
- Lowenstam, H.A.; Weiner, S. Mineralization by organisms and the evolution of biomineralization. In Biomineralization and Biological Metal Accumulation: Biological and Geological Perspectives Papers presented at the Fourth International Symposium on Biomineralization, Renesse, The Netherlands, 2–5 June 1982; Springer: Berlin/Heidelberg, Germany, 1983; pp. 191–203. [Google Scholar]
- Kasamesiri, P.; Meksumpun, S.; Meksumpun, C. Spicule network patterns of Phyllidia varicosa. ScienceAsia 2011, 37, 160. [Google Scholar]
- Lowenstam, H.A.; Weiner, S. On Biomineralization; Oxford University Press: New York, NY, USA; London, UK, 1989; 336p. [Google Scholar]
- Molnár, Z.; Dódony, I.; Pósfai, M. Transformation of amorphous calcium carbonate in the presence of magnesium, phosphate, and mineral surfaces. Geochim. Cosmochim. Acta 2023, 345, 90–101. [Google Scholar]




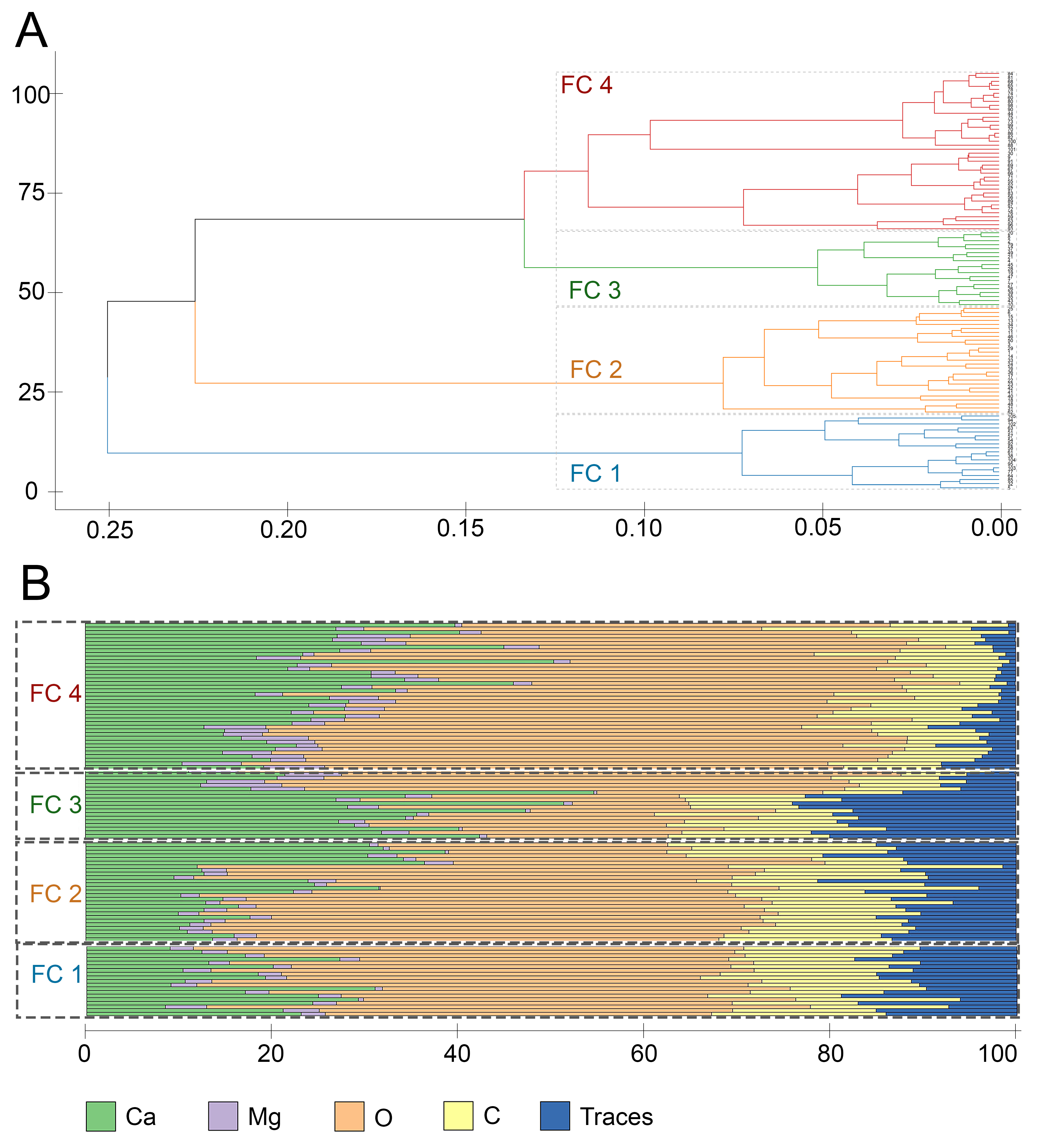
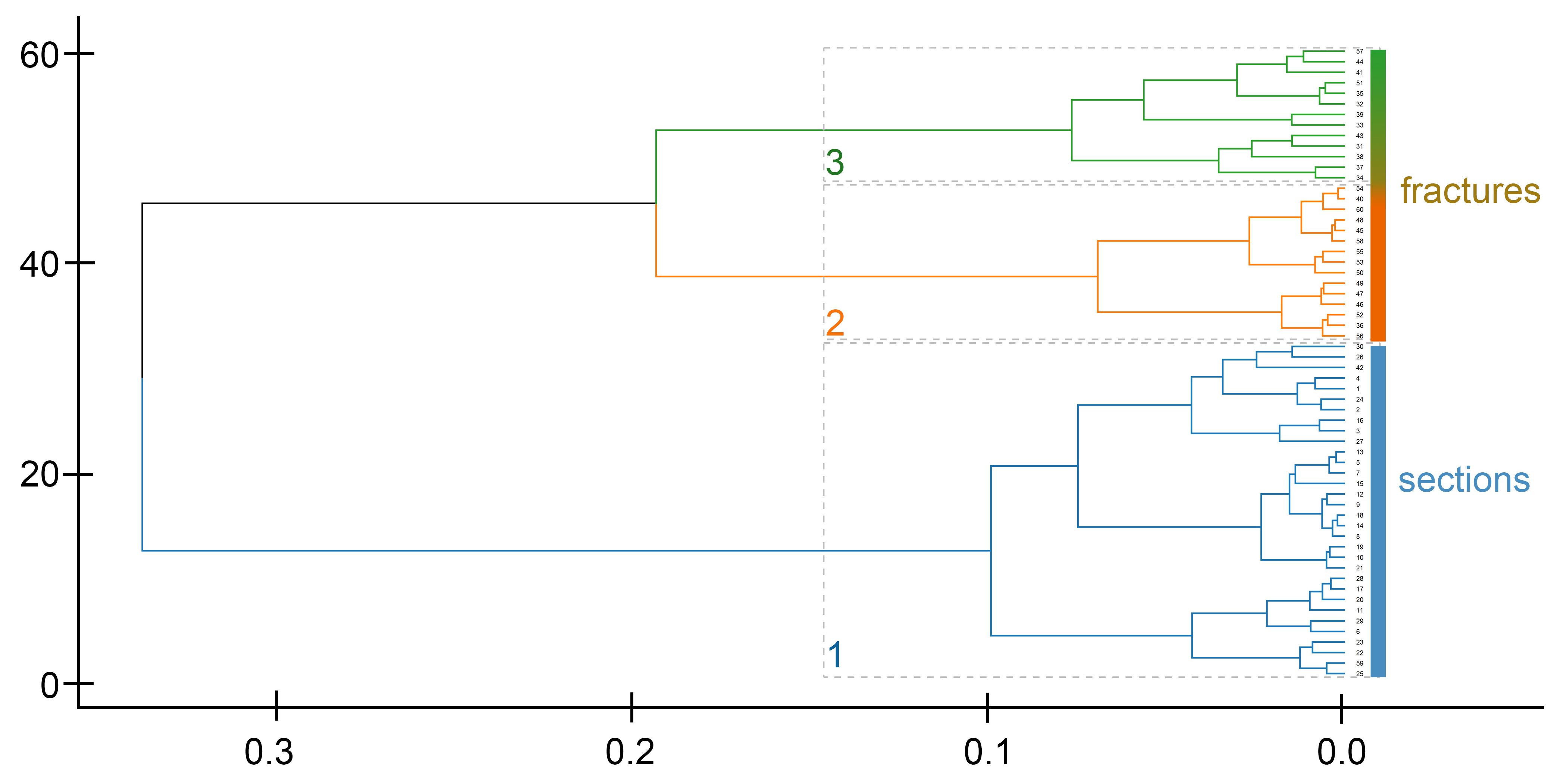

| Cluster | Number of Spicules | C | O | Ca | Mg | Traces | Total Variance |
|---|---|---|---|---|---|---|---|
| SC I | 14 | 20.07 | 46.05 | 19.80 | 1.60 | 12.48 | 0.1063 |
| SC II | 41 | 9.47 | 61.96 | 21.72 | 3.94 | 2.9 | 0.2553 |
| SC III | 45 | 8.06 | 53.49 | 33.82 | 2.88 | 1.74 | 0.2567 |
| FC I | 19 | 14.79 | 29.69 | 36.53 | 1.26 | 17.72 | 0.7958 |
| FC II | 27 | 11.58 | 53.55 | 29.59 | 3.20 | 2.07 | 0.7080 |
| FC III | 19 | 10.46 | 62.46 | 16.75 | 5.32 | 5.00 | 0.3288 |
| FC IV | 40 | 17.31 | 53.32 | 15.91 | 1.91 | 11.54 | 0.9920 |
| Ca | Mg | O | C | Traces | ||
|---|---|---|---|---|---|---|
| Ca | 1 | 7.13—12.36—23.54 | 0.29—0.43—0.57 | 0.55—0.99—1.42 | 1.21—1.59—1.89 | SC I |
| 3.02—5.51—9.08 | 0.25—0.35—0.48 | 1.12—2.29—3.99 | 2.1—7.48—17.71 | SC II | ||
| 6.36—11.74—21.67 | 0.43—0.63—0.9 | 1.94—4.2—8.82 | 7.31—19.41—45 | SC III | ||
| 8.49—29.01—160.74 | 0.77—1.23—4.12 | 1.36—2.47—6.38 | 1.21—2.06—4.49 | FC I | ||
| 3.45—9.24—56.77 | 0.29—0.56—1.48 | 1.03—2.56—9.02 | 4.84—14.28—79.89 | FC II | ||
| 1.42—3.15—9.82 | 0.16—0.27—0.40 | 0.85—1.60—3.04 | 1.30—3.35—8.17 | FC III | ||
| 1.91—8.32—597.5 | 0.13—0.3—0.73 | 0.40—0.92—2.12 | 0.86—1.38—8.48 | FC IV | ||
| Mg | 0.04—0.08—0.14 | 1 | 0.02—0.03—0.04 | 0.05—0.08—0.1 | 0.08—0.13—0.2 | SC I |
| 0.11—0.18—0.33 | 0.04—0.06—0.11 | 0.14—0.42—0.78 | 0.58—1.36—2.19 | SC II | ||
| 0.05—0.09—0.18 | 0.04—0.05—0.08 | 0.14—0.36—0.65 | 0.64—1.65—3.85 | SC III | ||
| 0.01—0.03—0.12 | 0.01—0.04—0.11 | 0.02—0.09—0.35 | 0.02—0.07—0.26 | FC I | ||
| 0.02—0.11—0.29 | 0.02—0.06—0.16 | 0.06—0.28—0.79 | 0.63—1.54—3.53 | FC II | ||
| 0.10—0.32—0.71 | 0.04—0.09—0.14 | 0.23—0.51—1.00 | 0.27—1.06—2.04 | FC III | ||
| 0.002—0.12—0.52 | 0.0003—0.06—0.09 | 0.0006—0.11—0.3 | 0.01—0.17—0.6 | FC IV | ||
| O | 1.75—2.33—3.42 | 23.84—28.76—41.19 | 1 | 1.71—2.29—2.69 | 2.98—3.69—5 | SC I |
| 2.09—2.85—3.94 | 8.99—15.73—25.53 | 2.97—6.55—9.44 | 5.2—21.34—41.01 | SC II | ||
| 1.11—1.58—2.35 | 12.—18.58—26.7 | 3.55—6.64—12.12 | 13.07—30.7—86.61 | SC III | ||
| 0.24—0.81—1.3 | 9.23—23.58—82.79 | 0.99—2.01—4.52 | 0.52—1.68—3.52 | FC I | ||
| 0.68—1.81—3.48 | 6.12—16.73—65.83 | 1.9—4.63—9.02 | 8.72—25.84—58.33 | FC II | ||
| 2.49—3.73—6.36 | 7.11—11.73—24.45 | 4.23—5.97—8.54 | 6.13—12.48—26.96 | FC III | ||
| 1.36—3.35—7.68 | 11.49—27.88—2854.5 | 1.93—3.08—4.43 | 1.81—4.62—40.49 | FC IV | ||
| C | 0.70—1.01—1.81 | 9.68—12.54—20.16 | 0.37—0.44—0.58 | 1 | 1.23—1.61—2.2 | SC I |
| 0.25—0.43—0.89 | 1.29—2.4—7.2 | 0.11—0.15—0.34 | 0.78—3.27—10.75 | SC II | ||
| 0.11—0.24—0.52 | 1.53—2.8—6.94 | 0.08—0.15—0.28 | 2.12—4.62—12.81 | SC III | ||
| 0.16—0.4—0.74 | 2.86—11.75—64.22 | 0.22—0.5—1.01 | 0.46—0.83—1.72 | FC I | ||
| 0.11—0.39—0.97 | 1.27—3.62—18.13 | 0.11—0.22—0.53 | 1.68—5.59—23.83 | FC II | ||
| 0.33—0.63—1.17 | 1.00—2.00—4.32 | 0.12—0.17—0.24 | 1.08—2.09—4.18 | FC III | ||
| 0.47—1.09—2.47 | 3.36—9.05—1476.0 | 0.22—0.32—0.52 | 0.61—1.5—20.97 | FC IV | ||
| Traces | 0.53—0.63—0.82 | 5.05—7.79—13.11 | 0.20—0.27—0.34 | 0.45—0.62—0.81 | 1 | SC I |
| 0.06—0.13—0.48 | 0.46—0.74—1.73 | 0.02—0.05—0.19 | 0.09—0.31—1.29 | SC II | ||
| 0.02—0.05—0.14 | 0.26—0.61—1.57 | 0.01—0.03—0.08 | 0.08—0.21—0.47 | SC III | ||
| 0.22—0.49—0.83 | 3.78—14.08—40.9 | 0.28—0.6—1.92 | 0.58—1.2—2.16 | FC I | ||
| 0.01—0.07—0.21 | 0.28—0.65—1.59 | 0.02—0.04—0.11 | 0.04—0.18—0.6 | FC II | ||
| 0.12—0.3—0.77 | 0.49—0.94—3.71 | 0.04—0.08—0.16 | 0.24—0.48—0.93 | FC III | ||
| 0.12—0.73—1.16 | 1.66—6.04—70.5 | 0.02—0.22—0.55 | 0.05—0.67—1.64 | FC IV |
| Ca | Mg | O | C | ||
|---|---|---|---|---|---|
| Mg | 0.1419 | SC I | |||
| 0.0886 | SC II | ||||
| 0.0819 | SC III | ||||
| 0.9440 | FC I | ||||
| 0.4438 | FC II | ||||
| 0.2235 | FC III | ||||
| 1.1959 | FC IV | ||||
| O | 0.0621 | 0.0259 | SC I | ||
| 0.0315 | 0.0554 | SC II | |||
| 0.0402 | 0.0586 | SC III | |||
| 0.2007 | 0.5723 | FC I | |||
| 0.1529 | 0.2652 | FC II | |||
| 0.0812 | 0.0863 | FC III | |||
| 0.2721 | 0.8717 | FC IV | |||
| C | 0.0912 | 0.0408 | 0.0177 | SC I | |
| 0.1026 | 0.1568 | 0.0723 | SC II | ||
| 0.1020 | 0.1176 | 0.1002 | SC III | ||
| 0.2209 | 0.8347 | 0.2060 | FC I | ||
| 0.2943 | 0.5389 | 0.2017 | FC II | ||
| 0.1248 | 0.1246 | 0.0369 | FC III | ||
| 0.2064 | 1.0989 | 0.0402 | FC IV | ||
| Traces | 0.0242 | 0.0680 | 0.0204 | 0.0391 | SC I |
| 0.2225 | 0.1005 | 0.2093 | 0.2370 | SC II | |
| 0.2162 | 0.1174 | 0.2242 | 0.2252 | SC III | |
| 0.1204 | 0.5289 | 0.1899 | 0.1610 | FC I | |
| 0.5520 | 0.2449 | 0.3380 | 0.5080 | FC II | |
| 0.2588 | 0.2654 | 0.2514 | 0.1912 | FC III | |
| 0.2885 | 0.3584 | 0.2756 | 0.3522 | FC IV |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ozerov, D.A.; Nikitenko, E.D.; Piryazev, A.A.; Lavrov, A.I.; Vortsepneva, E.V. Mineral Composition of Skeletal Elements in Dorid Nudibranchia Onchidoris muricata (Gastropoda, Mollusca). Biomimetics 2025, 10, 211. https://doi.org/10.3390/biomimetics10040211
Ozerov DA, Nikitenko ED, Piryazev AA, Lavrov AI, Vortsepneva EV. Mineral Composition of Skeletal Elements in Dorid Nudibranchia Onchidoris muricata (Gastropoda, Mollusca). Biomimetics. 2025; 10(4):211. https://doi.org/10.3390/biomimetics10040211
Chicago/Turabian StyleOzerov, Dmitry A., Ekaterina D. Nikitenko, Alexey A. Piryazev, Andrey I. Lavrov, and Elena V. Vortsepneva. 2025. "Mineral Composition of Skeletal Elements in Dorid Nudibranchia Onchidoris muricata (Gastropoda, Mollusca)" Biomimetics 10, no. 4: 211. https://doi.org/10.3390/biomimetics10040211
APA StyleOzerov, D. A., Nikitenko, E. D., Piryazev, A. A., Lavrov, A. I., & Vortsepneva, E. V. (2025). Mineral Composition of Skeletal Elements in Dorid Nudibranchia Onchidoris muricata (Gastropoda, Mollusca). Biomimetics, 10(4), 211. https://doi.org/10.3390/biomimetics10040211






