Water and Sediment Quality Changes in Mangrove Systems with Shrimp Farms in the Northern Ecuadorean Coast
Abstract
:1. Introduction
2. Materials and Methods
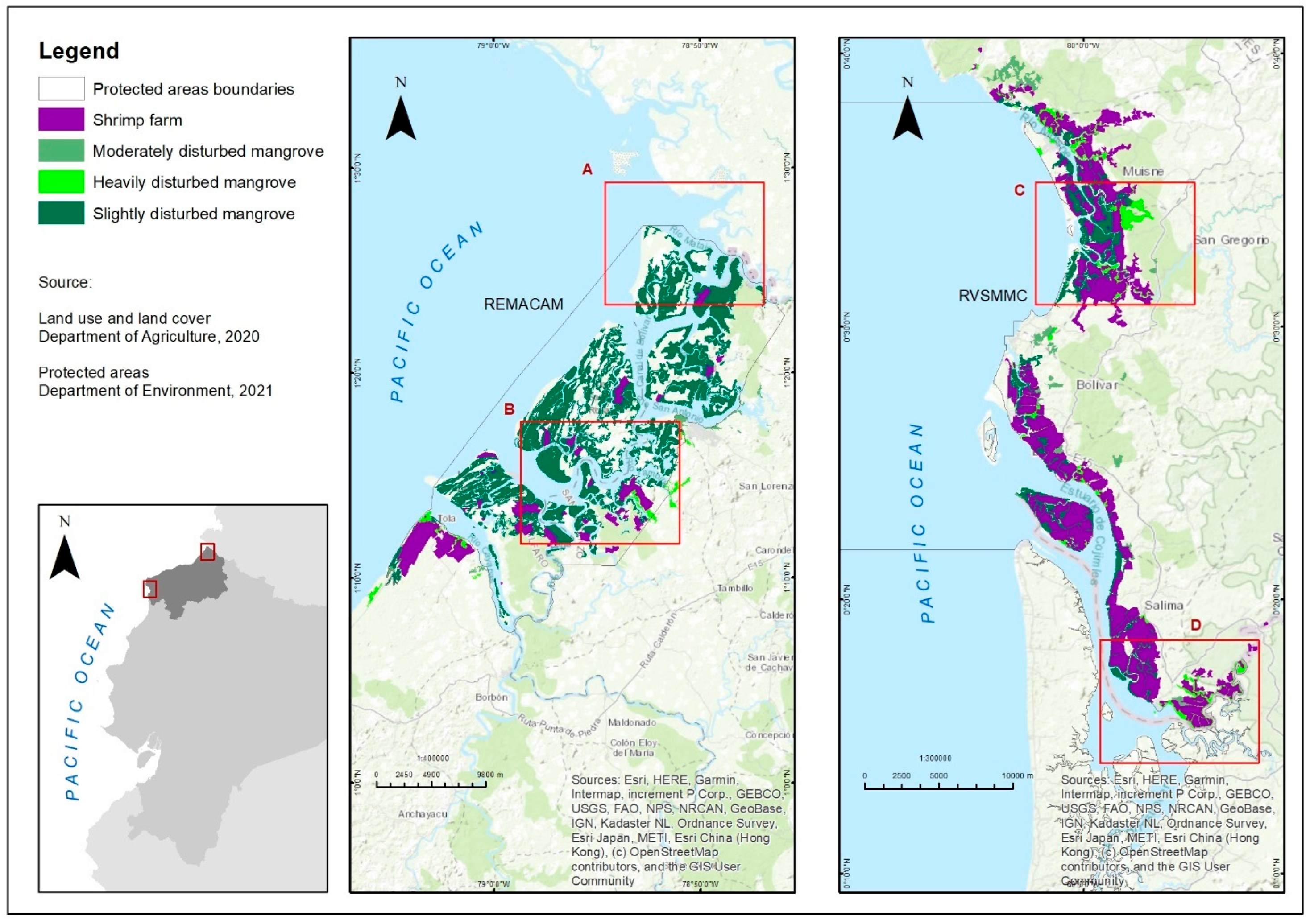
2.1. Study Area
2.2. Location of Sampling Sites
2.3. Water Analysis
2.4. Sediment Analysis
2.5. Benthic Assemblage Analysis
2.6. Data Treatment
3. Results
3.1. Water Quality
3.2. Sediment Quality
3.3. Benthic Assemblages
3.4. Multivariate Analyses
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- FAO. Ecuador. Available online: https://www.fao.org/fishery/en/countrysector/ec/en?lang=en (accessed on 21 June 2022).
- FAO. FishStatJ Version: 4.02.06. Available online: www.fao.org/fishery/statistics/software/fishstatj/es (accessed on 21 June 2022).
- Zhao, M.; Yao, D.; Li, S.; Zhang, Y.; Aweya, J.J. Effects of ammonia on shrimp physiology and immunity: A review. Rev. Aquac. 2020, 12, 2194–2211. [Google Scholar] [CrossRef]
- Sohel, S.I.; Ullah, H. Ecohydrology: A framework for overcoming the environmental impacts of shrimp aquaculture on the coastal zone of Bangladesh. Ocean Coast. Manag. 2012, 63, 67–78. [Google Scholar] [CrossRef]
- Ottinger, M.; Clauss, K.; Kuenzer, C. Aquaculture: Relevance, distribution, impacts and spatial assessments—A review. Ocean Coast. Manag. 2016, 119, 244–266. [Google Scholar] [CrossRef]
- Jayanthi, M.; Thirumurthy, S.; Muralidhar, M.; Ravichandran, P. Impact of shrimp aquaculture development on important ecosystems in India. Glob. Environ. Chang. 2018, 52, 10–21. [Google Scholar] [CrossRef]
- Treviño, M.; Murillo-Sandoval, P.J. Uneven consequences: Gendered impacts of shrimp aquaculture development on man-grove dependent communities. Ocean Coast Manag. 2021, 210, 105688. [Google Scholar] [CrossRef]
- Bunting, P.; Rosenqvist, A.; Lucas, R.M.; Rebelo, L.M.; Hilarides, L.; Thomas, N.; Hardy, A.; Itoh, T.; Shimada, M.; Finlayson, C.M. The global mangrove watch—A new 2010 global baseline of mangrove extent. Remote Sens. 2018, 10, 1669. [Google Scholar] [CrossRef] [Green Version]
- Leal, M.; Spalding, M.D. (Eds.) The State of the World’s Mangroves 2022. Global Mangrove Alliance. Available online: https://www.mangrovealliance.org/wp-content/uploads/2022/09/The-State-of-the-Worlds-Mangroves-Report_2022.pdf (accessed on 15 February 2023).
- Bhowmik, A.K.; Padmanaban, R.; Cabral, P.; Romeiras, M.M. Global Mangrove Deforestation and Its Interacting Social-Ecological Drivers: A Systematic Review and Synthesis. Sustainability 2022, 14, 4433. [Google Scholar] [CrossRef]
- CLIRSEN; PMRC. Actualización del Estudio Multitemporal de Manglares, Camaroneras y Áreas Salinas en la Costa Conti-Nental Ecuatoriana al Año 2006. 2007. Available online: http://cpps.dyndns.info/cpps-docs-web/planaccion/docs2013/manglares/Informe%20Final%20PMRC-CLIRSEN.PDF (accessed on 25 June 2018).
- CCONDEM 2020 Informe: Como la Certificación Ambiental Y Social Encubre la Violación de Derechos Humanos y de la Natura-leza en Ecuador. La Acuicultura Industrial del Camarón en el Periodo 2008–2018. Available online: https://www.wrm.org.uy/wp-content/uploads/2021/03/C-Condem-Ecuador-Certificacion-Acuacultura.pdf (accessed on 15 September 2021).
- Herbeck, L.S.; Krumme, U.; Andersen, T.J.; Jennerjahn, T.C. Decadal trends in mangrove and pond aquaculture cover on Hainan (China) since 1966: Mangrove loss, fragmentation and associated biogeochemical changes. Estuar. Coast. Shelf Sci. 2019, 233, 106531. [Google Scholar] [CrossRef]
- Santos-Andrade, M.; Hatje, V.; Arias-Ortiz, A.; Patire, V.F.; da Silva, L.A. Human disturbance drives loss of soil organic matter and changes its stability and sources in mangroves. Environ. Res. 2021, 202, 111663. [Google Scholar] [CrossRef]
- Bournazel, J.; Kumara, M.P.; Jayatissa, L.P.; Viergever, K.; Morel, V.; Huxham, M. The impacts of shrimp farming on land-use and carbon storage around Puttalam lagoon, Sri Lanka. Ocean Coast. Manag. 2015, 113, 18–28. [Google Scholar] [CrossRef] [Green Version]
- Morshed, M.; Islam, S.; Das Lohano, H.; Shyamsundar, P. Production externalities of shrimp aquaculture on paddy farming in coastal Bangladesh. Agric. Water Manag. 2020, 238, 106213. [Google Scholar] [CrossRef]
- Aldridge, D.; Salazar, M.; Serna, A.; Cock, J. Density-dependent effects of a new invasive false mussel, Mytilopsis trautwineana (Tryon 1866), on shrimp, Litopenaeus vannamei (Boone 1931), aquaculture in Colombia. Aquaculture 2008, 281, 34–42. [Google Scholar] [CrossRef]
- Khanjani, M.H.; da Silva, L.O.B.; Fóes, G.K.; Vieira, F.D.N.; Poli, M.A.; Santos, M.; Emerenciano, M.G.C. Synbiotics and aquamimicry as alternative microbial-based approaches in intensive shrimp farming and biofloc: Novel disruptive techniques or complementary management tools? A scientific-based overview. Aquaculture 2023, 567, 739273. [Google Scholar] [CrossRef]
- Camacho-Jiménez, L.; Álvarez-Sánchez, A.R.; Mejía-Ruíz, C.H. Silver nanoparticles (AgNPs) as antimicrobials in marine shrimp farming: A review. Aquac. Rep. 2020, 18, 100512. [Google Scholar] [CrossRef]
- Luu, Q.H.; Nguyen, T.B.T.; Nguyen, T.L.A.; Do, T.T.T.; Dao, T.H.T.; Padungtod, P. Antibiotics use in fish and shrimp farms in Vietnam. Aquac. Rep. 2021, 20, 100711. [Google Scholar] [CrossRef]
- Nunes, A.J.; Dalen, L.L.; Leonardi, G.; Burri, L. Developing sustainable, cost-effective and high-performance shrimp feed formulations containing low fish meal levels. Aquac. Rep. 2022, 27, 101422. [Google Scholar] [CrossRef]
- Latorre, S. Resisting environmental dispossession in Ecuador: Whom does the political category of “ancestral peoples of the mangrove ecosystem” include and aim to empower? J. Agrar. Chang. 2014, 14, 541–563. [Google Scholar] [CrossRef]
- Mialhe, F.; Gunnell, Y.; Mering, C. The impacts of shrimp farming on land use, employment and migration in Tumbes, northern Peru. Ocean Coast. Manag. 2013, 73, 1–12. [Google Scholar] [CrossRef]
- Arifanti, V.B.; Kauffman, J.B.; Hadriyanto, D.; Murdiyarso, D.; Diana, R. Carbon dynamics and land use carbon footprints in mangrove-converted aquaculture: The case of the Mahakam Delta, Indonesia. For. Ecol. Manag. 2018, 432, 17–29. [Google Scholar] [CrossRef]
- Tan, J.; Lichtfouse, E.; Luo, M.; Liu, Y.; Tan, F.; Zhang, C.; Chen, X.; Huang, J.; Xiao, L. Aquaculture drastically increases methane production by favoring acetoclastic rather than hydrogenotrophic methanogenesis in shrimp pond sediments. Aquaculture 2023, 563, 738999. [Google Scholar] [CrossRef]
- Queiroz, H.M.; Artur, A.G.; Taniguchi, C.A.K.; Silveira, S.A.K.; Nascimento, M.S.; Nóbrega, J.C.; Otero, G.N.; Ferreira, X.L.; Osorio, T. Hidden contribution of shrimp farming effluents to greenhouse gas emissions from mangrove soils. Estuar. Coast. Shelf Sci. 2019, 221, 8–14. [Google Scholar] [CrossRef]
- Thomas, Y.; Courties, C.; El Helwe, Y.; Herbland, A.; Lemonnier, H. Spatial and temporal extension of eutrophication associated with shrimp farm wastewater discharges in the New Caledonia lagoon. Mar. Pollut. Bull. 2010, 61, 387–398. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barraza-Guardado, R.H.; Arreola-Lizárraga, J.A.; Miranda-Baeza, A.; Juárez-García, M.; Juvera-Hoyos, A.; Casillas-Hernández, R. Enhancing Ecoefficiency in Shrimp Farming through Interconnected Ponds. BioMed Res. Int. 2015, 2015, 873748. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Medina-Galván, J.; Osuna-Martínez, C.C.; Padilla-Arredondo, G.; Frías-Espericueta, M.G.; Barraza-Guardado, R.H.; Arreola-Lizárraga, J.A. Comparing the biogeochemical functioning of two arid subtropical coastal lagoons: The effect of wastewater discharges. Ecosyst. Heal. Sustain. 2021, 7. [Google Scholar] [CrossRef]
- Avnimelech, Y.; Ritvo, G. Shrimp and fish pond soils: Processes and management. Aquaculture 2003, 220, 549–567. [Google Scholar] [CrossRef] [Green Version]
- Funge-Smith, S.J.; Briggs, M.R. Nutrient budgets in intensive shrimp ponds: Implications for sustainability. Aquaculture 1998, 164, 117–133. [Google Scholar] [CrossRef]
- Molnar, N.; Welsh, D.T.; Marchand, C.; Deborde, J.; Meziane, T. Impacts of shrimp farm effluent on water quality, benthic metabolism and N-dynamics in a mangrove forest (New Caledonia). Estuar. Coast. Shelf Sci. 2013, 117, 12–21. [Google Scholar] [CrossRef]
- Páez-Osuna, F. The environmental impact of shrimp aquaculture: Causes, effects, and mitigating alternatives. Environ. Manag. 2001, 28, 131–140. [Google Scholar] [CrossRef]
- Biao, X.; Ding, Z.; Wang, X. Impact of the intensive shrimp farming on the water quality of the adjacent coastal creeks from Eastern China. Mar. Pollut. Bull. 2004, 48, 543–553. [Google Scholar] [CrossRef]
- Bui, T.D.; Luong-Van, J.; Austin, C.M. Impact of shrimp farm effluent on water quality in coastal areas of the world heritage-listed Ha Long Bay. Am. J. Environ. Sci. 2012, 8, 104–116. [Google Scholar]
- Herbeck, L.S.; Unger, D.; Wu, Y.; Jennerjahn, T.C. Effluent, nutrient and organic matter export from shrimp and fish ponds causing eutrophication in coastal and back-reef waters of NE Hainan, tropical China. Cont. Shelf Res. 2013, 57, 92–104. [Google Scholar] [CrossRef]
- Cardoso-Mohedano, J.-G.; Bernardello, R.; Sanchez-Cabeza, J.-A.; Páez-Osuna, F.; Ruiz-Fernández, A.-C.; Molino-Minero-Re, E.; Cruzado, A. Reducing nutrient impacts from shrimp effluents in a subtropical coastal lagoon. Sci. Total Environ. 2016, 571, 388–397. [Google Scholar] [CrossRef]
- Barcellos, D.; Queiroz, H.M.; Nóbrega, G.N.; de Oliveira Filho, R.L.; Santaella, S.T.; Otero, X.L.; Ferreira, T.O. Phosphorus en-riched effluents increase eutrophication risks for mangrove systems in northeastern Brazil. Mar. Pollut. Bull. 2019, 142, 58–63. [Google Scholar] [CrossRef]
- Bravo Aguas, Y.M. Valoración Económica de Manglares del Sur de la Reserva REMACAM Próximos a Camaroneras Mediante el Método de Reposición de Daño. Master’s Thesis, Escuela de Gestión Ambiental, Pontificia Universidad Católica del Ecuador Sede Esmeraldas, Esmeraldas, Ecuador, 2018. [Google Scholar]
- Mohanty, R.K.; Ambast, S.; Panigrahi, P.; Mandal, K. Water quality suitability and water use indices: Useful management tools in coastal aquaculture of Litopenaeus vannamei. Aquaculture 2018, 485, 210–219. [Google Scholar] [CrossRef]
- Costa, B.G.B.; Soares, T.M.; Torres, R.F.; Lacerda, L.D. Mercury distribution in a mangrove tidal creek affected by intensive shrimp farming. Bull. Environ. Contam. Toxicol. 2013, 90, 537–541. [Google Scholar] [CrossRef]
- León-Cañedo, J.A.; Alarcón-Silvas, S.G.; Fierro-Sañudo, J.F.; Mariscal-Lagarda, M.M.; Díaz-Valdés, T.; Páez-Osuna, F. Assessment of environmental loads of Cu and Zn from intensive inland shrimp aquaculture. Environ. Monit. Assess. 2017, 189, 69. [Google Scholar] [CrossRef]
- Na Nakorn, A.; Chevakidagarn, P.; Danteravanich, S. Environmental impact of white shrimp culture during 2012–2013 at Bandon Bay, Surat Thani Province: A case study investigating farm size. Agric. Nat. Resour. 2017, 51, 109–116. [Google Scholar] [CrossRef]
- Bui, T.D.; Luong-Van, J.; Maier, S.W.; Austin, C.M. Assessment and monitoring of nutrient loading in the sediments of tidal creeks receiving shrimp farm effluent in Quang Ninh, Vietnam. Environ. Monit. Assess. 2013, 185, 8715–8731. [Google Scholar] [CrossRef]
- Nóbrega, G.N.; Ferreira, T.O.; Romero, R.E.; Marques, A.G.B.; Otero, X.L. Iron and sulfur geochemistry in semi-arid mangrove soils (Ceará, Brazil) in relation to seasonal changes and shrimp farming effluents. Environ. Monit. Assess. 2013, 185, 7393–7407. [Google Scholar] [CrossRef]
- Nuto Nóbrega, G.; Otero, X.L.; Macias, F.; Ferreira, T. Phosphorus geochemistry in a Brasilian semiarid mangrove soil af-fected by shrimp farm effluents. Environ. Monit. Assess. 2014, 186, 5749–5762. [Google Scholar] [CrossRef]
- Aschenbroich, A.; Marchand, C.; Molnar, N.; Deborde, J.; Hubas, C.; Rybarczyk, H.; Meziane, T. Spatio-temporal variations in the composition of organic matter in surface sediments of a mangrove receiving shrimp farm effluents (New Caledonia). Sci. Total Environ. 2015, 512–513, 296–307. [Google Scholar] [CrossRef] [PubMed]
- Suarez-Abelenda, M.; Ferreira, T.; Camps-Arbestain, M.; Rivera-Monroy, V.; Macias, F.; Nuto Nóbrega, G.; Otero, X.L. The effect of nutrient-rich effluents from shrimp farming on mangrove soil carbon storage and geochemistry under semi-arid climate conditions in Northern Brasil. Geoderma 2014, 213, 551–559. [Google Scholar] [CrossRef]
- Pérez, A.; Machado, W.; Gutiérrez, D.; Saldarriaga, M.S.; Sanders, C.J. Shrimp farming influence on carbon and nutrient accu-mulation within Peruvian mangroves sediments. Estuar. Coast. Shelf Sci. 2020, 243, 106879. [Google Scholar] [CrossRef]
- Queiroz, H.M.; Ferreira, T.O.; Taniguchi, C.A.K.; Barcellos, D.; Costa do Nascimento, N.; Otero, X.L.; Arthur, A.G. Nitrogen min-eralization and eutrophication risk in mangroves receiving shrimp farming effluents. Environ. Sci. Pollut. Res. 2020, 27, 34941–34950. [Google Scholar] [CrossRef]
- Hidayati, N.V.; Prudent, P.; Asis, L.; Vassalo, L.; Torre, F.; Widowati, I.; Sabdono, A.; Syakti, A.D.; Doumenq, P. Assesstment of the ecological and human health risks from metal in shrimp aquaculture environments in Central Java, Indonesia. Eviron. Sci. Pollut. Res. 2020, 27, 41668–41687. [Google Scholar] [CrossRef]
- Hong, A.H.; Hargan, K.E.; Williams, B.; Nuangsaeng, B.; Siriwong, S.; Tassawad, P.; Chaiharn, C.; Los Huertos, M. Mollusc as bioincicator of shrimp aquaculture effluent contamination in a southeast Asian mangrove. Ecol. Indic. 2020, 115, 106365. [Google Scholar] [CrossRef]
- de Lacerda, L.D.; Ward, R.D.; Godoy, M.D.P.; Meireles, A.J.D.A.; Borges, R.; Ferreira, A.C. 20-Years Cumulative Impact from Shrimp Farming on Mangroves of Northeast Brazil. Front. For. Glob. Chang. 2021, 4, 653096. [Google Scholar] [CrossRef]
- Ribeiro, L.; Eca, G.; Barros, F.; Hatje, V. Impacts of shrimp farming cultivation cycles on macrobenthic assemblages and chem-istry of sediments. Environ. Pollut. 2016, 211, 307–315. [Google Scholar] [CrossRef]
- Hatje, V.; de Souza, M.M.; Ribeiro, L.F.; Eça, G.F.; Barros, F. Detection of environmental impacts of shrimp farming through multiple lines of evidence. Environ. Pollut. 2016, 219, 672–684. [Google Scholar] [CrossRef]
- Hernandez, L.D.C. Subsecretario de Acuicultura. Oficio N° MAP-SUBACUA-2018-0392-O, Relativo a Predios ca-maroneros dirigido a la Cámara nacional de Acuacultura, Guayaquil 7 de marzo 2018.Ministerio de Acuacultura y Pesca del Ecuador, MAP 2018.
- Worthington, T.A.; Zu Ermgassen, P.S.E.; Friess, D.A.; Krauss, K.W.; Lovelock, C.E.; Thorley, J.; Tingey, R.; Woodroffe, C.D.; Bunting, P.; Cormier, N.; et al. A global biophysical typology of mangroves and its relevance for ecosystem structure and deforestation. Sci. Rep. 2020, 10, 14652. [Google Scholar] [CrossRef]
- Hamilton, S. Mangroves and Aquaculture, a Five-Decade Remote Sensing Analysis of Ecuador’s Estuarine Environmental; Coastal Reserach Library; Springer: Boca Raton, FL, USA, 2020; Volume 33, p. 211. [Google Scholar]
- Boyd, C.E.; Tucker, C.S. Water Quality and Pond Soil Analysis for Aquaculture; Alabama Agricultural Experiment Station, Auburn University: Auburn, AL, USA, 1992; p. 183. [Google Scholar]
- American Public Health Association; American Water Works Association; Water Environmental Federation. APHA Standard Methods for the Examination of Water and Wastewater, 20th ed.; American Public Health Association, American Water Works Association and Water Environmental Federation: Washington, DC, USA, 1998. [Google Scholar]
- Byers, S.C.; Mills, E.L.; Stewart, P.L. A comparison of methods of determining organic carbon in marine sediments, with suggestions for a standard method. Hydrobiologia 1978, 58, 43–47. [Google Scholar] [CrossRef]
- Borja, A.; Franco, J.; Pérez, V. A marine biotic index to establish the ecological quality of soft-bottom Bentos Within European Estuarine and coastal environments. Mar. Pollut. Bull. 2000, 40, 1100–1114. [Google Scholar] [CrossRef]
- Bald, J.; Borja, A.; Muxika, I.; Franco, J.; Valencia, V. Assessing reference conditions and physico-chemical status according to the European Water Framework Directive: A case-study from the Basque Country (Northern Spain). Mar. Pollut. Bull. 2005, 50, 1508–1522. [Google Scholar] [CrossRef]
- Muxika, I.; Borja, A.; Bald, J. Using historical data, expert judgement, and multivariate analysis in assessing reference condi-tions and benthic ecological status, according to the European Water Framework Directive. Mar. Pollut. Bull. 2007, 55, 16–29. [Google Scholar] [CrossRef]
- Royston, J.P. An extension of Shapiro and Wilk’s W test for normality to large samples. Appl. Stat. 1982, 31, 115–124. [Google Scholar] [CrossRef]
- Conover, W.; Johnson, M.E.; Johnson, M.M. A comparative study of tests for homogeneity of variances, with applications to the outer continental shelf bidding data. Technometrics 1981, 23, 351–361. [Google Scholar] [CrossRef]
- Forrest, W. Young. Scaling. Annual Review of Psychology. Aims Scope J. 1974, 35, 55–81. [Google Scholar]
- Ahsan, M.; Mahmud, M.A.P.; Saha, P.K.; Gupta, K.D.; Siddique, Z. Effect of Data Scaling Methods on Machine Learning Algorithms and Model Performance. Technologies 2021, 9, 52. [Google Scholar] [CrossRef]
- Past3x. The Past of the Future Natural History Museum and University of Oslo. 2013. Available online: https://www.nhm.uio.no/english/research/resources/past/ (accessed on 15 February 2020).
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2021; Available online: https://www.R-project.org/ (accessed on 20 March 2018).
- Reserva Ecológica Manglares Cayapas-Mataje. Available online: https://www.ambiente.gob.ec/reserva-ecologica-manglares-cayapas-mataje/ (accessed on 10 March 2023).
- Inpectoria de Acuicultura; (Base de Datos de Predios Camaroneros de la Provincia de Esmeraldas, Esmeraldas, Ecuador). Personal Communication, 2019.
- Refúgio de Vida Silvestre Manglares Rio Muisne. Available online: http://areasprotegidas.ambiente.gob.ec/es/areas-protegidas/refugio-de-vida-silvestre-manglar-el-estuario-del-r%C3%ADo-muisne (accessed on 10 March 2023).
- Ofori, S.A.; Kodikara, S.K.A.; Jayatissa, L.P.; Madarasinghe, S.K.; Mafaziya Nijamdeen, T.W.G.F.; Dahdouh-Guebas, F. What is the ecological footprint of aquaculture after 5 decades of competition between mangrove conservation and shrimp farm de-velopment. Aquat. Conserv. Mar. Freshw. Ecosyst. 2023, 33, 15–28. [Google Scholar] [CrossRef]
- Hamilton, S.E.; Lovette, J. Ecuador’s Mangrove Forest carbon Stocks: A Spatiotemporal Analysis of Living Carbon Holdings and Their depletion since the Asvent of Commercial Aquaculture. PLoS ONE 2015, 10, e0118880. [Google Scholar] [CrossRef]
- Wu, R. The environmental impact of marine fish culture: Towards a sustainable future. Mar. Pollut. Bull. 1995, 31, 159–166. [Google Scholar] [CrossRef]
- Jones, A.; O’Donohue, M.; Udy, J.; Dennison, W. Assessing ecological impacts of shrimp and sewage effluent: Biological indicators with standard water quality analyses. Estuar. Coast. Shelf Sci. 2001, 52, 91–109. [Google Scholar] [CrossRef]
- Alongi, D.M.; Boto, K.G.; Robertson, A.I. Nitrogen and phosphorus cycles. In Tropical Mangrove Ecosystems; Robertson, A.I., Alongi, D.M., Eds.; Coastal and estuarine studies 41; AGU: Washington, DC, USA, 1992; pp. 251–292. [Google Scholar]
- Rao, K.; Ramanathan, A.; Raju, N.J. Assessment of Blue Carbon Stock of Coringa mangroves: Climate change Perspective. J. Clim. Chang. 2022, 8, 41–58. [Google Scholar] [CrossRef]
- Adame, M.F.; Connolly, R.M.; Turschwell, M.P.; Lovelock, C.E.; Fatoyinbo, T.; Lagomasino, D.; Goldberg, L.A.; Holdorf, J.; Friess, D.A.; Sasmito, S.D.; et al. Future carbon emissions from global mangrove forest loss. Glob. Chang. Biol. 2021, 27, 2856–2866. [Google Scholar] [CrossRef] [PubMed]
- Donato, D.C.; Kauffman, J.B.; Murdiyarso, D.; Kurnianto, S.; Stidham, M.; Kanninen, M. Mangroves among the most carbon-rich forests in the tropics. Nat. Geosci. 2011, 4, 293–297. [Google Scholar] [CrossRef]
- Acuerdo 097-A, Anexo 1, Libro VI del Texto Unificado de Legislación Secundaria del Ministerio del Ambiente: Norma de Calidad Ambiental y de Descarga de Efluentes Recurso Agua. Available online: https://www.gob.ec/sites/default/files/regulations/2018-09/Documento_Registro-Oficial-No-387-04-noviembre-2015_0.pdf (accessed on 24 April 2021).
- Hargrave, B.T.; Phillips, G.A.; Doucette, L.I.; White, M.J.; Milligan, T.G.; Wildish, D.J.; Cranston, R.-E. Assessing benthic im-pacts of organic enrichment from marine aquaculture. Water Air Soil Pollut. 1997, 99, 641–650. [Google Scholar] [CrossRef]
- Hargrave, B.T.; Holmer, M.; Newcombe, C.P. Towards a classification of organic enrichment in marine sediments based on biochemical indicators. Mar. Pollut. Bull. 2008, 56, 810–824. [Google Scholar] [CrossRef]
- Normativa Sectorial: Reglamento Ambiental para la Acuicultura, Gobierno de Chile 2016. Available online: https://mma.gob.cl/wp-content/uploads/2017/09/16_Susana-Giglio_Subpesca.pdf (accessed on 24 December 2022).
- Breithaupt, L.; Steinmuller, H.E. Refining the Global Estimate of Mangrove Carbon Burial Rates Using Sedimentary and Geo-Morphic Settings. Geophys. Res. Lett. 2022, 49, 100177. [Google Scholar] [CrossRef]
- Buringh, P. Organic carbon in soils of the world. In The Role of Terrestrial Vegetation in the Global Carbon Cycle: Measurement by Remote Sensing; Woodwell, G.M., Ed.; John Wiley & Sons Ltd.: New York, NY, USA, 1984; pp. 91–109. [Google Scholar]
- Eid, E.M.; Arshad, M.; Shaltour, K.H.; El-Sheik, M.A.; Alfarhan, A.H.; Píco, Y.; Barcelo, D. Effect of the conversion of mangroves into shrimp farms on carbon stock in the sediment along the southern Red Sea coast, Saudi Arabia, 2019. Environ. Res. 2019, 176, 108536. [Google Scholar] [CrossRef]
- Merecí-Guamán, J.; Casanoves, F.; Delgado-Rodríguez, D.; Ochoa, P.; Cifuentes-Jara, M. Impact of shrimp ponds on Mangrove Blue Carbon Stocks in Ecuador. Forests 2021, 12, 816. [Google Scholar] [CrossRef]
- Tian, Y.; Chen, G.; Lu, H.; Zhu, H.; Ye, Y. Effects of shrimp ponds on stocks of organic carbon, nitrogen and phosphorus in soils of Kandelia obovata forest along Jiulong River Estuary. Mar. Pollut. Bull. 2019, 149, 110657. [Google Scholar] [CrossRef]
- Loiola, M.; Silva, A.E.T.; Krull, M.; Barbosa, F.A.; Galvão, E.H.; Patire, V.F.; Cruz, I.C.S.; Barros, F.; Hatje, V.; Meirelles, P.M. Mangrove microbial community recovery and their role in early stages of forest recolonization within shrimp ponds. Sci. Total. Environ. 2023, 855, 158863. [Google Scholar] [CrossRef]
- Adame, M.F.; Zakaria, R.M.; Fry, B.; Chong, V.C.; Then, Y.H.A.; Brown, C.J.; Lee, S.Y. Loss and recovery of carbon and nitrogen after mangrove clearing. Ocean. Coast. Manag. 2018, 161, 117–126. [Google Scholar] [CrossRef]
- Chinacalle-Martínez, N.; García-Rada, E.; López-Macías, J.; Pinoargote, S.; Loor, G.; Zevallos-Rosado, J.; Cruz, P.; Andrade, D.P.B.; Robalino-Mejia, C.; Añazco, F.; et al. Oceanic primary production trend pat-terns along coast of Ecuador. Neotrop. Biodivers. 2021, 7, 379–391. [Google Scholar] [CrossRef]
- Feebarani, J.; Joydas, T.; Damodaran, R.; Borja, A. Benthic quality assessment in a naturally- and human-stressed tropical estuary. Ecol. Indic. 2016, 67, 380–390. [Google Scholar] [CrossRef]
- Kamalifar, R.; Aeinjamshid, K.; Nurinejad, M.; Dehghan-Mediseh, S.; Vazirizadeh, A. Ecological status assessment of Bidkhun mangrove swamp from Bushehr province, Persian Gulf, using macrofauna community structure. AACL Aquac. Aquar. Conserv. Legis. 2016, 9, 8–19. [Google Scholar]
- Tweedley, J.R.; Warwick, R.M.; Robert Clarke, K.; Potter, I.C. Family-level AMBI is valid for use in the north-eastern Atlantic but not for assesing the health of microtidal Australian estuaries. Estuar. Coast. Shelf Sci. 2014, 141, 85–96. [Google Scholar] [CrossRef] [Green Version]
- Perfil para la zona del Estuario de Cojimíes. Centro Regional para el Manejo de Ecosistemas Costeros, EcoCostas. Agosto 2005. USAID. Available online: https://www.crc.uri.edu/download/Cojimies_final-1.pdf (accessed on 3 March 2023).
- Navarro, E. Ecuador: La codicia camaronera en Cojimíes. Notícias de Navarra. 2022. Available online: https://www.noticiasdenavarra.com/mundo/2022/09/25/ecuador-codicia-camaronera-cojimies-6026032.html (accessed on 3 March 2023).
- Reglamento General a la ley Orgánica para el Desarrollo de la Acuicultura y Pesca. Available online: https://www.produccion.gob.ec/wp-content/uploads/downloads/2022/03/Decreto-Ejecutivo-No.-362-Reglamento-General-a-la-Ley-Organica-para-el-Desarrollo-de-la-Acuicultura-y-Pesca.pdf (accessed on 6 June 2022).
- Gautier, D. The Integration of Mangrove and Shrimp Farming: A Case Study on the Caribbean Coast of Colombia; Department of fisheries and Allied Aquacultures, Auburn University, Alabama 36849 USA. In Report Prepared for the World Bank, Network of Aquaculture Centres in Asia-Pacific, World Wildlife Foundation and FAO Consortium Program on Shrimp Farming and the Environment; Work in Progress for Public Discussion; The Consortium: Singapore, 2002; p. 26. [Google Scholar]
- Primavera, J.; Altamirano, J.; Lebata, M.J.H.L.; de los Reyes, A.A., Jr.; Pitogo, C.L. Mangroves and shrimp pond culture effluents in Aklan, Panay Is., central Philippines. Bull. Mar. Sci. 2007, 80, 795–804. [Google Scholar]
- Zaldivar-Jimenez, A.; Herrera-Silveira, J.; Perez-Ceballos, R.; Teutli-Hernandez, C. Evaluación del uso de humedales de manglar como biofiltro de efluentes de camaroneras en Yucatán, México. Rev. Biol. Mar. Oceanogr. 2012, 47, 395–405. [Google Scholar] [CrossRef]
- Colette, M.; Guentas, L.; Gunkel-Grillon, P.; Callac, N.; Della Patrona, L. Is halophyte species growing in the vicinity of the shrimp ponds a promising agri-aquaculture system for shrimp ponds remediation in New Caledonia? Mar. Pollut. Bull. 2022, 177, 113563. [Google Scholar] [CrossRef]








| Parameter | Global Mean | SECTORS | SYSTEM | TYPE | SEASON | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| PR | TA | MU | CO | REM | RVS | SF | MG | SU | WI | ||
| Temperature (°C) | 27.07 ± 1.39 | 27.85 ± 0.62 a | 28.25 ± 0.95 a | 26.34 ± 1.14 b | 25.85 ± 1.18 b | 28.05 ± 0.81 a | 26.09 ± 1.15 b | 26.63 ± 1.48 b | 27.73 ± 0.96 a | 26.53 ± 1.54 | 27.62 ± 0.99 |
| Salinity (ppt) | 15.82 ± 7.29 | 17.07 ± 6.81 | 13.23 ± 1.07 | 15.51 ± 11.07 | 17.49 ± 6.92 | 15.15 ± 5.14 | 16.50 ± 9.04 | 15.91 ± 8.31 | 15.69 ± 5.68 | 16.72 ± 7.70 | 14.93 ± 6.94 |
| Dissolved oxigen * | 4.78 ± 1.36 | 4.62 ± 1.17 | 4.73 ± 1.31 | 5.18 ± 1.38 | 4.59 ± 1.66 | 4.68 ± 1.21 | 4.89 ± 1.52 | 5.13 ± 1.35 a | 4.26 ± 1.25 b | 5.17 ± 1.37 | 4.39 ± 1.27 |
| Oxigen saturation % | 60.30 ± 17.15 | 59.71 ± 15.68 | 60.65 ± 16.68 | 64.13 ± 17.55 | 56.72 ± 20.29 | 60.04 ± 17.05 | 58.71 ± 21.30 | 64.91 ± 19.02 | 55.14 ± 17.86 | 65.44 ± 18.45 a | 51.68 ± 16.98 b |
| DBO5 * | 11.01 ± 5.61 | 9.21 ± 3.47 a,b | 14.46 ± 8.46 a | 12.16 ± 4.22 a,b | 8.21 ± 2.75 b | 11.84 ± 6.85 | 10.18 ± 4.02 | 11.73 ± 5.56 | 9.93 ± 5.67 | 13.65 ± 6.49 a | 8.36 ± 2.74 b |
| Electrical conductivity (µS/cm) | 27.53 ± 10.89 | 29.55 ± 11.32 | 23.22 ± 2.24 | 28.33 ± 15.99 | 29.26 ± 11.87 | 26.38 ± 8.58 | 28.88 ± 13.24 | 27.79 ± 12.06 | 27.18 ± 9.47 | 29.85 ± 9.75 | 25.55 ± 11.63 |
| REDOX (mV) | 132.59 ± 60.29 | 112.11 ± 47.40 | 133.08 ± 90.40 | 110.12 ± 32.80 | 175.04 ± 35.01 | 122.59 ± 71.07 | 142.58 ± 46.90 | 144.21 ± 43.27 | 115.15 ± 77.71 | 120.28 ± 74.02 | 144.90 ± 40.77 |
| pH | 7.32 ± 0.49 a,b | 6.90 ± 0.52 b | 7.74 ± 0.16 a | 7.46 ± 0.36 a | 7.14 ± 0.53 b | 7.60 ± 0.31 a | 7.52 ± 0.40 a | 7.23 ± 0.50 b | 7.75 ± 0.17 a | 7.08 ± 0.44 b | |
| Ammonium * | 0.08 ± 0.07 | 0.05 ± 0.04 | 0.05 ± 0.04 | 0.11 ± 0.06 | 0.10 ± 0.10 | 0.05 ± 0.04 b | 0.11 ± 0.08 a | 0.10 ± 0.07 a | 0.04 ± 0.04 b | 0.10 ± 0.06 a | 0.06 ± 0.08 b |
| Nitrate * | 0.12 ± 0.16 | 0.03 ± 0.03 b | 0.05 ± 0.06 b | 0.14 ± 0.10 a,b | 0.270 ± 0.24 a | 0.04 ± 0.05 b | 0.21 ± 0.19 a | 0.15 ± 0.17 | 0.08 ± 0.14 | 0.14 ± 0.18 | 0.11 ± 0.14 |
| Nitrite * | 0.06 ± 0.09 | 0.02 ± 0.01 b | 0.06 ± 0.08 a,b | 0.02 ± 0.01 b | 0.13 ± 0.13 a | 0.04 ± 0.06 | 0.08 ± 0.11 | 0.06 ± 0.08 | 0.05 ± 0.09 | 0.04 ± 0.08 | 0.07 ± 0.09 |
| Total Nitrogen * | 4.31 ± 1.69 | 4.10 ± 1.51 a,b | 2.83 ± 0.34 b | 4.57 ± 1.85 a | 5.73 ± 1.33 a | 3.46 ± 1.25 b | 5.15 ± 1.68 a | 4.64 ± 1.83 | 3.80 ± 1.35 | 3.83 ± 1.26 | 4.79 ± 1.95 |
| Phosphate* | 0.12 ± 0.07 | 0.09 ± 0.02 b | 0.07 ± 0.03 b | 0.20 ± 0.08 a | 0.12 ± 0.04 b | 0.08 ± 0.03 b | 0.16 ± 0.07 a | 0.14 ± 0.08 a | 0.09 ± 0.03 b | 0.13 ± 0.09 | 0.11 ± 0.04 |
| Total Phosphorus * | 0.11 ± 0.11 | 0.04 ± 0.03 | 0.09 ± 0.14 | 0.16 ± 0.06 | 0.16 ± 0.15 | 0.07 ± 0.10 b | 0.16 ± 0.11 a | 0.15 ± 0.13 a | 0.06 ± 0.06 b | 0.11 ± 0.13 | 0.11 ± 0.10 |
| Chlorophyll a (µg/L) | 25.72 ± 12.95 | 18.16 ± 7.82 | 26.66 ± 15.17 | 26.59 ± 9.53 | 27.47 ± 16.76 | 22.41 ± 12.53 | 27.03 ± 13.27 | 29.27 ± 13.32 a | 17.89± 9.01 b | 22.67 ± 10.29 | 26.77 ± 15.16 |
| Settleable solids * | 0.14 ± 0.40 | 0.10 ± 0.25 | 0.10 ± 0.12 | 0.06 ± 0.10 | 0.31± 0.74 | 0.10 ± 0.19 | 0.19 ± 0.53 | 0.18 ± 0.48 | 0.08 ± 0.20 | 0.25 ± 0.54 | 0.04 ± 0.08 |
| Total suspended solids * | 51.08 ± 40.06 | 59.61 ± 43.41 | 33.46 ± 19.30 | 65.76 ± 54.18 | 39.92 ± 17.51 | 46.53 ± 35.34 | 57.15 ± 46.19 | 54.03 ± 41.24 | 47.14 ± 3 | 73.86 ± 50.98 a | 34 ± 15.47 b |
| Parameter | Global Mean | SECTOR | SYSTEM | TYPE | SEASON | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| PR | TA | MU | CO | REM | RVS | SF | MG | SU | WI | ||
| Electrical conductivity µS/cm | 7973 ± 3177 | 8563 ± 2747 | 7204 ± 1470 | 7344 ± 4373 | 8779 ± 3564 | 7883 ± 2255 | 8062 ± 3952 | 7772 ± 3626 | 8273 ± 2433 | 8108 ± 3050 | 7837 ± 3373 |
| REDOX mV | 3.29 ± 118.25 | 22.80 ± 101.17 | 32.29 ± 107.84 | 11.04 ± 142.76 | −52.96 ± 115.78 | 27.55 ± 101.89 | −20.96 ± 130.70 | −7.29 ± 124.04 | 19.17 ± 110.96 | 29.19 ± 100.63 | −22.61 ± 131 |
| pH | 6.72 ± 0.46 | 6.30 ± 0.39 b | 6.67 ± 0.51 a,b | 7.10 ± 0.35 a,b | 6.98 ± 0.19 a | 6.49 ± 0.48 b | 6.92 ± 0.52 a | 6.87 ± 0.52 a | 6.45 ± 0.47 b | 6.49 ± 0.61 b | 6.91 ± 0.37 a |
| Ammonium * | 6.91 ± 6.03 | 4.45 ± 2.87 b | 4.85 ± 3.96 b | 5.250 ± 3.92 b | 13.10 ± 7.82 a | 4.66 ± 3.37 b | 9.18 ± 7.24 a | 8.56 ± 7.07 a | 4.43 ± 2.63 b | 6.62 ± 6.72 | 7.20 ± 5.41 |
| Total Nitrogen * | 2052 ± 1213 | 3325 ± 981 a | 2189 ± 963 b | 1179 ± 755 b | 1516 ± 967 b | 2757 ± 1111 a | 1347 ± 861 b | 1567 ± 962 b | 2779 ± 1211 a | 1586 ± 1302 b | 2519 ± 931 a |
| Phosphate * | 4.30 ± 1.42 | 4.15 ± 1.39 | 4.5 ± 1.05 | 3.85 ± 1.92 | 4.70 ± 1.30 | 4.33 ± 1.22 | 4.28 ± 1.63 | 4.29 ± 1.54 | 4.31 ± 1.25 | 4.30 ± 1.23 | 4.30 ± 1.62 |
| TOM % | 15.25 ± 6.73 | 19.38 ± 4.57 a | 17.07 ± 10.17 a,b | 11.13 ± 3.66 b | 13.42 ± 3.58 a b | 18.23 ± 7.77 a | 12.27 ± 3.72 b | 11.74 ± 4.50 b | 20.53 ± 6.10 a | 14.02 ± 6.98 | 16.49 ± 6.39 |
| Coppe r * | 34.3 ± 11.78 | 34.2 ± 12.48 | 34.2 ± 15.51 | 33.9 ± 12.68 | 34.9 ± 6.57 | 34.2 ± 13.71 | 34.4 ± 9.85 | 35.75 ± 11.70 | 32.13 ± 11.93 | 36.75 ± 14.03 | 31.85 ± 8.67 |
| Mercury * | 0.05 ± 0.01 | 0.05 ± 0 | 0.06 ± 0.02 | 0.05 ± 0 | 0.05 ± 0 | 0.05 ± 0.01 | 0.05 ± 0 | 0.05 ± 0 | 0.05 ± 0.01 | 0.05 ± 0.01 | 0.05 ± 0 |
| Lead * | 3.69 ± 1.36 | 3.79 ± 1.77 a,b | 3.04 ± 1.19 b | 3.24 ± 0.99 a,b | 4.69 ± 0.80 a | 3.42 ± 1.52 | 3.97 ± 1.15 | 3.80 ± 1.11 | 3.52 ± 1.68 | 3.57 ± 1.48 | 3.81 ± 1.25 |
| Zinc * | 47.3 ± 14.03 | 43.4 ± 17.62 | 45.10 ± 17.66 | 49.30 ± 10.57 | 51.4 ± 8.57 | 44.25 ± 17.19 | 50.35 ± 9.43 | 50.38 ± 12.18 | 42.68 ± 15.69 | 55.20 ± 12.52 a | 39.04 ± 10.44 b |
| SYSTEM | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| REM | RVS | ||||||||||
| SECTORS | |||||||||||
| PR | TA | MU | CO | ||||||||
| Phyllum/Class | Family | Genus/Specie/Type | SU | WI | SU | WI | SU | WI | SU | WI | |
| 1 | Annellida, Polychaeta | Capitellidae | Capitella sp. | 6 | 1 | 4 | 5 | 28 | 27 | 11 | 9 |
| 2 | Notomastus sp. | - | - | - | - | 3 | - | - | - | ||
| 3 | Nephtyidae | Nepthys sp2 | 6 | 2 | 5 | 2 | 1 | 3 | 2 | - | |
| 4 | Nepthys sp1 | 18 | 1 | 2 | 4 | 4 | - | - | - | ||
| 5 | Nereididae | Perinereis sp. | 1 | - | 5 | 8 | 4 | 6 | 8 | - | |
| 6 | Nereis succinea | 1 | - | 1 | - | - | - | - | - | ||
| 7 | Hessionidae | Hesione picta | 11 | 1 | - | 2 | - | - | - | 2 | |
| 8 | Maldanidae | Maldane sp. | 12 | - | 4 | - | 1 | 3 | - | - | |
| 9 | Euclymene sp. | 2 | 12 | 18 | 21 | 5 | 2 | 2 | - | ||
| 10 | Terebellidae | Terebellidae | 1 | - | - | - | - | - | - | - | |
| 11 | Phyllodocidae | Phyllodoce sp1 | 4 | 1 | 4 | 1 | 1 | 6 | 3 | 24 | |
| 12 | Phyllodoce sp2 | 1 | - | 3 | - | 1 | - | 8 | - | ||
| 13 | Poecilochaeitidae | Poecilochaetus sp. | 2 | - | - | 1 | 3 | - | 2 | - | |
| 14 | Oenonidae | Arabella sp. | 6 | 7 | 14 | 2 | - | - | - | - | |
| 15 | Drilonereis sp. | 1 | - | 1 | 1 | 3 | - | - | - | ||
| 16 | Opheliidae | Armandia sp. | 5 | 3 | 1 | - | 1 | - | - | - | |
| 17 | Spionidae | Prionosprio sp. | 13 | 7 | 13 | 14 | 8 | 34 | 4 | - | |
| 18 | Gonionidae | Glycinde sp. | 2 | - | 4 | 3 | - | - | - | - | |
| 19 | Cossuridae | Cossura rostrata | - | - | 1 | - | 9 | 107 | - | 6 | |
| 20 | Cirratulidae | Caulleriella sp. | - | - | - | - | 1 | - | 2 | - | |
| 21 | Glyceridae | Glycera sp. | 1 | - | 1 | - | 1 | - | - | - | |
| 22 | Oweniidae | Oweniidae | 1 | - | 2 | - | - | - | 1 | - | |
| 23 | Pecteneridae | Amphictene sp. | - | - | - | 2 | - | - | - | - | |
| 24 | Sternaspidae | Sternaspis sp. | - | 2 | - | - | - | - | - | - | |
| 25 | Mollusca, Bivalvia | Cyrenidae | Corbicula fluminea | - | - | - | - | - | - | 3 | - |
| 26 | Mytilidae | Modiolus sp. | - | - | - | - | - | - | 1 | - | |
| 27 | Veneridae | Leukoma asperrima | 1 | - | - | - | - | - | 2 | - | |
| 28 | Tellinidae | Tellina ecuadoriana | - | - | 6 | - | 20 | - | - | - | |
| 29 | Tellina recurvata | - | - | - | - | 2 | - | - | - | ||
| 30 | Solecurtidae | Tagelus bourgeoisae | 1 | - | - | 1 | 3 | 1 | - | - | |
| 31 | Mytilidae | Mytella guyanensis | - | - | 2 | 2 | - | - | - | - | |
| 32 | Cyrenidae | Polymesoda inflata | 1 | 1 | 4 | 2 | - | - | - | - | |
| 33 | Crassatellidae | Crasinella pacifica | - | - | 4 | - | - | - | - | - | |
| 34 | Molusca, Gastropods | Columbellidae | Cosmioconcha modesta | 4 | - | - | - | - | - | - | - |
| 35 | Polinicinae | Polinices grayi | - | - | 1 | - | - | - | - | - | |
| 36 | Nassariinae | Nassarius versicolor | 2 | - | - | - | - | - | - | - | |
| 37 | Arthropoda, Malacostraca | Panopeidae | Panopeus sp. | - | - | - | - | - | - | 5 | - |
| 38 | Penaeidae | Penaeus vannamei | 1 | - | 1 | - | - | - | 1 | - | |
| 49 | Protrachypene precipua | 1 | 1 | - | 1 | - | - | - | - | ||
| 39 | Portunidae | Callinectes sp. | - | - | - | - | - | - | 1 | - | |
| 40 | Mithracidae | Microphrys bicornutus | - | - | - | - | - | - | 2 | - | |
| 41 | Ocipodidae | Uca sp. | - | - | - | - | 4 | - | - | - | |
| 42 | Alpheidae | Alpheus sp. | 2 | - | 4 | 2 | 1 | - | - | - | |
| 43 | Leptocheliidae | Chondrochelia sp. | - | 10 | 2 | - | - | - | - | - | |
| 44 | Paratanaidae | Pseudoparatanais sp. | - | - | - | - | 5 | - | - | - | |
| 45 | Amphilochidae | Amphilochus sp. | - | 12 | - | - | - | 1 | - | - | |
| 46 | Goneplacidae | Carcinoplax sp. | 4 | 3 | 2 | 2 | 1 | 1 | - | - | |
| 47 | Balanidae | Balanus sp. | - | - | - | - | - | - | 1 | - | |
| 48 | Hexapodidae | Mariaplax sp. | 1 | - | - | - | 1 | - | - | - | |
| 53 | Arthropoda, Insecta | Diptera | NA | - | - | - | - | 1 | - | - | - |
| 54 | Chironomidae | Chironomus sp. | - | 2 | - | 15 | - | 2 | - | - | |
| 50 | Echinodermata, Ophiuroidea | Ophiotrichidae | Ophiotrix spiculata | - | - | 1 | - | 1 | - | - | - |
| 51 | Ophiodermatidae | Ophioderma panamense | 1 | - | 3 | - | 1 | - | 2 | - | |
| 52 | Platyhelminthes | Planariidae | Planaria sp. | 2 | - | 1 | 2 | 2 | - | - | - |
| 55 | Bryozoa | Stenolaemata | Briozoa | - | - | - | - | - | - | 1 | - |
| 56 | Cnidaria | Anthozoa | Anemone 1 | - | - | - | 1 | - | - | - | - |
| Descriptor/ Index | Global Mean | SECTOR | SYSTEM | TYPE | SEASON | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| PR | TA | MU | CO | REM | RVS | SF | MG | SU | WI | ||
| Richness | 7 ± 5 | 8 ± 5 a,b | 10 ± 5 a | 7 ± 4 a,b | 4 ± 4 b | 9 ± 5 a | 5 ± 4 b | 6 ± 5 | 8 ± 5 | 9 ± 5a | 5 ± 4 b |
| Abundance | 20 ± 18 | 18 ±14 a,b | 21 ± 13 a,b | 31 ± 27 a | 10 ± 10 b | 19 ± 13 | 21 ± 22 | 21 ± 21 | 18 ± 12 | 20 ± 14 | 20 ± 22 |
| Dominance | 0.313 ± 0.229 | 0.302 ± 0.261 | 0.213 ± 0.102 | 0.383 ± 0.226 | 0.371 ± 0.304 | 0.257 ± 0.198 | 0.378 ± 0.252 | 0.335 ± 0.239 | 0.283 ± 0.220 | 0.222 ± 0.127 | 0.420 ± 0.277 |
| Shannon | 1.552 ± 0.687 | 1.627 ± 0.746 | 1.888 ± 0.513 | 1.314 ± 0.663 | 1.306 ± 0.759 | 1.758 ± 0.637 a | 1.311 ± 0.681 b | 1.485 ± 0.700 | 1.640 ± 0.681 | 1.824 ± 0.605 | 1.233 ± 0.652 |
| AMBI | 2.481 ± 1.670 | 1.269 ± 0.698 b | 1.416 ± 0.584 b | 3.175 ± 0.808 a | 4.062 ± 2.199 a | 1.343 ± 0.631 | 3.619 ± 1.676 | 2.977 ± 1.861 | 1.737 ± 1.103 | 2.145± 1.022 | 2.817 ± 2.156 |
| M-AMBI | 0.476 ± 0.234 | 0.580 ± 0.173 | 0.632 ± 0.151 | 0.425 ± 0.171 | 0.273 ± 0.262 | 0.606 ± 0.160 | 0.349 ± 0.229 | 0.426 ± 0.254 | 0.555 ± 0.183 | 0.581 ± 0.198 | 0.374 ± 0.226 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rebolledo Monsalve, E.; Verduga Vergara, L. Water and Sediment Quality Changes in Mangrove Systems with Shrimp Farms in the Northern Ecuadorean Coast. Appl. Sci. 2023, 13, 7749. https://doi.org/10.3390/app13137749
Rebolledo Monsalve E, Verduga Vergara L. Water and Sediment Quality Changes in Mangrove Systems with Shrimp Farms in the Northern Ecuadorean Coast. Applied Sciences. 2023; 13(13):7749. https://doi.org/10.3390/app13137749
Chicago/Turabian StyleRebolledo Monsalve, Eduardo, and Lita Verduga Vergara. 2023. "Water and Sediment Quality Changes in Mangrove Systems with Shrimp Farms in the Northern Ecuadorean Coast" Applied Sciences 13, no. 13: 7749. https://doi.org/10.3390/app13137749





